Chlojaponilactone B Attenuates THP-1 Macrophage Pyroptosis by Inhibiting the TLR/MyD88/NF-κB Pathway
Abstract
1. Introduction
2. Results
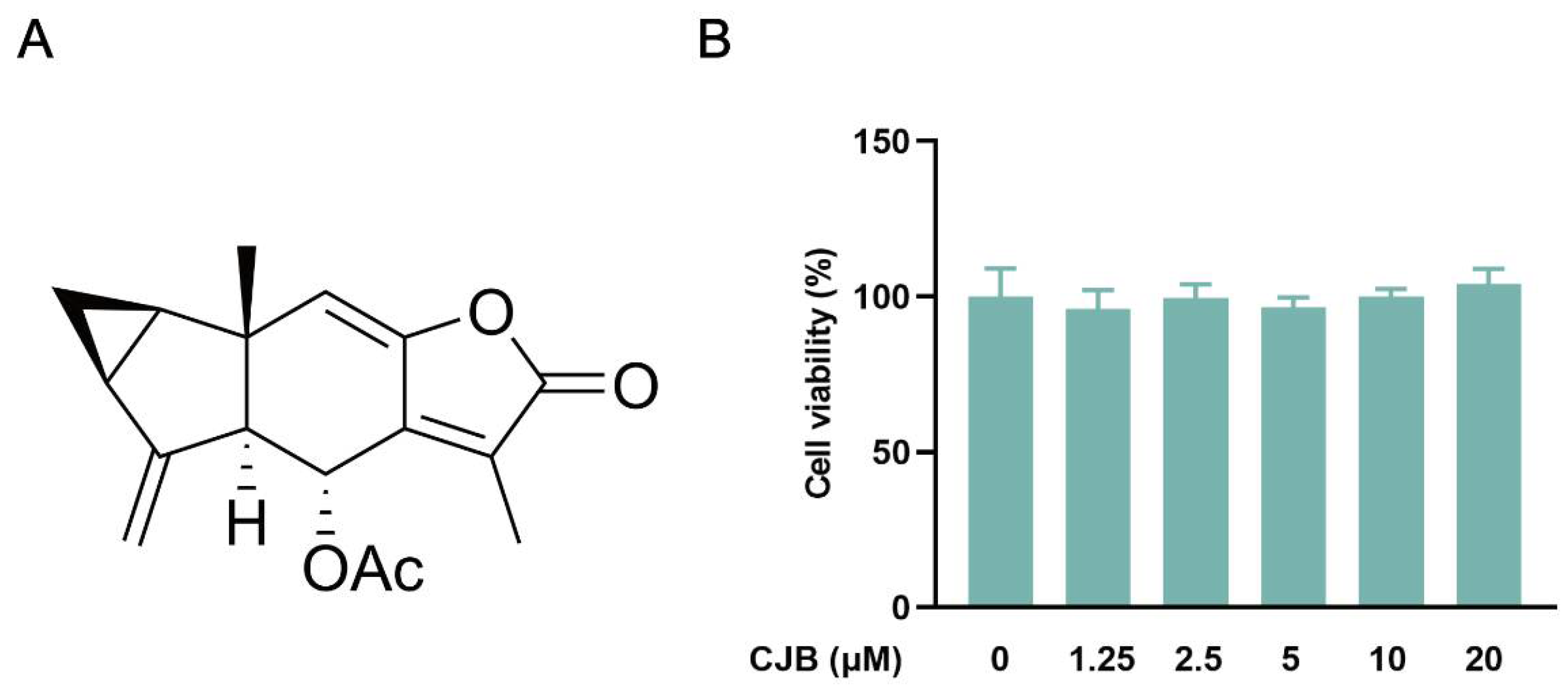
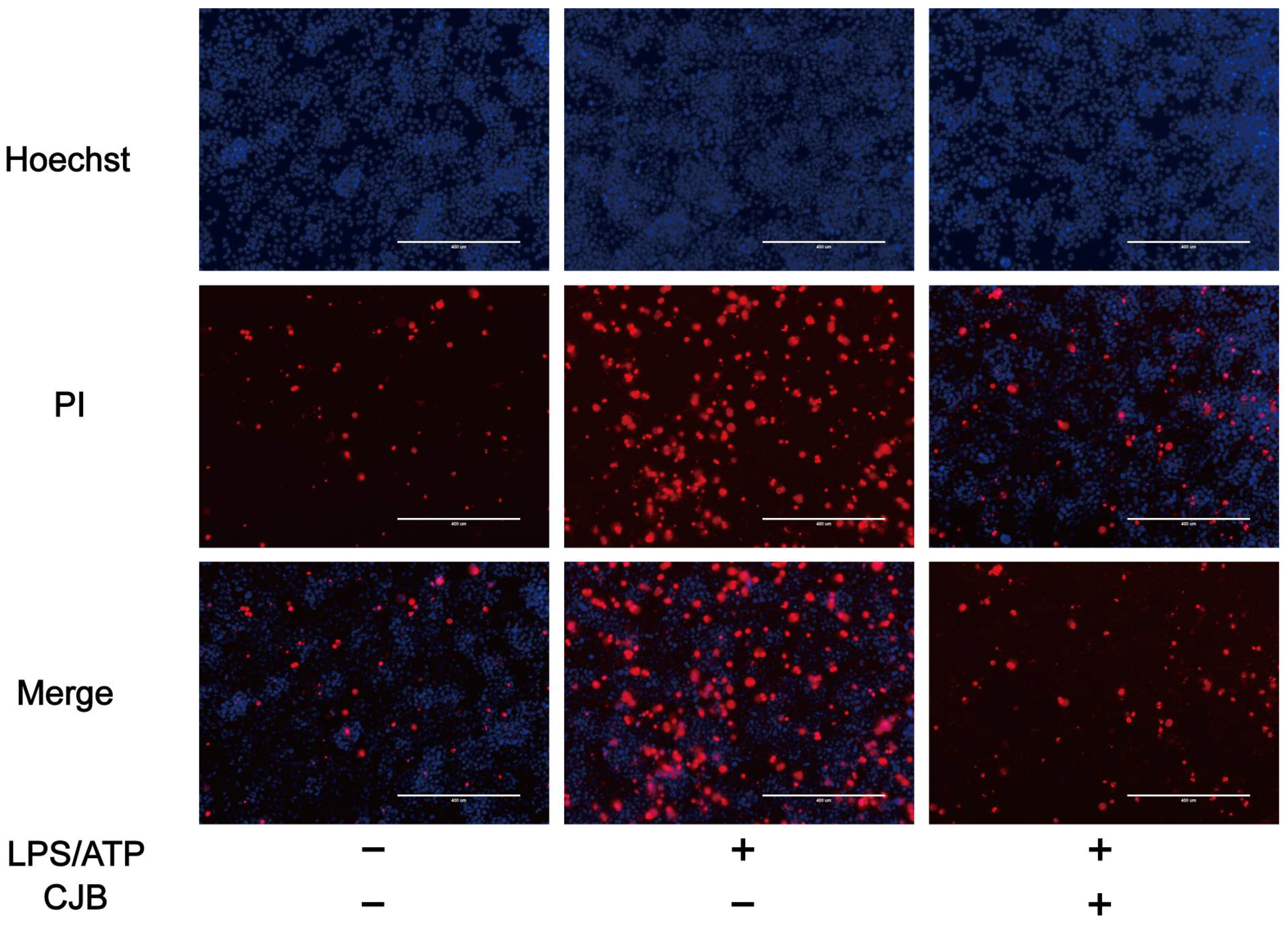
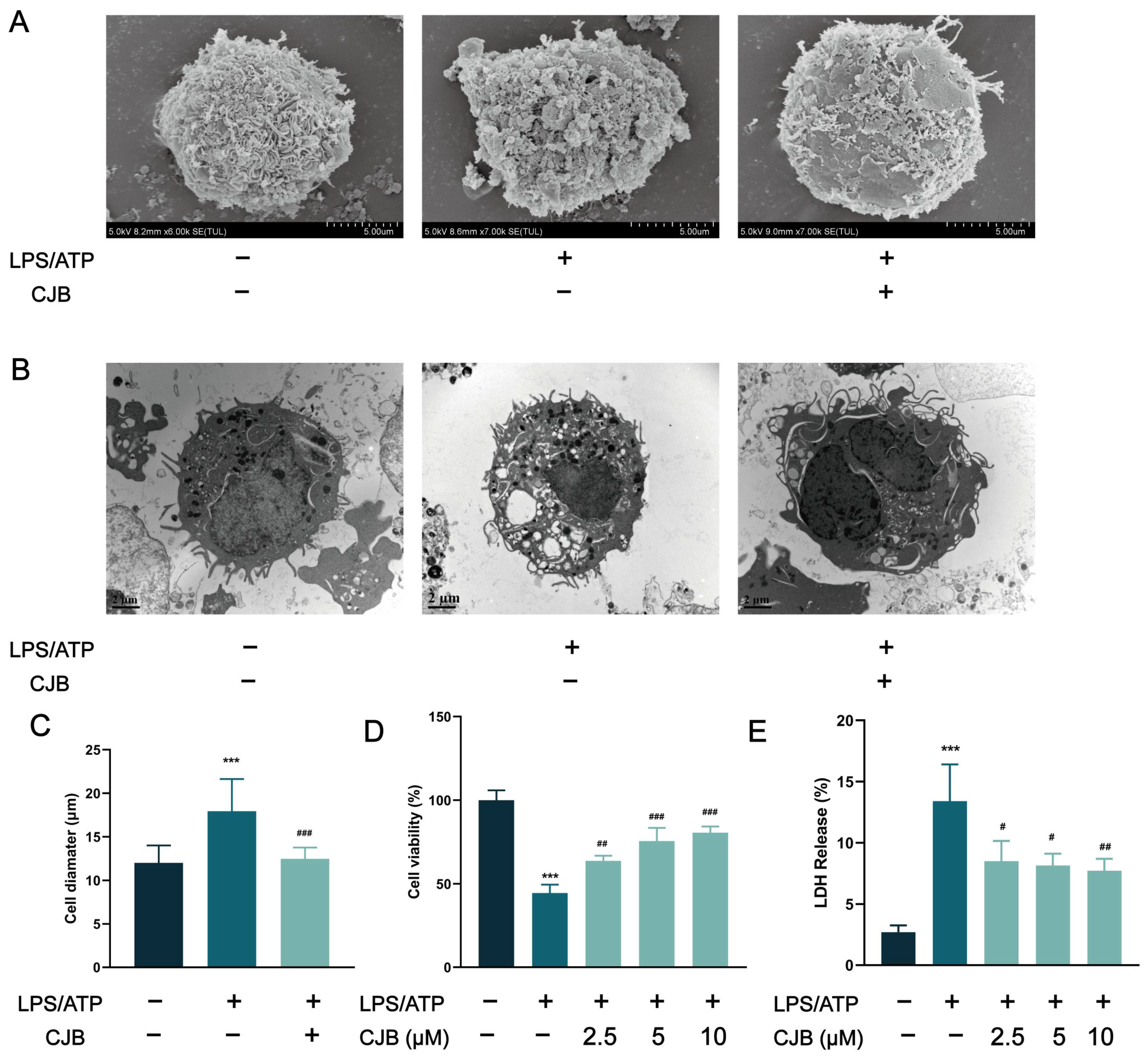
2.1. CJB Suppressed the Pyroptosis of THP-1 Macrophages
2.2. CJB Suppressed the NLRP3 Activation Stimulated by LPS/ATP
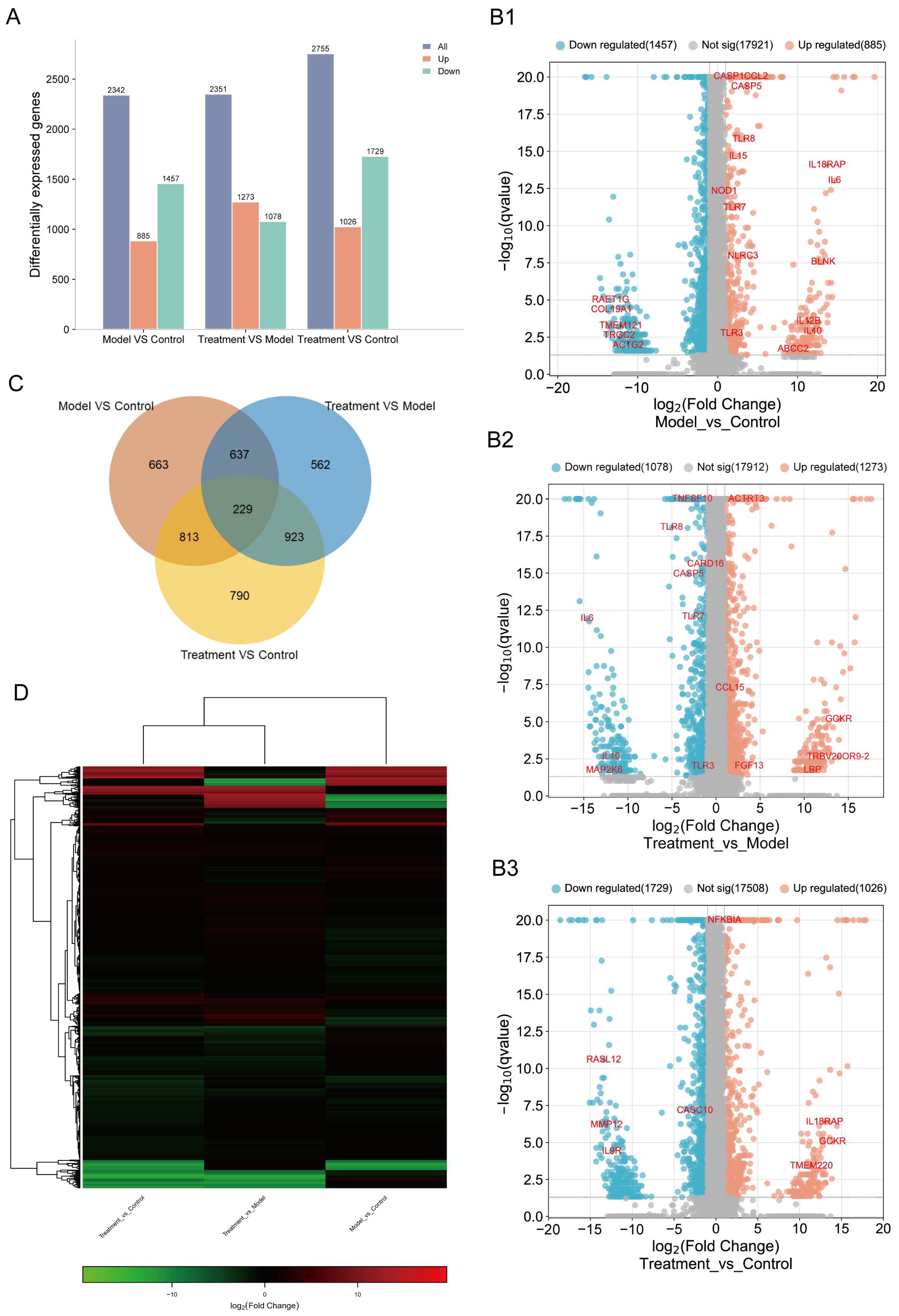
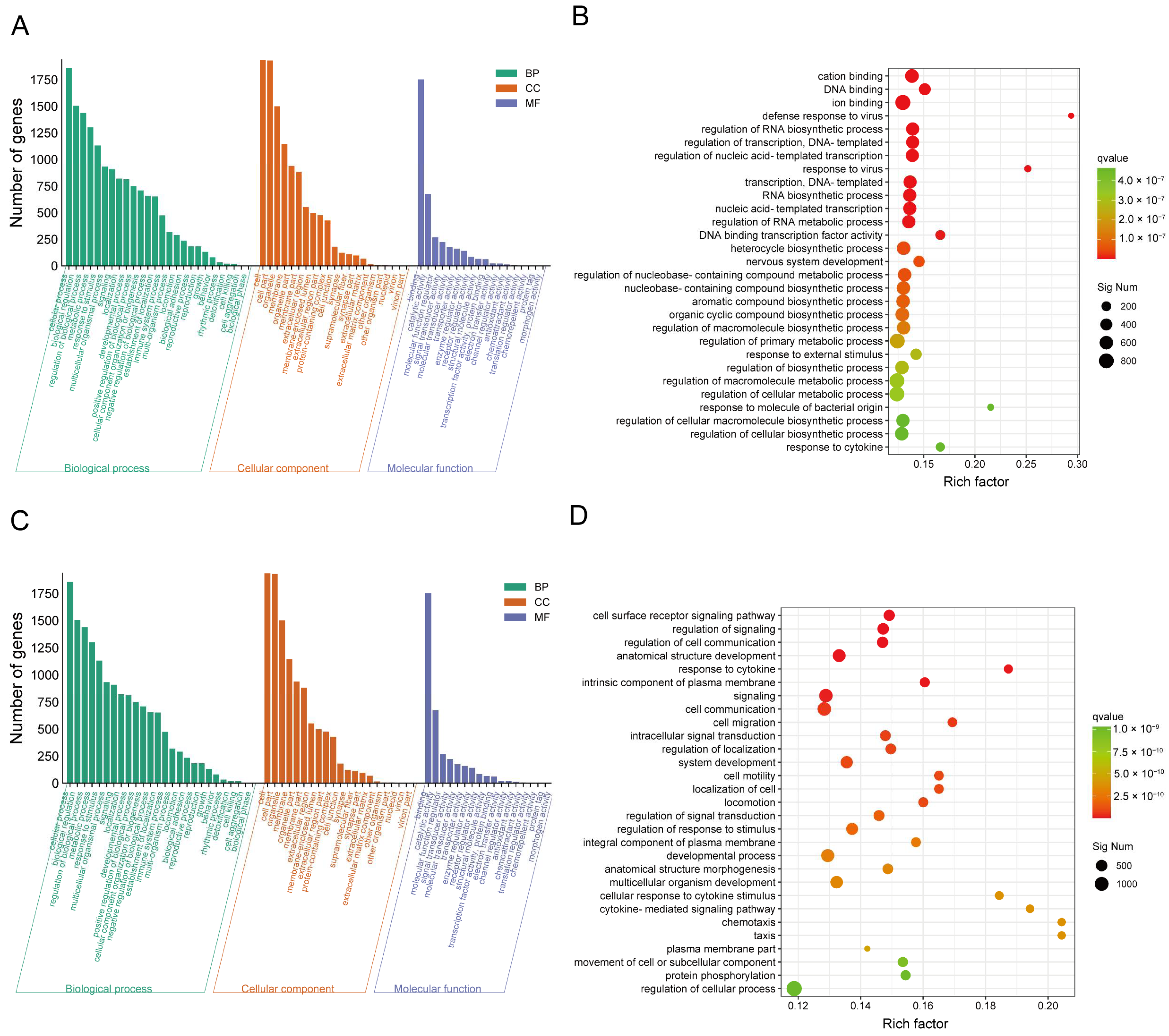
2.3. CJB Interferes with the LPS/ATP-Induced Transcriptome Sequencing of THP-1 Macrophages
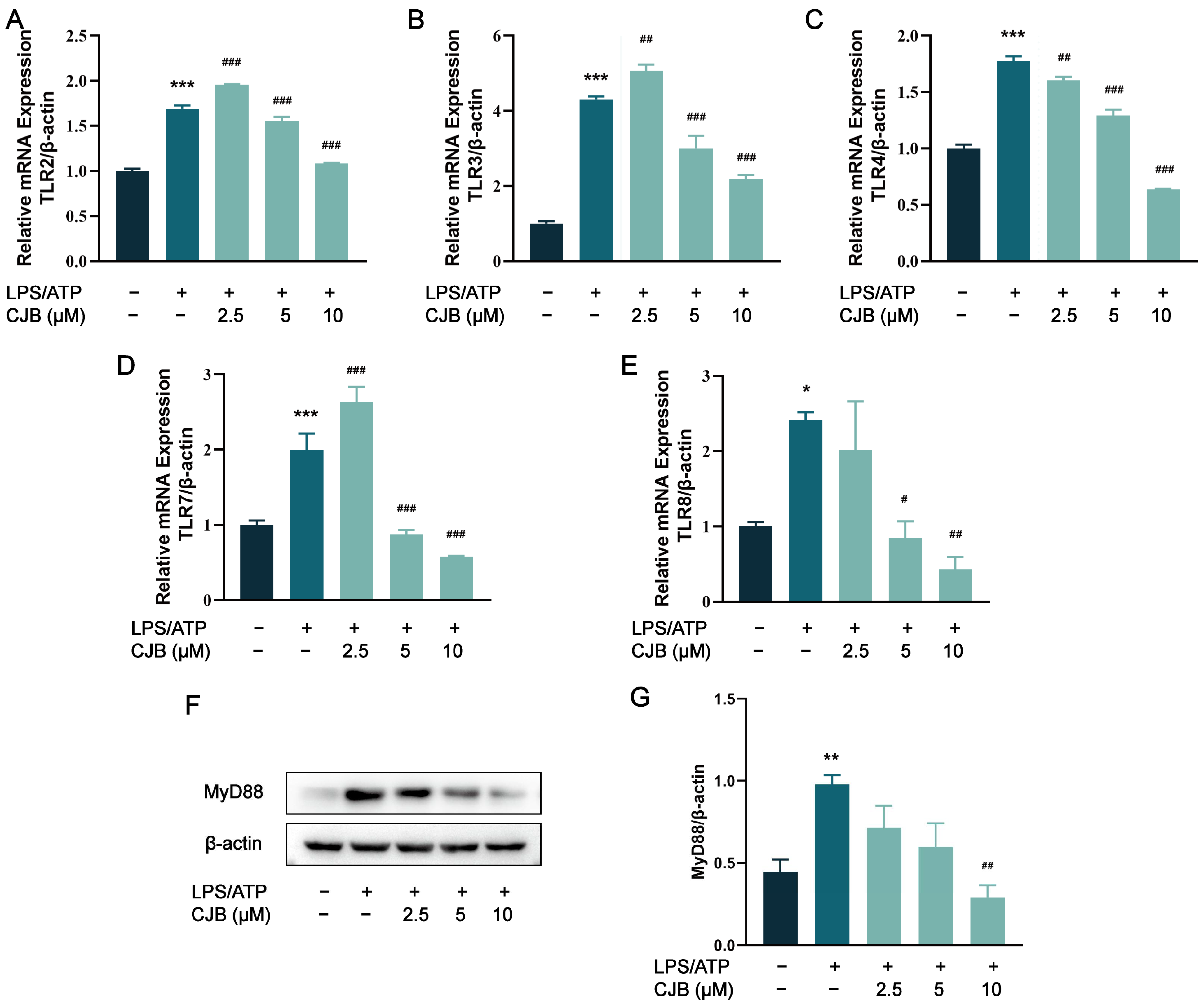
2.4. DEG Profiles and Functional Prediction Highlighted That CJB Suppressed LPS/ATP-Induced THP-1 Macrophages’ Pyroptosis through the TLR/NF-κB Signaling Pathway
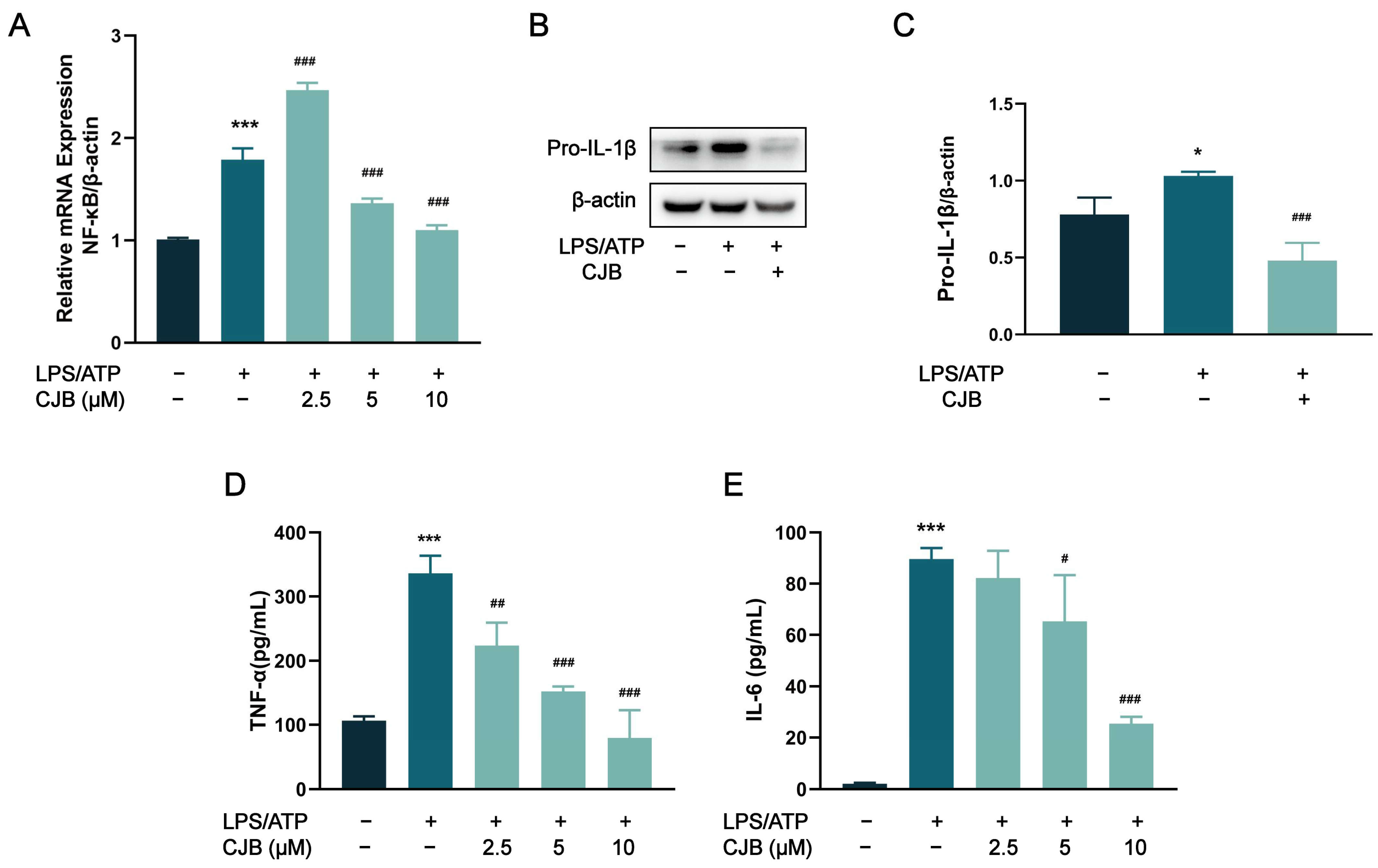
2.5. CJB Suppressed the TLR/NF-κB Signaling Pathway
3. Discussion
4. Materials and Methods
4.1. Reagents and Antibodies
4.2. Material
4.3. Cell Culture and Induction
4.4. Cell Viability Assay
4.5. LDH Assay
4.6. Propidium Iodide Staining
4.7. Western Blotting
4.8. ELISA Assay
4.9. NF-κB p65 Nuclear Translocation
4.10. RNA-Seq Analysis
4.11. qRT-PCR
4.12. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Frantz, S.; Ducharme, A.; Sawyer, D.; Rohde, L.E.; Kobzik, L.; Fukazawa, R.; Tracey, D.; Allen, H.; Lee, R.T.; Kelly, R.A. Targeted deletion of caspase-1 reduces early mortality and left ventricular dilatation following myocardial infarction. J. Mol. Cell. Cardiol. 2003, 35, 685–694. [Google Scholar] [CrossRef]
- Fink, S.L.; Cookson, B.T. Caspase-1-dependent pore formation during pyroptosis leads to osmotic lysis of infected host macrophages. Cell. Microbiol. 2006, 8, 1812–1825. [Google Scholar] [CrossRef]
- Sun, G.W.; Lu, J.; Pervaiz, S.; Cao, W.P.; Gan, Y.H. Caspase-1 dependent macrophage death induced by Burkholderia pseudomallei. Cell. Microbiol. 2005, 7, 1447–1458. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Xu, W.; Zhou, R. NLRP3 inflammasome activation and cell death. Cell. Mol. Immunol. 2021, 18, 2114–2127. [Google Scholar] [CrossRef]
- Shi, J.; Zhao, Y.; Wang, K.; Shi, X.; Wang, Y.; Huang, H.; Zhuang, Y.; Cai, T.; Wang, F.; Shao, F. Cleavage of GSDMD by inflammatory caspases determines pyroptotic cell death. Nature 2015, 526, 660–665. [Google Scholar] [CrossRef]
- Xia, S.; Zhang, Z.; Magupalli, V.G.; Pablo, J.L.; Dong, Y.; Vora, S.M.; Wang, L.; Fu, T.M.; Jacobson, M.P.; Greka, A.; et al. Gasdermin D pore structure reveals preferential release of mature interleukin-1. Nature 2021, 593, 607–611. [Google Scholar] [CrossRef]
- Baker, P.J.; Boucher, D.; Bierschenk, D.; Tebartz, C.; Whitney, P.G.; D’Silva, D.B.; Tanzer, M.C.; Monteleone, M.; Robertson, A.A.; Cooper, M.A.; et al. NLRP3 inflammasome activation downstream of cytoplasmic LPS recognition by both caspase-4 and caspase-5. Eur. J. Immunol. 2015, 45, 2918–2926. [Google Scholar] [CrossRef] [PubMed]
- Aglietti, R.A.; Estevez, A.; Gupta, A.; Ramirez, M.G.; Liu, P.S.; Kayagaki, N.; Ciferri, C.; Dixit, V.M.; Dueber, E.C. GsdmD p30 elicited by caspase-11 during pyroptosis forms pores in membranes. Proc. Natl. Acad. Sci. USA 2016, 113, 7858–7863. [Google Scholar] [CrossRef]
- Fang, Y.; Tian, S.; Pan, Y.; Li, W.; Wang, Q.; Tang, Y.; Yu, T.; Wu, X.; Shi, Y.; Ma, P.; et al. Pyroptosis: A new frontier in cancer. Biomed. Pharmacother. 2020, 121, 109595. [Google Scholar] [CrossRef] [PubMed]
- Hao, K.; Jiang, W.; Zhou, M.; Li, H.; Chen, Y.; Jiang, F.; Hu, Q. Targeting BRD4 prevents acute gouty arthritis by regulating pyroptosis. Int. J. Biol. Sci. 2020, 16, 3163–3173. [Google Scholar] [CrossRef]
- Zhaolin, Z.; Guohua, L.; Shiyuan, W.; Zuo, W. Role of pyroptosis in cardiovascular disease. Cell Prolif. 2019, 52, e12563. [Google Scholar] [CrossRef]
- Wang, Y.; Fang, C.; Xu, L.; Yang, B.; Song, E.; Song, Y. Polybrominated Diphenyl Ether Quinone Exposure Induces Atherosclerosis Progression via CD36-Mediated Lipid Accumulation, NLRP3 Inflammasome Activation, and Pyroptosis. Chem. Res. Toxicol. 2021, 34, 2125–2134. [Google Scholar] [CrossRef]
- Sharma, A.; Choi, J.S.Y.; Stefanovic, N.; Al-Sharea, A.; Simpson, D.S.; Mukhamedova, N.; Jandeleit-Dahm, K.; Murphy, A.J.; Sviridov, D.; Vince, J.E.; et al. Specific NLRP3 Inhibition Protects against Diabetes-Associated Atherosclerosis. Diabetes 2021, 70, 772–787. [Google Scholar] [CrossRef]
- Xu, S.; Chen, H.; Ni, H.; Dai, Q. Targeting HDAC6 attenuates nicotine-induced macrophage pyroptosis via NF-κB/NLRP3 pathway. Atherosclerosis 2021, 317, 1–9. [Google Scholar] [CrossRef]
- Wei, Y.Y.; Lan, B.D.; Zheng, T.; Yang, L.; Zhang, X.X.; Cheng, L.L.; Tuerhongjiang, G.; Yuan, Z.Y.; Wu, Y. GSDME-mediated pyroptosis promotes the progression and associated inflammation of atherosclerosis. Nat. Commun. 2023, 14, 929. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Wang, H.; Yu, Q.Q.; Ge, J.R.; Zhang, X.Z.; Mei, D.; Liang, F.Q.; Cai, X.Y.; Zhu, Y.; Shu, J.L.; et al. The monomer derivative of paeoniflorin inhibits macrophage pyroptosis via regulating TLR4/NLRP3/GSDMD signaling pathway in adjuvant arthritis rats. Int. Immunopharmacol. 2021, 101, 12. [Google Scholar] [CrossRef] [PubMed]
- Song, M.Y.; Wang, J.X.; Sun, Y.L.; Pang, J.L.; Li, X.N.; Liu, Y.; Zhou, Y.T.; Yang, P.R.; Fan, T.H.; Liu, Y.; et al. Inhibition of gasdermin D-dependent pyroptosis attenuates the progression of silica-induced pulmonary inflammation and fibrosis. Acta Pharm. Sin. B 2022, 12, 1213–1224. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Li, X.-H.; Zheng, X.-F.; Sun, C.-L.; Liu, H.-Y. Chlojaponilactones B—E, Four New Lindenane Sesquiterpenoid Lactones from Chloranthus japonicus. Helv. Chim. Acta 2013, 96, 1386–1391. [Google Scholar] [CrossRef]
- Ye, S.; Zheng, Q.; Zhou, Y.; Bai, B.; Yang, D.; Zhao, Z. Chlojaponilactone B Attenuates Lipopolysaccharide-Induced Inflammatory Responses by Suppressing TLR4-Mediated ROS Generation and NF-kappa B Signaling Pathway. Molecules 2019, 24, 3731. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.-J.; Guo, Y.-Q.; Yang, D.-P.; Xue, X.; Liu, Q.; Zhu, L.-P.; Yin, S.; Zhao, Z.-M. Chlojaponilactone B from Chloranthus japonicus: Suppression of Inflammatory Responses via Inhibition of the NF-kappa B Signaling Pathway. J. Nat. Prod. 2016, 79, 2257–2263. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Zhou, Z.; Wu, X.; Wang, Y.; Zuo, L.; Zheng, R.; Liu, Y.; Liu, Z.; Lai, X.; Zhou, L.; et al. Vitamin K3 Suppresses Pyroptosis in THP-1 Cells through Inhibition of NF-κB and JNK Signaling Pathways. Biol. Pharm. Bull. 2023, 46, 52–60. [Google Scholar] [CrossRef]
- Wang, X.; Wu, F.P.; Huang, Y.R.; Li, H.D.; Cao, X.Y.; You, Y.; Meng, Z.F.; Sun, K.Y.; Shen, X.Y. Matrine suppresses NLRP3 inflammasome activation via regulating PTPN2/JNK/SREBP2 pathway in sepsis. Phytomed. Int. J. Phytother. Phytopharm. 2023, 109, 154574. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, S.; Vargas, J.; Hoffmann, A. Signaling via the NFκB system. Wiley Interdiscip. Rev. Syst. Biol. Med. 2016, 8, 227–241. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Jia, L.; Shen, J.; Wang, Y.; Fu, Z.; Su, S.A.; Cai, Z.; Wang, J.A.; Xiang, M. Cathepsin B aggravates coxsackievirus B3-induced myocarditis through activating the inflammasome and promoting pyroptosis. PLoS Pathog. 2018, 14, e1006872. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.; Sun, X.; Liu, S.; Tang, Y.; Shi, Y.; Bai, Y.; Wang, Y.; Yang, Q.; Yang, Q.; Jiang, W.; et al. Caspase-11-Gasdermin D-Mediated Pyroptosis Is Involved in the Pathogenesis of Atherosclerosis. Front. Pharmacol. 2021, 12, 657486. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Su, H.; Zhang, Y.; Liu, Z.; Kong, J. Cholecalciterol cholesterol emulsion attenuates experimental autoimmune myocarditis in mice via inhibition of the pyroptosis signaling pathway. Biochem. Biophys. Res. Commun. 2017, 493, 422–428. [Google Scholar] [CrossRef] [PubMed]
- Ge, G.; Bai, J.; Wang, Q.; Liang, X.; Tao, H.; Chen, H.; Wei, M.; Niu, J.; Yang, H.; Xu, Y.; et al. Punicalagin ameliorates collagen-induced arthritis by downregulating M1 macrophage and pyroptosis via NF-κB signaling pathway. Sci. China Life Sci. 2022, 65, 588–603. [Google Scholar] [CrossRef]
- Zhou, Q.; Li, T.; Fang, G.; Pang, Y.; Wang, X. Bioactive Molecules against Rheumatoid Arthritis by Suppressing Pyroptosis. Pharmaceuticals 2023, 16, 952. [Google Scholar] [CrossRef]
- Hu, Y.; Gui, Z.; Zhou, Y.; Xia, L.; Lin, K.; Xu, Y. Quercetin alleviates rat osteoarthritis by inhibiting inflammation and apoptosis of chondrocytes, modulating synovial macrophages polarization to M2 macrophages. Free Radic. Biol. Med. 2019, 145, 146–160. [Google Scholar] [CrossRef]
- Wu, X.Y.; Li, K.T.; Yang, H.X.; Yang, B.; Lu, X.; Zhao, L.D.; Fei, Y.Y.; Chen, H.; Wang, L.; Li, J.; et al. Complement C1q synergizes with PTX3 in promoting NLRP3 inflammasome over-activation and pyroptosis in rheumatoid arthritis. J. Autoimmun. 2020, 106, 102336. [Google Scholar] [CrossRef]
- Yang, S.; Chen, S. Chloranthus japonicus: Activating blood to remove dampness. Plant J. 2001, 1, 18. [Google Scholar]
- Ye, S.; Wen, Q.; Zhu, L.; Qian, C.; Yang, D.; Zhao, Z. Neuroprotective Effects of a New Derivative of Chlojaponilactone B against Oxidative Damaged Induced by Hydrogen Peroxide in PC12 Cells. Molecules 2022, 27, 6049. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Luo, X.; Feng, Y.; Fang, S.; Tian, J.; Yu, B.; Li, J. Luteolin prevents THP-1 macrophage pyroptosis by suppressing ROS production via Nrf2 activation. Chem. Biol. Interact. 2021, 345, 109573. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Bao, X.; Weng, X.; Bai, X.; Feng, Y.; Huang, J.; Liu, S.; Jia, H.; Yu, B. The protective effect of quercetin on macrophage pyroptosis via TLR2/Myd88/NF-κB and ROS/AMPK pathway. Life Sci. 2022, 291, 120064. [Google Scholar] [CrossRef] [PubMed]
- Bergsbaken, T.; Fink, S.L.; Cookson, B.T. Pyroptosis: Host cell death and inflammation. Nat. Rev. Microbiol. 2009, 7, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Kelley, N.; Jeltema, D.; Duan, Y.; He, Y. The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation. Int. J. Mol. Sci. 2019, 20, 3328. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Chai, Y.; Fu, Y.; Wang, Y.; Zhang, Y.; Zhang, X.; Zhu, L.; Miao, M.; Yan, T. Salidroside Ameliorates Alzheimer’s Disease by Targeting NLRP3 Inflammasome-Mediated Pyroptosis. Front. Aging Neurosci. 2021, 13, 809433. [Google Scholar] [CrossRef]
- Ding, S.; Xu, S.; Ma, Y.; Liu, G.; Jang, H.; Fang, J. Modulatory Mechanisms of the NLRP3 Inflammasomes in Diabetes. Biomolecules 2019, 9, 850. [Google Scholar] [CrossRef]
- Shao, B.Z.; Xu, H.Y.; Zhao, Y.C.; Zheng, X.R.; Wang, F.; Zhao, G.R. NLRP3 Inflammasome in Atherosclerosis: Putting Out the Fire of Inflammation. Inflammation 2023, 46, 35–46. [Google Scholar] [CrossRef]
- Sharma, B.R.; Kanneganti, T.D. NLRP3 inflammasome in cancer and metabolic diseases. Nat. Immunol. 2021, 22, 550–559. [Google Scholar] [CrossRef]
- Mangan, M.S.J.; Olhava, E.J.; Roush, W.R.; Seidel, H.M.; Glick, G.D.; Latz, E. Targeting the NLRP3 inflammasome in inflammatory diseases. Nat. Rev. Drug Discov. 2018, 17, 588–606. [Google Scholar] [CrossRef]
- Ramsey, I.S.; Delling, M.; Clapham, D.E. An introduction to TRP channels. Annu. Rev. Physiol. 2006, 68, 619–647. [Google Scholar] [CrossRef]
- Parenti, A.; De Logu, F.; Geppetti, P.; Benemei, S. What is the evidence for the role of TRP channels in inflammatory and immune cells? Br. J. Pharmacol. 2016, 173, 953–969. [Google Scholar] [CrossRef]
- Miller, B.A.; Zhang, W. TRP channels as mediators of oxidative stress. Adv. Exp. Med. Biol. 2011, 704, 531–544. [Google Scholar] [CrossRef]
- El Karim, I.; McCrudden, M.T.; Linden, G.J.; Abdullah, H.; Curtis, T.M.; McGahon, M.; About, I.; Irwin, C.; Lundy, F.T. TNF-α-induced p38MAPK activation regulates TRPA1 and TRPV4 activity in odontoblast-like cells. Am. J. Pathol. 2015, 185, 2994–3002. [Google Scholar] [CrossRef] [PubMed]
- Kochukov, M.Y.; McNearney, T.A.; Yin, H.; Zhang, L.; Ma, F.; Ponomareva, L.; Abshire, S.; Westlund, K.N. Tumor necrosis factor-alpha (TNF-alpha) enhances functional thermal and chemical responses of TRP cation channels in human synoviocytes. Mol. Pain 2009, 5, 49. [Google Scholar] [CrossRef]
- Lim, K.H.; Staudt, L.M. Toll-like receptor signaling. Cold Spring Harb. Perspect. Biol. 2013, 5, a011247. [Google Scholar] [CrossRef]
- Duan, T.; Du, Y.; Xing, C.; Wang, H.Y.; Wang, R.F. Toll-Like Receptor Signaling and Its Role in Cell-Mediated Immunity. Front. Immunol. 2022, 13, 812774. [Google Scholar] [CrossRef] [PubMed]
- Kinra, M.; Nampoothiri, M.; Arora, D.; Mudgal, J. Reviewing the importance of TLR-NLRP3-pyroptosis pathway and mechanism of experimental NLRP3 inflammasome inhibitors. Scand. J. Immunol. 2022, 95, e13124. [Google Scholar] [CrossRef]
- Chen, Y.; Lin, J.; Zhao, Y.; Ma, X.; Yi, H. Toll-like receptor 3 (TLR3) regulation mechanisms and roles in antiviral innate immune responses. J. Zhejiang Univ. Sci. B 2021, 22, 609–632. [Google Scholar] [CrossRef]
- Lu, J.; Zhong, X.; Guo, C.; Tang, L.; Yu, N.; Peng, C.; Ding, Y.; Bao, X.; Zhou, J.; Shi, Y. TLR7-MyD88-DC-CXCL16 axis results neutrophil activation to elicit inflammatory response in pustular psoriasis. Cell Death Dis. 2023, 14, 315. [Google Scholar] [CrossRef]
- Simón-Fuentes, M.; Herrero, C.; Acero-Riaguas, L.; Nieto, C.; Lasala, F.; Labiod, N.; Luczkowiak, J.; Alonso, B.; Delgado, R.; Colmenares, M.; et al. TLR7 Activation in M-CSF-Dependent Monocyte-Derived Human Macrophages Potentiates Inflammatory Responses and Prompts Neutrophil Recruitment. J. Innate Immun. 2023, 15, 517–530. [Google Scholar] [CrossRef]
- Chamberlain, N.D.; Kim, S.J.; Vila, O.M.; Volin, M.V.; Volkov, S.; Pope, R.M.; Arami, S.; Mandelin, A.M., 2nd; Shahrara, S. Ligation of TLR7 by rheumatoid arthritis synovial fluid single strand RNA induces transcription of TNFα in monocytes. Ann. Rheum. Dis. 2013, 72, 418–426. [Google Scholar] [CrossRef]
- Martínez-Espinoza, I.; Guerrero-Plata, A. The Relevance of TLR8 in Viral Infections. Pathogens 2022, 11, 134. [Google Scholar] [CrossRef]
- Yang, W.; Sun, X.; Liu, S.; Xu, Y.; Li, Y.; Huang, X.; Liu, K.; Mao, L.; Min, S.; Liu, L.; et al. TLR8 agonist Motolimod-induced inflammatory death for treatment of acute myeloid leukemia. Biomed. Pharmacother. 2023, 163, 114759. [Google Scholar] [CrossRef]
- Ma, Y.; Haynes, R.L.; Sidman, R.L.; Vartanian, T. TLR8: An innate immune receptor in brain, neurons and axons. Cell Cycle 2007, 6, 2859–2868. [Google Scholar] [CrossRef]
- Forsbach, A.; Nemorin, J.G.; Montino, C.; Müller, C.; Samulowitz, U.; Vicari, A.P.; Jurk, M.; Mutwiri, G.K.; Krieg, A.M.; Lipford, G.B.; et al. Identification of RNA sequence motifs stimulating sequence-specific TLR8-dependent immune responses. J. Immunol. 2008, 180, 3729–3738. [Google Scholar] [CrossRef]
- Hu, W.; Spaink, H.P. The Role of TLR2 in Infectious Diseases Caused by Mycobacteria: From Cell Biology to Therapeutic Target. Biology 2022, 11, 246. [Google Scholar] [CrossRef]
- Zhang, Y.; Liang, X.; Bao, X.; Xiao, W.; Chen, G. Toll-like receptor 4 (TLR4) inhibitors: Current research and prospective. Eur. J. Med. Chem. 2022, 235, 114291. [Google Scholar] [CrossRef]
- Dong, Q.; Li, Y.; Chen, J.; Wang, N. Azilsartan Suppressed LPS-Induced Inflammation in U937 Macrophages through Suppressing Oxidative Stress and Inhibiting the TLR2/MyD88 Signal Pathway. ACS Omega 2021, 6, 113–118. [Google Scholar] [CrossRef]
- Chen, S.N.; Tan, Y.; Xiao, X.C.; Li, Q.; Wu, Q.; Peng, Y.Y.; Ren, J.; Dong, M.L. Deletion of TLR4 attenuates lipopolysaccharide-induced acute liver injury by inhibiting inflammation and apoptosis. Acta Pharmacol. Sin. 2021, 42, 1610–1619. [Google Scholar] [CrossRef]
- Takeda, K.; Akira, S. TLR signaling pathways. Semin. Immunol. 2004, 16, 3–9. [Google Scholar] [CrossRef]
- Qian, C.; Cao, X. Regulation of Toll-like receptor signaling pathways in innate immune responses. Ann. N. Y. Acad. Sci. 2013, 1283, 67–74. [Google Scholar] [CrossRef]
- Laird, M.H.; Rhee, S.H.; Perkins, D.J.; Medvedev, A.E.; Piao, W.; Fenton, M.J.; Vogel, S.N. TLR4/MyD88/PI3K interactions regulate TLR4 signaling. J. Leukoc. Biol. 2009, 85, 966–977. [Google Scholar] [CrossRef]
- Deguine, J.; Barton, G.M. MyD88: A central player in innate immune signaling. F1000prime Rep. 2014, 6, 97. [Google Scholar] [CrossRef]
- Place, D.E.; Kanneganti, T.D. Recent advances in inflammasome biology. Curr. Opin. Immunol. 2018, 50, 32–38. [Google Scholar] [CrossRef]
- Swanson, K.V.; Deng, M.; Ting, J.P. The NLRP3 inflammasome: Molecular activation and regulation to therapeutics. Nat. Rev. Immunol. 2019, 19, 477–489. [Google Scholar] [CrossRef] [PubMed]
- Hanada, T.; Yoshimura, A. Regulation of cytokine signaling and inflammation. Cytokine Growth Factor Rev. 2002, 13, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Umezawa, K.; Chaicharoenpong, C. Molecular design and biological activities of NF-kappaB inhibitors. Mol. Cells 2002, 14, 163–167. [Google Scholar] [CrossRef]
- Yin, K.; Sun, X.; Zheng, Y.; Zhang, W.; Lin, H. Bisphenol A exacerbates selenium deficiency-induced pyroptosis via the NF-κB/NLRP3/Caspase-1 pathway in chicken trachea. Comp. Biochem. Physiol. Toxicol. Pharmacol. CBP 2023, 263, 109488. [Google Scholar] [CrossRef]
- García, J.A.; Volt, H.; Venegas, C.; Doerrier, C.; Escames, G.; López, L.C.; Acuña-Castroviejo, D. Disruption of the NF-κB/NLRP3 connection by melatonin requires retinoid-related orphan receptor-α and blocks the septic response in mice. FASEB J. 2015, 29, 3863–3875. [Google Scholar] [CrossRef] [PubMed]
- Shen, A.; Kim, H.J.; Oh, G.S.; Lee, S.B.; Lee, S.H.; Pandit, A.; Khadka, D.; Choe, S.K.; Kwak, S.C.; Yang, S.H.; et al. NAD(+) augmentation ameliorates acute pancreatitis through regulation of inflammasome signalling. Sci. Rep. 2017, 7, 3006. [Google Scholar] [CrossRef] [PubMed]
- Gilmore, T.D. Introduction to NF-kappaB: Players, pathways, perspectives. Oncogene 2006, 25, 6680–6684. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb. Perspect. Biol. 2009, 1, a001651. [Google Scholar] [CrossRef] [PubMed]
- Oeckinghaus, A.; Ghosh, S. The NF-kappaB family of transcription factors and its regulation. Cold Spring Harb. Perspect. Biol. 2009, 1, a000034. [Google Scholar] [CrossRef]
- Liu, X.; Lin, Z.; Yin, X. Pellino2 accelerate inflammation and pyroptosis via the ubiquitination and activation of NLRP3 inflammation in model of pediatric pneumonia. Int. Immunopharmacol. 2022, 110, 108993. [Google Scholar] [CrossRef]



| Gene Symbol | Primer Sequences |
|---|---|
| β-actin | F 5′-CACGAAACTACCTTCAACTCC-3′ R 5′-CATACTCCTGCTTGCTGATC-3′ |
| TLR2 | F 5′-TCCGTCTTTTTGATGAGAACAATG-3′ F 5′-ACTCCAGGTAGGTCTTGGTGTTCA-3′ |
| TLR3 | F 5′-AGTGCCGTCTATTTGCCACA-3′ F 5′-GCATCCCAAAGGGCAAAAGG-3′ |
| TLR4 | F 5′-TGCTTCTTGCTGGCTGCATA-3′ F 5′-CCAGTCCTCATCCTGGCTTG-3′ |
| TLR7 | F 5′-CACAGCCGTCCCTACTGTTT-3′ F 5′-TTTTTACACGGCGCACAAGG-3′ |
| TLR8 | F 5′-ACATCAGCAAGACCCATCCC-3′ F 5′-TCTTCGGCGCATAACTCACA-3′ |
| NF-κB | F 5′-TGCAGCAGACCAAGGAGATG-3′ F 5′-TGCATTGGGGGCTTTACTGT-3′ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wen, Q.; Zhan, B.; Jin, L.; Peng, Z.; Liu, J.; Zhu, L.; Yang, D.; Xu, X.; Zhang, L.; Li, G.; et al. Chlojaponilactone B Attenuates THP-1 Macrophage Pyroptosis by Inhibiting the TLR/MyD88/NF-κB Pathway. Pharmaceuticals 2024, 17, 402. https://doi.org/10.3390/ph17030402
Wen Q, Zhan B, Jin L, Peng Z, Liu J, Zhu L, Yang D, Xu X, Zhang L, Li G, et al. Chlojaponilactone B Attenuates THP-1 Macrophage Pyroptosis by Inhibiting the TLR/MyD88/NF-κB Pathway. Pharmaceuticals. 2024; 17(3):402. https://doi.org/10.3390/ph17030402
Chicago/Turabian StyleWen, Qiyin, Bingjinfeng Zhan, Lu Jin, Zijing Peng, Ju Liu, Longping Zhu, Depo Yang, Xinjun Xu, Lixia Zhang, Ge Li, and et al. 2024. "Chlojaponilactone B Attenuates THP-1 Macrophage Pyroptosis by Inhibiting the TLR/MyD88/NF-κB Pathway" Pharmaceuticals 17, no. 3: 402. https://doi.org/10.3390/ph17030402
APA StyleWen, Q., Zhan, B., Jin, L., Peng, Z., Liu, J., Zhu, L., Yang, D., Xu, X., Zhang, L., Li, G., & Zhao, Z. (2024). Chlojaponilactone B Attenuates THP-1 Macrophage Pyroptosis by Inhibiting the TLR/MyD88/NF-κB Pathway. Pharmaceuticals, 17(3), 402. https://doi.org/10.3390/ph17030402






