Abstract
The thermal stability of aqueous polyurethanes has been measured applying the thermogravimetric analysis. The aqueous polyurethanes (aqPUR) with catalysts of different selectivity have been studied by use of the dynamic method. To obtain degradations of 0.025, 0.05, and 0.10, employing the dynamic method, the heating rates of 0.5, 1, 2, 5, and 10 °C min-1 have been used in the range of 30-500 °C. Using the more selective catalysts in the aqueous polyurethanes, the total resulting time of the decompositon has been on the increase at all degrees of the degradation and at the particular starting temperature. This paper shows that the dynamic method based on the thermogravimetric analysis can be used to assess the thermal stability of the aqueous polyurethanes using the catalysts of different selectivity.
Introduction
Two-component paint systems are used in various segments of the industrial coatings market. The performances of these systems are, in general, surprisingly good. The gloss, looks, chemical resistance and mechanical properties have the standard of solvent based products. However, the application of these aqueous two-component polyurethanes is rather less intense and it prevents the breakthrough of this technology into the market of the industrial coatings. It is, for instance, difficult to obtain greater layer thickness without surface defects using these two-component systems [1]. Likewise, the procedure itself of obtaining the polyols is rather essential and is reflected on characteristics of the obtained film [2,3].
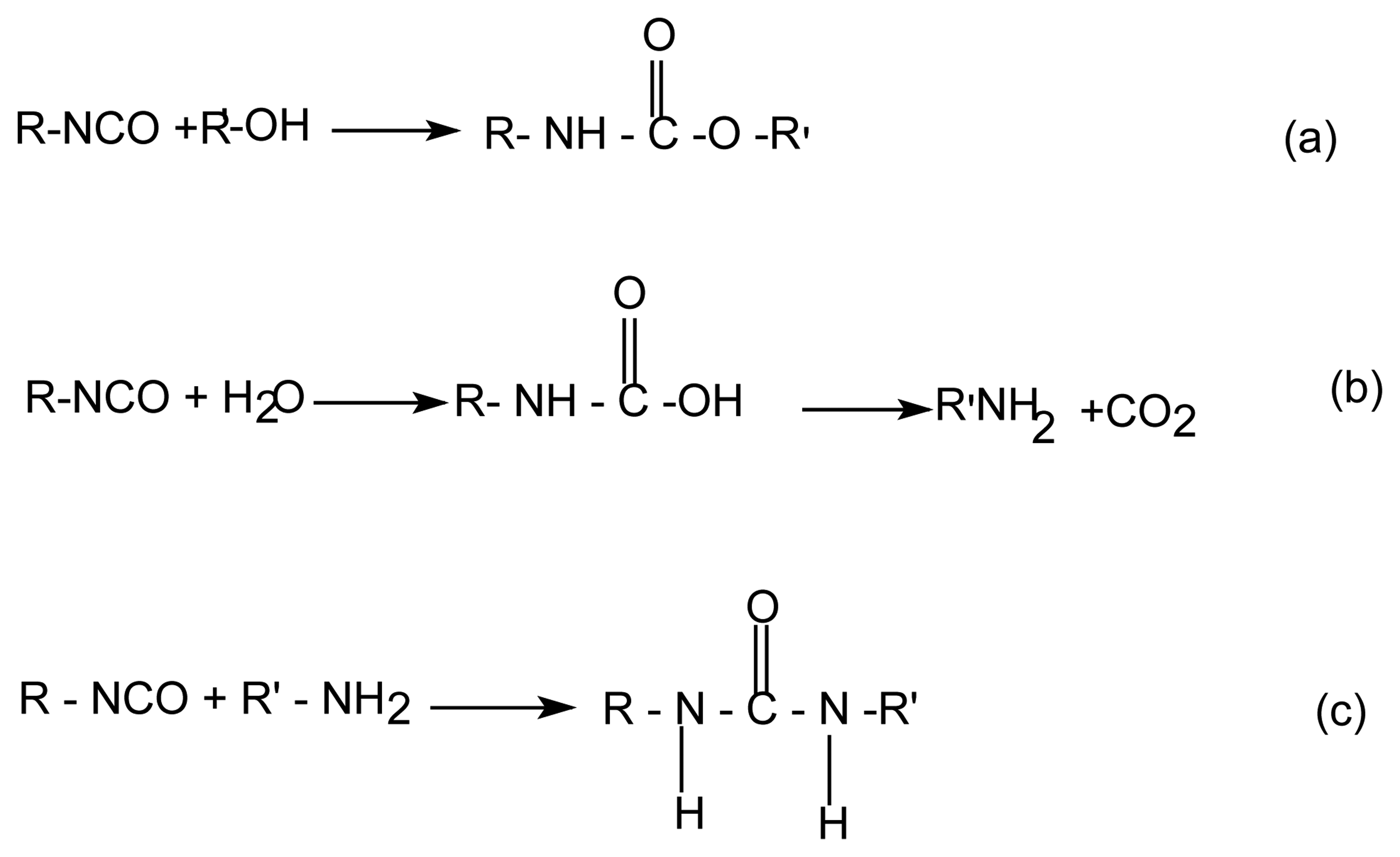
The main objective in the development of the aqueous polyurethanes is, in the first place, to find methods for preventing the undesired secondary reactions with water and achieving the best crosslinking. This reaction is reduced to a minimum by use of the non-tin catalysts [4]. One novel approach to control the water side reaction is the use of catalysts that selectively catalyze the isocyanate-polyol reaction but not the isocyanate-water reaction as shown in the following reactions (Sheme 1).
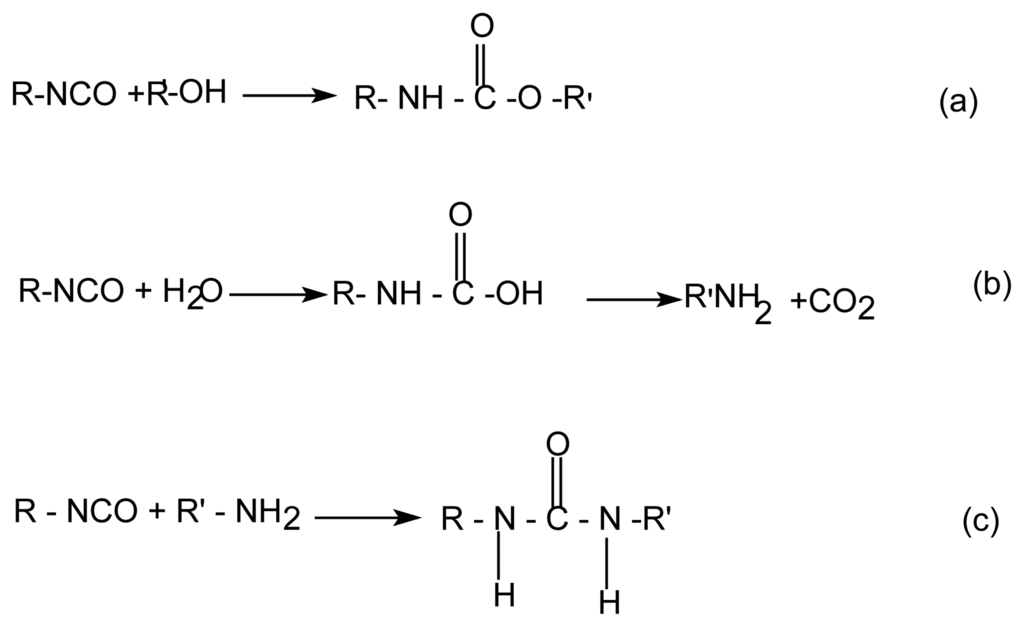
Sheme 1.
Isocyanate reactions with alcohol (a) and with water (b,c).
The reactions of isocyanates, especially the aliphatic isocyanates, with the hydroxyl group or water are relatively slow at the ambient temperature in the absence of a catalyst. Organo metallic compounds or tertiary amines are normally needed to catalyze the crosslinking of the isocyanates with the hydroxyl groups in the ambient conditions [4,5].
In the researches of Werner Blank [4,5], the selectivity of various compounds compared to model compounds has been examined. Butyl isocyanate and 2-ethyl-1-hexanol have been selected as model compounds due to their similarity to the aliphatic polyisocyanates and the hydroxyl resins as well as because of the simplicity of their FTIR spectra.
The relative selectivity (S), can be obtained by the method of Werner Blank [5], from the Equation 1, by measuring the ratio of the urethane IR peak area (Purethan) and the urea IR peak area (Purea):
Upon the integration of the characteristic absorption maximums of the urethane and the urea, the relative selectivity has been calculated.
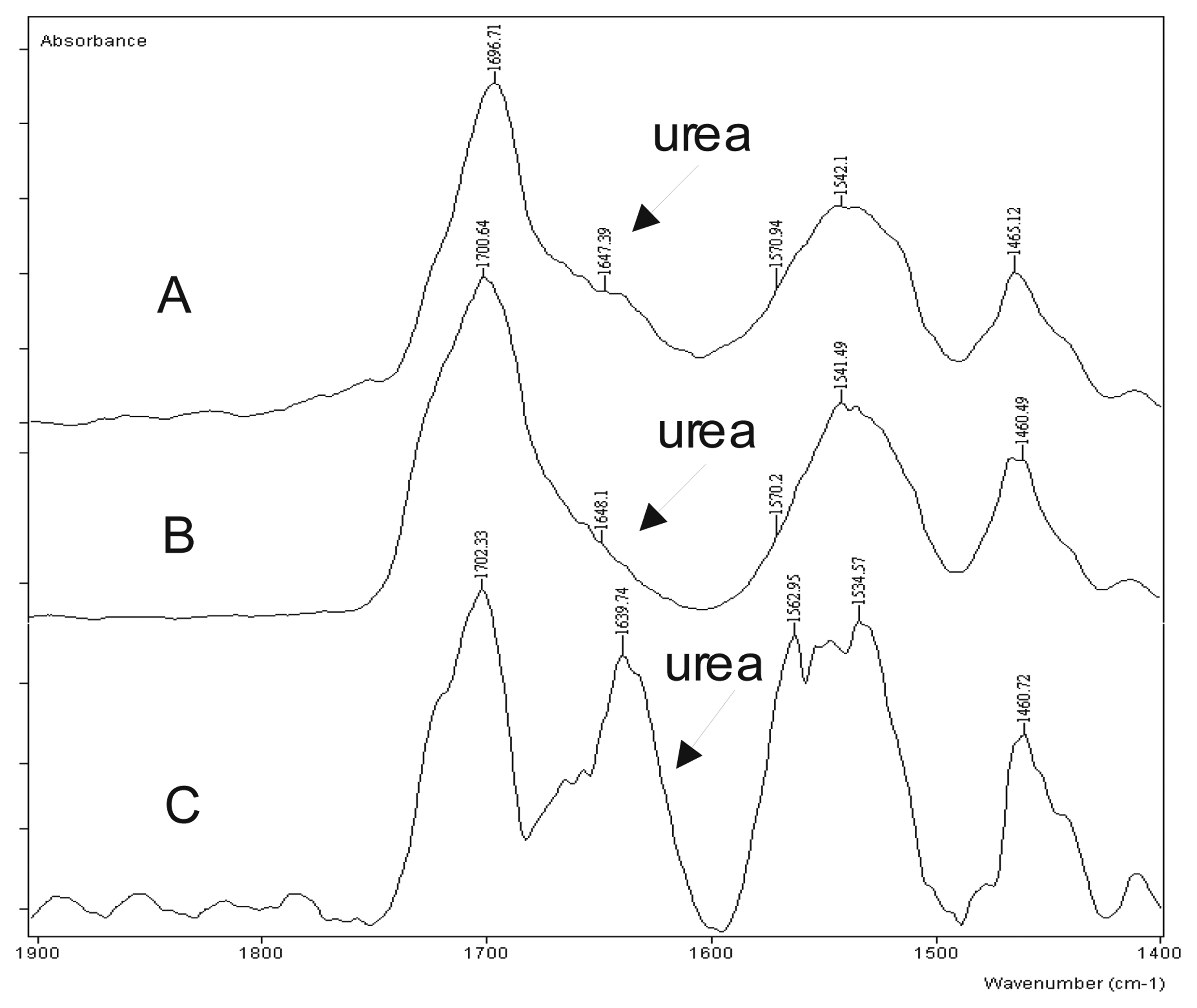
The complex of Mn(III)-diacetylacetonatomaleate with various ligands based on the acetylacetonate and the maleic acid, used in some of the experiments [6,7], has showed a high selectivity for the isocyanate-hydroxyl reaction (Figure 1).
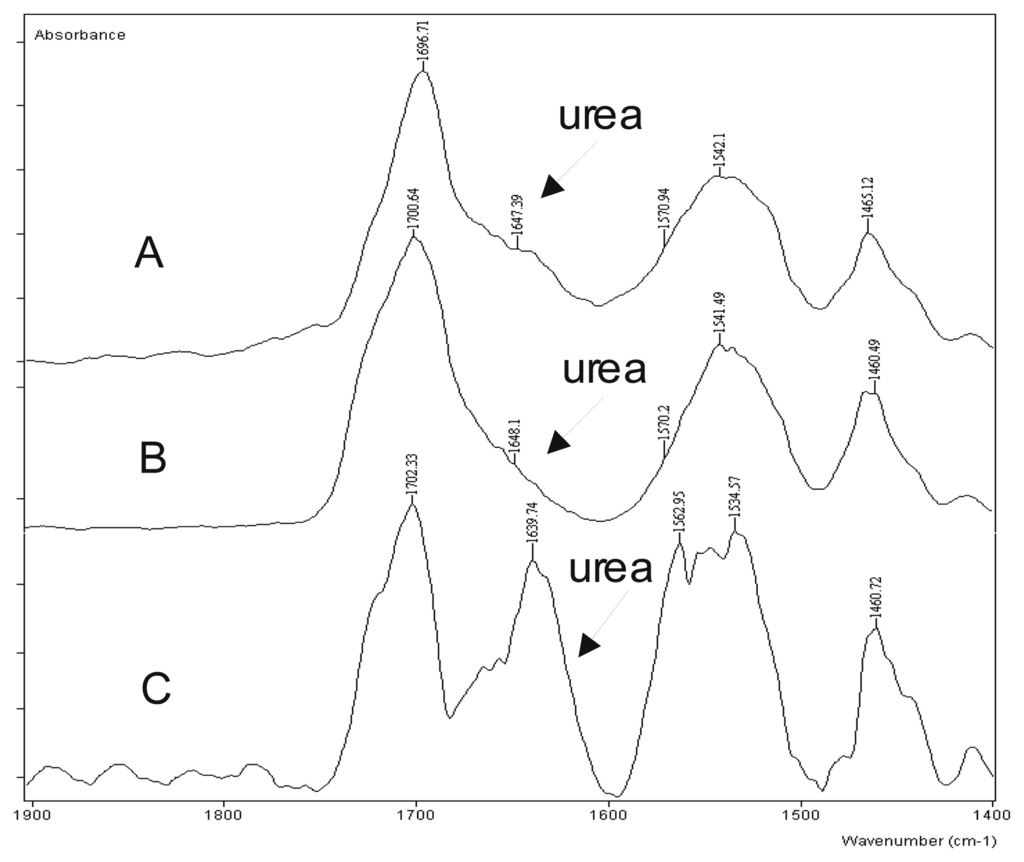
Figure 1.
The comparative FTIR spectrum of the catalyzed model system NCO/OH/H2O =1.0/1.0/2.0 with: A)- the catalyst of zirconium K-KAT®XC-6212 (King industries, Norwalk-USA); B)- the complex of Mn(III)-diacetylacetonatomaleate; C)- Co-octoat.
Different thermogravimetric methods can be used to study the thermal stability of polymeric materials both in oxidative and nonoxidative conditions. The evaluation of the kinetic parameters can be derived from the isothermal and dynamic data, as reported by various researchers [8-18].
The thermal stability of the aqueous polyurethanes measured by using the dynamic method of the thermogravimetric analysis has been studied by Fambi and Pegoretti [19]. They have studied the thermal stability of polyurethanes based on a solvent and on an aqueous base in the process of which the polyester and the polyether components without the catalysts have been used as polyol components.
The objective of the this paper is to evaluate the thermal stability of the aqueous polyurethanes (aqPUR) by means of the dynamic method based on the TGA, applying employing the polyol components based on different acrylic copolymers and the catalysts of different selectivity.
The thermoxidation experiments are generally determined by the percentage of weight loss, W%, and the degree of degradation, α, defined as:
where wo and w are the initial and the actual weight of the sample, respectively.
Several heating rates, β, have been used in the dynamic method [20,21] to obtain the temperature at which the defined percentage of weight loss occurs, according to the following equation:
where ADYN is the pre-exponential factor, Eact-DYN is the activation energy and Tα is the absolute temperature at which the degree of degradation, α, is reached.
Experimental
The polyols and polyisocyanates, being the commercial materials, have been used during the study without further purification. The two polyol components, with the hydroxyl number above 130, have been employed in this experiment and they are: the acrylic dispersion in the mixture water/butyl alcohol (Macrynal VSM 2521 w/42 WAB, Solutia Austria GmbH) and also the acrylic resin as the water dispersion (Macrynal VSM 6299 w/42 WA, Solutia Austria GmbH). Macrynal VSM 6299 is a pure acrylic copolymer and Macrynal VSM 2521 is the acrylic copolymer modified by fatty acids.The content of dry matter has been 42% in these dispersions.
Being suitable hardeners for these dispersions, the aliphatic polyfunctional isocyanates based on hexamethylen diisocyanates have been employed and they are as follows: Bayhydur VP LS 2319 (18.2% NCO), Bayhydur VP LS 2336 (16.2% NCO), Bayhydur VP LS 2150 (13.4% NCO) as well as Desmodur 3600 (23% NCO), purchased from Bayer AG Germany. All the applied hardeners have been without the organic solvents (100%), except for Bayhydur VP LS 2150 (70%).
The zirconium catalyst (ZrCAT) is a proprietary zirconium tetra-dionato complex [22] in the reactive solvent with a metal content of 0.4%.
The manganese catalyst, the complex of Mn(III)-diacetylacetonatomaleate, has shown the unusually high selectivity for the isocyanate-polyol reaction in comparison with the commercially available zirconium catalyst [8,9]. The manganese catalyst (MnCAT) has been used in the reactive diluent with the metal content of 0.4%.
The catalyst, concentrated 2% and 4% relating to the coating hardness, has been added to the B component. The Table 1 and Table 2 show the composition of the employed components.

Table 1.
The composition of the two-component aqueous polyurethane sample (aqPUR1) based on the polyol component Macrynal VSM 6299 w/42 WA (coating hardness 32.5 %).

Table 2.
The composition of the two-component aqueous polyurethane (aqPUR2) sample based on the polyol component Macrynal VSM 2521 w/42 WAB (coating hardness 40.2 %).
The thermograms have been recorded on the instrument DSC 4 from Perkin-Elmer. The samples have been prepared with the organic solvents and water, and therefore the laced up aluminium ampoules with a lid gap for gases release have been used, the sample weight being 360 mg.
In order to examine the the degradations of 0.025, 0.05 and 0.10, the heating rates of 0.5, 1, 2, 5 and 10 °C min-1, ranged 30-500 °C, have been employed during the dynamic method.
The instrument has the autocalibration and the linear regulation of a settled temperature gradient.
Results and Discussion
Table 3 and Table 4 sum up the experimental data of the thermal analysis of the different aqueous polyurethanes without the catalysts and with the catalysts of different selectivity measured by the dynamic method.

Table 3.
The obtained temperatures of the sample aqPUR1, with the different concentrations of the catalysts, at the degradations of 0.025, 0.05 and 0.10 in the function of the different heating rates.

Table 4.
The obtained temperatures of the sample aqPUR2, with the different concentrations of the catalysts, at the degradations of 0.025; 0.05 and 0.10 in the function of the different heating rates.
The lower thermal stability in aqueous polyurethanes without the catalyst is immediately obvious from the dynamic degradation, requiring a lower temperature to reach the determined degree of degradation.
During the dynamic thermooxidation, it is also possible to detect the presence of different degradation stages as shown in the case of aqPUR1 and aqPUR2 in Figure 2 and Figure 3, respectively. The change of the slope in the interval 100-300 °C is due to a change in the mechanism and contributes to the different activation energy measured at the degradations of 0.025, 0.05 and 0.10. The weight loss of about 1%, observed below the temperature of 100 °C, is the result of the evaporation of the equilibrium moisture and/or other volatile products [23].
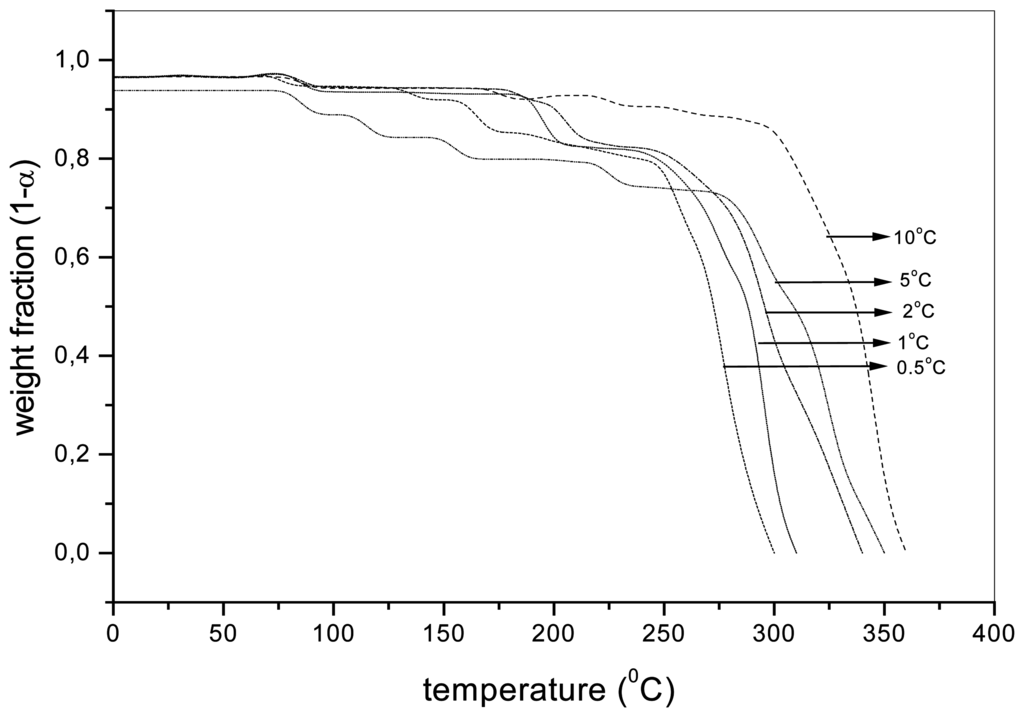
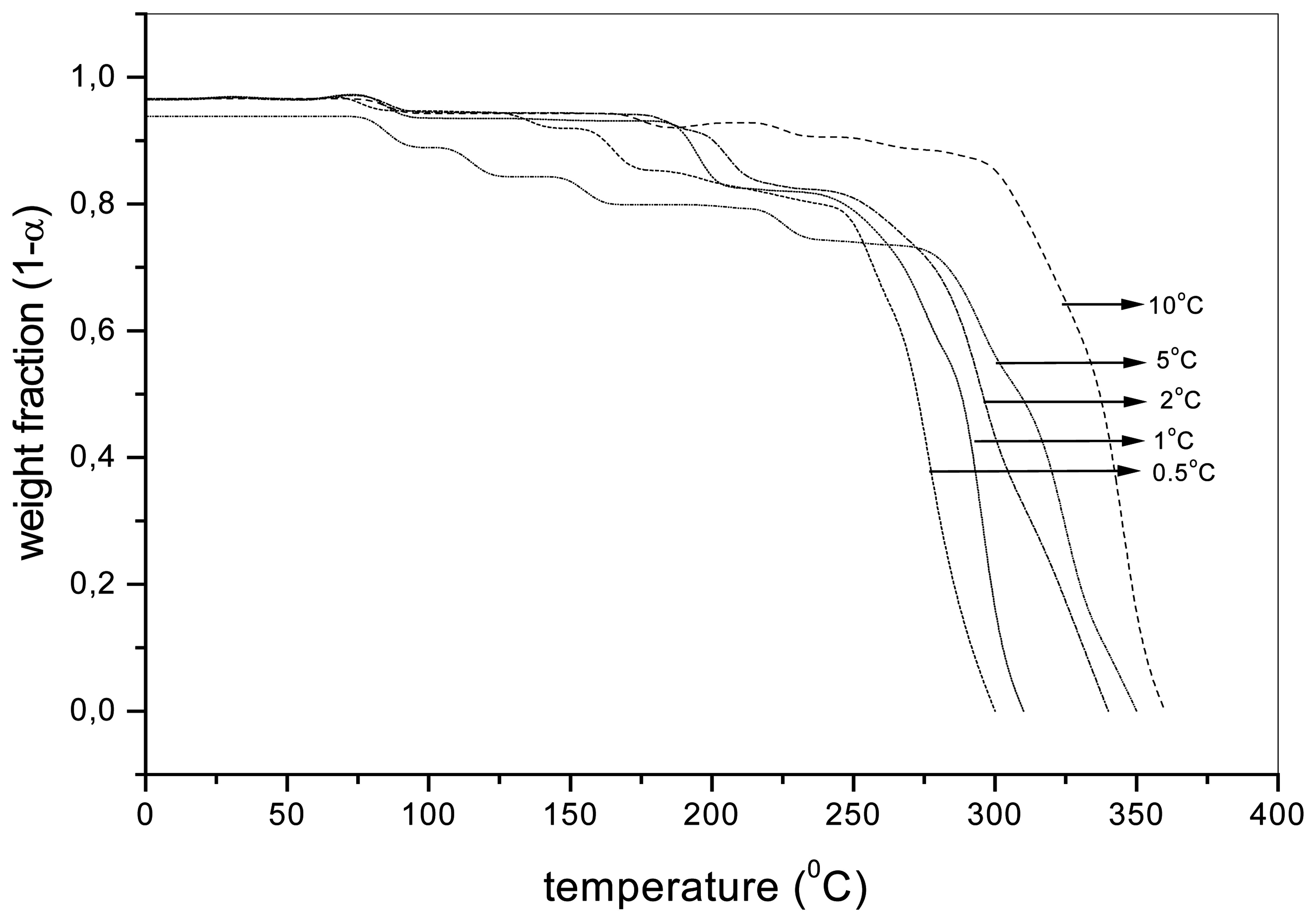
Figure 2.
The weight fraction (1-α) of the sample aqPUR1 with the MnCAT (2%) depending on the temperature during the dynamic thermooxidation at different heating rates.
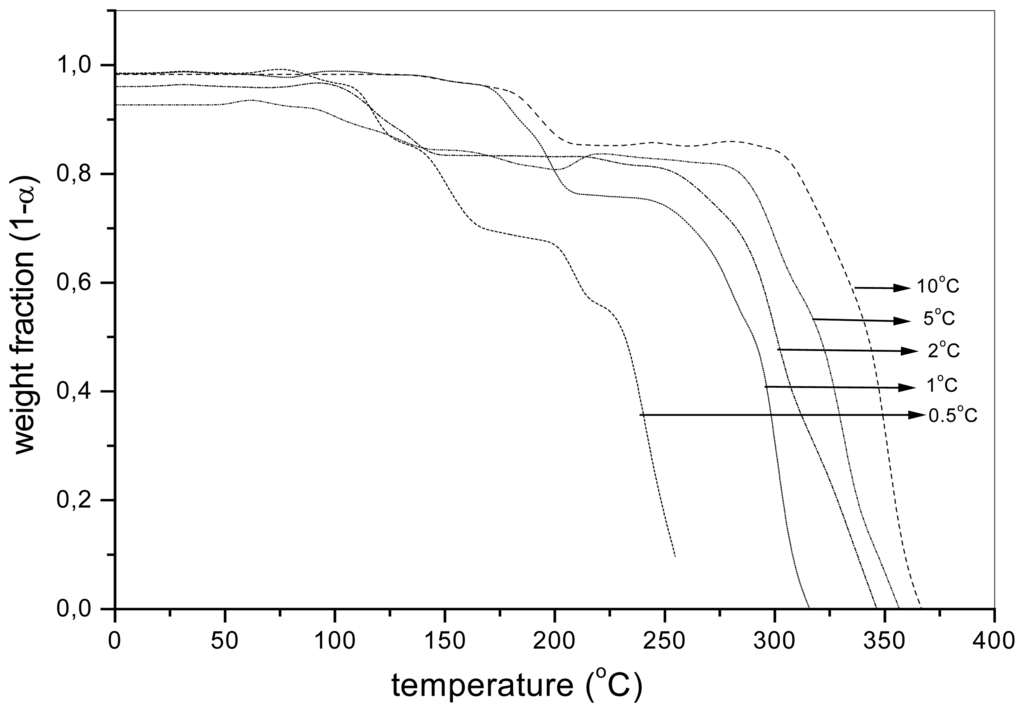
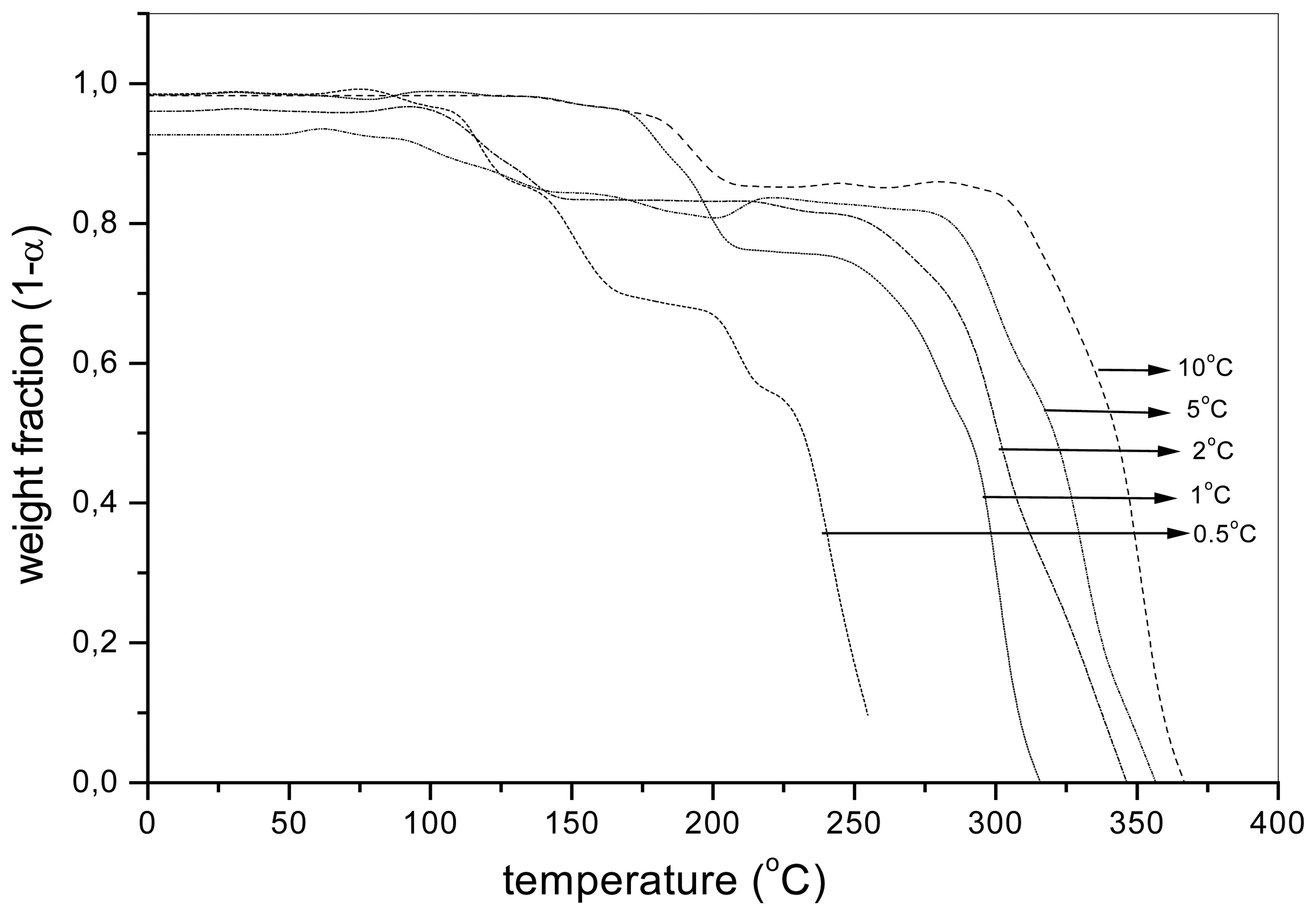
Figure 3.
The weight fraction (1-α) of the sample aqPUR2 with the MnCAT (2%) depending on the temperature during the dynamic thermooxidation, at different heating rates.
The total experimental time periods required to collect the TGA data during the kinetic analyses of the aqueous polyurethanes without the catalysts and with the catalysts of different selectivity have been compared by means of the dynamic method [19]. Table 5 and Table 6 show the decompositon time of the dynamic experiments at which the degradations of 0.025, 0.05 and 0.10 have occurred.

Table 5.
The total time of the decomposition required to calculate the thermal stability of the sample aqPUR1 at various degrees of the degradation.

Table 6.
The total time of the decomposition required to calculate the thermal stability of the sample aqPUR2 at various degrees of the degradation.
The overall time of the decompositon for the dynamic experiments, tdyn, for each degradation degree can be evaluated from the data presented in Table 3 and Table 4, taking into consideration the temperature at which a certain degradation, Tr, occurs, the heating rate, r (°C min-1) and the starting temperature, Ts (30 or 100 °C), according to the following equation:
For instance, in the case of the sample aqPUR1, the control test (no catalyst) of the degradation of 0.025 at the heating rate of 0.5 °C min-1 has been obtained at the temperature of 127 °C, taking into account the various heating rates, the total resulting time being 395.4 min.
By using the catalyst MnCAT (2% and 4%), which is more selective than the ZrCAT, the total resulting time of the decompositon has increased at all degradation degrees as shown in Table 5 and Table 6.
Likewise, the use of the more selective manganese catalyst with the sample aqPUR2 has resulted in obtaining the similar increase of the total resulting decomposition time. The relatively shorter total resulting time of the decomposition that has occurred with the sample aqPUR1 in relation to the aqPUR2 sample shows that the sample aqPUR1 is less thermally stable.
Conclusion
The thermogravimetric dynamic method has been used to study the thermal stability of the aqueous polyurethane materials. Different degradation stages can be observed by using the derivative of the degree of degradation after the dynamic experiments.
The relatively shorter total resulting time of the decompositon with the sample aqPUR1 than with the sample aqPUR2 indicates that the sample aqPUR1 has the lesser thermal stability.
The aqueous polyurethanes (aqPUR2) based on the polyol Macrynal 2521 component, with the more selective catalyst (MnCAT), demonstrate the higher thermal stability than the aqueous polyurethanes (aqPUR1) based on the polyol Macrynal 6299 component, also with the more selective catalyst (MnCAT).
Acknowledgments
We gratefully acknowledge the financial support from the Ministry of Science, Technology and Development, Republic of Serbia (grant number ON 142039, BTN 351002.B and BTN 351004.B) for support of the work.
References
- Brinkman, E.; Vandevoorde, P. Waterborne two-pack isocyanate-free systems for industrial coatings. Progr. Org. Coatings 1997, 34, 21–25. [Google Scholar]
- Billiani, J.; Wilfinger, W. New low VOC acrylic polyol dispersions for two-component polyurethane coatings. 79th Annual meeting of the FSCT, Atalanta, 5-7.november, 2001.
- Billiani, J.; Wilfinger, W. Waterborne acrylic polyols for 2k waterborne polyuethane coatings. European coatings show, Nurnberg, 2-5. april, 2001.
- Blank, W.J.; He, Z.A.; Hessell, E.T. Catalysis of the isocyanate-hydroxyl reaction by non-tin catalysts. Progr. Org. Coatings 1999, 35, 19–29. [Google Scholar]
- He, Z.A.; Blank, W.J.; Picci, M.E. A selective catalyst for two-component waterborne polyurethane coatings. J. Coatings Tech. 2002, 74, 31–36. [Google Scholar]
- Stamenković, J.; Cakić, S.; Nikolić, G. Study of the catalytic selectivity of an aqueous two-component polyurethane system by FTIR spectroscopy. Chem. Industry 2003, 57, 559–569. [Google Scholar]
- Stamenković, J.; Cakić, S.; Konstantinović, S.; Stoilković, S. Catalysis of the isocyanate-hydroxyl reaction by non-tin catalysts in waterborne two-component polyurethane coatings. Facta Univesitatis 2004, 2, 243–250. [Google Scholar]
- Petrović, Z.S.; Zavargo, Z. Reliability of methods for determination of kinetic parameters from thermogravimetry and DSC measurements. J. Appl. Polym. Sci. 1986, 32, 4353–4368. [Google Scholar]
- Jimenez, A.; Berenguer, V.; Lopez, J.; Sanchez, A. Thermal degradation study of poly(vinyl chloride). Kinetic analysis of thermogravimetric data. J. Appl. Polym. Sci. 1993, 50, 1565–1573. [Google Scholar]
- Petrović, Z.S.; Zavargo, Z.; Flynn, J.H.; Macknight, W. Thermal degradation of segmented polyurethanes. J. Appl. Polym. Sci. 1994, 51, 1087–1095. [Google Scholar]
- Denq, B.L.; Chiu, W.Y.; Lin, K.F. Kinetic model of thermal degradation of polymers for nonisothermal process. J. Appl. Polym. Sci. 1997, 66, 1855–1868. [Google Scholar]
- Pegoretti, A.; Penati, A.; Kolarik, J. Effect of hydrolysis on molar mass and thermal properties of poly(ester urethanes). J. Therm. Anal. 1944, 41, 1441–1452. [Google Scholar]
- Fambri, L.; Pegoretti, A.; Kolarik, J.; Gavazza, C.; Penati, A. Thermal stabilities of different polyurethanes after hydrolytic treatment. J. Therm. Anal. 1998, 52, 789–797. [Google Scholar]
- Park, J.W.; Oh, S.C.; Lee, H.P.; Hee, T.K.; Yoo, K.O. A kinetic analysis of thermal degradation of polymers using a dynamic method. Polym. Degrad. Stab. 2000, 67, 535–540. [Google Scholar]
- Cakic, S.; Lacnjevac, C.; Rajkovic, M.B.; Raskovic, Lj.; Stamenkovic, J. Reticulation of Agueous Polyurethane Systems Controlled by DSC Method. Sensors 2006, 6, 536–545. [Google Scholar]
- Agic, A.; Bajsic, E.G.; Rek, V. Kinetic parameters estimation for thermal degradation of polyurethane elastomers. J. Elast. Plast. 2006, 38(2), 105–118. [Google Scholar]
- Coutinho, F.M.B.; Delpech, M.C. Degradation profile of films cast from aqueous polyurethane dispersions. Polym. Degrad.Stab. 2000, 70, 49–57. [Google Scholar]
- Chattopadhyay, D.K.; Sreedhar, B.; Raju, K.V.S.N. Thermal stability of chemically crosslinked moisture-cured polyurethane coatings. J. Appl. Polym. Sci. 2005, 96(6), 1509–1518. [Google Scholar]
- Fambri, L.; Pegoretti, A.; Gavazza, C.; Penati, A. Thermooxidative stability of different polyurethanes evaluated by isothermal and dynamic methods. J. Appl. Polym. Sci. 2001, 81, 1216–1225. [Google Scholar]
- Flynn, J.H. Temperature dependence of the rate of reaction in thermal analysis. The Arrhenius equation in condensed phase kinetics. J.Therm. Anal. 1990, 36(4), 1579–1593. [Google Scholar]
- Flynn, J.H. General differential technique for the determination of parameters for d(α)/dt = f(α)A exp (-E/RT). Energy of activation, preexponential factor and order of reaction (when applicable). J. Therm. Anal. 1991, 37, 293–305. [Google Scholar]
- K-KAT®XC-6212 a product of King Industries Inc. US patent 5.846.897.
- Barendregt, R.B.; Berg, P. The degradation of polyurethane. Thermochim. Acta 1980, 38, 181–195. [Google Scholar]
© 2006 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.