Nematode-Applied Technology for Human Tumor Microenvironment Research and Development
Abstract
:1. Introduction
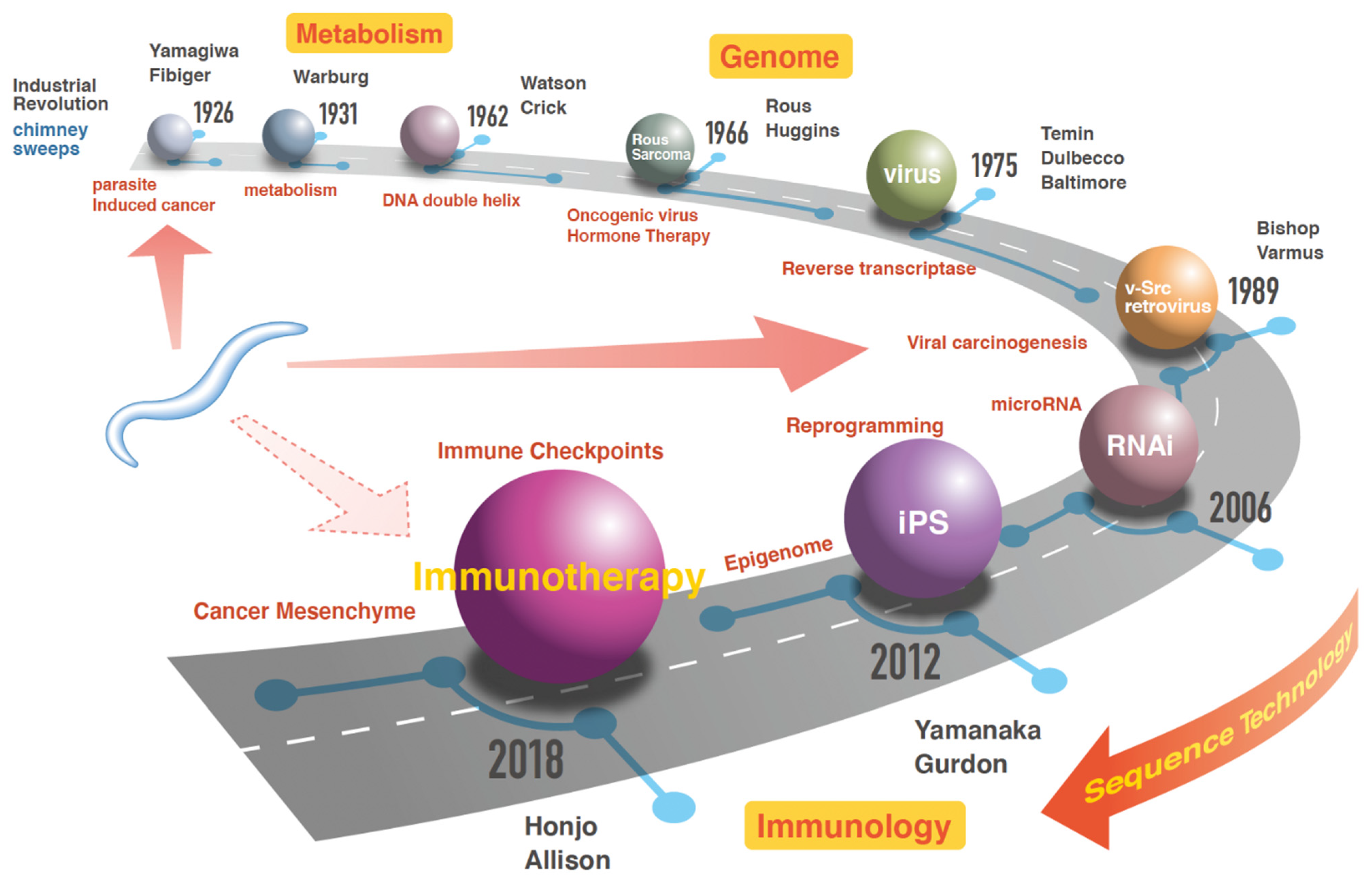
2. Milestones in Cancer Research
3. MicroRNA
4. Smell Research
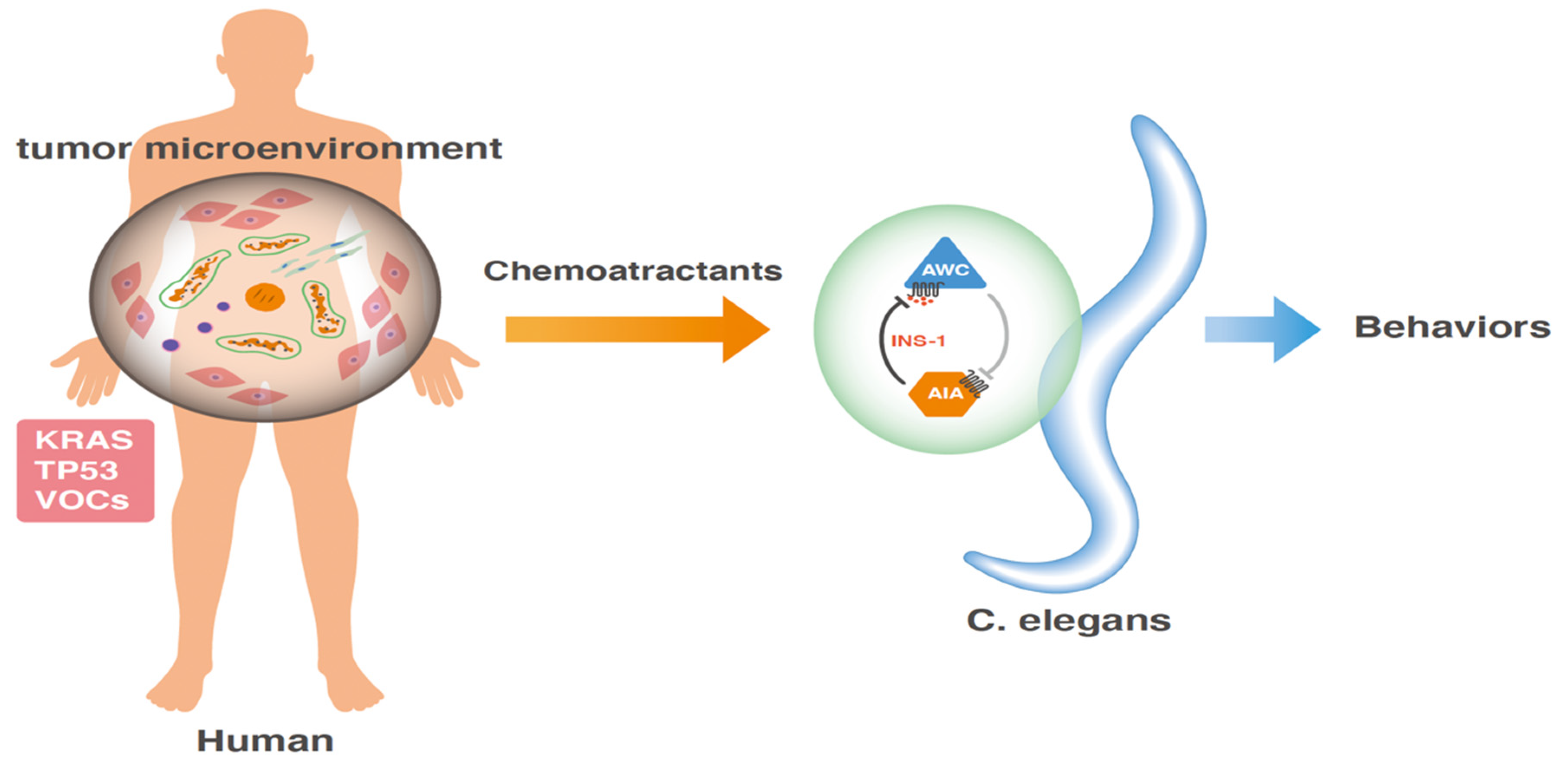
5. Innovative Method for Studying the Tumor Microenvironment
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Makarova, A.A.; Polilov, A.A.; Chklovskii, D.B. Small brains for big science. Curr. Opin. Neurobiol. 2021, 71, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Tuli, M.A.; Daul, A.; Schedl, T. Caenorhabditis nomenclature. WormBook 2018, 2018, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ribatti, D. Rudolf Virchow, the founder of cellular pathology. Rom. J. Morphol. Embryol. 2019, 60, 1381–1382. [Google Scholar] [PubMed]
- Listed, N.A. Rudolf Ludwig Karl Virchow (1821–1902) “Omnis Cellula E Cellula”. Minn. Med. 1966, 49, 533–534. [Google Scholar]
- Brotman, D.J.; Deitcher, S.R.; Lip, G.Y.; Matzdorff, A.C. Virchow’s triad revisited. South. Med. J. 2004, 97, 213–214. [Google Scholar] [CrossRef] [PubMed]
- Urbach, F. Environmental risk factors for skin cancer. In Skin Carcinogenesis in Man and in Experimental Models; Springer: Berlin/Heidelberg, Germany, 1993; Volume 128, pp. 243–262. [Google Scholar]
- Stolt, C.-M.; Klein, G.; Jansson, A.T. An Analysis of a Wrong Nobel Prize—Johannes Fibiger, 1926: A Study in the Nobel Archives. Adv. Cancer Res. 2004, 92, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Urbano, A.M. Otto Warburg: The journey towards the seminal discovery of tumor cell bioenergetic reprogramming. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2021, 1867, 165965. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. The hallmarks of cancer. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef] [Green Version]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [Green Version]
- Watson, J.D.; Crick, F.H.C. Molecular Structure of Nucleic Acids: A Structure for Deoxyribose Nucleic Acid. Nature 1953, 171, 737–738. [Google Scholar] [CrossRef]
- Raju, T.N. The Nobel chronicles. 1966: Francis Peyton Rous (1879–1970) and Charles Brenton Huggins (1901–1997). Lancet 1999, 354, 520. [Google Scholar] [CrossRef]
- Raju, T.N. The Nobel chronicles. 1975: Renato Dulbecco (b 1914), David Baltimore (b 1938), and Howard Martin Temin (1934–1994). Lancet 1999, 354, 1308. [Google Scholar] [CrossRef]
- Raju, T.N. The Nobel Chronicles. 1989: John Michael Bishop (b 1936) and Harold Eliot Varmus (b 1939). Lancet 2000, 355, 1106. [Google Scholar] [CrossRef]
- Takahashi, K.; Yamanaka, S. Induction of Pluripotent Stem Cells from Mouse Embryonic and Adult Fibroblast Cultures by Defined Factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wolchok, J. Putting the Immunologic Brakes on Cancer. Cell 2018, 175, 1452–1454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ishida, Y. PD-1: Its Discovery, Involvement in Cancer Immunotherapy, and Beyond. Cells 2020, 9, 1376. [Google Scholar] [CrossRef] [PubMed]
- Ishida, Y.; Agata, Y.; Shibahara, K.; Honjo, T. Induced expression of PD-1, a novel member of the immunoglobulin gene superfamily, upon programmed cell death. EMBO J. 1992, 11, 3887–3895. [Google Scholar] [CrossRef]
- Weinberg, R. Tumor suppressor genes. Science 1991, 254, 1138–1146. [Google Scholar] [CrossRef]
- Fire, A.; Xu, S.; Montgomery, M.K.; Kostas, S.A.; Driver, S.E.; Mello, C.C. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 1998, 391, 806–811. [Google Scholar] [CrossRef]
- Gregory, R.I.; Chendrimada, T.P.; Cooch, N.; Shiekhattar, R. Human RISC Couples MicroRNA Biogenesis and Posttranscriptional Gene Silencing. Cell 2005, 123, 631–640. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Kolb, F.A.; Jaskiewicz, L.; Westhof, E.; Filipowicz, W. Single Processing Center Models for Human Dicer and Bacterial RNase III. Cell 2004, 118, 57–68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, R.C.; Feinbaum, R.L.; Ambros, V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 1993, 75, 843–854. [Google Scholar] [CrossRef]
- Wightman, B.; Ha, I.; Ruvkun, G. Posttranscriptional regulation of the heterochronic gene lin-14 by lin-4 mediates temporal pattern formation in C. elegans. Cell 1993, 75, 855–862. [Google Scholar] [CrossRef]
- Sulston, J.; Horvitz, H. Post-embryonic cell lineages of the nematode, Caenorhabditis elegans. Dev. Biol. 1977, 56, 110–156. [Google Scholar] [CrossRef]
- Sulston, J.; Schierenberg, E.; White, J.; Thomson, J. The embryonic cell lineage of the nematode Caenorhabditis elegans. Dev. Biol. 1983, 100, 64–119. [Google Scholar] [CrossRef]
- Kaufman, E.J.; Miska, E.A. The microRNAs of Caenorhabditis elegans. Semin. Cell Dev. Biol. 2010, 21, 728–737. [Google Scholar] [CrossRef]
- Calin, G.A.; Dumitru, C.D.; Shimizu, M.; Bichi, R.; Zupo, S.; Noch, E.; Aldler, H.; Rattan, S.; Keating, M.; Rai, K.; et al. Frequent deletions and down-regulation of micro- RNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc. Natl. Acad. Sci. USA 2002, 99, 15524–15529. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calin, G.; Ferracin, M.; Cimmino, A.; Di Leva, G.; Shimizu, M.; Wojcik, S.E.; Iorio, M.; Visone, R.; Sever, N.I.; Fabbri, M.; et al. A MicroRNA Signature Associated with Prognosis and Progression in Chronic Lymphocytic Leukemia. N. Engl. J. Med. 2005, 353, 1793–1801. [Google Scholar] [CrossRef]
- Takamizawa, J.; Konishi, H.; Yanagisawa, K.; Tomida, S.; Osada, H.; Endoh, H.; Harano, T.; Yatabe, Y.; Nagino, M.; Nimura, Y.; et al. Reduced Expression of the let-7 MicroRNAs in Human Lung Cancers in Association with Shortened Postoperative Survival. Cancer Res. 2004, 64, 3753–3756. [Google Scholar] [CrossRef] [Green Version]
- Lu, J.; Getz, G.; Miska, E.A.; Alvarez-Saavedra, E.; Lamb, J.; Peck, D.; Sweet-Cordero, A.; Ebert, B.L.; Mak, R.H.; Ferrando, A.A.; et al. MicroRNA expression profiles classify human cancers. Nature 2005, 435, 834–838. [Google Scholar] [CrossRef]
- Konno, M.; Asai, A.; Kitagawa, T.; Yabumoto, M.; Ofusa, K.; Arai, T.; Hirotsu, T.; Doki, Y.; Eguchi, H.; Ishii, H. State-of-the-Art Technology of Model Organisms for Current Human Medicine. Diagnostics 2020, 10, 392. [Google Scholar] [CrossRef] [PubMed]
- Lo, G.K.-H.; Macpherson, K.; MacDonald, H.; Roberts, W.A. A comparative study of memory for olfactory discriminations: Dogs (Canis familiaris), rats (Rattus norvegicus), and humans (Homo sapiens). J. Comp. Psychol. 2020, 134, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Flores, H.; Apresa-García, T.; Garay-Villar, Ó.; Sánchez-Pérez, A.; Flores-Villegas, D.; Bandera-Calderón, A.; García-Palacios, R.; Rojas-Sánchez, T.; Romero-Morelos, P.; Sánchez-Albor, V.; et al. A non-invasive tool for detecting cervical cancer odor by trained scent dogs. BMC Cancer 2017, 17, 79. [Google Scholar] [CrossRef] [Green Version]
- Seo, I.-S.; Lee, H.-G.; Koo, B.; Koh, C.S.; Park, H.-Y.; Im, C.; Shin, H.-C. Cross detection for odor of metabolic waste between breast and colorectal cancer using canine olfaction. PLoS ONE 2018, 13, e0192629. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirotsu, T.; Sonoda, H.; Uozumi, T.; Shinden, Y.; Mimori, K.; Maehara, Y.; Ueda, N.; Hamakawa, M. A Highly Accurate Inclusive Cancer Screening Test Using Caenorhabditis elegans Scent Detection. PLoS ONE 2015, 10, e0118699. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kusumoto, H.; Tashiro, K.; Shimaoka, S.; Tsukasa, K.; Baba, Y.; Furukawa, S.; Furukawa, J.; Suenaga, T.; Kitazono, M.; Tanaka, S.; et al. Behavioural Response Alteration in Caenorhabditis elegans to Urine After Surgical Removal of Cancer: Nematode-NOSE (N-NOSE) for Postoperative Evaluation. Biomark. Cancer 2019, 11, 1179299x19896551. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kusumoto, H.; Tashiro, K.; Shimaoka, S.; Tsukasa, K.; Baba, Y.; Furukawa, S.; Furukawa, J.; Niihara, T.; Hirotsu, T.; Uozumi, T. Efficiency of Gastrointestinal Cancer Detection by Nematode-NOSE (N-NOSE). In Vivo 2019, 34, 73–80. [Google Scholar] [CrossRef]
- Asai, A.; Konno, M.; Ozaki, M.; Kawamoto, K.; Chijimatsu, R.; Kondo, N.; Hirotsu, T.; Ishii, H. Scent test using Caenorhabditis elegans to screen for early-stage pancreatic cancer. Oncotarget 2021, 12, 1687–1696. [Google Scholar] [CrossRef]
- Ueda, Y.; Kawamoto, K.; Konno, M.; Noguchi, K.; Kaifuchi, S.; Satoh, T.; Eguchi, H.; Doki, Y.; Hirotsu, T.; Mori, M.; et al. Application of C. elegans cancer screening test for the detection of pancreatic tumor in genetically engineered mice. Oncotarget 2019, 10, 5412–5418. [Google Scholar] [CrossRef] [Green Version]
- Rengarajan, S.; Hallem, E.A. Olfactory circuits and behaviors of nematodes. Curr. Opin. Neurobiol. 2016, 41, 136–148. [Google Scholar] [CrossRef] [Green Version]
- Chalasani, S.H.; Kato, S.; Albrecht, D.R.; Nakagawa, T.; Abbott, L.F.; Bargmann, C.I. Neuropeptide feedback modifies odor-evoked dynamics in Caenorhabditis elegans olfactory neurons. Nat. Neurosci. 2010, 13, 615–621. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Z.; Li, Y.; Yi, Y.; Huang, W.; Yang, S.; Niu, W.; Zhang, L.; Xu, Z.; Qu, A.; Wu, Z.; et al. Dissecting a central flip-flop circuit that integrates contradictory sensory cues in C. elegans feeding regulation. Nat. Commun. 2012, 3, 776. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirotsu, T.; Saeki, S.; Yamamoto, M.; Iino, Y. The Ras-MAPK pathway is important for olfaction in Caenorhabditis elegans. Nature 2000, 404, 289–293. [Google Scholar] [CrossRef] [PubMed]
- Nissinen, S.I.; Roine, A.; Hokkinen, L.; Karjalainen, M.; Venäläinen, M.; Helminen, H.; Niemi, R.; Lehtimäki, T.; Rantanen, T.; Oksala, N. Detection of Pancreatic Cancer by Urine Volatile Organic Compound Analysis. Anticancer Res. 2018, 39, 73–79. [Google Scholar] [CrossRef] [Green Version]
- Daulton, E.; Wicaksono, A.N.; Tiele, A.; Kocher, H.M.; Debernardi, S.; Crnogorac-Jurcevic, T.; Covington, J.A. Volatile organic compounds (VOCs) for the non-invasive detection of pancreatic cancer from urine. Talanta 2021, 221, 121604. [Google Scholar] [CrossRef]
- Thompson, M.; Feria, N.S.; Yoshioka, A.; Tu, E.; Civitci, F.; Estes, S.; Wagner, J.T. A Caenorhabditis elegans behavioral assay distinguishes early stage prostate cancer patient urine from controls. Biol. Open 2021, 10, bio057398. [Google Scholar] [CrossRef]
- Chen, X.; Xu, F.; Wang, Y.; Pan, Y.; Lu, D.; Wang, P.; Ying, K.; Chen, E.; Zhang, W. A study of the volatile organic compounds exhaled by lung cancer cells in vitro for breath diagnosis. Cancer 2007, 110, 835–844. [Google Scholar] [CrossRef]
- Phillips, M.; Altorki, N.; Austin, J.H.; Cameron, R.B.; Cataneo, R.N.; Kloss, R.; Maxfield, R.A.; Munawar, M.I.; Pass, H.; Rashid, A.; et al. Detection of lung cancer using weighted digital analysis of breath biomarkers. Clin. Chim. Acta 2008, 393, 76–84. [Google Scholar] [CrossRef] [Green Version]
- Dragonieri, S.; Annema, J.T.; Schot, R.; van der Schee, M.P.; Spanevello, A.; Carratù, P.; Resta, O.; Rabe, K.F.; Sterk, P.J. An electronic nose in the discrimination of patients with non-small cell lung cancer and COPD. Lung Cancer 2009, 64, 166–170. [Google Scholar] [CrossRef]
- Mazzone, P.J.; Wang, X.-F.; Xu, Y.; Mekhail, T.; Beukemann, M.C.; Na, J.; Kemling, J.W.; Suslick, K.; Sasidhar, M. Exhaled Breath Analysis with a Colorimetric Sensor Array for the Identification and Characterization of Lung Cancer. J. Thorac. Oncol. 2012, 7, 137–142. [Google Scholar] [CrossRef] [Green Version]
- Amann, A.; Corradi, M.; Mazzone, P.; Mutti, A. Lung cancer biomarkers in exhaled breath. Expert Rev. Mol. Diagn. 2011, 11, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Hakim, M.; Broza, Y.Y.; Barash, O.; Peled, N.; Phillips, M.; Amann, A.; Haick, H. Volatile Organic Compounds of Lung Cancer and Possible Biochemical Pathways. Chem. Rev. 2012, 112, 5949–5966. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.P.A.; Barash, O.; Jeries, R.; Peled, N.; Ilouze, M.; Hyde, R.; Marcus, M.W.; Field, J.; Haick, H. Unique volatolomic signatures of TP53 and KRAS in lung cells. Br. J. Cancer 2014, 111, 1213–1221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peled, N.; Barash, O.; Tisch, U.; Ionescu, R.; Broza, Y.Y.; Ilouze, M.; Mattei, J.; Bunn, P.A., Jr.; Hirsch, F.R.; Haick, H. Volatile fingerprints of cancer specific genetic mutations. Nanomedicine 2013, 9, 758–766. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Makohon-Moore, A.; Iacobuzio-Donahue, C.A. Pancreatic cancer biology and genetics from an evolutionary perspective. Nat. Rev. Cancer 2016, 16, 553–565. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Son, J.; Lyssiotis, C.A.; Ying, H.; Wang, X.; Hua, S.; Ligorio, M.; Perera, R.M.; Ferrone, C.R.; Mullarky, E.; Shyh-Chang, N.; et al. Glutamine supports pancreatic cancer growth through a KRAS-regulated metabolic pathway. Nature 2013, 496, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Murphy, D.A.; Courtneidge, S.A. The ‘ins’ and ‘outs’ of podosomes and invadopodia: Characteristics, formation and function. Nat. Rev. Mol. Cell Biol. 2011, 12, 413–426. [Google Scholar] [CrossRef] [Green Version]
- Hoshino, D.; Branch, K.M.; Weaver, A.M. Signaling inputs to invadopodia and podosomes. J. Cell Sci. 2013, 126, 2979–2989. [Google Scholar] [CrossRef] [Green Version]
- Schwab, M. Cathepsins, Encyclopedia of Cancer; Springer: Berlin/Heidelberg, Germany, 2017. [Google Scholar] [CrossRef]
- Génot, E.; Gligorijevic, B. Invadosomes in their natural habitat. Eur. J. Cell Biol. 2014, 93, 367–379. [Google Scholar] [CrossRef] [Green Version]
- Goicoechea, S.; Garcia-Mata, R.; Staub, J.; Valdivia, A.; Sharek, L.; McCulloch, C.G.; Hwang, R.F.; Urrutia, R.P.; Yeh, J.J.; Kim, H.J.; et al. Palladin promotes invasion of pancreatic cancer cells by enhancing invadopodia formation in cancer-associated fibroblasts. Oncogene 2013, 33, 1265–1273. [Google Scholar] [CrossRef] [Green Version]
- David-Pfeuty, T.; Singer, S.J. Altered distributions of the cytoskeletal proteins vinculin and α-actinin in cultured fibroblasts transformed by Rous sarcoma virus. Proc. Natl Acad. Sci. USA 1980, 77, 6687–6691. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shaye, D.D.; Greenwald, I. OrthoList: A Compendium of C. elegans Genes with Human Orthologs. PLoS ONE 2011, 6, e20085. [Google Scholar] [CrossRef] [PubMed]
- Saltel, F.; Daubon, T.; Juin, A.; Ganuza, I.E.; Veillat, V.; Génot, E. Invadosomes: Intriguing structures with promise. Eur. J. Cell Biol. 2011, 90, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Artym, V.V.; Zhang, Y.; Seillier-Moiseiwitsch, F.; Yamada, K.; Mueller, S.C. Dynamic Interactions of Cortactin and Membrane Type 1 Matrix Metalloproteinase at Invadopodia: Defining the Stages of Invadopodia Formation and Function. Cancer Res. 2006, 66, 3034–3043. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ayala, I.; Giacchetti, G.; Caldieri, G.; Attanasio, F.; Mariggiò, S.; Tetè, S.; Polishchuk, R.; Castronovo, V.; Buccione, R. Faciogenital dysplasia protein Fgd1 regulates invadopodia biogenesis and extracellular matrix degradation and is up-regulated in prostate and breast cancer. Cancer Res. 2009, 69, 747–752. [Google Scholar] [CrossRef] [Green Version]
- Lohmer, L.L.; Clay, M.R.; Naegeli, K.M.; Chi, Q.; Ziel, J.W.; Hagedorn, E.J.; Park, J.E.; Jayadev, R.; Sherwood, D.R. A Sensitized Screen for Genes Promoting Invadopodia Function In Vivo: CDC-42 and Rab GDI-1 Direct Distinct Aspects of Invadopodia Formation. PLoS Genet. 2016, 12, e1005786. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
di Luccio, E.; Kaifuchi, S.; Kondo, N.; Chijimatsu, R.; Vecchione, A.; Hirotsu, T.; Ishii, H. Nematode-Applied Technology for Human Tumor Microenvironment Research and Development. Curr. Issues Mol. Biol. 2022, 44, 988-997. https://doi.org/10.3390/cimb44020065
di Luccio E, Kaifuchi S, Kondo N, Chijimatsu R, Vecchione A, Hirotsu T, Ishii H. Nematode-Applied Technology for Human Tumor Microenvironment Research and Development. Current Issues in Molecular Biology. 2022; 44(2):988-997. https://doi.org/10.3390/cimb44020065
Chicago/Turabian Styledi Luccio, Eric, Satoru Kaifuchi, Nobuaki Kondo, Ryota Chijimatsu, Andrea Vecchione, Takaaki Hirotsu, and Hideshi Ishii. 2022. "Nematode-Applied Technology for Human Tumor Microenvironment Research and Development" Current Issues in Molecular Biology 44, no. 2: 988-997. https://doi.org/10.3390/cimb44020065
APA Styledi Luccio, E., Kaifuchi, S., Kondo, N., Chijimatsu, R., Vecchione, A., Hirotsu, T., & Ishii, H. (2022). Nematode-Applied Technology for Human Tumor Microenvironment Research and Development. Current Issues in Molecular Biology, 44(2), 988-997. https://doi.org/10.3390/cimb44020065





