MECP2 Dysfunction in Rett Syndrome: Molecular Mechanisms, Multisystem Pathology, and Emerging Therapeutic Strategies
Abstract
1. Introduction
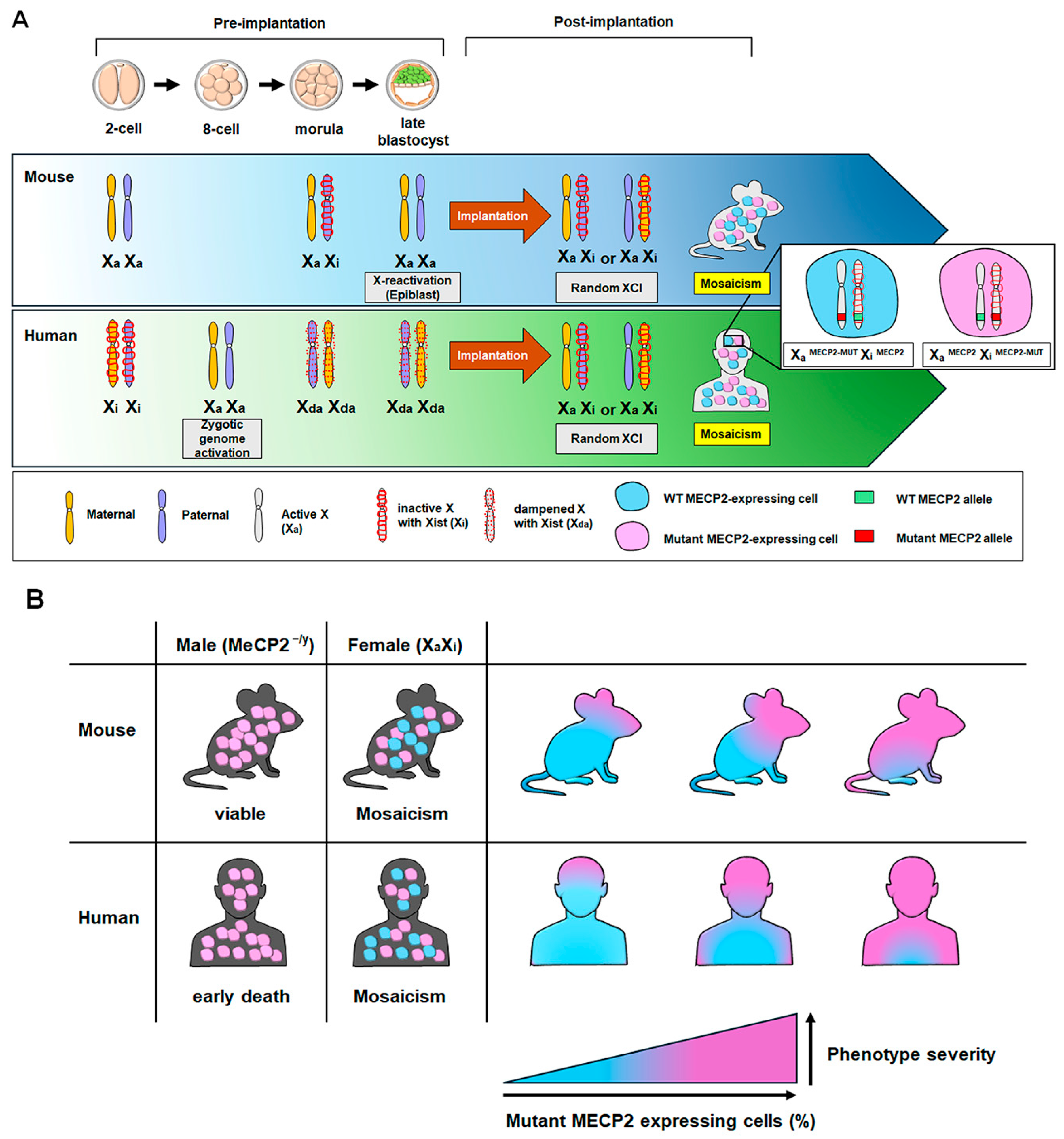
2. X Chromosome Inactivation and MECP2 Mosaicism in Rett Syndrome Pathogenesis
3. Molecular Mechanism of MECP2 Function
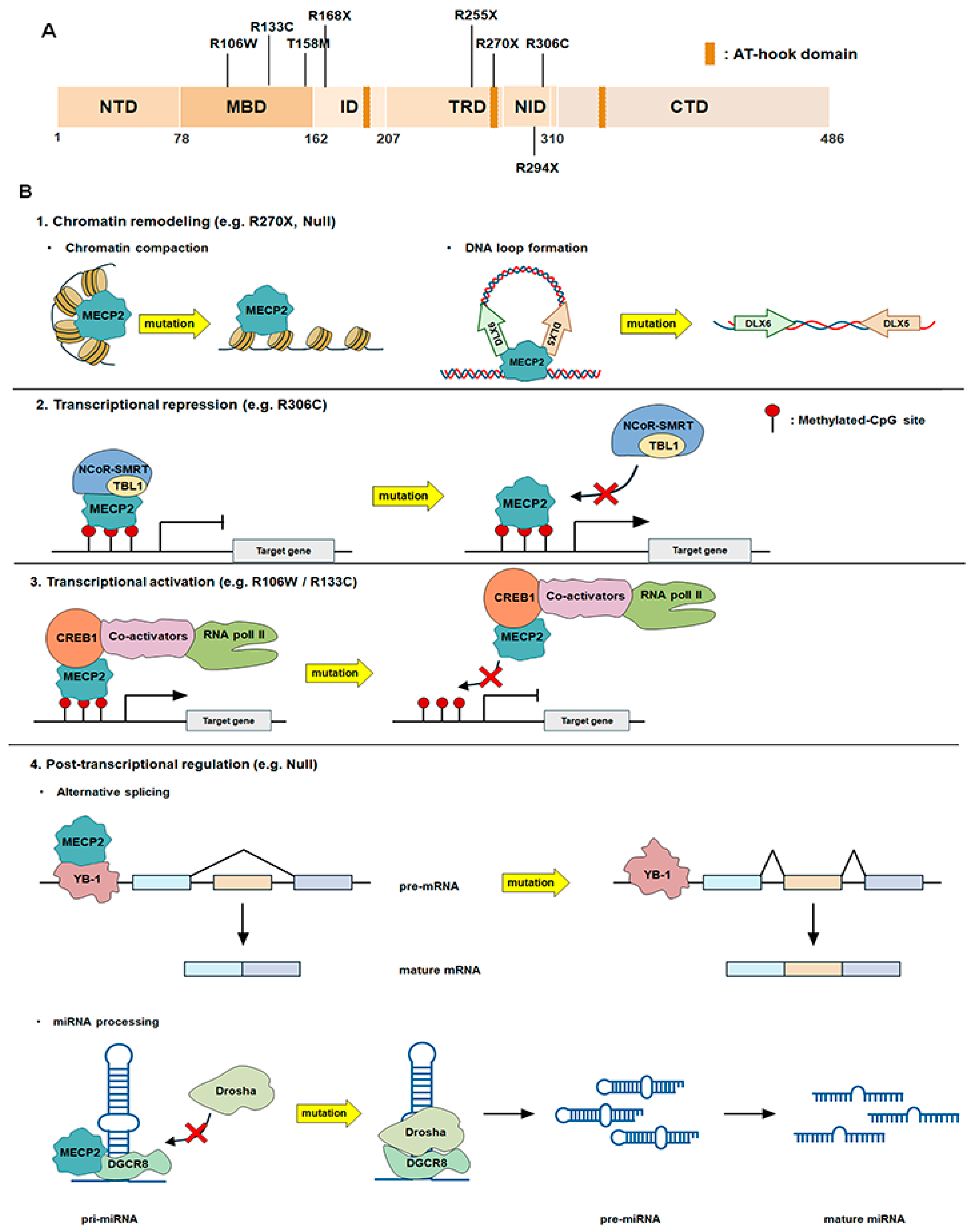
3.1. MECP2 Structure and Its Mechanism of Gene Expression Regulation
3.2. MECP2 Mutations Affecting Gene Expression
3.3. Post-Translational Modification Regulation by MECP2
3.3.1. Histone Deacetylation and Chromatin Remodeling via MECP2–HDAC Interactions
3.3.2. Histone Methylation
3.3.3. DNA Methylation
3.3.4. MECP2 Phosphorylation and SUMOylation
4. Nervous Systems Affected by MECP2 Mutations
4.1. MECP2 Function in Early Neurogenesis
4.2. Neuronal Defects in Cerebral Cortex
4.2.1. Clinical Alteration of Brain Structure in Rett Syndrome
4.2.2. Clinical Alteration of Neurons in Rett Syndrome
4.2.3. MECP2-Regulated Genes Involved in Neuronal Function
- Excitatory Neurotransmission-Related Genes
- Inhibitory Neurotransmission-Related Genes
- Other Genes Regulated by MECP2
4.2.4. Neuronal Imbalance
4.2.5. Neuronal Types Affected by MECP2 Mutations
4.2.6. Cerebellar Cortex Dysfunction in Rett Syndrome
4.2.7. Hippocampal Dysfunction in Rett Syndrome
4.2.8. Olfactory Bulb
4.2.9. Autonomous Nervous System
4.3. Glial Cells Affected by MECP2 Mutations and Rett Syndrome
4.4. Other Organs Affected by MECP2 Mutation and Rett Syndrome
5. Rett Syndrome Treatment
5.1. AAV9-Based Gene Replacement Therapy
5.2. MECP2 Gene Editing and Xi Reactivation
5.3. Anti-Inflammation and Immunomodulation
5.4. Pharmacological Therapy
5.5. Other Therapeutic Approaches
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ADAR2 | Adenosine deaminases acting on RNA 2 |
| ANS | Autonomous nervous system |
| BDNF | Brain-derived neurotrophic factor |
| CREB1 | cAMP-responsive element-binding protein 1 |
| CRF | Corticotropic releasing factor |
| CTD | C-terminal domain |
| DAN | Dorsal attention network |
| DBS | Deep brain stimulation |
| ERK | Extracellular signal-regulated kinase |
| GABA | γ-aminobutyric acid |
| GABAA | γ-aminobutyric acid a |
| GFAP | Glial fibrillary acidic protein |
| GIRK | G protein-gated inwardly rectifying potassium channel |
| GO | Gene Ontology |
| GPE | Glycine-Proline-Glutamate |
| GS | Glutamine synthetase |
| HAT | Histone acetyltransferase |
| HDAC | Histone deacetylases |
| ID | Intervening domain |
| IGF | Insulin-like growth factor |
| LTP | Long-term potentiation |
| MBD | Methyl-CpG-binding domain |
| MECP2 | Methyl-CpG-binding protein 2 |
| NCoR | Nuclear receptor co-repressor |
| NGF | Neurogliaform |
| NID | NCoR–SMRT interaction domain |
| NMDA | N-methyl-D-aspartate |
| NMDAR | NMDA receptor |
| NPC | Neural Progenitor Cell |
| NSC | Neural stem cells |
| OMP | Olfactory marker protein |
| ORN | Olfactory receptor neurons |
| PC | Purkinje cell |
| PTEN | Phosphatase and tensin homolog |
| PTZ | Neural progenitor cells |
| SIN3A | Switch-independent 3A |
| SMRT | Silencing mediator of retinoic acid and thyroid hormone receptor |
| TrkB | Tropomyosin receptor kinase B |
| TRD | Transcriptional repression domain |
| TSS | Transcription start site |
| WT | Wild type |
| XCI | X chromosome inactivation |
References
- Zoghbi, H.Y. MeCP2 dysfunction in humans and mice. J. Child Neurol. 2005, 20, 736–740. [Google Scholar] [CrossRef]
- Evans, J.C.; Archer, H.L.; Colley, J.P.; Ravn, K.; Nielsen, J.B.; Kerr, A.; Williams, E.; Christodoulou, J.; Gécz, J.; Jardine, P.E. Early onset seizures and Rett-like features associated with mutations in CDKL5. Eur. J. Hum. Genet. 2005, 13, 1113–1120. [Google Scholar] [CrossRef]
- Philippe, C.; Amsallem, D.; Francannet, C.; Lambert, L.; Saunier, A.; Verneau, F.; Jonveaux, P. Phenotypic variability in Rett syndrome associated with FOXG1 mutations in females. J. Med. Genet. 2010, 47, 59–65. [Google Scholar] [CrossRef][Green Version]
- Petriti, U.; Dudman, D.C.; Scosyrev, E.; Lopez-Leon, S. Global prevalence of Rett syndrome: Systematic review and meta-analysis. Syst. Rev. 2023, 12, 5. [Google Scholar] [CrossRef]
- Akbarian, S.; Jiang, Y.; Laforet, G. The molecular pathology of Rett syndrome: Synopsis and update. Neuromol. Med. 2006, 8, 485–494. [Google Scholar] [CrossRef]
- Chahrour, M.; Zoghbi, H.Y. The story of Rett syndrome: From clinic to neurobiology. Neuron 2007, 56, 422–437. [Google Scholar] [CrossRef]
- Krishnaraj, R.; Ho, G.; Christodoulou, J. RettBASE: Rett syndrome database update. Hum. Mutat. 2017, 38, 922–931. [Google Scholar] [CrossRef] [PubMed]
- Signorini, C.; Leoncini, S.; Durand, T.; Galano, J.-M.; Guy, A.; Bultel-Poncé, V.; Oger, C.; Lee, J.C.-Y.; Ciccoli, L.; Hayek, J. Circulating 4-F4t-Neuroprostane and 10-F4t-Neuroprostane Are Related to MECP2 Gene Mutation and Natural History in Rett Syndrome. Int. J. Mol. Sci. 2021, 22, 4240. [Google Scholar] [CrossRef] [PubMed]
- Neul, J.; Fang, P.; Barrish, J.; Lane, J.; Caeg, E.; Smith, E.; Zoghbi, H.; Percy, A.; Glaze, D. Specific mutations in methyl-CpG-binding protein 2 confer different severity in Rett syndrome. Neurology 2008, 70, 1313–1321. [Google Scholar] [CrossRef]
- Cuddapah, V.A.; Pillai, R.B.; Shekar, K.V.; Lane, J.B.; Motil, K.J.; Skinner, S.A.; Tarquinio, D.C.; Glaze, D.G.; McGwin, G.; Kaufmann, W.E. Methyl-CpG-binding protein 2 (MECP2) mutation type is associated with disease severity in Rett syndrome. J. Med. Genet. 2014, 51, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Trappe, R.; Laccone, F.; Cobilanschi, J.; Meins, M.; Huppke, P.; Hanefeld, F.; Engel, W. MECP2 mutations in sporadic cases of Rett syndrome are almost exclusively of paternal origin. Am. J. Hum. Genet. 2001, 68, 1093–1101. [Google Scholar] [CrossRef]
- Girard, M.; Couvert, P.; Carrié, A.; Tardieu, M.; Chelly, J.; Beldjord, C.; Bienvenu, T. Parental origin of de novo MECP2 mutations in Rett syndrome. Eur. J. Hum. Genet. 2001, 9, 231–236. [Google Scholar] [CrossRef]
- Zhu, X.; Li, M.; Pan, H.; Bao, X.; Zhang, J.; Wu, X. Analysis of the parental origin of de novo MECP2 mutations and X chromosome inactivation in 24 sporadic patients with Rett syndrome in China. J. Child Neurol. 2010, 25, 842–848. [Google Scholar] [CrossRef]
- Shah, J.; Patel, H.; Jain, D.; Sheth, F.; Sheth, H. A rare case of a male child with post-zygotic de novo mosaic variant c. 538C> T in MECP2 gene: A case report of Rett syndrome. BMC Neurol. 2021, 21, 469. [Google Scholar] [CrossRef]
- Schüle, B.; Armstrong, D.; Vogel, H.; Oviedo, A.; Francke, U. Severe congenital encephalopathy caused by MECP2 null mutations in males: Central hypoxia and reduced neuronal dendritic structure. Clin. Genet. 2008, 74, 116–126. [Google Scholar] [CrossRef]
- Cheung, A.Y.; Horvath, L.M.; Grafodatskaya, D.; Pasceri, P.; Weksberg, R.; Hotta, A.; Carrel, L.; Ellis, J. Isolation of MECP2-null Rett Syndrome patient hiPS cells and isogenic controls through X-chromosome inactivation. Hum. Mol. Genet. 2011, 20, 2103–2115. [Google Scholar] [CrossRef] [PubMed]
- Guy, J.; Hendrich, B.; Holmes, M.; Martin, J.E.; Bird, A. A mouse Mecp2-null mutation causes neurological symptoms that mimic Rett syndrome. Nat. Genet. 2001, 27, 322–326. [Google Scholar] [CrossRef] [PubMed]
- Tukiainen, T.; Villani, A.-C.; Yen, A.; Rivas, M.A.; Marshall, J.L.; Satija, R.; Aguirre, M.; Gauthier, L.; Fleharty, M.; Kirby, A.; et al. Landscape of X chromosome inactivation across human tissues. Nature 2017, 550, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Patrat, C.; Ouimette, J.-F.; Rougeulle, C. X chromosome inactivation in human development. Development 2020, 147, dev183095. [Google Scholar] [CrossRef]
- van den Berg, I.M.; Laven, J.S.; Stevens, M.; Jonkers, I.; Galjaard, R.-J.; Gribnau, J.; van Doorninck, J.H. X chromosome inactivation is initiated in human preimplantation embryos. Am. J. Hum. Genet. 2009, 84, 771–779. [Google Scholar] [CrossRef]
- Thorpe, J.; Osei-Owusu, I.A.; Avigdor, B.E.; Tupler, R.; Pevsner, J. Mosaicism in human health and disease. Annu. Rev. Genet. 2020, 54, 487–510. [Google Scholar] [CrossRef]
- Renthal, W.; Boxer, L.D.; Hrvatin, S.; Li, E.; Silberfeld, A.; Nagy, M.A.; Griffith, E.C.; Vierbuchen, T.; Greenberg, M.E. Characterization of human mosaic Rett syndrome brain tissue by single-nucleus RNA sequencing. Nat. Neurosci. 2018, 21, 1670–1679. [Google Scholar] [CrossRef]
- Armstrong, D.D. Neuropathology of Rett syndrome. J. Child Neurol. 2005, 20, 747–753. [Google Scholar] [CrossRef]
- Young, J.I.; Zoghbi, H.Y. X-chromosome inactivation patterns are unbalanced and affect the phenotypic outcome in a mouse model of rett syndrome. Am. J. Hum. Genet. 2004, 74, 511–520. [Google Scholar] [CrossRef]
- Schaevitz, L.; Gómez, N.; Zhen, D.; Berger-Sweeney, J. MeCP2 R168X male and female mutant mice exhibit Rett-like behavioral deficits. Genes Brain Behav. 2013, 12, 732–740. [Google Scholar] [CrossRef] [PubMed]
- Cheung, A.Y.; Horvath, L.M.; Carrel, L.; Ellis, J. X-chromosome inactivation in rett syndrome human induced pluripotent stem cells. Front. Psychiatry 2012, 3, 24. [Google Scholar] [CrossRef]
- Archer, H.; Evans, J.; Leonard, H.; Colvin, L.; Ravine, D.; Christodoulou, J.; Williamson, S.; Charman, T.; Bailey, M.E.; Sampson, J. Correlation between clinical severity in patients with Rett syndrome with a p.R168X or p.T158M MECP2 mutation, and the direction and degree of skewing of X-chromosome inactivation. J. Med. Genet. 2007, 44, 148–152. [Google Scholar] [CrossRef]
- Pohodich, A.E.; Zoghbi, H.Y. Rett syndrome: Disruption of epigenetic control of postnatal neurological functions. Hum. Mol. Genet. 2015, 24, R10–R16. [Google Scholar] [CrossRef] [PubMed]
- D’Mello III, S.R. MECP2 and the biology of MECP2 duplication syndrome. J. Neurochem. 2021, 159, 29–60. [Google Scholar] [CrossRef] [PubMed]
- Shahbazian, M.D.; Young, J.I.; Yuva-Paylor, L.A.; Spencer, C.M.; Antalffy, B.A.; Noebels, J.L.; Armstrong, D.L.; Paylor, R.; Zoghbi, H.Y. Mice with truncated MeCP2 recapitulate many Rett syndrome features and display hyperacetylation of histone H3. Neuron 2002, 35, 243–254. [Google Scholar] [CrossRef]
- Chen, R.Z.; Akbarian, S.; Tudor, M.; Jaenisch, R. Deficiency of methyl-CpG binding protein-2 in CNS neurons results in a Rett-like phenotype in mice. Nat. Genet. 2001, 27, 327–331. [Google Scholar] [CrossRef]
- Li, Y.; Wang, H.; Muffat, J.; Cheng, A.W.; Orlando, D.A.; Lovén, J.; Kwok, S.-m.; Feldman, D.A.; Bateup, H.S.; Gao, Q. Global transcriptional and translational repression in human-embryonic-stem-cell-derived Rett syndrome neurons. Cell Stem Cell 2013, 13, 446–458. [Google Scholar] [CrossRef] [PubMed]
- Cheng, T.-L.; Chen, J.; Wan, H.; Tang, B.; Tian, W.; Liao, L.; Qiu, Z. Regulation of mRNA splicing by MeCP2 via epigenetic modifications in the brain. Sci. Rep. 2017, 7, 42790. [Google Scholar] [CrossRef] [PubMed]
- Young, J.I.; Hong, E.P.; Castle, J.C.; Crespo-Barreto, J.; Bowman, A.B.; Rose, M.F.; Kang, D.; Richman, R.; Johnson, J.M.; Berget, S. Regulation of RNA splicing by the methylation-dependent transcriptional repressor methyl-CpG binding protein 2. Proc. Natl. Acad. Sci. USA 2005, 102, 17551–17558. [Google Scholar] [CrossRef]
- Tsujimura, K.; Irie, K.; Nakashima, H.; Egashira, Y.; Fukao, Y.; Fujiwara, M.; Itoh, M.; Uesaka, M.; Imamura, T.; Nakahata, Y. miR-199a links MeCP2 with mTOR signaling and its dysregulation leads to Rett syndrome phenotypes. Cell Rep. 2015, 12, 1887–1901. [Google Scholar] [CrossRef]
- Cheng, T.-L.; Wang, Z.; Liao, Q.; Zhu, Y.; Zhou, W.-H.; Xu, W.; Qiu, Z. MeCP2 suppresses nuclear microRNA processing and dendritic growth by regulating the DGCR8/Drosha complex. Dev. Cell 2014, 28, 547–560. [Google Scholar] [CrossRef]
- Pejhan, S.; Rastegar, M. Role of DNA methyl-CpG-binding protein MeCP2 in Rett syndrome pathobiology and mechanism of disease. Biomolecules 2021, 11, 75. [Google Scholar] [CrossRef]
- Georgel, P.T.; Horowitz-Scherer, R.A.; Adkins, N.; Woodcock, C.L.; Wade, P.A.; Hansen, J.C. Chromatin compaction by human MeCP2: Assembly of novel secondary chromatin structures in the absence of DNA methylation. J. Biol. Chem. 2003, 278, 32181–32188. [Google Scholar] [CrossRef] [PubMed]
- Skene, P.J.; Illingworth, R.S.; Webb, S.; Kerr, A.R.; James, K.D.; Turner, D.J.; Andrews, R.; Bird, A.P. Neuronal MeCP2 is expressed at near histone-octamer levels and globally alters the chromatin state. Mol. Cell 2010, 37, 457–468. [Google Scholar] [CrossRef]
- Wang, L.; Hu, M.; Zuo, M.-Q.; Zhao, J.; Wu, D.; Huang, L.; Wen, Y.; Li, Y.; Chen, P.; Bao, X. Rett syndrome-causing mutations compromise MeCP2-mediated liquid–liquid phase separation of chromatin. Cell Res. 2020, 30, 393–407. [Google Scholar] [CrossRef]
- Horike, S.-i.; Cai, S.; Miyano, M.; Cheng, J.-F.; Kohwi-Shigematsu, T. Loss of silent-chromatin looping and impaired imprinting of DLX5 in Rett syndrome. Nat. Genet. 2005, 37, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Nan, X.; Ng, H.-H.; Johnson, C.A.; Laherty, C.D.; Turner, B.M.; Eisenman, R.N.; Bird, A. Transcriptional repression by the methyl-CpG-binding protein MeCP2 involves a histone deacetylase complex. Nature 1998, 393, 386–389. [Google Scholar] [CrossRef]
- Jones, P.L.; Veenstra, G.J.C.; Wade, P.A.; Vermaak, D.; Kass, S.U.; Landsberger, N.; Strouboulis, J.; Wolffe, A.P. Methylated DNA and MeCP2 recruit histone deacetylase to repress transcription. Nat. Genet. 1998, 19, 187–191. [Google Scholar] [CrossRef]
- Lyst, M.J.; Ekiert, R.; Ebert, D.H.; Merusi, C.; Nowak, J.; Selfridge, J.; Guy, J.; Kastan, N.R.; Robinson, N.D.; de Lima Alves, F. Rett syndrome mutations abolish the interaction of MeCP2 with the NCoR/SMRT co-repressor. Nat. Neurosci. 2013, 16, 898–902. [Google Scholar] [CrossRef]
- Chahrour, M.; Jung, S.Y.; Shaw, C.; Zhou, X.; Wong, S.T.; Qin, J.; Zoghbi, H.Y. MeCP2, a key contributor to neurological disease, activates and represses transcription. Science 2008, 320, 1224–1229. [Google Scholar] [CrossRef]
- Ben-Shachar, S.; Chahrour, M.; Thaller, C.; Shaw, C.A.; Zoghbi, H.Y. Mouse models of MeCP2 disorders share gene expression changes in the cerebellum and hypothalamus. Hum. Mol. Genet. 2009, 18, 2431–2442. [Google Scholar] [CrossRef]
- Li, R.; Dong, Q.; Yuan, X.; Zeng, X.; Gao, Y.; Chiao, C.; Li, H.; Zhao, X.; Keles, S.; Wang, Z. Misregulation of alternative splicing in a mouse model of Rett syndrome. PLoS Genet. 2016, 12, e1006129. [Google Scholar] [CrossRef] [PubMed]
- Lyabin, D.N.; Eliseeva, I.A.; Ovchinnikov, L.P. YB-1 protein: Functions and regulation. Wiley Interdiscip. Rev. RNA 2014, 5, 95–110. [Google Scholar] [CrossRef]
- Urdinguio, R.G.; Fernandez, A.F.; Lopez-Nieva, P.; Rossi, S.; Huertas, D.; Kulis, M.; Liu, C.-G.; Croce, C.M.; Calin, G.A.; Esteller, M. Disrupted microRNA expression caused by Mecp2 loss in a mouse model of Rett syndrome. Epigenetics 2010, 5, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Ballestar, E.; Ropero, S.; Alaminos, M.; Armstrong, J.; Setien, F.; Agrelo, R.; Fraga, M.F.; Herranz, M.; Avila, S.; Pineda, M. The impact of MECP2 mutations in the expression patterns of Rett syndrome patients. Hum. Genet. 2005, 116, 91–104. [Google Scholar] [CrossRef]
- Traynor, J.; Agarwal, P.; Lazzeroni, L.; Francke, U. Gene expression patterns vary in clonal cell cultures from Rett syndrome females with eight different MECP2 mutations. BMC Med. Genet. 2002, 3, 12. [Google Scholar] [CrossRef]
- Lyst, M.J.; Bird, A. Rett syndrome: A complex disorder with simple roots. Nat. Rev. Genet. 2015, 16, 261–275. [Google Scholar] [CrossRef] [PubMed]
- Baker, S.A.; Chen, L.; Wilkins, A.D.; Yu, P.; Lichtarge, O.; Zoghbi, H.Y. An AT-hook domain in MeCP2 determines the clinical course of Rett syndrome and related disorders. Cell 2013, 152, 984–996. [Google Scholar] [CrossRef]
- Xu, X.; Pozzo-Miller, L. A novel DNA-binding feature of MeCP2 contributes to Rett syndrome. Front. Cell. Neurosci. 2013, 7, 64. [Google Scholar] [CrossRef] [PubMed]
- Guy, J.; Alexander-Howden, B.; FitzPatrick, L.; DeSousa, D.; Koerner, M.V.; Selfridge, J.; Bird, A. A mutation-led search for novel functional domains in MeCP2. Hum. Mol. Genet. 2018, 27, 2531–2545. [Google Scholar] [CrossRef] [PubMed]
- Heckman, L.D.; Chahrour, M.H.; Zoghbi, H.Y. Rett-causing mutations reveal two domains critical for MeCP2 function and for toxicity in MECP2 duplication syndrome mice. eLife 2014, 3, e02676. [Google Scholar] [CrossRef][Green Version]
- Liu, Y.; Flamier, A.; Bell, G.W.; Diao, A.J.; Whitfield, T.W.; Wang, H.-C.; Wu, Y.; Schulte, F.; Friesen, M.; Guo, R. MECP2 directly interacts with RNA polymerase II to modulate transcription in human neurons. Neuron 2024, 112, 1943–1958.E10. [Google Scholar] [CrossRef]
- Bebbington, A.; Anderson, A.; Ravine, D.; Fyfe, S.; Pineda, M.; de Klerk, N.; Ben-Zeev, B.; Yatawara, N.; Percy, A.; Kaufmann, W.E.; et al. Investigating genotype-phenotype relationships in Rett syndrome using an international data set. Neurology 2008, 70, 868–875. [Google Scholar] [CrossRef]
- Colvin, L.; Leonard, H.; de Klerk, N.; Davis, M.; Weaving, L.; Williamson, S.; Christodoulou, J. Refining the phenotype of common mutations in Rett syndrome. J. Med. Genet. 2004, 41, 25–30. [Google Scholar] [CrossRef]
- Klose, R.J.; Sarraf, S.A.; Schmiedeberg, L.; McDermott, S.M.; Stancheva, I.; Bird, A.P. DNA binding selectivity of MeCP2 due to a requirement for A/T sequences adjacent to methyl-CpG. Mol. Cell 2005, 19, 667–678. [Google Scholar] [CrossRef]
- Kouzarides, T. Chromatin modifications and their function. Cell 2007, 128, 693–705. [Google Scholar] [CrossRef]
- Wang, Z.; Zang, C.; Rosenfeld, J.A.; Schones, D.E.; Barski, A.; Cuddapah, S.; Cui, K.; Roh, T.-Y.; Peng, W.; Zhang, M.Q. Combinatorial patterns of histone acetylations and methylations in the human genome. Nat. Genet. 2008, 40, 897–903. [Google Scholar] [CrossRef] [PubMed]
- Nishida, H.; Suzuki, T.; Kondo, S.; Miura, H.; Fujimura, Y.-i.; Hayashizaki, Y. Histone H3 acetylated at lysine 9 in promoter is associated with low nucleosome density in the vicinity of transcription start site in human cell. Chromosome Res. 2006, 14, 203–211. [Google Scholar] [CrossRef]
- Morris, S.A.; Rao, B.; Garcia, B.A.; Hake, S.B.; Diaz, R.L.; Shabanowitz, J.; Hunt, D.F.; Allis, C.D.; Lieb, J.D.; Strahl, B.D. Identification of histone H3 lysine 36 acetylation as a highly conserved histone modification. J. Biol. Chem. 2007, 282, 7632–7640. [Google Scholar] [CrossRef]
- Xu, Y.M.; Du, J.Y.; Lau, A.T. Posttranslational modifications of human histone H3: An update. Proteomics 2014, 14, 2047–2060. [Google Scholar] [CrossRef]
- Che, Y.-C.; Jiang, L.; Li, Q.-H. Molecular modification of a HSV-1 protein and its associated gene transcriptional regulation. Virol. Sin. 2008, 23, 394–398. [Google Scholar] [CrossRef]
- Ayissi, V.B.O.; Ebrahimi, A.; Schluesenner, H. Epigenetic effects of natural polyphenols: A focus on SIRT1-mediated mechanisms. Mol. Nutr. Food Res. 2014, 58, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Dandrea, M.; Donadelli, M.; Costanzo, C.; Scarpa, A.; Palmieri, M. MeCP2/H3meK9 are involved in IL-6 gene silencing in pancreatic adenocarcinoma cell lines. Nucleic Acids Res. 2009, 37, 6681–6690. [Google Scholar] [CrossRef]
- Thambirajah, A.A.; Ng, M.K.; Frehlick, L.J.; Li, A.; Serpa, J.J.; Petrotchenko, E.V.; Silva-Moreno, B.; Missiaen, K.K.; Borchers, C.H.; Adam Hall, J. MeCP2 binds to nucleosome free (linker DNA) regions and to H3K9/H3K27 methylated nucleosomes in the brain. Nucleic Acids Res. 2012, 40, 2884–2897. [Google Scholar] [CrossRef]
- Nan, X.; Meehan, R.R.; Bird, A. Dissection of the methyl-CpG binding domain from the chromosomal protein MeCP2. Nucleic Acids Res. 1993, 21, 4886–4892. [Google Scholar] [CrossRef]
- Chen, L.; Chen, K.; Lavery, L.A.; Baker, S.A.; Shaw, C.A.; Li, W.; Zoghbi, H.Y. MeCP2 binds to non-CG methylated DNA as neurons mature, influencing transcription and the timing of onset for Rett syndrome. Proc. Natl. Acad. Sci. USA 2015, 112, 5509–5514. [Google Scholar] [CrossRef]
- Mellén, M.; Ayata, P.; Dewell, S.; Kriaucionis, S.; Heintz, N. MeCP2 binds to 5hmC enriched within active genes and accessible chromatin in the nervous system. Cell 2012, 151, 1417–1430. [Google Scholar] [CrossRef]
- Lee, W.; Kim, J.; Yun, J.-M.; Ohn, T.; Gong, Q. MeCP2 regulates gene expression through recognition of H3K27me3. Nat. Commun. 2020, 11, 3140. [Google Scholar] [CrossRef]
- Kimura, H.; Shiota, K. Methyl-CpG-binding protein, MeCP2, is a target molecule for maintenance DNA methyltransferase, Dnmt1. J. Biol. Chem. 2003, 278, 4806–4812. [Google Scholar] [CrossRef]
- Baubec, T.; Ivánek, R.; Lienert, F.; Schübeler, D. Methylation-dependent and-independent genomic targeting principles of the MBD protein family. Cell 2013, 153, 480–492. [Google Scholar] [CrossRef] [PubMed]
- Ebert, D.H.; Gabel, H.W.; Robinson, N.D.; Kastan, N.R.; Hu, L.S.; Cohen, S.; Navarro, A.J.; Lyst, M.J.; Ekiert, R.; Bird, A.P. Activity-dependent phosphorylation of MeCP2 threonine 308 regulates interaction with NCoR. Nature 2013, 499, 341–345. [Google Scholar] [CrossRef] [PubMed]
- Shibasaki, F.; Hallin, U.; Uchino, H. Calcineurin as a multifunctional regulator. J. Biochem. 2002, 131, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Buchthal, B.; Lau, D.; Weiss, U.; Weislogel, J.-M.; Bading, H. Nuclear calcium signaling controls methyl-CpG-binding protein 2 (MeCP2) phosphorylation on serine 421 following synaptic activity. J. Biol. Chem. 2012, 287, 30967–30974. [Google Scholar] [CrossRef]
- Lin, Y.; Bloodgood, B.L.; Hauser, J.L.; Lapan, A.D.; Koon, A.C.; Kim, T.-K.; Hu, L.S.; Malik, A.N.; Greenberg, M.E. Activity-dependent regulation of inhibitory synapse development by Npas4. Nature 2008, 455, 1198–1204. [Google Scholar] [CrossRef]
- Banerjee, A.; Romero-Lorenzo, E.; Sur, M. MeCP2: Making sense of missense in Rett syndrome. Cell Res. 2013, 23, 1244–1246. [Google Scholar] [CrossRef][Green Version]
- Zhou, Z.; Hong, E.J.; Cohen, S.; Zhao, W.-n.; Ho, H.-y.H.; Schmidt, L.; Chen, W.G.; Lin, Y.; Savner, E.; Griffith, E.C. Brain-specific phosphorylation of MeCP2 regulates activity-dependent Bdnf transcription, dendritic growth, and spine maturation. Neuron 2006, 52, 255–269. [Google Scholar] [CrossRef]
- Cohen, S.; Gabel, H.W.; Hemberg, M.; Hutchinson, A.N.; Sadacca, L.A.; Ebert, D.H.; Harmin, D.A.; Greenberg, R.S.; Verdine, V.K.; Zhou, Z. Genome-wide activity-dependent MeCP2 phosphorylation regulates nervous system development and function. Neuron 2011, 72, 72–85. [Google Scholar] [CrossRef]
- Tao, J.; Hu, K.; Chang, Q.; Wu, H.; Sherman, N.E.; Martinowich, K.; Klose, R.J.; Schanen, C.; Jaenisch, R.; Wang, W. Phosphorylation of MeCP2 at Serine 80 regulates its chromatin association and neurological function. Proc. Natl. Acad. Sci. USA 2009, 106, 4882–4887. [Google Scholar] [CrossRef] [PubMed]
- Chao, H.-T.; Zoghbi, H.Y. The yin and yang of MeCP2 phosphorylation. Proc. Natl. Acad. Sci. USA 2009, 106, 4577–4578. [Google Scholar] [CrossRef]
- Schlüter, O.M.; Schmitz, F.; Jahn, R.; Rosenmund, C.; Südhof, T.C. A complete genetic analysis of neuronal Rab3 function. J. Neurosci. 2004, 24, 6629–6637. [Google Scholar] [CrossRef]
- Bezzi, P.; Gundersen, V.; Galbete, J.L.; Seifert, G.; Steinhäuser, C.; Pilati, E.; Volterra, A. Astrocytes contain a vesicular compartment that is competent for regulated exocytosis of glutamate. Nat. Neurosci. 2004, 7, 613–620. [Google Scholar] [CrossRef]
- Park, J.; Liu, B.; Chen, T.; Li, H.; Hu, X.; Gao, J.; Zhu, Y.; Zhu, Q.; Qiang, B.; Yuan, J. Disruption of Nectin-like 1 cell adhesion molecule leads to delayed axonal myelination in the CNS. J. Neurosci. 2008, 28, 12815–12819. [Google Scholar] [CrossRef]
- Stefanelli, G.; Gandaglia, A.; Costa, M.; Cheema, M.S.; Di Marino, D.; Barbiero, I.; Kilstrup-Nielsen, C.; Ausió, J.; Landsberger, N. Brain phosphorylation of MeCP2 at serine 164 is developmentally regulated and globally alters its chromatin association. Sci. Rep. 2016, 6, 28295. [Google Scholar] [CrossRef]
- Tai, D.J.; Liu, Y.C.; Hsu, W.L.; Ma, Y.L.; Cheng, S.J.; Liu, S.Y.; Lee, E.H. MeCP2 SUMOylation rescues Mecp2-mutant-induced behavioural deficits in a mouse model of Rett syndrome. Nat. Commun. 2016, 7, 10552. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Huang, M.; Zhu, Y.; Xin, Y.J.; Zhao, Y.K.; Huang, J.; Yu, J.X.; Zhou, W.H.; Qiu, Z. SUMO ylation of Me CP 2 is essential for transcriptional repression and hippocampal synapse development. J. Neurochem. 2014, 128, 798–806. [Google Scholar] [CrossRef] [PubMed]
- Kalani, L.; Kim, B.-H.; Vincent, J.B.; Ausió, J. MeCP2 ubiquitination and sumoylation, in search of a function. Hum. Mol. Genet. 2024, 33, 1–11. [Google Scholar] [CrossRef]
- Hsu, W.-L.; Ma, Y.-L.; Liu, Y.-C.; Tai, D.J.; Lee, E.H. Restoring Wnt6 signaling ameliorates behavioral deficits in MeCP2 T158A mouse model of Rett syndrome. Sci. Rep. 2020, 10, 1074. [Google Scholar] [CrossRef]
- Goffin, D.; Allen, M.; Zhang, L.; Amorim, M.; Wang, I.-T.J.; Reyes, A.-R.S.; Mercado-Berton, A.; Ong, C.; Cohen, S.; Hu, L. Rett syndrome mutation MeCP2 T158A disrupts DNA binding, protein stability and ERP responses. Nat. Neurosci. 2012, 15, 274–283. [Google Scholar] [CrossRef]
- Kishi, N.; Macklis, J.D. MECP2 is progressively expressed in post-migratory neurons and is involved in neuronal maturation rather than cell fate decisions. Mol. Cell. Neurosci. 2004, 27, 306–321. [Google Scholar] [CrossRef]
- Li, H.; Zhong, X.; Chau, K.F.; Williams, E.C.; Chang, Q. Loss of activity-induced phosphorylation of MeCP2 enhances synaptogenesis, LTP and spatial memory. Nat. Neurosci. 2011, 14, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.; Ash, R.T.; Baker, S.A.; Suter, B.; Ferguson, A.; Park, J.; Rudy, J.; Torsky, S.P.; Chao, H.-T.; Zoghbi, H.Y. Dendritic arborization and spine dynamics are abnormal in the mouse model of MECP2 duplication syndrome. J. Neurosci. 2013, 33, 19518–19533. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-Y.; Hysolli, E.; Park, I.-H. Neuronal maturation defect in induced pluripotent stem cells from patients with Rett syndrome. Proc. Natl. Acad. Sci. USA 2011, 108, 14169–14174. [Google Scholar] [CrossRef]
- Jung, B.P.; Jugloff, D.G.; Zhang, G.; Logan, R.; Brown, S.; Eubanks, J.H. The expression of methyl CpG binding factor MeCP2 correlates with cellular differentiation in the developing rat brain and in cultured cells. J. Neurobiol. 2003, 55, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Shahbazian, M.D.; Antalffy, B.; Armstrong, D.L.; Zoghbi, H.Y. Insight into Rett syndrome: MeCP2 levels display tissue-and cell-specific differences and correlate with neuronal maturation. Hum. Mol. Genet. 2002, 11, 115–124. [Google Scholar] [CrossRef]
- Mullaney, B.; Johnston, M.; Blue, M. Developmental expression of methyl-CpG binding protein 2 is dynamically regulated in the rodent brain. Neuroscience 2004, 123, 939–949. [Google Scholar] [CrossRef]
- Nakashima, H.; Tsujimura, K.; Irie, K.; Imamura, T.; Trujillo, C.A.; Ishizu, M.; Uesaka, M.; Pan, M.; Noguchi, H.; Okada, K. MeCP2 controls neural stem cell fate specification through miR-199a-mediated inhibition of BMP-Smad signaling. Cell Rep. 2021, 35, 109124. [Google Scholar] [CrossRef] [PubMed]
- Mellios, N.; Feldman, D.A.; Sheridan, S.D.; Ip, J.P.; Kwok, S.; Amoah, S.K.; Rosen, B.; Rodriguez, B.A.; Crawford, B.; Swaminathan, R. MeCP2-regulated miRNAs control early human neurogenesis through differential effects on ERK and AKT signaling. Mol. Psychiatry 2018, 23, 1051–1065. [Google Scholar] [CrossRef]
- Belichenko, P.V.; Hagberg, B.; Dahlström, A. Morphological study of neocortical areas in Rett syndrome. Acta Neuropathol. 1996, 93, 50–61. [Google Scholar] [CrossRef]
- Hou, J.; Lin, L.; Zhou, W.; Wang, Z.; Ding, G.; Dong, Q.; Qin, L.; Wu, X.; Zheng, Y.; Yang, Y. Identification of miRNomes in human liver and hepatocellular carcinoma reveals miR-199a/b-3p as therapeutic target for hepatocellular carcinoma. Cancer Cell 2011, 19, 232–243. [Google Scholar] [CrossRef]
- Penna, E.; Orso, F.; Taverna, D. miR-214 as a key hub that controls cancer networks: Small player, multiple functions. J. Investig. Dermatol. 2015, 135, 960–969. [Google Scholar] [CrossRef]
- Shu, P.; Fu, H.; Zhao, X.; Wu, C.; Ruan, X.; Zeng, Y.; Liu, W.; Wang, M.; Hou, L.; Chen, P. MicroRNA-214 modulates neural progenitor cell differentiation by targeting Quaking during cerebral cortex development. Sci. Rep. 2017, 7, 8014. [Google Scholar] [CrossRef]
- Pucilowska, J.; Puzerey, P.A.; Karlo, J.C.; Galán, R.F.; Landreth, G.E. Disrupted ERK signaling during cortical development leads to abnormal progenitor proliferation, neuronal and network excitability and behavior, modeling human neuro-cardio-facial-cutaneous and related syndromes. J. Neurosci. 2012, 32, 8663–8677. [Google Scholar] [CrossRef]
- Zhang, J.; Shemezis, J.R.; McQuinn, E.R.; Wang, J.; Sverdlov, M.; Chenn, A. AKT activation by N-cadherin regulates beta-catenin signaling and neuronal differentiation during cortical development. Neural Dev. 2013, 8, 7. [Google Scholar] [CrossRef] [PubMed]
- Davis, B.N.; Hilyard, A.C.; Lagna, G.; Hata, A. SMAD proteins control DROSHA-mediated microRNA maturation. Nature 2008, 454, 56–61. [Google Scholar] [CrossRef]
- Takeguchi, R.; Kuroda, M.; Tanaka, R.; Suzuki, N.; Akaba, Y.; Tsujimura, K.; Itoh, M.; Takahashi, S. Structural and functional changes in the brains of patients with Rett syndrome: A multimodal MRI study. J. Neurol. Sci. 2022, 441, 120381. [Google Scholar] [CrossRef] [PubMed]
- Szczepanski, S.M.; Pinsk, M.A.; Douglas, M.M.; Kastner, S.; Saalmann, Y.B. Functional and structural architecture of the human dorsal frontoparietal attention network. Proc. Natl. Acad. Sci. USA 2013, 110, 15806–15811. [Google Scholar] [CrossRef]
- Lanssens, A.; Pizzamiglio, G.; Mantini, D.; Gillebert, C.R. Role of the dorsal attention network in distracter suppression based on features. In The Cognitive Neuroscience of Attention; Routledge: Abingdon, UK, 2020; pp. 204–213. [Google Scholar]
- Rohr, C.S.; Arora, A.; Cho, I.Y.; Katlariwala, P.; Dimond, D.; Dewey, D.; Bray, S. Functional network integration and attention skills in young children. Dev. Cogn. Neurosci. 2018, 30, 200–211. [Google Scholar] [CrossRef]
- Rohr, C.S.; Vinette, S.A.; Parsons, K.A.; Cho, I.Y.; Dimond, D.; Benischek, A.; Lebel, C.; Dewey, D.; Bray, S. Functional connectivity of the dorsal attention network predicts selective attention in 4–7 year-old girls. Cereb. Cortex 2017, 27, 4350–4360. [Google Scholar] [CrossRef]
- Della Sala, G.; Pizzorusso, T. Synaptic plasticity and signaling in Rett syndrome. Dev. Neurobiol. 2014, 74, 178–196. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Miller, E.C.; Pozzo-Miller, L. Dendritic spine dysgenesis in Rett syndrome. Front. Neuroanat. 2014, 8, 97. [Google Scholar] [CrossRef] [PubMed]
- Johnston, M.V.; Blue, M.E.; Naidu, S. Rett syndrome and neuronal development. J. Child Neurol. 2005, 20, 759–763. [Google Scholar] [CrossRef] [PubMed]
- Santos, M.; Summavielle, T.; Teixeira-Castro, A.; Silva-Fernandes, A.; Duarte-Silva, S.; Marques, F.; Martins, L.; Dierssen, M.; Oliveira, P.; Sousa, N. Monoamine deficits in the brain of methyl-CpG binding protein 2 null mice suggest the involvement of the cerebral cortex in early stages of Rett syndrome. Neuroscience 2010, 170, 453–467. [Google Scholar] [CrossRef]
- Ip, J.P.; Mellios, N.; Sur, M. Rett syndrome: Insights into genetic, molecular and circuit mechanisms. Nat. Rev. Neurosci. 2018, 19, 368–382. [Google Scholar] [CrossRef]
- Fukuda, T.; Itoh, M.; Ichikawa, T.; Washiyama, K.; Goto, Y.-i. Delayed maturation of neuronal architecture and synaptogenesis in cerebral cortex of Mecp2-deficient mice. J. Neuropathol. Exp. Neurol. 2005, 64, 537–544. [Google Scholar] [CrossRef]
- Maezawa, I.; Jin, L.-W. Rett syndrome microglia damage dendrites and synapses by the elevated release of glutamate. J. Neurosci. 2010, 30, 5346–5356. [Google Scholar] [CrossRef]
- Kishi, N.; MacDonald, J.L.; Ye, J.; Molyneaux, B.J.; Azim, E.; Macklis, J.D. Reduction of aberrant NF-κB signalling ameliorates Rett syndrome phenotypes in Mecp2-null mice. Nat. Commun. 2016, 7, 10520. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, D.; Dunn, J.K.; Antalffy, B.; Trivedi, R. Selective dendritic alterations in the cortex of Rett syndrome. J. Neuropathol. Exp. Neurol. 1995, 54, 195–201. [Google Scholar] [CrossRef]
- Armstrong, D.D. Neuropathology of Rett syndrome. Ment. Retard. Dev. Disabil. Res. Rev. 2002, 8, 72–76. [Google Scholar] [CrossRef]
- Wang, L.; Chang, X.; She, L.; Xu, D.; Huang, W.; Poo, M.-m. Autocrine action of BDNF on dendrite development of adult-born hippocampal neurons. J. Neurosci. 2015, 35, 8384–8393. [Google Scholar] [CrossRef]
- Cheng, P.-L.; Song, A.-H.; Wong, Y.-H.; Wang, S.; Zhang, X.; Poo, M.-M. Self-amplifying autocrine actions of BDNF in axon development. Proc. Natl. Acad. Sci. USA 2011, 108, 18430–18435. [Google Scholar] [CrossRef]
- Larimore, J.L.; Chapleau, C.A.; Kudo, S.; Theibert, A.; Percy, A.K.; Pozzo-Miller, L. Bdnf overexpression in hippocampal neurons prevents dendritic atrophy caused by Rett-associated MECP2 mutations. Neurobiol. Dis. 2009, 34, 199–211. [Google Scholar] [CrossRef]
- Lengel, D.; Romm, Z.L.; Bostwick, A.; Huh, J.W.; Snyder, N.W.; Smith, G.M.; Raghupathi, R. Glucocorticoid receptor overexpression in the dorsal hippocampus attenuates spatial learning and synaptic plasticity deficits after pediatric traumatic brain injury. J. Neurotrauma 2022, 39, 979–998. [Google Scholar] [CrossRef]
- Steffke, E.E.; Kirca, D.; Mazei-Robison, M.S.; Robison, A.J. Serum-and glucocorticoid-inducible kinase 1 activity reduces dendritic spines in dorsal hippocampus. Neurosci. Lett. 2020, 725, 134909. [Google Scholar] [CrossRef] [PubMed]
- Sahin, P.; McCaig, C.; Jeevahan, J.; Murray, J.T.; Hainsworth, A.H. The cell survival kinase SGK 1 and its targets FOXO 3a and NDRG 1 in aged human brain. Neuropathol. Appl. Neurobiol. 2013, 39, 623–633. [Google Scholar] [CrossRef]
- Martin-Batista, E.; Maglio, L.E.; Armas-Capote, N.; Hernández, G.; de la Rosa, D.A.; Giraldez, T. SGK1. 1 limits brain damage after status epilepticus through M current-dependent and independent mechanisms. Neurobiol. Dis. 2021, 153, 105317. [Google Scholar] [CrossRef] [PubMed]
- Kwon, O.C.; Song, J.J.; Yang, Y.; Kim, S.H.; Kim, J.Y.; Seok, M.J.; Hwang, I.; Yu, J.W.; Karmacharya, J.; Maeng, H.J. SGK1 inhibition in glia ameliorates pathologies and symptoms in Parkinson disease animal models. EMBO Mol. Med. 2021, 13, e13076. [Google Scholar] [CrossRef]
- Nuber, U.A.; Kriaucionis, S.; Roloff, T.C.; Guy, J.; Selfridge, J.; Steinhoff, C.; Schulz, R.; Lipkowitz, B.; Ropers, H.H.; Holmes, M.C. Up-regulation of glucocorticoid-regulated genes in a mouse model of Rett syndrome. Hum. Mol. Genet. 2005, 14, 2247–2256. [Google Scholar] [CrossRef]
- Géranton, S.M.; Morenilla-Palao, C.; Hunt, S.P. A role for transcriptional repressor methyl-CpG-binding protein 2 and plasticity-related gene serum-and glucocorticoid-inducible kinase 1 in the induction of inflammatory pain states. J. Neurosci. 2007, 27, 6163–6173. [Google Scholar] [CrossRef] [PubMed]
- Itoh, M.; Ide, S.; Takashima, S.; Kudo, S.; Nomura, Y.; Segawa, M.; Kubota, T.; Mori, H.; Tanaka, S.; Horie, H. Methyl CpG-binding protein 2 (a mutation of which causes Rett syndrome) directly regulates insulin-like growth factor binding protein 3 in mouse and human brains. J. Neuropathol. Exp. Neurol. 2007, 66, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Payet, L.D.; Wang, X.-H.; Baxter, R.C.; Firth, S.M. Amino-and carboxyl-terminal fragments of insulin-like growth factor (IGF) binding protein-3 cooperate to bind IGFs with high affinity and inhibit IGF receptor interactions. Endocrinology 2003, 144, 2797–2806. [Google Scholar] [CrossRef]
- Cubbage, M.; Suwanichkul, A.; Powell, D. Insulin-like growth factor binding protein-3. Organization of the human chromosomal gene and demonstration of promoter activity. J. Biol. Chem. 1990, 265, 12642–12649. [Google Scholar] [CrossRef]
- Modric, T.; Silha, J.V.; Shi, Z.; Gui, Y.; Suwanichkul, A.; Durham, S.K.; Powell, D.R.; Murphy, L.J. Phenotypic manifestations of insulin-like growth factor-binding protein-3 overexpression in transgenic mice. Endocrinology 2001, 142, 1958–1967. [Google Scholar] [CrossRef]
- StühMer, T.; Anderson, S.A.; Ekker, M.; Rubenstein, J.L. Ectopic expression of the Dlx genes induces glutamic acid decarboxylase and Dlx expression. Development 2002, 129, 245–252. [Google Scholar] [CrossRef]
- Miyano, M.; Horike, S.-I.; Cai, S.; Oshimura, M.; Kohwi-Shigematsu, T. DLX5 expression is monoallelic and Dlx5 is up-regulated in the Mecp2-null frontal cortex. J. Cell. Mol. Med. 2008, 12, 1188. [Google Scholar] [CrossRef] [PubMed]
- Schüle, B.; Li, H.H.; Fisch-Kohl, C.; Purmann, C.; Francke, U. DLX5 and DLX6 expression is biallelic and not modulated by MeCP2 deficiency. Am. J. Hum. Genet. 2007, 81, 492–506. [Google Scholar] [CrossRef]
- Li, S.; Huang, H.; Wei, X.; Ye, L.; Ma, M.; Ling, M.; Wu, Y. The recycling of AMPA receptors/GABAa receptors is related to neuronal excitation/inhibition imbalance and may be regulated by KIF5A. Ann. Transl. Med. 2022, 10, 1103. [Google Scholar] [CrossRef]
- Davenport, E.C.; Szulc, B.R.; Drew, J.; Taylor, J.; Morgan, T.; Higgs, N.F.; López-Doménech, G.; Kittler, J.T. Autism and schizophrenia-associated CYFIP1 regulates the balance of synaptic excitation and inhibition. Cell Rep. 2019, 26, 2037–2051.e6. [Google Scholar] [CrossRef]
- Cox, O.H.; Song, H.Y.; Garrison-Desany, H.M.; Gadiwalla, N.; Carey, J.L.; Menzies, J.; Lee, R.S. Characterization of glucocorticoid-induced loss of DNA methylation of the stress-response gene Fkbp5 in neuronal cells. Epigenetics 2021, 16, 1377–1397. [Google Scholar] [CrossRef]
- Peddada, S.; Yasui, D.H.; LaSalle, J.M. Inhibitors of differentiation (ID1, ID2, ID3 and ID4) genes are neuronal targets of MeCP2 that are elevated in Rett syndrome. Hum. Mol. Genet. 2006, 15, 2003–2014. [Google Scholar] [CrossRef] [PubMed]
- Chu, Y.-H.; Lin, J.-d.; Nath, S.; Schachtrup, C. Id proteins: Emerging roles in CNS disease and targets for modifying neural stemcell behavior. Cell Tissue Res. 2022, 387, 433–449. [Google Scholar] [CrossRef]
- Jung, S.; Park, R.-H.; Kim, S.; Jeon, Y.-J.; Ham, D.-S.; Jung, M.-Y.; Kim, S.-S.; Lee, Y.-D.; Park, C.-H.; Suh-Kim, H. Id proteins facilitate self-renewal and proliferation of neural stem cells. Stem Cells Dev. 2010, 19, 831–841. [Google Scholar] [CrossRef]
- Langlands, K.; Yin, X.; Anand, G.; Prochownik, E.V. Differential interactions of Id proteins with basic-helix-loop-helix transcription factors. J. Biol. Chem. 1997, 272, 19785–19793. [Google Scholar] [CrossRef] [PubMed]
- Deng, V.; Matagne, V.; Banine, F.; Frerking, M.; Ohliger, P.; Budden, S.; Pevsner, J.; Dissen, G.A.; Sherman, L.S.; Ojeda, S.R. FXYD1 is an MeCP2 target gene overexpressed in the brains of Rett syndrome patients and Mecp2-null mice. Hum. Mol. Genet. 2007, 16, 640–650. [Google Scholar] [CrossRef] [PubMed]
- Matagne, V.; Wondolowski, J.; Frerking, M.; Shahidullah, M.; Delamere, N.A.; Sandau, U.S.; Budden, S.; Ojeda, S.R. Correcting deregulated Fxyd1 expression rescues deficits in neuronal arborization and potassium homeostasis in MeCP2 deficient male mice. Brain Res. 2018, 1697, 45–52. [Google Scholar] [CrossRef]
- Rotaru, D.C.; Wallaard, I.; de Vries, M.; van der Bie, J.; Elgersma, Y. UBE3A expression during early postnatal brain development is required for proper dorsomedial striatal maturation. JCI Insight 2023, 8, e166073. [Google Scholar] [CrossRef]
- Samaco, R.C.; Hogart, A.; LaSalle, J.M. Epigenetic overlap in autism-spectrum neurodevelopmental disorders: MECP2 deficiency causes reduced expression of UBE3A and GABRB3. Hum. Mol. Genet. 2005, 14, 483–492. [Google Scholar] [CrossRef]
- Makedonski, K.; Abuhatzira, L.; Kaufman, Y.; Razin, A.; Shemer, R. MeCP2 deficiency in Rett syndrome causes epigenetic aberrations at the PWS/AS imprinting center that affects UBE3A expression. Hum. Mol. Genet. 2005, 14, 1049–1058. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Chahrour, M.; Ben-Shachar, S.; Lim, J. Ube3a/E6AP is involved in a subset of MeCP2 functions. Biochem. Biophys. Res. Commun. 2013, 437, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Park, M.J.; Aja, S.; Li, Q.; Degano, A.L.; Penati, J.; Zhuo, J.; Roe, C.R.; Ronnett, G.V. Anaplerotic triheptanoin diet enhances mitochondrial substrate use to remodel the metabolome and improve lifespan, motor function, and sociability in MeCP2-null mice. PLoS ONE 2014, 9, e109527. [Google Scholar] [CrossRef]
- Senior, A.W.; Evans, R.; Jumper, J.; Kirkpatrick, J.; Sifre, L.; Green, T.; Qin, C.; Žídek, A.; Nelson, A.W.; Bridgland, A. Protein structure prediction using multiple deep neural networks in the 13th Critical Assessment of Protein Structure Prediction (CASP13). Proteins Struct. Funct. Bioinform. 2019, 87, 1141–1148. [Google Scholar] [CrossRef]
- Senior, A.W.; Evans, R.; Jumper, J.; Kirkpatrick, J.; Sifre, L.; Green, T.; Qin, C.; Žídek, A.; Nelson, A.W.; Bridgland, A. Improved protein structure prediction using potentials from deep learning. Nature 2020, 577, 706–710. [Google Scholar] [CrossRef]
- Mok, R.S.; Zhang, W.; Sheikh, T.I.; Pradeepan, K.; Fernandes, I.R.; DeJong, L.C.; Benigno, G.; Hildebrandt, M.R.; Mufteev, M.; Rodrigues, D.C. Wide spectrum of neuronal and network phenotypes in human stem cell-derived excitatory neurons with Rett syndrome-associated MECP2 mutations. Transl. Psychiatry 2022, 12, 450. [Google Scholar] [CrossRef]
- Lamothe, H.; Schreiweis, C.; Mondragón-González, L.S.; Rebbah, S.; Lavielle, O.; Mallet, L.; Burguière, E. The Sapap3−/− mouse reconsidered as a comorbid model expressing a spectrum of pathological repetitive behaviours. Transl. Psychiatry 2023, 13, 26. [Google Scholar] [CrossRef] [PubMed]
- Mahgoub, M.; Adachi, M.; Suzuki, K.; Liu, X.; Kavalali, E.T.; Chahrour, M.H.; Monteggia, L.M. MeCP2 and histone deacetylases 1 and 2 in dorsal striatum collectively suppress repetitive behaviors. Nat. Neurosci. 2016, 19, 1506–1512. [Google Scholar] [CrossRef]
- Chao, H.-T.; Chen, H.; Samaco, R.C.; Xue, M.; Chahrour, M.; Yoo, J.; Neul, J.L.; Gong, S.; Lu, H.-C.; Heintz, N. Dysfunction in GABA signalling mediates autism-like stereotypies and Rett syndrome phenotypes. Nature 2010, 468, 263–269. [Google Scholar] [CrossRef]
- Dani, V.S.; Chang, Q.; Maffei, A.; Turrigiano, G.G.; Jaenisch, R.; Nelson, S.B. Reduced cortical activity due to a shift in the balance between excitation and inhibition in a mouse model of Rett syndrome. Proc. Natl. Acad. Sci. USA 2005, 102, 12560–12565. [Google Scholar] [CrossRef]
- Medrihan, L.; Tantalaki, E.; Aramuni, G.; Sargsyan, V.; Dudanova, I.; Missler, M.; Zhang, W. Early defects of GABAergic synapses in the brain stem of a MeCP2 mouse model of Rett syndrome. J. Neurophysiol. 2008, 99, 112–121. [Google Scholar] [CrossRef]
- Samaco, R.C.; Mandel-Brehm, C.; Chao, H.-T.; Ward, C.S.; Fyffe-Maricich, S.L.; Ren, J.; Hyland, K.; Thaller, C.; Maricich, S.M.; Humphreys, P. Loss of MeCP2 in aminergic neurons causes cell-autonomous defects in neurotransmitter synthesis and specific behavioral abnormalities. Proc. Natl. Acad. Sci. USA 2009, 106, 21966–21971. [Google Scholar] [CrossRef] [PubMed]
- Thomas, A.; Burant, A.; Bui, N.; Graham, D.; Yuva-Paylor, L.A.; Paylor, R. Marble burying reflects a repetitive and perseverative behavior more than novelty-induced anxiety. Psychopharmacology 2009, 204, 361–373. [Google Scholar] [CrossRef]
- Fahn, S. The history of dopamine and levodopa in the treatment of Parkinson’s disease. Mov. Disord. 2008, 23, S497–S508. [Google Scholar] [CrossRef] [PubMed]
- Temudo, T.; Ramos, E.; Dias, K.; Barbot, C.; Vieira, J.P.; Moreira, A.; Calado, E.; Carrilho, I.; Oliveira, G.; Levy, A. Movement disorders in Rett syndrome: An analysis of 60 patients with detected MECP2 mutation and correlation with mutation type. Mov. Disord. 2008, 23, 1384–1390. [Google Scholar] [CrossRef]
- Crook, J.; Hendrickson, A.; Robinson, F. Co-localization of glycine and gaba immunoreactivity in interneurons in Macaca monkey cerebellar cortex. Neuroscience 2006, 141, 1951–1959. [Google Scholar] [CrossRef] [PubMed]
- Simat, M.; Parpan, F.; Fritschy, J.M. Heterogeneity of glycinergic and gabaergic interneurons in the granule cell layer of mouse cerebellum. J. Comp. Neurol. 2007, 500, 71–83. [Google Scholar] [CrossRef]
- Xing, H.; Cui, N.; Johnson, C.M.; Faisthalab, Z.; Jiang, C. Dual synaptic inhibitions of brainstem neurons by GABA and glycine with impact on Rett syndrome. J. Cell. Physiol. 2021, 236, 3615–3628. [Google Scholar] [CrossRef]
- Oldfors, A.; Sourander, P.; Armstrong, D.L.; Percy, A.K.; Witt-Engerström, I.; Hagberg, B.A. Rett syndrome: Cerebellar pathology. Pediatr. Neurol. 1990, 6, 310–314. [Google Scholar] [CrossRef]
- Murakami, J.W.; Courchesne, E.; Haas, R.; Press, G.; Yeung-Courchesne, R. Cerebellar and cerebral abnormalities in Rett syndrome: A quantitative MR analysis. AJR. Am. J. Roentgenol. 1992, 159, 177–183. [Google Scholar] [CrossRef]
- Ehrhart, F.; Coort, S.L.; Cirillo, E.; Smeets, E.; Evelo, C.T.; Curfs, L. New insights in Rett syndrome using pathway analysis for transcriptomics data. Wien. Med. Wochenschr. 2016, 166, 346. [Google Scholar] [CrossRef]
- Xu, F.-X.; Wang, X.-T.; Cai, X.-Y.; Liu, J.-Y.; Guo, J.-W.; Yang, F.; Chen, W.; Schonewille, M.; De Zeeuw, C.; Zhou, L. Purkinje-cell-specific MeCP2 deficiency leads to motor deficits and autistic-like behavior due to aberrations in PTP1B-TrkB-SK signaling. Cell Rep. 2023, 42, 113559. [Google Scholar] [CrossRef]
- Hagena, H.; Manahan-Vaughan, D. Interplay of hippocampal long-term potentiation and long-term depression in enabling memory representations. Philos. Trans. R. Soc. B Biol. Sci. 2024, 379, 20230229. [Google Scholar] [CrossRef] [PubMed]
- Belichenko, P.V.; Wright, E.E.; Belichenko, N.P.; Masliah, E.; Li, H.H.; Mobley, W.C.; Francke, U. Widespread changes in dendritic and axonal morphology in Mecp2-mutant mouse models of Rett syndrome: Evidence for disruption of neuronal networks. J. Comp. Neurol. 2009, 514, 240–258. [Google Scholar] [CrossRef]
- Nguyen, M.V.C.; Du, F.; Felice, C.A.; Shan, X.; Nigam, A.; Mandel, G.; Robinson, J.K.; Ballas, N. MeCP2 is critical for maintaining mature neuronal networks and global brain anatomy during late stages of postnatal brain development and in the mature adult brain. J. Neurosci. 2012, 32, 10021–10034. [Google Scholar] [CrossRef] [PubMed]
- Asaka, Y.; Jugloff, D.G.; Zhang, L.; Eubanks, J.H.; Fitzsimonds, R.M. Hippocampal synaptic plasticity is impaired in the Mecp2-null mouse model of Rett syndrome. Neurobiol. Dis. 2006, 21, 217–227. [Google Scholar] [CrossRef]
- Gogliotti, R.G.; Senter, R.K.; Fisher, N.M.; Adams, J.; Zamorano, R.; Walker, A.G.; Blobaum, A.L.; Engers, D.W.; Hopkins, C.R.; Daniels, J.S. mGlu7 potentiation rescues cognitive, social, and respiratory phenotypes in a mouse model of Rett syndrome. Sci. Transl. Med. 2017, 9, eaai7459. [Google Scholar] [CrossRef] [PubMed]
- Asgarihafshejani, A.; Raveendran, V.A.; Pressey, J.C.; Woodin, M.A. LTP is Absent in the CA1 Region of the Hippocampus of Male and Female Rett Syndrome Mouse Models. Neuroscience 2024, 537, 189–204. [Google Scholar] [CrossRef]
- Nelson, E.D.; Kavalali, E.T.; Monteggia, L.M. MeCP2-dependent transcriptional repression regulates excitatory neurotransmission. Curr. Biol. 2006, 16, 710–716. [Google Scholar] [CrossRef]
- Cohen, D.R.; Matarazzo, V.; Palmer, A.M.; Tu, Y.; Jeon, O.-H.; Pevsner, J.; Ronnett, G.V. Expression of MeCP2 in olfactory receptor neurons is developmentally regulated and occurs before synaptogenesis. Mol. Cell. Neurosci. 2003, 22, 417–429. [Google Scholar] [CrossRef] [PubMed]
- Palmer, A.; Qayumi, J.; Ronnett, G. MeCP2 mutation causes distinguishable phases of acute and chronic defects in synaptogenesis and maintenance, respectively. Molecular. Cell. Neurosci. 2008, 37, 794–807. [Google Scholar] [CrossRef]
- Ronnett, G.V.; Leopold, D.; Cai, X.; Hoffbuhr, K.C.; Moses, L.; Hoffman, E.P.; Naidu, S. Olfactory biopsies demonstrate a defect in neuronal development in Rett’s syndrome. Ann. Neurol. 2003, 54, 206–218. [Google Scholar] [CrossRef] [PubMed]
- Guideri, F.; Acampa, M. Sudden death and cardiac arrhythmias in Rett syndrome. Pediatr. Cardiol. 2005, 26, 111. [Google Scholar] [CrossRef]
- Axelrod, F.B.; Chelimsky, G.G.; Weese-Mayer, D.E. Pediatric autonomic disorders. Pediatrics 2006, 118, 309–321. [Google Scholar] [CrossRef]
- Lugaresi, E.; Cirignotta, F.; Montagna, P. Abnormal breathing in the Rett syndrome. Brain Dev. 1985, 7, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Schlüter, B.; Aguigah, G.; Buschatz, D.; Trowitzsch, E.; Aksu, F. Polysomnographic recordings of respiratory disturbances in Rett syndrome. J. Sleep Res. 1995, 4, 203–207. [Google Scholar] [CrossRef]
- Cirignotta, F.; Lugaresi, E.; Montagna, P.; Opitz, J.M.; Reynolds, J.F. Breathing impairment in Rett syndrome. Am. J. Med. Genet. 1986, 25, 167–173. [Google Scholar] [CrossRef]
- Weese-Mayer, D.E.; Lieske, S.P.; Boothby, C.M.; Kenny, A.S.; Bennett, H.L.; Silvestri, J.M.; Ramirez, J.-M. Autonomic nervous system dysregulation: Breathing and heart rate perturbation during wakefulness in young girls with Rett syndrome. Pediatr. Res. 2006, 60, 443–449. [Google Scholar] [CrossRef]
- Richter, D.W.; Manzke, T.; Wilken, B.; Ponimaskin, E. Serotonin receptors: Guardians of stable breathing. Trends Mol. Med. 2003, 9, 542–548. [Google Scholar] [CrossRef]
- Hodges, M.R.; Wehner, M.; Aungst, J.; Smith, J.C.; Richerson, G.B. Transgenic mice lacking serotonin neurons have severe apnea and high mortality during development. J. Neurosci. 2009, 29, 10341–10349. [Google Scholar] [CrossRef]
- Abdala, A.P.; Bissonnette, J.M.; Newman-Tancredi, A. Pinpointing brainstem mechanisms responsible for autonomic dysfunction in Rett syndrome: Therapeutic perspectives for 5-HT1A agonists. Front. Physiol. 2014, 5, 205. [Google Scholar] [CrossRef]
- He, F.; Sun, Y.E. Glial cells more than support cells? Int. J. Biochem. Cell Biol. 2007, 39, 661–665. [Google Scholar] [CrossRef]
- He, L.; Zhang, R.; Yang, M.; Lu, M. The role of astrocyte in neuroinflammation in traumatic brain injury. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2024, 1870, 166992. [Google Scholar] [CrossRef]
- Barres, B.A. The mystery and magic of glia: A perspective on their roles in health and disease. Neuron 2008, 60, 430–440. [Google Scholar] [CrossRef] [PubMed]
- Andoh-Noda, T.; Akamatsu, W.; Miyake, K.; Matsumoto, T.; Yamaguchi, R.; Sanosaka, T.; Okada, Y.; Kobayashi, T.; Ohyama, M.; Nakashima, K. Differentiation of multipotent neural stem cells derived from Rett syndrome patients is biased toward the astrocytic lineage. Mol. Brain 2015, 8, 31. [Google Scholar] [CrossRef] [PubMed]
- Forbes-Lorman, R.M.; Kurian, J.R.; Auger, A.P. MeCP2 regulates GFAP expression within the developing brain. Brain Res. 2014, 1543, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Messing, A.; Head, M.W.; Galles, K.; Galbreath, E.J.; Goldman, J.E.; Brenner, M. Fatal encephalopathy with astrocyte inclusions in GFAP transgenic mice. Am. J. Pathol. 1998, 152, 391–398. [Google Scholar]
- Maezawa, I.; Swanberg, S.; Harvey, D.; LaSalle, J.M.; Jin, L.-W. Rett syndrome astrocytes are abnormal and spread MeCP2 deficiency through gap junctions. J. Neurosci. 2009, 29, 5051–5061. [Google Scholar] [CrossRef]
- Sheldon, A.L.; Robinson, M.B. The role of glutamate transporters in neurodegenerative diseases and potential opportunities for intervention. Neurochem. Int. 2007, 51, 333–355. [Google Scholar] [CrossRef]
- Lehmann, C.; Bette, S.; Engele, J. High extracellular glutamate modulates expression of glutamate transporters and glutamine synthetase in cultured astrocytes. Brain Res. 2009, 1297, 1–8. [Google Scholar] [CrossRef]
- Eid, T.; Williamson, A.; Lee, T.S.W.; Petroff, O.A.; De Lanerolle, N.C. Glutamate and astrocytes—Key players in human mesial temporal lobe epilepsy? Epilepsia 2008, 49, 42–52. [Google Scholar] [CrossRef]
- Okabe, Y.; Takahashi, T.; Mitsumasu, C.; Kosai, K.-i.; Tanaka, E.; Matsuishi, T. Alterations of gene expression and glutamate clearance in astrocytes derived from an MeCP2-null mouse model of Rett syndrome. PLoS ONE 2012, 7, e35354. [Google Scholar] [CrossRef]
- Müller, M.; Can, K. Aberrant redox homoeostasis and mitochondrial dysfunction in Rett syndrome. Biochem. Soc. Trans. 2014, 42, 959–964. [Google Scholar] [CrossRef]
- Tomasello, D.L.; Barrasa, M.I.; Mankus, D.; Alarcon, K.I.; Lytton-Jean, A.K.; Liu, X.S.; Jaenisch, R. Mitochondrial dysfunction and increased reactive oxygen species production in MECP2 mutant astrocytes and their impact on neurons. Sci. Rep. 2024, 14, 20565. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.-W.; Horiuchi, M.; Wulff, H.; Liu, X.-B.; Cortopassi, G.A.; Erickson, J.D.; Maezawa, I. Dysregulation of glutamine transporter SNAT1 in Rett syndrome microglia: A mechanism for mitochondrial dysfunction and neurotoxicity. J. Neurosci. 2015, 35, 2516–2529. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, K.; Esposito, E.; Wang, X.; Terasaki, Y.; Liu, Y.; Xing, C.; Ji, X.; Lo, E.H. Transfer of mitochondria from astrocytes to neurons after stroke. Nature 2016, 535, 551–555. [Google Scholar] [CrossRef] [PubMed]
- Lampinen, R.; Belaya, I.; Saveleva, L.; Liddell, J.R.; Rait, D.; Huuskonen, M.T.; Giniatullina, R.; Sorvari, A.; Soppela, L.; Mikhailov, N. Neuron-astrocyte transmitophagy is altered in Alzheimer’s disease. Neurobiol. Dis. 2022, 170, 105753. [Google Scholar] [CrossRef]
- Sharma, K.; Singh, J.; Pillai, P.P. MeCP2 differentially regulate the myelin MBP and PLP protein expression in oligodendrocytes and C6 glioma. J. Mol. Neurosci. 2018, 65, 343–350. [Google Scholar] [CrossRef]
- Colantuoni, C.; Jeon, O.H.; Hyder, K.; Chenchik, A.; Khimani, A.H.; Narayanan, V.; Hoffman, E.P.; Kaufmann, W.E.; Naidu, S.; Pevsner, J. Gene expression profiling in postmortem Rett Syndrome brain: Differential gene expression and patient classification. Neurobiol. Dis. 2001, 8, 847–865. [Google Scholar] [CrossRef]
- Nguyen, M.V.; Felice, C.A.; Du, F.; Covey, M.V.; Robinson, J.K.; Mandel, G.; Ballas, N. Oligodendrocyte lineage cells contribute unique features to Rett syndrome neuropathology. J. Neurosci. 2013, 33, 18764–18774. [Google Scholar] [CrossRef] [PubMed]
- Alessio, N.; Riccitiello, F.; Squillaro, T.; Capasso, S.; Del Gaudio, S.; Di Bernardo, G.; Cipollaro, M.; Melone, M.A.B.; Peluso, G.; Galderisi, U. Neural stem cells from a mouse model of Rett syndrome are prone to senescence, show reduced capacity to cope with genotoxic stress, and are impaired in the differentiation process. Exp. Mol. Med. 2018, 50, 1. [Google Scholar] [CrossRef] [PubMed]
- Kahanovitch, U.; Patterson, K.C.; Hernandez, R.; Olsen, M.L. Glial Dysfunction in MeCP2 Deficiency Models: Implications for Rett Syndrome. Int. J. Mol. Sci. 2019, 20, 3813. [Google Scholar] [CrossRef] [PubMed]
- Lipi, B.; Jaldeep, L.; Prakash, P. Role of astrocytic MeCP2 in regulation of CNS myelination by affecting oligodendrocyte and neuronal physiology and axo–glial interactions. Exp. Brain Res. 2018, 236, 3015–3027. [Google Scholar] [CrossRef]
- Yoon, S.H.; Kim, G.Y.; Choi, G.T.; Do, J.T. Organ abnormalities caused by Turner syndrome. Cells 2023, 12, 1365. [Google Scholar] [CrossRef]
- McCauley, M.D.; Wang, T.; Mike, E.; Herrera, J.; Beavers, D.L.; Huang, T.-W.; Ward, C.S.; Skinner, S.; Percy, A.K.; Glaze, D.G. Pathogenesis of lethal cardiac arrhythmias in Mecp2 mutant mice: Implication for therapy in Rett syndrome. Sci. Transl. Med. 2011, 3, 113ra125. [Google Scholar] [CrossRef]
- Hara, M.; Takahashi, T.; Mitsumasu, C.; Igata, S.; Takano, M.; Minami, T.; Yasukawa, H.; Okayama, S.; Nakamura, K.; Okabe, Y. Disturbance of cardiac gene expression and cardiomyocyte structure predisposes Mecp2-null mice to arrhythmias. Sci. Rep. 2015, 5, 11204. [Google Scholar] [CrossRef]
- Ward, C.S.; Huang, T.-W.; Herrera, J.A.; Samaco, R.C.; Pitcher, M.R.; Herron, A.; Skinner, S.A.; Kaufmann, W.E.; Glaze, D.G.; Percy, A.K. Loss of MeCP2 causes urological dysfunction and contributes to death by kidney failure in mouse models of Rett syndrome. PLoS ONE 2016, 11, e0165550. [Google Scholar] [CrossRef]
- Wang, J.; Xiong, M.; Fan, Y.; Liu, C.; Wang, Q.; Yang, D.; Yuan, Y.; Huang, Y.; Wang, S.; Zhang, Y. Mecp2 protects kidney from ischemia-reperfusion injury through transcriptional repressing IL-6/STAT3 signaling. Theranostics 2022, 12, 3896–3910. [Google Scholar] [CrossRef]
- Cronk, J.C.; Derecki, N.C.; Ji, E.; Xu, Y.; Lampano, A.E.; Smirnov, I.; Baker, W.; Norris, G.T.; Marin, I.; Coddington, N. Methyl-CpG binding protein 2 regulates microglia and macrophage gene expression in response to inflammatory stimuli. Immunity 2015, 42, 679–691. [Google Scholar] [CrossRef]
- Zalosnik, M.; Fabio, M.; Bertoldi, M.; Castañares, C.; Degano, A. MeCP2 deficiency exacerbates the neuroinflammatory setting and autoreactive response during an autoimmune challenge. Sci. Rep. 2021, 11, 10997. [Google Scholar] [CrossRef]
- Kida, H.; Takahashi, T.; Nakamura, Y.; Kinoshita, T.; Hara, M.; Okamoto, M.; Okayama, S.; Nakamura, K.; Kosai, K.I.; Taniwaki, T.; et al. Pathogenesis of Lethal Aspiration Pneumonia in Mecp2-null Mouse Model for Rett Syndrome. Sci. Rep. 2017, 7, 12032. [Google Scholar] [CrossRef] [PubMed]
- De Felice, C.; Guazzi, G.; Rossi, M.; Ciccoli, L.; Signorini, C.; Leoncini, S.; Tonni, G.; Latini, G.; Valacchi, G.; Hayek, J. Unrecognized lung disease in classic Rett syndrome: A physiologic and high-resolution CT imaging study. Chest 2010, 138, 386–392. [Google Scholar] [CrossRef]
- Vashi, N.; Ackerley, C.; Post, M.; Justice, M.J. Aberrant lung lipids cause respiratory impairment in a Mecp2-deficient mouse model of Rett syndrome. Hum. Mol. Genet. 2021, 30, 2161–2176. [Google Scholar] [CrossRef] [PubMed]
- Panayotis, N.; Ehinger, Y.; Felix, M.S.; Roux, J.c. State-of-the-art therapies for Rett syndrome. Dev. Med. Child Neurol. 2023, 65, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Matagne, V.; Ehinger, Y.; Saidi, L.; Borges-Correia, A.; Barkats, M.; Bartoli, M.; Villard, L.; Roux, J.C. A codon-optimized Mecp2 transgene corrects breathing deficits and improves survival in a mouse model of Rett syndrome. Neurobiol. Dis. 2017, 99, 1–11. [Google Scholar] [CrossRef]
- Matagne, V.; Borloz, E.; Ehinger, Y.; Saidi, L.; Villard, L.; Roux, J.-C. Severe offtarget effects following intravenous delivery of AAV9-MECP2 in a female mouse model of Rett syndrome. Neurobiol. Dis. 2021, 149, 105235. [Google Scholar] [CrossRef]
- Gadalla, K.K.E.; Vudhironarit, T.; Hector, R.D.; Sinnett, S.; Bahey, N.G.; Bailey, M.E.S.; Gray, S.J.; Cobb, S.R. Development of a Novel AAV Gene Therapy Cassette with Improved Safety Features and Efficacy in a Mouse Model of Rett Syndrome. Mol. Ther. Methods Clin. Dev. 2017, 5, 180–190. [Google Scholar] [CrossRef]
- Deverman, B.E.; Pravdo, P.L.; Simpson, B.P.; Kumar, S.R.; Chan, K.Y.; Banerjee, A.; Wu, W.L.; Yang, B.; Huber, N.; Pasca, S.P.; et al. Cre-dependent selection yields AAV variants for widespread gene transfer to the adult brain. Nat. Biotechnol. 2016, 34, 204–209. [Google Scholar] [CrossRef]
- Morabito, G.; Giannelli, S.G.; Ordazzo, G.; Bido, S.; Castoldi, V.; Indrigo, M.; Cabassi, T.; Cattaneo, S.; Luoni, M.; Cancellieri, C.; et al. AAV-PHP.B-Mediated Global-Scale Expression in the Mouse Nervous System Enables GBA1 Gene Therapy for Wide Protection from Synucleinopathy. Mol. Ther. 2017, 25, 2727–2742. [Google Scholar] [CrossRef]
- Luoni, M.; Giannelli, S.; Indrigo, M.T.; Niro, A.; Massimino, L.; Iannielli, A.; Passeri, L.; Russo, F.; Morabito, G.; Calamita, P.; et al. Whole brain delivery of an instability-prone Mecp2 transgene improves behavioral and molecular pathological defects in mouse models of Rett syndrome. eLife 2020, 9, e52629. [Google Scholar] [CrossRef]
- Goertsen, D.; Flytzanis, N.C.; Goeden, N.; Chuapoco, M.R.; Cummins, A.; Chen, Y.; Fan, Y.; Zhang, Q.; Sharma, J.; Duan, Y.; et al. AAV capsid variants with brain-wide transgene expression and decreased liver targeting after intravenous delivery in mouse and marmoset. Nat. Neurosci. 2022, 25, 106–115. [Google Scholar] [CrossRef]
- Sadhu, C.; Lyons, C.; Oh, J.; Jagadeeswaran, I.; Gray, S.J.; Sinnett, S.E. The Efficacy of a Human-Ready miniMECP2 Gene Therapy in a Pre-Clinical Model of Rett Syndrome. Genes 2023, 15, 31. [Google Scholar] [CrossRef]
- Nishikura, K. Functions and regulation of RNA editing by ADAR deaminases. Annu. Rev. Biochem. 2010, 79, 321–349. [Google Scholar] [CrossRef]
- Paul, M.S.; Bass, B.L. Inosine exists in mRNA at tissue-specific levels and is most abundant in brain mRNA. EMBO J. 1998, 17, 1120–1127. [Google Scholar] [CrossRef]
- Sinnamon, J.R.; Kim, S.Y.; Corson, G.M.; Song, Z.; Nakai, H.; Adelman, J.P.; Mandel, G. Site-directed RNA repair of endogenous Mecp2 RNA in neurons. Proc. Natl. Acad. Sci. USA 2017, 114, E9395–E9402. [Google Scholar] [CrossRef]
- Sinnamon, J.R.; Kim, S.Y.; Fisk, J.R.; Song, Z.; Nakai, H.; Jeng, S.; McWeeney, S.K.; Mandel, G. In Vivo Repair of a Protein Underlying a Neurological Disorder by Programmable RNA Editing. Cell Rep. 2020, 32, 107878. [Google Scholar] [CrossRef]
- Sinnamon, J.R.; Jacobson, M.E.; Yung, J.F.; Fisk, J.R.; Jeng, S.; McWeeney, S.K.; Parmelee, L.K.; Chan, C.N.; Yee, S.P.; Mandel, G. Targeted RNA editing in brainstem alleviates respiratory dysfunction in a mouse model of Rett syndrome. Proc. Natl. Acad. Sci. USA 2022, 119, e2206053119. [Google Scholar] [CrossRef]
- Gadalla, K.K.; Bailey, M.E.; Cobb, S.R. MeCP2 and Rett syndrome: Reversibility and potential avenues for therapy. Biochem. J. 2011, 439, 1–14. [Google Scholar] [CrossRef]
- Carrette, L.L.G.; Wang, C.Y.; Wei, C.; Press, W.; Ma, W.; Kelleher, R.J., 3rd; Lee, J.T. A mixed modality approach towards Xi reactivation for Rett syndrome and other X-linked disorders. Proc. Natl. Acad. Sci. USA 2018, 115, E668–E675. [Google Scholar] [CrossRef]
- Przanowski, P.; Wasko, U.; Zheng, Z.; Yu, J.; Sherman, R.; Zhu, L.J.; McConnell, M.J.; Tushir-Singh, J.; Green, M.R.; Bhatnagar, S. Pharmacological reactivation of inactive X-linked Mecp2 in cerebral cortical neurons of living mice. Proc. Natl. Acad. Sci. USA 2018, 115, 7991–7996. [Google Scholar] [CrossRef]
- Qian, J.; Guan, X.; Xie, B.; Xu, C.; Niu, J.; Tang, X.; Li, C.H.; Colecraft, H.M.; Jaenisch, R.; Liu, X.S. Multiplex epigenome editing of MECP2 to rescue Rett syndrome neurons. Sci. Transl. Med. 2023, 15, eadd4666. [Google Scholar] [CrossRef]
- De Felice, C.; Della Ragione, F.; Signorini, C.; Leoncini, S.; Pecorelli, A.; Ciccoli, L.; Scalabri, F.; Marracino, F.; Madonna, M.; Belmonte, G.; et al. Oxidative brain damage in Mecp2-mutant murine models of Rett syndrome. Neurobiol. Dis. 2014, 68, 66–77. [Google Scholar] [CrossRef]
- Leoncini, S.; De Felice, C.; Signorini, C.; Zollo, G.; Cortelazzo, A.; Durand, T.; Galano, J.M.; Guerranti, R.; Rossi, M.; Ciccoli, L.; et al. Cytokine Dysregulation in MECP2- and CDKL5-Related Rett Syndrome: Relationships with Aberrant Redox Homeostasis, Inflammation, and omega-3 PUFAs. Oxid. Med. Cell. Longev. 2015, 2015, 421624. [Google Scholar] [CrossRef] [PubMed]
- Nance, E.; Kambhampati, S.P.; Smith, E.S.; Zhang, Z.; Zhang, F.; Singh, S.; Johnston, M.V.; Kannan, R.M.; Blue, M.E.; Kannan, S. Dendrimer-mediated delivery of N-acetyl cysteine to microglia in a mouse model of Rett syndrome. J. Neuroinflamm. 2017, 14, 252. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Min, X.; Xie, X.; Huang, M.; Liu, Y.; Sun, W.; Xu, G.; He, M.; He, K.; Li, Y.; et al. RIPK1 activation in Mecp2-deficient microglia promotes inflammation and glutamate release in RTT. Proc. Natl. Acad. Sci. USA 2024, 121, e2320383121. [Google Scholar] [CrossRef]
- Neul, J.L.; Percy, A.K.; Benke, T.A.; Berry-Kravis, E.M.; Glaze, D.G.; Marsh, E.D.; Lin, T.; Stankovic, S.; Bishop, K.M.; Youakim, J.M. Trofinetide for the treatment of Rett syndrome: A randomized phase 3 study. Nat. Med. 2023, 29, 1468–1475. [Google Scholar] [CrossRef] [PubMed]
- Hudu, S.A.; Elmigdadi, F.; Qtaitat, A.A.; Almehmadi, M.; Alsaiari, A.A.; Allahyani, M.; Aljuaid, A.; Salih, M.; Alghamdi, A.; Alrofaidi, M.A. Trofinetide for Rett syndrome: Highlights on the development and related inventions of the first USFDA-approved treatment for rare pediatric unmet medical need. J. Clin. Med. 2023, 12, 5114. [Google Scholar] [CrossRef]
- Mohammed, H.E.; Bady, Z.; Haseeb, M.E.; Aboeldahab, H.; Sharaf-Eldin, W.E.; Zaki, M.S. Is trofinetide a future treatment for Rett syndrome? A comprehensive systematic review and meta-analysis of randomized controlled trials. BMC Med. 2024, 22, 299. [Google Scholar] [CrossRef]
- Sansal, I.; Sellers, W.R. The biology and clinical relevance of the PTEN tumor suppressor pathway. J. Clin. Oncol. 2004, 22, 2954–2963. [Google Scholar] [CrossRef]
- Ricciardi, S.; Boggio, E.M.; Grosso, S.; Lonetti, G.; Forlani, G.; Stefanelli, G.; Calcagno, E.; Morello, N.; Landsberger, N.; Biffo, S. Reduced AKT/mTOR signaling and protein synthesis dysregulation in a Rett syndrome animal model. Hum. Mol. Genet. 2011, 20, 1182–1196. [Google Scholar] [CrossRef]
- Blue, M.E.; Kaufmann, W.E.; Bressler, J.; Eyring, C.; O’Driscoll, C.; Naidu, S.; Johnston, M.V. Temporal and regional alterations in NMDA receptor expression in Mecp2-null mice. Anat. Rec. 2011, 294, 1624–1634. [Google Scholar] [CrossRef]
- Patrizi, A.; Picard, N.; Simon, A.J.; Gunner, G.; Centofante, E.; Andrews, N.A.; Fagiolini, M. Chronic Administration of the N-Methyl-D-Aspartate Receptor Antagonist Ketamine Improves Rett Syndrome Phenotype. Biol. Psychiatry 2016, 79, 755–764. [Google Scholar] [CrossRef]
- Chen, W.G.; Chang, Q.; Lin, Y.; Meissner, A.; West, A.E.; Griffith, E.C.; Jaenisch, R.; Greenberg, M.E. Derepression of BDNF transcription involves calcium-dependent phosphorylation of MeCP2. Science 2003, 302, 885–889. [Google Scholar] [CrossRef] [PubMed]
- Ogier, M.; Wang, H.; Hong, E.; Wang, Q.; Greenberg, M.E.; Katz, D.M. Brain-derived neurotrophic factor expression and respiratory function improve after ampakine treatment in a mouse model of Rett syndrome. J. Neurosci. 2007, 27, 10912–10917. [Google Scholar] [CrossRef] [PubMed]
- Mohanta, S.K.; Yin, C.; Weber, C.; Godinho-Silva, C.; Veiga-Fernandes, H.; Xu, Q.J.; Chang, R.B.; Habenicht, A.J.R. Cardiovascular Brain Circuits. Circ. Res. 2023, 132, 1546–1565. [Google Scholar] [CrossRef]
- Adams, I.; Yang, T.; Longo, F.M.; Katz, D.M. Restoration of motor learning in a mouse model of Rett syndrome following long-term treatment with a novel small-molecule activator of TrkB. Dis. Models Mech. 2020, 13, dmm044685. [Google Scholar] [CrossRef]
- Fazzari, M.; Lunghi, G.; Carsana, E.V.; Valsecchi, M.; Spiombi, E.; Breccia, M.; Casati, S.R.; Pedretti, S.; Mitro, N.; Mauri, L. GM1 Oligosaccharide Ameliorates Rett Syndrome Phenotypes In Vitro and In Vivo via Trk Receptor Activation. Int. J. Mol. Sci. 2024, 25, 11555. [Google Scholar] [CrossRef] [PubMed]
- Schmid, D.A.; Yang, T.; Ogier, M.; Adams, I.; Mirakhur, Y.; Wang, Q.; Massa, S.M.; Longo, F.M.; Katz, D.M. A TrkB small molecule partial agonist rescues TrkB phosphorylation deficits and improves respiratory function in a mouse model of Rett syndrome. J. Neurosci. 2012, 32, 1803–1810. [Google Scholar] [CrossRef]
- Levitt, E.S.; Hunnicutt, B.J.; Knopp, S.J.; Williams, J.T.; Bissonnette, J.M. A selective 5-HT1a receptor agonist improves respiration in a mouse model of Rett syndrome. J. Appl. Physiol. 2013, 115, 1626–1633. [Google Scholar] [CrossRef]
- Newman-Tancredi, A. Biased agonism at serotonin 5-HT1A receptors: Preferential postsynaptic activity for improved therapy of CNS disorders. Neuropsychiatry 2011, 1, 149. [Google Scholar] [CrossRef]
- Frasca, A.; Miramondi, F.; Butti, E.; Indrigo, M.; Balbontin Arenas, M.; Postogna, F.M.; Piffer, A.; Bedogni, F.; Pizzamiglio, L.; Cambria, C.; et al. Neural precursor cells rescue symptoms of Rett syndrome by activation of the Interferon gamma pathway. EMBO Mol. Med. 2024, 16, 3218–3246. [Google Scholar] [CrossRef] [PubMed]
- Hao, S.; Tang, B.; Wu, Z.; Ure, K.; Sun, Y.; Tao, H.; Gao, Y.; Patel, A.J.; Curry, D.J.; Samaco, R.C.; et al. Forniceal deep brain stimulation rescues hippocampal memory in Rett syndrome mice. Nature 2015, 526, 430–434. [Google Scholar] [CrossRef]
- Tang, B.; Wu, Z.; Wang, Q.; Tang, J. Neuronal Network Activation Induced by Forniceal Deep Brain Stimulation in Mice. Genes 2025, 16, 210. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, G.; Lee, S.; Yoo, S.; Do, J.T. MECP2 Dysfunction in Rett Syndrome: Molecular Mechanisms, Multisystem Pathology, and Emerging Therapeutic Strategies. Int. J. Mol. Sci. 2025, 26, 8277. https://doi.org/10.3390/ijms26178277
Choi G, Lee S, Yoo S, Do JT. MECP2 Dysfunction in Rett Syndrome: Molecular Mechanisms, Multisystem Pathology, and Emerging Therapeutic Strategies. International Journal of Molecular Sciences. 2025; 26(17):8277. https://doi.org/10.3390/ijms26178277
Chicago/Turabian StyleChoi, Gyutae, Sanghyo Lee, Seungjae Yoo, and Jeung Tae Do. 2025. "MECP2 Dysfunction in Rett Syndrome: Molecular Mechanisms, Multisystem Pathology, and Emerging Therapeutic Strategies" International Journal of Molecular Sciences 26, no. 17: 8277. https://doi.org/10.3390/ijms26178277
APA StyleChoi, G., Lee, S., Yoo, S., & Do, J. T. (2025). MECP2 Dysfunction in Rett Syndrome: Molecular Mechanisms, Multisystem Pathology, and Emerging Therapeutic Strategies. International Journal of Molecular Sciences, 26(17), 8277. https://doi.org/10.3390/ijms26178277




