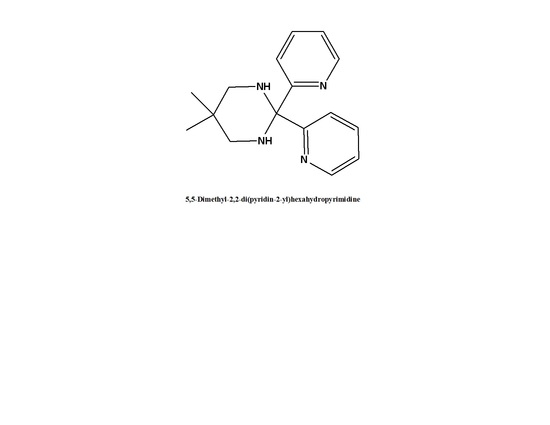
Introduction
The six-membered heterocyclic hexahydropyrimidine unit is present in several natural products and pharmaceutical agents of various pharmacological activities [
1]. Hexahydropyrimidines are also found in many bioactive compounds, like antiinflammatory and analgesic agents, fungicides, antibacterials, parasiticides, and antivirals [
2]. They also behave as prodrugs of pharmacologically active di- and polyamines [
3,
4]. Several derivatives of hexahydropyrimidines are used as polymers stabilizers [
5]. Due to their facile cleavage under mild acidic conditions, hexahydopyrimidines have been employed in organic synthesis as protective groups in the selective acylation by addition of 1,3-diamines [
6]. Furthermore, hexahydropyrimidines and some of their derivatives form good polydentate nitrogen donor complexes, they coordinate the transition metal ions in mono-, di-, or poly-coordination mode [
7,
8,
9]. The classic synthetic route leading to hexahydropyrimidine heterocyclic compounds involves the condensation of substituted propane-1,3-diamines with aldehydes and ketones [
2,
3,
4]. In this work, we prepared and characterized 5,5-dimethyl-2,2-di(pyridin-2-yl)hexahydropyrimidine, the X-ray structures of this compound and other derivatives which were previously published by our group [
9,
10,
11].
Result and Discussion
The title compound was synthesized by mixing equimolar amounts of 2,2-dimethylpropane-1,3-diamine and di(pyridin-2-yl)methanone in dichloromethane as shown in
Scheme 1. The product is colorless and soluble in CH
2Cl
2, partially in ROH, insoluble in water and non-polar solvents like
n-hexane.
The IR spectrum of 5,5-dimethyl-2,2-di(pyridin-2-yl)hexahydropyrimidine shows the absorption bands of the functional groups present as displayed in
Figure 1. The most significant absorption bands in the 3390, 3080, 2980–2700 cm
−1 region can be assigned to N-H aromatic C–H, aliphatic C-H stretching vibrations, respectively. Other characteristic bands due to other functional groups are also present in the expected regions, the formation of the product was confirmed by N-H stretching vibration shifts and C=O (of the ketone) disappearance, as seen in
Figure 1.
The elemental analysis of the compound is consistent with the proposed molecular formula (Calcd. for C
16H
20N
4: C, 71.61; H, 7.51; N, 20.88. Found: C, 71.25; H, 7.31; N, 20.55). EI-MS spectrum of the compound is in good agreement with the assigned structure and shows the experimental molecular ion [M
+]
m/z = 270 (269.2 theoretical) as shown in
Figure 2.
The electronic absorption spectrum of the compound was acquired in CH
2Cl
2,
Figure 3 shows the electronic absorption as expected (on the UV region only) at λ
max = 215 and 265 nm which assigned to intra-ligand π-π* / n-π* transitions.
1H-NMR and the 13C-NMR spectra data are in a good agreement with its assigned structure, signals of aromatic and aliphatic protons and carbon are cited to their positions.
Experimental Section
The IR spectra for samples were recorded using Perkin Elmer Spectrum 1000 FT-IR Spectrometer. The Uv-visible spectrum was measured by using a TU-1901 double-beam UV–visible spectrophotometer. High-resolution 1H, and 13C{1H} were recorded on Bruker DRX 250 spectrometer (Bruker, Mainz, Germany) (1H, 250 MHz and 13C, 62.5 MHz frequency) at 298 K. EI-MS data was obtained on a Finnigan 711A (8 kV) (PerkinElmer Inc., Waltham, MA, USA).
A solution of 2-dipyridlketone (0.25 g, 1.45 mmol) in dichloromethane (20 mL) was mixed with 2,2-dimethyl-1,3-propanediamine (0.11 mL, 1.5 mmoL) and allowed to stand for 1 h. The resulting mixture was concentrated under reduced pressure and the title compound was precipitated by the addition of 70 mL of n-hexane. The precipitates were filtered off, washed three times with 80 mL of distilled water.
Yield 88%, Colorless, Mp: 103 °C; Molecular formula C16H20N4; 1H-NMR (250 MHz, CDCl3): (ppm) 0.99 (s, 6H, CH3), 2.69 (br, 4H, CH2), 3.24 (br, 2H, NH), 7.10–8.20 (3m, 10H, Py–H), 13C-NMR (62.5 MHz, CDCl3): (ppm) 24.83 (2CH3), 28.29 (C), 53.43 (2CH2), 75.82 (CN2) 122.09, 122.15 (p-CH and m-CH), 136.70 (o-CH), 149.04 (m-CHN), 162.23 (i-C-Py). Calcd. for C16H20N4: C, 71.61; H, 7.51; N, 20.88. Found: C, 71.25; H, 7.31; N, 20.55). [M+] = 270 m/z. IR: 3390 cm−1N-H, 3080 cm−1C–H Py, 2980–2700 cm−1C-H aliphatic.
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Acknowledgments
I. Warad would like to thank AN-Najah National for their research support and Mr. Nafith Dweik for his kind collaboration.
Author Contributions
A. Abu-Obaid and A.I. Asadi analyzed the NMR data; A. Alruwaili, H. Atieh and Sh. Khlaif performed the experiments; T. B. Hadda, S. Radi and B. Hammouti helped in the results and discussion and writing the manuscript; and I. Warad wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Perillo, I.A.; Garcia, M.B.; Bisceglia, J.A.; Orelli, L.R. N-Arylhexahydropyrimidines. Part 1. Synthesis and 1H NMR Characterization of 1,3-Di and 1,2,3-Trisubstituted Derivatives. J. Heterocycl. Chem. 2002, 39, 655–661. [Google Scholar] [CrossRef]
- Drandarov, K.; Guggisberg, A.; Hesse, M. Macrocyclic Spermine Alkaloids from Verbascum: The (E/Z)-Isomeric Pairs (−)-(S)-Verbasitrine/(−)-(S)-Isoverbasitrine and (+)-(S)-Verbametrine/(+)-(S)-Isoverbametrine: Isolation, Structure Elucidation, and Synthesis. Helv. Chim. Acta 1999, 82, 229–237. [Google Scholar] [CrossRef]
- Hrvath, D.J. A Virtual Screening Approach Applied to the Search for Trypanothione Reductase Inhibitors. J. Med. Chem. 1999, 40, 2412–2423. [Google Scholar] [CrossRef] [PubMed]
- Mayr, M.; Wurst, K.; Ongania, K.-H.; Buchmester, M.R. 1,3-Dialkyl- and 1,3-Diaryl-3,4,5,6-tetrahydropyrimidin-2-ylidene Rhodium(i) and Palladium(II) Complexes: Synthesis, Structure, and Reactivity. Chem.-Eur. J. 2004, 10, 1256–1266. [Google Scholar] [CrossRef] [PubMed]
- Mayr, M.; Buchmeister, M.R. Rapid Screening of New Polymer-Supported Palladium(II) Bis(3,4,5,6-tetrahydropyrimidin-2-ylidenes. Macromol. Rapid Commun. 2004, 25, 231–236. [Google Scholar] [CrossRef]
- Katritzky, A.R.; Singh, S.K.; He, H.-Y. Novel Syntheses of Hexahydropyrimidines and Tetrahydroquinazolines. J. Org. Chem. 2002, 67, 3115–3117. [Google Scholar] [CrossRef] [PubMed]
- Ren, C.-X.; Ye, B.-H.; Zhu, H.-L.; Shi, J.-X.; Chen, X.-M. Syntheses, Structures and Photo luminescentProperties of Silver(I) Complexes with in situ Generated Hexahydropyrimidine Derivatives. Inorg. Chim. Acta 2004, 357, 443–450. [Google Scholar] [CrossRef]
- Schmidt, M.; Wiedemann, D.; Grohmann, A. First-row Transition Metal Complexes of a Novel Pentadentate Amine/Imine Ligand Containing a Hexahydropyrimidine Core. Inorg. Chim. Acta 2011, 374, 514–520. [Google Scholar] [CrossRef]
- Azam, M.; Ismail Warad, I.; Al-Resayes, S.; Alzaqri, N.; Khan, M.; Pallepogu, R.; Dwivedi, S.; Musarrat, J.; Shakir, M. Synthesis and structural characterization of Pd(II) complexes derived from perimidine ligand and their in vitro antimicrobial studies. J. Mol. Struct. 2013, 1047, 48–54. [Google Scholar] [CrossRef]
- Warad, I.; Alruwaili, A.; Al-Resayes, S.; Choudhary, M.I.; Yousuf, Y. 5,5-Dimethyl-2,2-bis(pyridin-2-yl)-1,3-diazinane. Acta Crystallogr. Sect. E: Struct. Rep. Online 2012, 68, o1786. [Google Scholar] [CrossRef] [PubMed]
- Haddad, S.F.; Warad, I.; Jodeh, S.; Ben Hadda, T. 2,2-Bis(pyridin-2-yl)-1,3-diazinane. Acta Crystallogr. Sect. E: Struct. Rep. Online 2013, 69, o569. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).