Plasma Globotriaosylsphingosine and α-Galactosidase A Activity as a Combined Screening Biomarker for Fabry Disease in a Large Japanese Cohort
Abstract
:1. Introduction
2. Materials and Methods
2.1. Lyso-Gb3 and GLA Screening
2.2. Patient Enrollment
2.3. Sample Collection
2.4. Measurement of Plasma Lyso-Gb3
2.5. Measurement of Plasma GLA Activity
2.6. Genetic Counseling
2.7. Gene Analysis
2.8. Defining FD
2.9. Statistical Analyses
3. Results
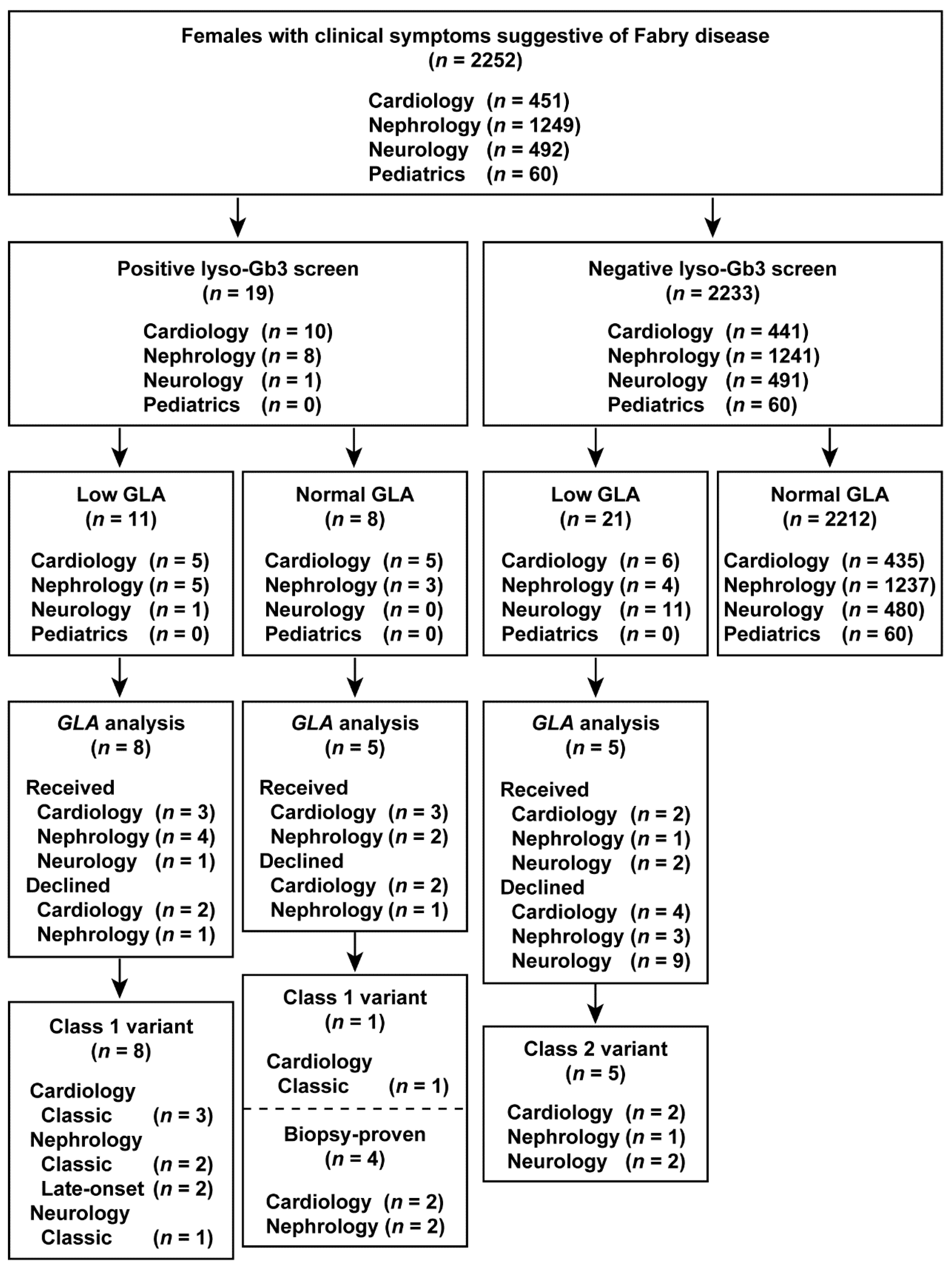
3.1. Study Population
3.2. Plasma Lyso-Gb3 Levels
3.3. Plasma GLA Activity
3.4. Classification of FD Probands with Class 1 Variants
3.5. Frequency of Positive Lyso-Gb3 Screens and FD Diagnosis by Clinical Department
3.6. Prosaposin (PSAP) Analysis for Probands with Late-Onset Biopsy-Proven FD
3.7. Comparison of Lyso-Gb3 Screening and GLA Analysis between Cohorts
3.8. Differences in Plasma Lyso-Gb3 Levels and GLA Activity between Cohorts
3.9. ROC Curve Analysis for Plasma Lyso-Gb3 Levels, GLA Activity, and GLA/lyso-Gb3 Ratio
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brady, R.O.; Gal, A.E.; Bradley, R.M.; Martensson, E.; Warshaw, A.L.; Laster, L. Enzymatic defect in Fabry’s disease ceramidetrihexosidase deficiency. N. Engl. J. Med. 1967, 276, 1163–1167. [Google Scholar] [CrossRef] [PubMed]
- Schiffmann, R.; Hughes, D.A.; Linthorst, G.E.; Ortiz, A.; Svarstad, E.; Warnock, D.G.; West, M.L.; Wanner, C. Conference Participants: Screening, diagnosis, and management of patients with Fabry disease: Conclusions from a “Kidney Disease: Improving Global Outcomes” (KDIGO) Controversies Conference. Kidney Int. 2017, 91, 284–293. [Google Scholar] [CrossRef]
- Oliveira, J.P.; Ferreira, S. Multiple phenotypic domains of Fabry disease and their relevance for establishing genotype-phenotype correlations. Appl. Clin. Genet. 2019, 12, 35–50. [Google Scholar] [CrossRef] [Green Version]
- Aerts, J.M.; Groener, J.E.; Kuiper, S.; Donker-Koopman, W.E.; Strijland, A.; Ottenhoff, R.; van Roomen, C.; Mirzaian, M.; Wijburg, F.A.; Linthorst, G.E.; et al. Elevated globotriaosylsphingosine is a hallmark of Fabry disease. Proc. Natl. Acad. Sci. USA 2008, 26, 2812–2817. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nowak, A.; Mechtler, T.; Kasper, D.C.; Desnick, R.J. Correlation of lyso-Gb3 levels in dried blood spots and sera from patients with classic and later-onset Fabry disease. Mol. Genet. Metab. 2017, 121, 320–324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stiles, A.R.; Zhang, H.; Dai, J.; McCaw, P.; Beasley, J.; Rehder, C.; Koeberl, D.D.; McDonald, M.; Bali, D.S.; Young, S.P. A comprehensive testing algorithm for the diagnosis of Fabry disease in males and females. Mol. Genet. Metab. 2020, 130, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Maruyama, H.; Miyata, K.; Mikame, M.; Taguchi, A.; Guili, C.; Shimura, M.; Murayama, K.; Inoue, T.; Yamamoto, S.; Sugimura, K.; et al. Effectiveness of plasma lyso-Gb3 as a biomarker for selecting high-risk patients with Fabry disease from multispecialty clinics for genetic analysis. Genet. Med. 2019, 21, 44–52. [Google Scholar] [CrossRef]
- Gal, A.; Hughes, D.A.; Winchester, B. Toward a consensus in the laboratory diagnostics of Fabry disease—Recommendations of a European expert group. J. Inherit. Metab. Dis. 2011, 34, 509–514. [Google Scholar] [CrossRef] [Green Version]
- Caudron, E.; Prognon, P.; Germain, D.P. Enzymatic diagnosis of Fabry disease using a fluorometric assay on dried blood spots: An alternative methodology. Eur. J. Med. Genet. 2015, 58, 681–684. [Google Scholar] [CrossRef]
- Ferreira, S.; Auray-Blais, C.; Boutin, M.; Lavoie, P.; Nunes, J.P.; Martins, E.; Garman, S.; Oliveira, J.P. Variations in the GLA gene correlate with globotriaosylceramide and globotriaosylsphingosine analog levels in urine and plasma. Clin. Chim. Acta 2015, 447, 96–104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smid, B.E.; van der Tol, L.; Cecchi, F.; Elliott, P.M.; Hughes, D.A.; Linthorst, G.E.; Timmermans, J.; Weidemann, F.; West, M.L.; Biegstraaten, M.; et al. Uncertain diagnosis of Fabry disease: Consensus recommendation on diagnosis in adults with left ventricular hypertrophy and genetic variants of unknown significance. Int. J. Cardiol. 2014, 177, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Kase, R.; Bierfreund, U.; Klein, A.; Kolter, T.; Itoh, K.; Suzuki, M.; Hashimoto, Y.; Sandhoff, K.; Sakuraba, H. Only sphingolipid activator protein B (SAP-B or saposin B) stimulates the degradation of globotriaosylceramide by recombinant human lysosomal α-galactosidase in a detergent-free liposomal system. FEBS Lett. 1996, 393, 74–76. [Google Scholar] [CrossRef] [Green Version]
- Motta, M.; Tatti, M.; Furlan, F.; Celato, A.; Di Fruscio, G.; Polo, G.; Manara, R.; Nigro, V.; Tartaglia, M.; Burlina, A.; et al. Clinical, biochemical and molecular characterization of prosaposin deficiency. Clin. Genet. 2016, 90, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Kuchař, L.; Ledvinová, J.; Hřebíček, M.; Myšková, H.; Dvořáková, L.; Berná, L.; Chrastina, P.; Asfaw, B.; Elleder, M.; Petermöller, M.; et al. Prosaposin deficiency and saposin B deficiency (activator-deficient metachromatic leukodystrophy): Report on two patients detected by analysis of urinary sphingolipids and carrying novel PSAP gene mutations. Am. J. Med. Genet. Part A 2009, 149A, 613–621. [Google Scholar] [CrossRef] [Green Version]
- Germain, D.P.; Oliveira, J.P.; Bichet, D.G.; Yoo, H.W.; Hopkin, R.J.; Lemay, R.; Politei, J.; Wanner, C.; Wilcox, W.R.; Warnock, D.G. Use of a rare disease registry for establishing phenotypic classification of previously unassigned GLA variants: A consensus classification system by a multispecialty Fabry disease genotype–phenotype workgroup. J. Med. Genet. 2020, 57, 542–551. [Google Scholar] [CrossRef] [Green Version]
- Maruyama, H.; Takata, T.; Tsubata, Y.; Tazawa, R.; Goto, K.; Tohyama, J.; Narita, I.; Yoshioka, H.; Ishii, S. Screening of male dialysis patients for Fabry disease by plasma globotriaosylsphingosine. Clin. J. Am. Soc. Nephrol. 2013, 8, 629–636. [Google Scholar] [CrossRef] [Green Version]
- Hulková, H.; Cervenková, M.; Ledvinová, J.; Tochácková, M.; Hrebícek, M.; Poupetová, H.; Befekadu, A.; Berná, L.; Paton, B.C.; Harzer, K.; et al. A novel mutation in the coding region of the prosaposin gene leads to a complete deficiency of prosaposin and saposins, and is associated with a complex sphingolipidosis dominated by lactosylceramide accumulation. Hum. Mol. Genet. 2001, 10, 927–940. [Google Scholar] [CrossRef] [Green Version]
- den Dunnen, J.T.; Dalgleish, R.; Maglott, D.R.; Hart, R.K.; Greenblatt, M.S.; McGowan-Jordan, J.; Roux, A.F.; Smith, T.; Antonarakis, S.E.; Taschner, P.E. HGVS recommendations for the description of sequence variants: 2016 update. Hum. Mutat. 2016, 37, 564–569. [Google Scholar] [CrossRef] [Green Version]
- Koskenvuo, J.W.; Engblom, E.; Kantola, I.M.; Hartiala, J.J.; Saraste, A.; Kiviniemi, T.O.; Mononen, I.; Saraste, M. Echocardiography in Fabry disease: Diagnostic value of endocardial border binary appearance. Clin. Physiol. Funct. Imaging 2009, 29, 177–180. [Google Scholar] [CrossRef] [PubMed]
- Shabbeer, J.; Yasuda, M.; Luca, E.; Desnick, R.J. Fabry disease: 45 novel mutations in the alpha-galactosidase A gene causing the classical phenotype. Mol. Genet. Metab. 2002, 76, 23–30. [Google Scholar] [CrossRef]
- Varela, P.; Mastroianni Kirsztajn, G.; Motta, F.L.; Martin, R.P.; Turaça, L.T.; Ferrer, H.L.F.; Gomes, C.P.; Nicolicht, P.; Mara Marins, M.; Pessoa, J.G.; et al. Correlation between GLA variants and alpha-Galactosidase A profile in dried blood spot: An observational study in Brazilian patients. Orphanet J. Rare Dis. 2020, 15, 30. [Google Scholar] [CrossRef] [Green Version]
- Eng, C.M.; Niehaus, D.J.; Enriquez, A.L.; Burgert, T.S.; Ludman, M.D.; Desnick, R.J. Fabry disease: Twenty-three mutations including sense and antisense CpG alterations and identification of a deletional hot-spot in the alpha-galactosidase A gene. Hum. Mol. Genet. 1994, 3, 1795–1799. [Google Scholar] [CrossRef] [PubMed]
- Fukutomi, M.; Tanaka, N.; Uchinoumi, H.; Kanemoto, M.; Nakao, F.; Yamada, J.; Kamei, T.; Takenaka, T.; Fujii, T. Japanese patients with Fabry disease predominantly showing cardiac and neurological manifestation with novel missense mutation: R220P. J. Cardiol. 2013, 62, 63–69. [Google Scholar] [CrossRef] [Green Version]
- Redonnet-Vernhet, I.; van Amstel, J.K.P.; Jansen, R.P.; Wevers, R.A.; Salvayre, R.; Levade, T. Uneven X inactivation in a female monozygotic twin pair with Fabry disease and discordant expression of a novel mutation in the alpha-galactosidase A gene. J. Med. Genet. 1996, 33, 682–688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eng, C.M.; Ashley, G.A.; Burgert, T.S.; Enriquez, A.L.; D’Souza, M.; Desnick, R.J. Fabry disease: Thirty-five mutations in the alpha-galactosidase A gene in patients with classic and variant phenotypes. Mol. Med. 1997, 3, 174–182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sakuraba, H.; Oshima, A.; Fukuhara, Y.; Shimmoto, M.; Nagao, Y.; Bishop, D.F.; Desnick, R.J.; Suzuki, Y. Identification of point mutations in the alpha-galactosidase A gene in classical and atypical hemizygotes with Fabry disease. Am. J. Hum. Genet. 1990, 47, 784–789. [Google Scholar] [PubMed]
- Shimotori, M.; Maruyama, H.; Nakamura, G.; Suyama, T.; Sakamoto, F.; Itoh, M.; Miyabayashi, S.; Ohnishi, T.; Sakai, N.; Wataya-Kaneda, M.; et al. Novel mutations of the GLA gene in Japanese patients with Fabry disease and their functional characterization by active site specific chaperone. Hum. Mutat. 2008, 29, 331. [Google Scholar] [CrossRef]
- Sakuraba, H.; Tsukimura, T.; Togawa, T.; Tanaka, T.; Ohtsuka, T.; Sato, A.; Shiga, T.; Saito, S.; Ohno, K. Fabry disease in a Japanese population-molecular and biochemical characteristics. Mol. Genet. Metab. Rep. 2018, 17, 73–79. [Google Scholar] [CrossRef]
- Wakakuri, H.; Nakamura, S.; Utsumi, K.; Shimizu, W.; Yasutake, M. Novel α-galactosidase A mutation (K391E) in a young woman with severe cardiac and renal manifestations of Fabry disease. Int. Heart J. 2016, 57, 637–639. [Google Scholar] [CrossRef] [Green Version]
- Rozenfeld, P.A.; Tarabuso, A.; Ebner, R.; Ramallo, G.; Fossati, C.A. A successful approach for the detection of Fabry patients in Argentina. Clin. Genet. 2006, 69, 344–348. [Google Scholar] [CrossRef]
- Sahashi, K.; Masuda, A.; Matsuura, T.; Shinmi, J.; Zhang, Z.; Takeshima, Y.; Matsuo, M.; Sobue, G.; Ohno, K. In Vitro and in silico analysis reveals an efficient algorithm to predict the splicing consequences of mutations at the 5’ splice sites. Nucleic Acids Res. 2007, 35, 5995–6003. [Google Scholar] [CrossRef]
- Nakai, K.; Sakamoto, H. Construction of a novel database containing aberrant splicing mutations of mammalian genes. Gene 1994, 141, 171–177. [Google Scholar] [CrossRef]
- Burset, M.; Seledtsov, I.A.; Solovyev, V.V. Analysis of canonical and non-canonical splice sites in mammalian genomes. Nucleic Acids Res. 2000, 28, 4364–4375. [Google Scholar] [CrossRef]
- Baydakova, G.V.; Ilyushkina, A.A.; Moiseev, S.; Bychkov, I.O.; Nikitina, N.V.; Buruleva, T.A.; Zakharova, E.Y. α-Galactosidase A/lysoGb3 ratio as a potential marker for Fabry disease in females. Clin. Chim. Acta 2020, 501, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Chien, Y.H.; Lee, N.C.; Chiang, S.C.; Desnick, R.J.; Hwu, W.L. Fabry disease: Incidence of the common later-onset α-galactosidase A IVS4+919G→A mutation in Taiwanese newborns—Superiority of DNA-based to enzyme-based newborn screening for common mutations. Mol. Med. 2012, 18, 780–784. [Google Scholar] [CrossRef]
- Johnstone, B.; Kaiser, A.; Injeyan, M.C.; Sappleton, K.; Chitayat, D.; Stephens, D.; Shuman, C. The relationship between burnout and occupational stress in genetic counselors. J. Genet. Counsel. 2016, 25, 731–741. [Google Scholar] [CrossRef] [Green Version]
- Abramowicz, A.; Gos, M. Splicing mutations in human genetic disorders: Examples, detection, and confirmation. J. Appl. Genet. 2018, 59, 253–268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Varela, P.; Caldas, M.M.; Pesquero, J.B. Novel GLA mutation promotes intron inclusion leading to Fabry disease. Front. Genet. 2019, 10, 783. [Google Scholar] [CrossRef] [Green Version]
- Ishii, S.; Nakao, S.; Minamikawa-Tachino, R.; Desnick, R.J.; Fan, J.Q. Alternative splicing in the α-galactosidase A gene: Increased exon inclusion results in the Fabry cardiac phenotype. Am. J. Hum. Genet. 2002, 70, 994–1002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rolfs, A.; Böttcher, T.; Zschiesche, M.; Morris, P.; Winchester, B.; Bauer, P.; Walter, U.; Mix, E.; Löhr, M.; Harzer, K.; et al. Prevalence of Fabry disease in patients with cryptogenic stroke: A prospective study. Lancet 2005, 366, 1794–1796. [Google Scholar] [CrossRef]
- Fancellu, L.; Borsini, W.; Romani, I.; Pirisi, A.; Deiana, G.A.; Sechi, E.; Doneddu, P.E.; Rassu, A.L.; Demurtas, R.; Scarabotto, A.; et al. Exploratory screening for Fabry’s disease in young adults with cerebrovascular disorders in northern Sardinia. BMC Neurol. 2015, 15, 256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferreira, S.; Reguenga, C.; Oliveira, J.P. The modulatory effects of the polymorphisms in GLA 5’-untranslated region upon gene expression are cell-type specific. JIMD Rep. 2015, 23, 27–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lanthier, S.; Saposnik, G.; Lebovic, G.; Pope, K.; Selchen, D.; Moore, D.F.; Canadian Fabry Stroke Screening Initiative Study Group. Prevalence of Fabry disease and outcomes in young Canadian patients with cryptogenic ischemic cerebrovascular events. Stroke 2017, 48, 1766–1772. [Google Scholar] [CrossRef] [PubMed]
- Kinoshita, N.; Hosomi, N.; Matsushima, H.; Nakamori, M.; Yagita, Y.; Yamawaki, T.; Torii, T.; Kitamura, T.; Sueda, Y.; Shimomura, R.; et al. Screening for Fabry disease in Japanese patients with young-onset stroke by measuring α-galactosidase A and globotriaosylsphingosine. J. Stroke Cerebrovasc. Dis. 2018, 27, 3563–3569. [Google Scholar] [CrossRef]
- Rolfs, A.; Fazekas, F.; Grittner, U.; Dichgans, M.; Martus, P.; Holzhausen, M.; Böttcher, T.; Heuschmann, P.U.; Tatlisumak, T.; Tanislav, C.; et al. Acute cerebrovascular disease in the young: The stroke in young Fabry patients study. Stroke 2013, 44, 340–349. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baptista, M.V.; Ferreira, S.; Pinho-e-Melo, T.; Carvalho, M.; Cruz, V.T.; Carmona, C.; Silva, F.A.; Tuna, A.; Rodrigues, M.; Ferreira, C.; et al. Mutations of the GLA gene in young patients with stroke: The PORTYSTROKE study—Screening genetic conditions in Portuguese young stroke patients. Stroke 2010, 41, 431–436. [Google Scholar] [CrossRef] [Green Version]
- Lukas, J.; Giese, A.K.; Markoff, A.; Grittner, U.; Kolodny, E.; Mascher, H.; Lackner, K.J.; Meyer, W.; Wree, P.; Saviouk, V.; et al. Functional characterisation of alpha-galactosidase a mutations as a basis for a new classification system in Fabry disease. PLoS Genet. 2013, 9, e1003632. [Google Scholar] [CrossRef] [Green Version]
- De Brabander, I.; Yperzeele, L.; Ceuterick-De Groote, C.; Brouns, R.; Baker, R.; Belachew, S.; Delbecq, J.; De Keulenaer, G.; Dethy, S.; Eyskens, F.; et al. Phenotypical characterization of α-galactosidase A gene mutations identified in a large Fabry disease screening program in stroke in the young. Clin. Neurol. Neurosurg. 2013, 115, 1088–1093. [Google Scholar] [CrossRef]


| Cardiology | Nephrology | Neurology | Pediatrics | Total | |
|---|---|---|---|---|---|
| Males | (n = 1006) | (n = 1771) | (n = 624) | (n = 38) | (n = 3439) |
| Median (IQR) age (years) | 64 (51–71) | 66 (56–74) | 54 (47–71) | 11 (9–13) | 64 (51–73) |
| Clinics (n) | 59 | 47 | 43 | 13 | 162 |
| Females | (n = 451) | (n = 1249) | (n = 492) | (n = 60) | (n = 2252) |
| Median (IQR) age (years) | 67 (53–75) | 69 (61–77) | 66 (47–81) | 12 (9–14) | 68 (54–77) |
| Clinics (n) | 74 | 57 | 57 | 26 | 214 |
| Patient No. | Lyso-Gb3 Levels (ng/mL) | GLA Activity (nmol/h/mL) | GLA Variants | Age (Years) | Early-Onset Classic Manifestations | Manifestations | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| DNA | Protein | Heart | Kidneys | CNS | ||||||
| Males; Classic type | ||||||||||
| Pediatrics | 1 1 | 190.2 | 0.4 | c.(202C>T) | p.Leu68Phe | 13 | Acroparesthesia Hypohidrosis | None | None | None |
| 2 1 | 172.2 | 0.3 | c.(254G>T) | p.Gly85Val | 9 | Acroparesthesia Hypohidrosis | None | None | None | |
| 3 | 230.1 | 0.3 | c.(658C>T) | p.Arg220* | 9 | Acroparesthesia Hypohidrosis | None | None | None | |
| 4 | 177.4 | 0.3 | c.1133G>T | p.Cys378Phe | 9 | Acroparesthesia Hypohidrosis | None | None | None | |
| Males; Late-onset type | ||||||||||
| Cardiology | 5 1 | 3.6 | 2.7 | c.(335G>A) | p.Arg112His | 61 | None | LVH | G5DA1 | None |
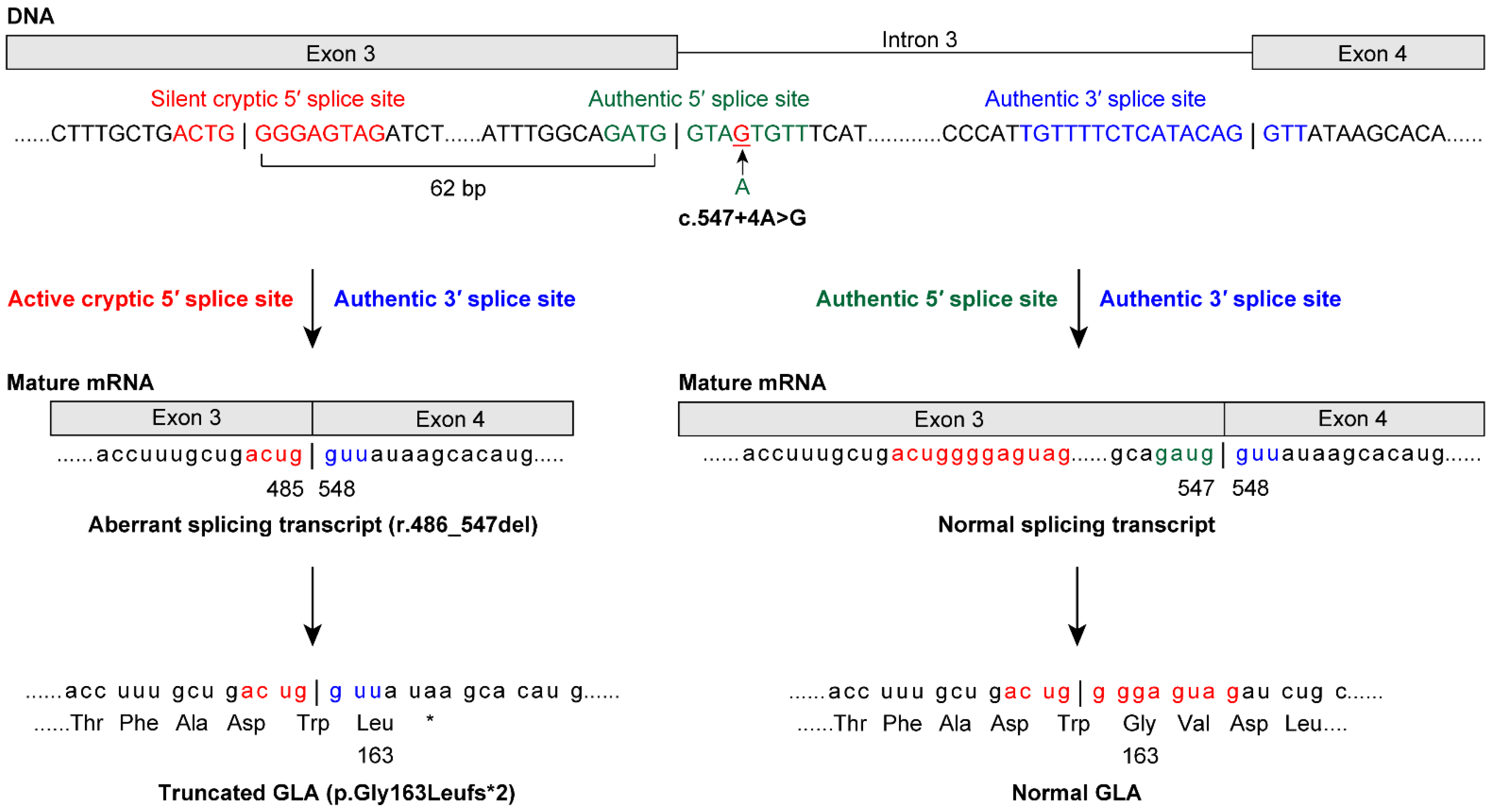
| 6 | 14.2 | 1.5 | c.547+4A>G r.486_547del | p.Gly163Leufs*2 | 74 | None | Arrhythmia LVH | G3a | None | |
| 7 1 | 15.6 | 1.1 | c.(902G>A) | p.Arg301Gln | 55 | None | LVH | None | None | |
| 8 1 | 14.5 | 1.0 | c.(935A>G) | p.Gln312Arg | 77 | None | LVH | None | None | |
| Nephrology | 9 | 14.7 | 1.9 | c.44C>G | p.(Ala15Gly) | 66 | None | Arrhythmia LVH | G3A2 | None |
| 10 1 | 4.1 | 0.6 | c.(335G>A) | p.Arg112His | 42 | None | None | G2A3 | None | |
| 11 1 | 4.0 | 1.3 | c.1171A>G | p.(Lys391Glu) | 75 | None | None | G5DA2 | Stroke | |
| Females; Classic type | ||||||||||
| Cardiology | 12 | 29.5 | 1.9 | c.(281G>A) | p.Cys94Tyr | 71 | None | Arrhythmia LVH | G3a | Stroke |
| 13 1 | 21.8 | 3.2 | c.559_560del | p.(Met187Valfs*6) | 65 | Acroparesthesia Gastrointestinal symptoms | LVH | None | Stroke | |
| 14 1 | 24.2 | 1.6 | c.(659G>C) | p.Arg220Pro | 65 | Angiokeratoma Cornea verticillata | LVH | G3bA1 | Stroke | |
| 15 1 | 4.0 | 8.0 | c.691G>A | p.Asp231Asn | 63 | None | Arrhythmia Heart failure | None | None | |
| Nephrology | 16 | 19.9 | 1.9 | c.334C>T | p.(Arg112Cys) | 44 | None | LVH | G2A3 | None |
| 17 1 | 15.0 | 2.6 | c.1244T>C | p.(Leu415Pro) | 32 | Acroparesthesia | LVH | G1A3 | None | |
| Neurology | 18 1 | 135.0 | 0.8 | c.[788A>G];[0] | p.(Asn263Ser) | 27 | Acroparesthesia Hypohidrosis Angiokeratoma | Arrhythmia | G1A3 | None |
| Females; Late-onset type | ||||||||||
| Nephrology | 19 | 15.8 | 2.2 | c.1163T>A | p.(Leu388His) | 59 | None | LVH | G3aA3 | None |
| 20 1 | 3.3 | 2.6 | c.1208T>C | p.(Leu403Ser) | 53 | None | LVH | G5A3 | None | |
| Cardiology | Nephrology | Neurology | Pediatrics | Total | ||
|---|---|---|---|---|---|---|
| Males | Lyso-Gb3-positive patients, n/N (%) | 4/1006 (0.4) | 6/1771 (0.3) | 0/624 (0) | 4/38 (10.5) | 14/3439 (0.4) |
| GLA variant, n/N (%) | 4/1006 (0.4) | 3/1771 (0.2) | 0/624 (0) | 4/38 (10.5) | 11/3439 (0.3) | |
| Classic type, n | 0 | 0 | 0 | 4 | 4 | |
| Late-onset type, n | 4 | 3 | 0 | 0 | 7 | |
| Females | Lyso-Gb3-positive patients, n/N (%) | 10/451 (2.2) | 8/1249 (0.6) | 1/492 (0.2) | 0/60 (0) | 19/2252 (0.8) |
| GLA variant, n/N (%) | 4/451 (0.9) | 4/1249 (0.3) | 1/492 (0.2) | 0/60 (0) | 9/2252 (0.4) | |
| Classic type, n | 4 | 2 | 1 | 0 | 7 | |
| Late-onset type, n | 0 | 2 | 0 | 0 | 2 |
| Department | Patient No. | Lyso-Gb3 Levels (ng/mL) | GLA Activity (nmol/h/mL) | GLA Class 1 Variants | Age (years) | Early-Onset Classic Manifestations | Manifestations | ||
|---|---|---|---|---|---|---|---|---|---|
| Heart | Kidneys | CNS | |||||||
| Male | |||||||||
| Nephrology | 1 | 3.2 | 8.4 | None | 75 | None | None | G2A1 Lamellar body | None |
| Females | |||||||||
| Cardiology | 2 1 | 8.4 | 8.5 | None | 59 | None | Arrhythmia LVH Lamellar body | G5A3 Lamellar body | None |
| 3 1 | 18.5 | 10.9 | None | 67 | None | Arrhythmia LVH Lamellar body | None | None | |
| Nephrology | 4 1 | 7.8 | 4.7 | None | 69 | None | Arrhythmia | G3aA3 Lamellar body | None |
| 5 | 5.6 | 5.9 | None | 66 | None | None | G3aA3 Lamellar body | None | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maruyama, H.; Taguchi, A.; Mikame, M.; Izawa, A.; Morito, N.; Izaki, K.; Seto, T.; Onishi, A.; Sugiyama, H.; Sakai, N.; et al. Plasma Globotriaosylsphingosine and α-Galactosidase A Activity as a Combined Screening Biomarker for Fabry Disease in a Large Japanese Cohort. Curr. Issues Mol. Biol. 2021, 43, 389-404. https://doi.org/10.3390/cimb43010032
Maruyama H, Taguchi A, Mikame M, Izawa A, Morito N, Izaki K, Seto T, Onishi A, Sugiyama H, Sakai N, et al. Plasma Globotriaosylsphingosine and α-Galactosidase A Activity as a Combined Screening Biomarker for Fabry Disease in a Large Japanese Cohort. Current Issues in Molecular Biology. 2021; 43(1):389-404. https://doi.org/10.3390/cimb43010032
Chicago/Turabian StyleMaruyama, Hiroki, Atsumi Taguchi, Mariko Mikame, Atsushi Izawa, Naoki Morito, Kazufumi Izaki, Toshiyuki Seto, Akifumi Onishi, Hitoshi Sugiyama, Norio Sakai, and et al. 2021. "Plasma Globotriaosylsphingosine and α-Galactosidase A Activity as a Combined Screening Biomarker for Fabry Disease in a Large Japanese Cohort" Current Issues in Molecular Biology 43, no. 1: 389-404. https://doi.org/10.3390/cimb43010032






