Abstract
Ovarian cancer (OC) is gynecological cancer, and diagnosis and treatment are continuously advancing. Next-generation sequencing (NGS)-based diagnoses have emerged as novel methods for identifying molecules and pathways in cancer research. The NGS-based applications have expanded in OC research for early detection and identification of aberrant genes and dysregulation pathways, demonstrating comprehensive views of the entire transcriptome, such as fusion genes, genetic mutations, and gene expression profiling. Coinciding with advances in NGS-based diagnosis, treatment strategies for OC, such as molecular targeted therapy and immunotherapy, have also advanced. Immunotherapy is effective against many other cancers, and its efficacy against OC has also been demonstrated at the clinical phase. In this review, we describe several NGS-based applications for therapeutic targets of OC, and introduce current immunotherapeutic strategies, including vaccines, checkpoint inhibitors, and chimeric antigen receptor (CAR)-T cell transplantation, for effective diagnosis and treatment of OC.
1. Introduction
Gynecological cancer is a type of cancer that occurs in female reproductive organs, such as the vulva, vagina, cervix, uterus, fallopian tubes, and ovaries, that is related to the secretion of sex hormones [1]. Ovarian cancer (OC) is the eighth most common cancer in women, with a 5-year survival rate of approximately 45% [2,3]. The incidence of OC is approximately 10% that of breast cancer. OC can be classified into several histological types, characterized by cancer risk factors, clinical features, and molecular expression [4], and they include high-grade serous carcinoma (HGSC), endometrioid carcinoma of the ovary (ECO), clear cell carcinoma (CCC), mucinous carcinoma (MC), and low-grade serous carcinoma (LGSC) [5]. LGSC is associated with serous borderline tumor mutations in KRAS and BRAF. In contrast, HGSCs are not associated with serous borderline tumor mutations and have TP53 and BRCA mutations [6,7]. As these OC types are distinct diseases, diagnosis or treatment requires methodological approaches, suggesting that histopathological diagnosis of reproducible tumor cell types is critical for the successful treatment of OC.
In general, OC that has progressed to the advanced stage of a malignant tumor shows intractability, relapse, and drug resistance, resulting in poor prognosis with conventional treatment. The first-line standard care of patients with OC is surgery with platinum-based agent-based chemotherapy [8]. The combination of taxane compounds, such as paclitaxel, and platinum-based agents, such as carboplatin, is mainly adopted as first-line chemotherapy in OC [9]. However, in most patients who receive first-line treatment, OC relapses within several years [10,11]. More than 25% of patients with relapse have a poor prognosis due to a platinum-resistant or platinum-refractory disease [12,13]. Although outcomes and response rates are low, platinum-resistant patients are treated with non-platinum agents, such as liposomal doxorubicin, melphalan, paclitaxel, gemcitabine, and topotecan [1,14,15]. With an increasing understanding of cancer progression mechanisms, targeted therapies are emerging as innovative and promising therapeutic strategies. Therefore, antiangiogenic molecules, immune checkpoint inhibitors, poly ADP-ribose polymerase (PARP) inhibitors, estrogen receptor inhibitors, and various inhibitors of intrinsic tumor signaling pathways are being used as potential therapeutic agents for OC patients [1].
In particular, PARP inhibitors, such as olaparib or niraparib, have been mainly used in OC patients with BRCA mutations [16,17,18,19], and new chemotherapy, that induces drug combinations, is constantly being developed. PARP inhibitors play a key role in the maintenance of therapy for recurrent OC and primary HGSC [20]. A study has demonstrated that PARP inhibitors remarkably extend progression-free survival (PFS) after response to platinum-based chemotherapy [21]. However, relapse after first-line treatment and toxicity of chemotherapeutic agents are still high, so it is necessary to find an effective and safe treatment method that reduces side effects.
Recent studies have shown that next-generation sequencing (NGS)-based approaches provide a rationale for sequence-matching therapies to improve the overall drug response and survival rates in cancer patients [22]. The genetic landscape of cancer patients, including mutational status and gene expression patterns, may provide new druggable targets and pathways, leading to a clinical advantage for cancer patients [23,24,25]. Thus, a variety of NGS-based applications, such as whole transcriptome sequencing (RNA-seq) and assays for transposase-accessible chromatin sequencing (scATAC-seq), have been used to investigate the detailed genetic landscape of OC. These efforts not only provide detailed information for the understanding of the genetic variation of OC but also form the basis for immunotherapy, a new therapeutic strategy for the treatment of OC.
In this review, we discuss how therapeutic targets were discovered through NGS-based techniques and outline the future direction by summarizing the findings of current clinical trials on the efficacy of immunotherapy in OC.
2. NGS-Based Identification of New Pathways and Targets in OC
2.1. Bulk RNA-Sequencing Analysis (RNA-Seq)
New approaches for the treatment of OC are being intensively developed. In OC patients with BRCA1/2 mutations, PARP inhibitors improved drug response rates and prognosis by preventing the repair of damaged DNA, offering a different mechanism of anticancer treatment from conventional therapy, and evaluating the status of BRCA to prevent resistance to treatments [26,27,28]. Also, a recent study reported that the landscape of germline alterations could enhance the diagnosis, prognosis, prediction, and strategy of therapy in OC [29]. This suggests that molecular-targeted therapy may provide an appropriate strategy for the design of new clinical trials on OC treatment. Thus, the identification of molecular markers and signaling pathways relevant to the prognosis of OC is a key step in molecular targeted therapy. Bulk RNA-seq has been widely used in transcriptome studies as a replacement for microarrays. It has an average expression level in different cell types of samples [30].
CD151 is one of the molecules that regulates the migration and invasion of OC cells, as identified by RNA-seq [31]. The global expression pattern of OC, which represents both the early and late stages of OC, was investigated using RNA-seq. Interestingly, CD151 was expressed at all stages and did not differ between stages or specific subtypes. CD151 is restricted to the cell membranes on the surface of normal ovaries, indicating that when it is internalized into the cytoplasmic endosomes, it can dissociate from the primary tumor by weakening its ability to cooperate with other cells [32]. In vivo study targeting CD151 would affect cell survival and anti-CD151 monoclonal antibodies, which, involved in metastasis, may facilitate preventing dissemination of tumor cell and peritoneal spread, which is a critical cause of lethality in HGSC [31]. Moreover, depending on its expression level, CD151 has functional significance, as it can silence or block cell migration and invasion, suggesting that it could be an important emerging therapeutic target in OC.
RNA-seq-based mutation analysis may also be useful for the detection of racial differences in genetic variants of OC. New mutations, such as TP53 (67%), BRCA1 (38%), BRCA2 (54%), and ARID1A (13%), were observed in 24 Japanese OC specimens [33]. Although >90% of HGSC cases had TP53 mutations [34], not all TP53 mutations were observed in recurrent samples, because of sample heterogeneity in the same patient. Nevertheless, the use of RNA-seq for the investigation of genes that are not transcriptionally expressed, and those that are silenced after transcription, is limited.
RNA-seq has also been used to identify long non-coding RNAs (lncRNAs), particularly long intergenic non-coding RNAs (lincRNAs), which are mainly expressed in OC tissues [35]. lincRNAs are transcribed non-coding RNAs longer than 200 nucleotides, which do not overlap annotated coding genes. They share several features with lncRNA transcripts and compose more than half of lncRNAs in humans [36]. RNA-seq analysis revealed that OC with intergenic noncoding RNA1 (OIN1) is often more expressed in OC than in normal ovarian tissue. In addition, OIN1 expression negatively correlated with the expression of apoptosis-associated genes, ras-associated domain family member 5 (RASSF5), and adenosine A1 receptor (ADORA1), which were upregulated by OIN1 knockdown in OC. Moreover, increased expression of an oncogenic gene, known as urothelial cancer-associated 1 (UCA1), enhanced cell migration, invasion, and cisplatin resistance in OC [37]. These results demonstrate that OIN1 may have an ameliorating effect on OC progression by modulating oncogenes, suggesting that it has the potential to play an important role in the diagnosis and treatment of OC.
However, the expression of fusion genes in OC has rarely been reported. A fusion gene is a subtype of a hybrid gene containing two or more independent genes [38]. Recently, RNA-seq approaches detected two fusion genes, DPP9-PPP6R3 and DPP9-PLIN3, in advanced OC [39]. DPP9 is involved in the regulation of proliferative and survival pathways, and has the ability to suppress tumor cells [40]. In addition, the MUC1-TRIM46-KRTCAP2 fusion gene was detected by RNA-seq in HGSC [39]. MUC1 overexpression represents a diagnostic marker in metastatic and advanced OC, and these three fusion genes are readily detected only in tumor samples, suggesting that they may be potential biomarkers for diagnostic and therapeutic targets of OC [41].
RNA-seq analysis can be used to identify important factors related to OC stem cells (CSC). ZIP4, a Zn transporter, is present in the stem cell niche and has intestinal integrity [42]. It has been reported that the loss of ZIP4, which is upregulated in aggressive OC, decreased some cancer stem cell-related activities, such as tumor proliferation, anoikis resistance, colony formation, and particularly, spheroid formation, suggesting that ZIP4 is a promising and significant CSC regulator in OC [43]. A mechanistic study revealed that ZIP4 promoted the activation of lipid lysophosphatic acid (LPA) in an extracellular Zn-dependent manner, acting as a ligand for the nuclear receptor peroxisome proliferator-activated receptor gamma (PPARγ) [44]. This study demonstrated that the LPA–PPARγ–ZIP4 pathway potentiated ZIP4-positive tumors by promoting CSCs in human OC, indicating that ZIP4 is an important therapeutic target.
In general, human tumor cells are heterogeneous and complex, as they require the ability to migrate to attack tissues. The PI3K/AKT/mTOR pathway activates cell proliferation and migration, and inhibits apoptosis; therefore, the PI3K pathway is a good predictor of invasive and migratory abilities of human OC [45]. High levels of AKT activation have been observed in high-grade and late-stage serous OC [46]. This study showed that AKT exhibited anti-apoptotic effects and interfered with cell cycle arrest through the mTOR pathway. One study identified five genes that were differentially expressed in OC using the RNA-seq approach, which included ECM1, GPCR, NUR 77, AKT3, and PRKCB; these genes are the main components of the PI3K/AKT/mTOR pathway [47]. The study implied that the activation of the PI3K/AKT/mTOR signaling pathway led to improved invasive and migratory capacities of human OC cells. Therefore, controlling this pathway could be an effective therapeutic strategy for OC.
2.2. Single-Cell RNA-Sequencing Analysis (scRNA-Seq)
Although bulk RNA-seq-based approaches can elucidate the mechanisms associated with cancer development, OC metastasis, and various therapeutic targets, these approaches do not allow for accurate single-cell unit analysis, because they analyze all the cells in cancer tissue [48]. Heterogeneity is also a hallmark of OC, indicating that OC cells are composed of diverse populations with distinct genotypes and phenotypes [49]. scRNA-seq approaches can determine gene expression levels for each transcript and the expression distribution in each subpopulation of cells, resulting in the precise characterization of tumor formation. scRNA-seq, through quantitative and qualitative analyses of complex tissue cells, is essential for better understanding of cellular heterogeneity in OC studies, such as those related to metastasis, drug resistance, and treatment [50].
CXCL12 is a biomarker of OC, and its overexpression indicates the growth of OC [51]. Matrix metalloproteinases (MMPs) play a key role in cancer cell invasion and metastasis by remodeling the tumor microenvironment [52]. scRNA-seq of OC tissue has revealed that the increased expression of some collagens and MMP genes mostly occurs in cancer fibroblasts and stromal cells [50]. C3, a trigger in the complement system that promotes tumor development and angiogenesis, is highly expressed by metastatic fibroblasts. Moreover, surface deposition of C3 is often observed in OC patients [53]. Metastatic fibroblasts also highly express CXCL12, which plays a pivotal role in CXCR4 signaling in OC and several cancers, creating an inflammatory environment implicated in the development and growth of OC [50].
Li et al. performed scRNA-seq analysis with 66 HGSC cells to identify 6000 variable and signature genes in each cluster, sorted by principal component analysis (PCA). Differentially expressed genes are predominantly abundant in metabolic and cancer-associated pathways, including the PI3K-Akt signaling pathway, which plays an essential role in malignant cancers and is involved in tumor proliferation, survival, and angiogenesis [54]. The expression of STAT1, ANP32E, GPRC5A, and EGFL6 was higher in OC tissues than in normal tissues. In addition, the expression levels of PMP22, FBXO21, and CYB5R3 were significantly lower in OC tissues than in normal tissues. High expression of ANP32E, STAT1, GPRC5A, EGFL6, and PMP22 implies low survival in patients with OC, whereas low expression of FBXO21, ANP32E, and CYB5R3 indicates a longer recurrence-free survival period in OC [55]. These genes are closely associated with progression, metastasis, and cancer stem cells in various solid cancers. For example, STAT1 and ANP32E are overexpressed in OC, and this leads to a worse prognosis. In addition, GPRC5A mutation is closely related to the induction of self-renewal in bladder cancer. Taken together, these reports demonstrate that the marker genes ANP32E, STAT1, GPRC5A, EGFL6, PMP22, FBXO21, and CYB5R3 are strongly associated with OC prognosis [55].
2.3. Transposase-Accessible Chromatin Analysis by Sequencing (ATAC-Seq)
ATAC-seq is a method for mapping chromatin accessibility to DNA sequences using Tn5 transposase at the genomic level. It is widely used to acquire open chromatin and transcription-factor-binding regions for the identification of specific chromatin accessibility conditions [56]. Methodologically, a transposase carrying a known DNA sequence tag must be attached to the nucleus, followed by the opening of the chromatin at the insertion site [57]. Open chromatin is then recognized as a label of a known sequence, which is used to construct a library for sequencing [58].
In cancer research, ATAC-seq can detect the tissue-specific chromatin activity of regulatory regions in tumors. Previous studies have shown that mutant cohesins can enhance the chromatin accessibility of binding sites for transcription factors, including ERG, GATA2, and RUNX1 [59]. In addition, the protein encoded by the well-known cancer suppressor gene p53 suppresses cancer in the normal state but promotes tumor development in the mutant state [60]. ATAC-seq was used to confirm the pre-binding of p53 to enhancers and promoters in normal fibroblasts. When DNA is damaged, inaccessible chromatin becomes accessible and p53 is activated to maintain stability [59].
Gallon et al. identified enhancers associated with platinum resistance and found SOX9 to be an important cluster of cis-regulatory elements that regulate transcription factors [61]. SOX9 also plays an important role in the chemical resistance of OC cells [62]. In addition, patients with OC treated with platinum chemotherapy, with a high expression of THBS1, a gene that is downregulated in resistant cell lines, showed longer survival [61]. Denser chromatin regions have more adduct formation during resistance, particularly in the intergenic region, indicating that more platinum-induced damage occurs in the clusters of cis-regulatory elements in chromatin regions that develop into denser nucleosomes as drug resistance progresses. Therefore, targeting chromatin structures in a specific genomic context could be an important strategy in epigenetic therapy to overcome drug resistance.
Tissue-resident macrophages (TRMs) are a heterogeneous population of immune cells that perform tissue-specific and niche-specific functions [63]. They are present in all tissues and participate in homeostasis, immunity, and tissue repair. In OC cells, ATAC-seq revealed that macrophages residing in serous cavity tissues were significantly enriched in retinoic X receptor (RXR) transcripts and RXR response elements [64]. It has also been shown that the peritoneal serous cavity TRMs (LPM) infiltrate early ovarian tumors and that RXR deletion reduces LPM accumulation in tumors, particularly in mice [64]. This suggests that the RXR signaling pathway regulates the maintenance of serous macrophages and that peritoneal LPM may be a good target for OC therapy.
2.4. Combined Sequencing Analysis
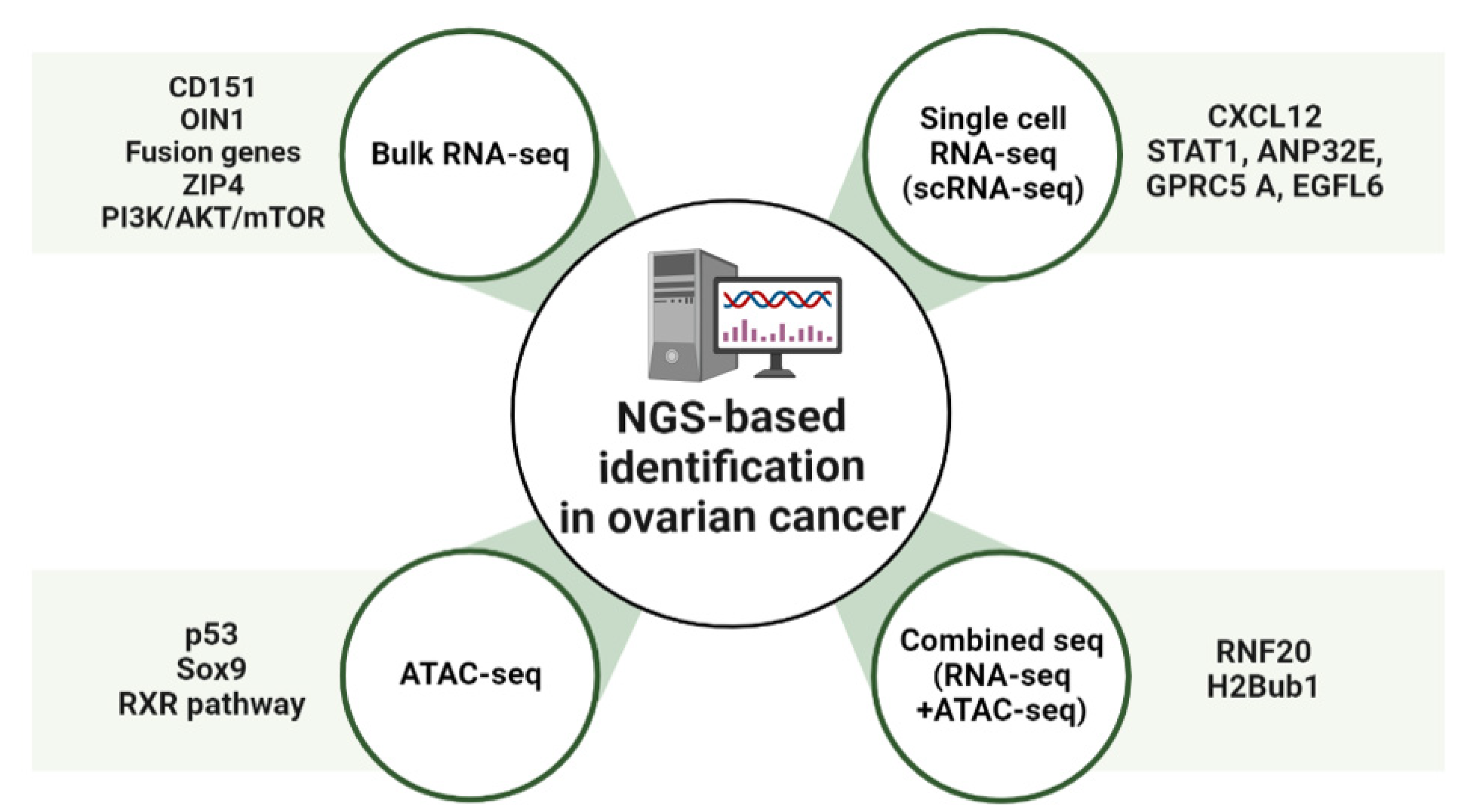
Advances in NGS-based applications using OC samples have elucidated the genetic landscape of patient tumors. Moreover, additional integrative analysis of NGS-based applications has provided further insight into tumor heterogeneity and detailed genetic information [65]. The role of the E3 ubiquitin ligase RNF20 and histone H2B monoubiquitination (H2Bub1), at the level of whole-genome changes in chromatin status, was investigated by gene expression analysis using ATAC-seq and RNA-seq [66]. The findings showed that in cells where RNF20 was deleted, the ATAC-seq read counts were enriched in the promoter region. Loss of H2Bub1 may lead to a more open chromatin structure in mammalian cells, implying a direct relationship between the open chromatin structure and loss of H2Bub1 [66]. In cells where RNF20 has been deleted, RNA-seq and ATAC-seq data showed a total of 1086 overlapping genes. The loss of H2Bub1, which is associated with immune pathways, can significantly alter IL-6-mediated immune signaling genes [66]. Moreover, the loss of H2Bub1 makes chromatin more accessible, suggesting that chromatin induces the upregulation of immunomodulators that promote the growth and migration of early HGSC states [66] (Figure 1 and Table 1).
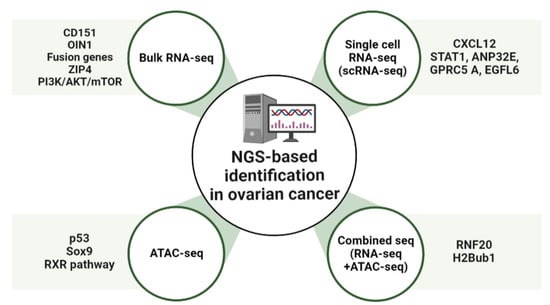
Figure 1.
Overview of NGS-based identification in ovarian cancer (OC). Schematic showing molecular targets of OC treatment. OIN1, Ovarian cancer long intergenic noncoding RNA 1; CXCL12, C-X-C motif chemokine 12; ANP32E, Acidic Nuclear Phosphoprotein 32 Family Member E; GPRC5A, G-protein coupled receptor family C group 5 member A; EGFL6, Epidermal growth factor-like domain-containing protein 6; RXR, Retinoid X receptor; RNF20, Ring finger protein 20.

Table 1.
Molecular targets discovered through NGS in ovarian cancer.
3. Immunotherapy in OC
In cancer research, immunotherapy enhances the immune response such that the patient’s immune cells can kill cancer cells. It is effective against several tumors and has innovative therapeutic potential to replace chemotherapy as the next-generation treatment for gynecological cancers [81]. Immunotherapy can be categorized into active and passive types. Active immunotherapy directly targets specific cancer antigens, and this involves treatment with immunostimulatory vaccines, genetically modified to recognize target antigens or chimeric antigen receptor T cells (CAR-T). Passive immunotherapy involves treatment with cytokines or checkpoint inhibitors, which indirectly activate a patient’s immune response [82]. These treatments are innovative, but according to the results of clinical trials, only some cancers can be treated, and side effects, such as cytokine syndrome and decreased immune function, can occur [81,83].
3.1. Immune-Stimulating Peptide Vaccines
Vaccine-mediated immunostimulatory therapy involves the use of a specific antigenic vaccine of the tumor, which is an immunotherapeutic agent that suppresses tumor growth or cancer recurrence by inducing an immune response [84,85]. Peptide vaccines consist of DNA vectors expressing dendritic cells, recognized by autologous tumor-specific antigens or tumor-specific peptides that induce immune activation, and the activity of which can be enhanced by various immune modulators [86,87].
3.1.1. P53 Peptide Vaccine
The most common mutant gene in human cancers is p53, with nearly 50% of OCs having p53 mutations [88]. Because p53 mutations mainly increase the expression and stability of the p53 protein, the amount of p53 in cancer cells is higher than that in normal cells [89]. Therefore, a p53 peptide vaccine could be a targeted therapeutic strategy for various cancer cells. It has been reported that p53-synthetic long peptide (p53-SLP) vaccine treatment in OC patients significantly induced p53-SLP-specific T-cell responses [90]. In addition, low-dose cyclophosphamide pre-treatment in patients with recurrent OC improved the immunogenicity of the p53-SLP vaccine, by enhancing the activity of regulatory T cells [91]. In a clinical study on platinum-resistant OC, p53 vaccine and gemcitabine significantly increased the reactivity of T cells, leading to better survival [92]. Recently, studies on the combination of various immunomodulators and p53 vaccines have been conducted to improve biochemical sensitivity and immune responses [93,94].
3.1.2. Wilms’ Tumor 1 (WT1) Peptide Vaccine
WT1 is often detected in various cancers, including leukemia, lung cancer, breast cancer, thyroid cancer, melanoma cancer, and OC [95]. Mouse and human WT1 genes have high homology, and hence, a murine model was used for WT1 vaccine development [95]. After confirming that the WT1 vaccine inhibited the growth of cancer cells by inducing a strong immune response, a WT1 vaccine using serum from cancer patients was evaluated for the first time [95]. HLA-A*24:02-restricted WT1-targeting vaccine promoted peptide-specific humoral immunity in refractory OC patients; therefore, WT1 levels can be used as a potential prognostic marker [96].
3.1.3. New York Esophageal Squamous Cell Carcinoma-1 (NYESO-1) Peptide Vaccine
NYESO-1 is a marker expressed in various cancer cells, including OC, and it is a significant target for vaccines [97]. Vaccine-mediated CD4+ and CD8+ T-cell responses, and the application of NY-ESO-1+ lymphocytes, have been reported [98]. The modified NY-ESO-1 vaccine rapidly induced a consistent and safe immune response in most vaccinated OC patients [99]. A recent study reported that the NYESO1 vaccine linked to the secretin-penetratin peptide induced a stronger and more specific T-cell immune response in a mouse animal model [100].
3.1.4. Glypican-3 (GPC3)-Derived Peptide Vaccine
GPC3, a heparan sulfate proteoglycan bound to the cell membrane by glycosylphosphatidylinositol, is specifically expressed in liver cancer, OC, lung cancer, and melanoma [101,102]. A recent report showed that the GPC3 vaccine improved the survival rate of chemoresistant OCs after chemotherapy [103]. In addition, it has been reported that adjuvant miRNA enhances the effect of GPC3 vaccine, and that the combination of miR-375, 193a, and 1128 can be used as predictive biomarkers [104]. The GPC3 vaccine was used in combination with CAR-T therapy, to significantly enhance the therapeutic effect against OC, suggesting that the GPC3 vaccine could be used in combination with several cancer treatments in the future [105].
3.1.5. Dendritic Cell (DC)-Based Peptide Vaccine
DCs are antigen-presenting cells of the immune system and are widely recognized as important cell types that can initiate antitumor responses [106]. DC-based vaccines are a novel strategy for effectively treating cancer patients, and hence, various clinical studies have been conducted using DC-related responses in cancer patients. Correll et al. reported that a DC vaccine could inhibit the activity of regulatory T cells in the blood of melanoma patients, and subsequently restore T-cell activity [107]. As a result of these observations, DC vaccines were evaluated in several carcinomas. Autologous DC-based vaccines with tumor lysates after chemotherapy in OC patients have also been reported to successfully reduce the rate of cancer progression and improve survival [108]. In addition, DC vaccines related to neoantigen peptides are being developed as clinical candidates for immune enhancement [109]. DC-based vaccines have great potential to improve the treatment of OC patients, and the advantage of using the immune system to induce a more sustained response, compared with that during cytotoxic chemotherapy [110]. However, further clinical studies of a cohort of patients with OC using DC vaccines are needed to confirm its safety and efficacy.
3.2. Blockade of Checkpoint
Checkpoint inhibitors are widely used to treat refractory tumors, including OC. Immune checkpoints are generally regulated by negative feedback inhibition, and they protect the host from autoimmunity and maintain self-tolerance by regulating the responses of various immune cells [111,112]. When activated T-cells recognize and bind to specific markers of tumor cells, immune checkpoints rapidly regulate the immune response through the T-cell receptor (TCR) signaling pathway [113].
3.2.1. Cytotoxic T-lymphocyte Antigen 4 (CTLA-4) Inhibitor
CTLA-4 inhibitors are widely used as immune checkpoint blockers to induce an immune response. PHI-101, a checkpoint kinase 2 inhibitor, showed a tumor-reducing effect on platinum-resistant, recurrent OC [114]. PHI-101 therapy was assessed in a phase IA clinical trial, and a phase II clinical trial has been recommended for the same. Ipilimumab, under the trade name Yervoy, is a CTLA-4 antibody generally used in the treatment of various cancers [115]. However, the anti-CTLA-4 antibodies need improvement, because immunotoxicity has often been reported in the liver, gastrointestinal tract, and endocrine system within the first few weeks of treatment [116].
3.2.2. Programed Cell Death Protein 1 (PD-1) and Programed Cell Death-Ligand 1 (PD-L1) Inhibitors
PD-1, a co-inhibitory receptor, inhibits T-cell activation, suggesting that it is a target for immunotherapy in cancer cells. PD-1 has two types of potential ligands: PD-L1 and PD-L2. PD-L1 is mainly expressed in most hematopoietic cells and vascular endothelial cells, whereas PD-L2 is expressed in some macrophages and dendritic cells [117]. PD-L1 is generally found in various malignancies, including OC [118,119]. PD-1–PD-L1 interaction normally inhibits the survival, proliferation, and functions of T cells [120]. Blocking the interaction of PD-1 with PD-L1 is widely used clinically for the treatment of tumors, and this strategy is less toxic than the use of CTLA-4 inhibitors, such as ipilimumab [121].
Pembrolizumab, also known as Keytruda, is a PD-1 inhibitor, and it has been effective in patients with recurrent OC [122,123]. A recent study showed that the combination of pembrolizumab and PEGylated liposomal doxorubicin has a potential therapeutic effect on platinum-resistant OC [124]. The combination of pembrolizumab with bevacizumab dramatically decreased the serum CA-125 level and regression of recurrent masses, with no marked side effects [125].
Nivolumab, under the trade name Opdivo, is a human monoclonal antibody approved for the treatment of several malignancies; it enhanced the anticancer activity of T cells by blocking the PD-1 and PD-L1/L2 pathways [118,126,127]. Monotherapy with nivolumab exhibited low tumor specificity and response in platinum-resistant OC [126]. Co-administration of nivolumab with various adjuvants has shown optimistic trends in the treatment of various cancers, including OC. In addition, the combination of nivolumab with the anti-CTLA4 antibody, ipilimumab, has been reported to improve the overall response to OC [128].
Atezolizumab (tecentriq; MPDL3280A) is an immunoglobulin mAb, that selectively interacts with PD-L1 in cancer cells, in the tumor microenvironment, and reactivates suppressed T cells to kill malignant tumors [129]. As PD-L1 expression is highly detectable in OC specimens, atezolizumab has been attracting attention as a potential immunotherapeutic agent. In addition, a combination of atezolizumab and other immunotherapeutic agents showed preliminary clinical activity in patients with OC [130]. For example, the combination of atezolizumab and bevacizumab induces a sustained response in some patients with platinum-resistant OC [131].
Abelumab, a monoclonal antibody capable of mediating antibody-dependent cytotoxicity, is a checkpoint inhibitor that interacts with PD-L1, especially when administered in combination with chemotherapeutic agents [132,133]. Several clinical studies have shown that avelumab monotherapy is effective in Merkel cell carcinoma (MCC) and urothelial cancer [134]. In addition, it has been reported that combination therapy with avelumab and axitinib increased the survival rate of urothelial cancer patients and showed significant activity when combined with docetaxel [134]. Several combination strategies have recently been evaluated, and further studies are underway to improve the response rate of avelumab in patients with OC [135,136,137].
3.3. Chimeric Antigen Receptor T (CAR-T) Cells
CAR-T cells are genetically modified patient-derived immune cells designed to activate the immune response by recognizing specific surface antigens on cancer cells. The inhibition of histone deacetylase (HDAC) activity plays an important role in CAR-T cell immune recognition. Co-treatment with sodium valproate (VPA), a representative HDAC inhibitor, and CAR-T enhanced the immune recognition of CAR-T cells in OC [138]. The identification of specific antigens overexpressed in cancer cells is an important strategy in CAR-T therapy. The most common target antigens for CAR-T in OC are mesothelin, mucin 16 (MUC16), folate receptor α, and human epidermal growth factor receptor 2 (HER2) [139].
Mesothelin is a new antigen that can be a target of CAR-T and is overexpressed in various cancers, including OC. However, it is also expressed in some normal tissues and has the disadvantage of causing off-target effects [140]. Mouse studies have shown that treatment with mesothelin-induced CAR-T cells is a potential therapy for OC. It was able to significantly prolong the survival of mice with OC [141].
Mucine 16 (MUC16), a glycoprotein of the mucin family, is expressed in various tumor cells and is involved in the proliferation and metastasis of cancer cells [142]. MUC16 is strongly expressed in most OCs and known well as a tumor marker (CA125), because it is cleaved and released from peripheral blood, suggesting that MUC16 is an ideal antigenic target for CAR-T. It has been reported that PD1-anti-MUC16 CAR-T cells have more potent anticancer activity than single MUC16-CAR-T cells in OC animal models [143,144]. The clinical application of MUC16 is still lacking due to several limitations, but immunotherapeutic studies using CAR-T cell construction are ongoing [145].
Folate receptor-α (FRα) protein is expressed at low levels in normal cells, specifically in OC [146]. T-cell activation using CAR targeting with FRα has been evaluated for use in OC treatment [147]. Recently, CAR-modified, cytokine-induced killer cells with FRα, enhanced anticancer immunity against OC [148].
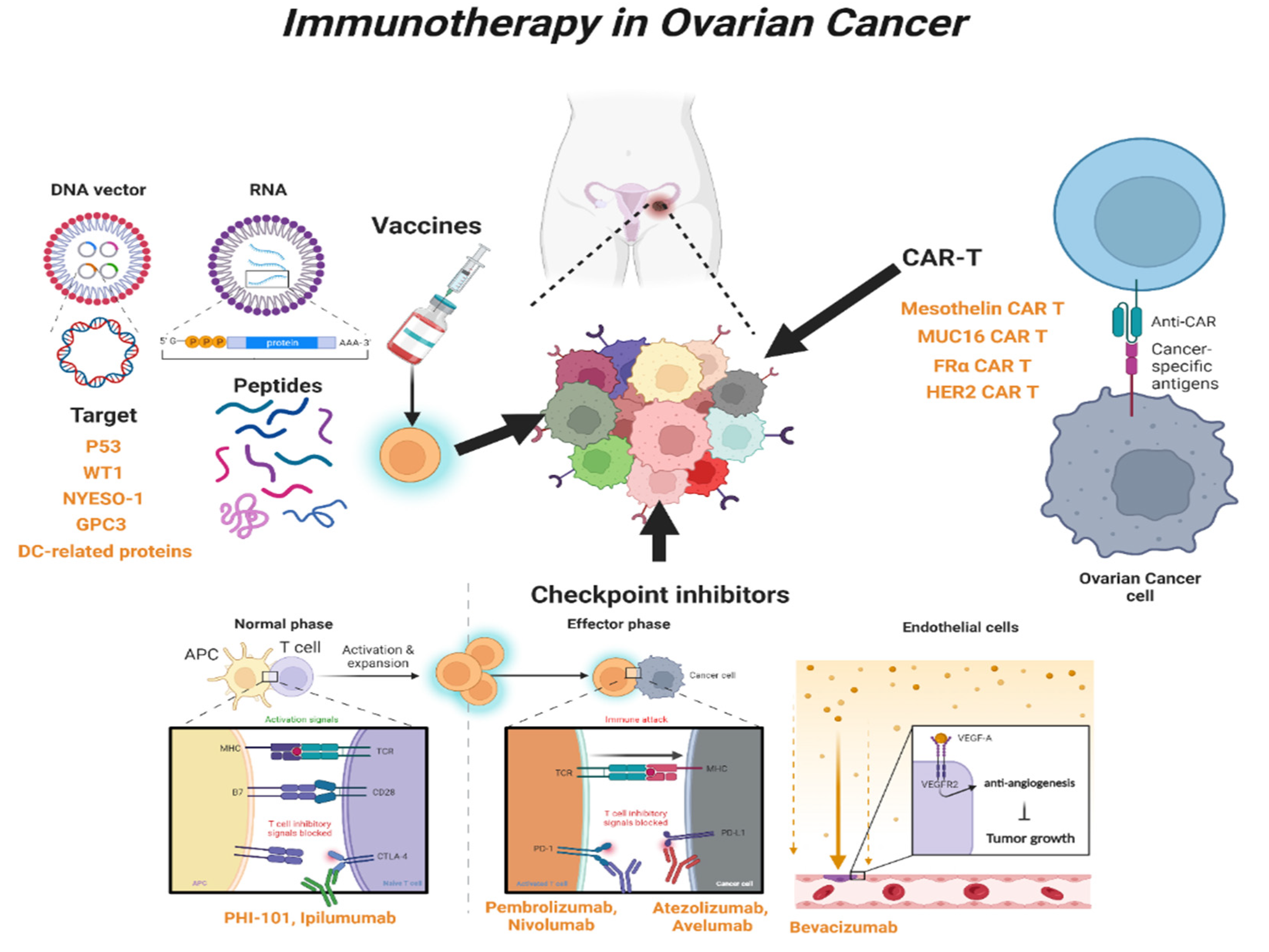
Human epidermal growth factor receptor 2 (HER-2) is overexpressed in breast cancer and OC [149]. One study reported that radiolabeled pertuzumab for HER2 imaging enables rapid and unambiguous delineation of OCs overexpressing HER2 [150]. The suppression of HER2 using shHER2-RNA treatment with cisplatin also enhanced the anticancer effect of OC [151]. The study of HER2-interact synthetic Notch CAR cells has been investigated in a mouse model, and it is expected that clinical therapeutics for HER2-CAR-T cells will also be developed in the near future [152] (Table 2 and Figure 2).

Table 2.
Immunotherapies for OC.
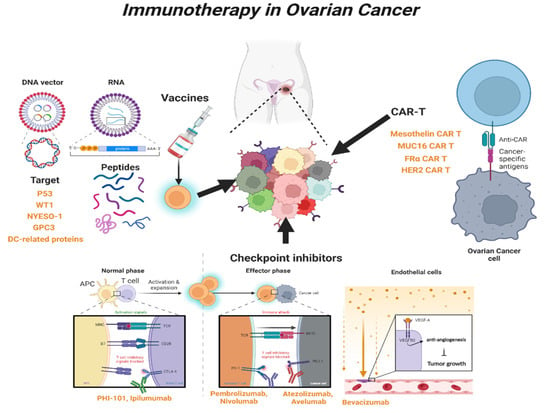
Figure 2.
Overview of immunotherapy in OC. Schematic showing the types and mechanisms of T-cell-mediated immunotherapy currently used in clinical practice. WT1, Wilms’ tumor 1; NYESO-1, New York esophageal squamous cell carcinoma-1; GPC3, Glypican-3; DC, Dendritic cell; CAR-T, Chimeric Antigen Receptor T; CTLA-4, Cytotoxic T-lymphocyte antigen 4; PD-1, Programed cell death protein 1; PD-L1, Programed cell death-ligand 1; MUC16, Mucin 16; FRα, Folate receptor-α; HER2, human epidermal growth factor receptor 2.
4. Conclusions
NGS-based applications have identified the detailed genetic landscape of cancer cells, including OC cells, providing different cancer treatment targets and strategies that can significantly improve patients’ prognoses. However, OC remains an incurable carcinoma that threatens the life of patients due to drug resistance and recurrence. The immune system plays an important role in the pathogenesis and treatment of OC, and hence, immunotherapy to suppress cancer cells by enhancing patients’ immunity has been continuously attempted in OC. The main directions of therapeutic approaches for OC reported so far are immune and vaccine-based treatment, checkpoint inhibition, and genetically engineered chimeric cells. Despite promising research results, the widespread clinical application of immunotherapy for OC is limited by insufficient experimental evidence. Therefore, epidemiological and clinical studies involving a broad population of OC patients, and expanded use of immunotherapeutic agents, are essential for improved patient clinical outcomes and enhanced survival of OC patients.
Author Contributions
H.Y. designed and wrote the manuscript; A.K. wrote the manuscript; H.J. supervised, organized, and wrote the manuscript; all authors revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Brain Korea 21 (BK21; # M2022B002600003), the Research Base Construction Fund Support Program funded by Jeonbuk National University in 2022, and the National Research Foundation of Korea (NRF), grant provided by the Korean government (MSIT) (No. 2021R1C1C1011346).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Wang, Q.; Peng, H.; Qi, X.; Wu, M.; Zhao, X. Targeted therapies in gynecological cancers: A comprehensive review of clinical evidence. Signal Transduct. Target. Ther. 2020, 5, 137. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Webb, P.M.; Jordan, S.J. Epidemiology of epithelial ovarian cancer. Best Pract. Res. Clin. Obstet. Gynaecol. 2017, 41, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Matulonis, U.; Sood, A.; Fallowfield, L.; Howitt, B.; Sehouli, J.; Karlan, B. Ovarian cancer. Nat. Rev. Dis. Prim. 2016, 2, 16061. [Google Scholar] [CrossRef] [PubMed]
- Prat, J. Ovarian carcinomas: Five distinct diseases with different origins, genetic alterations, and clinicopathological features. Virchows Arch. 2012, 460, 237–249. [Google Scholar] [CrossRef]
- Singer, G.; Oldt, R., 3rd; Cohen, Y.; Wang, B.G.; Sidransky, D.; Kurman, R.J.; Shih Ie, M. Mutations in BRAF and KRAS characterize the development of low-grade ovarian serous carcinoma. J. Natl. Cancer Inst. 2003, 95, 484–486. [Google Scholar] [CrossRef]
- Singer, G.; Stohr, R.; Cope, L.; Dehari, R.; Hartmann, A.; Cao, D.F.; Wang, T.L.; Kurman, R.J.; Shih Ie, M. Patterns of p53 mutations separate ovarian serous borderline tumors and low- and high-grade carcinomas and provide support for a new model of ovarian carcinogenesis: A mutational analysis with immunohistochemical correlation. Am. J. Surg. Pathol. 2005, 29, 218–224. [Google Scholar] [CrossRef]
- Reid, B.M.; Permuth, J.B.; Sellers, T.A. Epidemiology of ovarian cancer: A review. Cancer Biol. Med. 2017, 14, 9–32. [Google Scholar] [CrossRef]
- Katsumata, N.; Yasuda, M.; Isonishi, S.; Takahashi, F.; Michimae, H.; Kimura, E.; Aoki, D.; Jobo, T.; Kodama, S.; Terauchi, F.; et al. Long-term results of dose-dense paclitaxel and carboplatin versus conventional paclitaxel and carboplatin for treatment of advanced epithelial ovarian, fallopian tube, or primary peritoneal cancer (JGOG 3016): A randomised, controlled, open-label trial. Lancet Oncol. 2013, 14, 1020–1026. [Google Scholar] [CrossRef]
- Colombo, N.; Peiretti, M.; Parma, G.; Lapresa, M.; Mancari, R.; Carinelli, S.; Sessa, C.; Castiglione, M.; Group, E.G.W. Newly diagnosed and relapsed epithelial ovarian carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2010, 21, v23–v30. [Google Scholar] [CrossRef]
- Ledermann, J.A.; Raja, F.A.; Fotopoulou, C.; Gonzalez-Martin, A.; Colombo, N.; Sessa, C.; Group, E.G.W. Corrections to “Newly diagnosed and relapsed epithelial ovarian carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up”. Ann. Oncol. 2018, 29, iv259. [Google Scholar] [CrossRef]
- Christie, E.L.; Bowtell, D.D.L. Acquired chemotherapy resistance in ovarian cancer. Ann. Oncol. 2017, 28, viii13–viii15. [Google Scholar] [CrossRef]
- Pignata, S.; Pisano, C.; Di Napoli, M.; Cecere, S.C.; Tambaro, R.; Attademo, L. Treatment of recurrent epithelial ovarian cancer. Cancer 2019, 125, 4609–4615. [Google Scholar] [CrossRef]
- Conteduca, V.; Scarpi, E.; Farolfi, A.; Brighi, N.; Rossi, L.; Gurioli, G.; Lolli, C.; Schepisi, G.; Bleve, S.; Gianni, C.; et al. Melphalan as a Promising Treatment for BRCA-Related Ovarian Carcinoma. Front. Oncol. 2021, 11, 716467. [Google Scholar] [CrossRef]
- Kaern, J.; Baekelandt, M.; Trope, C.G. A phase II study of weekly paclitaxel in platinum and paclitaxel-resistant ovarian cancer patients. Eur. J. Gynaecol. Oncol. 2002, 23, 383–389. [Google Scholar]
- Ledermann, J.; Harter, P.; Gourley, C.; Friedlander, M.; Vergote, I.; Rustin, G.; Scott, C.L.; Meier, W.; Shapira-Frommer, R.; Safra, T.; et al. Olaparib maintenance therapy in patients with platinum-sensitive relapsed serous ovarian cancer: A preplanned retrospective analysis of outcomes by BRCA status in a randomised phase 2 trial. Lancet Oncol. 2014, 15, 852–861. [Google Scholar] [CrossRef]
- Moore, K.; Colombo, N.; Scambia, G.; Kim, B.G.; Oaknin, A.; Friedlander, M.; Lisyanskaya, A.; Floquet, A.; Leary, A.; Sonke, G.S.; et al. Maintenance Olaparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. N. Engl. J.Med. 2018, 379, 2495–2505. [Google Scholar] [CrossRef]
- Gonzalez-Martin, A.; Pothuri, B.; Vergote, I.; DePont Christensen, R.; Graybill, W.; Mirza, M.R.; McCormick, C.; Lorusso, D.; Hoskins, P.; Freyer, G.; et al. Niraparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. N. Engl. J. Med. 2019, 381, 2391–2402. [Google Scholar] [CrossRef]
- Lau, C.H.; Seow, K.M.; Chen, K.H. The Molecular Mechanisms of Actions, Effects, and Clinical Implications of PARP Inhibitors in Epithelial Ovarian Cancers: A Systematic Review. Int. J. Mol. Sci. 2022, 23, 8125. [Google Scholar] [CrossRef]
- McMullen, M.; Karakasis, K.; Madariaga, A.; Oza, A.M. Overcoming Platinum and PARP-Inhibitor Resistance in Ovarian Cancer. Cancers 2020, 12, 1607. [Google Scholar] [CrossRef]
- Klotz, D.M.; Wimberger, P. Overcoming PARP inhibitor resistance in ovarian cancer: What are the most promising strategies? Arch. Gynecol. Obstet. 2020, 302, 1087–1102. [Google Scholar] [CrossRef] [PubMed]
- Malone, E.R.; Oliva, M.; Sabatini, P.J.B.; Stockley, T.L.; Siu, L.L. Molecular profiling for precision cancer therapies. Genome. Med. 2020, 12, 8. [Google Scholar] [CrossRef] [PubMed]
- Garofalo, A.; Sholl, L.; Reardon, B.; Taylor-Weiner, A.; Amin-Mansour, A.; Miao, D.; Liu, D.; Oliver, N.; MacConaill, L.; Ducar, M.; et al. The impact of tumor profiling approaches and genomic data strategies for cancer precision medicine. Genome. Med. 2016, 8, 79. [Google Scholar] [CrossRef] [PubMed]
- Shyr, D.; Liu, Q. Next generation sequencing in cancer research and clinical application. Biol. Proced. Online 2013, 15, 4. [Google Scholar] [CrossRef]
- Morash, M.; Mitchell, H.; Beltran, H.; Elemento, O.; Pathak, J. The Role of Next-Generation Sequencing in Precision Medicine: A Review of Outcomes in Oncology. J. Pers. Med. 2018, 8, 30. [Google Scholar] [CrossRef]
- Tattersall, A.; Ryan, N.; Wiggans, A.J.; Rogozinska, E.; Morrison, J. Poly(ADP-ribose) polymerase (PARP) inhibitors for the treatment of ovarian cancer. Cochrane Database Syst. Rev. 2022, 2, CD007929. [Google Scholar] [CrossRef]
- Kyo, S.; Kanno, K.; Takakura, M.; Yamashita, H.; Ishikawa, M.; Ishibashi, T.; Sato, S.; Nakayama, K. Clinical Landscape of PARP Inhibitors in Ovarian Cancer: Molecular Mechanisms and Clues to Overcome Resistance. Cancers 2022, 14, 2504. [Google Scholar] [CrossRef]
- Palleschi, M.; Tedaldi, G.; Sirico, M.; Virga, A.; Ulivi, P.; De Giorgi, U. Moving beyond PARP Inhibition: Current State and Future Perspectives in Breast Cancer. Int. J. Mol. Sci. 2021, 22, 7884. [Google Scholar] [CrossRef]
- Gurioli, G.; Tedaldi, G.; Farolfi, A.; Petracci, E.; Casanova, C.; Comerci, G.; Danesi, R.; Arcangeli, V.; Ravegnani, M.; Calistri, D.; et al. Clinical Impact of Next-Generation Sequencing Multi-Gene Panel Highlighting the Landscape of Germline Alterations in Ovarian Cancer Patients. Int. J. Mol. Sci. 2022, 23, 5789. [Google Scholar] [CrossRef]
- Kuksin, M.; Morel, D.; Aglave, M.; Danlos, F.X.; Marabelle, A.; Zinovyev, A.; Gautheret, D.; Verlingue, L. Applications of single-cell and bulk RNA sequencing in onco-immunology. Eur. J. Cancer 2021, 149, 193–210. [Google Scholar] [CrossRef]
- Medrano, M.; Communal, L.; Brown, K.R.; Iwanicki, M.; Normand, J.; Paterson, J.; Sircoulomb, F.; Krzyzanowski, P.; Novak, M.; Doodnauth, S.A.; et al. Interrogation of Functional Cell-Surface Markers Identifies CD151 Dependency in High-Grade Serous Ovarian Cancer. Cell Rep. 2017, 18, 2343–2358. [Google Scholar] [CrossRef]
- Mosig, R.A.; Lin, L.; Senturk, E.; Shah, H.; Huang, F.; Schlosshauer, P.; Cohen, S.; Fruscio, R.; Marchini, S.; D’Incalci, M.; et al. Application of RNA-Seq transcriptome analysis: CD151 is an Invasion/Migration target in all stages of epithelial ovarian cancer. J. Ovarian. Res. 2012, 5, 4. [Google Scholar] [CrossRef]
- Nagasawa, S.; Ikeda, K.; Horie-Inoue, K.; Sato, S.; Takeda, S.; Hasegawa, K.; Inoue, S. Identification of novel mutations of ovarian cancer-related genes from RNA-sequencing data for Japanese epithelial ovarian cancer patients. Endocr. J. 2020, 67, 219–229. [Google Scholar] [CrossRef]
- Saleh, A.; Perets, R. Mutated p53 in HGSC-From a Common Mutation to a Target for Therapy. Cancers 2021, 13, 3465. [Google Scholar] [CrossRef]
- Pal, S.; Garg, M.; Pandey, A.K. Deciphering the Mounting Complexity of the p53 Regulatory Network in Correlation to Long Non-Coding RNAs (lncRNAs) in Ovarian Cancer. Cells 2020, 9, 527. [Google Scholar] [CrossRef]
- Ransohoff, J.D.; Wei, Y.; Khavari, P.A. The functions and unique features of long intergenic non-coding RNA. Nat. Rev. Mol. Cell Biol. 2018, 19, 143–157. [Google Scholar] [CrossRef]
- Takeiwa, T.; Mitobe, Y.; Ikeda, K.; Hasegawa, K.; Horie, K.; Inoue, S. Long Intergenic Noncoding RNA OIN1 Promotes Ovarian Cancer Growth by Modulating Apoptosis-Related Gene Expression. Int. J. Mol. Sci. 2021, 22, 1242. [Google Scholar] [CrossRef]
- Earp, M.A.; Raghavan, R.; Li, Q.; Dai, J.; Winham, S.J.; Cunningham, J.M.; Natanzon, Y.; Kalli, K.R.; Hou, X.; Weroha, S.J.; et al. Characterization of fusion genes in common and rare epithelial ovarian cancer histologic subtypes. Oncotarget 2017, 8, 46891–46899. [Google Scholar] [CrossRef]
- Wang, J.; Dean, D.C.; Hornicek, F.J.; Shi, H.; Duan, Z. RNA sequencing (RNA-Seq) and its application in ovarian cancer. Gynecol. Oncol. 2019, 152, 194–201. [Google Scholar] [CrossRef]
- Tang, Z.; Li, J.; Shen, Q.; Feng, J.; Liu, H.; Wang, W.; Xu, L.; Shi, G.; Ye, X.; Ge, M.; et al. Contribution of upregulated dipeptidyl peptidase 9 (DPP9) in promoting tumoregenicity, metastasis and the prediction of poor prognosis in non-small cell lung cancer (NSCLC). Int. J. Cancer 2017, 140, 1620–1632. [Google Scholar] [CrossRef]
- Deng, J.; Wang, L.; Chen, H.; Li, L.; Ma, Y.; Ni, J.; Li, Y. The role of tumour-associated MUC1 in epithelial ovarian cancer metastasis and progression. Cancer Metastasis Rev. 2013, 32, 535–551. [Google Scholar] [CrossRef]
- Fan, Q.; Zhang, W.; Emerson, R.E.; Xu, Y. ZIP4 Is a Novel Cancer Stem Cell Marker in High-Grade Serous Ovarian Cancer. Cancers 2020, 12, 3692. [Google Scholar] [CrossRef] [PubMed]
- Fan, Q.; Li, L.; Wang, T.L.; Emerson, R.E.; Xu, Y. A Novel ZIP4-HDAC4-VEGFA Axis in High-Grade Serous Ovarian Cancer. Cancers 2021, 13, 3821. [Google Scholar] [CrossRef] [PubMed]
- Fan, Q.; Cai, Q.; Li, P.; Wang, W.; Wang, J.; Gerry, E.; Wang, T.L.; Shih, I.M.; Nephew, K.P.; Xu, Y. The novel ZIP4 regulation and its role in ovarian cancer. Oncotarget 2017, 8, 90090–90107. [Google Scholar] [CrossRef] [PubMed]
- Ediriweera, M.K.; Tennekoon, K.H.; Samarakoon, S.R. Role of the PI3K/AKT/mTOR signaling pathway in ovarian cancer: Biological and therapeutic significance. Semin. Cancer Biol. 2019, 59, 147–160. [Google Scholar] [CrossRef]
- Hanrahan, A.J.; Schultz, N.; Westfal, M.L.; Sakr, R.A.; Giri, D.D.; Scarperi, S.; Janakiraman, M.; Olvera, N.; Stevens, E.V.; She, Q.B.; et al. Genomic complexity and AKT dependence in serous ovarian cancer. Cancer Discov. 2012, 2, 56–67. [Google Scholar] [CrossRef]
- Bai, H.; Li, H.; Li, W.; Gui, T.; Yang, J.; Cao, D.; Shen, K. The PI3K/AKT/mTOR pathway is a potential predictor of distinct invasive and migratory capacities in human ovarian cancer cell lines. Oncotarget 2015, 6, 25520–25532. [Google Scholar] [CrossRef]
- Svensson, V.; Natarajan, K.N.; Ly, L.H.; Miragaia, R.J.; Labalette, C.; Macaulay, I.C.; Cvejic, A.; Teichmann, S.A. Power analysis of single-cell RNA-sequencing experiments. Nat. Methods 2017, 14, 381–387. [Google Scholar] [CrossRef]
- Kossai, M.; Leary, A.; Scoazec, J.Y.; Genestie, C. Ovarian Cancer: A Heterogeneous Disease. Pathobiology 2018, 85, 41–49. [Google Scholar] [CrossRef]
- Shih, A.J.; Menzin, A.; Whyte, J.; Lovecchio, J.; Liew, A.; Khalili, H.; Bhuiya, T.; Gregersen, P.K.; Lee, A.T. Identification of grade and origin specific cell populations in serous epithelial ovarian cancer by single cell RNA-seq. PLoS ONE 2018, 13, e0206785. [Google Scholar] [CrossRef]
- Mao, T.L.; Fan, K.F.; Liu, C.L. Targeting the CXCR4/CXCL12 axis in treating epithelial ovarian cancer. Gene Ther. 2017, 24, 621–629. [Google Scholar] [CrossRef]
- Najafi, M.; Farhood, B.; Mortezaee, K. Extracellular matrix (ECM) stiffness and degradation as cancer drivers. J. Cell Biochem. 2019, 120, 2782–2790. [Google Scholar] [CrossRef]
- Bareke, H.; Akbuga, J. Complement system’s role in cancer and its therapeutic potential in ovarian cancer. Scand. J. Immunol. 2018, 88, e12672. [Google Scholar] [CrossRef]
- Jiang, N.; Dai, Q.; Su, X.; Fu, J.; Feng, X.; Peng, J. Role of PI3K/AKT pathway in cancer: The framework of malignant behavior. Mol. Biol. Rep. 2020, 47, 4587–4629. [Google Scholar] [CrossRef]
- Li, Y.; Wang, J.; Wang, F.; Gao, C.; Cao, Y.; Wang, J. Identification of Specific Cell Subpopulations and Marker Genes in Ovarian Cancer Using Single-Cell RNA Sequencing. Biomed. Res. Int. 2021, 2021, 1005793. [Google Scholar] [CrossRef]
- Li, Z.; Schulz, M.H.; Look, T.; Begemann, M.; Zenke, M.; Costa, I.G. Identification of transcription factor binding sites using ATAC-seq. Genome Biol. 2019, 20, 45. [Google Scholar] [CrossRef]
- Bysani, M.; Agren, R.; Davegardh, C.; Volkov, P.; Ronn, T.; Unneberg, P.; Bacos, K.; Ling, C. ATAC-seq reveals alterations in open chromatin in pancreatic islets from subjects with type 2 diabetes. Sci. Rep. 2019, 9, 7785. [Google Scholar] [CrossRef]
- Zhu, C.; Yu, M.; Huang, H.; Juric, I.; Abnousi, A.; Hu, R.; Lucero, J.; Behrens, M.M.; Hu, M.; Ren, B. An ultra high-throughput method for single-cell joint analysis of open chromatin and transcriptome. Nat. Struct. Mol. Biol. 2019, 26, 1063–1070. [Google Scholar] [CrossRef]
- Sun, Y.; Miao, N.; Sun, T. Detect accessible chromatin using ATAC-sequencing, from principle to applications. Hereditas 2019, 156, 29. [Google Scholar] [CrossRef]
- Boutelle, A.M.; Attardi, L.D. p53 and Tumor Suppression: It Takes a Network. Trends Cell Biol. 2021, 31, 298–310. [Google Scholar] [CrossRef]
- Gallon, J.; Loomis, E.; Curry, E.; Martin, N.; Brody, L.; Garner, I.; Brown, R.; Flanagan, J.M. Chromatin accessibility changes at intergenic regions are associated with ovarian cancer drug resistance. Clin. Epigenetics 2021, 13, 122. [Google Scholar] [CrossRef] [PubMed]
- Panda, M.; Tripathi, S.K.; Biswal, B.K. SOX9: An emerging driving factor from cancer progression to drug resistance. Biochim Biophys Acta. Rev. Cancer 2021, 1875, 188517. [Google Scholar] [CrossRef] [PubMed]
- Davies, L.C.; Jenkins, S.J.; Allen, J.E.; Taylor, P.R. Tissue-resident macrophages. Nat. Immunol. 2013, 14, 986–995. [Google Scholar] [CrossRef] [PubMed]
- Casanova-Acebes, M.; Menendez-Gutierrez, M.P.; Porcuna, J.; Alvarez-Errico, D.; Lavin, Y.; Garcia, A.; Kobayashi, S.; Le Berichel, J.; Nunez, V.; Were, F.; et al. RXRs control serous macrophage neonatal expansion and identity and contribute to ovarian cancer progression. Nat. Commun. 2020, 11, 1655. [Google Scholar] [CrossRef]
- Pongor, L.; Harami-Papp, H.; Mehes, E.; Czirok, A.; Gyorffy, B. Cell Dispersal Influences Tumor Heterogeneity and Introduces a Bias in NGS Data Interpretation. Sci. Rep. 2017, 7, 7358. [Google Scholar] [CrossRef]
- Hooda, J.; Novak, M.; Salomon, M.P.; Matsuba, C.; Ramos, R.I.; MacDuffie, E.; Song, M.; Hirsch, M.S.; Lester, J.; Parkash, V.; et al. Early Loss of Histone H2B Monoubiquitylation Alters Chromatin Accessibility and Activates Key Immune Pathways That Facilitate Progression of Ovarian Cancer. Cancer Res. 2019, 79, 760–772. [Google Scholar] [CrossRef]
- Kumari, S.; Devi, G.t.; Badana, A.; Dasari, V.R.; Malla, R.R. CD151-A Striking Marker for Cancer Therapy. Biomark Cancer 2015, 7, 7–11. [Google Scholar] [CrossRef]
- Zhu, J.; Cai, T.; Zhou, J.; Du, W.; Zeng, Y.; Liu, T.; Fu, Y.; Li, Y.; Qian, Q.; Yang, X.H.; et al. CD151 drives cancer progression depending on integrin alpha3beta1 through EGFR signaling in non-small cell lung cancer. J. Exp. Clin. Cancer Res. 2021, 40, 192. [Google Scholar] [CrossRef]
- Huang, J.; Zhou, N.; Watabe, K.; Lu, Z.; Wu, F.; Xu, M.; Mo, Y.Y. Long non-coding RNA UCA1 promotes breast tumor growth by suppression of p27 (Kip1). Cell Death Dis. 2014, 5, e1008. [Google Scholar] [CrossRef]
- Nath, S.; Mukherjee, P. MUC1: A multifaceted oncoprotein with a key role in cancer progression. Trends Mol. Med. 2014, 20, 332–342. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhan, H.; Zhang, Y.; Yang, J.; Liu, M.; Xu, C.; Fan, X.; Zhang, J.; Zhou, Z.; Shi, X.; et al. ZIP4 promotes non-small cell lung cancer metastasis by activating snail-N-cadherin signaling axis. Cancer Lett. 2021, 521, 71–81. [Google Scholar] [CrossRef]
- Ersahin, T.; Tuncbag, N.; Cetin-Atalay, R. The PI3K/AKT/mTOR interactive pathway. Mol. Biosyst. 2015, 11, 1946–1954. [Google Scholar] [CrossRef]
- Yang, J.; Nie, J.; Ma, X.; Wei, Y.; Peng, Y.; Wei, X. Targeting PI3K in cancer: Mechanisms and advances in clinical trials. Mol. Cancer 2019, 18, 26. [Google Scholar] [CrossRef]
- Janssens, R.; Struyf, S.; Proost, P. The unique structural and functional features of CXCL12. Cell Mol. Immunol. 2018, 15, 299–311. [Google Scholar] [CrossRef]
- Obri, A.; Ouararhni, K.; Papin, C.; Diebold, M.L.; Padmanabhan, K.; Marek, M.; Stoll, I.; Roy, L.; Reilly, P.T.; Mak, T.W.; et al. ANP32E is a histone chaperone that removes H2A.Z from chromatin. Nature 2014, 505, 648–653. [Google Scholar] [CrossRef]
- Meissl, K.; Macho-Maschler, S.; Muller, M.; Strobl, B. The good and the bad faces of STAT1 in solid tumours. Cytokine 2017, 89, 12–20. [Google Scholar] [CrossRef]
- Donehower, L.A.; Soussi, T.; Korkut, A.; Liu, Y.; Schultz, A.; Cardenas, M.; Li, X.; Babur, O.; Hsu, T.K.; Lichtarge, O.; et al. Integrated Analysis of TP53 Gene and Pathway Alterations in The Cancer Genome Atlas. Cell Rep. 2019, 28, 3010. [Google Scholar] [CrossRef]
- Jo, A.; Denduluri, S.; Zhang, B.; Wang, Z.; Yin, L.; Yan, Z.; Kang, R.; Shi, L.L.; Mok, J.; Lee, M.J.; et al. The versatile functions of Sox9 in development, stem cells, and human diseases. Genes Dis 2014, 1, 149–161. [Google Scholar] [CrossRef]
- Krezel, W.; Ruhl, R.; de Lera, A.R. Alternative retinoid X receptor (RXR) ligands. Mol. Cell Endocrinol. 2019, 491, 110436. [Google Scholar] [CrossRef]
- Wang, F.; El-Saafin, F.; Ye, T.; Stierle, M.; Negroni, L.; Durik, M.; Fischer, V.; Devys, D.; Vincent, S.D.; Tora, L. Histone H2Bub1 deubiquitylation is essential for mouse development, but does not regulate global RNA polymerase II transcription. Cell Death Differ. 2021, 28, 2385–2403. [Google Scholar] [CrossRef]
- Levinson, K.; Dorigo, O.; Rubin, K.; Moore, K. Immunotherapy in Gynecologic Cancers: What We Know Now and Where We Are Headed. Am. Soc. Clin. Oncol. Educ. Book 2019, 39, e126–e140. [Google Scholar] [CrossRef] [PubMed]
- Lobenwein, D.; Kocher, F.; Dobner, S.; Gollmann-Tepekoylu, C.; Holfeld, J. Cardiotoxic mechanisms of cancer immunotherapy—A systematic review. Int. J. Cardiol. 2021, 323, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Palaia, I.; Tomao, F.; Sassu, C.M.; Musacchio, L.; Benedetti Panici, P. Immunotherapy For Ovarian Cancer: Recent Advances And Combination Therapeutic Approaches. Onco. Targets Ther. 2020, 13, 6109–6129. [Google Scholar] [CrossRef] [PubMed]
- Seledtsov, V.I.; von Delwig, A. Clinically feasible and prospective immunotherapeutic interventions in multidirectional comprehensive treatment of cancer. Expert Opin. Biol. Ther. 2021, 21, 323–342. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, L.; Xu, Z.; Miao, L.; Huang, L. mRNA Vaccine with Antigen-Specific Checkpoint Blockade Induces an Enhanced Immune Response against Established Melanoma. Mol. Ther. 2018, 26, 420–434. [Google Scholar] [CrossRef]
- Yang, X.; Fan, J.; Wu, Y.; Ma, Z.; Huang, J.; Zhang, Y.; Zhou, Z.; Mo, F.; Liu, X.; Yuan, H.; et al. Synthetic multiepitope neoantigen DNA vaccine for personalized cancer immunotherapy. Nanomedicine 2021, 37, 102443. [Google Scholar] [CrossRef]
- Zamani, P.; Teymouri, M.; Nikpoor, A.R.; Navashenaq, J.G.; Gholizadeh, Z.; Darban, S.A.; Jaafari, M.R. Nanoliposomal vaccine containing long multi-epitope peptide E75-AE36 pulsed PADRE-induced effective immune response in mice TUBO model of breast cancer. Eur. J. Cancer 2020, 129, 80–96. [Google Scholar] [CrossRef]
- Marks, J.R.; Davidoff, A.M.; Kerns, B.J.; Humphrey, P.A.; Pence, J.C.; Dodge, R.K.; Clarke-Pearson, D.L.; Iglehart, J.D.; Bast, R.C., Jr.; Berchuck, A. Overexpression and mutation of p53 in epithelial ovarian cancer. Cancer Res. 1991, 51, 2979–2984. [Google Scholar]
- Havrilesky, L.; Darcy, K.M.; Hamdan, H.; Priore, R.L.; Leon, J.; Bell, J.; Berchuck, A.; Gynecologic Oncology Group, S. Prognostic significance of p53 mutation and p53 overexpression in advanced epithelial ovarian cancer: A Gynecologic Oncology Group Study. J. Clin. Oncol. 2003, 21, 3814–3825. [Google Scholar] [CrossRef]
- Leffers, N.; Lambeck, A.J.; Gooden, M.J.; Hoogeboom, B.N.; Wolf, R.; Hamming, I.E.; Hepkema, B.G.; Willemse, P.H.; Molmans, B.H.; Hollema, H.; et al. Immunization with a P53 synthetic long peptide vaccine induces P53-specific immune responses in ovarian cancer patients, a phase II trial. Int. J. Cancer 2009, 125, 2104–2113. [Google Scholar] [CrossRef]
- Vermeij, R.; Leffers, N.; Hoogeboom, B.N.; Hamming, I.L.; Wolf, R.; Reyners, A.K.; Molmans, B.H.; Hollema, H.; Bart, J.; Drijfhout, J.W.; et al. Potentiation of a p53-SLP vaccine by cyclophosphamide in ovarian cancer: A single-arm phase II study. Int. J. Cancer 2012, 131, E670–E680. [Google Scholar] [CrossRef]
- Hardwick, N.R.; Frankel, P.; Ruel, C.; Kilpatrick, J.; Tsai, W.; Kos, F.; Kaltcheva, T.; Leong, L.; Morgan, R.; Chung, V.; et al. p53-Reactive T Cells Are Associated with Clinical Benefit in Patients with Platinum-Resistant Epithelial Ovarian Cancer After Treatment with a p53 Vaccine and Gemcitabine Chemotherapy. Clin. Cancer Res. 2018, 24, 1315–1325. [Google Scholar] [CrossRef]
- Davodabadi, F.; Sarhadi, M.; Arabpour, J.; Sargazi, S.; Rahdar, A.; Diez-Pascual, A.M. Breast cancer vaccines: New insights into immunomodulatory and nano-therapeutic approaches. J. Control Release 2022, 349, 844–875. [Google Scholar] [CrossRef]
- Advani, S.; Kopetz, S. Ongoing and future directions in the management of metastatic colorectal cancer: Update on clinical trials. J. Surg. Oncol. 2019, 119, 642–652. [Google Scholar] [CrossRef]
- Gaiger, A.; Reese, V.; Disis, M.L.; Cheever, M.A. Immunity to WT1 in the animal model and in patients with acute myeloid leukemia. Blood 2000, 96, 1480–1489. [Google Scholar] [CrossRef]
- Nishida, S.; Morimoto, S.; Oji, Y.; Morita, S.; Shirakata, T.; Enomoto, T.; Tsuboi, A.; Ueda, Y.; Yoshino, K.; Shouq, A.; et al. Cellular and Humoral Immune Responses Induced by an HLA Class I-restricted Peptide Cancer Vaccine Targeting WT1 Are Associated With Favorable Clinical Outcomes in Advanced Ovarian Cancer. J. Immunother. 2022, 45, 56–66. [Google Scholar] [CrossRef]
- Odunsi, K.; Jungbluth, A.A.; Stockert, E.; Qian, F.; Gnjatic, S.; Tammela, J.; Intengan, M.; Beck, A.; Keitz, B.; Santiago, D.; et al. NY-ESO-1 and LAGE-1 cancer-testis antigens are potential targets for immunotherapy in epithelial ovarian cancer. Cancer Res. 2003, 63, 6076–6083. [Google Scholar]
- Odunsi, K.; Qian, F.; Matsuzaki, J.; Mhawech-Fauceglia, P.; Andrews, C.; Hoffman, E.W.; Pan, L.; Ritter, G.; Villella, J.; Thomas, B.; et al. Vaccination with an NY-ESO-1 peptide of HLA class I/II specificities induces integrated humoral and T cell responses in ovarian cancer. Proc. Natl. Acad. Sci. USA 2007, 104, 12837–12842. [Google Scholar] [CrossRef]
- Odunsi, K.; Matsuzaki, J.; Karbach, J.; Neumann, A.; Mhawech-Fauceglia, P.; Miller, A.; Beck, A.; Morrison, C.D.; Ritter, G.; Godoy, H.; et al. Efficacy of vaccination with recombinant vaccinia and fowlpox vectors expressing NY-ESO-1 antigen in ovarian cancer and melanoma patients. Proc. Natl. Acad. Sci. USA 2012, 109, 5797–5802. [Google Scholar] [CrossRef]
- Yang, Y.; Guo, X.; Hu, B.; He, P.; Jiang, X.; Wang, Z.; Zhu, H.; Hu, L.; Yu, M.; Feng, M. Generated SecPen_NY-ESO-1_ubiquitin-pulsed dendritic cell cancer vaccine elicits stronger and specific T cell immune responses. Acta. Pharm. Sin. B 2021, 11, 476–487. [Google Scholar] [CrossRef]
- Yu, S.; Yi, M.; Qin, S.; Wu, K. Next generation chimeric antigen receptor T cells: Safety strategies to overcome toxicity. Mol. Cancer 2019, 18, 125. [Google Scholar] [CrossRef] [PubMed]
- Nakatsura, T.; Nishimura, Y. Usefulness of the novel oncofetal antigen glypican-3 for diagnosis of hepatocellular carcinoma and melanoma. BioDrugs 2005, 19, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, S.; Sakata, J.; Utsumi, F.; Sekiya, R.; Kajiyama, H.; Shibata, K.; Kikkawa, F.; Nakatsura, T. Efficacy of glypican-3-derived peptide vaccine therapy on the survival of patients with refractory ovarian clear cell carcinoma. Oncoimmunology 2016, 5, e1238542. [Google Scholar] [CrossRef] [PubMed]
- Ukai, M.; Yokoi, A.; Yoshida, K.; Suzuki, S.; Shibata, K.; Kikkawa, F.; Nakatsura, T.; Kajiyama, H. Extracellular miRNAs as Predictive Biomarkers for Glypican-3-Derived Peptide Vaccine Therapy Response in Ovarian Clear Cell Carcinoma. Cancers 2021, 13, 550. [Google Scholar] [CrossRef]
- Pang, N.; Shi, J.; Qin, L.; Chen, A.; Tang, Y.; Yang, H.; Huang, Y.; Wu, Q.; Li, X.; He, B.; et al. IL-7 and CCL19-secreting CAR-T cell therapy for tumors with positive glypican-3 or mesothelin. J. Hematol. Oncol. 2021, 14, 118. [Google Scholar] [CrossRef]
- Hubbe, M.L.; Jaehger, D.E.; Andresen, T.L.; Andersen, M.H. Leveraging Endogenous Dendritic Cells to Enhance the Therapeutic Efficacy of Adoptive T-Cell Therapy and Checkpoint Blockade. Front. Immunol. 2020, 11, 578349. [Google Scholar] [CrossRef]
- Correll, A.; Tuettenberg, A.; Becker, C.; Jonuleit, H. Increased regulatory T-cell frequencies in patients with advanced melanoma correlate with a generally impaired T-cell responsiveness and are restored after dendritic cell-based vaccination. Exp. Dermatol. 2010, 19, e213–e221. [Google Scholar] [CrossRef]
- Tanyi, J.L.; Bobisse, S.; Ophir, E.; Tuyaerts, S.; Roberti, A.; Genolet, R.; Baumgartner, P.; Stevenson, B.J.; Iseli, C.; Dangaj, D.; et al. Personalized cancer vaccine effectively mobilizes antitumor T cell immunity in ovarian cancer. Sci. Transl. Med 2018, 10, eaao5931. [Google Scholar] [CrossRef]
- Morisaki, T.; Hikichi, T.; Onishi, H.; Morisaki, T.; Kubo, M.; Hirano, T.; Yoshimura, S.; Kiyotani, K.; Nakamura, Y. Intranodal Administration of Neoantigen Peptide-loaded Dendritic Cell Vaccine Elicits Epitope-specific T Cell Responses and Clinical Effects in a Patient with Chemorefractory Ovarian Cancer with Malignant Ascites. Immunol. Investig. 2021, 50, 562–579. [Google Scholar] [CrossRef]
- Caro, A.A.; Deschoemaeker, S.; Allonsius, L.; Coosemans, A.; Laoui, D. Dendritic Cell Vaccines: A Promising Approach in the Fight against Ovarian Cancer. Cancers 2022, 14, 4037. [Google Scholar] [CrossRef]
- Fares, C.M.; Van Allen, E.M.; Drake, C.G.; Allison, J.P.; Hu-Lieskovan, S. Mechanisms of Resistance to Immune Checkpoint Blockade: Why Does Checkpoint Inhibitor Immunotherapy Not Work for All Patients? Am. Soc. Clin. Oncol. Educ. Book 2019, 39, 147–164. [Google Scholar] [CrossRef]
- Gonzalez, N.L.; Puwanant, A.; Lu, A.; Marks, S.M.; Zivkovic, S.A. Myasthenia triggered by immune checkpoint inhibitors: New case and literature review. Neuromuscul. Disord. 2017, 27, 266–268. [Google Scholar] [CrossRef]
- Kreileder, M.; Barrett, I.; Bendtsen, C.; Brennan, D.; Kolch, W. Signaling Dynamics Regulating Crosstalks between T-Cell Activation and Immune Checkpoints. Trends Cell Biol. 2021, 31, 224–235. [Google Scholar] [CrossRef]
- Park, S.J.; Chang, S.J.; Suh, D.H.; Kong, T.W.; Song, H.; Kim, T.H.; Kim, J.W.; Kim, H.S.; Lee, S.J. A phase IA dose-escalation study of PHI-101, a new checkpoint kinase 2 inhibitor, for platinum-resistant recurrent ovarian cancer. BMC Cancer 2022, 22, 28. [Google Scholar] [CrossRef]
- Hodi, F.S.; O’Day, S.J.; McDermott, D.F.; Weber, R.W.; Sosman, J.A.; Haanen, J.B.; Gonzalez, R.; Robert, C.; Schadendorf, D.; Hassel, J.C.; et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 2010, 363, 711–723. [Google Scholar] [CrossRef]
- Chang, H.W.; Frey, G.; Liu, H.; Xing, C.; Steinman, L.; Boyle, W.J.; Short, J.M. Generating tumor-selective conditionally active biologic anti-CTLA4 antibodies via protein-associated chemical switches. Proc. Natl. Acad. Sci. USA 2021, 118, e2020606118. [Google Scholar] [CrossRef]
- Loke, P.; Allison, J.P. PD-L1 and PD-L2 are differentially regulated by Th1 and Th2 cells. Proc. Natl. Acad. Sci. USA 2003, 100, 5336–5341. [Google Scholar] [CrossRef]
- Hamanishi, J.; Mandai, M.; Iwasaki, M.; Okazaki, T.; Tanaka, Y.; Yamaguchi, K.; Higuchi, T.; Yagi, H.; Takakura, K.; Minato, N.; et al. Programmed cell death 1 ligand 1 and tumor-infiltrating CD8+ T lymphocytes are prognostic factors of human ovarian cancer. Proc. Natl. Acad. Sci. USA 2007, 104, 3360–3365. [Google Scholar] [CrossRef]
- Hino, R.; Kabashima, K.; Kato, Y.; Yagi, H.; Nakamura, M.; Honjo, T.; Okazaki, T.; Tokura, Y. Tumor cell expression of programmed cell death-1 ligand 1 is a prognostic factor for malignant melanoma. Cancer 2010, 116, 1757–1766. [Google Scholar] [CrossRef]
- Tseng, S.Y.; Otsuji, M.; Gorski, K.; Huang, X.; Slansky, J.E.; Pai, S.I.; Shalabi, A.; Shin, T.; Pardoll, D.M.; Tsuchiya, H. B7-DC, a new dendritic cell molecule with potent costimulatory properties for T cells. J. Exp. Med. 2001, 193, 839–846. [Google Scholar] [CrossRef]
- Fecher, L.A.; Agarwala, S.S.; Hodi, F.S.; Weber, J.S. Ipilimumab and its toxicities: A multidisciplinary approach. Oncologist 2013, 18, 733–743. [Google Scholar] [CrossRef] [PubMed]
- Matulonis, U.A.; Shapira-Frommer, R.; Santin, A.D.; Lisyanskaya, A.S.; Pignata, S.; Vergote, I.; Raspagliesi, F.; Sonke, G.S.; Birrer, M.; Provencher, D.M.; et al. Antitumor activity and safety of pembrolizumab in patients with advanced recurrent ovarian cancer: Results from the phase II KEYNOTE-100 study. Ann. Oncol. 2019, 30, 1080–1087. [Google Scholar] [CrossRef] [PubMed]
- Mehnert, J.M.; Varga, A.; Brose, M.S.; Aggarwal, R.R.; Lin, C.C.; Prawira, A.; de Braud, F.; Tamura, K.; Doi, T.; Piha-Paul, S.A.; et al. Safety and antitumor activity of the anti-PD-1 antibody pembrolizumab in patients with advanced, PD-L1-positive papillary or follicular thyroid cancer. BMC Cancer 2019, 19, 196. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.K.; Xiong, N.; Cheng, S.C.; Barry, W.T.; Penson, R.T.; Konstantinopoulos, P.A.; Hoffman, M.A.; Horowitz, N.; Dizon, D.S.; Stover, E.H.; et al. Combined pembrolizumab and pegylated liposomal doxorubicin in platinum resistant ovarian cancer: A phase 2 clinical trial. Gynecol. Oncol. 2020, 159, 72–78. [Google Scholar] [CrossRef]
- Lin, Y.C.; Wen, K.C.; Sung, P.L.; Chou, Y.T.; Liew, P.L.; Chen, L.Y.; Huang, R.L.; Lai, H.C.; Chang, L.T. Complete remission of heavily treated ovarian clear cell carcinoma with ARID1A mutations after pembrolizumab and bevacizumab combination therapy: A case report. J. Ovarian. Res. 2020, 13, 143. [Google Scholar] [CrossRef]
- Hamanishi, J.; Mandai, M.; Ikeda, T.; Minami, M.; Kawaguchi, A.; Murayama, T.; Kanai, M.; Mori, Y.; Matsumoto, S.; Chikuma, S.; et al. Safety and Antitumor Activity of Anti-PD-1 Antibody, Nivolumab, in Patients With Platinum-Resistant Ovarian Cancer. J. Clin. Oncol. 2015, 33, 4015–4022. [Google Scholar] [CrossRef]
- Iwai, Y.; Ishida, M.; Tanaka, Y.; Okazaki, T.; Honjo, T.; Minato, N. Involvement of PD-L1 on tumor cells in the escape from host immune system and tumor immunotherapy by PD-L1 blockade. Proc. Natl. Acad. Sci. USA 2002, 99, 12293–12297. [Google Scholar] [CrossRef]
- Zamarin, D.; Burger, R.A.; Sill, M.W.; Powell, D.J., Jr.; Lankes, H.A.; Feldman, M.D.; Zivanovic, O.; Gunderson, C.; Ko, E.; Mathews, C.; et al. Randomized Phase II Trial of Nivolumab Versus Nivolumab and Ipilimumab for Recurrent or Persistent Ovarian Cancer: An NRG Oncology Study. J. Clin. Oncol. 2020, 38, 1814–1823. [Google Scholar] [CrossRef]
- Herbst, R.S.; Soria, J.C.; Kowanetz, M.; Fine, G.D.; Hamid, O.; Gordon, M.S.; Sosman, J.A.; McDermott, D.F.; Powderly, J.D.; Gettinger, S.N.; et al. Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 2014, 515, 563–567. [Google Scholar] [CrossRef]
- Liu, J.F.; Gordon, M.; Veneris, J.; Braiteh, F.; Balmanoukian, A.; Eder, J.P.; Oaknin, A.; Hamilton, E.; Wang, Y.; Sarkar, I.; et al. Safety, clinical activity and biomarker assessments of atezolizumab from a Phase I study in advanced/recurrent ovarian and uterine cancers. Gynecol. Oncol. 2019, 154, 314–322. [Google Scholar] [CrossRef]
- Moroney, J.W.; Powderly, J.; Lieu, C.H.; Bendell, J.C.; Eckhardt, S.G.; Chang, C.W.; Molinero, L.; Spahn, J.; Williams, P.; Lin, Y.G.; et al. Safety and Clinical Activity of Atezolizumab Plus Bevacizumab in Patients with Ovarian Cancer: A Phase Ib Study. Clin. Cancer Res. 2020, 26, 5631–5637. [Google Scholar] [CrossRef]
- Boyerinas, B.; Jochems, C.; Fantini, M.; Heery, C.R.; Gulley, J.L.; Tsang, K.Y.; Schlom, J. Antibody-Dependent Cellular Cytotoxicity Activity of a Novel Anti-PD-L1 Antibody Avelumab (MSB0010718C) on Human Tumor Cells. Cancer Immunol. Res. 2015, 3, 1148–1157. [Google Scholar] [CrossRef]
- Kaufman, H.L.; Russell, J.; Hamid, O.; Bhatia, S.; Terheyden, P.; D’Angelo, S.P.; Shih, K.C.; Lebbe, C.; Linette, G.P.; Milella, M.; et al. Avelumab in patients with chemotherapy-refractory metastatic Merkel cell carcinoma: A multicentre, single-group, open-label, phase 2 trial. Lancet Oncol. 2016, 17, 1374–1385. [Google Scholar] [CrossRef]
- Hamilton, G. Avelumab: Search for combinations of immune checkpoint inhibition with chemotherapy. Expert Opin. Biol. Ther. 2021, 21, 311–322. [Google Scholar] [CrossRef]
- Disis, M.L.; Taylor, M.H.; Kelly, K.; Beck, J.T.; Gordon, M.; Moore, K.M.; Patel, M.R.; Chaves, J.; Park, H.; Mita, A.C.; et al. Efficacy and Safety of Avelumab for Patients With Recurrent or Refractory Ovarian Cancer: Phase 1b Results From the JAVELIN Solid Tumor Trial. JAMA Oncol. 2019, 5, 393–401. [Google Scholar] [CrossRef]
- Pujade-Lauraine, E.; Fujiwara, K.; Ledermann, J.A.; Oza, A.M.; Kristeleit, R.; Ray-Coquard, I.L.; Richardson, G.E.; Sessa, C.; Yonemori, K.; Banerjee, S.; et al. Avelumab alone or in combination with chemotherapy versus chemotherapy alone in platinum-resistant or platinum-refractory ovarian cancer (JAVELIN Ovarian 200): An open-label, three-arm, randomised, phase 3 study. Lancet Oncol. 2021, 22, 1034–1046. [Google Scholar] [CrossRef]
- Monk, B.J.; Colombo, N.; Oza, A.M.; Fujiwara, K.; Birrer, M.J.; Randall, L.; Poddubskaya, E.V.; Scambia, G.; Shparyk, Y.V.; Lim, M.C.; et al. Chemotherapy with or without avelumab followed by avelumab maintenance versus chemotherapy alone in patients with previously untreated epithelial ovarian cancer (JAVELIN Ovarian 100): An open-label, randomised, phase 3 trial. Lancet Oncol. 2021, 22, 1275–1289. [Google Scholar] [CrossRef]
- Song, D.G.; Ye, Q.; Santoro, S.; Fang, C.; Best, A.; Powell, D.J., Jr. Chimeric NKG2D CAR-expressing T cell-mediated attack of human ovarian cancer is enhanced by histone deacetylase inhibition. Hum. Gene Ther. 2013, 24, 295–305. [Google Scholar] [CrossRef]
- Yan, W.; Hu, H.; Tang, B. Advances Of Chimeric Antigen Receptor T Cell Therapy In Ovarian Cancer. Onco. Targets Ther. 2019, 12, 8015–8022. [Google Scholar] [CrossRef]
- Morello, A.; Sadelain, M.; Adusumilli, P.S. Mesothelin-Targeted CARs: Driving T Cells to Solid Tumors. Cancer Discov. 2016, 6, 133–146. [Google Scholar] [CrossRef]
- Schoutrop, E.; El-Serafi, I.; Poiret, T.; Zhao, Y.; Gultekin, O.; He, R.; Moyano-Galceran, L.; Carlson, J.W.; Lehti, K.; Hassan, M.; et al. Mesothelin-Specific CAR T Cells Target Ovarian Cancer. Cancer Res. 2021, 81, 3022–3035. [Google Scholar] [CrossRef] [PubMed]
- Felder, M.; Kapur, A.; Gonzalez-Bosquet, J.; Horibata, S.; Heintz, J.; Albrecht, R.; Fass, L.; Kaur, J.; Hu, K.; Shojaei, H.; et al. MUC16 (CA125): Tumor biomarker to cancer therapy, a work in progress. Mol. Cancer 2014, 13, 129. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Cheng, Z.; Luo, L.; Yang, Y.; Zhang, Z.; Ma, H.; Chen, T.; Huang, X.; Lin, S.Y.; Jin, M.; et al. C-terminus of MUC16 activates Wnt signaling pathway through its interaction with beta-catenin to promote tumorigenesis and metastasis. Oncotarget 2016, 7, 36800–36813. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Wang, J. Therapeutic effect of dual CAR-T targeting PDL1 and MUC16 antigens on ovarian cancer cells in mice. BMC Cancer 2020, 20, 678. [Google Scholar] [CrossRef] [PubMed]
- Coelho, R.; Marcos-Silva, L.; Ricardo, S.; Ponte, F.; Costa, A.; Lopes, J.M.; David, L. Peritoneal dissemination of ovarian cancer: Role of MUC16-mesothelin interaction and implications for treatment. Expert. Rev. Anticancer Ther. 2018, 18, 177–186. [Google Scholar] [CrossRef]
- Coney, L.R.; Tomassetti, A.; Carayannopoulos, L.; Frasca, V.; Kamen, B.A.; Colnaghi, M.I.; Zurawski, V.R., Jr. Cloning of a tumor-associated antigen: MOv18 and MOv19 antibodies recognize a folate-binding protein. Cancer Res. 1991, 51, 6125–6132. [Google Scholar]
- Kandalaft, L.E.; Powell, D.J., Jr.; Coukos, G. A phase I clinical trial of adoptive transfer of folate receptor-alpha redirected autologous T cells for recurrent ovarian cancer. J. Transl. Med. 2012, 10, 157. [Google Scholar] [CrossRef]
- Zuo, S.; Wen, Y.; Panha, H.; Dai, G.; Wang, L.; Ren, X.; Fu, K. Modification of cytokine-induced killer cells with folate receptor alpha (FRalpha)-specific chimeric antigen receptors enhances their antitumor immunity toward FRalpha-positive ovarian cancers. Mol. Immunol. 2017, 85, 293–304. [Google Scholar] [CrossRef]
- Berchuck, A.; Kamel, A.; Whitaker, R.; Kerns, B.; Olt, G.; Kinney, R.; Soper, J.T.; Dodge, R.; Clarke-Pearson, D.L.; Marks, P.; et al. Overexpression of HER-2/neu is associated with poor survival in advanced epithelial ovarian cancer. Cancer Res. 1990, 50, 4087–4091. [Google Scholar]
- Jiang, D.; Im, H.J.; Sun, H.; Valdovinos, H.F.; England, C.G.; Ehlerding, E.B.; Nickles, R.J.; Lee, D.S.; Cho, S.Y.; Huang, P.; et al. Radiolabeled pertuzumab for imaging of human epidermal growth factor receptor 2 expression in ovarian cancer. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1296–1305. [Google Scholar] [CrossRef]
- Song, X.; Sun, K.; Hu, J.; Zhou, J. Suppression of human epidermal growth factor receptor 2 via interference increases the chemosensitivity of ovarian carcinoma. Oncol. Lett. 2016, 11, 3028–3032. [Google Scholar] [CrossRef]
- Hyrenius-Wittsten, A.; Su, Y.; Park, M.; Garcia, J.M.; Alavi, J.; Perry, N.; Montgomery, G.; Liu, B.; Roybal, K.T. SynNotch CAR circuits enhance solid tumor recognition and promote persistent antitumor activity in mouse models. Sci. Transl. Med. 2021, 13, 591. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).