Covalent Conjugates of Allylbenzenes and Terpenoids as Antibiotics Enhancers with the Function of Prolonged Action
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis and Characterization of Covalent and Non-Covalent Antibacterial Formulations
- (1)
- Synthesis and characterization by FTIR and NMR spectroscopy of the covalent conjugates of MF, allylbenzenes (EG and apiol), terpenoids (linalool and limonene) with polymers (based on PEI, chitosan, mannan), with a variable type of covalent linkage (amide or ester bond) and with a variable crosslinking agent, to determine the most efficient and optimal composition of the antibacterial conjugate.
- (2)
- Incorporation of the drug composition into polymer nanogels followed by covalent crosslinking of the particles, without involving the drug in covalent bonds.
- (3)
- Investigation of the antibacterial activity (against E. coli and B. subtilis) of the developed prodrugs in terms of efficiency and duration of their action.
- (4)
- Elucidation of the mechanisms of the conjugate action in cells using FTIR spectroscopy in comparison with free antibacterials.
- (5)
- Study of the pharmacokinetics of the antibacterial covalent prodrugs in comparison with the antibacterial formulations included in nanogels (non-covalent prodrugs).
2.1.1. MF Covalent Prodrug Formulations Synthesis and Characterization by FTIR and NMR Spectroscopy
2.1.2. Allylbenzenes and Terpenoids Formulations
2.1.3. Non-Covalent Formulations of MF, Allylbenzenes and Terpenoids with Glycol–Chitosan Hydrogels Stabilized by Crosslinking Bifunctional Agents
2.2. Antibacterial Activity of MF, Allylbenzenes, Terpenoids
2.2.1. Primary High-Throughput Screening of the Antibacterial Activity of Prodrug Conjugates
2.2.2. Prolonged Antibacterial Experiment with the Most Promising Formulations
2.3. FTIR Spectroscopy as a Tool for Tracking Drug Penetration into Cells and Its Effectiveness
2.4. FTIR Spectroscopy as a Tool for Determining the Number of Cells in Antibacterial Experiments
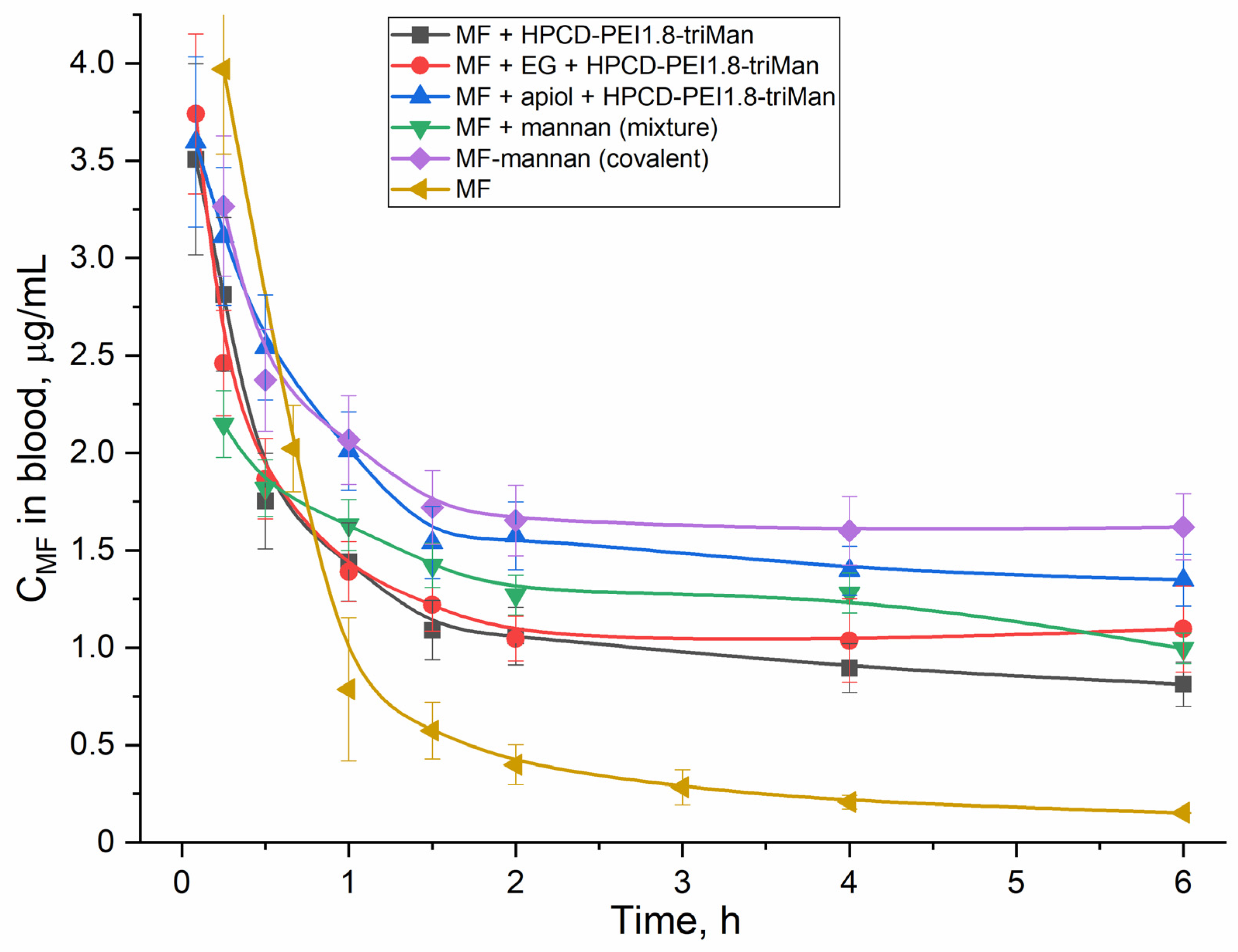
2.5. Pharmacokinetics of MF in Polymer Particles and Covalent Conjugates
3. Materials and Methods
3.1. Reagents
3.2. HPCD–PEI1.8–triMan Polymer Synthesis and Properties
3.3. MF Conjugation with Polymers
3.4. Preparation of MF Non-Concovalent Formulations
3.5. Adjuvant Conjugation with Polymers
3.6. FTIR Spectroscopy
3.7. NMR Spectroscopy
3.8. Antibacterial Activity Studies: FTIR Spectroscopy, Microbiology
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| EG | eugenol |
| GlycChit | glycol chitosan |
| HPCD | (2-hydroxypropyl)-β-cyclodextrin |
| IC50 | Half-maximal inhibitory concentration |
| MF | moxifloxacin |
| PEI | polyethyleneimine |
| triMan | mannotriose residue |
References
- Carvalho, M.F.; Maia, A.S.; Tiritan, M.E.; Castro, P.M.L. Bacterial Degradation of Moxifloxacin in the Presence of Acetate as a Bulk Substrate. J. Environ. Manag. 2016, 168, 219–228. [Google Scholar] [CrossRef]
- Tejashri, G.; Amrita, B.; Darshana, J. Cyclodextrin Based Nanosponges for Pharmaceutical Use: A Review. Acta Pharm. 2013, 63, 335–358. [Google Scholar] [CrossRef] [PubMed]
- Pereira, P.; Morgado, D.; Crepet, A.; David, L.; Gama, F.M. Glycol Chitosan-Based Nanogel as a Potential Targetable Carrier for SiRNA. Macromol. Biosci. 2013, 13, 1369–1378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohamed, S.H.; Arafa, A.S.; Mady, W.H.; Fahmy, H.A.; Omer, L.M.; Morsi, R.E. Preparation and Immunological Evaluation of Inactivated Avian Influenza Virus Vaccine Encapsulated in Chitosan Nanoparticles. Biologicals 2018, 51, 46–53. [Google Scholar] [CrossRef]
- Le-Deygen, I.M.; Skuredina, A.A.; Kudryashova, E.V. Drug Delivery Systems for Fluoroquinolones: New Prospects in Tuberculosis Treatment. Russ. J. Bioorganic Chem. 2017, 43, 487–501. [Google Scholar] [CrossRef]
- Zlotnikov, I.D.; Kudryashova, E.V. Spectroscopy Approach for Highly-Efficient Screening of Lectin-Ligand Interactions in Application for Mannose Receptor and Molecular Containers for Antibacterial Drugs. Pharmaceuticals 2022, 15, 625. [Google Scholar] [CrossRef] [PubMed]
- Ghotbi, Z.; Haddadi, A.; Hamdy, S.; Hung, R.W.; Samuel, J.; Lavasanifar, A. Active Targeting of Dendritic Cells with Mannan-Decorated PLGA Nanoparticles. J. Drug Target. 2011, 19, 281–292. [Google Scholar] [CrossRef]
- Ran, S.; Volk, L.; Hall, K.; Flister, M.J. Lymphangiogenesis and Lymphatic Metastasis in Breast Cancer. Pathophysiology 2010, 17, 229–251. [Google Scholar] [CrossRef] [Green Version]
- Hoffmann, S.; Gorzelanny, C.; Moerschbacher, B.; Goycoolea, F.M. Physicochemical Characterization of FRET-Labelled Chitosan Nanocapsules and Model Degradation Studies. Nanomaterials 2018, 8, 846. [Google Scholar] [CrossRef] [Green Version]
- Gheran, C.V.; Voicu, S.N.; Galateanu, B.; Callewaert, M.; Moreau, J.; Cadiou, C.; Chuburu, F.; Dinischiotu, A. In Vitro Studies Regarding the Safety of Chitosan and Hyaluronic Acid-Based Nanohydrogels Containing Contrast Agents for Magnetic Resonance Imaging. Int. J. Mol. Sci. 2022, 23, 3258. [Google Scholar] [CrossRef]
- Gordon, S. The Role of the Macrophage in Immune Regulation. Res. Immunol. 1998, 149, 685–688. [Google Scholar] [CrossRef]
- Paczkowska-Walendowska, M.; Cielecka-Piontek, J. Chitosan as a Functional Carrier for the Local Delivery Anti-Inflammatory Systems Containing Scutellariae Baicalensis Radix Extract. Pharmaceutics 2022, 14, 2148. [Google Scholar] [CrossRef]
- Chen, P.; Zhang, X.; Jia, L.; Prud’Homme, R.K.; Szekely, Z.; Sinko, P.J. Optimal structural design of mannosylated nanocarriers for macrophage targeting. J. Control. Release 2014, 194, 341–349. [Google Scholar] [CrossRef] [Green Version]
- Di Benedetto, P.; Ruscitti, P.; Vadasz, Z.; Toubi, E.; Giacomelli, R. Macrophages with Regulatory Functions, a Possible New Therapeutic Perspective in Autoimmune Diseases. Autoimmun. Rev. 2019, 18, 102369. [Google Scholar] [CrossRef]
- Serra, P.; Santamaria, P. Nanoparticle-Based Autoimmune Disease Therapy. Clin. Immunol. 2015, 160, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Sato, A.; Kawakami, S.; Yamada, M.; Yamashita, F.; Hashida, M. Enhanced Gene Transfection in Macrophages Using Mannosylated Cationic Liposome-Polyethylenimine-Plasmid DNA Complexes. J. Drug Target. 2001, 9, 201–207. [Google Scholar] [CrossRef]
- Le-Deygen, I.M.; Mamaeva, P.V.; Skuredina, A.A.; Kudryashova, E.V. A Spectral Approach to Study Interaction between Chitosan Modified with Mannose and Concavalin A for the Creation of Address Delivery Systems of Antituberculosis Drugs. Mosc. Univ. Chem. Bull. 2020, 75, 213–217. [Google Scholar] [CrossRef]
- Alizadeh, D.; Zhang, L.; Hwang, J.; Schluep, T.; Badie, B. Tumor-Associated Macrophages Are Predominant Carriers of Cyclodextrin-Based Nanoparticles into Gliomas. Nanomed. Nanotechnol. Biol. Med. 2010, 6, 382–390. [Google Scholar] [CrossRef] [Green Version]
- Zlotnikov, I.D.; Vigovskiy, M.A.; Davydova, M.P.; Danilov, M.R.; Dyachkova, U.D.; Grigorieva, O.A.; Kudryashova, E.V. Mannosylated Systems for Targeted Delivery of Antibacterial Drugs to Activated Macrophages. Int. J. Mol. Sci. 2022, 23, 6144. [Google Scholar] [CrossRef] [PubMed]
- Raviv, L.; Jaron-Mendelson, M.; David, A. Mannosylated Polyion Complexes for in Vivo Gene Delivery into CD11c+ Dendritic Cells. Mol. Pharm. 2015, 12, 453–462. [Google Scholar] [CrossRef]
- Nahar, M.; Dubey, V.; Mishra, D.; Mishra, P.K.; Dube, A.; Jain, N.K. In Vitro Evaluation of Surface Functionalized Gelatin Nanoparticles for Macrophage Targeting in the Therapy of Visceral Leishmaniasis. J. Drug Target. 2010, 18, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.; Wang, X.; Nie, S.; Chen, Z.; Shin, D.M. Therapeutic Nanoparticles for Drug Delivery in Cancer. Clin. Cancer Res. 2008, 14, 1310–1316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amiji, M.M.; Kalariya, M.; Jain, S.; Attarwala, H. U.S. Patent No. 10,500,156, 2019.
- Piotrowski-Daspit, A.S.; Kauffman, A.C.; Bracaglia, L.G.; Saltzman, W.M. Polymeric Vehicles for Nucleic Acid Delivery. Adv. Drug Deliv. Rev. 2020, 156, 119–132. [Google Scholar] [CrossRef]
- Gao, J.Q.; Zhao, Q.Q.; Lv, T.F.; Shuai, W.P.; Zhou, J.; Tang, G.P.; Liang, W.Q.; Tabata, Y.; Hu, Y.L. Gene-Carried Chitosan-Linked-PEI Induced High Gene Transfection Efficiency with Low Toxicity and Significant Tumor-Suppressive Activity. Int. J. Pharm. 2010, 387, 286–294. [Google Scholar] [CrossRef]
- Qindeel, M.; Ahmed, N.; Khan, G.M.; Rehman, A. Ligand Decorated Chitosan as an Advanced Nanocarrier for Targeted Delivery: A Critical Review. Nanomedicine 2019, 14, 1623–1642. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, A.; Marchesi, F.; Malesci, A.; Laghi, L.; Europe PMC Funders Group. Tumor-Associated Macrophages as Treatment Targets in Oncology. Nat. Rev. Clin. Oncol. 2018, 14, 399–416. [Google Scholar] [CrossRef]
- Zlotnikov, I.D.; Ezhov, A.A.; Petrov, R.A.; Vigovskiy, M.A.; Grigorieva, O.A.; Belogurova, N.G.; Kudryashova, E.V. Mannosylated Polymeric Ligands for Targeted Delivery of Antibacterials and Their Adjuvants to Macrophages for the Enhancement of the Drug Efficiency. Pharmaceuticals 2022, 15, 1172. [Google Scholar] [CrossRef]
- Zlotnikov, I.D.; Vanichkin, D.A.; Kudryashova, E.V. Methods for Determining the Parameters of Receptor-Ligand Interactions on the Model of Concanavalin A and Mannosylated Chitosans Promising Carriers for Drug Delivery to Alveolar Macrophages. Biotekhnologiya 2021, 37, 28–40. [Google Scholar] [CrossRef]
- Zlotnikov, I.D.; Kudryashova, E.V. Mannose Receptors of Alveolar Macrophages as a Target for the Addressed Delivery of Medicines to the Lungs. Russ. J. Bioorganic Chem. 2022, 48, 46–75. [Google Scholar] [CrossRef]
- Zlotnikov, I.D.; Kudryashova, E.V. Computer Simulation of the Receptor–Ligand Interactions of Mannose Receptor CD206 in Comparison with the Lectin Concanavalin A Model. Biochem. 2022, 87, 54–69. [Google Scholar] [CrossRef]
- Zlotnikov, I.D.; Ezhov, A.A.; Vigovskiy, M.A.; Grigorieva, O.A.; Dyachkova, U.D.; Belogurova, N.G.; Kudryashova, E.V. Application Prospects of FTIR Spectroscopy and CLSM to Monitor the Drugs Interaction with Bacteria Cells Localized in Macrophages for Diagnosis and Treatment Control of Respiratory Diseases. Diagnostics 2023, 13, 698. [Google Scholar] [CrossRef]
- Bhat, A.R.; Wani, F.A.; Behera, K.; Khan, A.B.; Patel, R. Formulation of Biocompatible Microemulsions for Encapsulation of Anti-TB Drug Rifampicin: A Physicochemical and Spectroscopic Study. Colloids Surf. A Physicochem. Eng. Asp. 2022, 645, 128846. [Google Scholar] [CrossRef]
- Jafri, H.; Banerjee, G.; Khan, M.S.A.; Ahmad, I.; Abulreesh, H.H.; Althubiani, A.S. Synergistic Interaction of Eugenol and Antimicrobial Drugs in Eradication of Single and Mixed Biofilms of Candida Albicans and Streptococcus Mutans. AMB Express 2020, 10, 185. [Google Scholar] [CrossRef] [PubMed]
- Weisheimer, V.; Miron, D.; Silva, C.B.; Guterres, S.S.; Schapoval, E.E.S. Microparticles Containing Lemongrass Volatile Oil: Preparation, Characterization and Thermal Stability. Pharmazie 2010, 65, 885–890. [Google Scholar] [CrossRef]
- Leite, A.M.; Lima, E.D.O.; De Souza, E.L.; Diniz, M.D.F.F.M.; Trajano, V.N.; De Medeiros, I.A. Inhibitory Effect of β-Pinene, α-Pinene and Eugenol on the Growth of Potential Infectious Endocarditis Causing Gram-Positive Bacteria. Rev. Bras. Cienc. Farm. J. Pharm. Sci. 2007, 43, 121–126. [Google Scholar] [CrossRef] [Green Version]
- Tsyganov, D.V.; Samet, A.V.; Silyanova, E.A.; Ushkarov, V.I.; Varakutin, A.E.; Chernysheva, N.B.; Chuprov-Netochin, R.N.; Khomutov, A.A.; Volkova, A.S.; Leonov, S.V.; et al. Synthesis and Antiproliferative Activity of Triphenylphosphonium Derivatives of Natural Allylpolyalkoxybenzenes. ACS Omega 2022, 7, 3369–3383. [Google Scholar] [CrossRef] [PubMed]
- Elshafie, H.S.; Sakr, S.H.; Sadeek, S.A.; Camele, I. Biological Investigations and Spectroscopic Studies of New Moxifloxacin/Glycine-Metal Complexes. Chem. Biodivers. 2019, 16, e1800633. [Google Scholar] [CrossRef]
- Semenov, V.V.; Kiselyov, A.S.; Titov, I.Y.; Sagamanova, I.K.; Ikizalp, N.N.; Chernysheva, N.B.; Tsyganov, D.V.; Konyushkin, L.D.; Firgang, S.I.; Semenov, R.V.; et al. Synthesis of Antimitotic Polyalkoxyphenyl Derivatives of Combretastatin Using Plant Allylpolyalkoxybenzenes (1). J. Nat. Prod. 2010, 73, 1796–1802. [Google Scholar] [CrossRef] [PubMed]
- Kost, B.; Brzeziński, M.; Zimnicka, M.; Socka, M.; Wielgus, E.; Słowianek, M.; Biela, T. PLA Stereocomplexed Microspheres Modified with Methyl-β-Cyclodextrin as an Atropine Delivery System. Synthesis and Characterization. Mater. Today Commun. 2020, 25, 101605. [Google Scholar] [CrossRef]
- Pereira de Lira, M.H.; Fernandes Queiroga Moraes, G.; Macena Santos, G.; Patrício de Andrade Júnior, F.; De Oliveira Pereira, F.; Oliveira Lima, I. Synergistic Antibacterial Activity of Monoterpenes in Combination with Conventional Antimicrobials against Gram-Positive and Gram-Negative Bacteria. Rev. Ciências Médicas E Biológicas 2020, 19, 258. [Google Scholar] [CrossRef]
- Zlotnikov, I.D.; Belogurova, N.G.; Krylov, S.S.; Semenova, M.N.; Semenov, V.V.; Kudryashova, E.V. Plant Alkylbenzenes and Terpenoids in the Form of Cyclodextrin Inclusion Complexes as Antibacterial Agents and Levofloxacin Synergists. Pharmaceuticals 2022, 15, 861. [Google Scholar] [CrossRef]
- Tadtong, S.; Watthanachaiyingcharoen, R.; Kamkaen, N. Antimicrobial Constituents and Synergism Effect of the Essential Oils from Cymbopogon Citratus and Alpinia Galanga. Nat. Prod. Commun. 2014, 9, 277–280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cardoso, N.N.R.; Alviano, C.S.; Blank, A.F.; Romanos, M.T.V.; Fonseca, B.B.; Rozental, S.; Rodrigues, I.A.; Alviano, D.S. Synergism Effect of the Essential Oil from Ocimum Basilicum Var. Maria Bonita and Its Major Components with Fluconazole and Its Influence on Ergosterol Biosynthesis. Evidence-based Complement. Altern. Med. 2016, 2016, 5647182. [Google Scholar] [CrossRef] [Green Version]
- Ng, W.C.; Lokanathan, Y.; Fauzi, M.B.; Baki, M.M.; Zainuddin, A.A.; Phang, S.J.; Azman, M. In vitro evaluation of genipin-crosslinked gelatin hydrogels for vocal fold injection. Sci. Rep. 2023, 13, 5128. [Google Scholar] [CrossRef]
- Bhattarai, N.; Ramay, H.R.; Gunn, J.; Matsen, F.A.; Zhang, M. PEG-Grafted Chitosan as an Injectable Thermosensitive Hydrogel for Sustained Protein Release. J. Control. Release 2005, 103, 609–624. [Google Scholar] [CrossRef]
- Singh, P.; Medronho, B.; Alves, L.; da Silva, G.J.; Miguel, M.G.; Lindman, B. Development of Carboxymethyl Cellulose-Chitosan Hybrid Micro- and Macroparticles for Encapsulation of Probiotic Bacteria. Carbohydr. Polym. 2017, 175, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Zuo, R.; Shi, J.; Jiang, S.; Chu, M.; Wang, Q.; Kong, L.; Guan, J. Promotion of the genipin crosslinked chitosan-fiber hydrogel loaded with sustained release of clemastine fumarate in diabetic wound repair. Int. J. Biol. Macromol. 2023, 226, 900–914. [Google Scholar] [CrossRef]
- Mi, F.L.; Sung, H.W.; Shyu, S.S.; Su, C.C.; Peng, C.K. Synthesis and Characterization of Biodegradable TPP/Genipin Co-Crosslinked Chitosan Gel Beads. Polymer 2003, 44, 6521–6530. [Google Scholar] [CrossRef]
- Muzzarelli, R.A.A. Genipin-Crosslinked Chitosan Hydrogels as Biomedical and Pharmaceutical Aids. Carbohydr. Polym. 2009, 77, 1–9. [Google Scholar] [CrossRef]
- Rusu, A.; Tóth, G.; Szocs, L.; Kökösi, J.; Kraszni, M.; Gyéresi, Á.; Noszál, B. Triprotic Site-Specific Acid-Base Equilibria and Related Properties of Fluoroquinolone Antibacterials. J. Pharm. Biomed. Anal. 2012, 66, 50–57. [Google Scholar] [CrossRef]
- Vinogradov, E.; Petersen, B.; Bock, K. Structural Analysis of the Intact Polysaccharide Mannan from Saccharomyces Cerevisiae Yeast Using 1H and 13C NMR Spectroscopy at 750 MHz. Carbohydr. Res. 1998, 307, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Buranachai, T.; Praphairaksit, N.; Muangsin, N. Chitosan/Polyethylene Glycol Beads Crosslinked with Tripolyphosphate and Glutaraldehyde for Gastrointestinal Drug Delivery. AAPS PharmSciTech 2010, 11, 1128–1137. [Google Scholar] [CrossRef] [Green Version]
- Bratskaya, S.; Skatova, A.; Privar, Y.; Boroda, A.; Kantemirova, E.; Maiorova, M.; Pestov, A. Stimuli-Responsive Dual Cross-Linked n-Carboxyethylchitosan Hydrogels with Tunable Dissolution Rate. Gels 2021, 7, 188. [Google Scholar] [CrossRef] [PubMed]
- Skuredina, A.A.; Tychinina, A.S.; Le-Deygen, I.M.; Golyshev, S.A.; Belogurova, N.G.; Kudryashova, E.V. The Formation of Quasi-Regular Polymeric Network of Cross-Linked Sulfobutyl Ether Derivative of β-Cyclodextrin Synthesized with Moxifloxacin as a Template. React. Funct. Polym. 2021, 159, 104811. [Google Scholar] [CrossRef]
- Larsson, M.; Huang, W.C.; Hsiao, M.H.; Wang, Y.J.; Nydén, M.; Chiou, S.H.; Liu, D.M. Biomedical Applications and Colloidal Properties of Amphiphilically Modified Chitosan Hybrids. Prog. Polym. Sci. 2013, 38, 1307–1328. [Google Scholar] [CrossRef]
- Prabaharan, M.; Mano, J.F. Chitosan-Based Particles as Controlled Drug Delivery Systems. Drug Deliv. J. Deliv. Target. Ther. Agents 2005, 12, 41–57. [Google Scholar] [CrossRef]
- He, Z.; Liu, C.; Zhao, J.; Guo, F.; Wang, Y. Enhanced gelling properties and hydration capacity of ginkgo seed proteins by genipin cross-linking. Food Chem. 2023, 399, 133924. [Google Scholar] [CrossRef] [PubMed]
- Vasile, C.; Pamfil, D.; Stoleru, E.; Baican, M. New Developments in Medical Applications of Hybrid Hydrogels Containing Natural Polymers. Molecules 2020, 25, 1539. [Google Scholar] [CrossRef] [Green Version]
- Charhouf, I.; Bennamara, A.; Abourriche, A.; Chenite, A.; Zhu, J.; Berrada, M. Characterization of a dialdehyde chitosan generated by periodate oxidation. Biosensors 2014, 16, 18. [Google Scholar]
- Samal, S.K.; Dash, M.; Van Vlierberghe, S.; Kaplan, D.L.; Chiellini, E.; van Blitterswijk, C.; Moroni, L.; Dubruel, P. Cationic Polymers and Their Therapeutic Potential. Chem. Soc. Rev. 2012, 41, 7147–7194. [Google Scholar] [CrossRef]
- Prabaharan, M. Chitosan-Based Nanoparticles for Tumor-Targeted Drug Delivery. Int. J. Biol. Macromol. 2015, 72, 1313–1322. [Google Scholar] [CrossRef]
- Bhattarai, N.; Gunn, J.; Zhang, M. Chitosan-Based Hydrogels for Controlled, Localized Drug Delivery. Adv. Drug Deliv. Rev. 2010, 62, 83–99. [Google Scholar] [CrossRef]
- Chung, Y.C.; Su, Y.P.; Chen, C.C.; Jia, G.; Wang, H.L.; Wu, J.C.G.; Lin, J.G. Relationship between Antibacterial Activity of Chitosan and Surface Characteristics of Cell Wall. Acta Pharmacol. Sin. 2004, 25, 932–936. [Google Scholar] [PubMed]
- Zlotnikov, I.D.; Ezhov, A.A.; Ferberg, A.S.; Krylov, S.S.; Semenova, M.N.; Semenov, V.V.; Kudryashova, E.V. Polymeric Micelles Formulation of Combretastatin Derivatives with Enhanced Solubility, Cytostatic Activity and Selectivity against Cancer Cells. Pharmaceutics 2023, 15, 1613. [Google Scholar] [CrossRef]
- Alvarez-Ordóñez, A.; Mouwen, D.J.M.; López, M.; Prieto, M. Fourier Transform Infrared Spectroscopy as a Tool to Characterize Molecular Composition and Stress Response in Foodborne Pathogenic Bacteria. J. Microbiol. Methods 2011, 84, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Razzaghi-Abyaneh, M.; Yoshinari, T.; Shams-Ghahfarokhi, M.; Rezaee, M.B.; Nagasawa, H.; Sakuda, S. Dillapiol and Apiol as Specific Inhibitors of the Biosynthesis of Aflatoxin G1 in Aspergillus Parasiticus. Biosci. Biotechnol. Biochem. 2007, 71, 2329–2332. [Google Scholar] [CrossRef] [Green Version]
- Jacobson, T.A. Comparative Pharmacokinetic Interaction Profiles of Pravastatin, Simvastatin, and Atorvastatin When Coadministered with Cytochrome P450 Inhibitors. Am. J. Cardiol. 2004, 94, 1140–1146. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, E. Clinically Important Pharmacokinetic Drug-Drug Interactions: Role of Cytochrome P450 Enzymes. J. Clin. Pharm. Ther. 1998, 23, 403–416. [Google Scholar] [CrossRef]
- Zlotnikov, I.D.; Malashkeevich, S.M.; Belogurova, N.G.; Kudryashova, E.V. Thermoreversible Gels Based on Chitosan Copolymers as “Intelligent” Drug Delivery System with Prolonged Action for Intramuscular Injection. Pharmaceutics 2023, 15, 1478. [Google Scholar] [CrossRef]
- Amin, M.; Abbas, N.S.; Hussain, M.A.; Edgar, K.J.; Tahir, M.N.; Tremel, W.; Sher, M. Cellulose Ether Derivatives: A New Platform for Prodrug Formation of Fluoroquinolone Antibiotics. Cellulose 2015, 22, 2011–2022. [Google Scholar] [CrossRef]
- Abbas, N.S.; Amin, M.; Hussain, M.A.; Edgar, K.J.; Tahir, M.N.; Tremel, W. Extended Release and Enhanced Bioavailability of Moxifloxacin Conjugated with Hydrophilic Cellulose Ethers. Carbohydr. Polym. 2016, 136, 1297–1306. [Google Scholar] [CrossRef]
- Riederer, M.S.; Requist, B.D.; Payne, K.A.; Way, J.D.; Krebs, M.D. Injectable and Microporous Scaffold of Densely-Packed, Growth Factor-Encapsulating Chitosan Microgels. Carbohydr. Polym. 2016, 152, 792–801. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zlotnikov, I.D.; Streltsov, D.A.; Ezhov, A.A. Smart PH- and Temperature-Sensitive Micelles Based on Chitosan Grafted with Fatty Acids to Increase the Efficiency and Selectivity of Doxorubicin and Its Adjuvant Regarding the Tumor Cells. Pharmaceutics 2023, 15, 1135. [Google Scholar] [CrossRef] [PubMed]
- Zlotnikov, I.D.; Streltsov, D.A.; Belogurova, N.G.; Kudryashova, E.V. Chitosan or Cyclodextrin Grafted with Oleic Acid Self-Assemble into Stabilized Polymeric Micelles with Potential of Drug Carriers. Life 2023, 13, 446. [Google Scholar] [CrossRef] [PubMed]
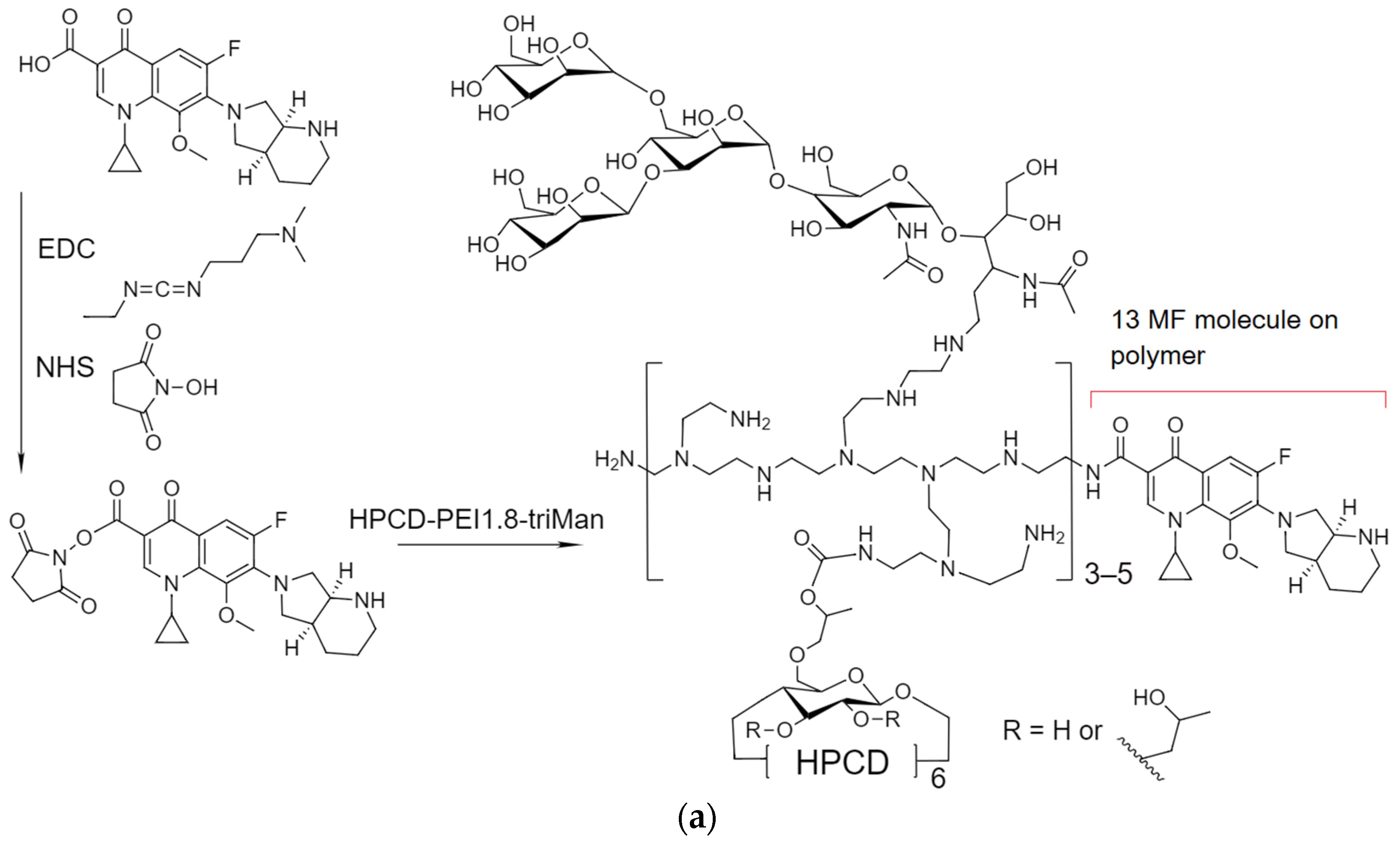
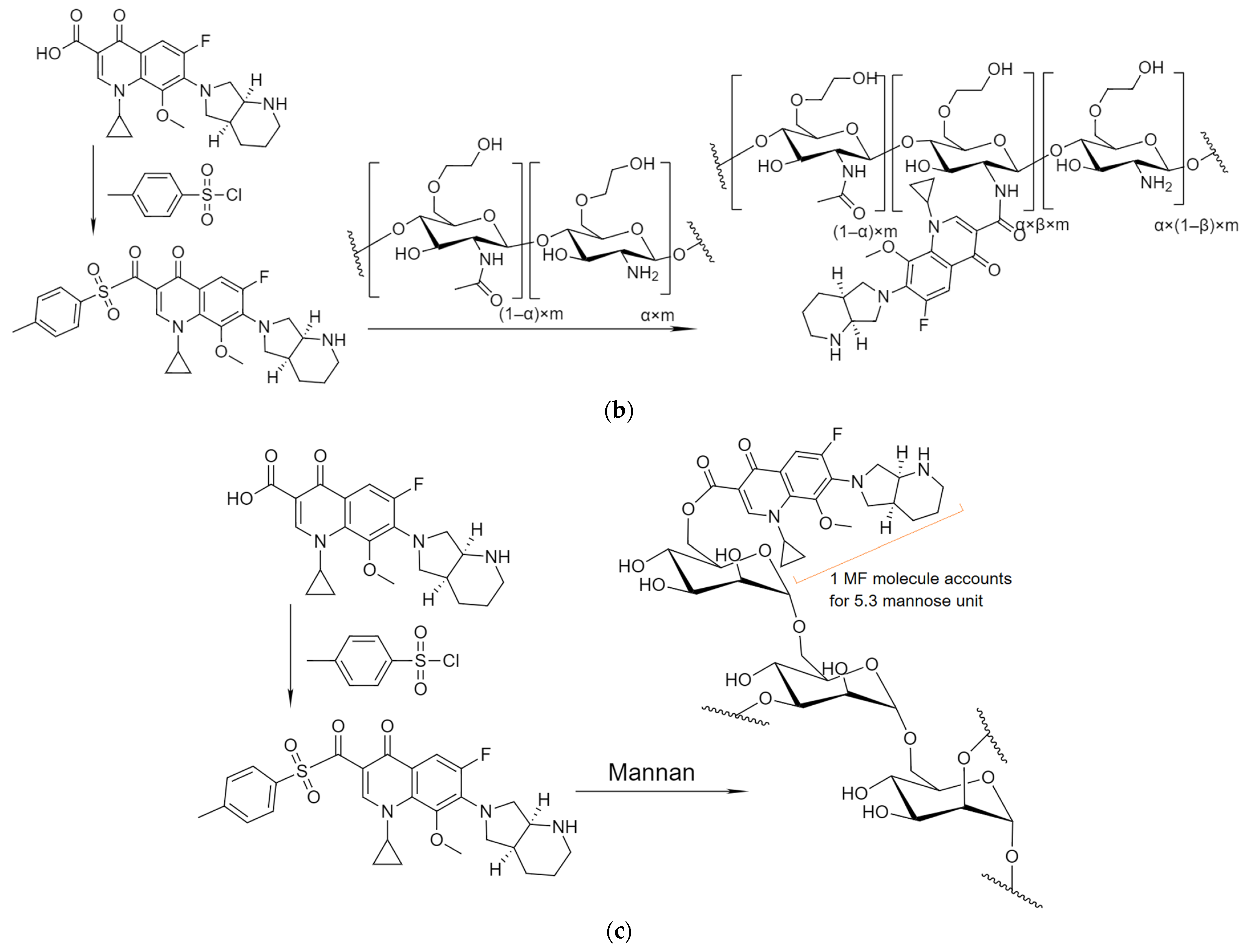

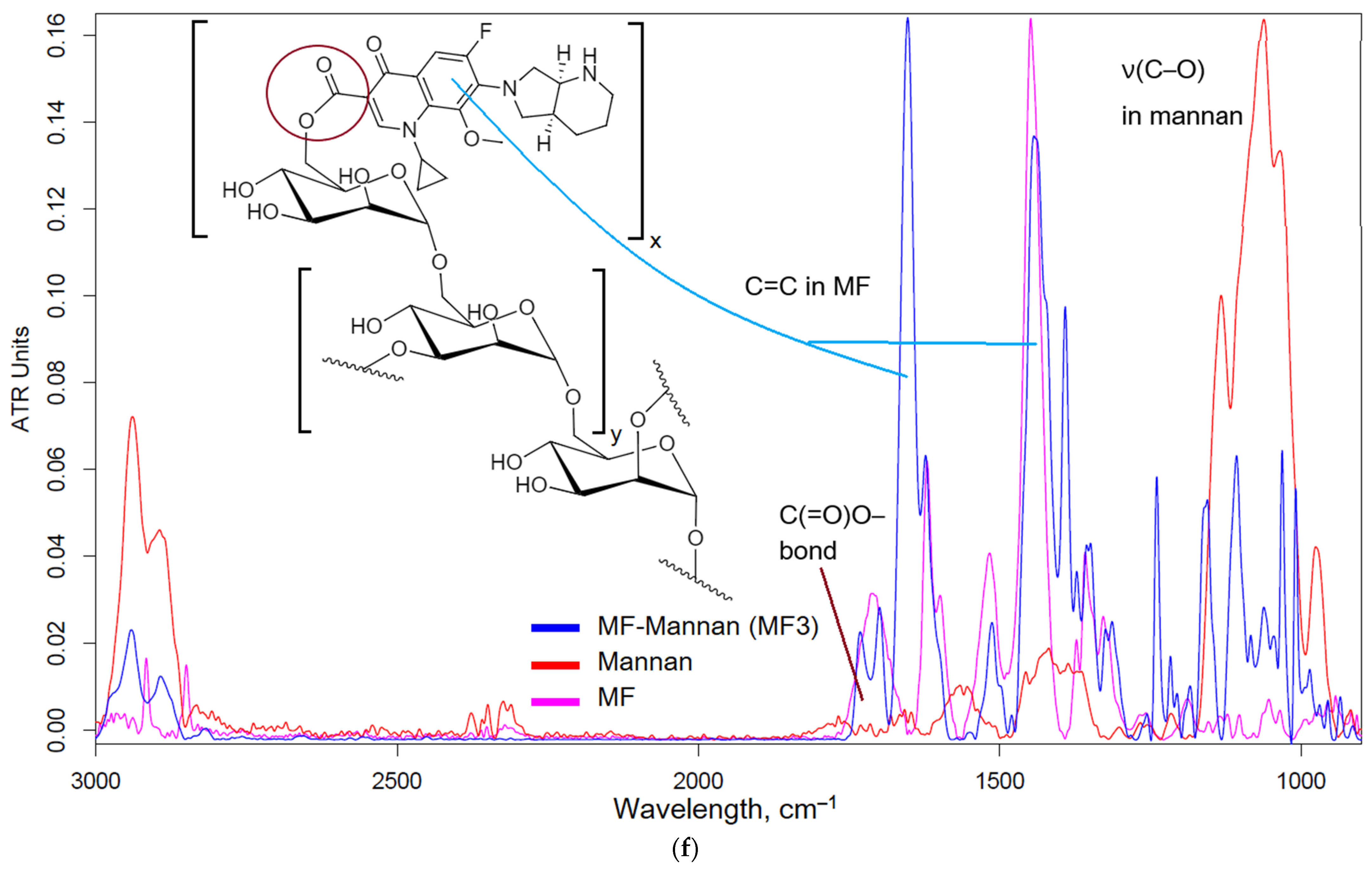
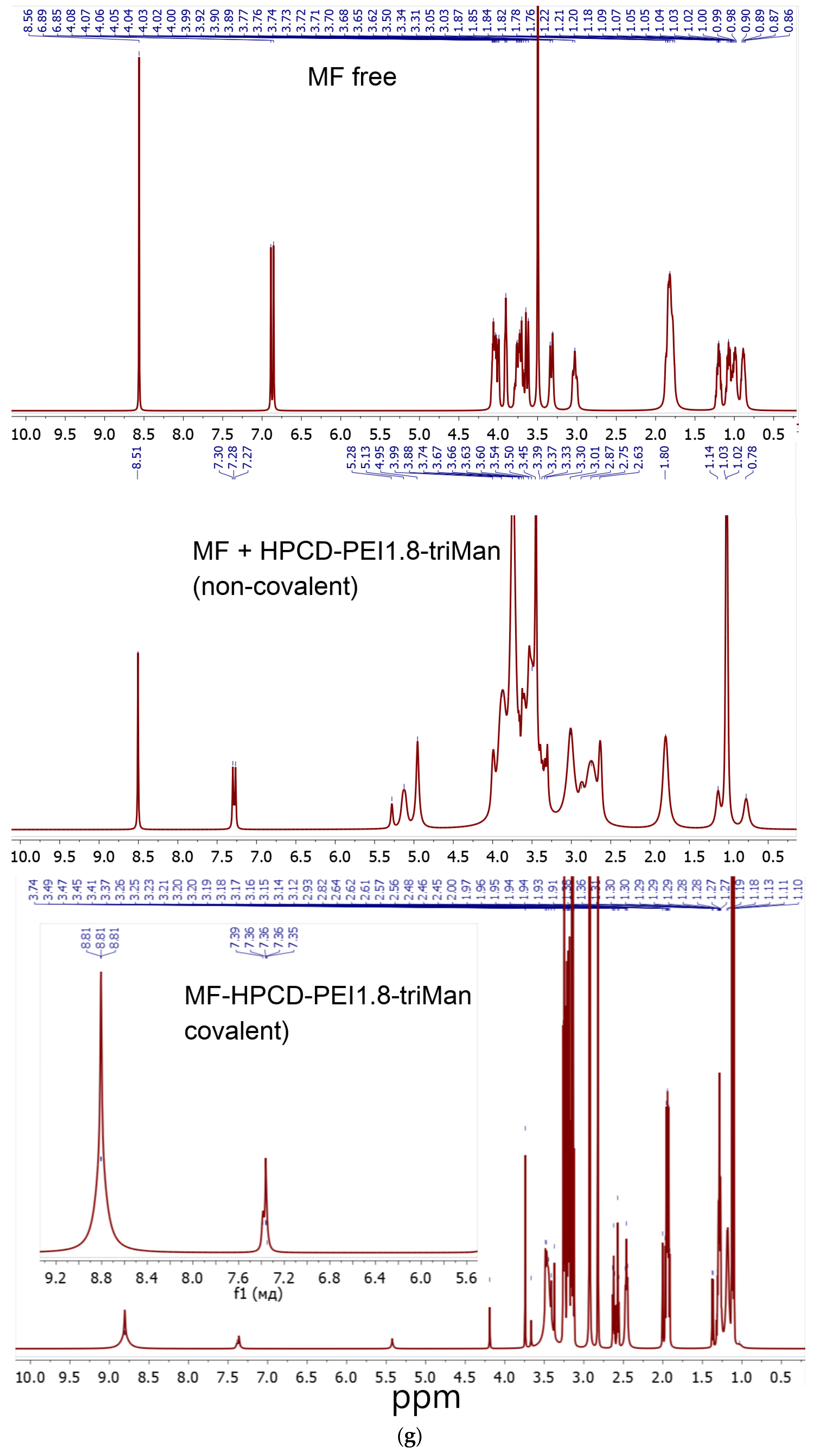
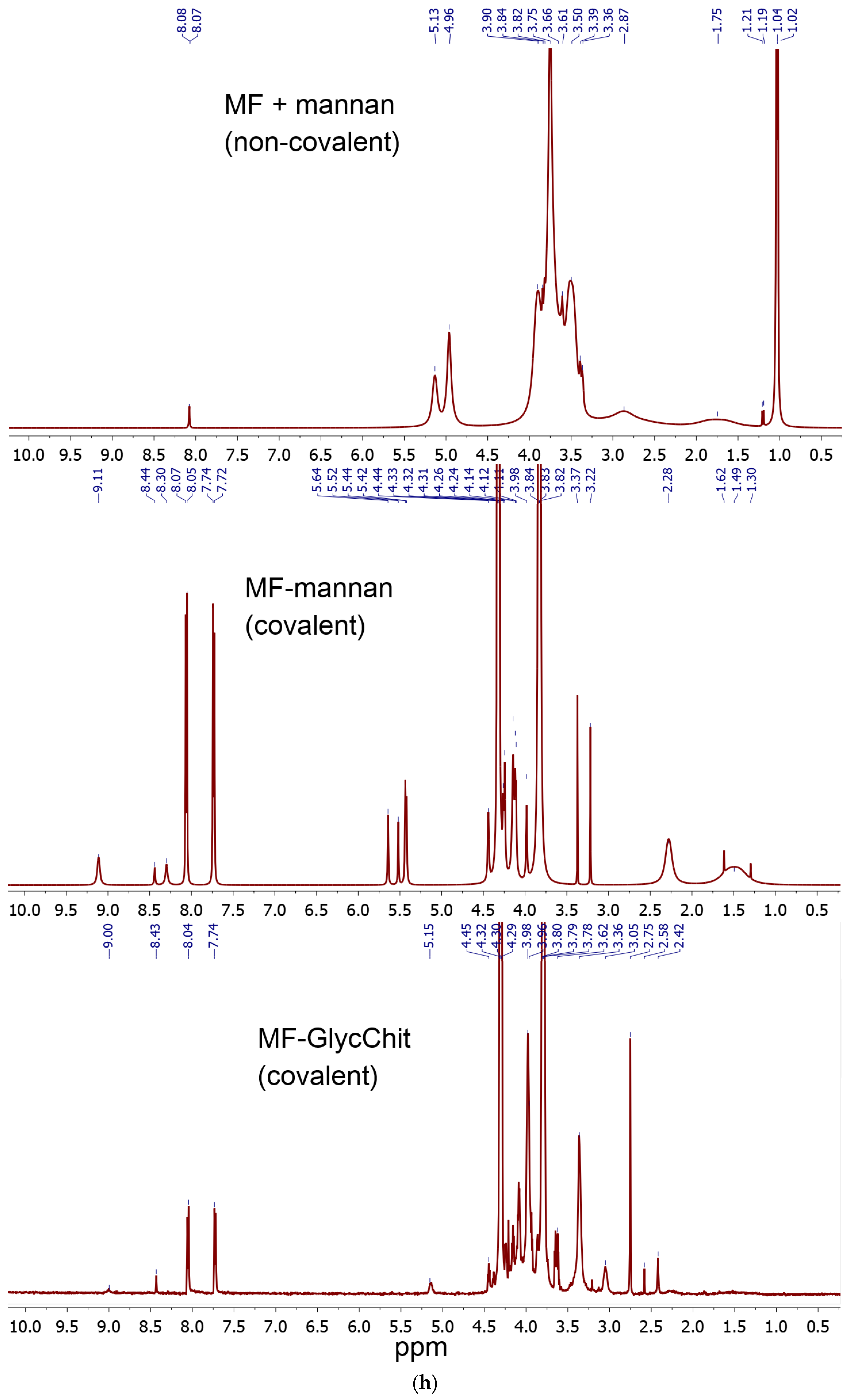
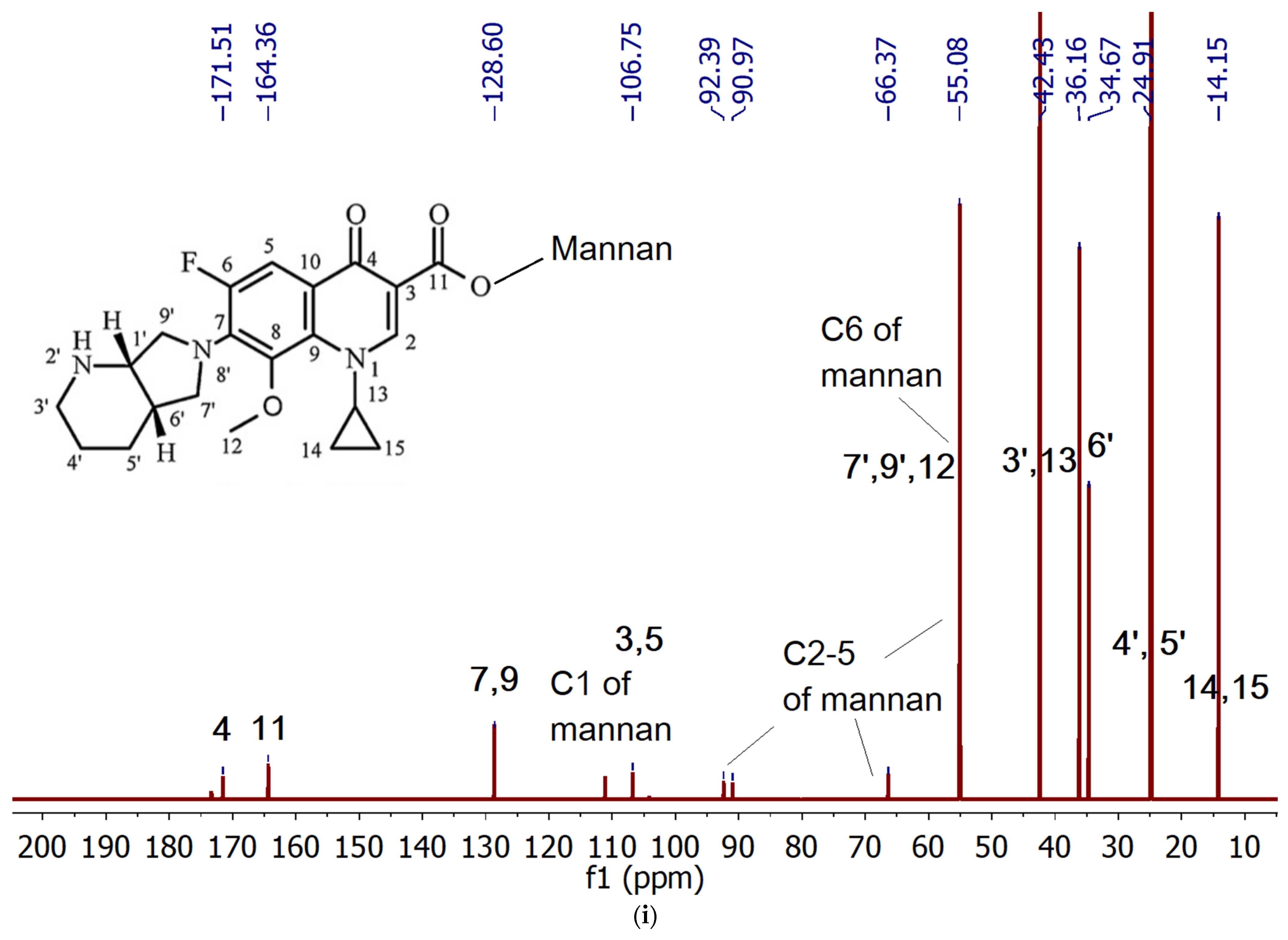

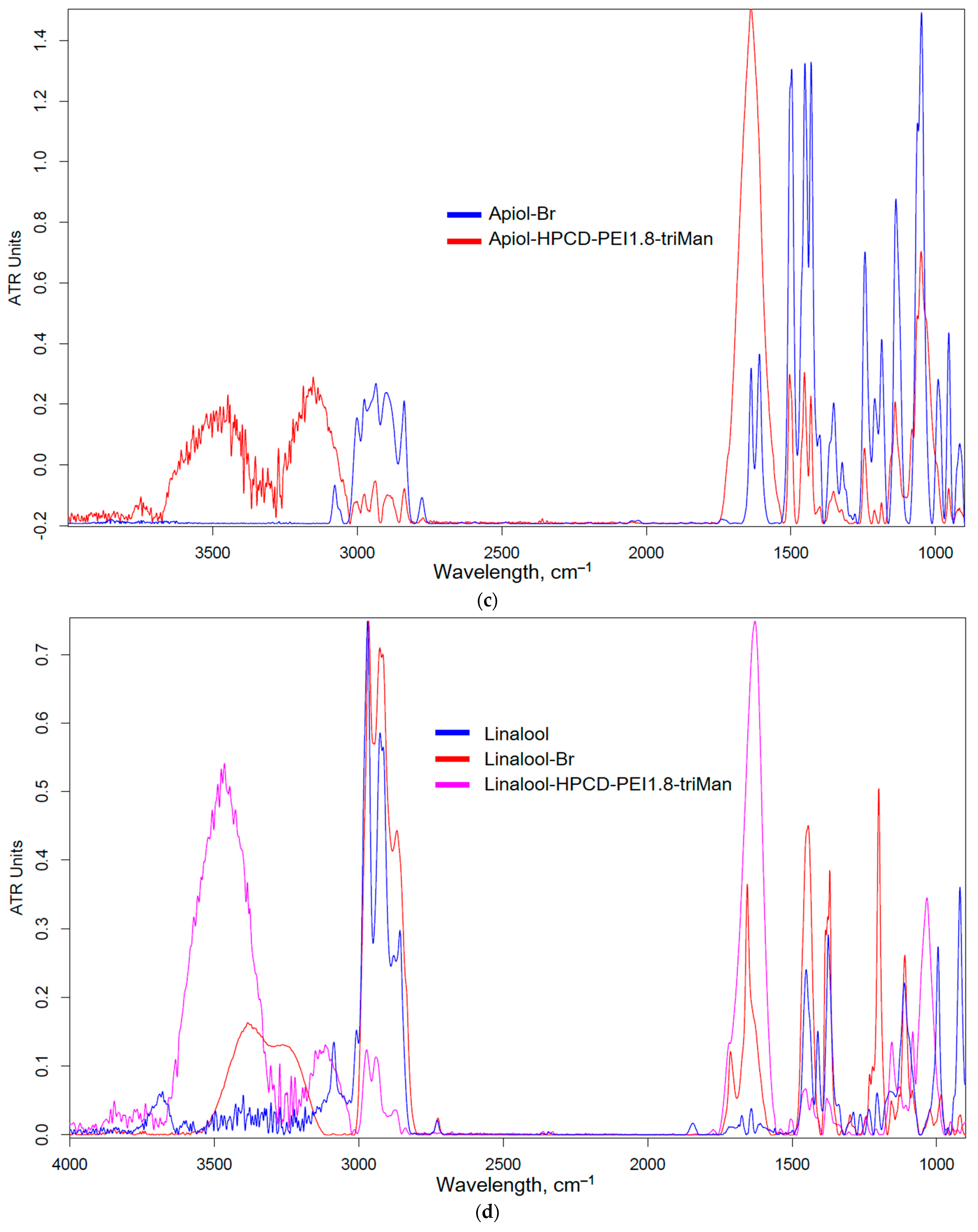
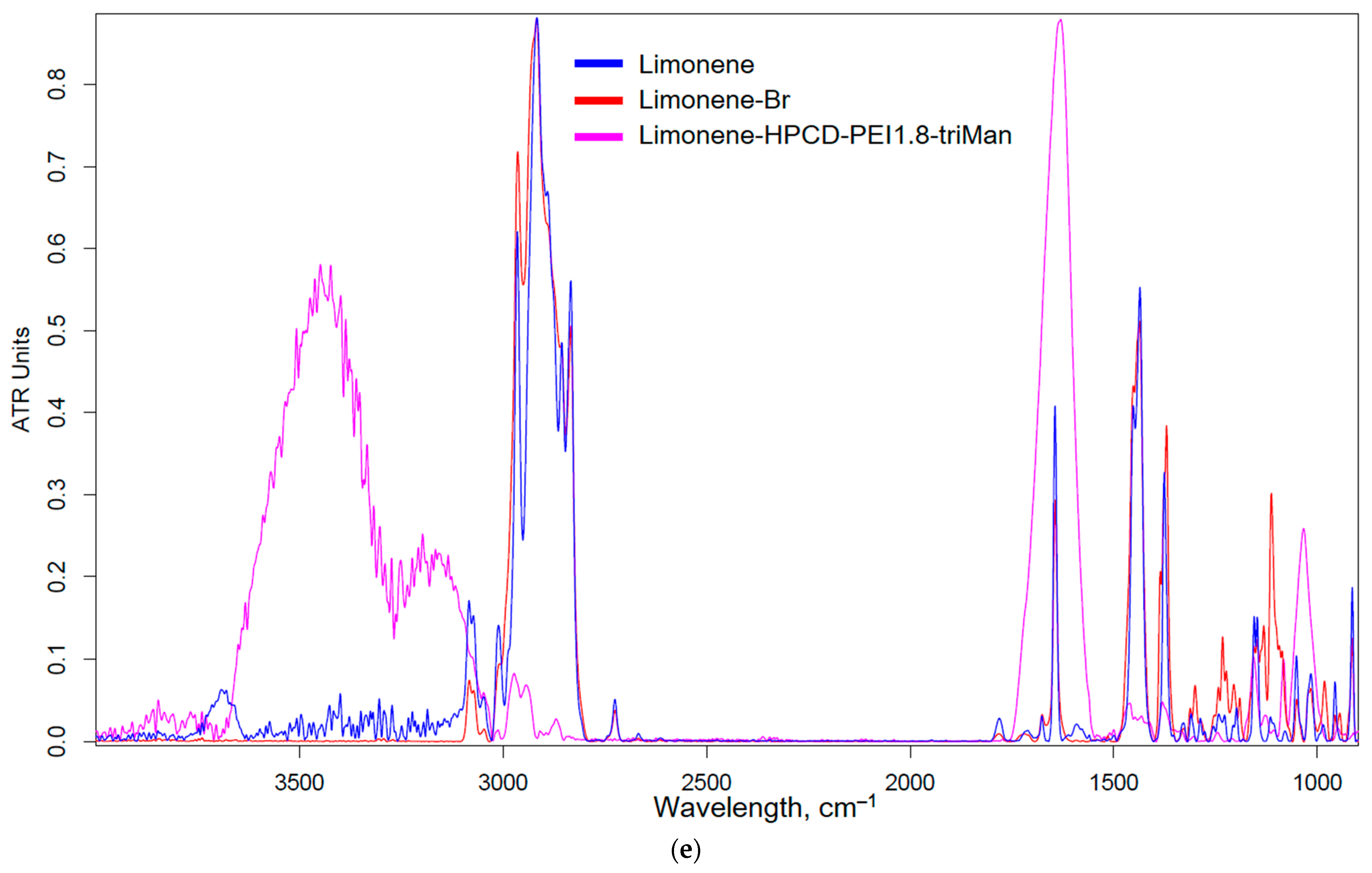
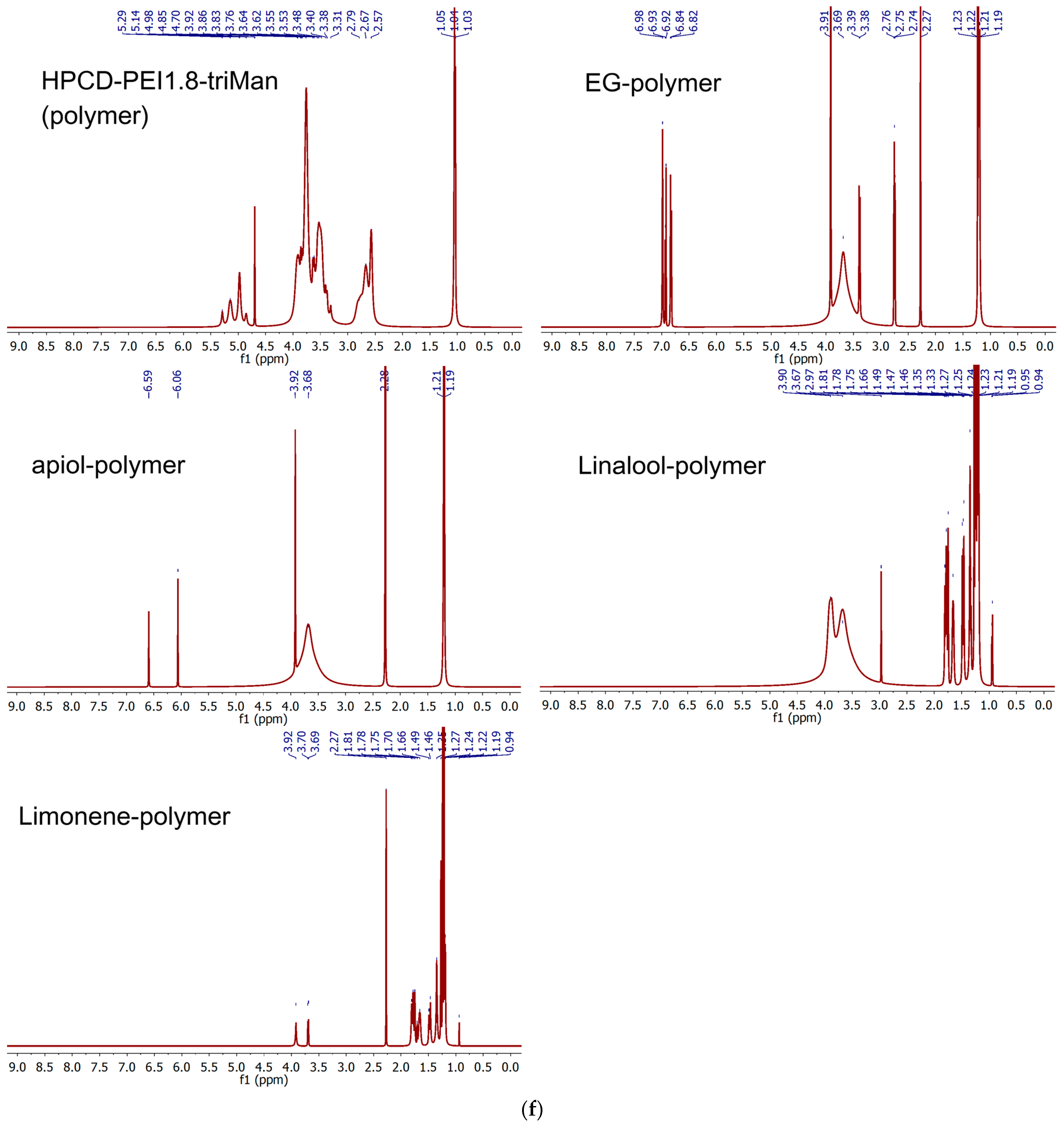
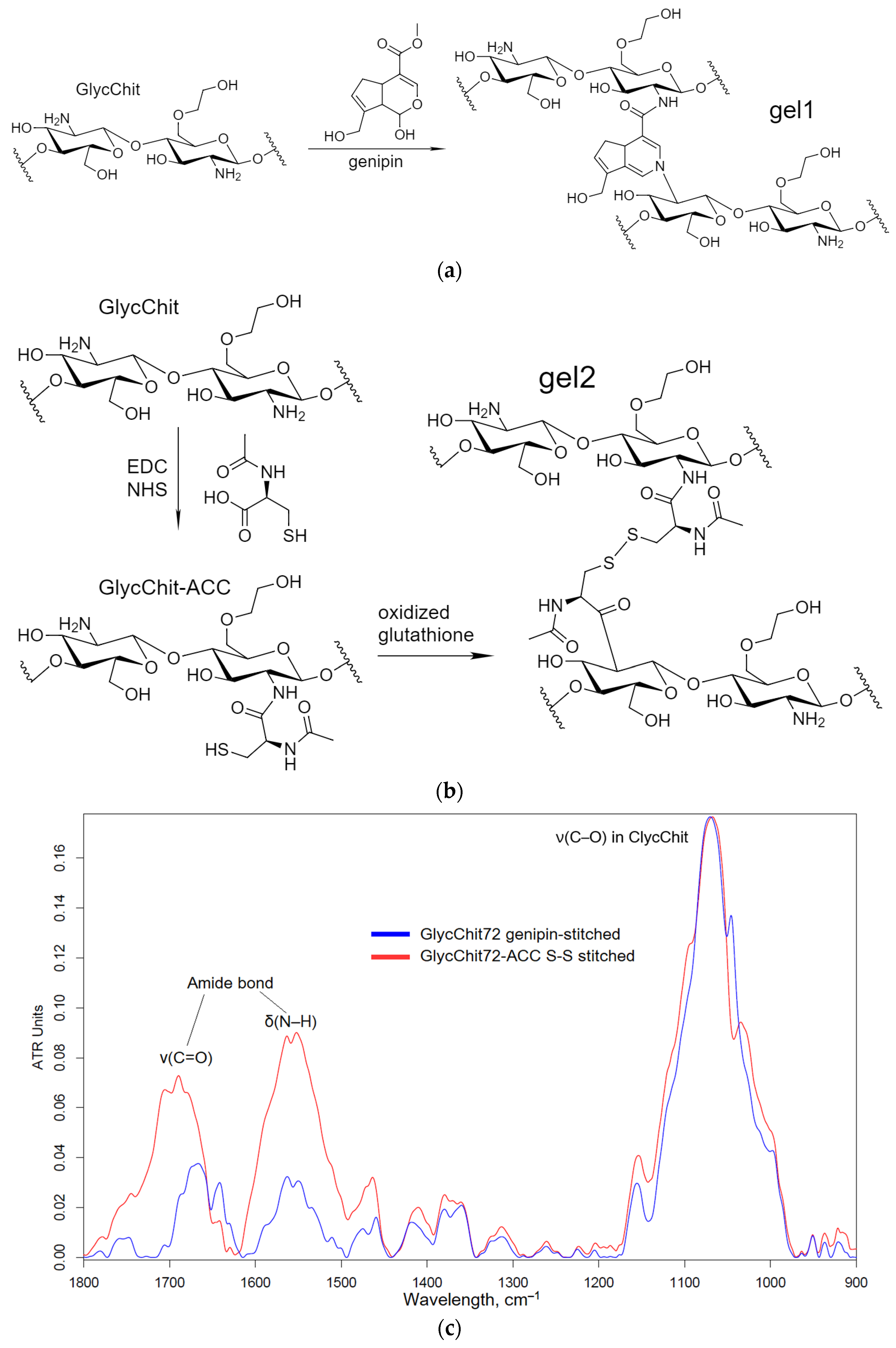
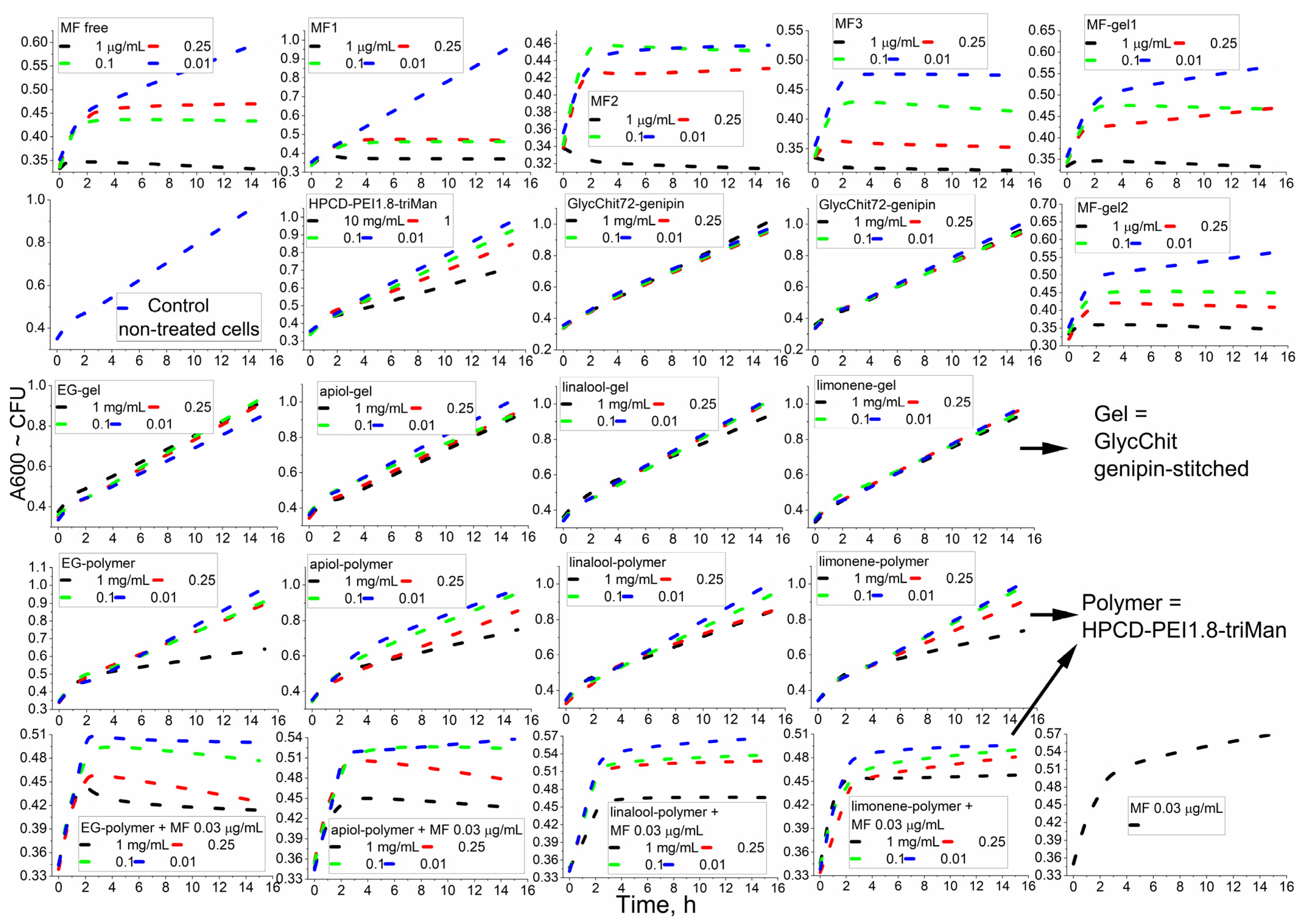


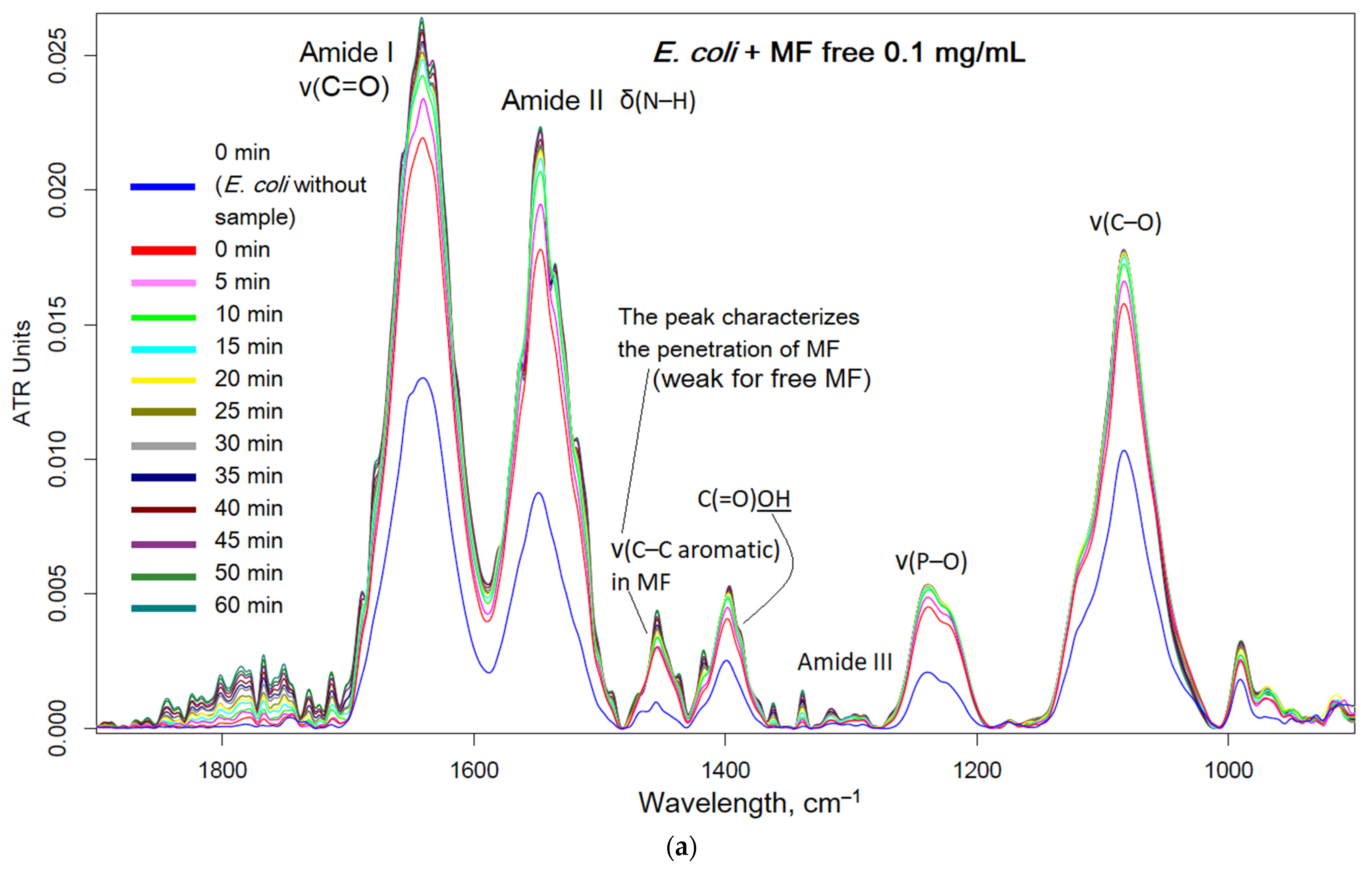
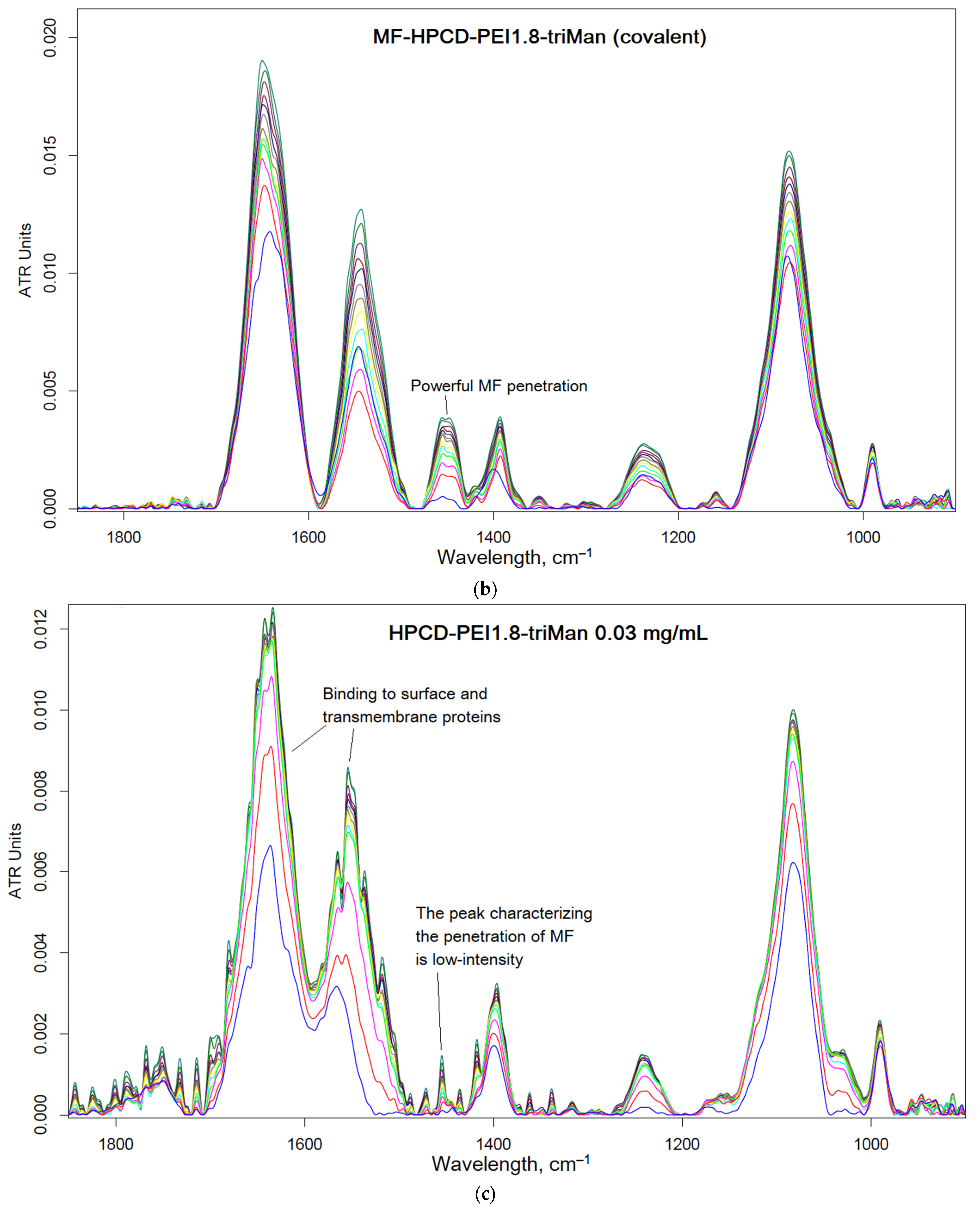



| Designation | Chemical Composition | MF Percentage *, % |
|---|---|---|
| MF | MF free | 100 |
| MF1 | MF–HPCD–PEI1.8-triMan (covalent), 13 MF molecule on polymer | 34 ± 5 |
| MF2 | MF–GlycChit (covalent), 1 MF molecule accounts for 2.4 glucosamine unit | 45 ± 3 |
| MF3 | MF–Mannan (covalent), 1 MF molecule accounts for 5.3 mannose unit | 32 ± 2 |
| MF-gel1 | MF + GlycChit genipin-stitched (non-covalent) | 72 ± 4 |
| MF-gel2 | MF + GlycChit-acetylcysteine S-S stitched (non-covalent) | 69 ± 3 |
| Designation * | IC50 against E. coli, ng/mL | IC50 against B. subtilis, ng/mL |
|---|---|---|
| MF | 8 ± 2 | 800 ± 300 |
| MF1 | 40 ± 7 | 11 ± 3 |
| MF2 | 4 ± 1 | 9 ± 2 |
| MF3 | 3 ± 1 | 10 ± 3 |
| MF-gel1 | 7 ± 2 | 20 ± 5 |
| MF-gel2 | 7 ± 2 | 15 ± 4 |
| E. coli | B. subtilis | |||||
|---|---|---|---|---|---|---|
| Compound X | X-Covalent with HPCD–PEI1.8–triMan (X-Polymer) | MF Free 30 ng/mL * + X-Covalent with HPCD–PEI1.8–triMan | X + GlycChit Genipin-Stitched (X-gel) | X-Covalent with HPCD–PEI1.8–triMan (X-Polymer) | MF Free 30 ng/mL ** + X-Covalent with HPCD–PEI1.8–triMan | X + GlycChit Genipin-Stitched (X-Gel) |
| EG | 64 ± 7 | 41 ± 3 | 85 ± 11 | 81 ± 8 | 60 ± 7 | 94 ± 3 |
| Apiol | 75 ± 9 | 44 ± 5 | 92 ± 8 | 81 ± 7 | 59 ± 7 | 93 ± 4 |
| Linalool | 85 ± 6 | 47 ± 4 | 93 ± 7 | 85 ± 9 | 62 ± 5 | 97 ± 2 |
| Limonene | 74 ± 9 | 46 ± 2 | 94 ± 5 | 83 ± 6 | 59 ± 7 | 89 ± 5 |
| Parameters | MF Free | MF + HPCD–PEI1.8–triMan (Non-Covalent) | MF + EG + HPCD–PEI1.8–triMan (Non-Covalent) | MF + Apiol + HPCD–PEI1.8–triMan (Non-Covalent) | MF–Mannan (Covalent) | MF + Mannan (Mixture) |
|---|---|---|---|---|---|---|
| Half-distribution period, min | 20 ± 2 | 15 ± 1 | 12 ± 2 | 25 ± 4 | 15 ± 2 | 14 ± 2 |
| Half-elimination period, h | 6.5 ± 0.5 | 10 ± 1 | 22 ± 2 | 45 ± 6 | 42 ± 7 | 24 ± 3 |
| Kinetical distribution volume, L | 8 ± 1 | 4 ± 1 | 4 ± 1 | 3 ± 1 | 3 ± 1 | 3 ± 1 |
| Stationary distribution volume, L | 4 ± 1 | 4 ± 1 | 4 ± 1 | 3 ± 1 | 3 ± 1 | 3 ± 1 |
| Clearance, mL/min | 14 ± 2 | 4.5 ± 0.6 | 2.0 ± 0.4 | 0.9 ± 0.2 | 0.8 ± 0.1 | 1.3 ± 0.3 |
| Area under curve | 360 ± 70 | 1100 ± 200 | 2400 ± 300 | 5800 ± 700 | 6500 ± 900 | 3800 ± 500 |
| Mean residence time, h | 5 ± 1 | 14 ± 2 | 28 ± 4 | 35 ± 6 | 35 ± 4 | 29 ± 3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zlotnikov, I.D.; Davydova, M.P.; Danilov, M.R.; Krylov, S.S.; Belogurova, N.G.; Kudryashova, E.V. Covalent Conjugates of Allylbenzenes and Terpenoids as Antibiotics Enhancers with the Function of Prolonged Action. Pharmaceuticals 2023, 16, 1102. https://doi.org/10.3390/ph16081102
Zlotnikov ID, Davydova MP, Danilov MR, Krylov SS, Belogurova NG, Kudryashova EV. Covalent Conjugates of Allylbenzenes and Terpenoids as Antibiotics Enhancers with the Function of Prolonged Action. Pharmaceuticals. 2023; 16(8):1102. https://doi.org/10.3390/ph16081102
Chicago/Turabian StyleZlotnikov, Igor D., Maria P. Davydova, Milan R. Danilov, Sergey S. Krylov, Natalya G. Belogurova, and Elena V. Kudryashova. 2023. "Covalent Conjugates of Allylbenzenes and Terpenoids as Antibiotics Enhancers with the Function of Prolonged Action" Pharmaceuticals 16, no. 8: 1102. https://doi.org/10.3390/ph16081102







