1. Introduction
Sleep is a periodic physiological phenomenon of human beings, which is vital to health and life conditions. However, there are many people with sleep disorders, which have a massive impact on daily life and work [
1]. Insomnia is a common sleep disorder, which means trouble falling or staying asleep, poor sleep quality, and decreased total sleep time [
2]. Insomnia can affect not only energy and emotion but also health, work performance, and quality of life, which has become an urgent social and medical problem [
3]. Currently, medication is the primary treatment for insomnia. Most medicines are helpful but have side effects. Therefore, exploring the neural and cellular mechanisms is significant in curing insomnia [
4].
The paraventricular thalamus (PVT) is located in the subependymal of the third ventricle, which is one of the midline nuclei of the thalamus [
5]. A recent study has shown that the PVT is important in regulating sleep and awakening [
6]. There are some spinal projections of the PVT to regulate sleep–wake cycles, which are involved in the regulation of physiological and pathological function. The PVT receives the hypocretin (Hcrt) secreted by the lateral hypothalamus (LH) [
7,
8], serotonin secreted by the dorsal raphe nucleus (DRN) [
9,
10], and norepinephrine (NE) secreted by the locus coeruleus (LC) [
5,
11] to regulate the excitability of the PVT neurons and promote the degree of arousal. On the other hand, previous studies revealed that the fibers of the PVT project to the nucleus accumbens (NAc), and this pathway was essential to the regulation of the arousal state [
6,
12]. Melatonin is considered the drug to help with falling asleep and treat sleep disorders, and the PVT is the regulatory site for melatonin. Melatonin production can regulate the activity of PVT neurons by acting on melatonin receptors in the PVT through cerebrospinal fluid [
13]. At the behavioral level, melatonin administration in the PVT promotes sleep occurrence, which is mediated primarily by MTR1 activation. Therefore, the PVT is a critical nucleus in the sleep–wake cycle, which needs to be detected and studied by high-resolution tools.
Currently, much research on deep brain nuclei is adopted via traditional microwire electrodes. Traditional nerve detection electrodes lacked enough temporal and spatial resolution. With the development of the micro-electro-mechanical system (MEMS), the microelectrode array (MEA) appeared and is now widely used in detecting electrophysiology and electrochemical signals of the brain [
14]. As a multichannel and high-resolution detection tool, the MEA can be positioned more accurately in relation to the target and cause less trauma during implantation [
15]. As metallic nanoparticles, platinum nanoparticles (PtNPs) are widely applied in medicine, electronics, and biotechnology due to their biocompatibility, antioxygenation, and surface chemistry [
16]. Modifying PtNPs can effectively reduce the impedance of recording sites and improve the SNR (signal-to-noise ratio) because PtNPs effectively increase the specific area.
Based on the above background, we designed an experiment using an MEA to detect neural signals in the PVT of rat brains before and after insomnia, which attempted to explain the relationship of insomnia to the PVT nucleus from the perspective of electrophysiology. The MEA fit with the PVT nucleus was designed and fabricated by the MEMS to record the neuronal signals in the PVT of control and insomnia rats. The result showed neurons of the PVT were activated in insomnia. Through further analysis, in the insomnia state, the synchronicity between adjacent neurons decreased, and burst-like firing appeared in the same neurons. Our research explored the changes in neural signals of the PVT before and after insomnia from the perspective of electrophysiology. It provided new ideas for the study of the PVT and the treatment of sleep disorders.
2. Materials and Methods
2.1. Reagents and Apparatus
The saline was purchased from Shuanghe Corporation (Beijing, China). P-Chloro-DL-phenylalanine (PCPA), Arabic gum, and phosphate-buffered saline (PBS) were purchased from Sigma-Aldrich (Shanghai, China), and 1,1′-Dioctadecyl-3,3,3′,3′-tetramethylindocarbocyanine perchlorate (DiI) was purchased from Beyotime Institute of Biotechnology (Shanghai, China). The chloroplatinic acid (H2PtCl6) and lead acetate [(CH3COO)2Pb] were purchased from Sinopharm Chemical Reagent (Shanghai, China). The isoflurane and anesthesia machine for small animals were purchased from RWD Life Science Co., Ltd. (Shenzhen, China).
The stereotaxic frame (51600) was purchased from Stoelting (Wood Dale, IL, USA). A micropositioner (model 2662) was purchased from David KOPF instrument (Tujunga, CA, USA). A 128-channel neuroelectrophysiological recording system was purchased from Blackrock Microsystems (Salt Lake City, UT, USA). A clinical cryostat (Leica CM1950) for cutting brain slices was purchased from Leica Biosystems (Barrington, IL, USA). The microscopes were Olympus BX51 (Olympus Corporation, Tokyo, Japan) and Leica M205C (Leica Biosystems, IL, USA), which were used for observing the MEA and brain slices.
2.2. Animals
Four male Sprague Dawley (SD) rats (weighted 350 g) were used for experiments. These rats were purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd. (Beijing, China). All rats were fed in the individual feeding cage within a 12/12 h dark–light cycle. The temperature was maintained at 25 ± 3 °C, and the humidity was maintained at 50–70%. The food and water were available ad libitum. All animal experiments were carried out with the permission of the Beijing Association on Laboratory Animal Care (Beijing, China) and approved by Institutional Animal Care and Use Committee at the Aerospace Information Research Institute, Chinese Academy of Sciences (AIRCAS, Beijing, China).
2.3. Design, Fabrication, and Modification of MEA
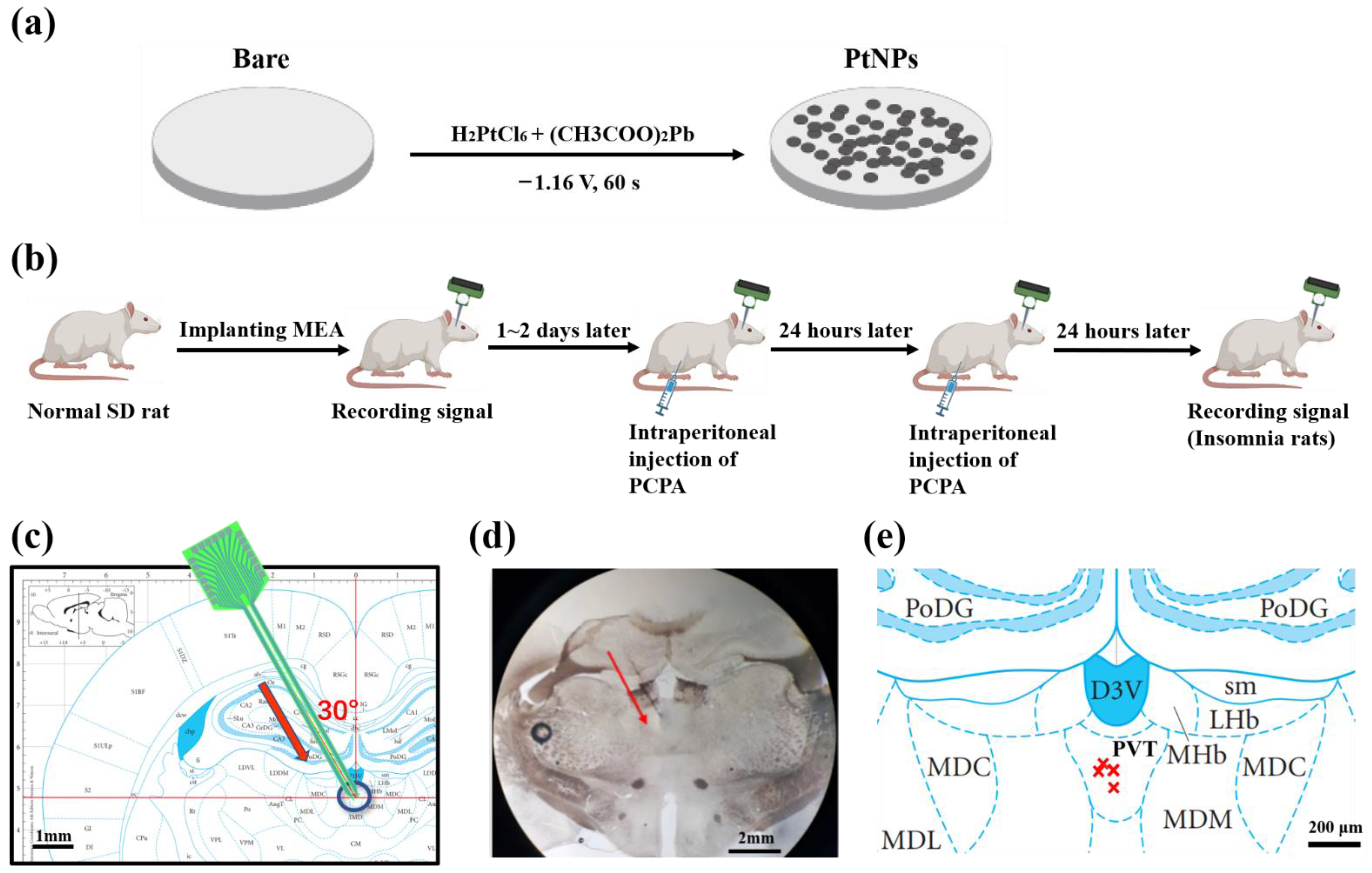
Based on the MEMS, we designed and fabricated a particular MEA to detect neural signals. The PVT area is located at a depth of 5.2 mm on the midline of the cerebral cross-section, and the shape of it is irregular. To match the depth, shape, and size of the PVT, an MEA with two tips was designed. There were eight equidistant electrophysiological recording sites and a 200 μm-long ground site on each tip (16 recording sites and 2 ground sites in total). Implanting the electrode vertically to the PVT broke the middle cerebral artery (MCA), which caused the rat to bleed to death. Therefore, we choose to tilt 30° for implantation.
The MEA consisted of three layers of material, including the base layer of Si (25 μm), the metal layer of Ti/Pt (30 nm/250 nm), and the insulating layer of SiO
2/Si
3N
4 (300 nm/500 nm). Silicon-on-insulator (SOI) wafer was used as the base of MEA.
Figure S1 shows the whole technological process of microelectrode preparation. First, a layer of 500 nm SiO
2 was deposited in the wafer by thermal oxidation. Next, the Ti/Pt metal layer was deposited by photolithography, sputtering, and lift-off. Then, SiO
2/Si
3N
4 insulating layer was deposited by plasma-enhanced chemical vapor deposition (PECVD). Through photolithography and deep reactive ion etching (DRIE), the conductive pads and sites were exposed, and the back oxide layer was etched. Finally, the etch of the back Si layer and the release of the MEA were accomplished by KOH wet etching. The individual released electrodes were connected to the printed circuit board (PCB) by an aluminum wire bond and then coated with protective room temperature vulcanization (RTV) silicone to complete the package.
The electrolyte solution was prepared by mixing chloroplatinic acid (48 mM in PBS) and lead acetate (4.2 mM in PBS) in a 1:1 volume ratio, then kept for 12 h [
17,
18]. As shown in
Figure 1a, using an electrochemical workstation (Gamry 600, Gamry Instrument, Warminster, PA, USA), the electrode tip was inserted into the electrolyte solution. PtNPs were electrodeposited on the electrode sites by chronoamperometry (−1.18 V, 60 s).
2.4. Surgical Operation
During the experiment, rats were anesthetized by isoflurane (3–5% for induction and 0.8–1.5% for maintenance) and fixed in the stereotaxic frame. The rat hair and scalp were cut off by a razer to expose the skull. The coordinate of craniotomy was 2.64 mm posterior and 2.8 mm lateral from the midline. Three sites of the skull were chosen to turn the skull screw as the grounding electrode and the stress point for the fixation of dental cement. To obtain firm fixation and less damage to the skull, two sites were contralateral with the skull window and one site was ipsilateral with the skull window. The positions of the sites were set to avoid the skull window. Then, the target locations of the skull were drilled by skull drill, and the scraps were cleaned by aurilave. Next, the rat and all apparatus were placed into an electromagnetic shielding box to eliminate external noise interference. The MEA was implanted at 30 degrees from the skull window (DV: 4.85 mm). Metal wires connected the shielded box (ground) and skull screws to improve the SNR. After being implanted in the target position, the MEA was fixed with dental cement.
2.5. Rat Model of Insomnia
PCPA was used as a medicine to induce rat models of insomnia, which is internationally recognized [
19,
20]. PCPA depletes 5-HT (5-hydroxytryptamine, an important neurotransmitter that regulates the sleep–wake cycle) in the central nervous system to induce insomnia. The decrease in 5-HT caused severe insomnia symptoms. PCPA was gradually metabolized in the body, and the insomnia symptoms slowly disappeared. Therefore, signals were detected periodically after modeling the insomnia disorder.
PCPA was dissolved in PBS solution (30 mg/mL), and Arabic gum (0.1%) was added to promote dissolution. After weighing the rat, the turbid liquid of PCPA was intraperitoneally injected (1 mL/100 g of rat weight). The above operation was repeated 24 h later. On the third day, the rats developed an insomniac state. Then, spikes and LFPs were recorded as the experimental group.
2.6. Experimental Strategy
As shown in
Figure 1b, the MEA was implanted into the PVT nucleus of the control rats. Then electrophysiological signals of the PVT neurons were recorded as the control group. After 1–2 days to recover from wounds and regain physical condition, PCPA was intraperitoneally injected twice, as described above. After the rats were in the insomnia state, electrophysiological signals of the PVT neurons were recorded again as the experimental group. Finally, the detected electrophysiological signals of the two groups were analyzed and compared to draw a reliable conclusion.
2.7. Signal Recording and Data Processing
Multichannel electrophysiological signals were acquired by a homemade electrophysiological recording system. The system was developed in the previous work of our group [
21], which included an implantable sensor (microelectrode array), a dual-function head-stage, and a low noise detection instrument.
The MEA was connected with the homemade electrophysiological recording system. Through amplifying circuit, filter circuit, and analog to digital converter (ADC), recorded electrophysiological signals were transmitted to the upper computer. The sampling frequency was set at 30 kHz to obtain a real-time neural signal. Spikes were extracted from measured data by a high pass filter (≥250 Hz), and LFPs were extracted by a low pass filter (≤250 Hz). The special algorithm was designed for spike sorting. Cluster analysis was used for sorting according to the waveform, peak, and duration of the signals. Moreover, the single unit with a refractory period greater than 1 ms was regarded as a neuron signal. Then, different types of spikes were sorted and the noise was separated from the signals.
NeuroExplorer 4 (Nex Technologies, Hays, KS, USA) was used for further statistics and analysis of spikes and LFPs. The results were exported, and Origin (OriginLab, Northampton, MA, USA) was used to draw graphs. All data were processed as mean ± SEM. The result p < 0.05 (ANOVA test) was regarded as statistical significance.
2.8. Histological Verification of Implant Position
As the PVT was located in the deep brain region, and the oblique implantation of the MEA could easily cause errors, we used the histological section to verify that the MEA was implanted into the PVT after in vivo detection. Before implantation, the cusp of the MEA was coated with fluorochrome dye (DiI). The above step was repeated twice every half hour to ensure that DiI adhered to the MEA. After finishing the electrophysiological recording, chloral hydrate was dissolved in saline (300 mg/kg body weight) to deeply narcotize rats by intraperitoneal injection. Then, the thoracic cavity of the rat was opened with a razor and hemostatic forceps under deep anesthesia. The brain was dehydrated by injecting saline (0.9%) and paraformaldehyde (4%) into the heart artery and put in the sucrose solution (20% and 30%) in turn to fixation. Coronal sections were cut into 50 μm by a clinical cryostat. The brain slices were taken onto the glass slide. The glass slide was covered with a drop of saline in advance to spread the brain slices. Next, excess saline was removed with absorbent paper. The glass slide was observed under the stereomicroscope. After adjusting the appropriate vision to show the whole brain slices, the image of the brain slices was captured.
The targeted position was marked on the rat brain atlas (
Figure 1c), and brain slices were observed under the microscope (
Figure 1d). By comparing the sections and rat brain atlas, we confirmed the rationality of the implantation method and the precision of implanting location.
Figure 2e shows the projection of recording points in the atlas, which reflected the electrode implantation accuracy.
4. Discussion
Accurately acquiring electrophysiological signals from the deep brain is critical in neurosciences. As a minimally invasive tool to detect neural signals, the MEA has significant advantages for deep brain detection in vivo. The characteristics of the MEA are high precision and high temporal-spatial resolution, which help detect the signal at the cellular level (μm). The electroplated PtNPs increase the specific surface area of the conductive site and the contact with neurons, improving SNR, the performance of impedance and phase for MEA.
Recent studies have shown that the PVT is involved in sleep–wake regulation, especially in regulating and maintaining awakening. Moreover, neurons of the PVT fired more frequently during wakefulness than sleep. This finding makes us interested in the PVT and sleep–wake regulation. We chose the classical insomnia model to probe the neural signal of the PVT. The symptoms of insomnia include fatigue, an inability to concentrate, or irritability [
25,
26]. Compared with control rats, the average spike firing rate and spike power increased for insomnia rats. In other words, PVT neurons were activated to fire frequently in insomnia, which confirmed that the PVT was the nucleus to regulate wakefulness, and insomnia led to less sleep.
LFP sampled the activities of a relatively localized population of neurons, which meant the signal was the concentrated expression of all local neuronal discharges. We analyzed the PSD of LFP in control and insomnia rats and found that the LFP power increased at 0–50 Hz after sleeplessness. Meanwhile, the proportion of LFP power decreased in the delta frequency band and increased in the beta frequency band. Delta waves help the brain to reduce awareness of the outside environment and focus inwardly, and sleep quality is directly related to delta waves [
27]. The activity of beta waves is evidently for processing information, associated with alertness, focus, and even agitation. For insomniacs, the LFP power of the delta frequency band decreased [
28]. The LFP power of the beta frequency band increased and extended to the neighboring frequency bands [
29]. Furthermore, there were also peaks of the average spike power in the delta frequency band, which corresponded with the peak of LFP power in the delta band. During insomnia, the spike firing rate increased, which increased spike power. It was speculated that the spike of higher power of the beta frequency in the insomnia state accumulated to the peak of LFP power in the beta frequency band. Our results also showed a lower proportion of LFP power at low frequency and a higher proportion of LFP power at high frequency in the insomnia state than in the control state. In the insomnia state, PVT neurons were highly activated at the cellular level, consistent with the macroscopical characteristics of LFP and apparent symptoms of insomnia disorder.
All physiological activities of the body were controlled and regulated by the brain, which was reflected in the neuron discharge pattern. Different physiological activities were regulated by one or more different kinds of neurons to meet body requirements. When most of the neurons were in the resting state, or they were activated in cooperation for the same physiological activity, there would be a strong synchronization between the spike firings of the neurons. The correlation coefficient was an important index to measure this synchronization. The increase in correlation coefficients meant the consistency of firing frequency, waveform, or amplitude, which indicated that the neurons maintained a similar or correlated state of physiological activities. The decline in correlation coefficients means the diversity of neuronal activity characteristics, which indicated that different kinds of neurons were activated and generated action potentials with various patterns at different times. In our research, the reduction of the cross-correlation coefficient indicated that the synchronism of discharges from different neurons in the PVT declined in the insomnia state. During sleep or anesthesia state, the body was inactive, and most neurons were at the resting stage, which induced high synchronism between neurons due to low neural activities in the brain. In the non-sleep state, more neurons were activated for more physiological activities. Therefore, there were more physiological activities of PVT neurons in the insomnia state, which meant difficulty maintaining the state of sleep.
The characteristics of insomnia were drowsiness and fatigue [
30]. When the individual is at a stage of fatigue, neurons discharge by burst firing to reduce energy consumption. By current gradient stimulation to the PVT, the firing patterns of PVT neurons were observed, which included tonic firing, initial bursting, delayed firing, single spiking, and reluctant firing [
31]. In our research, the observation of burst or burst-like firing is well explained.
There are two methods of signal transmission in the brain: electrical impulses firing on a single neuron (action potential) and neurotransmitters released between neurons (electrochemical signal). The neurons in the PVT are mainly excitatory glutamatergic neurons, which express vesicular glutamate transporter-2 (VGluT2). Meanwhile, there are no inhibitory GABAergic neurons in the PVT [
32]. To investigate the relationship between insomnia and neurotransmitters, the changes in the concentration of glutamate in the PVT during wakefulness are worth detecting and analyzing from the electrochemical perspective. The treatment for insomnia is also an important research direction. Moreover, many nuclei associated with sleep and wakefulness were located in the deep brain region. As a regulation method for many brain diseases, DBS (deep brain stimulus) is worthy of further study regarding the DBS regulation of the sleep–wake cycle and insomnia disorder.
5. Conclusions
In this study, we designed and fabricated a special implantable MEA to detect the electrophysiological neural signal in the PVT and modified PtNPs on the microelectrodes to improve detection performance, which made the impedance of recording sites decrease from 979.34 ± 387.77 kΩ to 41.6 ± 16.4 kΩ at 1 kHz. Our study showed that some neurons of the PVT became activated after insomnia modeling. The electrophysiological characteristics of PVT neurons were different in control and insomnia rats. Compared with control rats, the spike firing rate increased from 5.48 ± 0.28 spike/s to 7.39 ± 0.65 spike/s, and the spike power increased. The proportion of LFP power decreased in the delta frequency band and grew in the beta frequency band. The synchronism of discharge from neighboring neurons declined in insomnia rats. These results showed that the characteristics of insomnia were consistent with macroscopic insomnia symptoms. The signals at the cellular level were detected by the fabricated MEA, which conformed with the characteristic of LFP and insomnia symptoms.
Moreover, the synchronism of different neurons was reduced. The shortened discharge interval of the same neuron and the appearance of burst-like firing indicated the generation of a fatigued state, which proved the cellular mechanism in the PVT for insomnia. Our research provided a special MEA, a tool for studying the deep brain nuclei and the nuclei associated with insomnia. Meanwhile, we explored the neuron activity of the PVT in insomnia, which provided some thoughts for further research on the sleep–wake mechanisms and the treatment of sleep disorders in the future.