Recent Advances in Polysaccharide-Based Electrospun Nanofibers for Food Safety Detection
Abstract
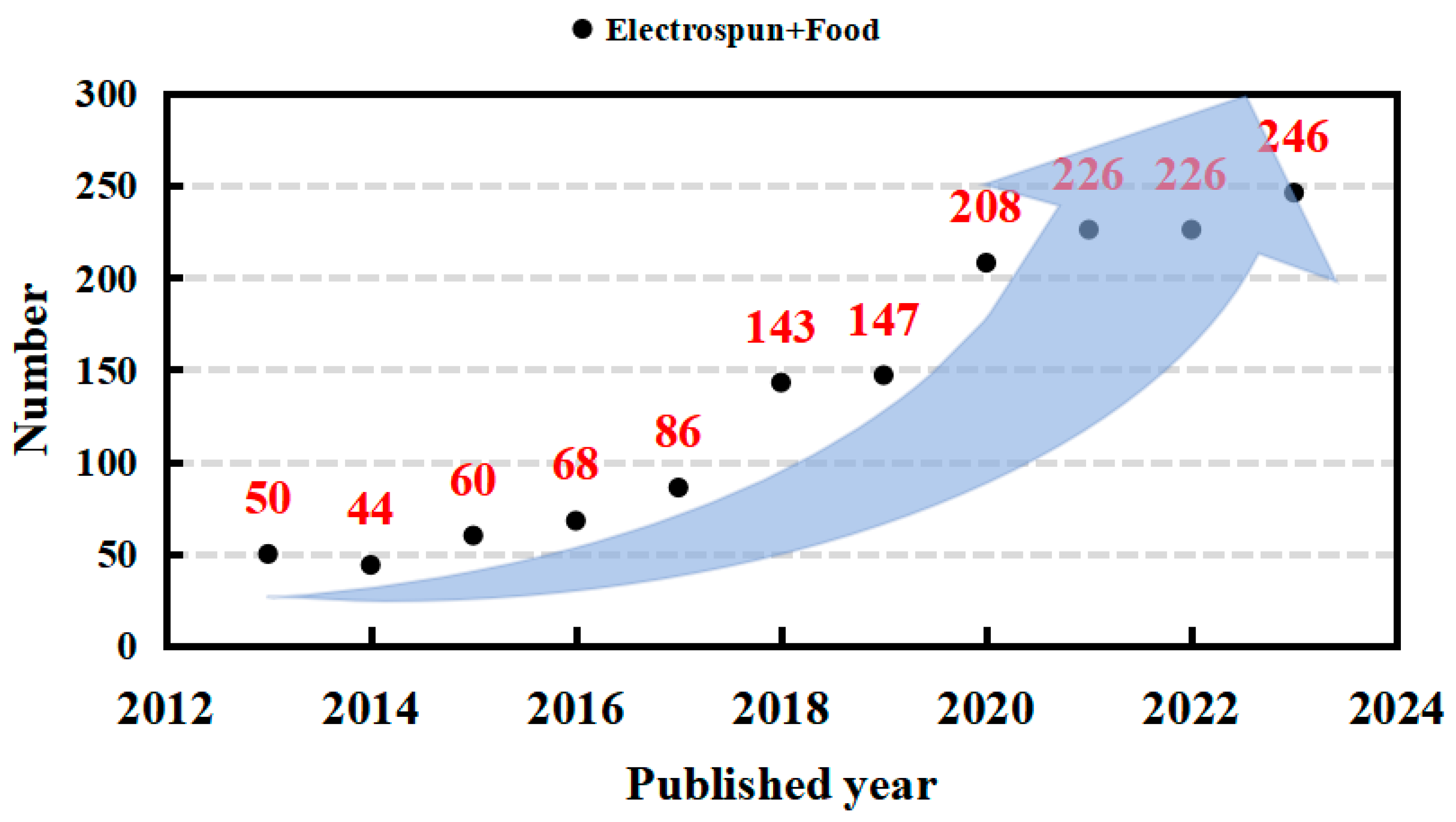
1. Introduction
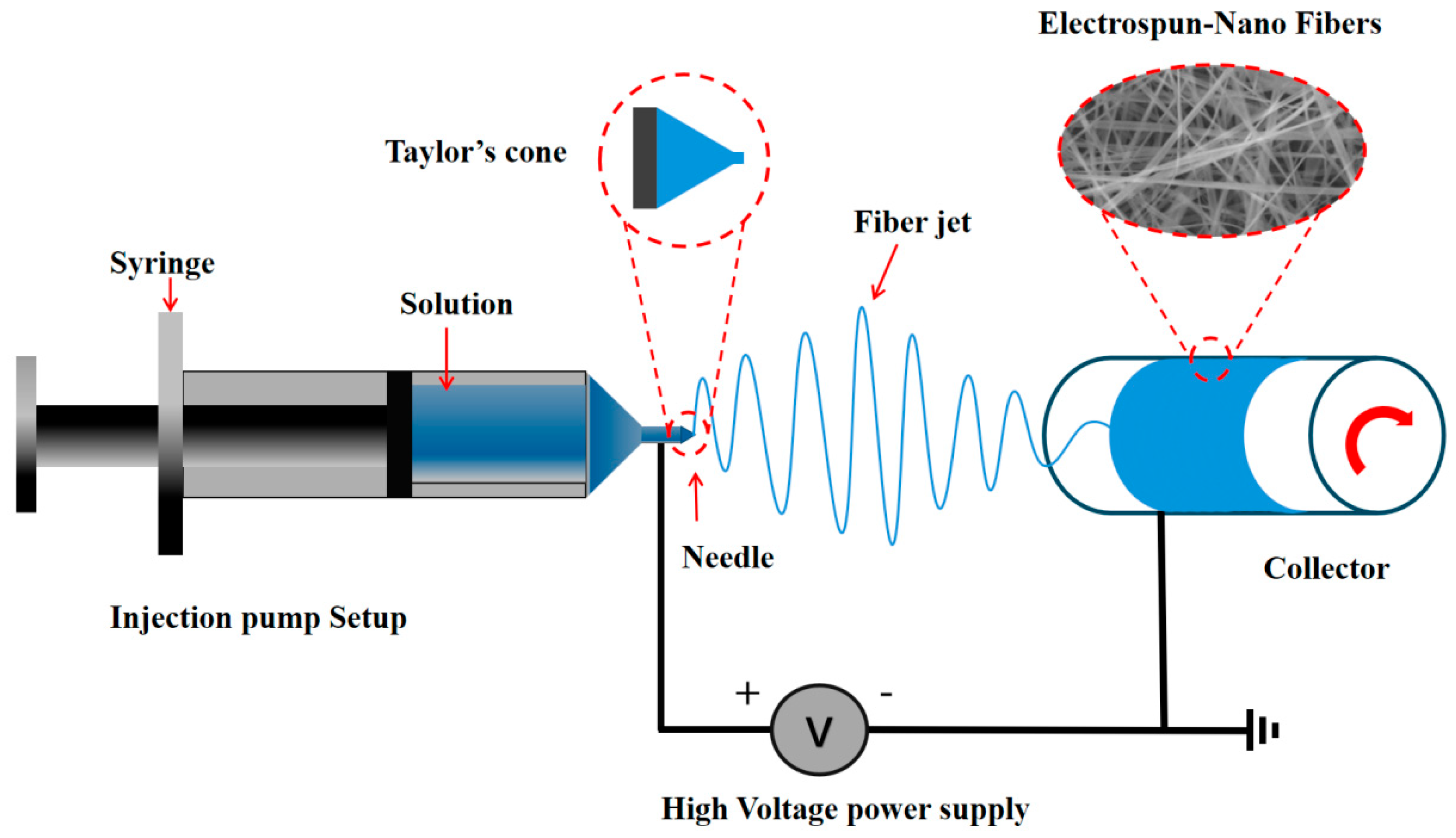
2. Electrospinning
3. Factors Influencing Electrospinning of Polysaccharides
3.1. Process Parameters
3.2. Properties of Polysaccharides Solution
3.3. Environmental Parameters
| Polysaccharide Polymer | Solvent System | Variable Parameter | Effect on Fiber Morphology | Account | References | |
|---|---|---|---|---|---|---|
| Process parameters | CS-Gelatin | Acetic acid | ↑High voltage | Diameter decrease | The flow rate and spinning distance are constant, the voltage is increased, and the fiber diameter is increased | Nafise Amiri [23] |
| Cellulose acetate (CA) | Acetone | ↑High voltage | Diameter decrease | The spinning distance is constant, decreasing or increasing the voltage at 12 kV will reduce the fiber diameter | Nicole Angel [21] | |
| CA/Ethyl cellulose | Acetone-DMF | ↑High voltage | Diameter increase | The spinning distance is constant and the diameter of nanofibers increases with the increase of voltage | Abdallah Refate [24] | |
| CS-Gelatin | Acetic acid | ↑Feed flow rate | Diameter increase | The voltage is constant and the diameter of the nanofibers increases with the increase of the flow rate | Nafise Amiri [23] | |
| Solution parameters | CS/PEO | Phosphoric acid | Add Polymer | More stable | The spinning stability of CS was improved by electrospinning by blending CS with PEO | Wei Pan [28] |
| SA/PEO/CaCl2/Glutaraldehyde/trifluoroacetic acid | Water | Add Polymer | More stable | SA was blended with other polymers for electrospinning to improve spinning stability | Qian Wang [29] | |
| CA/bark oil/clove bud oil | Acetone and dimethyl formaldehyde | Add Polymer | More balanced | The blending of cellulose and essential oil improves the evenness of spinning fibers | Maheshika Sethunga [34] | |
| CS | Acetic acid and trifluoroacetic acid | Add NaCl | More stable | Adding NaCl to CS solution can increase the conductivity and decrease the viscosity | Hengjie Su [27] | |
| CS/PEO | Acetic acid | Add NaCl | More stable | Adding NaCl to CS/PEO solution can increase the conductivity and decrease the viscosity | Varnaitė-Žuravliova [35] | |
| Environmental Parameters | CS | trifluoroacetic acid and dichloromethane | ↑Temperature | More balanced | The nanofibers are most uniform at 32 °C | Dan-Thuy Van-Phan [30] |
| CA | Acetone and dimethyl formamide | ↑Temperature | Load increase | Increasing the temperature from 22 °C to 52 °C can increase the yield | Awais Khatri [31] | |
| CA/Polyethylene | Acetone | ↑Temperature | More stable | CA/PEI films show an increase in thermal stability | Jinsoo Yoon [32] | |
| Silk | CaCl2/H2O/EtOH | ↑Humidity | Diameter decrease | The higher the humidity, the worse the fiber performance | Bo Kyung Park [33] |
4. Electrospinning of Polysaccharide
4.1. Starch
4.2. Chitosan/Chitin
4.3. Cellulose
4.4. Sodium Alginate
4.5. Pullulan
4.6. Dextran
4.7. Hyaluronic Acid
4.8. Cyclodextrin
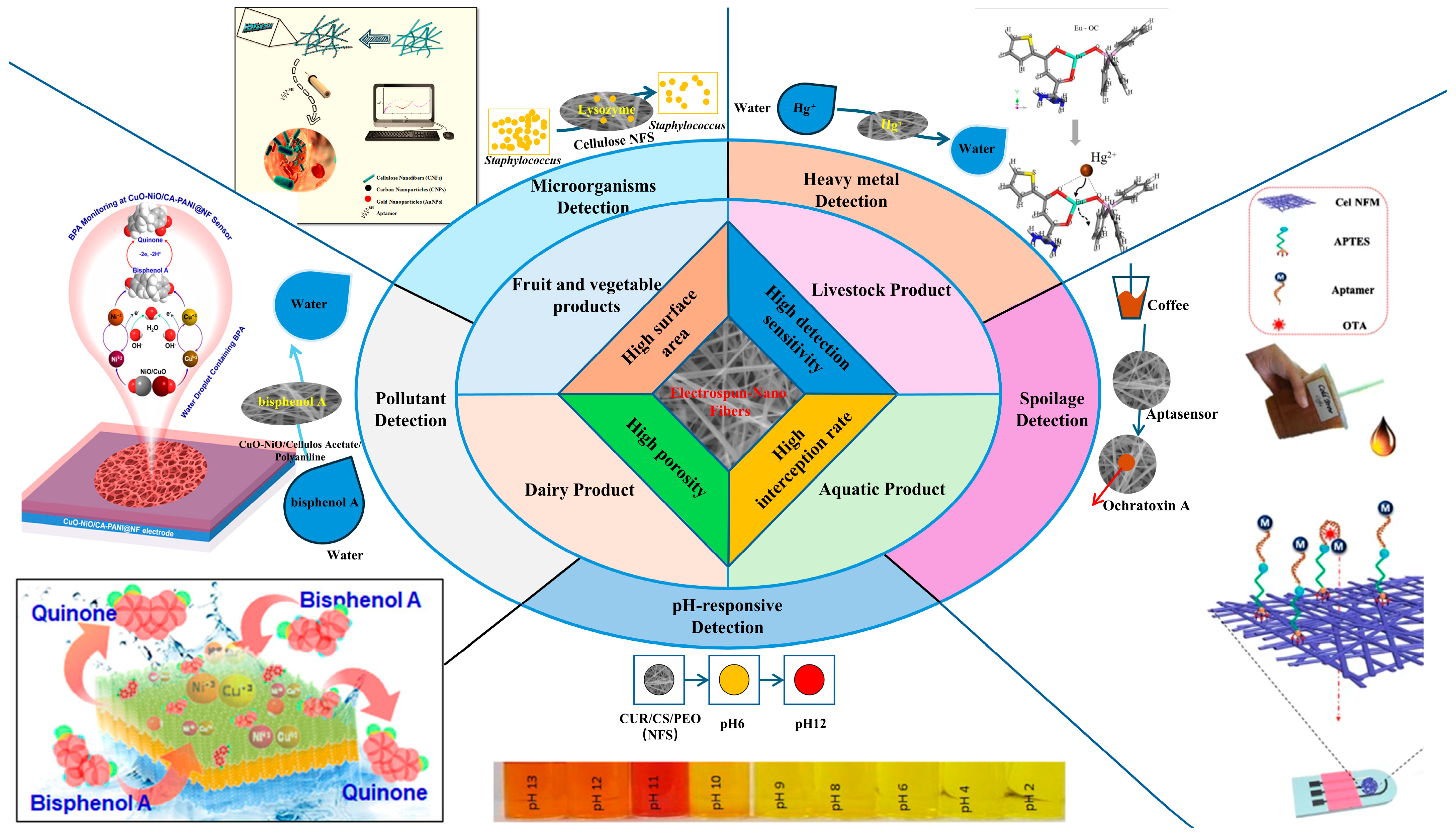
5. Application of Polysaccharide-Based Electrospun Nanofibers in Food Safety Detection
5.1. Livestock Products
5.2. Aquatic Products
5.3. Fruit and Vegetable Products
5.4. Dairy Product
6. Summary and Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nerín, C.; Aznar, M.; Carrizo, D. Food contamination during food process. Trends Food Sci. Technol. 2016, 48, 63–68. [Google Scholar] [CrossRef]
- Zhang, K.; Banerjee, K. A Review: Sample Preparation and Chromatographic Technologies for Detection of Aflatoxins in Foods. Toxins 2020, 12, 539. [Google Scholar] [CrossRef] [PubMed]
- Godebo, T.R.; Stoner, H.; Pechilis, M.; Taylor-Arnold, H.; Ashmead, J.; Claman, L.; Guest, L.; Consolati, W.; DiMatteo, O.; Johnson, M.; et al. Toxic metals and essential elements contents in commercially available fruit juices and other non-alcoholic beverages from the United States. J. Food Compos. Anal. 2023, 119, 105230. [Google Scholar] [CrossRef]
- Qian, J.; Huang, D.; Ni, D.; Zhao, J.; Shi, Z.; Fang, M.; Xu, Z. A portable CRISPR Cas12a based lateral flow platform for sensitive detection of Staphylococcus aureus with double insurance. Food Control 2022, 132, 108485. [Google Scholar] [CrossRef]
- Keirouz, A.; Wang, Z.; Reddy, V.S.; Nagy, Z.K.; Vass, P.; Buzgo, M.; Ramakrishna, S.; Radacsi, N. The History of Electrospinning: Past, Present, and Future Developments. Adv. Mater. Technol. 2023, 8, 2201723. [Google Scholar] [CrossRef]
- Wang, Y.; Khan, M.A.; Chen, K.; Zhang, L.; Chen, X. Electrospinning of Natural Biopolymers for Innovative Food Applications: A Review. Food Bioprocess Technol. 2023, 16, 704–725. [Google Scholar] [CrossRef]
- Dedhia, N.; Marathe, S.J.; Singhal, R.S. Food polysaccharides: A review on emerging microbial sources, bioactivities, nanoformulations and safety considerations. Carbohydr. Polym. 2022, 287, 119355. [Google Scholar] [CrossRef]
- Su, W.; Chang, Z.; E, Y.; Feng, Y.; Yao, X.; Wang, M.; Ju, Y.; Wang, K.; Jiang, J.; Li, P.; et al. Electrospinning and electrospun polysaccharide-based nanofiber membranes: A review. Int. J. Biol. Macromol. 2024, 263, 130335. [Google Scholar] [CrossRef]
- Kerwald, J.; de Moura Junior, C.F.; Freitas, E.D.; de Moraes Segundo, J.d.D.P.; Vieira, R.S.; Beppu, M.M. Cellulose-based electrospun nanofibers: A review. Cellulose 2022, 29, 25–54. [Google Scholar] [CrossRef]
- Amorim, L.F.A.; Mouro, C.; Gouveia, I.C. Electrospun fiber materials based on polysaccharides and natural colorants for food packaging applications. Cellulose 2024, 31, 6043–6069. [Google Scholar] [CrossRef]
- Ma, Y.; Morozova, S.M.; Kumacheva, E. From Nature-Sourced Polysaccharide Particles to Advanced Functional Materials. Adv. Mater. 2024, 36, 2312707. [Google Scholar] [CrossRef]
- Ding, B.; Wang, M.; Wang, X.; Yu, J.; Sun, G. Electrospun nanomaterials for ultrasensitive sensors. Mater. Today 2010, 13, 16–27. [Google Scholar] [CrossRef]
- Maruccio, C.; Quaranta, G.; Lorenzis, L.D.; Monti, G. Energy harvesting from electrospun piezoelectric nanofibers for structural health monitoring of a cable-stayed bridge. Smart Mater. Struct. 2016, 25, 085040. [Google Scholar] [CrossRef]
- Ghosh, S.K.; Adhikary, P.; Jana, S.; Biswas, A.; Sencadas, V.; Gupta, S.D.; Tudu, B.; Mandal, D. Electrospun gelatin nanofiber based self-powered bio-e-skin for health care monitoring. Nano Energy 2017, 36, 166–175. [Google Scholar] [CrossRef]
- Wang, Y.; Hao, J.; Huang, Z.; Zheng, G.; Dai, K.; Liu, C.; Shen, C. Flexible electrically resistive-type strain sensors based on reduced graphene oxide-decorated electrospun polymer fibrous mats for human motion monitoring. Carbon 2018, 126, 360–371. [Google Scholar] [CrossRef]
- Maftoonazad, N.; Ramaswamy, H. Design and testing of an electrospun nanofiber mat as a pH biosensor and monitor the pH associated quality in fresh date fruit (Rutab). Polym. Test. 2019, 75, 76–84. [Google Scholar] [CrossRef]
- Choi, J.; Chen, Y.; Abbel, R.; Visagie, I.; Parker, K. Flexible humidity sensors for wireless monitoring based on electrospun sulfonated polyether ether ketone (SPEEK) nanofibres. Sens. Actuators B Chem. 2020, 324, 128704. [Google Scholar] [CrossRef]
- Zhang, X.; Li, L.; Ouyang, J.; Zhang, L.; Xue, J.; Zhang, H.; Tao, W. Electroactive electrospun nanofibers for tissue engineering. Nano Today 2021, 39, 101196. [Google Scholar] [CrossRef]
- Graham, S.A.; Patnam, H.; Manchi, P.; Paranjape, M.V.; Kurakula, A.; Yu, J.S. Biocompatible electrospun fibers-based triboelectric nanogenerators for energy harvesting and healthcare monitoring. Nano Energy 2022, 100, 107455. [Google Scholar] [CrossRef]
- Liu, L.; Li, R.; Liu, F.; Huang, L.; Liu, W.; Wang, J.; Wu, Z.; Reddy, N.; Cui, W.; Jiang, Q. Highly Elastic and Strain Sensing Corn Protein Electrospun Fibers for Monitoring of Wound Healing. ACS Nano 2023, 17, 9600–9610. [Google Scholar] [CrossRef]
- Angel, N.; Guo, L.; Yan, F.; Wang, H.; Kong, L. Effect of processing parameters on the electrospinning of cellulose acetate studied by response surface methodology. J. Agric. Food Res. 2020, 2, 100015. [Google Scholar] [CrossRef]
- Matinfar, G.; Ye, H.; Bashiry, M.; Hashami, Z.; Yang, T. Electrospinning-based sensing technologies: Opportunities for food applications. Compr. Rev. Food Sci. Food Saf. 2024, 23, e13415. [Google Scholar] [CrossRef] [PubMed]
- Amiri, N.; Rozbeh, Z.; Afrough, T.; Sajadi Tabassi, S.A.; Moradi, A.; Movaffagh, J. Optimization of Chitosan-Gelatin Nanofibers Production: Investigating the Effect of Solution Properties and Working Parameters on Fibers Diameter. BioNanoScience 2018, 8, 778–789. [Google Scholar] [CrossRef]
- Refate, A.; Mohamed, Y.; Mohamed, M.; Sobhy, M.; Samhy, K.; Khaled, O.; Eidaroos, K.; Batikh, H.; El-Kashif, E.; El-Khatib, S.; et al. Influence of electrospinning parameters on biopolymers nanofibers, with emphasis on cellulose & chitosan. Heliyon 2023, 9, e17051. [Google Scholar] [CrossRef]
- Cao, P.; Wu, G.; Yao, Z.; Wang, Z.; Li, E.; Yu, S.; Liu, Q.; Gilbert, R.G.; Li, S. Effects of amylose and amylopectin molecular structures on starch electrospinning. Carbohydr. Polym. 2022, 296, 119959. [Google Scholar] [CrossRef]
- Lancuški, A.; Vasilyev, G.; Putaux, J.-L.; Zussman, E. Rheological Properties and Electrospinnability of High-Amylose Starch in Formic Acid. Biomacromolecules 2015, 16, 2529–2536. [Google Scholar] [CrossRef]
- Su, H.; Chen, X.; Mao, L.; Li, T. Enhancing Electrospinnability of Chitosan Membranes in Low-Humidity Environments by Sodium Chloride Addition. Mar. Drugs 2024, 22, 443. [Google Scholar] [CrossRef]
- Pan, W.; Wang, J.-P.; Sun, X.-B.; Wang, X.-X.; Jiang, J.-y.; Zhang, Z.-G.; Li, P.; Qu, C.-H.; Long, Y.-Z.; Yu, G.-F. Ultra uniform metal−organic framework-5 loading along electrospun chitosan/polyethylene oxide membrane fibers for efficient PM2.5 removal. J. Clean. Prod. 2021, 291, 125270. [Google Scholar] [CrossRef]
- Wang, Q.; Ju, J.; Tan, Y.; Hao, L.; Ma, Y.; Wu, Y.; Zhang, H.; Xia, Y.; Sui, K. Controlled synthesis of sodium alginate electrospun nanofiber membranes for multi-occasion adsorption and separation of methylene blue. Carbohydr. Polym. 2019, 205, 125–134. [Google Scholar] [CrossRef]
- Van-Pham, D.-T.; Thi Bich Quyen, T.; Van Toan, P.; Nguyen, C.-N.; Ho, M.H.; Van Hong Thien, D. Temperature effects on electrospun chitosan nanofibers. Green Process. Synth. 2020, 9, 488–495. [Google Scholar] [CrossRef]
- Khatri, A.; Ali, S.; Javeed, A. Indigo dyeing of cellulose electrospun nanofibrous mats. Cellulose 2023, 30, 7955–7963. [Google Scholar] [CrossRef]
- Yoon, J.; Lee, J.; Oh, S.-G. Preparation of electrospun cellulose acetate/polyethylene imine bicomponent nanofibers for CO2 capture. Polym. Bull. 2024, 81, 1389–1401. [Google Scholar] [CrossRef]
- Park, B.K.; Um, I.C. Effect of Relative Humidity on the Electrospinning Performance of Regenerated Silk Solution. Polymers 2021, 13, 2479. [Google Scholar] [CrossRef]
- Sethunga, M.; Gunathilake, K.D.P.P.; Ranaweera, K.K.D.S.; Munaweera, I. Antimicrobial and antioxidative electrospun cellulose acetate-essential oils nanofibrous membranes for active food packaging to extend the shelf life of perishable fruits. Innov. Food Sci. Emerg. Technol. 2024, 97, 103802. [Google Scholar] [CrossRef]
- Varnaitė-Žuravliova, S.; Savest, N.; Baltušnikaitė-Guzaitienė, J.; Abraitienė, A.; Krumme, A. The Investigation of the Production of Salt-Added Polyethylene Oxide/Chitosan Nanofibers. Materials 2024, 17, 132. [Google Scholar] [CrossRef]
- Fonseca, L.M.; Cruz, E.P.d.; Crizel, R.L.; Jansen-Alves, C.; Dias, A.R.G.; Zavareze, E.d.R. New advances of electrospun starch fibers, encapsulation, and food applications: A review. Trends Food Sci. Technol. 2024, 147, 104467. [Google Scholar] [CrossRef]
- Huang, X.; Teng, Z.; Xie, F.; Wang, G.; Li, Y.; Liu, X.; Li, S. Loading of cinnamon essential oil into electrospun octenylsuccinylated starch-pullulan nanofiber mats: Electrospinnability evaluation, structural characterization, and antibacterial potential. Food Hydrocoll. 2024, 148, 109426. [Google Scholar] [CrossRef]
- Hejabri Kandeh, S.; Amini, S.; Ebrahimzadeh, H. Development of poly(vinyl alcohol)/chitosan/aloe vera gel electrospun composite nanofibers as a novel sorbent for thin-film micro-extraction of pesticides in water and food samples followed by HPLC-UV analysis. New J. Chem. 2022, 46, 2431–2440. [Google Scholar] [CrossRef]
- Mu, R.; Hong, X.; Ni, Y.; Li, Y.; Pang, J.; Wang, Q.; Xiao, J.; Zheng, Y. Recent trends and applications of cellulose nanocrystals in food industry. Trends Food Sci. Technol. 2019, 93, 136–144. [Google Scholar] [CrossRef]
- Martín-Alfonso, M.A.; Rubio-Valle, J.F.; Hinestroza, J.P.; Martín-Alfonso, J.E.; Franco, J.M. Environmentally friendly tailor-made oleo-dispersions of electrospun cellulose acetate propionate nanostructures in castor oil for lubricant applications. Nano Mater. Sci. 2024. [Google Scholar] [CrossRef]
- Zhou, G.; Zhang, Z.; Meng, Z.; Qian, C.; Li, M.; Wang, Z.; Yang, Y. A highly specific chalcone derivative grafted ethylcellulose fluorescent probe for rapid and sensitive detection of Al3+ in actual environmental and food samples. Int. J. Biol. Macromol. 2023, 252, 126475. [Google Scholar] [CrossRef]
- Zhao, X.; Wang, X.; Lou, T. Simultaneous adsorption for cationic and anionic dyes using chitosan/electrospun sodium alginate nanofiber composite sponges. Carbohydr. Polym. 2022, 276, 118728. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhang, P.; Wang, Q.; Yu, N.; Zhang, X.; Su, S. Electrospinning Novel Sodium Alginate/MXene Nanofiber Membranes for Effective Adsorption of Methylene Blue. Polymers 2023, 15, 2110. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Lan, T.; Wang, L.; Wang, X.; Xu, Z.; Jiang, L.; Zhang, Y.; Sui, X. Development of composite electrospun films utilizing soy protein amyloid fibrils and pullulan for food packaging applications. Food Chem. X 2023, 20, 100995. [Google Scholar] [CrossRef] [PubMed]
- Amjadi, S.; Gholizadeh, S.; Ebrahimi, A.; Almasi, H.; Hamishehkar, H.; Taheri, R.A. Development and characterization of the carvone-loaded zein/pullulan hybrid electrospun nanofibers for food and medical applications. Ind. Crops Prod. 2022, 183, 114964. [Google Scholar] [CrossRef]
- Jiang, L.; Wang, C.; Zhao, F.; Li, S.; Sun, D.; Ma, Q.; Yu, Z.; Zhang, B.; Liu, Y.; Jiang, W. Development of electrospun nanofiber films based on pullulan/polyvinyl alcohol incorporating bayberry pomace anthocyanin extract for aquatic products freshness monitoring. Food Biosci. 2024, 58, 103717. [Google Scholar] [CrossRef]
- Phulmogare, G.; Rani, S.; Lodhi, S.; Patil, U.K.; Sinha, S.; Ajazuddin; Gupta, U. Fucoidan loaded PVA/Dextran blend electrospun nanofibers for the effective wound healing. Int. J. Pharm. 2024, 650, 123722. [Google Scholar] [CrossRef]
- Kenawy, E.-R.S.; Kamoun, E.A.; Eldin, M.S.; Soliman, H.M.A.; El-Moslamy, S.H.; El-Fakharany, E.M.; Shokr, A.-b.M. Electrospun PVA–Dextran Nanofibrous Scaffolds for Acceleration of Topical Wound Healing: Nanofiber Optimization, Characterization and In Vitro Assessment. Arab. J. Sci. Eng. 2023, 48, 205–222. [Google Scholar] [CrossRef]
- Gruppuso, M.; Iorio, F.; Turco, G.; Marsich, E.; Porrelli, D. Hyaluronic acid/lactose-modified chitosan electrospun wound dressings—Crosslinking and stability criticalities. Carbohydr. Polym. 2022, 288, 119375. [Google Scholar] [CrossRef]
- Yang, X.; Gu, P.; Jiang, Q.; Cheng, X.; Fan, J.; Bai, Y. Instant and Multifunctional Nanofibers Loaded with Proanthocyanidins and Hyaluronic Acid for Skincare Applications. Biomedicines 2024, 12, 1584. [Google Scholar] [CrossRef]
- Dodero, A.; Schlatter, G.; Hébraud, A.; Vicini, S.; Castellano, M. Polymer-free cyclodextrin and natural polymer-cyclodextrin electrospun nanofibers: A comprehensive review on current applications and future perspectives. Carbohydr. Polym. 2021, 264, 118042. [Google Scholar] [CrossRef] [PubMed]
- Yildiz, E.; Sumnu, G.; Kahyaoglu, L.N. Monitoring freshness of chicken breast by using natural halochromic curcumin loaded chitosan/PEO nanofibers as an intelligent package. Int. J. Biol. Macromol. 2021, 170, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Cai, Q.; Liu, K.; Yan, G.; Chen, J.; Wu, Y.; Li, Z.; Wang, L.; Li, H. Flexible and robust Eu/cellulose acetate nanofibrous membrane for the rapid detection of Hg2+ with ultra-stable recycle capability. Measurement 2023, 222, 113660. [Google Scholar] [CrossRef]
- Shoukat, N.; Anzar, S.; Asad, M.; Al-Sulami, A.I.; Khalid, H.; Choudhary, A.A.; Sherin, L.; Akhtar, N. Fabrication of CuO–NiO Wrapped Cellulose Acetate/Polyaniline Electrospun Nanofibers for Sensitive Monitoring of Bisphenol-A. ACS Sustain. Chem. Eng. 2023, 11, 4299–4307. [Google Scholar] [CrossRef]
- El-Moghazy, A.Y.; Amaly, N.; Istamboulie, G.; Nitin, N.; Sun, G. A signal-on electrochemical aptasensor based on silanized cellulose nanofibers for rapid point-of-use detection of ochratoxin A. Microchim. Acta 2020, 187, 535. [Google Scholar] [CrossRef]
- Ranjbar, S.; Shahrokhian, S. Design and fabrication of an electrochemical aptasensor using Au nanoparticles/carbon nanoparticles/cellulose nanofibers nanocomposite for rapid and sensitive detection of Staphylococcus aureus. Bioelectrochemistry 2018, 123, 70–76. [Google Scholar] [CrossRef]
- Bekhit, A.E.-D.A.; Holman, B.W.B.; Giteru, S.G.; Hopkins, D.L. Total volatile basic nitrogen (TVB-N) and its role in meat spoilage: A review. Trends Food Sci. Technol. 2021, 109, 280–302. [Google Scholar] [CrossRef]
- Li, B.; Chen, H.; Ma, Q.; Tang, T.; Bai, Y. A Novel Polyvinyl Alcohol/Chitosan-Based Anthocyanin Electrospun Colorimetric Film for Monitoring Chicken Breast Freshness. Food Anal. Methods 2025. [Google Scholar] [CrossRef]
- Guo, M.; Wang, H.; Wang, Q.; Chen, M.; Li, L.; Li, X.; Jiang, S. Intelligent double-layer fiber mats with high colorimetric response sensitivity for food freshness monitoring and preservation. Food Hydrocoll. 2020, 101, 105468. [Google Scholar] [CrossRef]
- Ur Rehman, M.U.; Alshammari, A.S.; Zulfiqar, A.; Zafar, F.; Khan, M.A.; Majeed, S.; Akhtar, N.; Sajjad, W.; Hanif, S.; Irfan, M.; et al. Machine learning powered CN-coordinated cobalt nanoparticles embedded cellulosic nanofibers to assess meat quality via clenbuterol monitoring. Biosens. Bioelectron. 2024, 261, 116498. [Google Scholar] [CrossRef]
- Wang, C.; Lu, Y.; An, X.; Wang, Y.; Wang, N.; Song, Y.; Hu, N.; Ren, M. Preparation, characterization, and application of pH-responsive biodegradable intelligent indicator film based on rose anthocyanins. LWT—Food. Sci. Technol. 2024, 200, 116156. [Google Scholar] [CrossRef]
- Duan, M.; Yu, S.; Sun, J.; Jiang, H.; Zhao, J.; Tong, C.; Hu, Y.; Pang, J.; Wu, C. Development and characterization of electrospun nanofibers based on pullulan/chitin nanofibers containing curcumin and anthocyanins for active-intelligent food packaging. Int. J. Biol. Macromol. 2021, 187, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Yurova, N.S.; Danchuk, A.; Mobarez, S.N.; Wongkaew, N.; Rusanova, T.; Baeumner, A.J.; Duerkop, A. Functional electrospun nanofibers for multimodal sensitive detection of biogenic amines in food via a simple dipstick assay. Anal. Bioanal. Chem. 2018, 410, 1111–1121. [Google Scholar] [CrossRef] [PubMed]
- Teodoro, K.B.R.; Migliorini, F.L.; Facure, M.H.M.; Correa, D.S. Conductive electrospun nanofibers containing cellulose nanowhiskers and reduced graphene oxide for the electrochemical detection of mercury(II). Carbohydr. Polym. 2019, 207, 747–754. [Google Scholar] [CrossRef]
- Wu, H.; Xie, R.; Hao, Y.; Pang, J.; Gao, H.; Qu, F.; Tian, M.; Guo, C.; Mao, B.; Chai, F. Portable smartphone-integrated AuAg nanoclusters electrospun membranes for multivariate fluorescent sensing of Hg2+, Cu2+ and l-histidine in water and food samples. Food Chem. 2023, 418, 135961. [Google Scholar] [CrossRef]
- Topsoy, O.K.; Muhammad, F.; Kolak, S.; Ulu, A.; Güngör, Ö.; Şimşek, M.; Köytepe, S.; Ateş, B. Fabrication of electrospun polycaprolactone/chitosan nanofiber-modified screen-printed electrode for highly sensitive detection of diazinon in food analysis. Measurement 2022, 187, 110250. [Google Scholar] [CrossRef]
- El-Moghazy, A.Y.; Soliman, E.A.; Ibrahim, H.Z.; Marty, J.L.; Istamboulie, G.; Noguer, T. Biosensor based on electrospun blended chitosan-poly (vinyl alcohol) nanofibrous enzymatically sensitized membranes for pirimiphos-methyl detection in olive oil. Talanta 2016, 155, 258–264. [Google Scholar] [CrossRef]
- Tsai, T.-Y.; Chen, S.-H.; Chen, L.-C.; Lin, S.-B.; Lou, S.-N.; Chen, Y.-H.; Chen, H.-H. Enzymatic Time-Temperature Indicator Prototype Developed by Immobilizing Laccase on Electrospun Fibers to Predict Lactic Acid Bacterial Growth in Milk during Storage. Nanomaterials 2021, 11, 1160. [Google Scholar] [CrossRef]
- Wang, P.; Zhang, C.; Zou, Y.; Li, Y.; Zhang, H. Immobilization of lysozyme on layer-by-layer self-assembled electrospun films: Characterization and antibacterial activity in milk. Food Hydrocoll. 2021, 113, 106468. [Google Scholar] [CrossRef]
- Beiranvand, R.; Sarlak, N. Electrospun nanofiber mat of graphene/mesoporous silica composite for wastewater treatment. Mater. Chem. Phys. 2023, 309, 128311. [Google Scholar] [CrossRef]
- Yalcinkaya, B.; Strejc, M.; Yalcinkaya, F.; Spirek, T.; Louda, P.; Buczkowska, K.E.; Bousa, M. An Innovative Approach for Elemental Mercury Adsorption Using X-ray Irradiation and Electrospun Nylon/Chitosan Nanofibers. Polymers 2024, 16, 1721. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.; Yoon, T.; Park, W.; You, J.; Na, S. Unraveling the Mechanical Property Decrease of Electrospun Spider Silk: A Molecular Dynamics Simulation Study. ACS Appl. Bio Mater. 2024, 7, 1968–1975. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Dong, Z.; Huan, K.; He, Z.; Zhang, Q.; Deng, D.; Luo, L. Application of the Electrospinning Technique in Electrochemical Biosensors: An Overview. Molecules 2024, 29, 2769. [Google Scholar] [CrossRef] [PubMed]
- Lu, G.; Tian, T.; Wang, Y. Advanced Electrospinning Technology Applied to Polymer-Based Sensors in Energy and Environmental Applications. Polymers 2024, 16, 839. [Google Scholar] [CrossRef]
- Zu, Y.; Duan, Z.; Yuan, Z.; Jiang, Y.; Tai, H. Electrospun nanofiber-based humidity sensors: Materials, devices, and emerging applications. J. Mater. Chem. A 2024, 12, 27157–27179. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, J.; Tang, J.; Zhang, M.; Zou, Y.; Pang, J.; Wu, C. Recent Advances in Polysaccharide-Based Electrospun Nanofibers for Food Safety Detection. Sensors 2025, 25, 2220. https://doi.org/10.3390/s25072220
Shi J, Tang J, Zhang M, Zou Y, Pang J, Wu C. Recent Advances in Polysaccharide-Based Electrospun Nanofibers for Food Safety Detection. Sensors. 2025; 25(7):2220. https://doi.org/10.3390/s25072220
Chicago/Turabian StyleShi, Jie, Junjie Tang, Mengfei Zhang, Yingqi Zou, Jie Pang, and Chunhua Wu. 2025. "Recent Advances in Polysaccharide-Based Electrospun Nanofibers for Food Safety Detection" Sensors 25, no. 7: 2220. https://doi.org/10.3390/s25072220
APA StyleShi, J., Tang, J., Zhang, M., Zou, Y., Pang, J., & Wu, C. (2025). Recent Advances in Polysaccharide-Based Electrospun Nanofibers for Food Safety Detection. Sensors, 25(7), 2220. https://doi.org/10.3390/s25072220







