Tetra(phenylethynyl)tin Is a New Reagent for Solvent-Free Alkynylation of Imines
Abstract
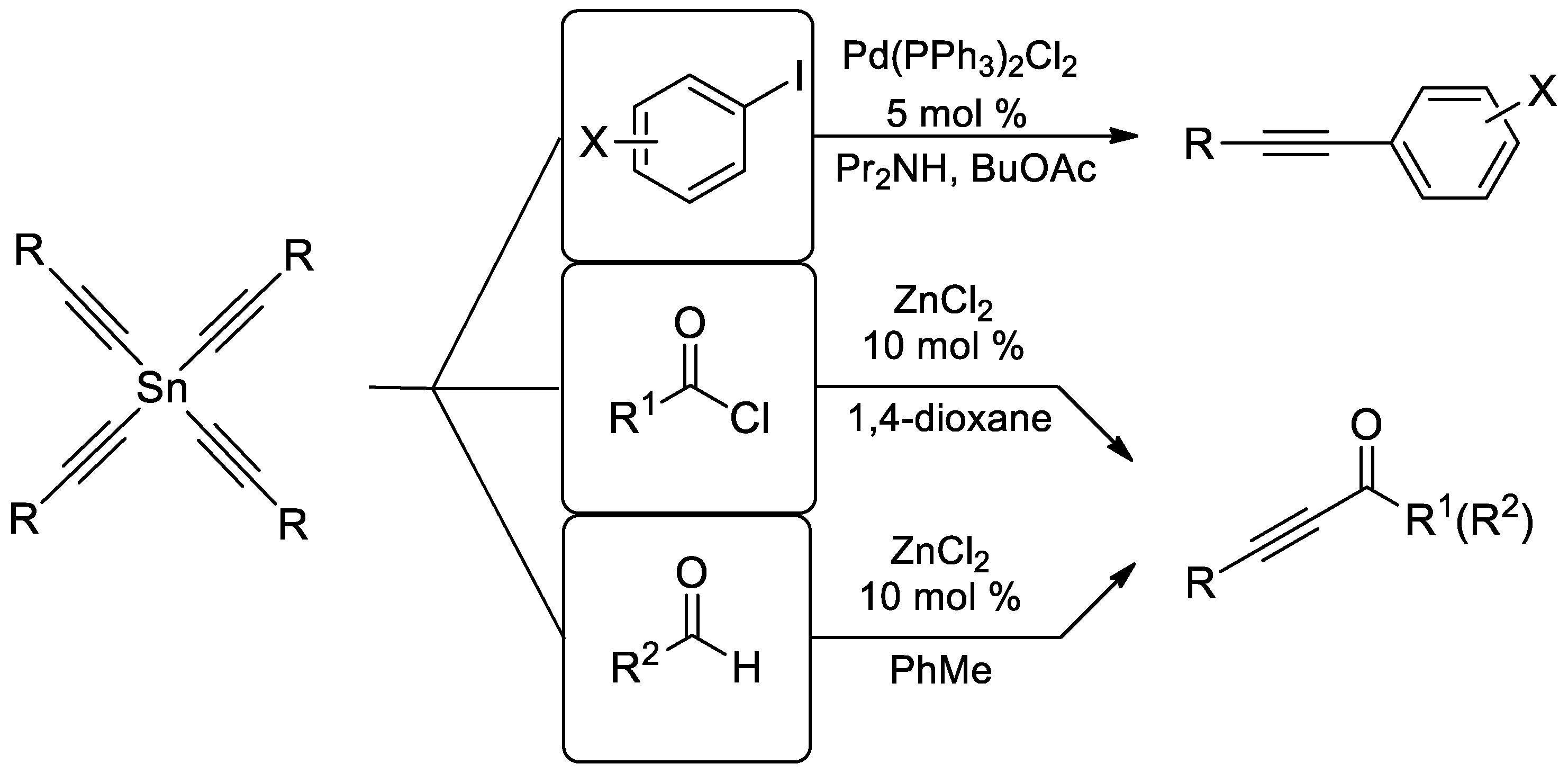
1. Introduction
2. Results and Discussion
3. Materials and Methods
General Procedure for the Synthesis of Propargylamine
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Diederich, F.; Stang, P.J.; Tykwinski, R.R. Acetylene Chemistry: Chemistry, Biology, and Material Science; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2005; pp. 1–508. [Google Scholar] [CrossRef]
- Trost, B.M.; Li, C.-J. Modern Alkyne Chemistry: Catalytic and Atom-Economic Transformations; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2005; pp. 1–402. [Google Scholar] [CrossRef]
- Rodygin, K.S.; Werner, G.; Kucherov, F.A.; Ananikov, V.P. Calcium carbide: A unique reagent for organic synthesis and nanotechnology. Chem. Asian J. 2016, 11, 965–976. [Google Scholar] [CrossRef] [PubMed]
- Rodygin, K.S.; Ledovskaya, M.S.; Voronin, V.V.; Lotsman, K.A.; Ananikov, V.P. Calcium carbide: Versatile synthetic applications, green methodology and sustainability. Eur. J. Org. Chem. 2021, 2021, 43–52. [Google Scholar] [CrossRef]
- Van Bonn, P.; Bolm, C. Mechanochemical synthesis of diarylethynes from aryl iodides and CaC2. Synlett 2022, 33, 893–897. [Google Scholar] [CrossRef]
- Nandy, S.; Paul, S.; Das, K.K.; Kumar, P.; Ghorai, D.; Panda, S. Synthesis and reactivity of alkynyl boron compounds. Org. Biomol. Chem. 2021, 19, 7276–7297. [Google Scholar] [CrossRef] [PubMed]
- Tani, T.; Sawatsugawa, Y.; Sano, Y.; Hirataka, Y.; Takahashi, N.; Hashimoto, S.; Sugiura, T.; Tsuchimoto, T. Alkynyl−B(dan)s in Various Palladium-Catalyzed Carbon−Carbon Bond-Forming Reactions Leading to Internal Alkynes, 1,4-Enynes, Ynones, and Multiply Substituted Alkenes. Adv. Synth. Catal. 2019, 361, 1815–1834. [Google Scholar] [CrossRef]
- Stefani, H.A.; Cella, R.; Vieira, A.S. Recent advances in organotrifluoroborates chemistry. Tetrahedron 2007, 63, 3623–3658. [Google Scholar] [CrossRef]
- Buendia, M.B.; Balin, J.-G.J.; Andersen, M.E.; Lian, Z.; Kramer, S. Copper-catalyzed alkynylation of benzylic C-H bonds with alkynylboronic esters. Synlett 2022, 33, 150–154. [Google Scholar] [CrossRef]
- Micouin, L.; Piccardi, R.; Turcaud, S.; Benedetti, E. Synthesis and Reactivity of Mixed Dimethylalkynylaluminum Reagents. Synthesis 2019, 50, 97–106. [Google Scholar] [CrossRef]
- Kafuta, K.; Rugen, C.J.; Heilmann, T.; Liu, T.; Golz, C.; Alcarazo, M. Reactivity of 5-(Alkynyl)dibenzothiophenium Salts: Synthesis of Diynes, Vinyl Sulfones, and Phenanthrenes. Eur. J. Org. Chem. 2021, 51, 4038–4048. [Google Scholar] [CrossRef]
- Waldecker, B.; Kraft, F.; Golz, C.; Alcarazo, M. 5-(alkynyl)dibenzothiophenium triflates: Sulfur-based reagents for electrophilic alkynylation. Angew. Chem. Int. Ed. 2018, 57, 12538–12542. [Google Scholar] [CrossRef]
- Amos, S.G.E.; Cavalli, D.; Le Vaillant, F.; Waser, J. Direct photoexcitation of ethynylbenziodoxolones: An alternative to photocatalysis for alkynylation reactions. Angew. Chem. Int. Ed. 2021, 60, 23827–23834. [Google Scholar] [CrossRef]
- Amos, S.G.E.; Waser, J. Radical alkynylations with EthynylBenziodoXolones: From photocatalysis to direct excitation. Chimia 2022, 76, 312–315. [Google Scholar] [CrossRef]
- García Ruano, J.L.; Alemán, J.; Parra, A.; Marzo, L. Sulfonyl Acetylenes as Alkynylating Reagents Under Radical or Anionic Conditions. Eur. J. Org. Chem. 2014, 2014, 1577–1588. [Google Scholar] [CrossRef]
- Andrei, M.; Undheim, K. Clarithromycin macrolides modified by unsaturation at the C10-position. Phytochem. Lett. 2022, 50, 128–133. [Google Scholar] [CrossRef]
- Makhloutah, A.; Hatych, D.; Chartier, T.; Rocard, L.; Goujon, A.; Felpin, F.-X.; Hudhomme, P. An investigation of palladium-catalyzed Stille-type cross-coupling of nitroarenes in perylenediimide series. Org. Biomol. Chem. 2022, 20, 362–365. [Google Scholar] [CrossRef]
- Yokose, D.; Nagashima, Y.; Kinoshita, S.; Nogami, J.; Tanaka, K. Enantioselective synthesis of axially chiral styrene-carboxylic esters by rhodium-catalyzed chelation-controlled [2+2+2] cycloaddition. Angew. Chem. Int. Ed. 2022, 61, e202202542. [Google Scholar] [CrossRef]
- Ouadoudi, O.; Kaehler, T.; Çevik, E.G.; Bolte, M.; Stöger, B.; Virovets, A.; Lerner, H.-W.; Wagner, M. Late-stage derivatization of a (B,O)2-doped perylene. Dalton Trans. 2022, 51, 13195–13198. [Google Scholar] [CrossRef]
- Lapkin, A.; Constable, D.J.C. Green Chemistry Metrics: Measuring and Monitoring Sustainable Processes. Green Chemistry Metrics: Measuring and Monitoring Sustainable Processes; Blackwell Publishing Ltd.: Hoboken, NJ, USA, 2009; pp. 1–324. [Google Scholar] [CrossRef]
- Levashov, A.S.; Andreev, A.A.; Konshin, V.V. Lewis acid promoted direct synthesis of tetraalkynylstannanes. Tetrahedron Lett. 2015, 56, 1870–1872. [Google Scholar] [CrossRef]
- Levashov, A.S.; Buryi, D.S.; Goncharova, O.V.; Konshin, V.V.; Dotsenko, V.V.; Andreev, A.A. Tetraalkynylstannanes in the stille cross coupling reaction: A new effective approach to arylalkynes. New J. Chem. 2017, 41, 2910–2918. [Google Scholar] [CrossRef]
- Levashov, A.S.; Aksenov, N.A.; Aksenova, I.V.; Konshin, V.V. Oxidative coupling of tetraalkynyltin with aldehydes leading to alkynyl ketones. New J. Chem. 2017, 41, 8297–8304. [Google Scholar] [CrossRef]
- Levashov, A.S.; Buryi, D.S. Lewis acid promoted reaction of tetraalkynylstannanes with acyl chlorides: An effective approach towards alkynyl ketones. Tetrahedron Lett. 2017, 58, 4476–4478. [Google Scholar] [CrossRef]
- Lauder, K.; Toscani, A.; Scalacci, N.; Castagnolo, D. Synthesis and reactivity of propargylamines in organic chemistry. Chem. Rev. 2017, 117, 14091–14200. [Google Scholar] [CrossRef] [PubMed]
- Jesin, I.; Nandi, G.C. Recent advances in the A3 coupling reactions and their applications. Eur. J. Org. Chem. 2019, 2019, 2704–2720. [Google Scholar] [CrossRef]
- Volkova, Y.; Baranin, S.; Zavarzin, I. A3 coupling reaction in the synthesis of heterocyclic compounds. Adv. Synth. Catal. 2021, 363, 40–61. [Google Scholar] [CrossRef]
- Monleón, A.; Blay, G.; Pedro, J.R. Catalytic enantioselective cyclopropylalkynylation of aldimines generated in situ from α-amido sulfones. Molecules 2022, 27, 3763. [Google Scholar] [CrossRef]
- Blay, G.; Monleon, A.; Pedro, J. Recent Developments in Asymmetric Alkynylation of Imines. Curr. Org. Chem. 2009, 13, 1498–1539. [Google Scholar] [CrossRef]
- Jiang, B.; Si, Y.-G. Lewis acid promoted alkynylation of imines with terminal alkynes: Simple, mild and efficient preparation of propargylic amines. Tetrahedron Lett. 2003, 44, 6767–6768. [Google Scholar] [CrossRef]
- Mokuolu, Q.F.; Duckmanton, P.A.; Hitchcock, P.B.; Wilson, C.; Blake, A.J.; Shukla, L.; Love, J.B. Early-late, mixed-metal compounds supported by amidophosphine ligands. Dalton Transactions 2004, 13, 1960–1970. [Google Scholar] [CrossRef]
- Nanni, D.; Pareschi, P.; Walton, J.C. An electron paramagnetic resonance study of intermediates generated from aromatic aldimines. J. Chem. Soc. Perkin Trans. 2002, 6, 1098–1104. [Google Scholar] [CrossRef]
- Yamashita, Y.; Noguchi, A.; Fushimi, S.; Hatanaka, M.; Kobayashi, S. Chiral metal salts as ligands for catalytic asymmetric mannich reactions with simple amides. J. Am. Chem. Soc. 2021, 143, 5598–5604. [Google Scholar] [CrossRef]
- Spesivaya, E.S.; Lupanova, I.A.; Konshina, D.N.; Konshin, V.V. Zn(OTf)2/i-Pr2NEt promoted synthesis of tetraalkynylsilanes. Tetrahedron Lett. 2021, 63, 152713. [Google Scholar] [CrossRef]
- Andreev, A.A.; Konshin, V.V.; Vinokurov, N.A.; Komarov, N.V. Synthesis of tri-and tetraalkynylgermanes. Russ. Chem. Bull. 2006, 55, 1430–1432. [Google Scholar] [CrossRef]
- Li, C.-J.; Wei, C. Highly efficient Grignard-type imine additions via C-H activation in water and under solvent-free conditions. Chem. Commun. 2002, 3, 268–269. [Google Scholar] [CrossRef]
- Zani, L.; Alesi, S.; Cozzi, P.G.; Bolm, C. Dimethylzinc-Mediated Alkynylation of Imines. J. Org. Chem. 2006, 71, 1558–1562. [Google Scholar] [CrossRef]


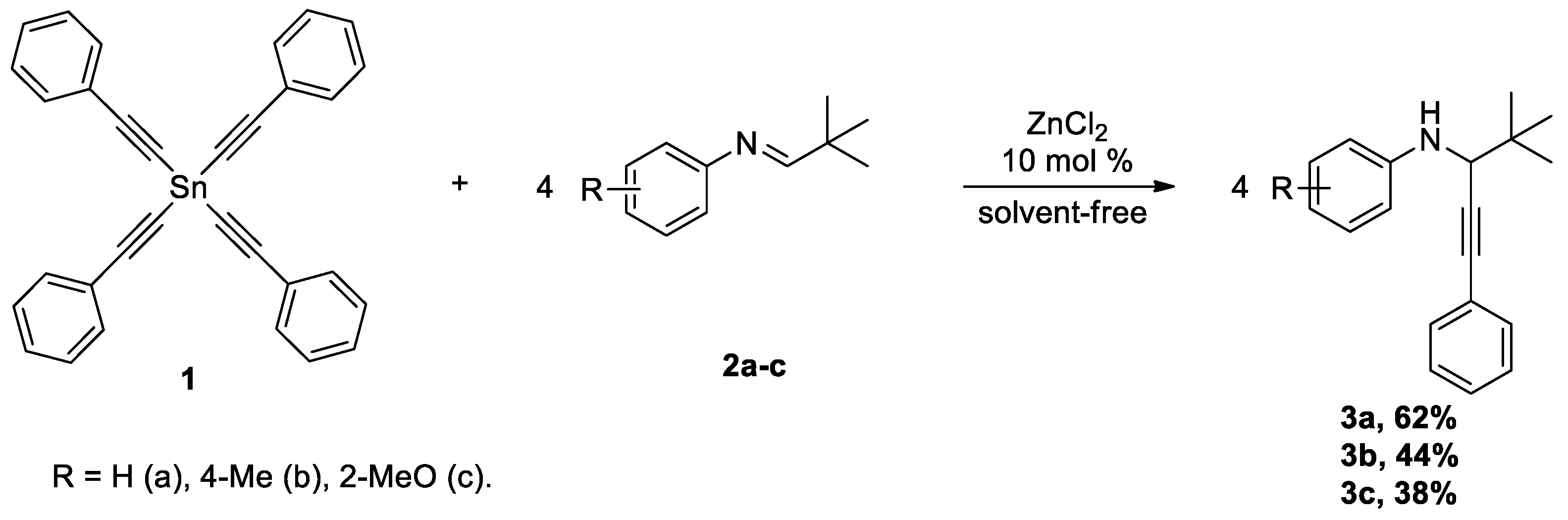
| Entry | Lewis Acid (10 mol %) | Solvent | Temp, °C | Time, h | Yield 3a,% (GS/MS) |
|---|---|---|---|---|---|
| 1 | ZnCl2 | PhMe | 100 | 3 | 36 |
| 2 | ZnCl2 | PhMe | 100 | 9 | 92 (7 **) |
| 3 | InCl3 | PhMe | 100 | 9 | 58 |
| 4 | AlCl3 | PhMe | 100 | 9 | 17 |
| 5 | BF3·OEt2 | PhMe | 100 | 9 | 52 |
| 6 | ZnCl2 | DCE | 80 | 9 | 30 |
| 7 | ZnCl2 | DCM | 30 | 9 | 3 |
| 8 | ZnCl2 | 1,4-dioxane | 100 | 9 | - |
| 9 | ZnCl2 | - | 100 | 12 | 98 |
| 10 | InBr3 | - | 100 | 9 | 25 |
| 11 | Sc(OTf)3 | - | 100 | 9 | 9 |
| 12 | Cu(OTf)2 | - | 100 | 9 | 18 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Levashov, A.S.; Dvirnaya, E.V.; Konshina, D.N.; Konshin, V.V. Tetra(phenylethynyl)tin Is a New Reagent for Solvent-Free Alkynylation of Imines. Molbank 2023, 2023, M1534. https://doi.org/10.3390/M1534
Levashov AS, Dvirnaya EV, Konshina DN, Konshin VV. Tetra(phenylethynyl)tin Is a New Reagent for Solvent-Free Alkynylation of Imines. Molbank. 2023; 2023(1):M1534. https://doi.org/10.3390/M1534
Chicago/Turabian StyleLevashov, Andrey S., Elena V. Dvirnaya, Dzhamilay N. Konshina, and Valery V. Konshin. 2023. "Tetra(phenylethynyl)tin Is a New Reagent for Solvent-Free Alkynylation of Imines" Molbank 2023, no. 1: M1534. https://doi.org/10.3390/M1534
APA StyleLevashov, A. S., Dvirnaya, E. V., Konshina, D. N., & Konshin, V. V. (2023). Tetra(phenylethynyl)tin Is a New Reagent for Solvent-Free Alkynylation of Imines. Molbank, 2023(1), M1534. https://doi.org/10.3390/M1534








