Microvascular Experimentation in the Chick Chorioallantoic Membrane as a Model for Screening Angiogenic Agents including from Gene-Modified Cells
Abstract
:1. Introduction
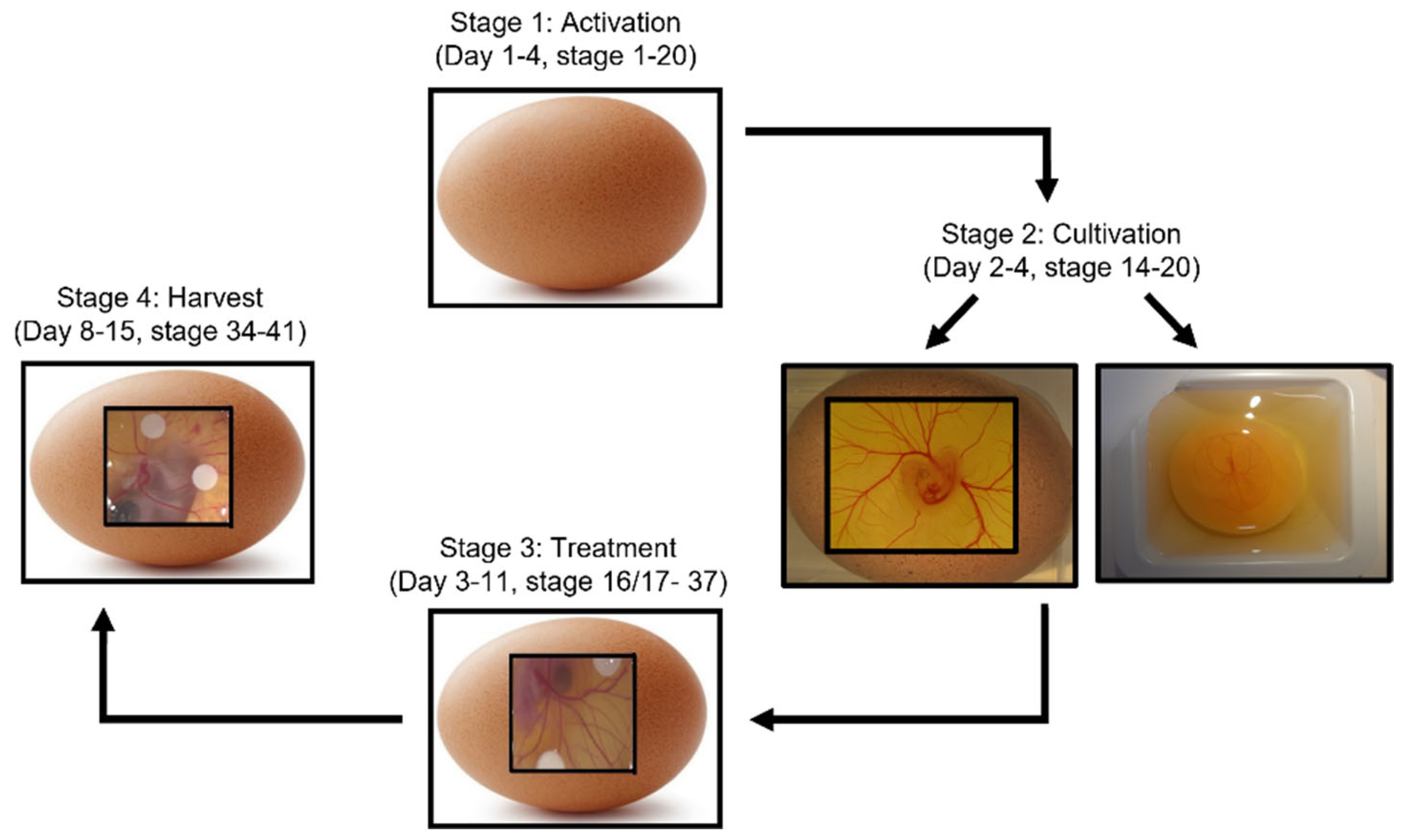
2. CAM and Chick Development
3. CAM Assay Procedures
4. CAM Experimental Treatments
4.1. Scaffolds and Delivery Methods
4.2. Drugs, Metabolites, miRNAs and Other Treatments
4.3. Cell and Gene Modified Cell On-Plants
5. CAM Analyses
5.1. Sectioning and Staining Techniques
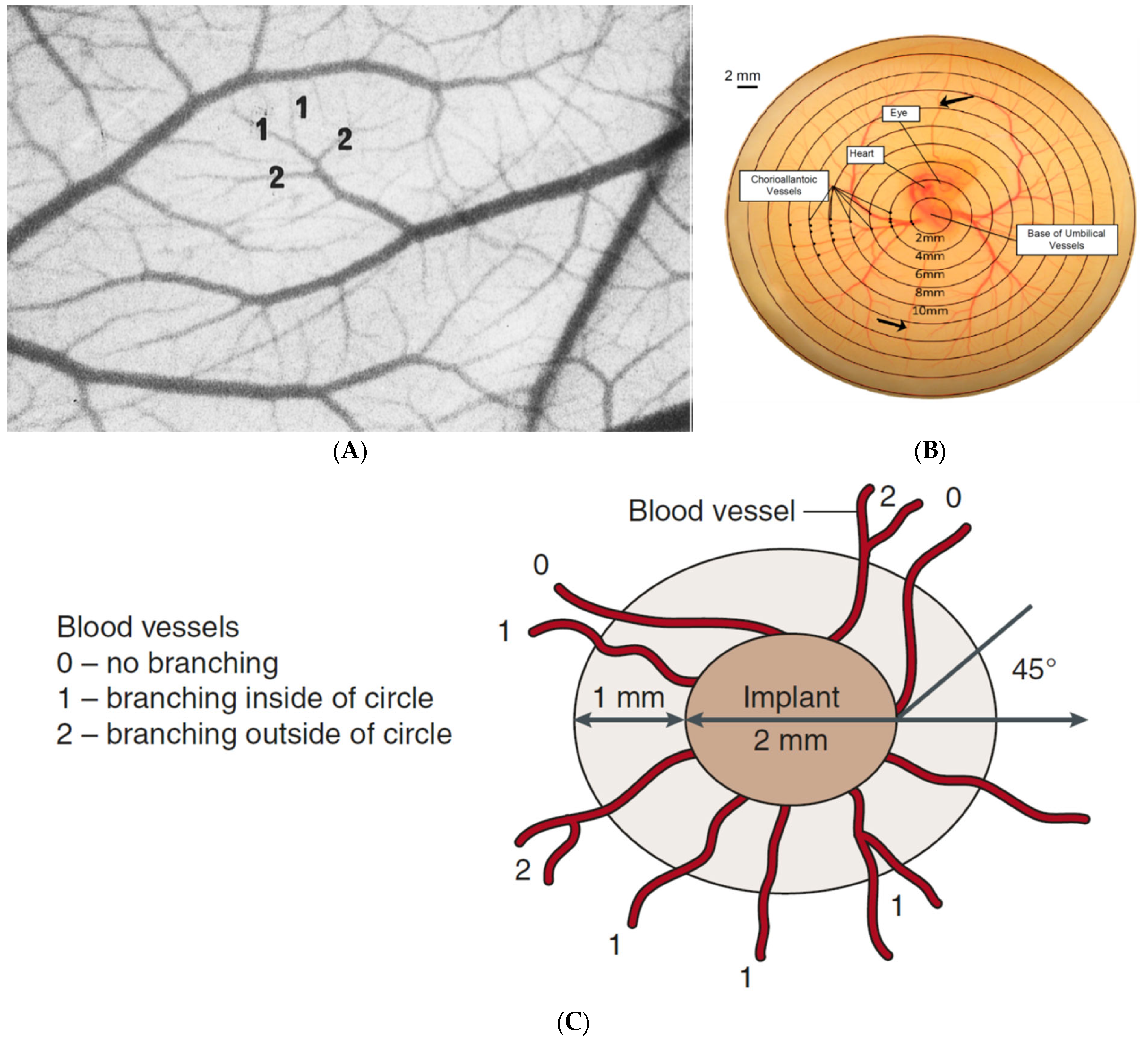
5.2. Image Quantification Techniques
5.3. Vascular Casting
5.4. Live Blood-Flow Observation
5.5. Molecular Analysis
6. Advantages and Limitations of the CAM to Study Microcirculation
7. Concluding Remarks
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Giaccia, A.J.; Simon, M.C.; Johnson, R. The biology of hypoxia: The role of oxygen sensing in development, normal function, and disease. Genes Dev. 2004, 18, 2183–2194. [Google Scholar] [CrossRef] [Green Version]
- Grace, P. Ischaemia-reperfusion injury. Br. J. Surg. 1994, 81, 637–647. [Google Scholar] [CrossRef]
- AS, M.N.; Deshpande, R.; Kale, V.P.; Bhonde, R.R.; Datar, S.P. Establishment of an in ovo chick embryo yolk sac membrane (YSM) assay for pilot screening of potential angiogenic and anti-angiogenic agents. Cell Biol. Int. 2018, 42, 1474–1483. [Google Scholar] [CrossRef] [PubMed]
- Ribatti, D.; Crivellato, E. “Sprouting angiogenesis”, a reappraisal. Dev. Biol. 2012, 372, 157–165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ausprunk, D.; Knighton, D.; Folkman, J. Vascularization of normal and neoplastic tissues grafted to the chick chorioallantois. Role of host and preexisting graft blood vessels. Am. J. Pathol. 1975, 79, 597. [Google Scholar] [PubMed]
- Djonov, V.; Schmid, M.; Tschanz, S.A.; Burri, P.H. Intussusceptive angiogenesis: Its role in embryonic vascular network formation. Circ. Res. 2000, 86, 286–292. [Google Scholar] [CrossRef]
- Montesano, R.; Vassalli, J.-D.; Baird, A.; Guillemin, R.; Orci, L. Basic fibroblast growth factor induces angiogenesis in vitro. Proc. Natl. Acad. Sci. USA 1986, 83, 7297–7301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baum, O.; Suter, F.; Gerber, B.; Tschanz, S.A.; Buergy, R.; Blank, F.; Hlushchuk, R.; Djonov, V. VEGF-A Promotes Intussusceptive Angiogenesis in the Developing Chicken Chorioallantoic Membrane. Microcirculation 2010, 17, 447–457. [Google Scholar] [CrossRef] [PubMed]
- Leker, R.R.; Toth, Z.E.; Shahar, T.; Cassiani-Ingoni, R.; Szalayova, I.; Key, S.; Bratincsák, A.; Mezey, E. Transforming growth factor α induces angiogenesis and neurogenesis following stroke. Neuroscience 2009, 163, 233–243. [Google Scholar] [CrossRef] [Green Version]
- Yang, E.Y.; Moses, H.L. Transforming growth factor beta 1-induced changes in cell migration, proliferation, and angiogenesis in the chicken chorioallantoic membrane. J. Cell Biol. 1990, 111, 731–741. [Google Scholar] [CrossRef]
- Bussolino, F.; Di Renzo, M.F.; Ziche, M.; Bocchietto, E.; Olivero, M.; Naldini, L.; Gaudino, G.; Tamagnone, L.; Coffer, A.; Comoglio, P. Hepatocyte growth factor is a potent angiogenic factor which stimulates endothelial cell motility and growth. J. Cell Biol. 1992, 119, 629–641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, L.; Chopp, M.; Teng, H.; Bolz, M.; Francisco, M.A.; Aluigi, D.M.; Wang, X.L.; Zhang, R.L.; Chrsitensen, S.; Sager, T.N. Tumor necrosis factor α primes cerebral endothelial cells for erythropoietin-induced angiogenesis. J. Cereb. Blood Flow Metab. 2011, 31, 640–647. [Google Scholar] [CrossRef] [Green Version]
- Volpert, O.V.; Tolsma, S.S.; Pellerin, S.; Feige, J.-J.; Chen, H.; Mosher, D.F.; Bouck, N. Inhibition of angiogenesis by thrombospondin-2. Biochem. Biophys. Res. Commun. 1995, 217, 326–332. [Google Scholar] [CrossRef] [PubMed]
- O’Reilly, M.; Holmgren, L.; Shing, Y.; Chen, C.; Rosenthal, R.; Cao, Y.; Moses, M.; Lane, W.; Sage, E.; Folkman, J. Angiostatin: A Circulating Endothelial Cell Inhibitor That Suppresses Angiogenesis and Tumor Growth; Cold Spring Harbor Symposia on Quantitative Biology; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 1994; pp. 471–482. [Google Scholar]
- O’Reilly, M.S.; Boehm, T.; Shing, Y.; Fukai, N.; Vasios, G.; Lane, W.S.; Flynn, E.; Birkhead, J.R.; Olsen, B.R.; Folkman, J. Endostatin: An endogenous inhibitor of angiogenesis and tumor growth. Cell 1997, 88, 277–285. [Google Scholar] [CrossRef] [Green Version]
- Watanabe, K.; Hasegawa, Y.; Yamashita, H.; Shimizu, K.; Ding, Y.; Abe, M.; Ohta, H.; Imagawa, K.; Hojo, K.; Maki, H. Vasohibin as an endothelium-derived negative feedback regulator of angiogenesis. J. Clin. Investig. 2004, 114, 898–907. [Google Scholar] [CrossRef] [Green Version]
- Deryugina, E.I.; Quigley, J.P. Chick embryo chorioallantoic membrane models to quantify angiogenesis induced by inflammatory and tumor cells or purified effector molecules. Methods Enzymol. 2008, 444, 21–41. [Google Scholar]
- DeCicco-Skinner, K.L.; Henry, G.H.; Cataisson, C.; Tabib, T.; Gwilliam, J.C.; Watson, N.J.; Bullwinkle, E.M.; Falkenburg, L.; O’Neill, R.C.; Morin, A. Endothelial cell tube formation assay for the in vitro study of angiogenesis. J. Vis. Exp. 2014, e51312. [Google Scholar] [CrossRef] [PubMed]
- Masson, V.; Devy, L.; Grignet-Debrus, C.; Bernt, S.; Bajou, K.; Blacher, S.; Roland, G.; Chang, Y.; Fong, T.; Carmeliet, P. Mouse aortic ring assay: A new approach of the molecular genetics of angiogenesis. Biol. Proced. Online 2002, 4, 24–31. [Google Scholar] [CrossRef] [Green Version]
- Wartenberg, M.; Günther, J.; Hescheler, J.; Sauer, H. The embryoid body as a novel in vitro assay system for antiangiogenic agents. Lab. Investig. 1998, 78, 1301–1314. [Google Scholar] [PubMed]
- Deckers, M.; van der Pluijm, G.; Dooijewaard, S.; Kroon, M.; van Hinsbergh, V.; Papapoulos, S.; Löwik, C. Effect of angiogenic and antiangiogenic compounds on the outgrowth of capillary structures from fetal mouse bone explants. Lab. Investig. 2001, 81, 5–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gimbrone, M.A., Jr.; Cotran, R.S.; Leapman, S.B.; Folkman, J. Tumor growth and neovascularization: An experimental model using the rabbit cornea. J. Natl. Cancer Inst. 1974, 52, 413–427. [Google Scholar] [CrossRef]
- Norrby, K.; Jakobsson, A.; Sörbo, J. Mast-cell-mediated angiogenesis: A novel experimental model using the rat mesentery. Virchows Arch. B 1986, 52, 195–206. [Google Scholar] [CrossRef]
- Lehr, H.-A.; Leunig, M.; Menger, M.D.; Nolte, D.; Messmer, K. Dorsal skinfold chamber technique for intravital microscopy in nude mice. Am. J. Pathol. 1993, 143, 1055. [Google Scholar] [PubMed]
- Brown, J.L.; Cao, Z.A.; Pinzon-Ortiz, M.; Kendrew, J.; Reimer, C.; Wen, S.; Zhou, J.Q.; Tabrizi, M.; Emery, S.; McDermott, B. A human monoclonal anti-ANG2 antibody leads to broad antitumor activity in combination with VEGF inhibitors and chemotherapy agents in preclinical models. Mol. Cancer Ther. 2010, 9, 145–156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kue, C.S.; Tan, K.Y.; LaM, M.L.; Lee, H.B. Chick embryo chorioallantoic membrane (CAM): An alternative predictive model in acute toxicological studies for anti-cancer drugs. Exp. Anim. 2015, 64, 129–138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ribatti, D.; Vacca, A.; Roncali, L.; Dammacco, F. The chick embryo chorioallantoic membrane as a model for in vivo research on angiogenesis. Int. J. Dev. Biol. 1996, 40, 1189–1197. [Google Scholar] [CrossRef] [PubMed]
- Dohle, D.S.; Pasa, S.D.; Gustmann, S.; Laub, M.; Wissler, J.H.; Jennissen, H.P.; Dünker, N. Chick ex ovo culture and ex ovo CAM assay: How it really works. J. Vis. Exp. 2009, e1620. [Google Scholar] [CrossRef] [Green Version]
- Blacher, S.; Devy, L.; Hlushchuk, R.; Larger, E.; Lamandé, N.; Burri, P.; Corvol, P.; Djonov, V.; Foidart, J.-M.; Noël, A. Quantification of angiogenesis in the chicken chorioallantoic membrane (CAM). Image Anal. Stereol. 2005, 24, 169–180. [Google Scholar] [CrossRef] [Green Version]
- Makanya, A.N.; Dimova, I.; Koller, T.; Styp-Rekowska, B.; Djonov, V. Dynamics of the developing chick chorioallantoic membrane assessed by stereology, allometry, immunohistochemistry and molecular analysis. PLoS ONE 2016, 11, e0152821. [Google Scholar] [CrossRef] [Green Version]
- Russell, W.M.S.; Burch, R.L. The Principles of Humane Experimental Technique; Methuen & Co., Ltd.: London, UK, 1959. [Google Scholar]
- Ribatti, D.; Nico, B.; Vacca, A.; Presta, M. The gelatin sponge–chorioallantoic membrane assay. Nat. Protoc. 2006, 1, 85. [Google Scholar] [CrossRef]
- Lokman, N.A.; Elder, A.S.; Ricciardelli, C.; Oehler, M.K. Chick chorioallantoic membrane (CAM) assay as an in vivo model to study the effect of newly identified molecules on ovarian cancer invasion and metastasis. Int. J. Mol. Sci. 2012, 13, 9959–9970. [Google Scholar] [CrossRef] [Green Version]
- Hamburger, V.; Hamilton, H.L. A series of normal stages in the development of the chick embryo. J. Morphol. 1951, 88, 49–92. [Google Scholar] [CrossRef] [PubMed]
- Chu, P.-Y.; Koh, A.P.-F.; Antony, J.; Huang, R.Y.-J. Applications of the Chick Chorioallantoic Membrane as an Alternative Model for Cancer Studies. Cells Tissues Organs 2021, 1–16. [Google Scholar] [CrossRef]
- Ribatti, D. The chick embryo chorioallantoic membrane (CAM). A multifaceted experimental model. Mech. Dev. 2016, 141, 70–77. [Google Scholar] [CrossRef]
- Barnhill, R.L.; Ryan, T.J. Biochemical modulation of angiogenesis in the chorioallantoic membrane of the chick embryo. J. Investig. Dermatol. 1983, 81, 485–488. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beckers, M.; Gladis-Villanueva, M.; Hamann, W.; Schmutzler, W.; Zwadlo-Klarwasser, G. The use of the chorio-allantoic membrane of the chick embryo as test for anti-inflammatory activity. Inflamm. Res. 1997, 46, 29–30. [Google Scholar] [CrossRef]
- Ribatti, D.; Frigeri, A.; Nico, B.; Nicchia, G.P.; De Giorgis, M.; Roncali, L.; Svelto, M. Aquaporin-1 expression in the chick embryo chorioallantoic membrane. Anat. Rec. 2002, 268, 85–89. [Google Scholar] [CrossRef]
- Yar, M.; Shahzad, S.; Shahzadi, L.; Shahzad, S.A.; Mahmood, N.; Chaudhry, A.A.; ur Rehman, I.; MacNeil, S. Heparin binding chitosan derivatives for production of pro-angiogenic hydrogels for promoting tissue healing. Mater. Sci. Eng. C 2017, 74, 347–356. [Google Scholar] [CrossRef]
- O’Dwyer, J.; Murphy, R.; González-Vázquez, A.; Kovarova, L.; Pravda, M.; Velebny, V.; Heise, A.; Duffy, G.P.; Cryan, S.A. Translational Studies on the Potential of a VEGF Nanoparticle-Loaded Hyaluronic Acid Hydrogel. Pharmaceutics 2021, 13, 779. [Google Scholar] [CrossRef] [PubMed]
- Naik, M.; Brahma, P.; Dixit, M. A cost-effective and efficient chick ex-ovo CAM assay protocol to assess angiogenesis. Methods Protoc. 2018, 1, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murphy, J.B. Transplantability of tissues to the embryo of foreign species: Its bearing on questions of tissue specificity and tumor immunity. J. Exp. Med. 1913, 17, 482–493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nowak-Sliwinska, P.; Segura, T.; Iruela-Arispe, M.L. The chicken chorioallantoic membrane model in biology, medicine and bioengineering. Angiogenesis 2014, 17, 779–804. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ribatti, D.; Annese, T.; Tamma, R. The use of the chick embryo CAM assay in the study of angiogenic activiy of biomaterials. Microvasc. Res. 2020, 131, 104026. [Google Scholar] [CrossRef] [PubMed]
- Zwadlo-Klarwasser, G.; Görlitz, K.; Hafemann, B.; Klee, D.; Klosterhalfen, B. The chorioallantoic membrane of the chick embryo as a simple model for the study of the angiogenic and inflammatory response to biomaterials. J. Mater. Sci. Mater. Med. 2001, 12, 195–199. [Google Scholar]
- Tomanek, R.J.; Schatteman, G.C. Angiogenesis: New insights and therapeutic potential. Anat. Rec. 2000, 261, 126–135. [Google Scholar] [CrossRef]
- Belle, J.; Ysasi, A.; Bennett, R.D.; Filipovic, N.; Nejad, M.I.; Trumper, D.L.; Ackermann, M.; Wagner, W.; Tsuda, A.; Konerding, M.A. Stretch-induced intussuceptive and sprouting angiogenesis in the chick chorioallantoic membrane. Microvasc. Res. 2014, 95, 60–67. [Google Scholar] [CrossRef] [Green Version]
- Mangır, N.; Eke, G.; Hasirci, N.; Chapple, C.R.; Hasirci, V.; MacNeil, S. An estradiol releasing, proangiogenic hydrogel as a candidate material for use in soft tissue interposition. Neurourol. Urodyn. 2019, 38, 1195–1202. [Google Scholar] [CrossRef] [PubMed]
- Sys, G.M.; Lapeire, L.; Stevens, N.; Favoreel, H.; Forsyth, R.; Bracke, M.; De Wever, O. The in ovo CAM-assay as a xenograft model for sarcoma. J. Vis. Exp. 2013, 77, e50522. [Google Scholar] [CrossRef]
- Kleinman, H.K.; Martin, G.R. Matrigel: Basement Membrane Matrix with Biological Activity; Seminars in Cancer Biology; Elsevier: Amsterdam, The Netherlands, 2005; pp. 378–386. [Google Scholar]
- Hughes, C.S.; Postovit, L.M.; Lajoie, G.A. Matrigel: A complex protein mixture required for optimal growth of cell culture. Proteomics 2010, 10, 1886–1890. [Google Scholar] [CrossRef]
- Aisenbrey, E.A.; Murphy, W.L. Synthetic alternatives to Matrigel. Nat. Rev. Mater. 2020, 5, 539–551. [Google Scholar] [CrossRef] [PubMed]
- Subauste, M.C.; Kupriyanova, T.A.; Conn, E.M.; Ardi, V.C.; Quigley, J.P.; Deryugina, E.I. Evaluation of metastatic and angiogenic potentials of human colon carcinoma cells in chick embryo model systems. Clin. Exp. Metastasis 2009, 26, 1033. [Google Scholar] [CrossRef] [Green Version]
- Valiulytė, I.; Curkūnavičiūtė, R.; Ribokaitė, L.; Kazlauskas, A.; Vaitkevičiūtė, M.; Skauminas, K.; Valančiūtė, A. The Anti-tumorigenic activity of Sema3C in the chick embryo chorioallantoic membrane model. Int. J. Mol. Sci. 2019, 20, 5672. [Google Scholar] [CrossRef] [Green Version]
- Hagenbuchner, J.; Rupp, M.; Salvador, C.; Meister, B.; Kiechl-Kohlendorfer, U.; Müller, T.; Geiger, K.; Sergi, C.; Obexer, P.; Ausserlechner, M.J. Nuclear FOXO3 predicts adverse clinical outcome and promotes tumor angiogenesis in neuroblastoma. Oncotarget 2016, 7, 77591. [Google Scholar] [CrossRef] [Green Version]
- Kavaliauskaitė, D.; Stakišaitis, D.; Martinkutė, J.; Šlekienė, L.; Kazlauskas, A.; Balnytė, I.; Lesauskaitė, V.; Valančiūtė, A. The effect of sodium valproate on the glioblastoma u87 cell line tumor development on the chicken embryo chorioallantoic membrane and on EZH2 and p53 expression. BioMed Res. Int. 2017, 2017, 6326053. [Google Scholar] [CrossRef] [PubMed]
- Hood, J.D.; Frausto, R.; Kiosses, W.B.; Schwartz, M.A.; Cheresh, D.A. Differential αv integrin–mediated Ras-ERK signaling during two pathways of angiogenesis. J. Cell Biol. 2003, 162, 933–943. [Google Scholar] [CrossRef] [Green Version]
- Da Costa, P.M.; da Costa, M.P.; Carvalho, A.A.; Cavalcanti, S.M.T.; de Oliveira Cardoso, M.V.; de Oliveira Filho, G.B.; de Araújo Viana, D.; Fechine-Jamacaru, F.V.; Leite, A.C.L.; de Moraes, M.O. Improvement of in vivo anticancer and antiangiogenic potential of thalidomide derivatives. Chemico-Biol. Interact. 2015, 239, 174–183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vimalraj, S.; Ashokkumar, T.; Saravanan, S. Biogenic gold nanoparticles synthesis mediated by Mangifera indica seed aqueous extracts exhibits antibacterial, anticancer and anti-angiogenic properties. Biomed. Pharmacother. 2018, 105, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Santos, C.J.; Ferreira Soares, D.C.; Ferreira, C.d.A.; de Barros, A.L.B.; Silva Cunha Junior, A.D.; Filho, F.M. Antiangiogenic evaluation of ZnWO4 nanoparticles synthesised through microwave-assisted hydrothermal method. J. Drug Target. 2018, 26, 806–817. [Google Scholar] [CrossRef] [PubMed]
- Boyineni, J.; Tanpure, S.; Gnanamony, M.; Antony, R.; Fernández, K.S.; Lin, J.; Pinson, D.; Gondi, C.S. SPARC overexpression combined with radiation retards angiogenesis by suppressing VEGF-A via miR-410 in human neuroblastoma cells. Int. J. Oncol. 2016, 49, 1394–1406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Michaeli, S.; Dakwar, V.; Weidenfeld, K.; Granski, O.; Gilon, O.; Schif-Zuck, S.; Mamchur, A.; Shams, I.; Barkan, D. Soluble mediators produced by pro-resolving macrophages inhibit angiogenesis. Front. Immunol. 2018, 9, 768. [Google Scholar] [CrossRef] [Green Version]
- Lemmens, S.; Kusters, L.; Bronckaers, A.; Geurts, N.; Hendrix, S. The β2-Adrenoceptor Agonist Terbutaline Stimulates Angiogenesis via Akt and ERK Signaling. J. Cell. Physiol. 2017, 232, 298–308. [Google Scholar] [CrossRef]
- Torres, P.; Díaz, J.; Arce, M.; Silva, P.; Mendoza, P.; Lois, P.; Molina-Berríos, A.; Owen, G.I.; Palma, V.; Torres, V.A. The salivary peptide histatin-1 promotes endothelial cell adhesion, migration, and angiogenesis. FASEB J. 2017, 31, 4946–4958. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ribatti, D.; De Falco, G.; Nico, B.; Ria, R.; Crivellato, E.; Vacca, A. In vivo time-course of the angiogenic response induced by multiple myeloma plasma cells in the chick embryo chorioallantoic membrane. J. Anat. 2003, 203, 323–328. [Google Scholar] [CrossRef]
- Shah, B.B.; Baksi, R.; Chaudagar, K.K.; Nivsarkar, M.; Mehta, A.A. Anti-leukemic and anti-angiogenic effects of d-Limonene on K562-implanted C57 BL/6 mice and the chick chorioallantoic membrane model. Anim. Models Exp. Med. 2018, 1, 328–333. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dewangan, J.; Kaushik, S.; Rath, S.K.; Balapure, A.K. Centchroman regulates breast cancer angiogenesis via inhibition of HIF-1α/VEGFR2 signalling axis. Life Sci. 2018, 193, 9–19. [Google Scholar] [CrossRef]
- Lamanuzzi, A.; Saltarella, I.; Desantis, V.; Frassanito, M.A.; Leone, P.; Racanelli, V.; Nico, B.; Ribatti, D.; Ditonno, P.; Prete, M. Inhibition of mTOR complex 2 restrains tumor angiogenesis in multiple myeloma. Oncotarget 2018, 9, 20563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Desantis, V.; Frassanito, M.A.; Tamma, R.; Saltarella, I.; Di Marzo, L.; Lamanuzzi, A.; Solimando, A.G.; Ruggieri, S.; Annese, T.; Nico, B. Rhu-Epo down-regulates pro-tumorigenic activity of cancer-associated fibroblasts in multiple myeloma. Ann. Hematol. 2018, 97, 1251–1258. [Google Scholar] [CrossRef]
- Manjunathan, R.; Devarajan, N.; Ragunathan, M. Possible Mechanism of Human Recombinant Leptin-Induced VEGF A Synthesis via PI3K/Akt/mTOR/S6 Kinase Signaling Pathway while Inducing Angiogenesis: An Analysis Using Chicken Chorioallantoic Membrane Model. J. Vasc. Res. 2021, 58, 343–360. [Google Scholar] [CrossRef]
- Gacche, R.; Meshram, R.; Shegokar, H.; Gond, D.; Kamble, S.; Dhabadge, V.; Utage, B.; Patil, K.; More, R. Flavonoids as a scaffold for development of novel anti-angiogenic agents: An experimental and computational enquiry. Arch. Biochem. Biophys. 2015, 577, 35–48. [Google Scholar] [CrossRef]
- Kamble, S.; Utage, B.; Mogle, P.; Kamble, R.; Hese, S.; Dawane, B.; Gacche, R. Evaluation of curcumin capped copper nanoparticles as possible inhibitors of human breast cancer cells and angiogenesis: A comparative study with native curcumin. AAPS PharmSciTech 2016, 17, 1030–1041. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eke, G.; Mangir, N.; Hasirci, N.; MacNeil, S.; Hasirci, V. Development of a UV crosslinked biodegradable hydrogel containing adipose derived stem cells to promote vascularization for skin wounds and tissue engineering. Biomaterials 2017, 129, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Mangir, N.; Raza, A.; Haycock, J.W.; Chapple, C.; Macneil, S. An improved in vivo methodology to visualise tumour induced changes in vasculature using the chick chorionic allantoic membrane assay. In Vivo 2018, 32, 461–472. [Google Scholar] [PubMed] [Green Version]
- Campbell, K.T.; Stilhano, R.S.; Silva, E.A. Enzymatically degradable alginate hydrogel systems to deliver endothelial progenitor cells for potential revasculature applications. Biomaterials 2018, 179, 109–121. [Google Scholar] [CrossRef]
- Shahzad, S.A.; Yar, M.; Khan, Z.A.; Shahzadi, L.; Naqvi, S.A.R.; Mahmood, A.; Ullah, S.; Shaikh, A.J.; Sherazi, T.A.; Bale, A.T. Identification of 1, 2, 4-triazoles as new thymidine phosphorylase inhibitors: Future anti-tumor drugs. Bioorganic Chem. 2019, 85, 209–220. [Google Scholar] [CrossRef]
- Papoutsi, M.; Sleeman, J.P.; Wilting, J. Interaction of rat tumor cells with blood vessels and lymphatics of the avian chorioallantoic membrane. Microsc. Res. Tech. 2001, 55, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Deryugina, E.I.; Zijlstra, A.; Partridge, J.J.; Kupriyanova, T.A.; Madsen, M.A.; Papagiannakopoulos, T.; Quigley, J.P. Unexpected effect of matrix metalloproteinase down-regulation on vascular intravasation and metastasis of human fibrosarcoma cells selected in vivo for high rates of dissemination. Cancer Res. 2005, 65, 10959–10969. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nik, M.E.; Malaekeh-Nikouei, B.; Amin, M.; Hatamipour, M.; Teymouri, M.; Sadeghnia, H.R.; Iranshahi, M.; Jaafari, M.R. Liposomal formulation of Galbanic acid improved therapeutic efficacy of pegylated liposomal Doxorubicin in mouse colon carcinoma. Sci. Rep. 2019, 9, 9527. [Google Scholar] [CrossRef] [PubMed]
- Iranmanesh, M.; Mohebbati, R.; Forouzanfar, F.; Roshan, M.K.; Ghorbani, A.; Nik, M.J.; Soukhtanloo, M. In vivo and In vitro effects of ethanolic extract of Trigonella foenum-graecum L. seeds on proliferation, angiogenesis and tube formation of endothelial cells. Res. Pharm. Sci. 2018, 13, 343. [Google Scholar]
- Nkembo, A.T.; Ntantie, E.; Salako, O.O.; Amissah, F.; Poku, R.A.; Latinwo, L.M.; Lamango, N.S. The antiangiogenic effects of polyisoprenylated cysteinyl amide inhibitors in HUVEC, chick embryo and zebrafish is dependent on the polyisoprenyl moiety. Oncotarget 2016, 7, 68194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumari, R.; Rawat, K.; Kumari, A.; Shrivastava, A. Amelioration of Dalton’s lymphoma–induced angiogenesis by melatonin. Tumor Biol. 2017, 39, 1–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, G.; Onur, M.A. Cellular localization and biological effects of 20 nm-gold nanoparticles. J. Biomed. Mater. Res. Part A 2018, 106, 1708–1721. [Google Scholar] [CrossRef]
- Weiss, A.; van Beijnum, J.R.; Bonvin, D.; Jichlinski, P.; Dyson, P.J.; Griffioen, A.W.; Nowak-Sliwinska, P. Low-dose angiostatic tyrosine kinase inhibitors improve photodynamic therapy for cancer: Lack of vascular normalization. J. Cell. Mol. Med. 2014, 18, 480–491. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.-Y.; Hung, C.-H.; Chang, C.-H.; Tseng, T.-H.; Wang, C.-J. Solanum nigrum suppress angiogenesis-mediated tumor growth through inhibition of the AKT/mTOR pathway. Am. J. Chin. Med. 2016, 44, 1273–1288. [Google Scholar] [CrossRef] [PubMed]
- Steinle, H.; Golombek, S.; Behring, A.; Schlensak, C.; Wendel, H.P.; Avci-Adali, M. Improving the angiogenic potential of EPCs via engineering with synthetic modified mRNAs. Mol. Ther. Nucleic Acids 2018, 13, 387–398. [Google Scholar] [CrossRef] [Green Version]
- Liu, L.-Z.; Ding, M.; Zheng, J.Z.; Zhu, Y.; Fenderson, B.A.; Li, B.; Jing, J.Y.; Jiang, B.-H. Tungsten carbide-cobalt nanoparticles induce reactive oxygen species, AKT, ERK, AP-1, NF-κB, VEGF, and angiogenesis. Biol. Trace Elem. Res. 2015, 166, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Su, C.-M.; Wang, I.-C.; Liu, S.-C.; Sun, Y.; Jin, L.; Wang, S.-W.; Lee, H.-P.; Tseng, W.-P.; Tang, C.-H. Hypoxia induced mitogenic factor (HIMF) triggers angiogenesis by increasing interleukin-18 production in myoblasts. Sci. Rep. 2017, 7, 7393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, W.; Wang, Q.; Qi, X.; Lu, H.; Chen, Y.; Shi, J.; Wang, F.; Wang, Z.; Lu, Y.; Lu, Z. An oncogenic viral interferon regulatory factor upregulates CUB domain-containing protein 1 to promote angiogenesis by hijacking transcription factor lymphoid enhancer-binding factor 1 and metastasis suppressor CD82. Cell Death Differ. 2020, 27, 3289–3306. [Google Scholar] [CrossRef]
- Adar, Y.; Stark, M.; Bram, E.E.; Nowak-Sliwinska, P.; Van Den Bergh, H.; Szewczyk, G.; Sarna, T.; Skladanowski, A.; Griffioen, A.; Assaraf, Y.G. Imidazoacridinone-dependent lysosomal photodestruction: A pharmacological Trojan horse approach to eradicate multidrug-resistant cancers. Cell Death Dis. 2012, 3, e293. [Google Scholar] [CrossRef] [Green Version]
- Berndsen, R.H.; Castrogiovanni, C.; Weiss, A.; Rausch, M.; Dallinga, M.G.; Miljkovic-Licina, M.; Klaassen, I.; Meraldi, P.; van Beijnum, J.R.; Nowak-Sliwinska, P. Anti-angiogenic effects of crenolanib are mediated by mitotic modulation independently of PDGFR expression. Br. J. Cancer 2019, 121, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Carrillo, P.; Martínez-Poveda, B.; Cheng-Sánchez, I.; Guerra, J.; Tobia, C.; López-Romero, J.M.; Sarabia, F.; Medina, M.Á.; Quesada, A.R. Exploring the antiangiogenic potential of solomonamide A bioactive precursors: In vitro and in vivo evidences of the inhibitory activity of solo F-OH during angiogenesis. Mar. Drugs 2019, 17, 228. [Google Scholar] [CrossRef] [Green Version]
- Dragostin, O.-M.; Tatia, R.; Samal, S.K.; Oancea, A.; Zamfir, A.S.; Dragostin, I.; Lisă, E.-L.; Apetrei, C.; Zamfir, C.L. Designing of Chitosan Derivatives Nanoparticles with Antiangiogenic Effect for Cancer Therapy. Nanomaterials 2020, 10, 698. [Google Scholar] [CrossRef] [Green Version]
- Paterson, T.E.; Gigliobianco, G.; Sherborne, C.; Green, N.H.; Dugan, J.M.; MacNeil, S.; Reilly, G.C.; Claeyssens, F. Porous microspheres support mesenchymal progenitor cell ingrowth and stimulate angiogenesis. APL Bioeng. 2018, 2, 026103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Del Gaudio, C.; Crognale, V.; Serino, G.; Galloni, P.; Audenino, A.; Ribatti, D.; Morbiducci, U. Natural polymeric microspheres for modulated drug delivery. Mater. Sci. Eng. C 2017, 75, 408–417. [Google Scholar] [CrossRef] [PubMed]
- Fergelot, P.; Bernhard, J.-C.; Soulet, F.; Kilarski, W.W.; Léon, C.; Courtois, N.; Deminiere, C.; Herbert, J.M.; Antczak, P.; Falciani, F. The experimental renal cell carcinoma model in the chick embryo. Angiogenesis 2013, 16, 181–194. [Google Scholar] [CrossRef]
- Soares, D.C.F.; de Paula Oliveira, D.C.; Barcelos, L.S.; Barbosa, A.S.; Vieira, L.C.; Townsend, D.M.; Rubello, D.; de Barros, A.L.B.; Duarte, L.P.; Silva-Cunha, A. Antiangiogenic activity of PLGA-Lupeol implants for potential intravitreal applications. Biomed. Pharmacother. 2017, 92, 394–402. [Google Scholar] [CrossRef]
- Doganci, S.; Yildirim, V.; Erol, G.; Yesildal, F.; Karabacak, K.; Kadan, M.; Ozkan, G.; Ince, M.; Ozgurtas, T. Polidocanol (Lauromacrogol 400) has anti-angiogenic effects in vitro and in vivo. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 1384–1389. [Google Scholar] [PubMed]
- Seifaddinipour, M.; Farghadani, R.; Namvar, F.; Mohamad, J.; Abdul Kadir, H. Cytotoxic effects and anti-angiogenesis potential of pistachio (Pistacia vera L.) hulls against MCF-7 human breast cancer cells. Molecules 2018, 23, 110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Y.-C.; Chen, P.-N.; Wang, S.-Y.; Liao, C.-Y.; Lin, Y.-Y.; Sun, S.-R.; Chiu, C.-L.; Hsieh, Y.-S.; Shieh, J.-C.; Chang, J.T. The differential roles of Slit2-exon 15 splicing variants in angiogenesis and HUVEC permeability. Angiogenesis 2015, 18, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, M.-J.; Liu, H.-T.; Wang, C.-N.; Huang, H.-Y.; Lin, Y.; Ko, Y.-S.; Wang, J.-S.; Chang, V.H.-S.; Pang, J.-H.S. Therapeutic potential of pro-angiogenic BPC157 is associated with VEGFR2 activation and up-regulation. J. Mol. Med. 2017, 95, 323–333. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Chen, L.H.; Niu, Y.H.; Geng, N.B.; Feng, C.J. AEG-1 promotes angiogenesis and may be a novel treatment target for tongue squamous cell carcinoma. Oral Dis. 2020, 26, 876–884. [Google Scholar] [CrossRef]
- Winter, R.; Dungel, P.; Reischies, F.M.J.; Rohringer, S.; Slezak, P.; Smolle, C.; Spendel, S.; Kamolz, L.-P.; Ghaffari-Tabrizi-Wizsy, N.; Schicho, K. Photobiomodulation (PBM) promotes angiogenesis in-vitro and in chick embryo chorioallantoic membrane model. Sci. Rep. 2018, 8, 17080. [Google Scholar] [CrossRef]
- Kim, H.J.; Ahn, M.-R. Apigenin Suppresses Angiogenesis by Inhibiting Tube Formation and Inducing Apoptosis. Nat. Prod. Commun. 2016, 11, 1433–1436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oliveira, C.; Granja, S.; Neves, N.M.; Reis, R.L.; Baltazar, F.; Silva, T.H.; Martins, A. Fucoidan from Fucus vesiculosus inhibits new blood vessel formation and breast tumor growth in vivo. Carbohydr. Polym. 2019, 223, 115034. [Google Scholar] [CrossRef] [Green Version]
- Bae, H.-S.; Kim, H.J.; Jeong, D.H.; Hosoya, T.; Kumazawa, S.; Jun, M.; Kim, O.-Y.; Kim, S.W.; Ahn, M.-R. In vitro and in vivo antiangiogenic activity of crowberry (Empetrum nigrum var. japonicum). Nat. Prod. Commun. 2016, 11. [Google Scholar] [CrossRef] [Green Version]
- Hirsch, T.; Laemmle, C.; Behr, B.; Lehnhardt, M.; Jacobsen, F.; Hoefer, D.; Kueckelhaus, M. Implant for autologous soft tissue reconstruction using an adipose-derived stem cell-colonized alginate scaffold. J. Plast. Reconstr. Aesthetic Surg. 2018, 71, 101–111. [Google Scholar] [CrossRef] [PubMed]
- He, R.-Q.; Li, X.-J.; Liang, L.; Xie, Y.; Luo, D.-Z.; Ma, J.; Peng, Z.-G.; Hu, X.-H.; Chen, G. The suppressive role of miR-542-5p in NSCLC: The evidence from clinical data and in vivo validation using a chick chorioallantoic membrane model. BMC Cancer 2017, 17, 655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gião, T.; Saavedra, J.; Vieira, J.R.; Pinto, M.T.; Arsequell, G.; Cardoso, I. Neuroprotection in early stages of Alzheimers Disease is promoted by Transthyretin angiogenic properties. Alzheimer’s Res. Ther. 2021, 13, 143. [Google Scholar] [CrossRef]
- Dikici, S.; Mangır, N.; Claeyssens, F.; Yar, M.; MacNeil, S. Exploration of 2-deoxy-D-ribose and 17β-Estradiol as alternatives to exogenous VEGF to promote angiogenesis in tissue-engineered constructs. Regen. Med. 2019, 14, 179–197. [Google Scholar] [CrossRef] [PubMed]
- Dikici, S.; Claeyssens, F.; MacNeil, S. Pre-Seeding of Simple Electrospun Scaffolds with a Combination of Endothelial Cells and Fibroblasts Strongly Promotes Angiogenesis. Tissue Eng. Regen. Med. 2020, 17, 445–458. [Google Scholar] [CrossRef]
- Zavala, G.; Prieto, C.P.; Villanueva, A.A.; Palma, V. Sonic hedgehog (SHH) signaling improves the angiogenic potential of Wharton’s jelly-derived mesenchymal stem cells (WJ-MSC). Stem Cell Res. Ther. 2017, 8, 203. [Google Scholar] [CrossRef]
- Gagliardi, F.; Narayanan, A.; Gallotti, A.L.; Pieri, V.; Mazzoleni, S.; Cominelli, M.; Rezzola, S.; Corsini, M.; Brugnara, G.; Altabella, L. Enhanced SPARCL1 expression in cancer stem cells improves preclinical modeling of glioblastoma by promoting both tumor infiltration and angiogenesis. Neurobiol. Dis. 2020, 134, 104705. [Google Scholar] [CrossRef] [PubMed]
- Strassburg, S.; Nienhueser, H.; Björn Stark, G.; Finkenzeller, G.; Torio-Padron, N. Co-culture of adipose-derived stem cells and endothelial cells in fibrin induces angiogenesis and vasculogenesis in a chorioallantoic membrane model. J. Tissue Eng. Regen. Med. 2016, 10, 496–506. [Google Scholar] [CrossRef]
- Watchararot, T.; Prasongchean, W.; Thongnuek, P. Angiogenic property of silk fibroin scaffolds with adipose-derived stem cells on chick chorioallantoic membrane. R. Soc. Open Sci. 2021, 8, 201618. [Google Scholar] [CrossRef]
- Shafaat, S.; Mangir, N.; Regureos, S.R.; Chapple, C.R.; MacNeil, S. Demonstration of improved tissue integration and angiogenesis with an elastic, estradiol releasing polyurethane material designed for use in pelvic floor repair. Neurourol. Urodyn. 2018, 37, 716–725. [Google Scholar] [CrossRef]
- Choi, S.-W.; Lee, K.-S.; Lee, J.H.; Kang, H.J.; Lee, M.J.; Kim, H.Y.; Park, K.-I.; Kim, S.-L.; Shin, H.K.; Seo, W.D. Suppression of Akt-HIF-1α signaling axis by diacetyl atractylodiol inhibits hypoxia-induced angiogenesis. BMB Rep. 2016, 49, 508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lim, H.N.; Jang, J.-P.; Han, J.M.; Jang, J.-H.; Ahn, J.S.; Jung, H.J. Antiangiogenic potential of microbial metabolite elaiophylin for targeting tumor angiogenesis. Molecules 2018, 23, 563. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, S.; Ullah, F.; Ayaz, M.; Zeb, A.; Ullah, F.; Sadiq, A. Antitumor and anti-angiogenic potentials of isolated crude saponins and various fractions of Rumex hastatus D. Don. Biol. Res. 2016, 49, 18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rabhi, C.; Arcile, G.; Cariel, L.; Lenoir, C.; Bignon, J.; Wdzieczak-Bakala, J.; Ouazzani, J. Antiangiogenic-like properties of fermented extracts of ayurvedic medicinal plants. J. Med. Food 2015, 18, 1065–1072. [Google Scholar] [CrossRef]
- Klagsbrun, M.; Knighton, D.; Folkman, J. Tumor angiogenesis activity in cells grown in tissue culture. Cancer Res. 1976, 36, 110–114. [Google Scholar] [PubMed]
- Knighton, D.; Ausprunk, D.; Tapper, D.; Folkman, J. Avascular and vascular phases of tumour growth in the chick embryo. Br. J. Cancer 1977, 35, 347–356. [Google Scholar] [CrossRef]
- Pink, D.B.; Schulte, W.; Parseghian, M.H.; Zijlstra, A.; Lewis, J.D. Real-time visualization and quantitation of vascular permeability in vivo: Implications for drug delivery. PLoS ONE 2012, 7, e33760. [Google Scholar] [CrossRef]
- Uloza, V.; Kuzminiene, A.; Palubinskiene, J.; Balnyte, I.; Uloziene, I.; Valanciute, A. Model of human recurrent respiratory papilloma on chicken embryo chorioallantoic membrane for tumor angiogenesis research. Histol. Histopathol. 2017, 32, 699–710. [Google Scholar] [PubMed]
- Auerbach, R.; Kubai, L.; Sidky, Y. Angiogenesis induction by tumors, embryonic tissues, and lymphocytes. Cancer Res. 1976, 36, 3435–3440. [Google Scholar] [PubMed]
- Murray, B.; Wilson, D. A study of metabolites as intermediate effectors in angiogenesis. Angiogenesis 2001, 4, 71–77. [Google Scholar] [CrossRef]
- Samad, N.A.; Abdul, A.B.; Rahman, H.S.; Rasedee, A.; Ibrahim, T.A.T.; Keon, Y.S. Zerumbone suppresses angiogenesis in HepG2 cells through inhibition of matrix metalloproteinase-9, vascular endothelial growth factor, and vascular endothelial growth factor receptor expressions. Pharmacogn. Mag. 2017, 13 (Suppl. S4), S731. [Google Scholar]
- Gurel-Gurevin, E.; Kiyan, H.T.; Esener, O.B.B.; Aydinlik, S.; Uvez, A.; Ulukaya, E.; Dimas, K.; Armutak, E.I. Chloroquine used in combination with chemotherapy synergistically suppresses growth and angiogenesis in vitro and in vivo. Anticancer Res. 2018, 38, 4011–4020. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Y.; Wang, Y.; Ma, Y.; Lan, Y.; Yang, X. Transforming growth factor β-regulated microRNA-29a promotes angiogenesis through targeting the phosphatase and tensin homolog in endothelium. J. Biol. Chem. 2013, 288, 10418–10426. [Google Scholar] [CrossRef] [Green Version]
- Bushati, N.; Cohen, S.M. microRNA functions. Annu. Rev. Cell Dev. Biol. 2007, 23, 175–205. [Google Scholar] [CrossRef] [PubMed]
- Anand, S. A brief primer on microRNAs and their roles in angiogenesis. Vasc. Cell 2013, 5, 2. [Google Scholar] [CrossRef] [Green Version]
- Huang, C.; Luo, W.; Wang, Q.; Ye, Y.; Fan, J.; Lin, L.; Shi, C.; Wei, W.; Chen, H.; Wu, Y. Human mesenchymal stem cells promote ischemic repairment and angiogenesis of diabetic foot through exosome miRNA-21-5p. Stem Cell Res. 2021, 52, 102235. [Google Scholar] [CrossRef] [PubMed]
- Roma-Rodrigues, C.; Fernandes, A.R.; Baptista, P.V. Counteracting the effect of leukemia exosomes by antiangiogenic gold nanoparticles. Int. J. Nanomed. 2019, 14, 6843. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Forough, R.; Wang, X.; Martinez-Lemus, L.A.; Thomas, D.; Sun, Z.; Motamed, K.; Parker, J.L.; Meininger, G.A. Cell-based and direct gene transfer-induced angiogenesis via a secreted chimeric fibroblast growth factor-1 (sp-FGF-1) in the chick chorioallantoic membrane (CAM). Angiogenesis 2003, 6, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Madrigal, J.L.; Sharma, S.N.; Campbell, K.T.; Stilhano, R.S.; Gijsbers, R.; Silva, E.A. Microgels produced using microfluidic on-chip polymer blending for controlled released of VEGF encoding lentivectors. Acta Biomater. 2018, 69, 265–276. [Google Scholar] [CrossRef]
- Augustine, R.; Zahid, A.A.; Hasan, A.; Wang, M.; Webster, T.J. CTGF loaded electrospun dual porous core-shell membrane for diabetic wound healing. Int. J. Nanomed. 2019, 14, 8573. [Google Scholar] [CrossRef] [Green Version]
- Lee, M.-S.; Ghim, J.; Kim, S.-J.; Yun, Y.S.; Yoo, S.-A.; Suh, P.-G.; Kim, W.-U.; Ryu, S.H. Functional interaction between CTGF and FPRL1 regulates VEGF-A-induced angiogenesis. Cell. Signal. 2015, 27, 1439–1448. [Google Scholar] [CrossRef]
- Wilting, J.; Christ, B.; Weich, H.A. The effects of growth factors on the day 13 chorioallantoic membrane (CAM): A study of VEGF 165 and PDGF-BB. Anat. Embryol. 1992, 186, 251–257. [Google Scholar] [CrossRef]
- Bai, Y.; Bai, L.; Zhou, J.; Chen, H.; Zhang, L. Sequential delivery of VEGF, FGF-2 and PDGF from the polymeric system enhance HUVECs angiogenesis in vitro and CAM angiogenesis. Cell. Immunol. 2018, 323, 19–32. [Google Scholar] [CrossRef]
- Miller, W.J.; Kayton, M.L.; Patton, A.; O’Connor, S.; He, M.; Vu, H.; Baibakov, G.; Lorang, D.; Knezevic, V.; Kohn, E. A novel technique for quantifying changes in vascular density, endothelial cell proliferation and protein expression in response to modulators of angiogenesis using the chick chorioallantoic membrane (CAM) assay. J. Transl. Med. 2004, 2, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, W.; Yalcin, M.; Bharali, D.J.; Lin, Q.; Godugu, K.; Fujioka, K.; Keating, K.A.; Mousa, S.A. Pharmacokinetics, biodistribution, and anti-angiogenesis efficacy of diamino propane tetraiodothyroacetic acid-conjugated biodegradable polymeric nanoparticle. Sci. Rep. 2019, 9, 9006. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Wang, Q.; Fei, T.; Han, J.-D.J.; Chen, Y.-G. MCP-1 mediates TGF-β–induced angiogenesis by stimulating vascular smooth muscle cell migration. Blood 2007, 109, 987–994. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shanmuganathan, S.; Angayarkanni, N. Chebulagic acid Chebulinic acid and Gallic acid, the active principles of Triphala, inhibit TNFα induced pro-angiogenic and pro-inflammatory activities in retinal capillary endothelial cells by inhibiting p38, ERK and NFkB phosphorylation. Vasc. Pharmacol. 2018, 108, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Taktak-Benamar, A.; Morjen, M.; Mabrouk, H.B.; Abdelmaksoud-Dammak, R.; Guerfali, M.; Fourati-Masmoudi, N.; Marrakchi, N.; Gargouri, A. Expression, purification and functionality of bioactive recombinant human vascular endothelial growth factor VEGF 165 in E. coli. AMB Express 2017, 7, 33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hlushchuk, R.; Ehrbar, M.; Reichmuth, P.; Heinimann, N.; Styp-Rekowska, B.; Escher, R.; Baum, O.; Lienemann, P.; Makanya, A.; Keshet, E. Decrease in VEGF expression induces intussusceptive vascular pruning. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 2836–2844. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilting, J.; Birkenhäger, R.; Eichmann, A.; Kurz, H.; Martiny-Baron, G.; Marmé, D.; McCarthy, J.E.; Christ, B.; Weich, H.A. VEGF 121 Induces Proliferation of Vascular Endothelial Cells and Expression of flk-1 without Affecting Lymphatic Vessels of the Chorioallantoic Membrane. Dev. Biol. 1996, 176, 76–85. [Google Scholar] [CrossRef]
- Abraham, S.; Rangaswamy, S.P.; Chinnaiah, A. Evaluation of recombinant human vascular endothelial growth factor VEGF 121-loaded poly-l-lactide microparticles as a controlled release delivery system. Turk. J. Biol. 2020, 44, 34–47. [Google Scholar]
- Yum, S.; Jeong, S.; Kim, D.; Lee, S.; Kim, W.; Yoo, J.-W.; Kim, J.; Kwon, O.S.; Kim, D.-D.; Min, D.S. Minoxidil induction of VEGF is mediated by inhibition of HIF-prolyl hydroxylase. Int. J. Mol. Sci. 2018, 19, 53. [Google Scholar] [CrossRef] [Green Version]
- Vázquez, F.; Hastings, G.; Ortega, M.-A.; Lane, T.F.; Oikemus, S.; Lombardo, M.; Iruela-Arispe, M.L. METH-1, a human ortholog of ADAMTS-1, and METH-2 are members of a new family of proteins with angio-inhibitory activity. J. Biol. Chem. 1999, 274, 23349–23357. [Google Scholar] [CrossRef] [Green Version]
- Fernandez, M.; Bonkovsky, H.L. Vascular endothelial growth factor increases heme oxygenase-1 protein expression in the chick embryo chorioallantoic membrane. Br. J. Pharmacol. 2003, 139, 634–640. [Google Scholar] [CrossRef]
- Campbell, K.T.; Hadley, D.J.; Kukis, D.L.; Silva, E.A. Alginate hydrogels allow for bioactive and sustained release of VEGF-C and VEGF-D for lymphangiogenic therapeutic applications. PLoS ONE 2017, 12, e0181484. [Google Scholar] [CrossRef] [Green Version]
- Cao, Y.; Linden, P.; Farnebo, J.; Cao, R.; Eriksson, A.; Kumar, V.; Qi, J.-H.; Claesson-Welsh, L.; Alitalo, K. Vascular endothelial growth factor C induces angiogenesis in vivo. Proc. Natl. Acad. Sci. USA 1998, 95, 14389–14394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malik, M.H.; Shahzadi, L.; Batool, R.; Safi, S.Z.; Khan, A.S.; Khan, A.F.; Chaudhry, A.A.; Rehman, I.U.; Yar, M. Thyroxine-loaded chitosan/carboxymethyl cellulose/hydroxyapatite hydrogels enhance angiogenesis in in-ovo experiments. Int. J. Biol. Macromol. 2020, 145, 1162–1170. [Google Scholar] [CrossRef]
- Nikhil, K.; Sharan, S.; Wishard, R.; Palla, S.R.; Peddinti, R.K.; Roy, P. Pterostilbene carboxaldehyde thiosemicarbazone, a resveratrol derivative inhibits 17β-Estradiol induced cell migration and proliferation in HUVECs. Steroids 2016, 108, 17–30. [Google Scholar] [CrossRef]
- Pullar, C.E.; Le Provost, G.S.; O’leary, A.P.; Evans, S.E.; Baier, B.S.; Isseroff, R.R. β2AR antagonists and β2AR gene deletion both promote skin wound repair processes. J. Investig. Dermatol. 2012, 132, 2076–2084. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gho, Y.S.; Chae, C.-B. Anti-angiogenin activity of the peptides complementary to the receptor-binding site of angiogenin. J. Biol. Chem. 1997, 272, 24294–24299. [Google Scholar] [CrossRef] [Green Version]
- Dusseau, J.W.; Hutchins, P.M.; Malbasa, D.S. Stimulation of angiogenesis by adenosine on the chick chorioallantoic membrane. Circ. Res. 1986, 59, 163–170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oranger, A.; Brunetti, G.; Colaianni, G.; Tamma, R.; Carbone, C.; Lippo, L.; Mori, G.; Pignataro, P.; Cirulli, N.; Zerlotin, R. Sclerostin stimulates angiogenesis in human endothelial cells. Bone 2017, 101, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, Y.; Lu, Q.; Xin, W.; Cui, W.; Zhu, J. Organoarsenic roxarsone promotes angiogenesis in vivo. Basic Clin. Pharmacol. Toxicol. 2016, 118, 259–270. [Google Scholar] [CrossRef]
- Soucy, N.V.; Ihnat, M.A.; Kamat, C.D.; Hess, L.; Post, M.J.; Klei, L.R.; Clark, C.; Barchowsky, A. Arsenic stimulates angiogenesis and tumorigenesis in vivo. Toxicol. Sci. 2003, 76, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Augustine, R.; Dalvi, Y.B.; Nath, V.Y.; Varghese, R.; Raghuveeran, V.; Hasan, A.; Thomas, S.; Sandhyarani, N. Yttrium oxide nanoparticle loaded scaffolds with enhanced cell adhesion and vascularization for tissue engineering applications. Mater. Sci. Eng. C 2019, 103, 109801. [Google Scholar] [CrossRef]
- Carmeliet, P.; Jain, R.K. Angiogenesis in cancer and other diseases. Nature 2000, 407, 249. [Google Scholar] [CrossRef]
- Nishida, N.; Yano, H.; Nishida, T.; Kamura, T.; Kojiro, M. Angiogenesis in cancer. Vasc. Health Risk Manag. 2006, 2, 213. [Google Scholar] [CrossRef] [PubMed]
- Gunasekaran, V.P.; Nishi, K.; Sivakumar, D.; Sivaraman, T.; Mathan, G. Identification of 2, 4-dihydroxy-5-pyrimidinyl imidothiocarbomate as a novel inhibitor to Y box binding protein-1 (YB-1) and its therapeutic actions against breast cancer. Eur. J. Pharm. Sci. 2018, 116, 2–14. [Google Scholar] [CrossRef]
- Homayouni-Tabrizi, M.; Soltani, M.; Karimi, E.; Namvar, F.; Pouresmaeil, V.; Es-Haghi, A. Putative mechanism for anticancer properties of Ag–PP (NPs) extract. IET Nanobiotechnol. 2019, 13, 617–620. [Google Scholar] [CrossRef]
- Ferrara, N.; Hillan, K.J.; Novotny, W. Bevacizumab (Avastin), a humanized anti-VEGF monoclonal antibody for cancer therapy. Biochem. Biophys. Res. Commun. 2005, 333, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Feflea, S.; Cimpean, A.M.; Ceausu, R.A.; Gaje, P.; Raica, M. Effects of antibodies to EG-VEGF on angiogenesis in the chick embryo chorioallantoic membrane. In Vivo 2012, 26, 793–797. [Google Scholar]
- Vitaliti, A.; Wittmer, M.; Steiner, R.; Wyder, L.; Neri, D.; Klemenz, R. Inhibition of tumor angiogenesis by a single-chain antibody directed against vascular endothelial growth factor. Cancer Res. 2000, 60, 4311–4314. [Google Scholar]
- Arezumand, R.; Mahdian, R.; Zeinali, S.; Hassanzadeh-Ghassabeh, G.; Mansouri, K.; Khanahmad, H.; Namvar-Asl, N.; Rahimi, H.; Behdani, M.; Cohan, R.A. Identification and characterization of a novel nanobody against human placental growth factor to modulate angiogenesis. Mol. Immunol. 2016, 78, 183–192. [Google Scholar] [CrossRef]
- Sanz, L.; Kristensen, P.; Blanco, B.; Facteau, S.; Russell, S.; Winter, G.; Alvarez-Vallina, L. Single-chain antibody-based gene therapy: Inhibition of tumor growth by in situ production of phage-derived human antibody fragments blocking functionally active sites of cell-associated matrices. Gene Ther. 2002, 9, 1049–1053. [Google Scholar] [CrossRef] [Green Version]
- Chiang, K.-C.; Sun, C.-C.; Chen, M.-H.; Huang, C.-Y.; Hsu, J.-T.; Yeh, T.-S.; Chen, L.-W.; Kuo, S.-F.; Juang, H.-H.; Takano, M. MART-10, the new brand of 1α, 25 (OH) 2D3 analog, is a potent anti-angiogenic agent in vivo and in vitro. J. Steroid Biochem. Mol. Biol. 2016, 155, 26–34. [Google Scholar] [CrossRef]
- Kim, A.; Ma, J.Y. Rhaponticin decreases the metastatic and angiogenic abilities of cancer cells via suppression of the HIF-1α pathway. Int. J. Oncol. 2018, 53, 1160–1170. [Google Scholar] [CrossRef] [PubMed]
- Azhar, A.; Khan, M.S.; Swaminathan, A.; Naseem, A.; Chatterjee, S.; Jairajpuri, M.A. Oxidized antithrombin is a dual inhibitor of coagulation and angiogenesis: Importance of low heparin affinity. Int. J. Biol. Macromol. 2016, 82, 541–550. [Google Scholar] [CrossRef]
- Babae, N.; Bourajjaj, M.; Liu, Y.; Van Beijnum, J.R.; Cerisoli, F.; Scaria, P.V.; Verheul, M.; Van Berkel, M.P.; Pieters, E.H.; Van Haastert, R.J. Systemic miRNA-7 delivery inhibits tumor angiogenesis and growth in murine xenograft glioblastoma. Oncotarget 2014, 5, 6687. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brandwijk, R.J.; Nesmelova, I.; Dings, R.P.; Mayo, K.H.; Thijssen, V.L.; Griffioen, A.W. Cloning an artificial gene encoding angiostatic anginex: From designed peptide to functional recombinant protein. Biochem. Biophys. Res. Commun. 2005, 333, 1261–1268. [Google Scholar] [CrossRef]
- Griffioen, A.; van der Schaft, D.; Barendsz-Janson, A.; Cox, A.; Boudier, H.S.; Hillen, H.; Mayo, K. Anginex, a designed peptide that inhibits angiogenesis. Biochem. J. 2001, 354, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Célérier, J.; Cruz, A.; Lamandé, N.; Gasc, J.-M.; Corvol, P. Angiotensinogen and its cleaved derivatives inhibit angiogenesis. Hypertension 2002, 39, 224–228. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghazaryan, N.; Movsisyan, N.; Macedo, J.C.; Vaz, S.; Ayvazyan, N.; Pardo, L.; Logarinho, E. The antitumor efficacy of monomeric disintegrin obtustatin in S-180 sarcoma mouse model. Investig. New Drugs 2019, 37, 1044–1051. [Google Scholar] [CrossRef] [Green Version]
- Loizzi, V.; Del Vecchio, V.; Gargano, G.; De Liso, M.; Kardashi, A.; Naglieri, E.; Resta, L.; Cicinelli, E.; Cormio, G. Biological pathways involved in tumor angiogenesis and bevacizumab based anti-angiogenic therapy with special references to ovarian cancer. Int. J. Mol. Sci. 2017, 18, 1967. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chaplain, M. The mathematical modelling of tumour angiogenesis and invasion. Acta Biotheor. 1995, 43, 387–402. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [Green Version]
- Becker, J.; Covelo-Fernandez, A.; von Bonin, F.; Kube, D.; Wilting, J. Specific tumor-stroma interactions of EBV-positive Burkitt’s lymphoma cells in the chick chorioallantoic membrane. Vasc. Cell 2012, 4, 3. [Google Scholar] [CrossRef] [Green Version]
- Vilahur, G.; Oñate, B.; Cubedo, J.; Béjar, M.T.; Arderiu, G.; Peña, E.; Casaní, L.; Gutiérrez, M.; Capdevila, A.; Pons-Lladó, G. Allogenic adipose-derived stem cell therapy overcomes ischemia-induced microvessel rarefaction in the myocardium: Systems biology study. Stem Cell Res. Ther. 2017, 8, 52. [Google Scholar] [CrossRef] [Green Version]
- Reuter, A.; Sckell, A.; Brandenburg, L.-O.; Burchardt, M.; Kramer, A.; Stope, M.B. Overexpression of MicroRNA-1 in Prostate Cancer Cells Modulates the Blood Vessel System of an In Vivo Hen’s Egg Test–Chorioallantoic Membrane Model. In Vivo 2019, 33, 41–46. [Google Scholar] [CrossRef] [Green Version]
- Wolint, P.; Bopp, A.; Woloszyk, A.; Tian, Y.; Evrova, O.; Hilbe, M.; Giovanoli, P.; Calcagni, M.; Hoerstrup, S.P.; Buschmann, J. Cellular self-assembly into 3D microtissues enhances the angiogenic activity and functional neovascularization capacity of human cardiopoietic stem cells. Angiogenesis 2019, 22, 37–52. [Google Scholar] [CrossRef] [PubMed]
- Ribatti, D.; Nico, B.; Cimpean, A.M.; Raica, M.; Crivellato, E.; Ruggieri, S.; Vacca, A. B16-F10 melanoma cells contribute to the new formation of blood vessels in the chick embryo chorioallantoic membrane through vasculogenic mimicry. Clin. Exp. Med. 2013, 13, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Isachenko, V.; Mallmann, P.; Petrunkina, A.M.; Rahimi, G.; Nawroth, F.; Hancke, K.; Felberbaum, R.; Genze, F.; Damjanoski, I.; Isachenko, E. Comparison of in vitro-and chorioallantoic membrane (CAM)-culture systems for cryopreserved medulla-contained human ovarian tissue. PLoS ONE 2012, 7, e32549. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kunzi-Rapp, K.; Kaskel, P.; Steiner, R.; Uwe Peter, R.; Krahn, G. Increased blood levels of Human S100 in melanoma chick embryo xenografts’ circulation. Pigment Cell Res. 2001, 14, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Marzullo, A.; Vacca, A.; Roncali, L.; Pollice, L.; Ribatti, D. Angiogenesis in hepatocellular carcinoma: An experimental study in the chick embryo chorioallantoic membrane. Int. J. Oncol. 1998, 13, 17–38. [Google Scholar] [CrossRef]
- Ismail, M.; Torsten, U.; Dressler, C.; Diederichs, J.; Hüske, S.; Weitzel, H.; Berlien, H.-P. Photodynamic therapy of malignant ovarian tumours cultivated on CAM. Lasers Med. Sci. 1999, 14, 91–96. [Google Scholar] [CrossRef]
- Jiang, W.; Zhu, P.; Zhang, T.; Liao, F.; Yu, Y.; Liu, Y.; Shen, H.; Zhao, Z.; Huang, X.; Zhou, N. MicroRNA-205 mediates endothelial progenitor functions in distraction osteogenesis by targeting the transcription regulator NOTCH2. Stem Cell Res. Ther. 2021, 12, 101. [Google Scholar] [CrossRef]
- Huang, C.-C.; Tseng, T.-T.; Liu, S.-C.; Lin, Y.-Y.; Law, Y.-Y.; Hu, S.-L.; Wang, S.-W.; Tsai, C.-H.; Tang, C.-H. S1P Increases VEGF Production in Osteoblasts and Facilitates Endothelial Progenitor Cell Angiogenesis by Inhibiting miR-16-5p Expression via the c-Src/FAK Signaling Pathway in Rheumatoid Arthritis. Cells 2021, 10, 2168. [Google Scholar] [CrossRef]
- Zhang, T.; Liu, W.; Zeng, X.-C.; Jiang, N.; Fu, B.-S.; Guo, Y.; Yi, H.-M.; Li, H.; Zhang, Q.; Chen, W.-J. Down-regulation of microRNA-338-3p promoted angiogenesis in hepatocellular carcinoma. Biomed. Pharmacother. 2016, 84, 583–591. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Yu, W.; Qu, X.; Wang, R.; Xu, J.; Zhang, Q.; Xu, J.; Li, J.; Chen, L. Argonaute 2 promotes myeloma angiogenesis via microRNA dysregulation. J. Hematol. Oncol. 2014, 7, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Han, Z.-P.; Zhang, S.-S.; Jing, Y.-Y.; Bu, X.-X.; Wang, C.-Y.; Sun, K.; Jiang, G.-C.; Zhao, X.; Li, R. Effects of inflammatory factors on mesenchymal stem cells and their role in the promotion of tumor angiogenesis in colon cancer. J. Biol. Chem. 2011, 286, 25007–25015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zacchigna, S.; Zentilin, L.; Morini, M.; Dell’Eva, R.; Noonan, D.M.; Albini, A.; Giacca, M. AAV-mediated gene transfer of tissue inhibitor of metalloproteinases-1 inhibits vascular tumor growth and angiogenesis in vivo. Cancer Gene Ther. 2004, 11, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Zhang, Z.; Zhao, H.; Zhu, D.; Zhang, Y.; Li, D. Anti-angiogenesis by lentivirus-mediated small interfering RNA silencing of angiopoietin-2 gene in pancreatic carcinoma. Technol. Cancer Res. Treat. 2011, 10, 361–369. [Google Scholar] [CrossRef]
- Liu, S.-C.; Chuang, S.; Hsu, C.; Tsai, C.; Wang, S.; Tang, C. CTGF increases vascular endothelial growth factor-dependent angiogenesis in human synovial fibroblasts by increasing miR-210 expression. Cell Death Dis. 2014, 5, e1485. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, J.; Dong, X.; Xu, Z.; Jiang, X.; Jiang, H.; Krissansen, G.W.; Sun, X. Endostatin gene therapy enhances the efficacy of paclitaxel to suppress breast cancers and metastases in mice. J. Biomed. Sci. 2008, 15, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Gu, M.; Zhang, K.; Yao, H.-J.; Zhou, J.; Peng, Y.-B.; Xu, M.-X.; Wang, Z. RNAi-mediated knockdown of vascular endothelial growth factor inhibits vascularization and tumor growth in renal cell carcinoma. Int. J. Mol. Med. 2015, 36, 1063–1072. [Google Scholar] [CrossRef]
- Liu, G.-T.; Huang, Y.-L.; Tzeng, H.-E.; Tsai, C.-H.; Wang, S.-W.; Tang, C.-H. CCL5 promotes vascular endothelial growth factor expression and induces angiogenesis by down-regulating miR-199a in human chondrosarcoma cells. Cancer Lett. 2015, 357, 476–487. [Google Scholar] [CrossRef] [PubMed]
- Xiao, T.; Fan, J.K.; Huang, H.L.; Gu, J.F.; Li, L.-Y.; Liu, X.Y. VEGI-armed oncolytic adenovirus inhibits tumor neovascularization and directly induces mitochondria-mediated cancer cell apoptosis. Cell Res. 2010, 20, 367–378. [Google Scholar] [CrossRef]
- Hu, C.C.; Ji, H.M.; Chen, S.L.; Zhang, H.W.; Wang, B.Q.; Zhou, L.Y.; Zhang, Z.P.; Sun, X.L.; Chen, Z.Z.; Cai, Y.Q. Investigation of a plasmid containing a novel immunotoxin VEGF165-PE38 gene for antiangiogenic therapy in a malignant glioma model. Int. J. Cancer 2010, 127, 2222–2229. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Kuscu, C.; Banach, A.; Zhang, Q.; Pulkoski-Gross, A.; Kim, D.; Liu, J.; Roth, E.; Li, E.; Shroyer, K.R. miR-181a-5p inhibits cancer cell migration and angiogenesis via downregulation of matrix metalloproteinase-14. Cancer Res. 2015, 75, 2674–2685. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, T.-H.; Hur, E.-g.; Kang, S.-J.; Kim, J.-A.; Thapa, D.; Lee, Y.M.; Ku, S.K.; Jung, Y.; Kwak, M.-K. NRF2 blockade suppresses colon tumor angiogenesis by inhibiting hypoxia-induced activation of HIF-1α. Cancer Res. 2011, 71, 2260–2275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bernard, H.; Garmy-Susini, B.; Ainaoui, N.; Van Den Berghe, L.; Peurichard, A.; Javerzat, S.; Bikfalvi, A.; Lane, D.P.; Bourdon, J.-C.; Prats, A.-C. The p53 isoform, Δ133p53α, stimulates angiogenesis and tumour progression. Oncogene 2013, 32, 2150–2160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, S.; Guo, R.; Li, P.Z.; Li, K.; Yan, Y.; Chen, J.; Wang, G.; Brand-Saberi, B.; Yang, X.; Cheng, X. Dexamethasone interferes with osteoblasts formation during osteogenesis through altering IGF-1-mediated angiogenesis. J. Cell. Physiol. 2019, 234, 15167–15181. [Google Scholar] [CrossRef]
- Rao, D.D.; Vorhies, J.S.; Senzer, N.; Nemunaitis, J. siRNA vs. shRNA: Similarities and differences. Adv. Drug Deliv. Rev. 2009, 61, 746–759. [Google Scholar] [CrossRef] [PubMed]
- Balke, M.; Neumann, A.; Kersting, C.; Agelopoulos, K.; Gebert, C.; Gosheger, G.; Buerger, H.; Hagedorn, M. Morphologic characterization of osteosarcoma growth on the chick chorioallantoic membrane. BMC Res. Notes 2010, 3, 58. [Google Scholar] [CrossRef]
- Jilani, S.M.; Murphy, T.J.; Thai, S.N.; Eichmann, A.; Alva, J.A.; Iruela-Arispe, M.L. Selective binding of lectins to embryonic chicken vasculature. J. Histochem. Cytochem. 2003, 51, 597–604. [Google Scholar] [CrossRef] [Green Version]
- Shereema, R.; Sruthi, T.; Kumar, V.S.; Rao, T.; Shankar, S.S. Angiogenic profiling of synthesized carbon quantum dots. Biochemistry 2015, 54, 6352–6356. [Google Scholar] [CrossRef]
- Isachenko, V.; Orth, I.; Isachenko, E.; Mallmann, P.; Peters, D.; Schmidt, T.; Morgenstern, B.; Foth, D.; Hanstein, B.; Rahimi, G. Viability of human ovarian tissue confirmed 5 years after freezing with spontaneous ice-formation by autografting and chorio-allantoic membrane culture. Cryobiology 2013, 66, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Larger, E.; Marre, M.; Corvol, P.; Gasc, J.-M. Hyperglycemia-induced defects in angiogenesis in the chicken chorioallantoic membrane model. Diabetes 2004, 53, 752–761. [Google Scholar] [CrossRef] [Green Version]
- Kleibeuker, E.A.; ten Hooven, M.A.; Castricum, K.C.; Honeywell, R.; Griffioen, A.W.; Verheul, H.M.; Slotman, B.J.; Thijssen, V.L. Optimal treatment scheduling of ionizing radiation and sunitinib improves the antitumor activity and allows dose reduction. Cancer Med. 2015, 4, 1003–1015. [Google Scholar] [CrossRef]
- Jiang, X.; Xiong, Q.; Xu, G.; Lin, H.; Fang, X.; Cui, D.; Xu, M.; Chen, F.; Geng, H. VEGF-loaded nanoparticle-modified BAMAs enhance angiogenesis and inhibit graft shrinkage in tissue-engineered bladder. Ann. Biomed. Eng. 2015, 43, 2577–2586. [Google Scholar] [CrossRef] [PubMed]
- Brash, J.T.; Ruhrberg, C.; Fantin, A. Evaluating vascular hyperpermeability-inducing agents in the skin with the Miles assay. J. Vis. Exp. 2018, 136, 57524. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, V.; Kim, D.; Durán, W.N.; DeFouw, D.O. Ontogeny of microvascular permeability to macromolecules in the chick chorioallantoic membrane during normal angiogenesis. Microvasc. Res. 1995, 49, 49–63. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, V.; DeFouw, D.O. Capillary sprouts restrict macromolecular extravasation during normal angiogenesis in the chick chorioallantoic membrane. Microvasc. Res. 1996, 52, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, V.; De Fouw, D. Microvascular permselectivity in the chick chorioallantoic membrane during endothelial cell senescence. Int. J. Microcirc. 1997, 17, 75–79. [Google Scholar] [CrossRef]
- Saleh, A.I.; Mohamed, I.; Mohamed, A.A.; Abdelkader, M.; Yalcin, H.C.; Aboulkassim, T.; Batist, G.; Yasmeen, A.; Moustafa, A.-E.A. Elaeagnus angustifolia plant extract inhibits angiogenesis and downgrades cell invasion of human oral cancer cells via Erk1/Erk2 inactivation. Nutr. Cancer 2018, 70, 297–305. [Google Scholar] [CrossRef]
- Aanhane, E.; Schulkens, I.A.; Heusschen, R.; Castricum, K.; Leffler, H.; Griffioen, A.W.; Thijssen, V.L. Different angioregulatory activity of monovalent galectin-9 isoforms. Angiogenesis 2018, 21, 545–555. [Google Scholar] [CrossRef] [Green Version]
- Borjan, B.; Steiner, N.; Karbon, S.; Kern, J.; Francesch, A.; Hermann, M.; Willenbacher, W.; Gunsilius, E.; Untergasser, G. The Aplidin analogs PM01215 and PM02781 inhibit angiogenesis in vitro and in vivo. BMC Cancer 2015, 15, 738. [Google Scholar] [CrossRef] [Green Version]
- Heuberger, D.M.; Harankhedkar, S.; Morgan, T.; Wolint, P.; Calcagni, M.; Lai, B.; Fahrni, C.J.; Buschmann, J. High-affinity Cu (I) chelator PSP-2 as potential anti-angiogenic agent. Sci. Rep. 2019, 9, 14055. [Google Scholar] [CrossRef] [Green Version]
- Melkonian, G.; Munoz, N.; Chung, J.; Tong, C.; Marr, R.; Talbot, P. Capillary plexus development in the day five to day six chick chorioallantoic membrane is inhibited by cytochalasin D and ssuramin. J. Exp. Zool. 2002, 292, 241–254. [Google Scholar] [CrossRef]
- Magalhães, L.F.; Mello-Andrade, F.; Pires, W.C.; Silva, H.D.; da Silva, P.F.F.; Macedo, L.M.; de Castro, C.H.; Carneiro, C.C.; Cardoso, C.G.; de Melo Reis, P.R. cis-[RuCl (BzCN)(bipy)(dppe)] PF6 induces anti-angiogenesis and apoptosis by a mechanism of caspase-dependent involving DNA damage, PARP activation, and Tp53 induction in Ehrlich tumor cells. Chemico-Biol. Interact. 2017, 278, 101–113. [Google Scholar] [CrossRef]
- Prieto, C.P.; Ortiz, M.C.; Villanueva, A.; Villarroel, C.; Edwards, S.S.; Elliott, M.; Lattus, J.; Aedo, S.; Meza, D.; Lois, P. Netrin-1 acts as a non-canonical angiogenic factor produced by human Wharton’s jelly mesenchymal stem cells (WJ-MSC). Stem Cell Res. Ther. 2017, 8, 43. [Google Scholar] [CrossRef] [Green Version]
- de Castro, J.V.; Gomes, E.D.; Granja, S.; Anjo, S.I.; Baltazar, F.; Manadas, B.; Salgado, A.J.; Costa, B.M. Impact of mesenchymal stem cells’ secretome on glioblastoma pathophysiology. J. Transl. Med. 2017, 15, 200. [Google Scholar] [CrossRef]
- Rizzo, V.; Defouw, D.O. Mast cell activation accelerates the normal rate of angiogenesis in the chick chorioallantoic membrane. Microvasc. Res. 1996, 52, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Burggren, W.; Rojas Antich, M. Angiogenesis in the Avian Embryo Chorioallantoic Membrane: A Perspective on Research Trends and a Case Study on Toxicant Vascular Effects. J. Cardiovasc. Dev. Dis. 2020, 7, 56. [Google Scholar] [CrossRef]
- DeFouw, D.O.; Rizzo, V.J.; Steinfeld, R.; Feinberg, R.N. Mapping of the microcirculation in the chick chorioallantoic membrane during normal angiogenesis. Microvasc. Res. 1989, 38, 136–147. [Google Scholar] [CrossRef]
- Dimitropoulou, C.; Malkusch, W.; Fait, E.; Maragoudakis, M.; Konerding, M. The vascular architecture of the chick chorioallantoic membrane: Sequential quantitative evaluation using corrosion casting. Angiogenesis 1998, 2, 255–263. [Google Scholar] [CrossRef]
- Cornillie, P.; Casteleyn, C.; von Horst, C.; Henry, R. Corrosion casting in anatomy: Visualizing the architecture of hollow structures and surface details. Anatomia Histol. Embryol. 2019, 48, 591–604. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hossler, F.E.; Douglas, J.E. Vascular corrosion casting: Review of advantages and limitations in the application of some simple quantitative methods. Microsc. Microanal. 2001, 7, 253–264. [Google Scholar] [CrossRef]
- Ackermann, M.; Konerding, M.A. Vascular casting for the study of vascular morphogenesis. In Vascular Morphogenesis; Springer: Berlin/Heidelberg, Germany, 2015; pp. 49–66. [Google Scholar]
- Krucker, T.; Lang, A.; Meyer, E.P. New polyurethane-based material for vascular corrosion casting with improved physical and imaging characteristics. Microsc. Res. Tech. 2006, 69, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Djonov, V.G.; Kurz, H.; Burri, P.H. Optimality in the developing vascular system: Branching remodeling by means of intussusception as an efficient adaptation mechanism. Dev. Dyn. 2002, 224, 391–402. [Google Scholar] [CrossRef]
- Siamblis, D.; Karnabatidis, D.; Hatjikondi, O.; Kalogeropoulou, C.; Kardamakis, D.; Dimopoulos, J. A novel radiological approach for the experimental study of angiogenesis: Angiography of the chick embryo and its chorioallantoic membrane. Eur. J. Radiol. 1996, 21, 220–224. [Google Scholar] [CrossRef]
- Nikiforidis, G.; Papazafiropoulos, D.; Siablis, D.; Karnabatidis, D.; Hatjikondi, O.; Dimopoulos, J. Quantitative assessment of angiogenesis in the chick embryo and its chorioallantoic membrane by computerised analysis of angiographic images. Eur. J. Radiol. 1999, 29, 168–179. [Google Scholar] [CrossRef]
- Rovithi, M.; Avan, A.; Funel, N.; Leon, L.G.; Gomez, V.E.; Wurdinger, T.; Griffioen, A.W.; Verheul, H.M.; Giovannetti, E. Development of bioluminescent chick chorioallantoic membrane (CAM) models for primary pancreatic cancer cells: A platform for drug testing. Sci. Rep. 2017, 7, 44686. [Google Scholar] [CrossRef]
- Zhang, P.; Zhong, S.; Wang, G.; Zhang, S.-Y.; Chu, C.; Zeng, S.; Yan, Y.; Cheng, X.; Bao, Y.; Hocher, B. N-acetylcysteine suppresses LPS-induced pathological angiogenesis. Cell. Physiol. Biochem. 2018, 49, 2483–2495. [Google Scholar] [CrossRef] [PubMed]
- Maacha, S.; Saule, S. Evaluation of tumor cell invasiveness in vivo: The chick chorioallantoic membrane assay. In Cell Migration; Humana Press: New York, NY, USA, 2018; pp. 71–77. [Google Scholar]
- Cardelli, M. Alu pcr. In PCR Protocols; Springer: Berlin/Heidelberg, Germany, 2011; pp. 221–229. [Google Scholar]
- Mira, E.; Lacalle, R.A.; Gómez-Moutón, C.; Leonardo, E.; Mañes, S. Quantitative determination of tumor cell intravasation in a real-time polymerase chain reaction-based assay. Clin. Exp. Metastasis 2002, 19, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Yu, W.; Kovalski, K.; Ossowski, L. Requirement for specific proteases in cancer cell intravasation as revealed by a novel semiquantitative PCR-based assay. Cell 1998, 94, 353–362. [Google Scholar] [CrossRef] [Green Version]
- Zijlstra, A.; Mellor, R.; Panzarella, G.; Aimes, R.T.; Hooper, J.D.; Marchenko, N.D.; Quigley, J.P. A quantitative analysis of rate-limiting steps in the metastatic cascade using human-specific real-time polymerase chain reaction. Cancer Res. 2002, 62, 7083–7092. [Google Scholar] [PubMed]
- Horst, E.H.v.d.; Leupold, J.H.; Schubbert, R.; Ullrich, A.; Allgayer, H. TaqMan®-based quantification of invasive cells in the chick embryo metastasis assay. Biotechniques 2004, 37, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Zijlstra, A.; Seandel, M.; Kupriyanova, T.A.; Partridge, J.J.; Madsen, M.A.; Hahn-Dantona, E.A.; Quigley, J.P.; Deryugina, E.I. Proangiogenic role of neutrophil-like inflammatory heterophils during neovascularization induced by growth factors and human tumor cells. Blood 2006, 107, 317–327. [Google Scholar] [CrossRef] [Green Version]
- Mangieri, D.; Nico, B.; Benagiano, V.; De Giorgis, M.; Vacca, A.; Ribatti, D. Angiogenic activity of multiple myeloma endothelial cells in vivo in the chick embryo chorioallantoic membrane assay is associated to a down-regulation in the expression of endogenous endostatin. J. Cell. Mol. Med. 2008, 12, 1023–1028. [Google Scholar] [CrossRef] [PubMed]
- Ribatti, D.; Urbinati, C.; Nico, B.; Rusnati, M.; Roncali, L.; Presta, M. Endogenous basic fibroblast growth factor is implicated in the vascularization of the chick embryo chorioallantoic membrane. Dev. Biol. 1995, 170, 39–49. [Google Scholar] [CrossRef] [Green Version]
- Eliceiri, B.P.; Klemke, R.; Strömblad, S.; Cheresh, D.A. Integrin αvβ3 requirement for sustained mitogen-activated protein kinase activity during angiogenesis. J. Cell Biol. 1998, 140, 1255–1263. [Google Scholar] [CrossRef] [Green Version]
- Brooks, P.C.; Silletti, S.; von Schalscha, T.L.; Friedlander, M.; Cheresh, D.A. Disruption of angiogenesis by PEX, a noncatalytic metalloproteinase fragment with integrin binding activity. Cell 1998, 92, 391–400. [Google Scholar] [CrossRef] [Green Version]


| Scaffold/Delivery Method | Reference |
|---|---|
| Collagen | [54,55,56,57] |
| Filter disc | [58,59,60,61,62,63,64,65] |
| Gelatin sponge | [66,67,68,69,70,71] |
| Glass discs | [72,73] |
| Hydrogel | [41,74,75,76,77] |
| Injected | [78,79,80,81,82,83,84] |
| Matrigel | [16,85,86,87,88,89,90] |
| Methylcellulose disc | [15,91,92,93,94] |
| Microspheres | [95,96] |
| Pipetted onto surface | [97,98,99,100,101,102,103] |
| Plastic ring | [75,104,105,106,107,108,109,110,111] |
| Scaffold | [108,112,113,114,115,116,117] |
| Thermanox coverslip | [118,119,120,121,122] |
| Tumour | [5,123,124,125,126] |
| Pellet | [127,128,129] |
| Treatment | Delivery Method | Angiogenic Outcome | Ref. |
|---|---|---|---|
| Connective tissue growth factor (CTGF) | Scaffold | Significant increase in blood vessel number and diameter following software quantification | [137] |
| Thermanox Coverslips | A dose dependent increase seen by appearance of spoke wheel pattern of blood vessels radiating from on-plants | [138] | |
| Platelet-derived growth factor (PDGF) | Thermanox Coverslips | Macroscopic observations indicated thickening of CAM, but no vascular response | [139] |
| Scaffold | An increased blood vessel density converging towards on-plant observed along with thickening of CAM membrane | [140] | |
| Basic Fibroblast Growth Factor (bFGF/FGF-2) | Scaffold | ||
| Plastic ring | Significant increase in number of blood vessels converging towards on-plant | [110] | |
| Filter disc | Significant increase in mean fluorescent vascular density, measured by pixel intensity | [141] | |
| Increased number of branch points in a region around on-plants | [142] | ||
| Transforming growth factor-β (TGF-β) | Filter disc | Radial formation of new vessels seen in area around on-plants | [143] |
| TNFα | Filter disc | Significant increase in tube length and size as measured by angiogenic software | [144] |
| VEGF-165 | Filter disc | A dose responsive increase in blood vessels in defined area observed | [145] |
| Hydrogel | A time-dependent increase in blood vessel diameter and branching points, measured using angiogenic software | [146] | |
| Thermanox Coverslips | Macroscopic observations saw a dose dependent increase in angiogenesis | [139] | |
| VEGF-121 | Thermanox Coverslips | Macroscopic observations noticed a change in vascular pattern under the treatment area | [147] |
| Filter disc | Software quantified a dose responsive increase in total blood vessel network length | [148] | |
| VEGF-A | Filter disc | Significant increase in sprouting blood vessels within a defined area | [149] |
| Scaffold | Increased blood vessel density observed within a defined area | [140] | |
| Matrigel | Microvascular mapping of the blood vessel network following FITC injection resulted in increased blood vessel density | [150] | |
| Glass fibre filter disc | Significant increase in vessels number quantified in a random square areas of CAM surface | [151] | |
| Plastic ring | Angiogenic software indicated a significant increase in number of branchpoints and average vessel length | [111] | |
| Hydrogel | Significant increase in vessel length, number and vasculogenic index | [74] | |
| Significant increase in vessel number in a region around on-plant | [152] | ||
| VEGF-C | Hydrogel | Significant increase in vessel number in a region around on-plant | [152] |
| Methylcellulose disc | Increase in sprouting blood vessels present within a defined area | [153] | |
| Thyroxine | Hydrogel | Significant increase in vascular penetration of on-plants | [154] |
| Heparin | Hydrogel | Significant increase in vessel number in a region around on-plant | [40] |
| VEGF-D | Hydrogel | Significant increase in sprouting blood vessels present within a defined area | [152] |
| Estradiol | Plastic ring | Calculation of percentage of CAM surface covered by endothelial cells resulted in a significant increase in the mean vessels count | [49] |
| Angiogenic software quantified a significant increase in number of branchpoints and average vessel length | [111] | ||
| Scaffold | Increase in angiogenic response seen by measurement of vasculogenic index | [117] | |
| Estradiol | Filter disc | Increased vascular branching observed within a defined area | [155] |
| L-Arginine | Filter disc | The number of primary, secondary, tertiary, and quaternary blood vessels counted with a significant increase in number of quaternary blood vessels | [3] |
| TGF-β induced miR-29a upregulation | Pipetted | Significant increase in number of blood vessels around on-plant observed | [130] |
| Fibroblast growth factor-1 expression plasmid | Pipetted | Significant increase in number of blood vessels in a region around on-plant observed | [135] |
| VEGF-GFP LV | Microgels | Increased blood vessel development quantified | [136] |
| MSCs-exomes | Not mentioned | Significant promotion of new blood vessel formation | [133] |
| miR-21-5p OE exomes | |||
| Transthyretin | Plastic ring | Significant increase in number of blood vessels growing towards on-plants | [110] |
| Terbutaline | Plastic disc | Counting blood vessels which intersected a concentric circle projected around on-plants observed a significant increase in number of blood vessels | [64] |
| β2AR antagonist | Coverslip | Increased number of blood vessel branch points observed within on-plants | [156] |
| Angiogenin | Thermonox discs | Visibly increased number of blood vessel spoke wheel pattern seen radiating from on-plants | [157] |
| Adenosine | Elvax Polymer pellet | Dose dependent increase in vascular density observed in a region around on-plant | [158] |
| Observation of spoke wheel pattern of blood vessels radiating from on-plants, with a positive result observed in majority of samples | [127] | ||
| ADP | |||
| ATP | |||
| Lactic Acid | |||
| Malate | |||
| Exosomes derived from chronic myeloid leukaemia cells (K562) | Plastic ring | Treatment with a higher concentration resulted in an increase in neovasculature | [134] |
| 2-deoxy-D-ribose | Plastic ring | Angiogenic software calculated a significant increase in number of branchpoints and average vessel length | [111] |
| Sclerostin | Gelatin sponge | Increased number of blood vessels converging towards on-plants observed | [159] |
| Roxarsone | Gelatin sponge | Increased number of neovessels and blood vessel length | [160] |
| Leptin | Gelatin sponge | Software measured significantly increased blood vessel tube length and size | [71] |
| Arsenic | Filter disc | Dose dependent increase in blood vessel number observed, however higher doses resulted in negative effects | [161] |
| Y2O3 nanoparticles | Scaffold | Improved blood vessel formation, vascular branching and blood vessel diameter within the area around scaffolds | [162] |
| Treatment | Delivery Method | Angiogenic Outcome | Ref. |
|---|---|---|---|
| Nicotinamide adenine dinucleotide (NAD) | 10% EVA copolymer Pellet | No spoke wheel pattern was observed radiating from on-plants | [127] |
| Pyruvate | |||
| Succinate Fumarate citrate | |||
| Avastin (Bevacizumab) | Injected | Significantly less vascular nodes and branches were quantified within a defined area | [80] |
| EG-VEGF Antibodies | No significant differences in vessel density observed, but dilated medium and large vessels observed | [168] | |
| Methyl blue | Microspheres | No spoke wheel pattern was observed radiating from on-plants | [96] |
| Chloroquine & Doxorubicin | Agarose pellet | Combination of doxorubicin and chloroquine resulted in strong anti-angiogenic effect on capillaries near on-plants | [129] |
| Avastin (Bevacizumab) | Pipetted | Significant decrease in percentage of surface area occupied by microvessels | [165] |
| Vitamin C | Pipetted | The number of primary, secondary, tertiary, and quaternary blood vessels was counted, with decrease in quaternary blood vessels quantified | [3] |
| MART-10 (Vitamin D analog) | Pipetted | Reduced vessel branch point numbers observed within a defined area | [172] |
| Green nanoparticles | Gelatin sponge | Decrease in vessels length and branch number within a defined area | [166] |
| Rhaponticin | Filter disc | Software determined a significant reduction in total blood vessel length | [173] |
| Thalidomide derivatives | Filter disc | Reduction in vessel number, branch points, neovascularization and total length of vessels | [59] |
| High affinity PGF-specific Nanobody | Filter disc | Significant inhibition of angiogenesis within a defined area | [170] |
| Antithrombin | Filter disc | Potent antiangiogenic activity in blood vessel tubules, networks and branching points | [174] |
| Zinc tungstate nanoparticles | Filter disc | A dose dependent reduction in percentage of surface area occupied by blood vessels was calculated | [61] |
| Gold nanoparticles | Filter disc | Software determined a dose dependent reduction in blood vessel size, length and branch points | [60] |
| Injected | Software determined a significant reduction in vessel length and number of junctions and complexes | [84] | |
| miR-7 mimics | Nitrocellulose rings | A reduction in vascular density within a defined area was visible | [175] |
| Sunitinib (receptor tyrosine kinase inhibitor) | Nitrocellulose rings | A reduction in vascular density within a defined area was visible | [175] |
| Vasohibin Adenovirus | Matrigel | Macroscopic observations saw inhibition of blood vessel growth | [16] |
| Chitosan derivatives nanoparticles | Methylcellulose disc | Reduction in number of blood vessels in contact with on-plants observed | [94] |
| Anti-VEGF Antibody | Methylcellulose disc | Visible anti-angiogenic activity observed through semi-quantitative evaluation | [169] |
| Anti-laminin antibody | Methylcellulose disc | Macroscopic observations saw a delay in capillary network development | [171] |
| Anginex | Plastic ring | Significant decrease in intersections of blood vessels with concentric rings projected onto images | [176,177] |
| Angiotensinogen | Plastic ring | First and second order centripetal blood vessels around on-plants were counted, with inhibition of smaller blood vessels observed | [178] |
| Following FITC injection, blood vessel density, length and number of branch points were quantified highlighting inhibition of smaller blood vessels | [29] | ||
| Obtustatin (α1β1 inhibitor) | Decrease in the number of small new vessels growing towards on-plants | [179] |
| Response | Treatment | Delivery Method | Angiogenic Outcome | Ref. |
|---|---|---|---|---|
| Pro-angiogenic | Glioblastoma cancer stem cells | Alginate scaffold | Increased blood vessel number converging towards on-plants | [114] |
| Human umbilical vein endothelial cells (HUVECs) | Cylindrical scaffold | Increased number of blood vessels and blood vessel penetration into on-plant | [115] | |
| Adipose derived stem cells | Cylindrical scaffold | Increased number of blood vessels and blood vessel penetration into on-plant | [115] | |
| Hydrogel | Significant increase in vessel number, vessel length and vasculogenic index | [74] | ||
| Seeded on a scaffold | Increased number of blood vessels converging towards on-plants | [108] | ||
| Matrigel | Following von Willebrand factor staining and semi quantitative scoring, a significant increase in angiogenesis | [184] | ||
| Burkitt’s Lymphoma cell lines (BL2B95 and BL74) | Matrigel | Following tissue sectioning increase in blood vessel diameter determined | [183] | |
| Human Liver Cancer (HepG2) cells | Matrigel | Increased number of blood vessels converging towards on-plants | [86] | |
| Prostate Cancer Cells (LNCaP) | Matrigel | A change in blood vessel number within a defined area observed | [185] | |
| Colon carcinoma (SW620) | Matrigel | Increase in angiogenic index was observed | [54] | |
| Neuroblastoma (NB15/FOXO3 cells) | Matrigel | Following desmin staining, increased micro-vessel formation was observed | [56] | |
| Glioblastoma (U87 MG) Cell lines | Matrigel | Increased observation of spoke wheel pattern of blood vessels radiating from on-plants | [55] | |
| Human Cardiopoietic Stem Cells | Scaffold | Blood vessel density within a defined area was increased | [186] | |
| Multiple myeloma plasma cells | Gelatin sponge | Induction of an increased vasculogenic index was calculated | [66] | |
| Mouse Melanoma (B-16) | Plastic ring | Development of visible spoke wheel pattern of blood vessels converging towards on-plants | [187] | |
| Human Melanoma (C8161) | Plastic ring | Significant increase in area occupied by endothelial cells observed within a defined area | [75] | |
| Hydrogel | ||||
| Human Prostate Cancer (PC3) | ||||
| Plastic ring | ||||
| Skin graft | Plastic ring | Photobiomodulation along with cell application resulted in increased number of vascular junctions within a defined area | [104] | |
| Human Ovarian Tissue | Plastic ring | Visual estimation of area occupied by blood vessels compared to total surface area resulted increased angiogenesis and neovascularisation | [188] | |
| Melanoma Tumour Tissue | Tumour | Spoke wheel pattern of capillaries converging towards on-plants observed | [189] | |
| Recurrent respiratory papilloma tissue (RRP) | Tumour | Increase in blood vessel number within a defined area observed | [125] | |
| Hepatocellular Carcinoma Tumour tissue | Tumour | Increased micro vessel density within a defined area observed | [190] | |
| Human Malignant Ovarian tumours | Tumour | Increase in the pattern, density, and size of the CAM blood vessels near the tumour implants visible | [191] | |
| Adenocarcinoma Tumour Tissue | Tumour | Increase in the pattern, density, and size of the CAM blood vessels near the tumour implants visible | [191] | |
| Glioma cells (C6) | Injected | Macroscopic observations indicated tumours became vascularised by CAM blood vessels | [78] | |
| Pancreatic carcinoma (10AS) | Injectí | Macroscopic observations indicated tumours became vascularised by CAM blood vessels | [78] | |
| Anti-angiogenic | Colon carcinoma (SW480) | Collagen | No Induction of angiogenesis or increased angiogenic index | [54] |
| Burkitt’s Lymphoma cell lines (BL2) | Matrigel | Following tissue sectioning reduced blood vessel diameter observed | [183] |
| Gene Modification | Cell Type | Delivery Method | Angiogenic Response | Ref. | |
|---|---|---|---|---|---|
| Pro-angiogenic | FGF-1 expression plasmid | Bovine Endothelial Cells | Gelatin sponge | Following tissue sectioning, and staining for von Willebrand Factor, a twofold increase in capillary number was quantified | [135] |
| LV miR-205 inhibition | Endothelial colony-forming cell CM | Not mentioned | Software quantified significant increase in blood vessel density within a defined area | [192] | |
| Sphingosine-1-phosphate treated | Osteoblast cell (MG-63) CM | Increase in blood vessel number within a defined area was quantified | [193] | ||
| miR-338-3p inhibition plasmid | Hepatocellular carcinoma (HCC) CM | Filter disc | Visual inspection of second and third order blood vessels inferred increased blood vessel formation | [194] | |
| AGO2 expression plasmid | Myeloma cell CM | Significant increase in on-plant infiltrating blood vessels observed | [195] | ||
| IFN-γ treated | Mesenchymal stem cell CM | Pipetted | A significant increase in number of small blood vessels (diameter less than 1 mm) | [196] | |
| TNF-α treated | |||||
| IFN-γ and TNF-α treated | A significant increase in both small and large blood vessels | ||||
| Anti-angiogenic | AAV-Timp1- transduced | Chinese hamster ovary cells | Gelatin sponge | No spoke wheel pattern of blood vessels radiating from on-plants | [197] |
| LV mediated Angiopoietin-2 shRNA | Pancreatic carcinoma cells | Pipetted | Decrease in number of blood vessel branch points | [198] | |
| Connective tissue growth factor (CTGF)-shRNA | OASF cell CM | Pipetted | Significant reduction in blood vessel count | [199] | |
| Endostatin expression plasmid | COS-1 cell CM | Pipetted | Significant reduction in blood vessel branch points | [200] | |
| LV VEGF shRNA | Hypertriploid renal cell carcinoma CM | Pipetted | Significant decrease in blood vessel counts and total blood vessel length | [201] | |
| CCL5-shRNA | Chondrosarcoma cells (JJ012) | Matrigel | Significant decrease in blood vessel branches | [202] | |
| Sema3C transfected | Glioblastoma cell line (U87 MG) | Collagen | Diminished observation of a spoke wheel pattern of blood vessels radiating from on-plants | [55] | |
| AGO2-shRNA | Myeloma cell CM | Filter disc | Lower blood vessel densities infiltrating the on-plants observed | [195] | |
| Vascular endothelial cell growth inhibitor (VEGI) expression plasmid | HeLa cell CM | Filter disc | Significant inhibition of neovascularization | [203] | |
| Novel immunotoxin (VEGF165-PE38) expression plasmid | HEK293 cell CM | Not mentioned | Inhibition in growth of capillary-like structures | [204] | |
| LV miR-205 OE | Endothelial colony-forming cell CM | Visual inspection saw reduced blood vessel formation | [192] | ||
| miR-181a-5p expression plasmid | Fibrosarcoma (HT1080) cell CM | Gelatin sponge | Impairment of new blood vessel formation observed | [205] | |
| Nuclear Factor-Erythroid 2 (NRF2) shRNA | Human colon cancer cell CM | Matrigel | Significant reduction in blood vessel branch points in circular region around on-plants | [206] | |
| P53 Isoform (Δ133p53) deletion | Human Glioblastoma (U87) cell CM | Silicon ring | Following tissue sectioning and staining, reduced blood vessels quantified | [207] | |
| LV miR-542-5p | Non-small cell lung cancer CM | Silicon ring | Significant reduction in percentage vascular density | [109] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kennedy, D.C.; Coen, B.; Wheatley, A.M.; McCullagh, K.J.A. Microvascular Experimentation in the Chick Chorioallantoic Membrane as a Model for Screening Angiogenic Agents including from Gene-Modified Cells. Int. J. Mol. Sci. 2022, 23, 452. https://doi.org/10.3390/ijms23010452
Kennedy DC, Coen B, Wheatley AM, McCullagh KJA. Microvascular Experimentation in the Chick Chorioallantoic Membrane as a Model for Screening Angiogenic Agents including from Gene-Modified Cells. International Journal of Molecular Sciences. 2022; 23(1):452. https://doi.org/10.3390/ijms23010452
Chicago/Turabian StyleKennedy, Donna C., Barbara Coen, Antony M. Wheatley, and Karl J. A. McCullagh. 2022. "Microvascular Experimentation in the Chick Chorioallantoic Membrane as a Model for Screening Angiogenic Agents including from Gene-Modified Cells" International Journal of Molecular Sciences 23, no. 1: 452. https://doi.org/10.3390/ijms23010452






