Current Evidence and Perspectives of Cluster of Differentiation 44 in the Liver’s Physiology and Pathology
Abstract
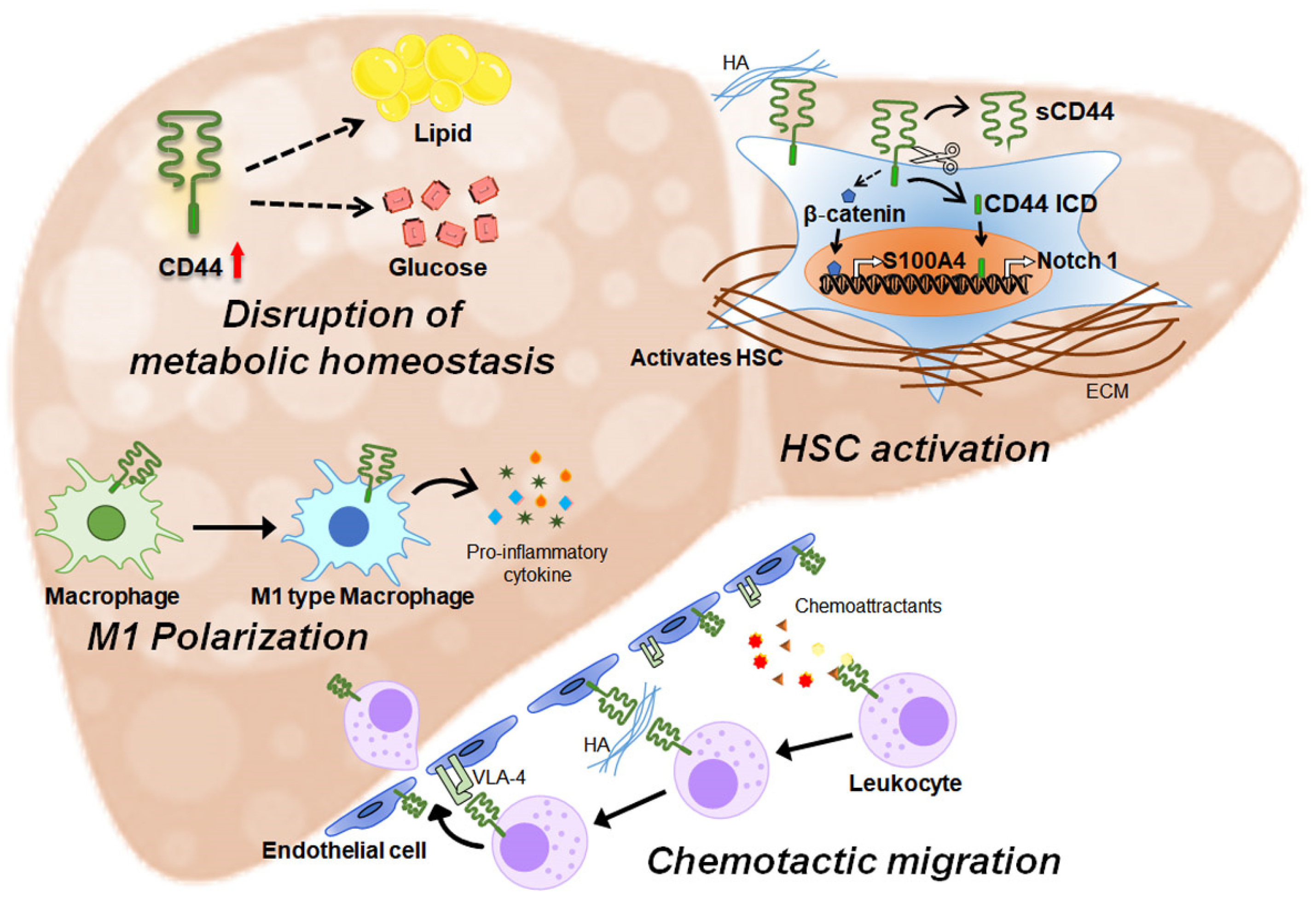
:1. Introduction
2. General Information on CD44
3. CD44 in Liver Disease
3.1. CD44 Is Involved in Liver Metabolism
3.2. CD44 Regulates Inflammation
3.3. CD44 Promotes Liver Fibrosis
4. CD44 Influences Cell Proliferation and Differentiation in the Liver
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Rui, L. Energy metabolism in the liver. Compr. Physiol. 2014, 4, 177–197. [Google Scholar] [CrossRef] [PubMed]
- Bechmann, L.P.; Hannivoort, R.A.; Gerken, G.; Hotamisligil, G.S.; Trauner, M.; Canbay, A. The interaction of hepatic lipid and glucose metabolism in liver diseases. J. Hepatol. 2012, 56, 952–964. [Google Scholar] [CrossRef] [PubMed]
- van den Berghe, G. The role of the liver in metabolic homeostasis: Implications for inborn errors of metabolism. J. Inherit. Metab. Dis. 1991, 14, 407–420. [Google Scholar] [CrossRef] [PubMed]
- Shu, W.; Yang, M.; Yang, J.; Lin, S.; Wei, X.; Xu, X. Cellular crosstalk during liver regeneration: Unity in diversity. Cell Commun. Signal 2022, 20, 117. [Google Scholar] [CrossRef] [PubMed]
- Malarkey, D.E.; Johnson, K.; Ryan, L.; Boorman, G.; Maronpot, R.R. New insights into functional aspects of liver morphology. Toxicol. Pathol. 2005, 33, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Itoh, T.; Tanimizu, N.; Miyajima, A. Liver stem/progenitor cells: Their characteristics and regulatory mechanisms. J. Biochem. 2011, 149, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Ishibashi, H.; Nakamura, M.; Komori, A.; Migita, K.; Shimoda, S. Liver architecture, cell function, and disease. Semin. Immunopathol. 2009, 31, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Kennedy, L.; Liangpunsakul, S.; Kusumanchi, P.; Yang, Z.; Meng, F.; Glaser, S.; Francis, H.; Alpini, G. Intercellular Communication between Hepatic Cells in Liver Diseases. Int. J. Mol. Sci. 2019, 20, 2180. [Google Scholar] [CrossRef] [PubMed]
- Malhi, H.; Gores, G.J. Cellular and molecular mechanisms of liver injury. Gastroenterology 2008, 134, 1641–1654. [Google Scholar] [CrossRef]
- Ozaki, M. Cellular and molecular mechanisms of liver regeneration: Proliferation, growth, death and protection of hepatocytes. Semin. Cell Dev. Biol. 2020, 100, 62–73. [Google Scholar] [CrossRef]
- Tao, Y.; Wang, M.; Chen, E.; Tang, H. Liver Regeneration: Analysis of the Main Relevant Signaling Molecules. Mediat. Inflamm. 2017, 2017, 4256352. [Google Scholar] [CrossRef] [PubMed]
- Luedde, T.; Kaplowitz, N.; Schwabe, R.F. Cell death and cell death responses in liver disease: Mechanisms and clinical relevance. Gastroenterology 2014, 147, 765–783.e4. [Google Scholar] [CrossRef] [PubMed]
- Malhi, H.; Guicciardi, M.E.; Gores, G.J. Hepatocyte death: A clear and present danger. Physiol. Rev. 2010, 90, 1165–1194. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.C.; Zhang, Q.B.; Qiao, L. Pathogenesis of liver cirrhosis. World J. Gastroenterol. 2014, 20, 7312–7324. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Poulsen, K.L.; Wu, L.; Liu, S.; Miyata, T.; Song, Q.; Wei, Q.; Zhao, C.; Lin, C.; Yang, J. Targeted therapeutics and novel signaling pathways in non-alcohol-associated fatty liver/steatohepatitis (NAFL/NASH). Signal Transduct. Target. Ther. 2022, 7, 287. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Gea, V.; Friedman, S.L. Pathogenesis of liver fibrosis. Annu. Rev. Pathol. 2011, 6, 425–456. [Google Scholar] [CrossRef] [PubMed]
- Block, L.H.; Tenschert, W.; Locher, R.; Siegenthaler, W.; Vetter, W. Receptor dysfunctions in human disease. Klin. Wochenschr. 1981, 59, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Jordan, A.R.; Racine, R.R.; Hennig, M.J.; Lokeshwar, V.B. The Role of CD44 in Disease Pathophysiology and Targeted Treatment. Front. Immunol. 2015, 6, 182. [Google Scholar] [CrossRef]
- Sneath, R.J.; Mangham, D.C. The normal structure and function of CD44 and its role in neoplasia. Mol. Pathol. 1998, 51, 191–200. [Google Scholar] [CrossRef]
- Ponta, H.; Sherman, L.; Herrlich, P.A. CD44: From adhesion molecules to signalling regulators. Nat. Rev. Mol. Cell Biol. 2003, 4, 33–45. [Google Scholar] [CrossRef]
- Weng, X.; Maxwell-Warburton, S.; Hasib, A.; Ma, L.; Kang, L. The membrane receptor CD44: Novel insights into metabolism. Trends Endocrinol. Metab. 2022, 33, 318–332. [Google Scholar] [CrossRef] [PubMed]
- Falleti, E.; Pirisi, M.; Fabris, C.; Bortolotti, N.; Soardo, G.; Gonano, F.; Bartoli, E. Circulating standard CD44 isoform in patients with liver disease: Relationship with other soluble adhesion molecules and evaluation of diagnostic usefulness. Clin. Biochem. 1997, 30, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Osawa, Y.; Kawai, H.; Tsunoda, T.; Komatsu, H.; Okawara, M.; Tsutsui, Y.; Yoshida, Y.; Yoshikawa, S.; Mori, T.; Yamazoe, T.; et al. Cluster of Differentiation 44 Promotes Liver Fibrosis and Serves as a Biomarker in Congestive Hepatopathy. Hepatol. Commun. 2021, 5, 1437–1447. [Google Scholar] [CrossRef] [PubMed]
- Rozeik, M.S.; Hammam, O.A.; Ali, A.I.; Magdy, M.; Khalil, H.; Anas, A.; Abo El Hassan, A.A.; Rahim, A.A.; El-Shabasy, A.I. Evaluation of CD44 and CD133 as markers of liver cancer stem cells in Egyptian patients with HCV-induced chronic liver diseases versus hepatocellular carcinoma. Electron. Physician 2017, 9, 4708–4717. [Google Scholar] [CrossRef] [PubMed]
- Skandalis, S.S. CD44 Intracellular Domain: A Long Tale of a Short Tail. Cancers 2023, 15, 5041. [Google Scholar] [CrossRef]
- Hassn Mesrati, M.; Syafruddin, S.E.; Mohtar, M.A.; Syahir, A. CD44: A Multifunctional Mediator of Cancer Progression. Biomolecules 2021, 11, 1850. [Google Scholar] [CrossRef]
- Underhill, C. CD44: The hyaluronan receptor. J. Cell Sci. 1992, 103 Pt 2, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Naor, D.; Sionov, R.V.; Ish-Shalom, D. CD44: Structure, function, and association with the malignant process. Adv. Cancer Res. 1997, 71, 241–319. [Google Scholar] [CrossRef] [PubMed]
- Goodison, S.; Urquidi, V.; Tarin, D. CD44 cell adhesion molecules. Mol. Pathol. 1999, 52, 189–196. [Google Scholar] [CrossRef]
- Chen, C.; Zhao, S.; Karnad, A.; Freeman, J.W. The biology and role of CD44 in cancer progression: Therapeutic implications. J. Hematol. Oncol. 2018, 11, 64. [Google Scholar] [CrossRef]
- Bourguignon, L.Y.; Zhu, H.; Shao, L.; Chen, Y.W. CD44 interaction with c-Src kinase promotes cortactin-mediated cytoskeleton function and hyaluronic acid-dependent ovarian tumor cell migration. J. Biol. Chem. 2001, 276, 7327–7336. [Google Scholar] [CrossRef] [PubMed]
- Dzwonek, J.; Wilczynski, G.M. CD44: Molecular interactions, signaling and functions in the nervous system. Front. Cell. Neurosci. 2015, 9, 175. [Google Scholar] [CrossRef] [PubMed]
- Cortes-Dericks, L.; Schmid, R.A. CD44 and its ligand hyaluronan as potential biomarkers in malignant pleural mesothelioma: Evidence and perspectives. Respir. Res. 2017, 18, 58. [Google Scholar] [CrossRef] [PubMed]
- Senbanjo, L.T.; Chellaiah, M.A. CD44: A Multifunctional Cell Surface Adhesion Receptor Is a Regulator of Progression and Metastasis of Cancer Cells. Front. Cell Dev. Biol. 2017, 5, 18. [Google Scholar] [CrossRef] [PubMed]
- Kothapalli, D.; Flowers, J.; Xu, T.; Puré, E.; Assoian, R.K. Differential activation of ERK and Rac mediates the proliferative and anti-proliferative effects of hyaluronan and CD44. J. Biol. Chem. 2008, 283, 31823–31829. [Google Scholar] [CrossRef] [PubMed]
- Kothapalli, D.; Zhao, L.; Hawthorne, E.A.; Cheng, Y.; Lee, E.; Puré, E.; Assoian, R.K. Hyaluronan and CD44 antagonize mitogen-dependent cyclin D1 expression in mesenchymal cells. J. Cell Biol. 2007, 176, 535–544. [Google Scholar] [CrossRef] [PubMed]
- Singleton, P.A.; Bourguignon, L.Y. CD44 interaction with ankyrin and IP3 receptor in lipid rafts promotes hyaluronan-mediated Ca2+ signaling leading to nitric oxide production and endothelial cell adhesion and proliferation. Exp. Cell Res. 2004, 295, 102–118. [Google Scholar] [CrossRef] [PubMed]
- Kawano, Y.; Okamoto, I.; Murakami, D.; Itoh, H.; Yoshida, M.; Ueda, S.; Saya, H. Ras oncoprotein induces CD44 cleavage through phosphoinositide 3-OH kinase and the rho family of small G proteins. J. Biol. Chem. 2000, 275, 29628–29635. [Google Scholar] [CrossRef]
- Brown, L.F.; Berse, B.; Van de Water, L.; Papadopoulos-Sergiou, A.; Perruzzi, C.A.; Manseau, E.J.; Dvorak, H.F.; Senger, D.R. Expression and distribution of osteopontin in human tissues: Widespread association with luminal epithelial surfaces. Mol. Biol. Cell 1992, 3, 1169–1180. [Google Scholar] [CrossRef]
- Denhardt, D.T.; Noda, M.; O’Regan, A.W.; Pavlin, D.; Berman, J.S. Osteopontin as a means to cope with environmental insults: Regulation of inflammation, tissue remodeling, and cell survival. J. Clin. Investig. 2001, 107, 1055–1061. [Google Scholar] [CrossRef]
- Nagano, O.; Saya, H. Mechanism and biological significance of CD44 cleavage. Cancer Sci. 2004, 95, 930–935. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, I.; Kawano, Y.; Matsumoto, M.; Suga, M.; Kaibuchi, K.; Ando, M.; Saya, H. Regulated CD44 cleavage under the control of protein kinase C, calcium influx, and the Rho family of small G proteins. J. Biol. Chem. 1999, 274, 25525–25534. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, I.; Kawano, Y.; Tsuiki, H.; Sasaki, J.; Nakao, M.; Matsumoto, M.; Suga, M.; Ando, M.; Nakajima, M.; Saya, H. CD44 cleavage induced by a membrane-associated metalloprotease plays a critical role in tumor cell migration. Oncogene 1999, 18, 1435–1446. [Google Scholar] [CrossRef]
- Kamarajan, P.; Shin, J.M.; Qian, X.; Matte, B.; Zhu, J.Y.; Kapila, Y.L. ADAM17-mediated CD44 cleavage promotes orasphere formation or stemness and tumorigenesis in HNSCC. Cancer Med. 2013, 2, 793–802. [Google Scholar] [CrossRef] [PubMed]
- Kajita, M.; Itoh, Y.; Chiba, T.; Mori, H.; Okada, A.; Kinoh, H.; Seiki, M. Membrane-type 1 matrix metalloproteinase cleaves CD44 and promotes cell migration. J. Cell Biol. 2001, 153, 893–904. [Google Scholar] [CrossRef] [PubMed]
- De Falco, V.; Tamburrino, A.; Ventre, S.; Castellone, M.D.; Malek, M.; Manié, S.N.; Santoro, M. CD44 proteolysis increases CREB phosphorylation and sustains proliferation of thyroid cancer cells. Cancer Res. 2012, 72, 1449–1458. [Google Scholar] [CrossRef] [PubMed]
- Johansson, E.; Grassi, E.S.; Pantazopoulou, V.; Tong, B.; Lindgren, D.; Berg, T.J.; Pietras, E.J.; Axelson, H.; Pietras, A. CD44 Interacts with HIF-2α to Modulate the Hypoxic Phenotype of Perinecrotic and Perivascular Glioma Cells. Cell Rep. 2017, 20, 1641–1653. [Google Scholar] [CrossRef] [PubMed]
- Patouraux, S.; Rousseau, D.; Bonnafous, S.; Lebeaupin, C.; Luci, C.; Canivet, C.M.; Schneck, A.S.; Bertola, A.; Saint-Paul, M.C.; Iannelli, A.; et al. CD44 is a key player in non-alcoholic steatohepatitis. J. Hepatol. 2017, 67, 328–338. [Google Scholar] [CrossRef] [PubMed]
- Bertola, A.; Deveaux, V.; Bonnafous, S.; Rousseau, D.; Anty, R.; Wakkach, A.; Dahman, M.; Tordjman, J.; Clément, K.; McQuaid, S.E.; et al. Elevated expression of osteopontin may be related to adipose tissue macrophage accumulation and liver steatosis in morbid obesity. Diabetes 2009, 58, 125–133. [Google Scholar] [CrossRef]
- Saponaro, C.; Gaggini, M.; Carli, F.; Gastaldelli, A. The Subtle Balance between Lipolysis and Lipogenesis: A Critical Point in Metabolic Homeostasis. Nutrients 2015, 7, 9453–9474. [Google Scholar] [CrossRef]
- Paschos, P.; Paletas, K. Non alcoholic fatty liver disease and metabolic syndrome. Hippokratia 2009, 13, 9–19. [Google Scholar] [PubMed]
- Kang, H.S.; Liao, G.; DeGraff, L.M.; Gerrish, K.; Bortner, C.D.; Garantziotis, S.; Jetten, A.M. CD44 plays a critical role in regulating diet-induced adipose inflammation, hepatic steatosis, and insulin resistance. PLoS ONE 2013, 8, e58417. [Google Scholar] [CrossRef] [PubMed]
- Samblas, M.; Mansego, M.L.; Zulet, M.A.; Milagro, F.I.; Martinez, J.A. An integrated transcriptomic and epigenomic analysis identifies CD44 gene as a potential biomarker for weight loss within an energy-restricted program. Eur. J. Nutr. 2019, 58, 1971–1980. [Google Scholar] [CrossRef] [PubMed]
- Kodama, K.; Toda, K.; Morinaga, S.; Yamada, S.; Butte, A.J. Anti-CD44 antibody treatment lowers hyperglycemia and improves insulin resistance, adipose inflammation, and hepatic steatosis in diet-induced obese mice. Diabetes 2015, 64, 867–875. [Google Scholar] [CrossRef] [PubMed]
- Katayama, Y.; Hidalgo, A.; Chang, J.; Peired, A.; Frenette, P.S. CD44 is a physiological E-selectin ligand on neutrophils. J. Exp. Med. 2005, 201, 1183–1189. [Google Scholar] [CrossRef] [PubMed]
- Puré, E.; Cuff, C.A. A crucial role for CD44 in inflammation. Trends Mol. Med. 2001, 7, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Hou, W.; Kong, L.; Hou, Z.; Ji, H. CD44 is a prognostic biomarker and correlated with immune infiltrates in gastric cancer. BMC Med. Genom. 2022, 15, 225. [Google Scholar] [CrossRef] [PubMed]
- Egan, C.E.; Daugherity, E.K.; Rogers, A.B.; Abi Abdallah, D.S.; Denkers, E.Y.; Maurer, K.J. CCR2 and CD44 promote inflammatory cell recruitment during fatty liver formation in a lithogenic diet fed mouse model. PLoS ONE 2013, 8, e65247. [Google Scholar] [CrossRef]
- McDonald, B.; Kubes, P. Interactions between CD44 and Hyaluronan in Leukocyte Trafficking. Front. Immunol. 2015, 6, 68. [Google Scholar] [CrossRef]
- Filippi, M.D. Neutrophil transendothelial migration: Updates and new perspectives. Blood 2019, 133, 2149–2158. [Google Scholar] [CrossRef]
- Zhu, B.; Suzuki, K.; Goldberg, H.A.; Rittling, S.R.; Denhardt, D.T.; McCulloch, C.A.; Sodek, J. Osteopontin modulates CD44-dependent chemotaxis of peritoneal macrophages through G-protein-coupled receptors: Evidence of a role for an intracellular form of osteopontin. J. Cell. Physiol. 2004, 198, 155–167. [Google Scholar] [CrossRef] [PubMed]
- Alstergren, P.; Zhu, B.; Glogauer, M.; Mak, T.W.; Ellen, R.P.; Sodek, J. Polarization and directed migration of murine neutrophils is dependent on cell surface expression of CD44. Cell. Immunol. 2004, 231, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Nandi, A.; Estess, P.; Siegelman, M. Bimolecular complex between rolling and firm adhesion receptors required for cell arrest; CD44 association with VLA-4 in T cell extravasation. Immunity 2004, 20, 455–465. [Google Scholar] [CrossRef]
- Graham, V.A.; Marzo, A.L.; Tough, D.F. A role for CD44 in T cell development and function during direct competition between CD44+ and CD44− cells. Eur. J. Immunol. 2007, 37, 925–934. [Google Scholar] [CrossRef] [PubMed]
- Hansel, C.; Erschfeld, S.; Baues, M.; Lammers, T.; Weiskirchen, R.; Trautwein, C.; Kroy, D.C.; Drescher, H.K. The Inhibitory T Cell Receptors PD1 and 2B4 Are Differentially Regulated on CD4 and CD8 T Cells in a Mouse Model of Non-alcoholic Steatohepatitis. Front. Pharmacol. 2019, 10, 244. [Google Scholar] [CrossRef]
- Guidotti, L.G.; Inverso, D.; Sironi, L.; Di Lucia, P.; Fioravanti, J.; Ganzer, L.; Fiocchi, A.; Vacca, M.; Aiolfi, R.; Sammicheli, S.; et al. Immunosurveillance of the liver by intravascular effector CD8+ T cells. Cell 2015, 161, 486–500. [Google Scholar] [CrossRef] [PubMed]
- Funken, D.; Ishikawa-Ankerhold, H.; Uhl, B.; Lerchenberger, M.; Rentsch, M.; Mayr, D.; Massberg, S.; Werner, J.; Khandoga, A. In situ targeting of dendritic cells sets tolerogenic environment and ameliorates CD4+ T-cell response in the postischemic liver. FASEB J. 2017, 31, 4796–4808. [Google Scholar] [CrossRef] [PubMed]
- Cruise, M.W.; Melief, H.M.; Lukens, J.; Soguero, C.; Hahn, Y.S. Increased Fas ligand expression of CD4+ T cells by HCV core induces T cell-dependent hepatic inflammation. J. Leukoc. Biol. 2005, 78, 412–425. [Google Scholar] [CrossRef] [PubMed]
- Baaten, B.J.; Li, C.R.; Bradley, L.M. Multifaceted regulation of T cells by CD44. Commun. Integr. Biol. 2010, 3, 508–512. [Google Scholar] [CrossRef]
- Do, Y.; Nagarkatti, P.S.; Nagarkatti, M. Role of CD44 and hyaluronic acid (HA) in activation of alloreactive and antigen-specific T cells by bone marrow-derived dendritic cells. J. Immunother. 2004, 27, 1–12. [Google Scholar] [CrossRef]
- Hegde, V.L.; Singh, N.P.; Nagarkatti, P.S.; Nagarkatti, M. CD44 mobilization in allogeneic dendritic cell-T cell immunological synapse plays a key role in T cell activation. J. Leukoc. Biol. 2008, 84, 134–142. [Google Scholar] [CrossRef] [PubMed]
- McDonald, B.; McAvoy, E.F.; Lam, F.; Gill, V.; de la Motte, C.; Savani, R.C.; Kubes, P. Interaction of CD44 and hyaluronan is the dominant mechanism for neutrophil sequestration in inflamed liver sinusoids. J. Exp. Med. 2008, 205, 915–927. [Google Scholar] [CrossRef] [PubMed]
- Volkov, Y.; Long, A.; Freeley, M.; Golden-Mason, L.; O’Farrelly, C.; Murphy, A.; Kelleher, D. The hepatitis C envelope 2 protein inhibits LFA-1-transduced protein kinase C signaling for T-lymphocyte migration. Gastroenterology 2006, 130, 482–492. [Google Scholar] [CrossRef] [PubMed]
- Masson, D.; Denis, M.G.; Denis, M.; Blanchard, D.; Loirat, M.J.; Cassagnau, E.; Lustenberger, P. Soluble CD44: Quantification and molecular repartition in plasma of patients with colorectal cancer. Br. J. Cancer 1999, 80, 1995–2000. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Narayanswamy, S.; Ramesh, A.V. Comparative evaluation of salivary soluble CD44 levels in periodontal health and disease. J. Indian Soc. Periodontol. 2014, 18, 734–738. [Google Scholar] [CrossRef] [PubMed]
- Franzmann, E.J.; Reategui, E.P.; Pedroso, F.; Pernas, F.G.; Karakullukcu, B.M.; Carraway, K.L.; Hamilton, K.; Singal, R.; Goodwin, W.J. Soluble CD44 is a potential marker for the early detection of head and neck cancer. Cancer Epidemiol. Biomark. Prev. 2007, 16, 1348–1355. [Google Scholar] [CrossRef] [PubMed]
- Heymann, F.; Tacke, F. Immunology in the liver—From homeostasis to disease. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 88–110. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T.A. Cellular and molecular mechanisms of fibrosis. J. Pathol. 2008, 214, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Koyama, Y.; Brenner, D.A. Liver inflammation and fibrosis. J. Clin. Investig. 2017, 127, 55–64. [Google Scholar] [CrossRef]
- Friedman, S.L. Hepatic stellate cells: Protean, multifunctional, and enigmatic cells of the liver. Physiol. Rev. 2008, 88, 125–172. [Google Scholar] [CrossRef]
- Lee, U.E.; Friedman, S.L. Mechanisms of hepatic fibrogenesis. Best Pract. Res. Clin. Gastroenterol. 2011, 25, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Hagenstein, J.; Burkhardt, S.; Sprezyna, P.; Tasika, E.; Tiegs, G.; Diehl, L. CD44 expression on murine hepatic stellate cells promotes the induction of monocytic and polymorphonuclear myeloid-derived suppressor cells. J. Leukoc. Biol. 2024, qiae053. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, S.; Griffin, C.T.; Wang, S.S.; Bissell, D.M. Role of CD44 in epithelial wound repair: Migration of rat hepatic stellate cells utilizes hyaluronic acid and CD44v6. J. Biol. Chem. 2005, 280, 15398–15404. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Kim, J.; Lee, C.; Jung, Y. Tumor necrosis factor-inducible gene 6 interacts with CD44, which is involved in fate-change of hepatic stellate cells. BMB Rep. 2020, 53, 425–430. [Google Scholar] [CrossRef]
- He, Y.; Wu, G.D.; Sadahiro, T.; Noh, S.I.; Wang, H.; Talavera, D.; Wang, H.; Vierling, J.M.; Klein, A.S. Interaction of CD44 and hyaluronic acid enhances biliary epithelial proliferation in cholestatic livers. Am. J. Physiol. Gastrointest. Liver Physiol. 2008, 295, G305–G312. [Google Scholar] [CrossRef] [PubMed]
- Uno, K.; Miyajima, K.; Toma, M.; Suzuki-Kemuriyama, N.; Nakae, D. CD44 expression in the bile duct epithelium is related to hepatic fibrosis in nonalcoholic steatohepatitis rats induced by a choline-deficient, methionine-lowered, L-amino acid diet. J. Toxicol. Pathol. 2022, 35, 149–157. [Google Scholar] [CrossRef]
- Kim, J.; Seki, E. Hyaluronan in liver fibrosis: Basic mechanisms, clinical implications, and therapeutic targets. Hepatol. Commun. 2023, 7, e0083. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.M.; Noureddin, M.; Liu, C.; Ohashi, K.; Kim, S.Y.; Ramnath, D.; Powell, E.E.; Sweet, M.J.; Roh, Y.S.; Hsin, I.F.; et al. Hyaluronan synthase 2-mediated hyaluronan production mediates Notch1 activation and liver fibrosis. Sci. Transl. Med. 2019, 11, eaat9284. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Phan, S.H. Notch in fibrosis and as a target of anti-fibrotic therapy. Pharmacol. Res. 2016, 108, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Dhar, D.; Antonucci, L.; Nakagawa, H.; Kim, J.Y.; Glitzner, E.; Caruso, S.; Shalapour, S.; Yang, L.; Valasek, M.A.; Lee, S.; et al. Liver Cancer Initiation Requires p53 Inhibition by CD44-Enhanced Growth Factor Signaling. Cancer Cell 2018, 33, 1061–1077.e6. [Google Scholar] [CrossRef]
- Mitaka, T.; Ichinohe, N.; Tanimizu, N. “Small Hepatocytes” in the Liver. Cells 2023, 12, 2718. [Google Scholar] [CrossRef] [PubMed]
- Kon, J.; Ooe, H.; Oshima, H.; Kikkawa, Y.; Mitaka, T. Expression of CD44 in rat hepatic progenitor cells. J. Hepatol. 2006, 45, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, K.; Kon, J.; Mizuguchi, T.; Chen, Q.; Ooe, H.; Oshima, H.; Hirata, K.; Mitaka, T. Proliferation of hepatocyte progenitor cells isolated from adult human livers in serum-free medium. Cell Transplant. 2008, 17, 1221–1230. [Google Scholar] [CrossRef] [PubMed]
- Ichinohe, N.; Kon, J.; Sasaki, K.; Nakamura, Y.; Ooe, H.; Tanimizu, N.; Mitaka, T. Growth ability and repopulation efficiency of transplanted hepatic stem cells, progenitor cells, and mature hepatocytes in retrorsine-treated rat livers. Cell Transplant. 2012, 21, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.; Lozoya, O.; Wang, Y.; Cardinale, V.; Gaudio, E.; Alpini, G.; Mendel, G.; Wauthier, E.; Barbier, C.; Alvaro, D.; et al. Human hepatic stem cell and maturational liver lineage biology. Hepatology 2011, 53, 1035–1045. [Google Scholar] [CrossRef] [PubMed]
- Turner, W.S.; Schmelzer, E.; McClelland, R.; Wauthier, E.; Chen, W.; Reid, L.M. Human hepatoblast phenotype maintained by hyaluronan hydrogels. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 82, 156–168. [Google Scholar] [CrossRef] [PubMed]
- Della Fazia, M.A.; Pettirossi, V.; Ayroldi, E.; Riccardi, C.; Magni, M.V.; Servillo, G. Differential expression of CD44 isoforms during liver regeneration in rats. J. Hepatol. 2001, 34, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.Y.; Baek, G.H.; Lee, W.; Lee, Y.J.; Shim, W.S.; Choi, Y.J.; Lee, B.H.; Kim, S.K.; Kang, K.W. CD44 is involved in liver regeneration through enhanced uptake of extracellular cystine. Clin. Transl. Med. 2022, 12, e873. [Google Scholar] [CrossRef] [PubMed]
- Stanger, B.Z. Cellular homeostasis and repair in the mammalian liver. Annu. Rev. Physiol. 2015, 77, 179–200. [Google Scholar] [CrossRef]
- Di-Iacovo, N.; Pieroni, S.; Piobbico, D.; Castelli, M.; Scopetti, D.; Ferracchiato, S.; Della-Fazia, M.A.; Servillo, G. Liver Regeneration and Immunity: A Tale to Tell. Int. J. Mol. Sci. 2023, 24, 1176. [Google Scholar] [CrossRef]
- Jeong, H.; Lee, C.; Lee, M.J.; Jung, Y. Therapeutic strategies to improve liver regeneration after hepatectomy. Exp. Biol. Med. 2023, 248, 1313–1318. [Google Scholar] [CrossRef]
- Chang, G.; Zhang, H.; Wang, J.; Zhang, Y.; Xu, H.; Wang, C.; Zhang, H.; Ma, L.; Li, Q.; Pang, T. CD44 targets Wnt/β-catenin pathway to mediate the proliferation of K562 cells. Cancer Cell Int. 2013, 13, 117. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.; Takasu, T.; Henke, C. Functional role of cyclin A on induction of fibroblast apoptosis due to ligation of CD44 matrix receptor by anti-CD44 antibody. Exp. Cell Res. 2000, 257, 135–144. [Google Scholar] [CrossRef] [PubMed]
- Shang, X.Z.; Zhu, H.; Lin, K.; Tu, Z.; Chen, J.; Nelson, D.R.; Liu, C. Stabilized beta-catenin promotes hepatocyte proliferation and inhibits TNFalpha-induced apoptosis. Lab. Investig. 2004, 84, 332–341. [Google Scholar] [CrossRef]
- Perugorria, M.J.; Olaizola, P.; Labiano, I.; Esparza-Baquer, A.; Marzioni, M.; Marin, J.J.G.; Bujanda, L.; Banales, J.M. Wnt-β-catenin signalling in liver development, health and disease. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 121–136. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Behari, J.; Cieply, B.; Michalopoulos, G.K.; Monga, S.P. Conditional deletion of beta-catenin reveals its role in liver growth and regeneration. Gastroenterology 2006, 131, 1561–1572. [Google Scholar] [CrossRef]
- Isono, M.; Takeuchi, J.; Maehara, A.; Nakagawa, Y.; Katagiri, H.; Miyatake, K.; Sekiya, I.; Koga, H.; Asou, Y.; Tsuji, K. Effect of CD44 signal axis in the gain of mesenchymal stem cell surface antigens from synovial fibroblasts in vitro. Heliyon 2022, 8, e10739. [Google Scholar] [CrossRef] [PubMed]
- Márquez, C.; Trigueros, C.; Fernández, E.; Toribio, M.L. The development of T and non-T cell lineages from CD34+ human thymic precursors can be traced by the differential expression of CD44. J. Exp. Med. 1995, 181, 475–483. [Google Scholar] [CrossRef]
- Guan, H.; Nagarkatti, P.S.; Nagarkatti, M. Role of CD44 in the differentiation of Th1 and Th2 cells: CD44-deficiency enhances the development of Th2 effectors in response to sheep RBC and chicken ovalbumin. J. Immunol. 2009, 183, 172–180. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, J.; Lee, C.; Jung, Y. Current Evidence and Perspectives of Cluster of Differentiation 44 in the Liver’s Physiology and Pathology. Int. J. Mol. Sci. 2024, 25, 4749. https://doi.org/10.3390/ijms25094749
Han J, Lee C, Jung Y. Current Evidence and Perspectives of Cluster of Differentiation 44 in the Liver’s Physiology and Pathology. International Journal of Molecular Sciences. 2024; 25(9):4749. https://doi.org/10.3390/ijms25094749
Chicago/Turabian StyleHan, Jinsol, Chanbin Lee, and Youngmi Jung. 2024. "Current Evidence and Perspectives of Cluster of Differentiation 44 in the Liver’s Physiology and Pathology" International Journal of Molecular Sciences 25, no. 9: 4749. https://doi.org/10.3390/ijms25094749





