Association between WHO First-Step Analgesic Use and Risk of Breast Cancer in Women of Working Age
Abstract
:1. Introduction
2. Results
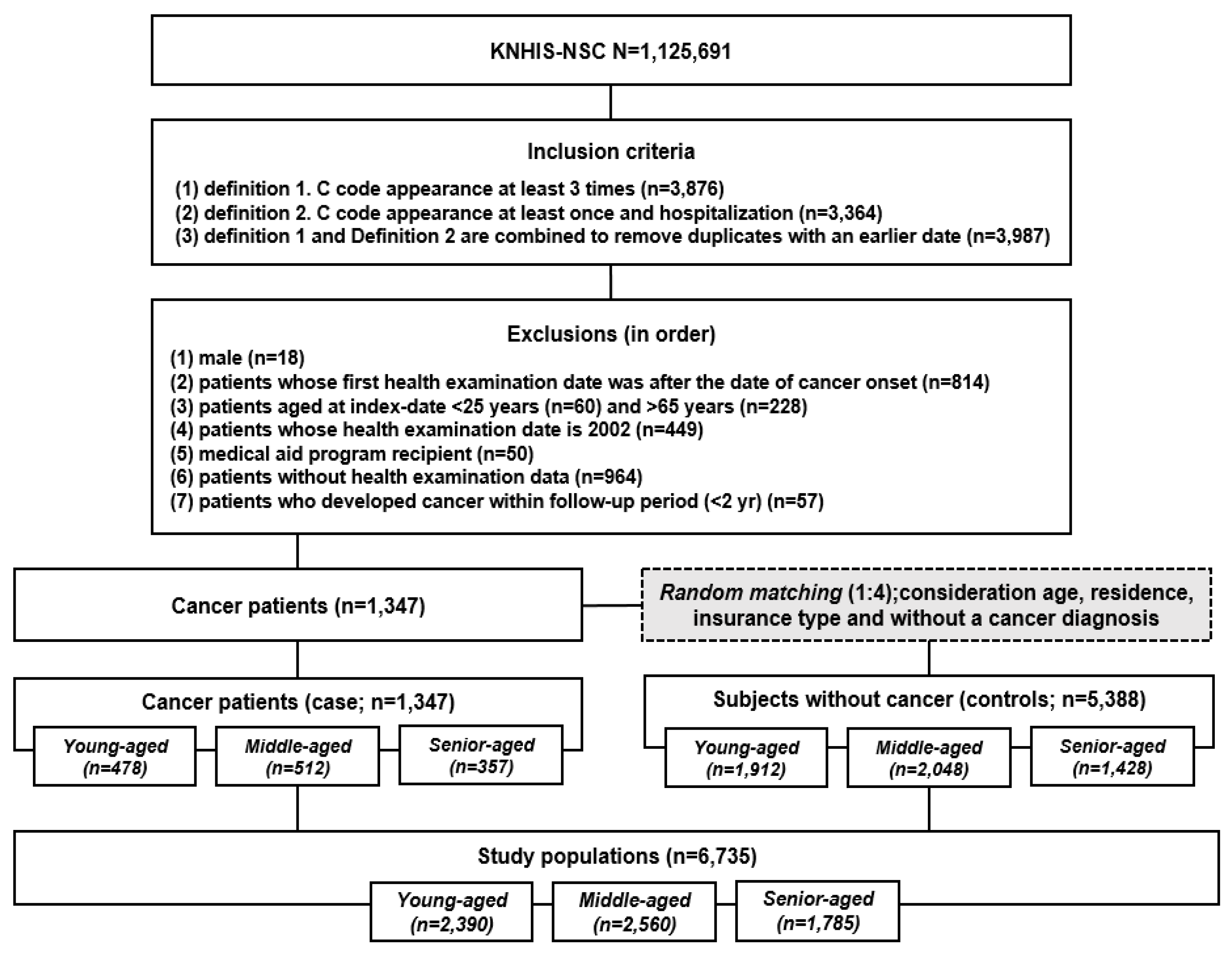
2.1. Participant Characteristics
2.2. Risk Factors Associated with Cancer Development
2.2.1. Hazard Ratios (HRs) Associated with Cancer Development
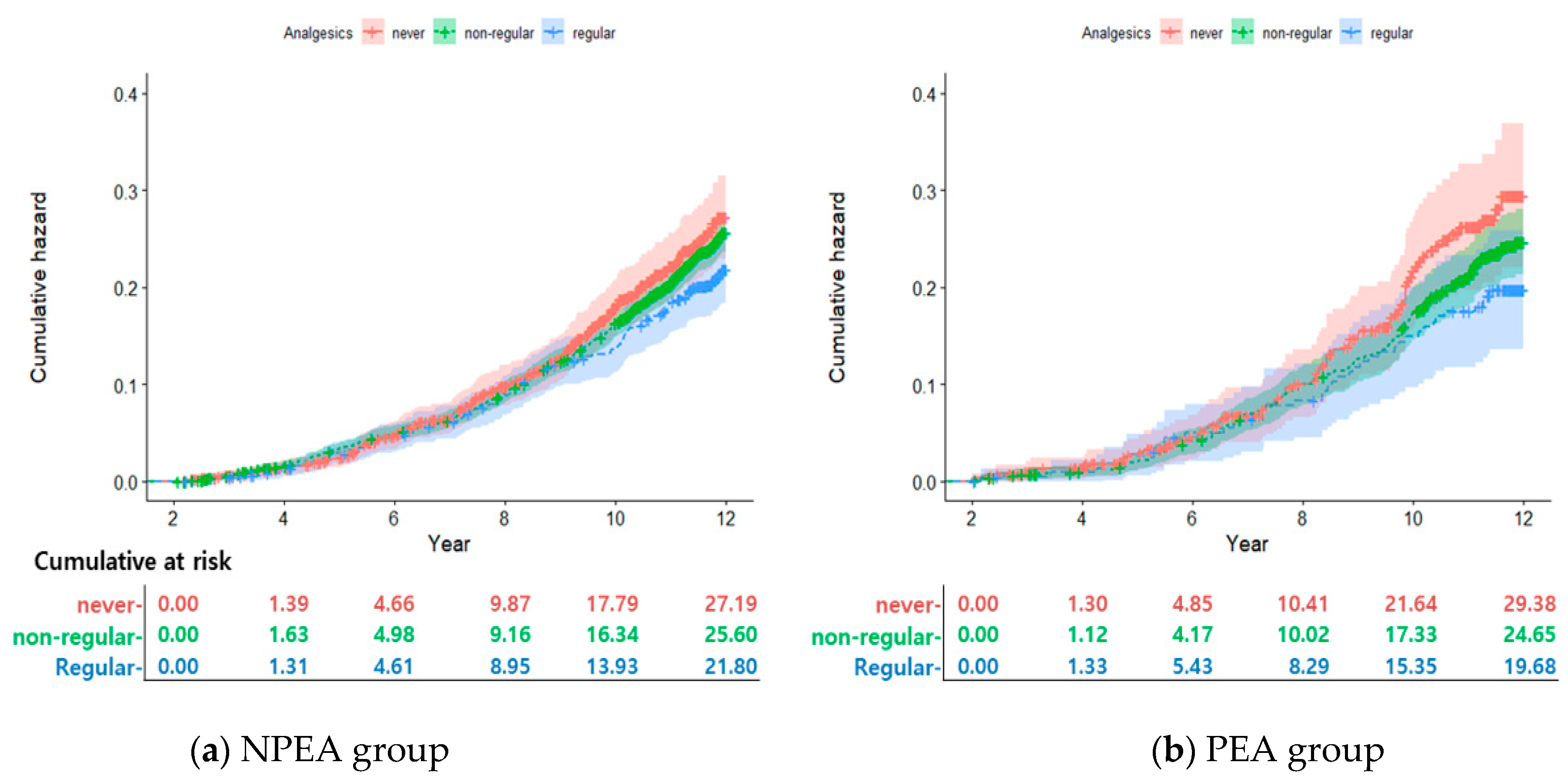
2.2.2. Cumulative Incidence According to Analgesic Use
2.3. Association between Cancer and Analgesic Use by Working-Age
3. Discussion
4. Materials and Methods
4.1. Study Design and Data Collection
4.2. Criteria and Definitions
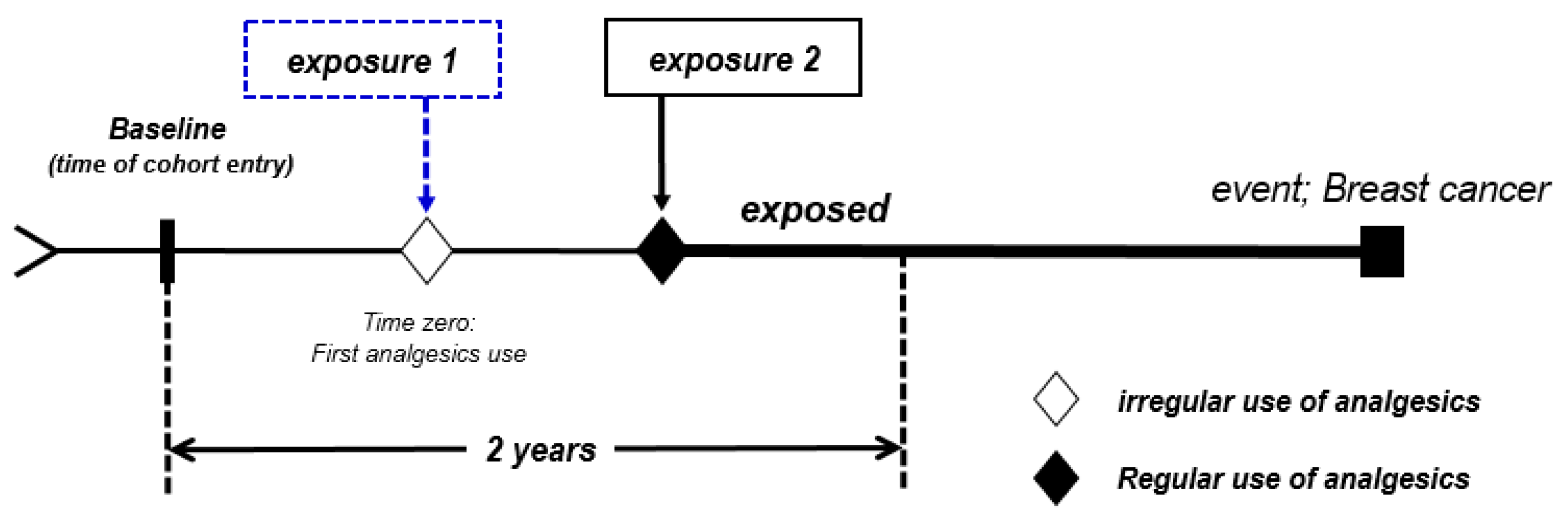
4.3. Assessment of First-Step Analgesic Use
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bank of Korea. Background and Implications of the Increase in Women’s Economic Activity in Major Countries. 2018. Available online: https://www.bok.or.kr/portal/bbs/P0000528/view.do?nttId=236370&menuNo=200434 (accessed on 8 November 2022).
- National Cancer Information Center. Annual Report of Cancer Statistics in Korea in 2016. 2018. Available online: https://www.cancer.go.kr/lay1/bbs/S1T674C680/B/26/view.do?article_seq=21129 (accessed on 8 November 2022).
- National Cancer Institute. Cancer Stat Facts: Female Breast Cancer. 2019. Available online: https://seer.cancer.gov/statfacts/html/breast.html (accessed on 8 November 2022).
- Korean Breast Cancer Society. Breast cancer facts & figures 2018. Korean Breast Cancer Soc. 2018, 3–9. [Google Scholar]
- Seo, J.S.; Park, H.A.; Kang, J.H.; Kim, K.W.; Cho, Y.G.; Hur, Y.I.; Park, Y.R. Obesity and obesity-related lifestyles of Korean breast cancer survivors. Korean J. Health Promot. 2015, 14, 93–102. [Google Scholar] [CrossRef] [Green Version]
- Yoo, K.Y.; Shin, H.R.; Park, S.K.; Yoon, H.J.; Shin, A.S.; Kang, D.H.; Noh, D.Y.; Choe, K.J. Is breast cancer incidence rate further increasing in Korea? Korean J. Epidemiol. 2001, 23, 1–7. [Google Scholar]
- Statistics Korea. A Woman’s Life in Statistics 2018. 2018. Available online: https://www.kostat.go.kr/portal/korea/kor_nw/1/6/1/index.board?bmode=read&aSeq=368636 (accessed on 8 November 2022).
- Duijts, S.F.; Zeegers, M.P.; Borne, B.V. The association between stressful life events and breast cancer risk: A meta-analysis. Int. J. Cancer 2003, 107, 1023–1029. [Google Scholar] [CrossRef]
- Kim, H.S.; Kwon, M.; Lee, J. Job Stress and mental health of female household head workers. Korean J. Occup. Health Nurs. 2016, 25, 249–258. [Google Scholar] [CrossRef] [Green Version]
- Chang, S.J.; Koh, S.B.; Kang, M.G.; Cha, B.S.; Park, J.K.; Hyun, S.J.; Park, J.H.; Kim, S.A.; Kang, D.M.; Chang, S.S.; et al. Epidemiology of psychosocial distress in Korea employees. J. Prev. Med. Public Health 2005, 38, 25–37. [Google Scholar] [PubMed]
- Lee, S.I.; Kim, S.G.; Seo, M.G. Analysis of relationship between stress and demographic characteristic: From the 2015 Korea national health and nutrition examination survey. J. Korean Off. Stat. 2018, 23, 1–13. [Google Scholar]
- Yu, E.J.; Choi, S.O.; Kang, Y.S. Study of working women’s psychosocial stress. J. Korean Data Anal. Soc. 2006, 8, 1793–1806. [Google Scholar]
- Jun, E.M.; Cho, D.S. The relationship of symptoms of stress, cancer prevention behavior and the quality of life in women. Korean J. Women Health Nurs. 2005, 11, 156–162. [Google Scholar] [CrossRef] [Green Version]
- Lim, S.A. Status of health and health-related risk factors of Korean women; from the perspective of Korean working women. J. Kyung Hee Univ. Med. Cent. 2014, 29, 27–31. [Google Scholar]
- Bittoni, M.A.; Carbone, D.P.; Harris, R.E. Ibuprofen and fatal lung cancer: A brief report of the prospective results from the Third National Health and Nutrition Examination Survey (NHANES III). Mol. Clin. Oncol. 2017, 6, 917–920. [Google Scholar] [CrossRef] [Green Version]
- Baris, D.; Karagas, M.R.; Koutros, S.; Colt, J.S.; Johnson, A.; Schwenn, M.; Fischer, A.H.; Figueroa, J.D.; Berndt, S.I.; Han, S.; et al. Nonsteroidal anti-inflammatory drugs and other analgesic use and bladder cancer in northern New England. Int. J. Cancer 2013, 132, 162–173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ulrich, C.M.; Bigler, J.; Potter, J.D. Non-steroidal anti-inflammatory drugs for cancer prevention: Promise, perils and pharmacogenetics. Nat. Rev. Cancer 2006, 6, 130–410. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.S. Analysis of changes in economic activity of married women and implications. KERI Insight 2021. [Google Scholar]
- Shacter, E.; Weitzman, S.A. Chronic inflammation and cancer. Oncology 2002, 16, 217–226. [Google Scholar]
- Yoo, Y.G.; Choi, S.K.; Hwang, S.J.; Lim, H.S. Risk factors of breast cancer according to life style. J. Korea Contents Assoc. 2013, 13, 262–272. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.S.; Zhu, S.; Li, X.W.; Wang, F.; Hu, F.L.; Li, D.D.; Zhang, W.C.; Li, X. Association between NSAIDs use and breast cancer risk: A systematic review and meta-analysis. Breast Cancer Res. Treat 2009, 117, 141–150. [Google Scholar] [CrossRef]
- Jacobs, E.J.; Newton, C.C.; Stevens, V.L.; Gapstur, S.M. A large cohort study of long-term acetaminophen use and prostate cancer incidence. Cancer Epidemiol. Biomark. Prev. 2011, 20, 1322–1328. [Google Scholar] [CrossRef] [Green Version]
- Khuder, S.A.; Mutgi, A.B. Breast cancer and NSAID use: A meta-analysis. Br. J. Cancer 2001, 84, 1188–1192. [Google Scholar] [CrossRef] [Green Version]
- Agrawal, A.; Fentiman, I.S. NSAIDs and breast cancer: A possible prevention and treatment strategy. Int. J. Clin. Pract. 2008, 62, 444–449. [Google Scholar] [CrossRef]
- De Pedro, M.; Baeza, S.; Escudero, M.T.; Dierssen-Sotos, T.; Gómez-Acebo, I.; Pollán, M.; Llorca, J. Effect of COX-2 inhibitors and other non-steroidal inflammatory drugs on breast cancer risk: A meta-analysis. Breast Cancer Res. Treat 2015, 149, 525–536. [Google Scholar] [CrossRef] [PubMed]
- Egan, K.M.; Stampfer, M.J.; Giovannucci, E.; Rosner, B.A.; Colditz, G.A. Prospective study of regular aspirin use and the risk of breast cancer. J. Natl. Cancer Inst. 1996, 88, 988–993. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hwangbo, Y.; Kang, D.; Kang, M.; Kim, S.; Lee, E.K.; Kim, Y.A.; Chang, Y.J.; Choi, K.S.; Jung, S.Y.; Woo, S.M.; et al. Incidence of diabetes after cancer develo.pment: A Korean national cohort study. JAMA Oncol. 2018, 4, 1099–1105. [Google Scholar] [CrossRef] [PubMed]
- Larsson, S.C.; Mantzoros, C.S.; Wolk, A. Diabetes mellitus and risk of breast cancer: A meta-analysis. Int. J. Cancer 2007, 121, 856–862. [Google Scholar] [CrossRef] [PubMed]
- Wong, F.Y.; Tham, W.Y.; Nei, W.L.; Lim, C.; Miao, H. Age exerts a continuous effect in the outcomes of Asian breast cancer patients treated with breast-conserving therapy. Cancer Commun. 2018, 38, 39. [Google Scholar] [CrossRef] [Green Version]
- Winchester, D.P.; Osteen, R.T.; Menck, H.R. The national cancer data base report on breast carcinoma characteristics and outcome in relation to age. Cancer 1996, 78, 1838–1843. [Google Scholar] [CrossRef]
- McNeil, J.J.; Gibbs, P.; Orchard, S.G.; Lockery, J.E.; Bernstein, W.B.; Cao, Y.; Ford, L.; Haydon, A.; Kirpach, B.; Macrae, F.; et al. Effect of aspirin on cancer incidence and mortality in older adults. J. Natl. Cancer Inst. 2021, 113, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Loomans-Kropp, H.A.; Pinsky, P.; Cao, Y.; Chan, A.T.; Umar, A. Association of aspirin use with mortality risk among older adult participants in the prostate, lung, colorectal, and ovarian cancer screening trial. JAMA Netw. Open 2019, 2, e1916729. [Google Scholar] [CrossRef] [Green Version]
- Jin, J. Use of aspirin to prevent cardiovascular disease. JAMA 2022, 327, 1624. [Google Scholar] [CrossRef]
- U.S. Preventive Services TASK FORCE. Preventive Services TASK FORCE. Aspirin Use to Prevent Cardiovascular Disease: Preventive Medication. 2022. Available online: https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/aspirin-to-prevent-cardiovascular-disease-preventive-medication (accessed on 8 November 2022).
- Gaudet, M.M.; Gapstur, S.M.; Sun, J.; Diver, W.R.; Hannan, L.M.; Thun, M.J. Active smoking and breast cancer risk: Original cohort data and meta-analysis. J. Natl. Cancer Inst. 2013, 105, 515–525. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.; Lee, J.S.; Park, S.H.; Shin, S.A.; Kim, K.W. Cohort profile: The National Health Insurance Service-national sample cohort (NHIS-NSC), South Korea. Int. J. Epidemiol. 2017, 46, e15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ernster, V.L. Nested case-control studies. Prev. Med. 1994, 23, 587–590. [Google Scholar] [CrossRef] [PubMed]
- Yaghjyan, L.; Eliassen, A.H.; Colditz, G.; Rosner, B.; Schedin, P.; Wijayabahu, A.; Tamimi, R.M. Associations of aspirin and other anti-inflammatory medications with breast cancer risk by the status of COX-2 expression. Breast Cancer Res. 2022, 24, 89. [Google Scholar] [CrossRef] [PubMed]
- Ballotari, P.; Vicentini, M.; Manicardi, V.; Gallo, M.; Ranieri, S.C.; Greci, M.; Rossi, P.G. Diabetes and risk of cancer incidence: Results from a population-based cohort study in northern Italy. BMC Cancer 2017, 17, 703. [Google Scholar] [CrossRef] [PubMed]
- National Health Insurance Sharing Service. Sample Cohort DB Manual. 2021. Available online: https://nhiss.nhis.or.kr/bd/ab/bdaba002cv.do# (accessed on 8 November 2022).
- Wan Ahmad, W.N.; Sakri, F.; Mokhsin, A.; Rahman, T.; Mohd Nasir, N.; Abdul-Razak, S.; Md Yasin, M.; Mohd Ismail, A.; Ismail, Z.; Nawawi, H. Low serum high density lipoprotein cholesterol concentration is an independent predictor for enhanced inflammation and endothelial activation. PLoS ONE 2015, 10, e0116867. [Google Scholar] [CrossRef] [Green Version]
- Jo, A.; Oh, H. Related to cigarette smoking and alcohol consumption in adults with metabolic syndrome: Prospective cohort study. J. Korean Acad. Nurs. 2019, 49, 713–723. [Google Scholar] [CrossRef]
- Charlson, M.E.; Pompei, P.; Ales, K.L.; MacKenzie, C.R. A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. J. Chronic Dis. 1987, 40, 373–383. [Google Scholar] [CrossRef]
- Sundararajan, V.; Henderson, T.; Perry, C.; Muggivan, A.; Quan, H.; Ghali, W.A. New ICD-10 version of the Charlson comorbidity index predicted in-hospital mortality. J. Clin. Epidemiol. 2004, 57, 1288–12894. [Google Scholar] [CrossRef]
- Hu, K.; Sjölander, A.; Lu, D.; Walker, A.K.; Sloan, E.K.; Fall, K.; Valdimarsdóttir, U.; Hall, P.; Smedby, K.E.; Fang, F. Aspirin and other non-steroidal anti-inflammatory drugs and depression, anxiety, and stress-related disorders following a cancer diagnosis: A nationwide register-based cohort study. BMC Med. 2020, 18, 238. [Google Scholar] [CrossRef]
- Chen, S.W.; Tsan, Y.T.; Chen, J.D.; Hsieh, H.I.; Lee, C.H.; Lin, H.H.; Wang, J.D.; Chen, P.C.; Health Data Analysis in Taiwan Research Group. Use of thiazolidinediones and the risk of colorectal cancer in patients with diabetes: A nationwide, population-based, case-control study. Diabetes Care 2013, 36, 369–375. [Google Scholar] [CrossRef] [Green Version]
- Anekar, A.A.; Cascella, M. WHO Analgesic Ladder. 2021. Available online: https://www.ncbi.nlm.nih.gov/books/NBK554435/ (accessed on 8 November 2022).
- Ruder, E.H.; Laiyemo, A.O.; Graubard, B.I.; Hollenbeck, A.R.; Schatzkin, A.; Cross, A.J. Non-steroidal anti-inflammatory drugs and colorectal cancer risk in a large, prospective cohort. Am. J. Gastroenterol. 2011, 106, 1340–1350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Variables | Descriptions | Young-Aged (25~39) | Middle-Aged (40~49) | Senior-Aged (50~64) | x2 | p | |||
|---|---|---|---|---|---|---|---|---|---|
| N | % | N | % | N | % | ||||
| Insurance type | |||||||||
| Members (a) | 950 | 39.7 | 1125 | 43.9 | 500 | 28.0 | 475.50 | 0.000 | |
| Dependents (b) | 815 | 34.1 | 825 | 32.2 | 940 | 52.7 | |||
| Self-employed | 220 | 9.2 | 400 | 15.6 | 295 | 16.5 | |||
| Employee | 405 | 16.9 | 210 | 8.2 | 50 | 2.8 | |||
| Income | |||||||||
| Low (1~3) | 695 | 29.1 | 817 | 31.9 | 611 | 34.2 | 49.85 | 0.000 | |
| Middle (4~6) | 875 | 36.6 | 743 | 29.0 | 495 | 27.7 | |||
| High (7~10) | 820 | 34.3 | 1000 | 39.1 | 679 | 38.0 | |||
| Body mass index (BMI) | |||||||||
| <25 | 1820 | 76.2 | 1756 | 68.6 | 1026 | 57.5 | 164.75 | 0.000 | |
| ≥25 | 570 | 23.8 | 804 | 31.4 | 759 | 42.5 | |||
| Total cholesterol | |||||||||
| <240 | 2225 | 93.1 | 2220 | 86.7 | 1409 | 78.9 | 178.49 | 0.000 | |
| ≥240 | 165 | 6.9 | 360 | 14.1 | 376 | 21.1 | |||
| Alcohol intake | |||||||||
| Never | 1531 | 64.1 | 1855 | 72.5 | 1547 | 86.7 | 267.80 | 0.000 | |
| Ever | 859 | 35.9 | 705 | 27.5 | 238 | 13.3 | |||
| Smoking | |||||||||
| Never | 2161 | 90.4 | 2347 | 91.7 | 1686 | 94.5 | 22.99 | 0.000 | |
| Ever | 229 | 9.6 | 213 | 8.3 | 99 | 5.5 | |||
| Exercise | |||||||||
| Never | 1011 | 42.3 | 1242 | 48.5 | 945 | 52.9 | 48.15 | 0.000 | |
| Ever | 1379 | 57.7 | 1318 | 51.5 | 840 | 47.1 | |||
| Charlson Comorbidity Index (CCI) | |||||||||
| 0 | 1515 | 63.4 | 1392 | 54.4 | 639 | 35.8 | 517.46 | 0.000 | |
| 1 | 573 | 24.0 | 641 | 25.0 | 468 | 26.2 | |||
| 2 | 208 | 8.7 | 301 | 11.8 | 293 | 16.4 | |||
| ≥3 | 94 | 3.9 | 226 | 8.8 | 385 | 21.6 | |||
| Analgesics use | |||||||||
| Never | 536 | 22.4 | 600 | 23.4 | 299 | 16.8 | 201.95 | 0.000 | |
| Non-regular | 1572 | 65.8 | 1630 | 63.7 | 1013 | 56.8 | |||
| Regular | 282 | 11.8 | 330 | 12.9 | 473 | 26.5 | |||
| Variables | HR (a) | 95% CI (b) | p | |
|---|---|---|---|---|
| Economic activity | ||||
| Not participating | 1.000 | |||
| Participating | 1.542 | 1.345–1.768 | 0.000 *** | |
| Income | ||||
| Low (1~3) | 1.000 | |||
| Middle (4~6) | 1.673 | 1.419–1.973 | 0.000 *** | |
| High (7~10) | 3.089 | 2.647–3.605 | 0.000 *** | |
| Body mass index (BMI) | 0.990 | 0.975–1.006 | 0.478 | |
| Total cholesterol | 0.998 | 0.997–0.999 | 0.007 ** | |
| Alcohol intake | ||||
| Never | 1.000 | |||
| Ever | 0.967 | 0.853–1.096 | 0.600 ** | |
| Smoking | ||||
| Never | 1.000 | |||
| Ever | 0.638 | 0.497–0.819 | 0.000 *** | |
| Exercise | ||||
| Never | 1.000 | |||
| Ever | 0.836 | 0.751–0.931 | 0.001 ** | |
| Charlson Comorbidity Index (CCI) | ||||
| 0 | 1.000 | |||
| 1 | 0.993 | 0.870–1.134 | 0.919 | |
| 2 | 1.123 | 0.946–1.343 | 0.186 | |
| ≥3 | 1.113 | 0.921–1.345 | 0.268 | |
| First-step analgesics use | ||||
| Never | 1.000 | |||
| Non-regular | 0.905 | 0.785–1.043 | 0.168 | |
| Regular | 0.748 | 0.614–0.912 | 0.004 ** | |
| Model 1 (a) | Young-Aged (25~39) | Middle-Aged (40~49) | Senior-Aged (50~64) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| HR (b) | 95% CI (c) | p | HR | 95% CI | p | HR | 95% CI | p | ||
| Economic activity | ||||||||||
| Not-participating | 1.000 | 1.000 | 1.000 | |||||||
| Participating | 1.482 | 1.185–1.854 | 0.001 ** | 1.700 | 1.361–2.124 | 0.000 *** | 1.386 | 1.049–1.833 | 0.022 ** | |
| Income | ||||||||||
| Low (1~3) | 1.000 | 1.000 | 1.000 | |||||||
| Middle (4~6) | 1.731 | 1.314–2.281 | 0.000 *** | 1.763 | 1.334–2.330 | 0.000 *** | 1.556 | 1.142–2.120 | 0.005 ** | |
| High (7~10) | 3.161 | 2.415–4.138 | 0.000 *** | 3.504 | 2.712–4.529 | 0.000 *** | 2.503 | 1.895–3.306 | 0.000 *** | |
| Charlson Comorbidity Index (CCI) | ||||||||||
| 0 | 1.000 | 1.000 | 1.000 | |||||||
| 1 | 1.234 | 0.994–1.534 | 0.057 | 0.969 | 0.785–1.196 | 0.767 | 0.730 | 0.551–0.968 | 0.029 ** | |
| 2 | 1.555 | 1.149–2.105 | 0.005 ** | 0.815 | 0.603–1.100 | 0.181 | 1.000 | 0.739–1.354 | 0.999 | |
| ≥3 | 2.095 | 1.422–3.086 | 0.000 *** | 0.976 | 0.703–1.356 | 0.886 | 0.823 | 0.606–1.118 | 0.213 | |
| First-step analgesics use | ||||||||||
| Never | 1.000 | 1.000 | 1.000 | |||||||
| Non-regular | 0.915 | 0.723–1.157 | 0.458 | 0.912 | 0.728–1.142 | 0.423 | 0.912 | 0.678–1.227 | 0.542 | |
| Regular | 0.611 | 0.427–0.875 | 0.007 ** | 0.929 | 0.670–1.288 | 0.659 | 0.741 | 0.517–1.061 | 0.100 * | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oh, H.S.; Seo, H.J. Association between WHO First-Step Analgesic Use and Risk of Breast Cancer in Women of Working Age. Pharmaceuticals 2023, 16, 323. https://doi.org/10.3390/ph16020323
Oh HS, Seo HJ. Association between WHO First-Step Analgesic Use and Risk of Breast Cancer in Women of Working Age. Pharmaceuticals. 2023; 16(2):323. https://doi.org/10.3390/ph16020323
Chicago/Turabian StyleOh, Hyun Sook, and Hwa Jeong Seo. 2023. "Association between WHO First-Step Analgesic Use and Risk of Breast Cancer in Women of Working Age" Pharmaceuticals 16, no. 2: 323. https://doi.org/10.3390/ph16020323
APA StyleOh, H. S., & Seo, H. J. (2023). Association between WHO First-Step Analgesic Use and Risk of Breast Cancer in Women of Working Age. Pharmaceuticals, 16(2), 323. https://doi.org/10.3390/ph16020323






