Profiling the Phytochemicals of Orostachys margaritifolia: Biological Activities, LC-ESI/MS, and HPLC Analyses
Abstract
:1. Introduction
2. Results
2.1. LC-ESI/MS
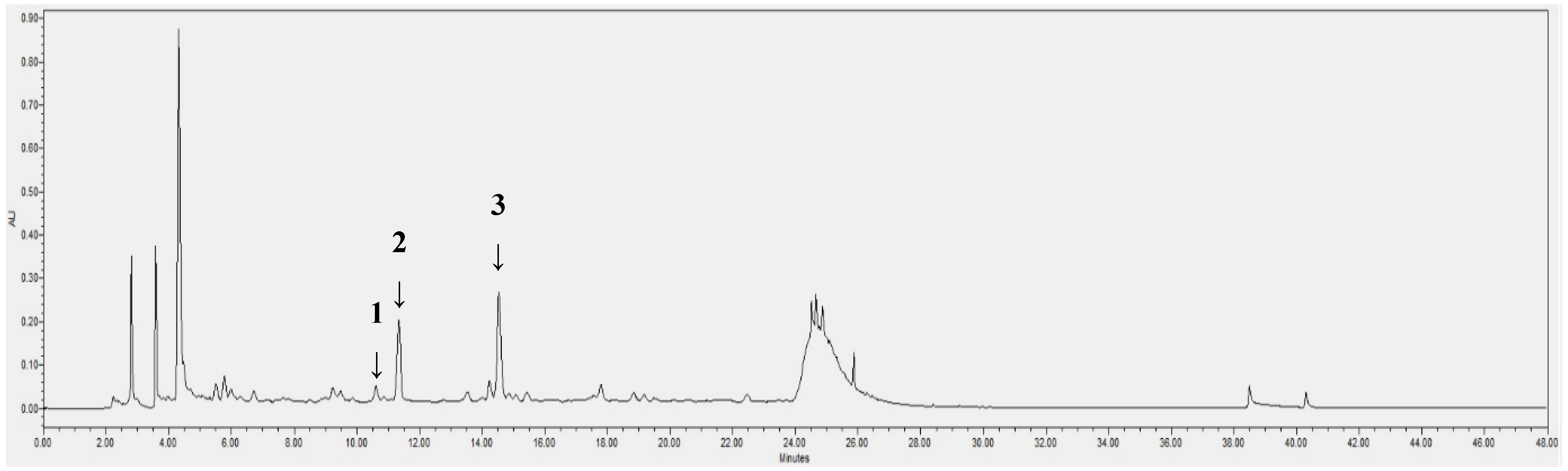
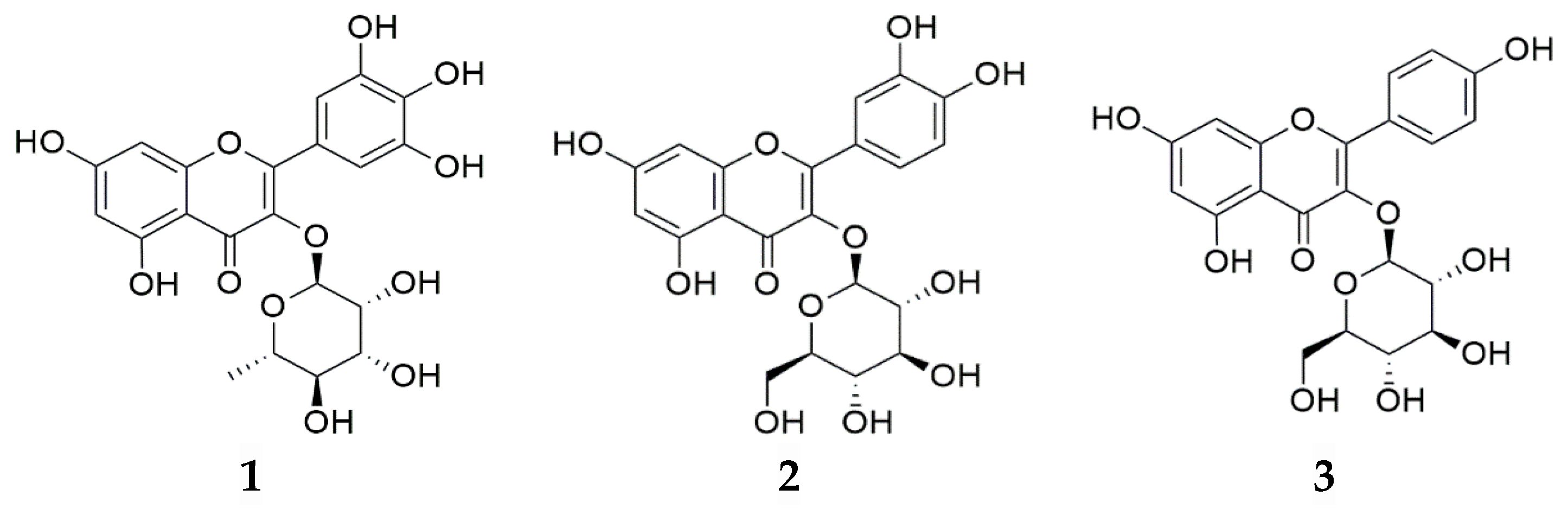
2.2. HPLC/PDA Analysis
2.3. Antioxidant Activity
2.4. NO Inhibitory Activity
3. Discussion
4. Materials and Methods
4.1. Plant Materials
4.2. Instruments and Reagents
4.3. Extraction from OMY Samples
4.4. Preparation of Samples and Standard Solutions for HPLC/PDA
4.5. LC-ESI/MS Conditions
4.6. HPLC/PDA Conditions
4.7. Calibration Curve
4.8. ABTS+ Radical Scavenging Activity
4.9. DPPH Radical Scavenging Activity
4.10. Nitric Oxide (NO) Inhibitory Activity
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lee, J.; Son, H.; Lee, K.H.; Kim, S.; Myagmar, G.; Kim, S.Y.; Chun, Y.; Yoo, H.Y. Identification and characterization of major flavonoids in extracts from an unexplored medicinal herb Orostachys fimbriata. Horticulturae 2022, 8, 1092. [Google Scholar] [CrossRef]
- Kwon, Y.K.; Kim, S.W.; Seo, J.M.; Liu, J.R. Prediction and discrimination of taxonomic relationship with Orostachys species using FT-IR spectroscopy combined by multivariate analysis. J. Plant Biol. 2011, 38, 9–14. [Google Scholar] [CrossRef]
- Nugroho, A.; Kim, M.H.; Han, Y.R.; Choi, J.S.; Park, H.J. High-performance liquid chromatographic quantification of flavonol glycosides in Orostachys species. Nat. Prod. Sci. 2012, 18, 32–38. [Google Scholar]
- Kim, H.D.; Park, K.R. Genetic variation in five species of Korean Orostachys (Crassulaceae). Korean J. Plant Taxon. 2005, 35, 295–311. [Google Scholar] [CrossRef]
- Hassan, M.H.A.; Elwekeel, A.; Moawad, A.; Afifi, N.; Amir, D.E. Phytochemical constituents and biological activity of selected genera of family Crassulaceae; a review. S. Afr. J. Bot. 2021, 141, 383–404. [Google Scholar] [CrossRef]
- Kim, I.S.; Pak, J.H.; Seo, B.B.; Song, S.D. Foliar ultrastructure of Korean Orostachys species. Korean J. Electron Microscopy 1995, 25, 52–61. [Google Scholar]
- Yin, X.; Zhang, J.; Wang, S.; Li, D. Magnetic separation hydroxynitrile glucoside of Orostachys malacophyllus. Microchem. J. 2021, 166, 106223. [Google Scholar] [CrossRef]
- Yang, X.; Cushman, J.C.; Borland, A.M.; Edwards, E.J.; Wullschleger, S.D.; Tuskan, G.A.; Owen, N.A.; Griffiths, H.; Smith, J.A.; De Paoli, H.C.; et al. A roadmap for research on crassulacean acid metabolism (CAM) to enhance sustainable food and bioenergy production in a hotter, drier world. New Phytol. 2015, 207, 491–504. [Google Scholar] [CrossRef]
- Lee, B.A.; Kim, H.H.; Lee, C.H. Morphological diversities of leaf in Korean native Orostachys species. Korean J. Plant Res. 2001, 14, 157–162. [Google Scholar]
- Lee, B.A.; Kim, H.H.; Cho, Y.G.; Lee, C.H. Analysis of genetic relationship among Korean native Orostachys species using RAPD. Hortic. Sci. Technol. 2001, 19, 159–162. [Google Scholar]
- Ma, C.J.; Jung, W.J.; Lee, K.Y.; Kim, Y.C.; Sung, S.H. Calpain inhibitory flavonoids isolated from Orostachys japonicus. J. Enzyme Inhib. Med. Chem. 2009, 24, 676–679. [Google Scholar]
- Jung, H.J.; Choi, J.; Nam, J.H.; Park, H.J. Anti-ulcerogenic effects of the flavonoid-rich fraction from the extract of Orostachys japonicus in mice. J. Med. Food 2007, 10, 702–706. [Google Scholar] [CrossRef] [PubMed]
- Sung, S.H.; Jung, W.J.; Kim, Y.C. A novel flavonol lyxoside of Orostachys japonicus herb. Nat. Prod. Lett. 2002, 16, 29–32. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Lee, S.J.; Park, S.; Kim, H.K.; Jeong, W.Y.; Choi, J.Y.; Sung, N.J.; Lee, W.S.; Lim, C.S.; Kim, G.S.; et al. Characterisation of flavonoids in Orostachys japonicus A. Berger using HPLC-MS/MS: Contribution to the overall antioxidant effect. Food Chem. 2011, 124, 1627–1633. [Google Scholar] [CrossRef]
- Yin, X.; Zhang, J.; Wang, S. Chemical constituents of the stems of Orostachys malacophyllus. Chem. Nat. Compd. 2020, 56, 740–742. [Google Scholar] [CrossRef]
- Zhongyi, F.; Yue, Z.; Guang, Y.; Wenwen, L.; Xiumei, Y.; Lili, J.; Sihong, W. Chemical constituents from Orostachys cartilaginea Borissova and their chemotaxonomic significance. Biochem. Syst. Ecol. 2022, 103, 104425. [Google Scholar] [CrossRef]
- Hur, S.; Jang, E.; Lee, J.H. Beneficial actions of Orostachys japonica and its compounds against tumors via MAPK signaling pathways. Nutrients 2021, 13, 555. [Google Scholar] [CrossRef]
- Park, J.C.; Han, W.D.; Park, J.R.; Choi, S.H.; Choi, J.W. Changes in hepatic drug metabolizing enzymes and lipid peroxidation by methanol extract and major compound of Orostachys japonicus. J. Ethnopharmacol. 2005, 102, 313–318. [Google Scholar] [CrossRef]
- Hur, J.M.; Park, J.C. Effects of the aerial parts of Orostachys japonicus and its bioactive component on hepatic alcohol-metabolizing enzyme system. J. Med. Food 2006, 9, 336–341. [Google Scholar] [CrossRef]
- Zhou, Z.; Qi, J.; Zhao, J.; Seo, J.H.; Shin, D.G.; Cha, J.D.; Lim, C.W.; Kim, J.W.; Kim, B. Orostachys japonicus ameliorates acetaminophen-induced acute liver injury in mice. J. Ethnopharmacol. 2021, 265, 113392. [Google Scholar] [CrossRef]
- Jeong, J.H.; Ryu, D.S.; Suk, D.H.; Lee, D.S. Anti-inflammatory effects of ethanol extract from Orostachys japonicus on modulation of signal pathways in LPS-stimulated RAW 264.7 cells. BMB Rep. 2011, 44, 399–404. [Google Scholar] [CrossRef]
- Lee, H.S.; Lee, G.S.; Kim, S.H.; Kim, H.K.; Suk, D.H.; Lee, D.S. Anti-oxidizing effect of the dichloromethane and hexane fractions from Orostachys japonicus in LPS-stimulated RAW 264.7 cells via upregulation of Nrf2 expression and activation of MAPK signaling pathway. BMB Rep. 2014, 47, 98–103. [Google Scholar] [CrossRef]
- Lee, S.G.; Kim, J.S.; Lee, H.S.; Lim, Y.M.; So, J.H.; Hahn, D.; Ha, Y.S.; Nam, J.O. Bioconverted Orostachys japonicas extracts suppress angiogenic activity of Ms-1 endothelial cells. Int. J. Mol. Sci. 2017, 18, 2615. [Google Scholar] [CrossRef]
- Jang, M.; Choi, H.Y.; Kim, G.H. Phenolic compounds rich ethyl acetate fraction of Orostachys japonicus inhibits lipid accumulation by regulating reactive oxygen species generation in adipogenesis. J. Food Biochem. 2019, 43, e12939. [Google Scholar] [CrossRef]
- Lee, S.J.; Zhang, G.F.; Sung, N.J. Hypolipidemic and hypoglycemic effects of Orostachys japonicus A. Berger extracts in streptozotocin-induced diabetic rats. Nutr. Res. Pract. 2011, 5, 301–307. [Google Scholar] [CrossRef]
- Park, H.J.; Yang, H.J.; Kim, K.H.; Kim, S.H. Aqueous extract of Orostachys japonicus A. Berger exerts immunostimulatory activity in RAW 264.7 macrophages. J. Ethnopharmacol. 2015, 170, 210–217. [Google Scholar] [CrossRef]
- Zehiroglu, C.; Ozturk Sarikaya, S.B. The importance of antioxidants and place in today’s scientific and technological studies. J. Food Sci. Technol. 2019, 56, 4757–4774. [Google Scholar] [CrossRef]
- Mucha, P.; Skoczyńska, A.; Małecka, M.; Hikisz, P.; Budzisz, E. Overview of the antioxidant and anti-inflammatory activities of selected plant compounds and their metal ions complexes. Molecules 2021, 26, 4886. [Google Scholar] [CrossRef]
- Moro, C.; Palacios, I.; Lozano, M.; D’Arrigo, M.; Guillamón, E.; Villares, A.; Martínez, J.A.; García-Lafuente, A. Anti-inflammatory activity of methanolic extracts from edible mushrooms in LPS activated RAW 264.7 macrophages. Food Chem. 2012, 130, 350–355. [Google Scholar] [CrossRef]
- Liu, T.; Zhang, M.; Mukosera, G.T.; Borchardt, D.; Li, Q.; Tipple, T.E.; Ahmed, A.S.I.; Power, G.G.; Blood, A.B. l-NAME releases nitric oxide and potentiates subsequent nitroglycerin-mediated vasodilation. Redox. Biol. 2019, 26, 101238. [Google Scholar] [CrossRef]
- Safdar, N.; Sarfaraz, A.; Kazmi, Z.; Yasmin, A. Ten different brewing methods of green tea: Comparative antioxidant study. J. Appl. Biol. Biotechnol. 2016, 4, 33–40. [Google Scholar]
- Kim, S.N.; Lee, C.M.; Kim, Y.C. Comparison in antioxidant activities of water extracts of green tea, white tea and black tea. J. Investig. Cosmetol. 2012, 8, 165–169. [Google Scholar]
- Zaiter, A.; Becker, L.; Karam, M.C.; Dicko, A. Effect of particle size on antioxidant activity and catechin content of green tea powders. J. Food Sci. Technol. 2016, 53, 2025–2032. [Google Scholar] [CrossRef]
- Im, E.K.; Yang, J.C. A study on the antioxidative effect of Orostachys japonicus A. Berger ethyl acetate fraction. Korean J. Food Sci. Technol. 2021, 38, 118–125. [Google Scholar]
- Kopincová, J.; Púzserová, A.; Bernátová, I. l-NAME in the cardiovascular system—nitric oxide synthase activator? Pharmacol. Rep. 2021, 64, 511–520. [Google Scholar] [CrossRef]
- Nakabayashi, T. Isolation of astragalin and isoquercitrin from Bracken, Pteridium aquilinum. Bull. Chem. Soc. Jpn. 1955, 19, 104–109. [Google Scholar] [CrossRef]
- Jia, Q.; Wang, T.; Wang, X.; Xu, H.; Liu, Y.; Wang, Y.; Shi, Q.; Liang, Q. Astragalin suppresses inflammatory responses and bone destruction in mice with collagen-induced arthritis and in human fibroblast-like synoviocytes. Front. Pharmacol. 2019, 10, 94. [Google Scholar] [CrossRef]
- Chen, J.; Zhong, K.; Qin, S.; Jing, Y.; Liu, S.; Li, D.; Peng, C. Astragalin: A food-origin flavonoid with therapeutic effect for multiple diseases. Front. Pharmacol. 2023, 14, 1265960. [Google Scholar] [CrossRef]
- Jung, S.H.; Kim, B.J.; Lee, E.H.; Osborne, N.N. Isoquercitrin is the most effective antioxidant in the plant Thuja orientalis and able to counteract oxidative-induced damage to a transformed cell line (RGC-5 cells). Neurochem. Int. 2010, 57, 713–721. [Google Scholar] [CrossRef]
- Kim, J.S.; Lim, J.H.; Cho, S.K. Effect of antioxidant and anti-inflammatory on bioactive components of carrot (Daucus carota L.) leaves from Jeju Island. Appl. Biol. Chem. 2023, 66, 34. [Google Scholar] [CrossRef]
- Han, Y.K.; Vinh, L.B.; Nam, M.H.; Lee, K.Y. Identification of compounds using HPLC-QTOF-MS online antioxidant activity mapping from aerial parts of Ligularia stenocephala. Appl. Biol. Chem. 2023, 66, 53. [Google Scholar] [CrossRef]
- Choi, J.; Lee, H.D.; Cho, H.; Lee, C.D.; Tran, G.H.; Kim, H.; Moon, S.K.; Lee, S. Antioxidative phenolic compounds from the aerial parts of Cyperus exaltatus var. iwasakii and their HPLC analysis. Appl. Biol. Chem. 2023, 66, 61. [Google Scholar] [CrossRef]
- Kim, J.; Kim, J.H.; Bang, S.I.; Shin, H.; Cho, E.J.; Lee, S. Antioxidant activity of edible sprouts and phytosterol contents by HPLC/UV analysis. Hortic. Environ. Biotechnol. 2022, 63, 769–778. [Google Scholar] [CrossRef]
- Li, R.; Yuan, C.; Dong, C.; Shuang, S.; Choi, M.M.F. In vivo antioxidative effect of isoquercitrin on cadmium-induced oxidative damage to mouse liver and kidney. Naunyn-Schmied. Arch. Pharmacol. 2011, 383, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Yang, L. Myricitrin decreases traumatic injury of the spinal cord and exhibits antioxidant and anti-inflammatory activities in a rat model via inhibition of COX-2, TGF β1, p53 and elevation of Bcl-2/Bax signaling pathway. Mol. Med. Rep. 2017, 16, 7699–7705. [Google Scholar]
- Domitrović, R.; Rashed, K.; Cvijanović, O.; Vladimir-Knežević, S.; Škoda, M.; Višnić, A. Myricitrin exhibits antioxidant, anti-inflammatory and antifibrotic activity in carbon tetrachloride-intoxicated mice. Chem. Biol. Interact. 2015, 230, 21–29. [Google Scholar] [CrossRef] [PubMed]
- He, N.; Wang, P.; Niu, Y.; Chen, J.; Li, C.; Kang, W. Evaluation antithrombotic activity and action mechanism of myricitrin. Ind. Crops Prod. 2019, 129, 536–541. [Google Scholar] [CrossRef]
- Kim, H.; Jeon, B.; Kim, W.J.; Chung, D.K. Effect of paraprobiotic prepared from Kimchi-derived Lactobacillus plantarum K8 on skin moisturizing activity in human keratinocyte. J. Funct. Foods 2020, 75, 104244. [Google Scholar] [CrossRef]
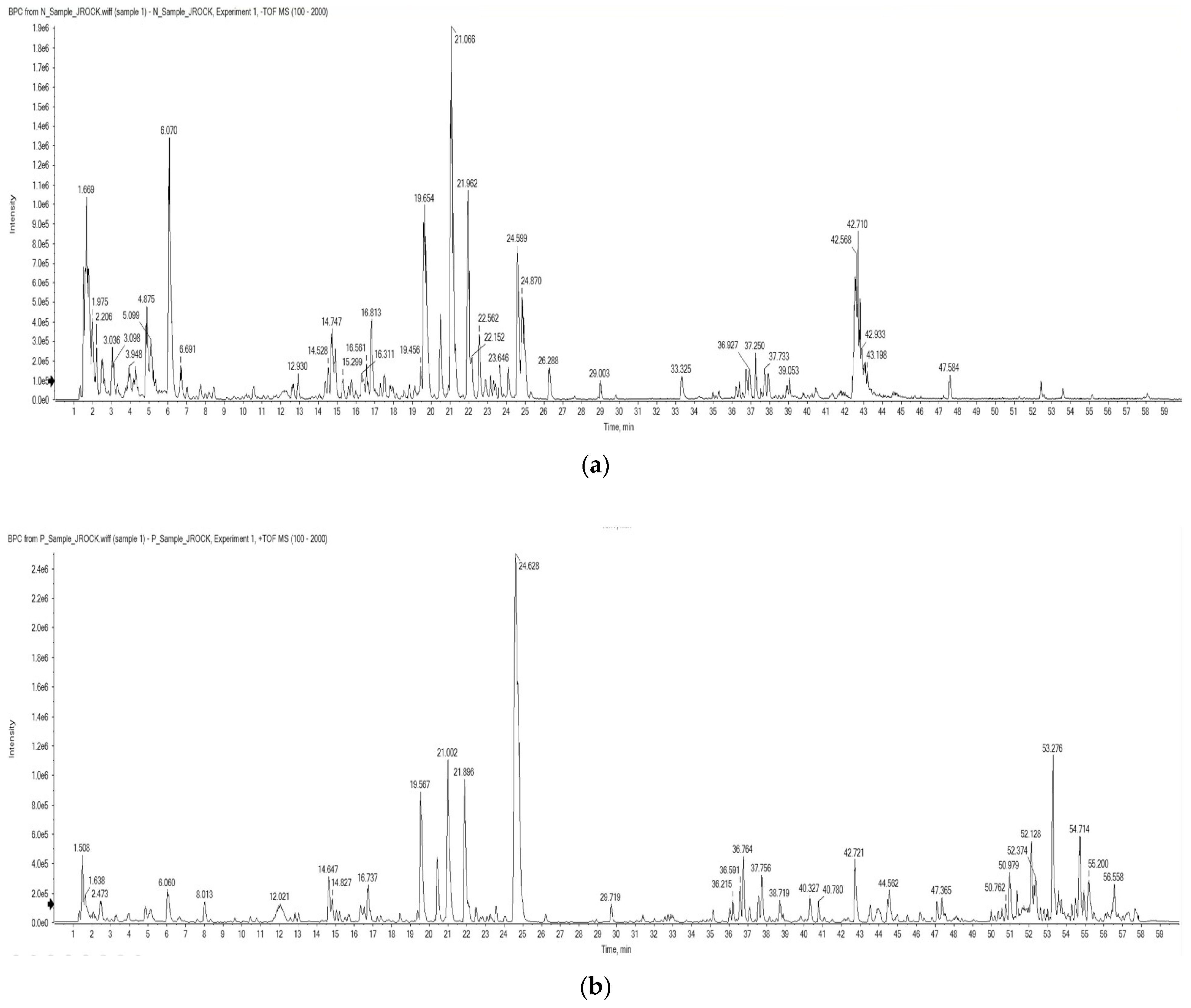
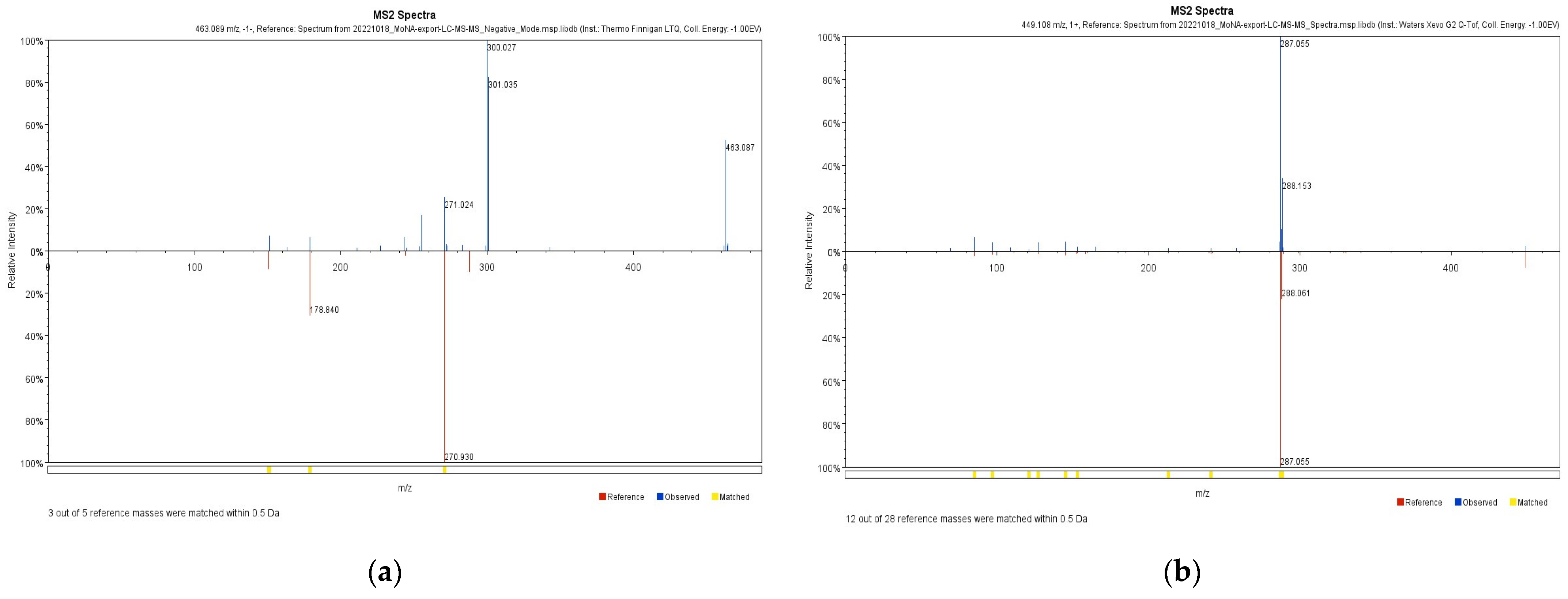



| Retention Time (min) | Molecular Weight | Proposed Compound |
|---|---|---|
| 2.51 | 332.1 | Galloyl glucose 2 |
| 5.23 | 332.1 | Gallic acid hexoside 2 |
| 12.05 | 449.1 | Cyanidijin-3-O-glucoside 1 |
| 12.60 | 356.1 | 1-O-Feruloylglucose 2 |
| 12.88 | 626.1 | Quercetin-3,4′-diglucoside 1,2 |
| 14.69 | 610.2 | Luteolin-3′,7-di-O-glucoside 1,2 |
| 14.90 | 610.2 | Luteolin-3′,7-di-O-glucoside 2 |
| 16.19 | 626.1 | Herbacetin-3,8-diglucoside 2 |
| 16.50 | 596.1 | Peltatoside 1 |
| 16.56 | 596.1 | Rhodalidin 2 |
| 16.80 | 594.2 | Kaempferol-3-neohesperidoside 1 |
| 17.11 | 464.1 | Myrtillin 2 |
| 17.26 | 596.1 | Peltatoside 1,2 |
| 19.62 | 464.1 | Myricitrin 1,2 |
| 19.82 | 610.2 | Luteolin-3′,7-di-O-glucoside 2 |
| 20.48 | 550.1 | Quercetin-3-O-malonylglucoside 2 |
| 20.91 | 506.1 | Quercetin-3-(6″-acetylglucoside) 2 |
| 21.02 | 448.1 | Astragalin 1,2 |
| 21.12 | 448.1 | Kaempferol-7-O-glucoside 2 |
| 21.94 | 534.1 | Luteolin-7-(6″-malonylglucoside) 1,2 |
| 22.52 | 432.1 | Afzelin 1,2 |
| 24.11 | 302.0 | Quercetin 2 |
| 26.29 | 286.0 | Kaempferol 2 |
| Compound | tR a | Calibration Equation b | R-Value c |
|---|---|---|---|
| 1 | 10.6 | - | - |
| 2 | 11.4 | Y = 26,898X − 13,035 | 1 |
| 3 | 14.5 | Y = 21,602X − 25,971 | 1 |
| Compound | mg/g Extract | mg/g DW | mg/g FW |
|---|---|---|---|
| 1 | tr a | tr | tr |
| 2 | 3.74 ± 0.01 | 1.18 ± 0.00 | 0.07 ± 0.00 |
| 3 | 3.19 ± 0.02 | 1.01 ± 0.01 | 0.06 ± 0.00 |
| Sample | Concentration (mg/mL) | Scavenging Activity (%) | IC50 (mg/mL) |
|---|---|---|---|
| Green tea | 0.05 | 19.32 ± 3.90 | 0.15 ± 0.01 |
| 0.10 | 36.57 ± 4.27 | ||
| 0.20 | 62.84 ± 2.68 | ||
| 0.39 | 92.86 ± 0.46 | ||
| OMY | 3.13 | 25.65 ± 3.39 | 10.49 ± 1.32 |
| 6.25 | 40.75 ± 3.13 | ||
| 12.50 | 55.22 ± 1.91 | ||
| 25.00 | 78.20 ± 2.37 | ||
| AA | 0.04 | 15.80 ± 4.07 | 0.11 ± 0.01 |
| 0.08 | 34.60 ± 1.57 | ||
| 0.12 | 56.44 ± 3.81 | ||
| 0.16 | 75.57 ± 4.33 | ||
| 0.20 | 93.75 ± 1.53 |
| Sample | Concentration (mg/mL) | Scavenging Activity (%) | IC50 (mg/mL) |
|---|---|---|---|
| Green tea | 0.05 | 10.00 ± 0.90 | 0.21 ± 0.00 |
| 0.10 | 25.30 ± 1.81 | ||
| 0.20 | 51.71 ± 1.07 | ||
| 0.39 | 84.74 ± 0.89 | ||
| OMY | 1.56 | 16.83 ± 3.00 | 10.31 ± 0.35 |
| 3.13 | 24.56 ± 0.82 | ||
| 6.25 | 36.17 ± 1.11 | ||
| 12.50 | 57.59 ± 1.59 | ||
| AA | 0.04 | 4.63 ± 1.67 | 0.17 ± 0.00 |
| 0.08 | 17.22 ± 0.41 | ||
| 0.12 | 34.37 ± 0.53 | ||
| 0.16 | 46.41 ± 3.18 | ||
| 0.20 | 63.09 ± 0.94 |
| Sample | NO Inhibition (IC50, μg/mL) |
|---|---|
| l-NAME | 41.1 ± 2.6 |
| OMY | 202.6 ± 8.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoon, N.; Yu, H.; Tran, G.H.; Ko, C.H.; Kim, H.; Yoon, M.J.; Lee, S. Profiling the Phytochemicals of Orostachys margaritifolia: Biological Activities, LC-ESI/MS, and HPLC Analyses. Pharmaceuticals 2024, 17, 290. https://doi.org/10.3390/ph17030290
Yoon N, Yu H, Tran GH, Ko CH, Kim H, Yoon MJ, Lee S. Profiling the Phytochemicals of Orostachys margaritifolia: Biological Activities, LC-ESI/MS, and HPLC Analyses. Pharmaceuticals. 2024; 17(3):290. https://doi.org/10.3390/ph17030290
Chicago/Turabian StyleYoon, Nari, Hyeonjun Yu, Gia Han Tran, Chung Ho Ko, Hoon Kim, Mi Jeong Yoon, and Sanghyun Lee. 2024. "Profiling the Phytochemicals of Orostachys margaritifolia: Biological Activities, LC-ESI/MS, and HPLC Analyses" Pharmaceuticals 17, no. 3: 290. https://doi.org/10.3390/ph17030290
APA StyleYoon, N., Yu, H., Tran, G. H., Ko, C. H., Kim, H., Yoon, M. J., & Lee, S. (2024). Profiling the Phytochemicals of Orostachys margaritifolia: Biological Activities, LC-ESI/MS, and HPLC Analyses. Pharmaceuticals, 17(3), 290. https://doi.org/10.3390/ph17030290







