Potential Application of Small Interfering RNA in Gastro-Intestinal Tumors
Abstract
1. Gastrointestinal Cancers
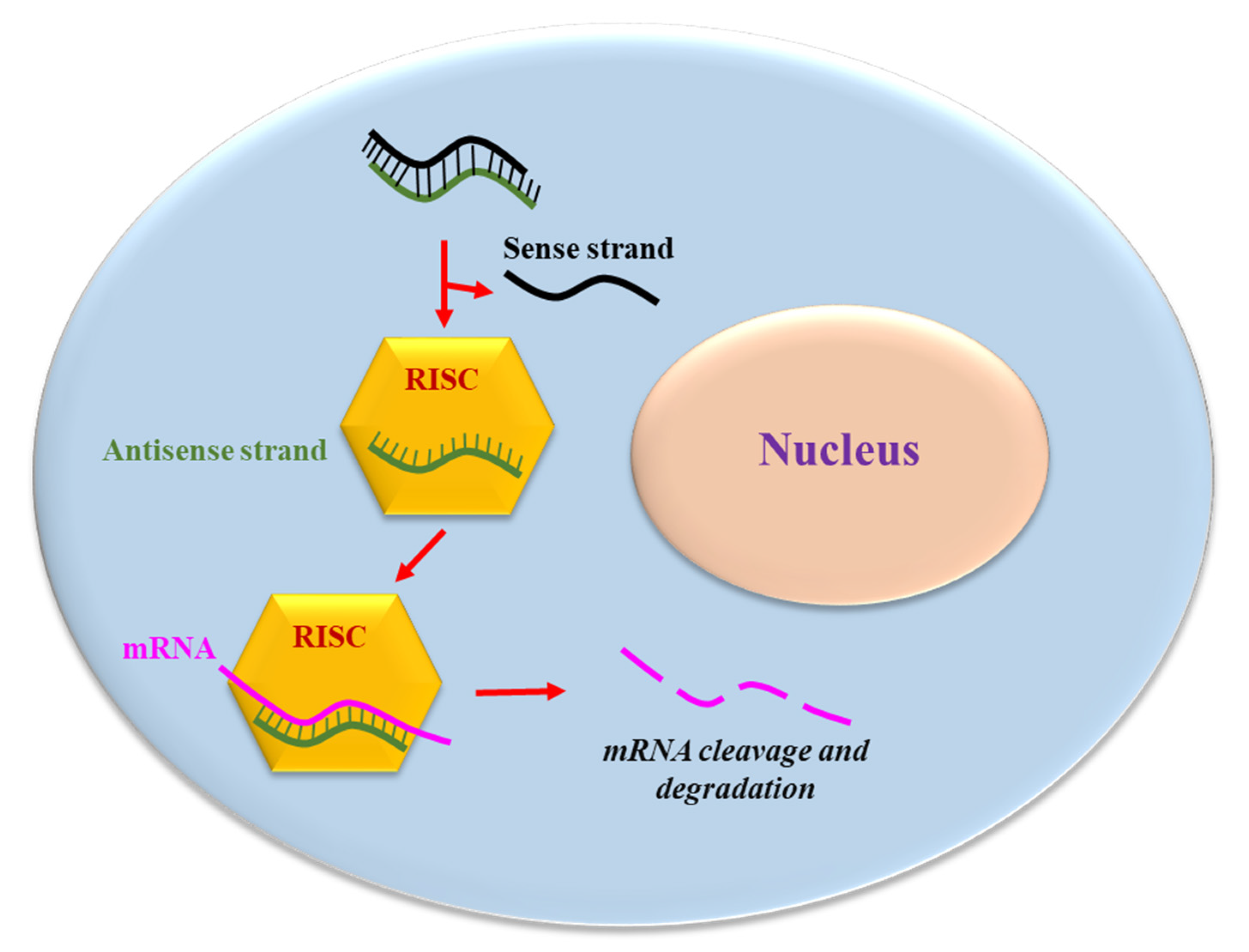
2. siRNA Structure, Function, and Delivery
2.1. siRNA Delivery to the Target Cell
2.2. Strategies to Optimize siRNA Delivery
2.2.1. Lipid-Based Delivery Materials
2.2.2. Polymer-Based Delivery Materials
2.2.3. Other Delivery Materials
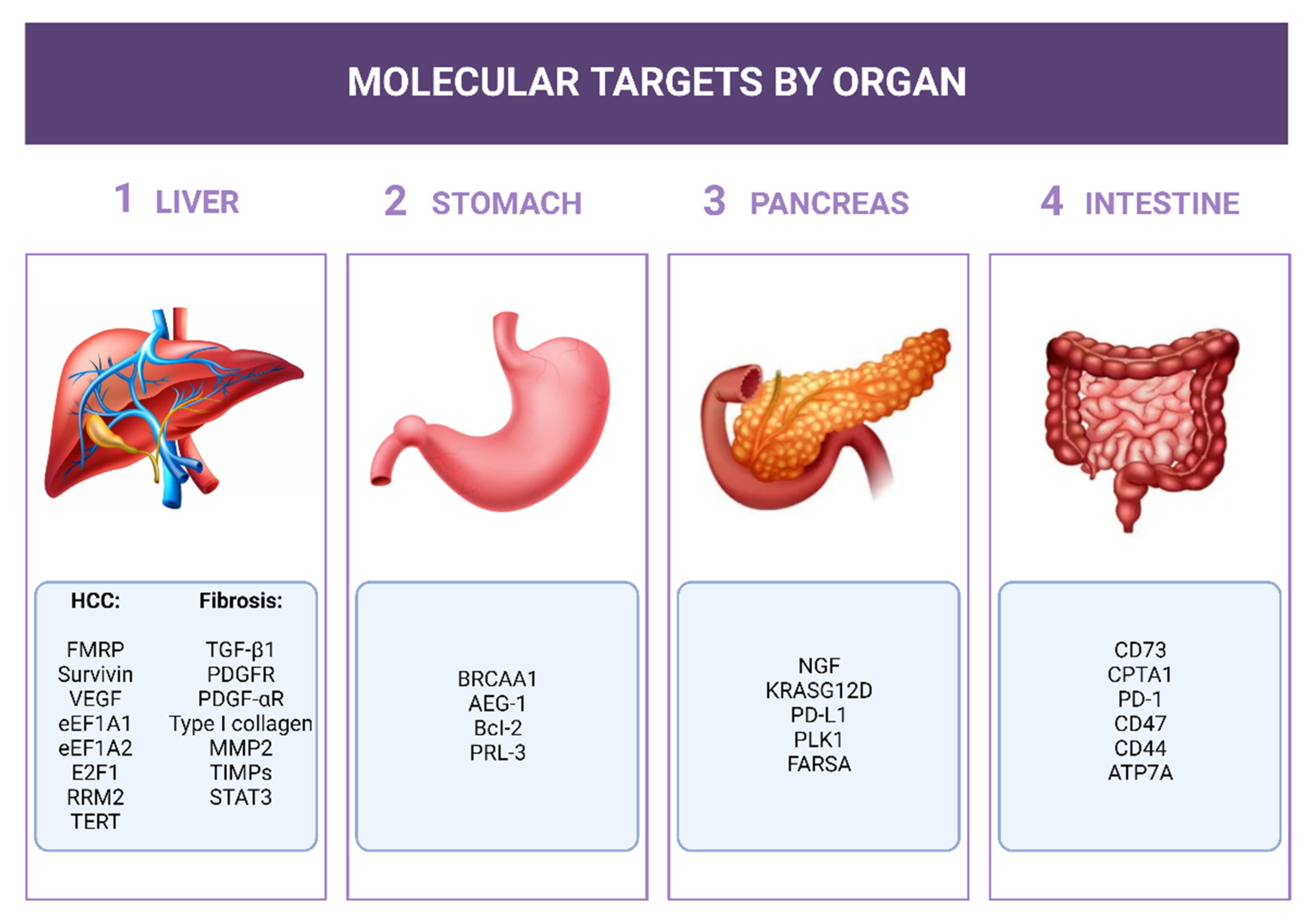
3. siRNAs for the GI Cancers
3.1. Potential Role of siRNAs in Upper-GI Cancers
3.1.1. Stomach
3.1.2. Pancreas
3.1.3. Liver
Liver Fibrosis
Hepatocellular Carcinoma
3.2. Roles of siRNAs Target Therapy in Lower-GI Cancers
Colorectal Cancer
4. Clinical Trials
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.A.; Tavilla, A.; Brenner, H.; Luttmann, S.; Navarro, C.; Gavin, A.T.; Holleczek, B.; Johnston, B.T.; Cook, M.B.; Bannon, F.; et al. Survival for oesophageal, stomach and small intestine cancers in Europe 1999-2007: Results from EUROCARE-5. Eur. J. Cancer 2015, 51, 2144–2157. [Google Scholar] [CrossRef] [PubMed]
- Chidambaranathan-Reghupaty, S.; Fisher, P.B.; Sarkar, D. Hepatocellular carcinoma (HCC): Epidemiology, etiology and molecular classification. Adv. Cancer Res. 2021, 149, 1–61. [Google Scholar] [CrossRef] [PubMed]
- Kulik, L.; El-Serag, H.B. Epidemiology and Management of Hepatocellular Carcinoma. Gastroenterology 2019, 156, 477–491. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Jemal, A.; Siegel, R.; Ward, E.; Murray, T.; Xu, J.; Smigal, C.; Thun, M.J. Cancer statistics, 2006. CA Cancer J. Clin. 2006, 56, 106–130. [Google Scholar] [CrossRef]
- Bakkevold, K.E.; Arnesjo, B.; Dahl, O.; Kambestad, B. Adjuvant combination chemotherapy (AMF) following radical resection of carcinoma of the pancreas and papilla of Vater—Results of a controlled, prospective, randomised multicentre study. Eur. J. Cancer 1993, 29A, 698–703. [Google Scholar] [CrossRef]
- Trede, M.; Schwall, G.; Saeger, H.D. Survival after pancreatoduodenectomy. 118 consecutive resections without an operative mortality. Ann. Surg. 1990, 211, 447–458. [Google Scholar] [CrossRef]
- Losurdo, P.; Giacca, M.; Biloslavo, A.; Fracon, S.; Sereni, E.; Giudici, F.; Generali, D.; de Manzini, N. Colorectal cancer-screening program improves both short- and long-term outcomes: A single-center experience in Trieste. Updates Surg. 2020, 72, 89–96. [Google Scholar] [CrossRef]
- Noone, A.M.; Cronin, K.A.; Altekruse, S.F.; Howlader, N.; Lewis, D.R.; Petkov, V.I.; Penberthy, L. Cancer Incidence and Survival Trends by Subtype Using Data from the Surveillance Epidemiology and End Results Program, 1992–2013. Cancer Epidemiol. Biomark. Prev. 2017, 26, 632–641. [Google Scholar] [CrossRef]
- Arnold, M.; Sierra, M.S.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global patterns and trends in colorectal cancer incidence and mortality. Gut 2017, 66, 683–691. [Google Scholar] [CrossRef]
- Allemani, C.; Weir, H.K.; Carreira, H.; Harewood, R.; Spika, D.; Wang, X.S.; Bannon, F.; Ahn, J.V.; Johnson, C.J.; Bonaventure, A.; et al. Global surveillance of cancer survival 1995–2009: Analysis of individual data for 25,676,887 patients from 279 population-based registries in 67 countries (CONCORD-2). Lancet 2015, 385, 977–1010. [Google Scholar] [CrossRef]
- Scaggiante, B.; Dapas, B.; Farra, R.; Grassi, M.; Pozzato, G.; Giansante, C.; Fiotti, N.; Grassi, G. Improving siRNA bio-distribution and minimizing side effects. Curr. Drug Metab. 2011, 12, 11–23. [Google Scholar] [CrossRef]
- Elbashir, S.M.; Harborth, J.; Lendeckel, W.; Yalcin, A.; Weber, K.; Tuschl, T. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature 2001, 411, 494–498. [Google Scholar] [CrossRef]
- Elsner, M. Single-stranded siRNAs for in vivo gene silencing. Nat. Biotechnol. 2012, 30, 1063. [Google Scholar] [CrossRef]
- Poliseno, L.; Evangelista, M.; Mercatanti, A.; Mariani, L.; Citti, L.; Rainaldi, G. The energy profiling of short interfering RNAs is highly predictive of their activity. Oligonucleotides 2004, 14, 227–232. [Google Scholar] [CrossRef]
- Reynolds, A.; Leake, D.; Boese, Q.; Scaringe, S.; Marshall, W.S.; Khvorova, A. Rational siRNA design for RNA interference. Nat. Biotechnol. 2004, 22, 326–330. [Google Scholar] [CrossRef]
- Grassi, G.; Dawson, P.; Guarnieri, G.; Kandolf, R.; Grassi, M. Therapeutic potential of hammerhead ribozymes in the treatment of hyper-proliferative diseases. Curr. Pharm. Biotechnol. 2004, 5, 369–386. [Google Scholar] [CrossRef]
- Grassi, G.; Marini, J.C. Ribozymes: Structure, function, and potential therapy for dominant genetic disorders. Ann. Med. 1996, 28, 499–510. [Google Scholar] [CrossRef]
- Farra, R.; Musiani, F.; Perrone, F.; Cemazar, M.; Kamensek, U.; Tonon, F.; Abrami, M.; Rucigaj, A.; Grassi, M.; Pozzato, G.; et al. Polymer-Mediated Delivery of siRNAs to Hepatocellular Carcinoma: Variables Affecting Specificity and Effectiveness. Molecules 2018, 23, 777. [Google Scholar] [CrossRef]
- Scarabel, L.; Perrone, F.; Garziera, M.; Farra, R.; Grassi, M.; Musiani, F.; Russo Spena, C.; Salis, B.; De Stefano, L.; Toffoli, G.; et al. Strategies to optimize siRNA delivery to hepatocellular carcinoma cells. Expert Opin. Drug Deliv. 2017, 14, 797–810. [Google Scholar] [CrossRef] [PubMed]
- Barba, A.A.; Lamberti, G.; Sardo, C.; Dapas, B.; Abrami, M.; Grassi, M.; Farra, R.; Tonon, F.; Forte, G.; Musiani, F.; et al. Novel Lipid and Polymeric Materials as Delivery Systems for Nucleic Acid Based Drugs. Curr. Drug Metab. 2015, 16, 427–452. [Google Scholar] [CrossRef] [PubMed]
- Grassi, G.; Schneider, A.; Engel, S.; Racchi, G.; Kandolf, R.; Kuhn, A. Hammerhead ribozymes targeted against cyclin E and E2F1 cooperate to down-regulate coronary smooth muscle cell proliferation. J. Gene Med. 2005, 7, 1223–1234. [Google Scholar] [CrossRef] [PubMed]
- Juliano, R.L. Chemical Manipulation of the Endosome Trafficking Machinery: Implications for Oligonucleotide Delivery. Biomedicines 2021, 9, 512. [Google Scholar] [CrossRef]
- Roberts, T.C.; Langer, R.; Wood, M.J.A. Advances in oligonucleotide drug delivery. Nat. Rev. Drug Discov. 2020, 19, 673–694. [Google Scholar] [CrossRef]
- Huang, Y.; Hong, J.; Zheng, S.; Ding, Y.; Guo, S.; Zhang, H.; Zhang, X.; Du, Q.; Liang, Z. Elimination pathways of systemically delivered siRNA. Mol. Ther. 2011, 19, 381–385. [Google Scholar] [CrossRef]
- Kaneda, Y. Gene therapy: A battle against biological barriers. Curr. Mol. Med. 2001, 1, 493–499. [Google Scholar] [CrossRef]
- Roberts, M.J.; Bentley, M.D.; Harris, J.M. Chemistry for peptide and protein PEGylation. Adv. Drug Deliv. Rev. 2002, 54, 459–476. [Google Scholar] [CrossRef]
- Kalluri, R.; LeBleu, V.S. The biology, function, and biomedical applications of exosomes. Science 2020, 367, eaau6977. [Google Scholar] [CrossRef]
- Natasha, G.; Gundogan, B.; Tan, A.; Farhatnia, Y.; Wu, W.; Rajadas, J.; Seifalian, A.M. Exosomes as immunotheranostic nanoparticles. Clin. Ther. 2014, 36, 820–829. [Google Scholar] [CrossRef]
- Farra, R.; Maruna, M.; Perrone, F.; Grassi, M.; Benedetti, F.; Maddaloni, M.; El Boustani, M.; Parisi, S.; Rizzolio, F.; Forte, G.; et al. Strategies for Delivery of siRNAs to Ovarian Cancer Cells. Pharmaceutics 2019, 11, 547. [Google Scholar] [CrossRef]
- Halib, N.; Pavan, N.; Trombetta, C.; Dapas, B.; Farra, R.; Scaggiante, B.; Grassi, M.; Grassi, G. An Overview of siRNA Delivery Strategies for Urological Cancers. Pharmaceutics 2022, 14, 718. [Google Scholar] [CrossRef]
- Park, T.G.; Jeong, J.H.; Kim, S.W. Current status of polymeric gene delivery systems. Adv. Drug Deliv. Rev. 2006, 58, 467–486. [Google Scholar] [CrossRef]
- York, A.W.; Kirkland, S.E.; McCormick, C.L. Advances in the synthesis of amphiphilic block copolymers via RAFT polymerization: Stimuli-responsive drug and gene delivery. Adv. Drug Deliv. Rev. 2008, 60, 1018–1036. [Google Scholar] [CrossRef]
- Makadia, H.K.; Siegel, S.J. Poly Lactic-co-Glycolic Acid (PLGA) as Biodegradable Controlled Drug Delivery Carrier. Polymers 2011, 3, 1377–1397. [Google Scholar] [CrossRef]
- Dufes, C.; Uchegbu, I.F.; Schatzlein, A.G. Dendrimers in gene delivery. Adv. Drug Deliv. Rev. 2005, 57, 2177–2202. [Google Scholar] [CrossRef]
- Khaitov, M.; Nikonova, A.; Shilovskiy, I.; Kozhikhova, K.; Kofiadi, I.; Vishnyakova, L.; Nikolskii, A.; Gattinger, P.; Kovchina, V.; Barvinskaia, E.; et al. Silencing of SARS-CoV-2 with modified siRNA-peptide dendrimer formulation. Allergy 2021, 76, 2840–2854. [Google Scholar] [CrossRef]
- Vivero-Escoto, J.L.; Slowing, I.I.; Trewyn, B.G.; Lin, V.S.Y. Mesoporous silica nanoparticles for intracellular controlled drug delivery. Small 2010, 6, 1952–1967. [Google Scholar] [CrossRef]
- Hanafi-Bojd, M.Y.; Ansari, L.; Malaekeh-Nikouei, B. Codelivery of anticancer drugs and siRNA by mesoporous silica nanoparticles. Ther. Deliv. 2016, 7, 649–655. [Google Scholar] [CrossRef]
- Singh, P.; Pandit, S.; Mokkapati, V.; Garg, A.; Ravikumar, V.; Mijakovic, I. Gold Nanoparticles in Diagnostics and Therapeutics for Human Cancer. Int. J. Mol. Sci. 2018, 19, 1979. [Google Scholar] [CrossRef]
- Rahme, K.; Guo, J.; Holmes, J.D. Bioconjugated Gold Nanoparticles Enhance siRNA Delivery in Prostate Cancer Cells. Methods Mol. Biol. 2019, 1974, 291–301. [Google Scholar] [CrossRef]
- Soutschek, J.; Akinc, A.; Bramlage, B.; Charisse, K.; Constien, R.; Donoghue, M.; Elbashir, S.; Geick, A.; Hadwiger, P.; Harborth, J.; et al. Therapeutic silencing of an endogenous gene by systemic administration of modified siRNAs. Nature 2004, 432, 173–178. [Google Scholar] [CrossRef]
- Khan, T.; Weber, H.; DiMuzio, J.; Matter, A.; Dogdas, B.; Shah, T.; Thankappan, A.; Disa, J.; Jadhav, V.; Lubbers, L.; et al. Silencing Myostatin Using Cholesterol-conjugated siRNAs Induces Muscle Growth. Mol. Ther. Nucleic Acids 2016, 5, e342. [Google Scholar] [CrossRef]
- McNamara, J.O., 2nd; Andrechek, E.R.; Wang, Y.; Viles, K.D.; Rempel, R.E.; Gilboa, E.; Sullenger, B.A.; Giangrande, P.H. Cell type-specific delivery of siRNAs with aptamer-siRNA chimeras. Nat. Biotechnol. 2006, 24, 1005–1015. [Google Scholar] [CrossRef]
- Moschos, S.A.; Jones, S.W.; Perry, M.M.; Williams, A.E.; Erjefalt, J.S.; Turner, J.J.; Barnes, P.J.; Sproat, B.S.; Gait, M.J.; Lindsay, M.A. Lung delivery studies using siRNA conjugated to TAT(48-60) and penetratin reveal peptide induced reduction in gene expression and induction of innate immunity. Bioconjug. Chem. 2007, 18, 1450–1459. [Google Scholar] [CrossRef]
- Sardh, E.; Harper, P.; Balwani, M.; Stein, P.; Rees, D.; Bissell, D.M.; Desnick, R.; Parker, C.; Phillips, J.; Bonkovsky, H.L.; et al. Phase 1 Trial of an RNA Interference Therapy for Acute Intermittent Porphyria. N. Engl. J. Med. 2019, 380, 549–558. [Google Scholar] [CrossRef]
- Ray, K.K.; Landmesser, U.; Leiter, L.A.; Kallend, D.; Dufour, R.; Karakas, M.; Hall, T.; Troquay, R.P.; Turner, T.; Visseren, F.L.; et al. Inclisiran in Patients at High Cardiovascular Risk with Elevated LDL Cholesterol. N. Engl. J. Med. 2017, 376, 1430–1440. [Google Scholar] [CrossRef]
- Cui, D.; Zhang, C.; Liu, B.; Shu, Y.; Du, T.; Shu, D.; Wang, K.; Dai, F.; Liu, Y.; Li, C.; et al. Regression of Gastric Cancer by Systemic Injection of RNA Nanoparticles Carrying both Ligand and siRNA. Sci. Rep. 2015, 5, 10726. [Google Scholar] [CrossRef]
- Jian-bo, X.; Hui, W.; Yu-long, H.; Chang-hua, Z.; Long-juan, Z.; Shi-rong, C.; Wen-hua, Z. Astrocyte-elevated gene-1 overexpression is associated with poor prognosis in gastric cancer. Med. Oncol. 2011, 28, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.T.; Lu, C.L. Effect of silencing Bcl-2 expression by small interfering RNA on radiosensitivity of gastric cancer BGC823 cells. Asian Pac. J. Trop. Med. 2013, 6, 49–52. [Google Scholar] [CrossRef]
- Cao, Y.; Tu, Y.; Mei, J.; Li, Z.; Jie, Z.; Xu, S.; Xu, L.; Wang, S.; Xiong, Y. RNAimediated knockdown of PRL3 inhibits cell invasion and downregulates ERK 1/2 expression in the human gastric cancer cell line, SGC7901. Mol. Med. Rep. 2013, 7, 1805–1811. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ying, Z.; Li, J.; Li, M. Astrocyte elevated gene 1: Biological functions and molecular mechanism in cancer and beyond. Cell Biosci. 2011, 1, 36. [Google Scholar] [CrossRef]
- Zhu, Z.; Kleeff, J.; Kayed, H.; Wang, L.; Korc, M.; Buchler, M.W.; Friess, H. Nerve growth factor and enhancement of proliferation, invasion, and tumorigenicity of pancreatic cancer cells. Mol. Carcinog. 2002, 35, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Lei, Y.; Tang, L.; Xie, Y.; Xianyu, Y.; Zhang, L.; Wang, P.; Hamada, Y.; Jiang, K.; Zheng, W.; Jiang, X. Gold nanoclusters-assisted delivery of NGF siRNA for effective treatment of pancreatic cancer. Nat. Commun. 2017, 8, 15130. [Google Scholar] [CrossRef]
- Kamerkar, S.; LeBleu, V.S.; Sugimoto, H.; Yang, S.; Ruivo, C.F.; Melo, S.A.; Lee, J.J.; Kalluri, R. Exosomes facilitate therapeutic targeting of oncogenic KRAS in pancreatic cancer. Nature 2017, 546, 498–503. [Google Scholar] [CrossRef]
- Jung, J.Y.; Ryu, H.J.; Lee, S.H.; Kim, D.Y.; Kim, M.J.; Lee, E.J.; Ryu, Y.M.; Kim, S.Y.; Kim, K.P.; Choi, E.Y.; et al. siRNA Nanoparticle Targeting PD-L1 Activates Tumor Immunity and Abrogates Pancreatic Cancer Growth in Humanized Preclinical Model. Cells 2021, 10, 2734. [Google Scholar] [CrossRef]
- Tang, S.; Hang, Y.; Ding, L.; Tang, W.; Yu, A.; Zhang, C.; Sil, D.; Xie, Y.; Oupicky, D. Intraperitoneal siRNA Nanoparticles for Augmentation of Gemcitabine Efficacy in the Treatment of Pancreatic Cancer. Mol. Pharm. 2021, 18, 4448–4458. [Google Scholar] [CrossRef]
- Yuan, H.; Huang, X.; Li, Q.; Luo, C.; Lin, C.; Zhang, S.; Zhang, Y.; Yan, Z.; Du, N.; Liu, Z.; et al. SiRNA-circFARSA-loaded porous silicon nanomaterials for pancreatic cancer treatment via inhibition of CircFARSA expression. Biomed. Pharm. 2022, 147, 112672. [Google Scholar] [CrossRef]
- Hang, Y.; Tang, S.; Tang, W.; Vetvicka, D.; Zhang, C.; Xie, Y.; Yu, F.; Yu, A.; Sil, D.; Li, J.; et al. Polycation fluorination improves intraperitoneal siRNA delivery in metastatic pancreatic cancer. J. Control. Release 2021, 333, 139–150. [Google Scholar] [CrossRef]
- Zhao, Z.J.; Shen, J. Circular RNA participates in the carcinogenesis and the malignant behavior of cancer. RNA Biol. 2017, 14, 514–521. [Google Scholar] [CrossRef]
- Acharya, P.; Chouhan, K.; Weiskirchen, S.; Weiskirchen, R. Cellular Mechanisms of Liver Fibrosis. Front. Pharm. 2021, 12, 671640. [Google Scholar] [CrossRef]
- Marcellin, P.; Kutala, B.K. Liver diseases: A major, neglected global public health problem requiring urgent actions and large-scale screening. Liver Int. 2018, 38 (Suppl. 1), 2–6. [Google Scholar] [CrossRef]
- Kanda, T.; Goto, T.; Hirotsu, Y.; Moriyama, M.; Omata, M. Molecular Mechanisms Driving Progression of Liver Cirrhosis towards Hepatocellular Carcinoma in Chronic Hepatitis B and C Infections: A Review. Int. J. Mol. Sci. 2019, 20, 1358. [Google Scholar] [CrossRef]
- Higashi, T.; Friedman, S.L.; Hoshida, Y. Hepatic stellate cells as key target in liver fibrosis. Adv. Drug Deliv. Rev. 2017, 121, 27–42. [Google Scholar] [CrossRef]
- Parola, M.; Pinzani, M. Liver fibrosis: Pathophysiology, pathogenetic targets and clinical issues. Mol. Asp. Med. 2019, 65, 37–55. [Google Scholar] [CrossRef]
- Kim, K.H.; Kim, H.C.; Hwang, M.Y.; Oh, H.K.; Lee, T.S.; Chang, Y.C.; Song, H.J.; Won, N.H.; Park, K.K. The antifibrotic effect of TGF-beta1 siRNAs in murine model of liver cirrhosis. Biochem. Biophys. Res. Commun. 2006, 343, 1072–1078. [Google Scholar] [CrossRef]
- Cheng, K.; Yang, N.; Mahato, R.I. TGF-beta1 gene silencing for treating liver fibrosis. Mol. Pharm. 2009, 6, 772–779. [Google Scholar] [CrossRef]
- Chen, S.W.; Chen, Y.X.; Zhang, X.R.; Qian, H.; Chen, W.Z.; Xie, W.F. Targeted inhibition of platelet-derived growth factor receptor-beta subunit in hepatic stellate cells ameliorates hepatic fibrosis in rats. Gene Ther. 2008, 15, 1424–1435. [Google Scholar] [CrossRef]
- Lim, B.J.; Lee, W.K.; Lee, H.W.; Lee, K.S.; Kim, J.K.; Chang, H.Y.; Lee, J.I. Selective deletion of hepatocyte platelet-derived growth factor receptor alpha and development of liver fibrosis in mice. Cell Commun. Signal. 2018, 16, 93. [Google Scholar] [CrossRef]
- Jimenez Calvente, C.; Sehgal, A.; Popov, Y.; Kim, Y.O.; Zevallos, V.; Sahin, U.; Diken, M.; Schuppan, D. Specific hepatic delivery of procollagen alpha1(I) small interfering RNA in lipid-like nanoparticles resolves liver fibrosis. Hepatology 2015, 62, 1285–1297. [Google Scholar] [CrossRef] [PubMed]
- Toriyabe, N.; Sakurai, Y.; Kato, A.; Yamamoto, S.; Tange, K.; Nakai, Y.; Akita, H.; Harahsima, H. The Delivery of Small Interfering RNA to Hepatic Stellate Cells Using a Lipid Nanoparticle Composed of a Vitamin A-Scaffold Lipid-Like Material. J. Pharm. Sci. 2017, 106, 2046–2052. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, F.; Ding, F.; Chen, P.; Tang, M. Inhibition of liver fibrosis using vitamin A-coupled liposomes to deliver matrix metalloproteinase-2 siRNA in vitro. Mol. Med. Rep. 2015, 12, 3453–3461. [Google Scholar] [CrossRef] [PubMed]
- Fowell, A.J.; Collins, J.E.; Duncombe, D.R.; Pickering, J.A.; Rosenberg, W.M.; Benyon, R.C. Silencing tissue inhibitors of metalloproteinases (TIMPs) with short interfering RNA reveals a role for TIMP-1 in hepatic stellate cell proliferation. Biochem. Biophys. Res. Commun. 2011, 407, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.; Chen, Y.; Li, B.; Sugimoto, H.; Yang, S.; Yang, C.; LeBleu, V.S.; McAndrews, K.M.; Kalluri, R. Therapeutic targeting of STAT3 with small interference RNAs and antisense oligonucleotides embedded exosomes in liver fibrosis. FASEB J. 2021, 35, e21557. [Google Scholar] [CrossRef]
- Zhao, X.; Yang, J.; Zhang, J.; Wang, X.; Chen, L.; Zhang, C.; Shen, Z. Inhibitory effect of aptamer-carbon dot nanomaterial-siRNA complex on the metastasis of hepatocellular carcinoma cells by interfering with FMRP. Eur. J. Pharm. Biopharm. 2022, 174, 47–55. [Google Scholar] [CrossRef]
- Han, L.; Tang, C.; Yin, C. Oral delivery of shRNA and siRNA via multifunctional polymeric nanoparticles for synergistic cancer therapy. Biomaterials 2014, 35, 4589–4600. [Google Scholar] [CrossRef]
- Perrone, F.; Craparo, E.F.; Cemazar, M.; Kamensek, U.; Drago, S.E.; Dapas, B.; Scaggiante, B.; Zanconati, F.; Bonazza, D.; Grassi, M.; et al. Targeted delivery of siRNAs against hepatocellular carcinoma-related genes by a galactosylated polyaspartamide copolymer. J. Control. Release 2021, 330, 1132–1151. [Google Scholar] [CrossRef]
- Zhao, B.; Zhou, B.; Shi, K.; Zhang, R.; Dong, C.; Xie, D.; Tang, L.; Tian, Y.; Qian, Z.; Yang, L. Sustained and targeted delivery of siRNA/DP7-C nanoparticles from injectable thermosensitive hydrogel for hepatocellular carcinoma therapy. Cancer Sci. 2021, 112, 2481–2492. [Google Scholar] [CrossRef]
- Gao, J.; Chen, H.; Yu, Y.; Song, J.; Song, H.; Su, X.; Li, W.; Tong, X.; Qian, W.; Wang, H.; et al. Inhibition of hepatocellular carcinoma growth using immunoliposomes for co-delivery of adriamycin and ribonucleotide reductase M2 siRNA. Biomaterials 2013, 34, 10084–10098. [Google Scholar] [CrossRef]
- Hu, Y.; Shen, Y.; Ji, B.; Wang, L.; Zhang, Z.; Zhang, Y. Combinational RNAi gene therapy of hepatocellular carcinoma by targeting human EGFR and TERT. Eur. J. Pharm. Sci. 2011, 42, 387–391. [Google Scholar] [CrossRef]
- Han, L.; Tang, C.; Yin, C. Enhanced antitumor efficacies of multifunctional nanocomplexes through knocking down the barriers for siRNA delivery. Biomaterials 2015, 44, 111–121. [Google Scholar] [CrossRef]
- Wu, C.; Gong, F.; Pang, P.; Shen, M.; Zhu, K.; Cheng, D.; Liu, Z.; Shan, H. An RGD-modified MRI-visible polymeric vector for targeted siRNA delivery to hepatocellular carcinoma in nude mice. PLoS ONE 2013, 8, e66416. [Google Scholar] [CrossRef]
- Craparo, E.F.; Sardo, C.; Serio, R.; Zizzo, M.G.; Bondi, M.L.; Giammona, G.; Cavallaro, G. Galactosylated polymeric carriers for liver targeting of sorafenib. Int. J. Pharm. 2014, 466, 172–180. [Google Scholar] [CrossRef]
- Farra, R.; Grassi, G.; Tonon, F.; Abrami, M.; Grassi, M.; Pozzato, G.; Fiotti, N.; Forte, G.; Dapas, B. The Role of the Transcription Factor E2F1 in Hepatocellular Carcinoma. Curr. Drug Deliv. 2017, 14, 272–281. [Google Scholar] [CrossRef]
- Farra, R.; Scaggiante, B.; Guerra, C.; Pozzato, G.; Grassi, M.; Zanconati, F.; Perrone, F.; Ferrari, C.; Trotta, F.; Grassi, G.; et al. Dissecting the role of the elongation factor 1A isoforms in hepatocellular carcinoma cells by liposome-mediated delivery of siRNAs. Int. J. Pharm. 2017, 525, 367–376. [Google Scholar] [CrossRef]
- Zhou, X.Z.; Lu, K.P. The isomerase PIN1 controls numerous cancer-driving pathways and is a unique drug target. Nat. Rev. Cancer 2016, 16, 463–478. [Google Scholar] [CrossRef]
- Sun, H.L.; Cui, R.; Zhou, J.; Teng, K.Y.; Hsiao, Y.H.; Nakanishi, K.; Fassan, M.; Luo, Z.; Shi, G.; Tili, E.; et al. ERK Activation Globally Downregulates miRNAs through Phosphorylating Exportin-5. Cancer Cell 2016, 30, 723–736. [Google Scholar] [CrossRef]
- Gao, J.; Yu, Y.; Zhang, Y.; Song, J.; Chen, H.; Li, W.; Qian, W.; Deng, L.; Kou, G.; Chen, J.; et al. EGFR-specific PEGylated immunoliposomes for active siRNA delivery in hepatocellular carcinoma. Biomaterials 2012, 33, 270–282. [Google Scholar] [CrossRef]
- Satow, R.; Shitashige, M.; Kanai, Y.; Takeshita, F.; Ojima, H.; Jigami, T.; Honda, K.; Kosuge, T.; Ochiya, T.; Hirohashi, S.; et al. Combined functional genome survey of therapeutic targets for hepatocellular carcinoma. Clin. Cancer Res. 2010, 16, 2518–2528. [Google Scholar] [CrossRef]
- Sutherland, R.; Delia, D.; Schneider, C.; Newman, R.; Kemshead, J.; Greaves, M. Ubiquitous cell-surface glycoprotein on tumor cells is proliferation-associated receptor for transferrin. Proc. Natl. Acad. Sci. USA 1981, 78, 4515–4519. [Google Scholar] [CrossRef]
- Yang, X.; Guo, X.; Chen, Y.; Chen, G.; Ma, Y.; Huang, K.; Zhang, Y.; Zhao, Q.; Winkler, C.A.; An, P.; et al. Telomerase reverse transcriptase promoter mutations in hepatitis B virus-associated hepatocellular carcinoma. Oncotarget 2016, 7, 27838–27847. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Liu, Y.; Wang, W.; Liu, K. Effect of integrin receptor-targeted liposomal paclitaxel for hepatocellular carcinoma targeting and therapy. Oncol. Lett. 2021, 21, 350. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B. CD73: A novel target for cancer immunotherapy. Cancer Res. 2010, 70, 6407–6411. [Google Scholar] [CrossRef] [PubMed]
- Salehi Khesht, A.M.; Karpisheh, V.; Sahami Gilan, P.; Melnikova, L.A.; Olegovna Zekiy, A.; Mohammadi, M.; Hojjat-Farsangi, M.; Majidi Zolbanin, N.; Mahmoodpoor, A.; Hassannia, H.; et al. Blockade of CD73 using siRNA loaded chitosan lactate nanoparticles functionalized with TAT-hyaluronate enhances doxorubicin mediated cytotoxicity in cancer cells both in vitro and in vivo. Int. J. Biol. Macromol. 2021, 186, 849–863. [Google Scholar] [CrossRef]
- Lin, D.; Zhang, H.; Liu, R.; Deng, T.; Ning, T.; Bai, M.; Yang, Y.; Zhu, K.; Wang, J.; Duan, J.; et al. iRGD-modified exosomes effectively deliver CPT1A siRNA to colon cancer cells, reversing oxaliplatin resistance by regulating fatty acid oxidation. Mol. Oncol. 2021, 15, 3430–3446. [Google Scholar] [CrossRef]
- Lu, S.; Gao, J.; Jia, H.; Li, Y.; Duan, Y.; Song, F.; Liu, Z.; Ma, S.; Wang, M.; Zhao, T.; et al. PD-1-siRNA Delivered by Attenuated Salmonella Enhances the Antitumor Effect of Chloroquine in Colon Cancer. Front. Immunol. 2021, 12, 707991. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Z.; Li, S.; Zhao, L.; Li, D.; Cao, Z.; Xu, X.; Yang, X. A siRNA-Assisted Assembly Strategy to Simultaneously Suppress "Self" and Upregulate “Eat-Me” Signals for Nanoenabled Chemo-Immunotherapy. ACS Nano 2021, 15, 16030–16042. [Google Scholar] [CrossRef]
- Zou, W.; Zhang, Y.; Bai, G.; Zhuang, J.; Wei, L.; Wang, Z.; Sun, M.; Wang, J. siRNA-induced CD44 knockdown suppresses the proliferation and invasion of colorectal cancer stem cells through inhibiting epithelial-mesenchymal transition. J. Cell. Mol. Med. 2022, 26, 1969–1978. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, Q.; Wang, M.; Huang, C.; Yao, X. Corrigendum: Effective Delivery of siRNA-Loaded Nanoparticles for Overcoming Oxaliplatin Resistance in Colorectal Cancer. Front. Oncol. 2022, 12, 916983. [Google Scholar] [CrossRef]
- Ma, Y.; Temkin, S.M.; Hawkridge, A.M.; Guo, C.; Wang, W.; Wang, X.Y.; Fang, X. Fatty acid oxidation: An emerging facet of metabolic transformation in cancer. Cancer Lett. 2018, 435, 92–100. [Google Scholar] [CrossRef]
- Hsu, J.; Hodgins, J.J.; Marathe, M.; Nicolai, C.J.; Bourgeois-Daigneault, M.C.; Trevino, T.N.; Azimi, C.S.; Scheer, A.K.; Randolph, H.E.; Thompson, T.W.; et al. Contribution of NK cells to immunotherapy mediated by PD-1/PD-L1 blockade. J. Clin. Investig. 2018, 128, 4654–4668. [Google Scholar] [CrossRef]
- Willingham, S.B.; Volkmer, J.P.; Gentles, A.J.; Sahoo, D.; Dalerba, P.; Mitra, S.S.; Wang, J.; Contreras-Trujillo, H.; Martin, R.; Cohen, J.D.; et al. The CD47-signal regulatory protein alpha (SIRPa) interaction is a therapeutic target for human solid tumors. Proc. Natl. Acad. Sci. USA 2012, 109, 6662–6667. [Google Scholar] [CrossRef]
- Chao, M.P.; Jaiswal, S.; Weissman-Tsukamoto, R.; Alizadeh, A.A.; Gentles, A.J.; Volkmer, J.; Weiskopf, K.; Willingham, S.B.; Raveh, T.; Park, C.Y.; et al. Calreticulin is the dominant pro-phagocytic signal on multiple human cancers and is counterbalanced by CD47. Sci. Transl. Med. 2010, 2, 63ra94. [Google Scholar] [CrossRef]
- Wang, L.; Zuo, X.; Xie, K.; Wei, D. The Role of CD44 and Cancer Stem Cells. Methods Mol. Biol. 2018, 1692, 31–42. [Google Scholar] [CrossRef]
- Petruzzelli, R.; Polishchuk, R.S. Activity and Trafficking of Copper-Transporting ATPases in Tumor Development and Defense against Platinum-Based Drugs. Cells 2019, 8, 1080. [Google Scholar] [CrossRef]
- Schultheis, B.; Strumberg, D.; Santel, A.; Vank, C.; Gebhardt, F.; Keil, O.; Lange, C.; Giese, K.; Kaufmann, J.; Khan, M.; et al. First-in-human phase I study of the liposomal RNA interference therapeutic Atu027 in patients with advanced solid tumors. J. Clin. Oncol. 2014, 32, 4141–4148. [Google Scholar] [CrossRef]
- Strumberg, D.; Schultheis, B.; Traugott, U.; Vank, C.; Santel, A.; Keil, O.; Giese, K.; Kaufmann, J.; Drevs, J. Phase I clinical development of Atu027, a siRNA formulation targeting PKN3 in patients with advanced solid tumors. Int. J. Clin. Pharmacol. Ther. 2012, 50, 76–78. [Google Scholar] [CrossRef]
- Zuckerman, J.E.; Gritli, I.; Tolcher, A.; Heidel, J.D.; Lim, D.; Morgan, R.; Chmielowski, B.; Ribas, A.; Davis, M.E.; Yen, Y. Correlating animal and human phase Ia/Ib clinical data with CALAA-01, a targeted, polymer-based nanoparticle containing siRNA. Proc. Natl. Acad. Sci. USA 2014, 111, 11449–11454. [Google Scholar] [CrossRef]
- Liu, J.; Guo, B. RNA-based therapeutics for colorectal cancer: Updates and future directions. Pharm. Res. 2020, 152, 104550. [Google Scholar] [CrossRef]
- Kulkarni, J.A.; Cullis, P.R.; van der Meel, R. Lipid Nanoparticles Enabling Gene Therapies: From Concepts to Clinical Utility. Nucleic Acid Ther. 2018, 28, 146–157. [Google Scholar] [CrossRef]
- Golan, T.; Khvalevsky, E.Z.; Hubert, A.; Gabai, R.M.; Hen, N.; Segal, A.; Domb, A.; Harari, G.; David, E.B.; Raskin, S.; et al. RNAi therapy targeting KRAS in combination with chemotherapy for locally advanced pancreatic cancer patients. Oncotarget 2015, 6, 24560–24570. [Google Scholar] [CrossRef]
- Schultheis, B.; Strumberg, D.; Kuhlmann, J.; Wolf, M.; Link, K.; Seufferlein, T.; Kaufmann, J.; Feist, M.; Gebhardt, F.; Khan, M.; et al. Safety, Efficacy and Pharcacokinetics of Targeted Therapy with The Liposomal RNA Interference Therapeutic Atu027 Combined with Gemcitabine in Patients with Pancreatic Adenocarcinoma. A Randomized Phase Ib/IIa Study. Cancers 2020, 12, 3130. [Google Scholar] [CrossRef]

| Material | Advantages | Disadvantages |
|---|---|---|
| Liposomes | Cheap synthesis Easy functionalization Biodegradable | Tendency to accumulate in the liver and lung if not functionalized Possible induction of mild inflammation |
| GalNac * | Targeting properties Biodegradable | |
| Exosomes | Excellent biodistribution and biocompatibility | Production by patient cells |
| PLGA * | Biocompatible biodegradable FDA approved | Not easy siRNA encapsulation due to PLGA hydrophobic nature |
| Chitosan | Easy functionalization | Low transfection efficiency and low solubility |
| Hyaluronic acid | Targeting ability toward cluster determinant 44, over-expressed in cancer cells Biodegradable Biocompatible | Functionalization required to bind siRNA due to its negative electric charge |
| Dendrimers | Great loading capacity related to the size | Possible toxicity related to particle size |
| Mesoporous silica | Biocompatible High surface area, the large pore volume, chemical and thermal stability | Functionalization required for targeting purposes |
| Gold nanoparticles | High surface area to volume ratio, possible multi-functionalization, easy synthesis, non-toxic non-immunogenic | Functionalization required to bind siRNA due to the negative electric charge |
| Target mRNA | Delivery System | Tumor Model | Ref. |
|---|---|---|---|
| BRCAA1 * | bacteriophage phi29 packaging motor with Folic acid targeting moiety | MGC803 cell line | Cui et al. [48] |
| AEG-1 * | Lipofectamine | SGC7901 cell line | Jian-bo et al. [49] |
| Bcl-2 * | Lipofectamine | BGC-823 cell line | Liu et al. [50] |
| PRL-3 * | Lipofectamine | SGC-7901 cell line | Cao et al. [51] |
| Target mRNA | Delivery System | Tumor Model | Ref. |
|---|---|---|---|
| NGF * | Gold nanoparticles | Panc-1 cell line; mouse subcutaneous model of pancreatic cancer; orthotopic patient-derived xenograft mouse model | Lei et al. [54] |
| KRASG12D * | Exosome | Panc-1 cell line; orthotopic mouse model | Kamerkar et al. [55] |
| PD-L1 * | PLGA | orthotopic model humanized mouse model | Jung et al. [56] |
| PLK1 * | Polymer | KPC8060 cell line; orthotopic syngenic mouse model (KPC8060); xenograft mouse model (S2-‘13) | Tang et al. [57] |
| FARSA * | Lipofectamine and porous silicon nanoparticles | SW1990 and Panc-1 cell lines; orthotopic patient-derived xenograft mouse model | Yuan et al. [58] |
| Target mRNA | Delivery System | Liver Fibrosis Model | Ref. |
|---|---|---|---|
| TGF-β1 * | Liposome | CCl4-induced murine model of liver fibrosis | Kim et al. [66] |
| TGF-β1 * | Lipofectamine | HSC-T6 | Cheng et al. [67] |
| PDGFR * | siRNA expression from glial fibrillary acidic protein promoter | CCl4-induced murine model of liver fibrosis and bile duct ligation induced chronic rat liver injury | Chen et al. [68] |
| PDGF α receptor * | Commercial liposome | LX2 | Lim et al. [69] |
| Type I collagen | Liposome | LX2; CCl4-induced murine model of liver fibrosis; spontaneous model of mouse biliary fibrosis | Calvente et al. [70] |
| Type I collagen | Lipid bound to vitamin A | LI-90; CCl4-induced murine model of liver fibrosis | Toriyabe et al. [71] |
| MMP2 * | Lipid bound to vitamin A | HSC-T6 | Li et al. [72] |
| TIMPs * | Electroporation | HSC isolated from normal livers of Sprague–Dawley rats. | Fowel et al. [73] |
| STAT3 * | Exosomes | HSC isolated from healthy mouse; CCl4-induced murine model of liver fibrosis | Tang et al. [74] |
| Target mRNA | Delivery System | Tumor Model | Ref. |
|---|---|---|---|
| FMRP * | Carbon dots conjugated with the aptamer AS1411 | HepG2 | Zhao et al. [75] |
| Survivin and VEGF * | Galactose-modified trimethyl chitosan-cysteine | Xenograft mouse model of HCC | Han et al. [76] |
| eEF1A1, eEF1A2 *, E2F1 | PDPG polymer bound to galactose | HuH7; Xenograft mouse model of HCC | Perrone et al. [77] |
| Pin1 | GalNac-siRNA embedded into a gel | Orthotopic mouse model | Zhao et al. [78] |
| RRM2 * | liposome-polycation-DNA complexes linked to anti-EGFR * Fab’ | Orthotopic mouse model | Gao et al. [79] |
| TERT * | PEGylated liposomes conjugated with antibodies against TfR * and HIR * | Xenograft mouse model of HCC | Hu et al. [80] |
| VEGF * | Polymer containing urocanic acid-modified galactosylated trimethyl chitosan | QGY-7703; mouse xenograft subcutaneous model | Han et al. [81] |
| Survivin | PEG/PEI conjugated with RGD * | Subcutaneous mouse model | Wu et al. [82] |
| Target mRNA | Delivery System | Tumor Model | Ref. |
|---|---|---|---|
| CD73 * | Chitosan linked to TAT *-Hyaluronic acid | CT26; subcutaneous xenograft mice model | Khesth et al. [94] |
| CPTA1 * | Exosome linked to iRGD * | HCT116; xenograft subcutaneous mouse model | Lin et al. [95] |
| PD-1 * | Attenuated Salmonella | Mouse xenograft subcutaneous model | Lu et al. [96] |
| CD47 * | PLGA * | Xenograft mouse model | Zhang et al. [97] |
| CD44 * | Liposome | HCT116-CSC; xenograft subcutaneous mouse model | Zou et al. [98] |
| ATP7A * | PEG-PLGA linked to a cationic lipid | HCT116 and LOVO; subcutaneous xenograft mouse model | Zhou et al. [99] |
| Target mRNA/Delivery System | Clinical Trial/Results | Disease | Number |
|---|---|---|---|
| PKN3 */liposome | Phase 1/well tolerated | Colorectal cancer | NCT00938574 |
| RRM2 */polymer | Phase 1a/well tolerated | Unresectable solid tumors | NCT00689065 |
| HSP47 */lipid | Phase I | Liver fibrosis | NCT02227459 |
| PLK1 */lipid | Phase I | Unresectable colorectal, pancreas, gastric, breast, ovarian and esophageal cancers with hepatic metastases | NCT01437007 |
| KRASG12D */ | Phase I/IIa Phase II | Pancreatic carcinoma | NCT01188785 NCT01676259 |
| PKN3 */polymer | Phase Ib/IIa | metastatic pancreatic adenocarcinoma | NCT018086389 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Losurdo, P.; de Manzini, N.; Palmisano, S.; Grassi, M.; Parisi, S.; Rizzolio, F.; Tierno, D.; Biasin, A.; Grassi, C.; Truong, N.H.; et al. Potential Application of Small Interfering RNA in Gastro-Intestinal Tumors. Pharmaceuticals 2022, 15, 1295. https://doi.org/10.3390/ph15101295
Losurdo P, de Manzini N, Palmisano S, Grassi M, Parisi S, Rizzolio F, Tierno D, Biasin A, Grassi C, Truong NH, et al. Potential Application of Small Interfering RNA in Gastro-Intestinal Tumors. Pharmaceuticals. 2022; 15(10):1295. https://doi.org/10.3390/ph15101295
Chicago/Turabian StyleLosurdo, Pasquale, Nicolò de Manzini, Silvia Palmisano, Mario Grassi, Salvatore Parisi, Flavio Rizzolio, Domenico Tierno, Alice Biasin, Chiara Grassi, Nhung Hai Truong, and et al. 2022. "Potential Application of Small Interfering RNA in Gastro-Intestinal Tumors" Pharmaceuticals 15, no. 10: 1295. https://doi.org/10.3390/ph15101295
APA StyleLosurdo, P., de Manzini, N., Palmisano, S., Grassi, M., Parisi, S., Rizzolio, F., Tierno, D., Biasin, A., Grassi, C., Truong, N. H., & Grassi, G. (2022). Potential Application of Small Interfering RNA in Gastro-Intestinal Tumors. Pharmaceuticals, 15(10), 1295. https://doi.org/10.3390/ph15101295








