3. Distribution of Edible Crickets
The history of insect consumption as human food has been widely documented. Half Hours with Insects, published in 1877 informed about the consumption of ants in India, bee pupae in China, grasshoppers in Arabia and Africa, bees and ants in Mexico and Sweden, silkworms and larvae of hawkmoths in China and palm weevils in the West Indies [
7,
8]. Nowadays, the global perspective of entomophagy is driven by specific conditions and cultures. Countries with warm and moist tropical climates are more renowned for insect rearing than those in temperate zones [
4]. Populations in the Asia Pacific region adapted to entomophagy as their culture, and have long acknowledged the nutritional and economical values of edible insects [
4,
9,
10]. Western populations are now showing increased awareness toward the positive impacts of edible insects on the environment and human health [
8].
Increasing human interest in entomophagy has led to the development of rearing processes and the export of insect foods, especially crickets [
11]. Countries in Australasia and Asia have applied three different techniques for insect rearing including wild harvesting, semi domestication and farming [
12]. Insect farming is the most applicable method in many developed countries because it is both cost-effective and profitable [
13]. For product marketing, Peru in South America is a renowned producer of cochineal, accounting for 85% of global production [
4]. The Central African Republic exports 8 tons of dried mopane caterpillars to Europe every year [
4], while HaoCheng Mealworm Inc. (Hunan, China) in China exports 200 tons of dried mealworms annually [
4]. In developing countries in Southeast Asia, insect rearing in rural areas is common in Thailand, Cambodia, Vietnam and the Lao People’s Democratic Republic (Lao PDR). In 2013, Thailand, a renowned country for edible insects, had an annual production of 7000 tons of crickets from over 20,000 cricket farmers [
11,
14]. Safety evaluation must be conducted before product marketing in some countries because edible insects are regarded as a novel food under EU regulation 2015/2283 for European countries [
15] and the Niger’s Décret No. 2011-616/PRN/MEL Republic of the Niger in Africa [
16].
The
Coleoptera,
Lepidoptera,
Hymenoptera,
Orthoptera,
Hemiptera,
Isoptera,
Odonate,
Diptera,
Dictyoptera and
Megaloptera are taxonomic orders of global edible insects, with the most renowned as
Coleoptera or the beetles [
4]. When considering nutritional value, cricket species in the
Gryllidae family,
Orthoptera are a high protein-rich source [
17,
18] and popular spotlights of the food industry [
10]. In food product development, species of edible crickets generally comply with rearing standards as short development cycle, low feed conversion ratio, high survival rate, high ability to live and high resistance to disease [
4].
Over 2400 species of crickets have been globally identified, and almost half belong to the
Gryllidae family [
19]. The
Gryllidae contains 62 popular edible species, mostly distributed in Asia (41 species) and Africa (26 species), while 5 species are found in America, 4 in Europe and 4 in Australia [
14].
Brachytrupes portentosus is the largest cricket in the family of
Gryllidae [
20], with availability limited to Africa [
14]. Most reared cricket species for consumption is
Acheta domesticus [
20].
Gryllus bimaculatus is mostly reared in Asia, Africa and Australia, while
A. domesticus is reared on all continents except Antarctica [
14]. Favorable conditions for crickets are warm temperature (29–35 °C) and high relative humidity (50%) [
21].
Figure 2 shows a representative image of
A. domesticus,
G. bimaculatus and cricket powder.
Crickets are farmed commercially worldwide, however, the rearing technology varies and depends on many factors, which may affect their development and nutritional profile as for instance rearing environment [
22] and diets. Regarding lighting, different light intensity and duration were applied to improve production efficiency [
4,
23,
24]. Various use of rearing materials as conventional (egg carton, cotton wool and plastic bucket) and improvised (bamboo, scrap blanket and plywood-based cage) materials has no effect on their growth [
25]. The composition of diet plays crucial role on growth and nutritional quality of crickets [
26]. Moreover, nutritive value of edible insect can be tailored by a supplement of dietary sources [
27,
28]. In Europe, insects are considered as farmed animals, and must be fed using safe diets. Regulations of the European Parliament and the EU Council obligate the use of animal feed and plant materials [
29].
To serve Western populations that are not familiar with entomophagy, cricket farms and industries in Western countries formulate various kinds of cricket-based foods in the form of dried cricket and cricket flour as for instance cricket bars [
11,
30,
31]. Many cricket farms and industries are listed in Europe and America. In North America, the first established farms were Armstrong Cricket Farm in 1945 and Ghann’s Cricket Farm in 1952. The number of cricket farms gradually increased including the Entomo Farm [
11], Big Cricket Farms, All Things Bugs LLC and Griopro [
31]. In the United States, the largest cricket farms in 2013 were Aspire Food Group and All Things Bugs [
11]. In Europe, the first cricket farm was established in the Netherlands as Kreka, followed by Entocube in Finland [
11] and Crickeatz in the United Kingdom [
31]. Europe and North America also have cricket industries including Crickets, Crickets Crackers, Cricket Flours, Cricket Foods, The Cricket Girl, Dimini Cricket and The Gourmet Cricket Co.
In Thailand, production of edible crickets began in 1998 and over 20,000 cricket farmers are now registered in the country [
20]. Initially, cricket rearing only focused on the three native species
G. bimaculatus,
Teleogryllus mitratus and
Teleogryllus occipitalis [
20]. However, recently, farmers have shifted their interest into mass rearing of
G. bimaculatus and
A. domesticus that offer a shorter production cycle and greater returns [
8]. These two species have a development cycle of 45 to 60 days [
32], with advantages of
A. domesticus production outweighing
G. bimaculatus.
G. bimaculatus stands out on economic factors (e.g., market value and sustainable development (SDG 12) [
15]), while
A. domesticus has a more acceptable taste (e.g., nutty and flavor of umami) [
20].
Acheta domesticus also has a better survival rate [
25,
33], with higher micronutrient and protein contents than
G. bimaculatus [
34,
35].
The Food and Agriculture Organization of the United Nations (FAO) reported abundant
A. domesticus reared in Thailand [
4] and exported to Western countries [
32]. Thai farmers conventionally rear crickets in their backyards using different types of pens including concrete cylinders, concrete blocks, plywood boxes and plastic drawers with covers to protect from other animals [
32]. Initially, farmers fed their crickets with commercial insect nourishment containing 14–21% protein to boost nutritional values [
32]. The formulated cricket feed market is constantly growing along with the increasing numbers of cricket farms in Thailand [
11]. Shortly before harvest, the crickets are fed with vegetables, vegetable by-products and fruits to improve taste and nutritional profile [
4,
36]. Nowadays, mass production of cricket flour and cricket snacks is conducted by Thailand Unique and Smile Bull Companies, while Global Bugs is a leader in cricket flour making [
11].
In 2017, the National Bureau of Agricultural Commodity and Food Standards, Ministry of Agriculture and Cooperatives, Thailand launched the Thai agricultural standard on Good Agricultural Practices (GAP) for Cricket Farming TAS 8202-2017 [
37]. This voluntary standard provided guidelines for the safe practice of cricket rearing. The GAP for cricket farming consists of five subsections related to farm components, farm management, animal health, the environment and record keeping. These requirements mitigate the occurrence of potential hazards and adverse effects of cricket rearing on the environment. The GAP also focuses on waste disposal handling. The Food and Agriculture Organization of the United Nations (FAO) in collaboration with an entomologist from Khon Kaen University, Thailand issued the ‘Guidance on sustainable cricket farming’ that details procedures for sustainable cricket rearing and farm inspections.
4. Nutritive Values of Edible Crickets
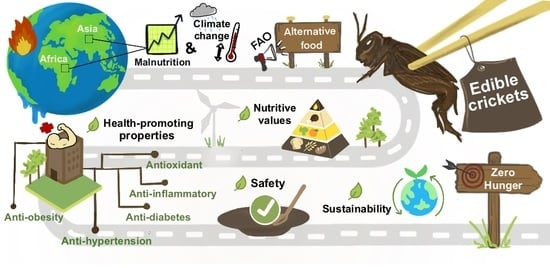
The FAO recommends edible insect consumption, including crickets, because of their high nutritive values, especially protein and fat [
4]. This review summarizes the proximate analysis, protein and fat contents of four edible crickets commonly consumed in Thailand including
A. domesticus,
B. portentosus and the two field crickets
G. bimaculatus and
Gryllus testaceus. Values in
Table 2 demonstrate edible crickets as rich sources of protein, especially
A. domesticus (64.1–71.7% dry matter (DM)), while
B. portentosus showed the lowest protein content. However, more research is required on cricket species with limited data. Besides species, the nutritional profiles of cricket were altered by developmental stage and rearing conditions (e.g., feed and temperature) [
26]. Interestingly, both field cricket species from different global regions (China, Korea, Thailand) showed similar protein contents ranging from 58.2 to 60.7% DM but more variation in fat content (10.30–23.40% DM). Adult
G. bimaculatus grown in Thailand exhibited the highest fat content (23.40% DM).
Gryllus bimaculatus had high protein content compared to egg, chicken, beef, pork, soybean and maize [
38,
39], while protein content was slightly lower than fish [
39]. The fat content of
G. bimaculatus was, as expected, higher than plant food such as soybean, maize and wheat [
39]. An intake of 200 g of
G. bimaculatus was sufficient to satisfy daily fat energy requirements [
40]; however, fat contents of different cricket species varied as shown in
Table 2.
Edible crickets contain high protein contents, and detailed investigations have been conducted on their amino acid profiles [
34,
38,
45]. Amino acids can be divided into two groups as essential and non-essential, based on bodily requirements. Essential amino acids cannot be synthesized by the body and must come from food intake, while non-essential amino acids are synthesized in the body. Amino acids play various vital roles. They are a major component of glutathione, the most abundant low molecular weight antioxidant in cells [
46], and also act as precursors to neurotransmitters and signaling regulators [
47,
48]. Protein deficiency or malnutrition leads to osmotic imbalance, resulting in kwashiorkor.
Table 3 presents the amino acid compositions of the four edible crickets. The high total protein and essential amino acid contents indicated good quality protein sources. Various amino acid compositions across species suggested that species and other factors (feed, sex and method of measurement) contributed to the diversity. Indeed, Kulma et al. (2019) reported that sex could be an important factor contributing to protein and lipid levels because female
A. domestica showed lower amount of protein, but higher in lipid compared to male
A. domestica [
49]. Regarding the protein contents, it has been revealed that crickets have chitin presented on their exocuticle and endocuticle. Chitin is a naturally polysaccharide binding to nitrogen, thus protein in crickets calculated as nitrogen content ×6.25 (nitrogen-to-protein conversion factor, k
p) by Kjeldahl or elemental analysis methods may be overestimated [
35,
50,
51,
52,
53,
54]. According to Boulos et al. (2020), the protein content of whole insects should be calculated using the k
p value of 5.33 rather than 6.25 in order to provide a suitable protein content in insects [
52]. In addition, Ritvanen et al. (2020) proposed a k
p value of 5.00 for
A. domesticus and
G. bimaculatus [
35]. Hence, in order to minimize overestimation of crude protein owing to chitin bound nitrogen, a precise measurement of insect chitin is required, Han and Heinonen (2021) developed an ultra-high performance liquid chromatographic and fluorescent method (UPLC/FLR) to analyze chitin from insects [
54]. After alkaline deproteinization and acid hydrolysis of chitin, the UPLC/FLR method exhibits sensitive and specific quantification of insect chitin [
54].
As well as proteins, fatty acids are crucial macromolecules that can be used as dietary energy, organ insulation and cell membrane components.
Table 4 shows the fatty acid profiles of four edible crickets, with limited data available for
B. portentosus and
G. testaceus. Crickets contain both saturated fatty acids (SFA) and unsaturated fatty acids (UFA). SFAs have been extensively reported for their adverse health effects including increased risk of cardiovascular disease (CVD) [
56], while polyunsaturated fatty acids (PUFA) contribute health benefits by reducing plasma cholesterol [
57] and increasing high-density lipoprotein (HDL) [
58], which is associated with lower risk of CVD. Palmitic acid and stearic acid were the predominant SFAs in crickets, particularly in
A. domesticus and
G. testaceus, while PUFA as oleic acid and linoleic acid were mainly presented, especially in
G. testaceus. Fatty acid profiles revealed that
G. bimaculatus had lower SFA compared to pork, beef and egg. Conversely, PUFA in
G. bimaculatus were higher than in pork, beef and egg [
38]. Furthermore,
G. testaceus showed the highest PUFA:SFA ratio (3.480) followed by
B. portentosus (1.554). Increasing consumption of fat with high PUFA:SFA ratio has been suggested for CVD prevention [
59]. The PUFA:SFA ratio in various species animals have been reported as follows, fish (0.50–1.79), chicken (0.93–0.94), pig (0.46–0.48), cattle (0.11–0.20) and lamp (0.13–0.37) [
60], implying the high quality of fat components of
G. testaceus and
B. portentosus. However, more information is still needed to conclude since several fatty acids are missing. Similar to amino acid profiles, the varied fatty acid compositions implied that species and other factors such as feed, sex and method of measurement contributed to the diversity [
61].
6. Safety Aspects of Crickets
To promote cricket consumption, knowledge of nutritive values, health benefits and safety procedures are paramount.
Acheta domesticus reared in a closed farming system have been considered safe by the EFSA panel [
102] and are used as a food ingredient in several food products such as crackers, breadsticks, meat imitates and snacks.
Acheta domesticus showed no cytotoxicity up to 250 µg/mL in the three human cell types HL60, HeLa and Caco-2) [
102]. Even though no report on toxicity and genotoxicity of
A. domesticus is reported [
102], EFSA panel raised concerns on biological and chemical hazards [
103].
The toxicity and genotoxicity of
G. bimaculatus have been intensively investigated. Oral toxicity testing using a repeated dose of the ethanolic extract of
G. bimaculatus in rats for 28 days was considered safe, with no-observed-adverse-effect level (NOAEL) at 1000 mg/kg/day according to the Organization for Economic Cooperation and Development (OECD) test guidelines No. 407 [
104]. The safety of
G. bimaculatus was also confirmed by conducting an oral toxicity test using a repeated dose of
G. bimaculatus powder in rats for 90 days. Results showed a NOAEL value at 5000 mg/kg/day with no histopathological changes [
105]. From these data, the acceptable daily intake (ADI) of
G. bimaculatus powder should be limited to 3000 mg/day, suggesting that
G. bimaculatus is considered safe for human consumption. As mentioned earlier, glycosaminoglycan isolated from field crickets possesses health benefits, and was also subjected to toxicity testing using a repeated dose for 28 days to determine oral toxicity [
106]. Results showed that the NOAEL value for glycosaminoglycan isolated from field crickets was 160 mg/kg/day, which was the highest dose used in the experiment [
106]. Beside sub-chronic or chronic toxicity testing, genotoxicity testing must also be employed and used as an indicator to detect mutagens and carcinogens. Genotoxicity testing determined by the Ames test in
Salmonella typhimurium, the chromosome aberration test in Chinese hamster ovary (CHO) cells and the micronucleus test in young male mice revealed that phosphate-buffered saline (PBS) extracted
G. bimaculatus showed no induction DNA damages in vitro and in vivo [
107]. Information on genome safety of glycosaminoglycan isolated from field crickets has also been reported [
106]. Thus, all the evidence indicated the safe consumption of
A. domesticus and
G. bimaculatus. However, safety information for
B. portentosus and
G. testaceus is still lacking.
Type I hypersensitivity (IgE-mediated hypersensitivity) including food allergy is associated with allergen-mediated histamine released from mast cells, leading to a red rash, face swelling and in severe cases, anaphylactic shock (
Figure 7) [
108]. Many food allergies cases are sensitive to arthropods, such as shrimp and crab [
109]. Edible insects, including crickets are classified as arthropods; thereby, possessing potential allergenicity. Some cases were reported for insect allergenicity in China and Thailand [
110,
111], together with cross-reactivity between
A. domesticus, shrimp and house dust mite [
112]. The IgE from patients allergic to shrimp or house dust mite can recognize
A. domesticus protein, confirming the potential allergenicity of crickets. The cross-reactivity of
G. bimaculatus and shrimp has also been demonstrated [
113]. Hence, crickets appear to be devoid of toxicity but their allergenicity should be considered, particularly for individuals allergic to shrimp and mite.
Crickets have also been tested for potential biological, chemical and physical hazards. Possible human hazards for both raw and processed crickets are summarized below to better control the quality and safety of cricket-based foods.
Raw crickets: Crickets, as a nutrient-rich source, are vulnerable to biological hazards including microbial loads [
114], pathogenic bacteria (
Salmonella sp. and
Shigella sp.) [
115], spore-forming bacteria (
Bacillus, Paenibacillus and
Psychrobacillus) [
116], high yeast and mold counts (
Debaryomycess) [
20], fungi (
Aspergillus flavus,
Aspergillus sydowii and
Cladosporium sphaerospermum) [
117] and parasites (
Balantidium sp. and
Cestoda sp.) [
118]. Crickets are also susceptible to virus infection such as cricket densovirus (AdDV) and cricket paralysis virus (CrPV); however, these viruses are unlikely threats to human health [
103]. Biological hazards in crickets such as viruses, fungi and parasites are categorized as low risk for human health because crickets have the ability to self-detoxify mycotoxin from fungi during the rearing process. These microorganisms can also be reduced through appropriate product processing using heat treatment [
117,
119,
120,
121]. However, spore-forming bacteria are categorized as medium risk since they can re-occur after the processing stage [
103].
Processed crickets: Biological hazards, especially pathogenic microorganisms in processed crickets rarely occur, except for fungi that can pose a risk during the manufacturing process [
103]. Unfortunately, there are no standardized harvest and processing protocols in the US for cricket powder production. High variation of microbial loads in cricket powder occur for aerobic mesophilic counts and fungi as well as for some pathogens, including
Staphylococcus and
Bacillus. For example, aerobic mesophilic counts varied from 1.0 to 6.0 log colony forming unit (CFU)/g [
122]. Another study on processed crickets focused on the bacterial count of dried, deep-fried and extruded products. Findings highlighted a higher possibility of bacterial reoccurrence in dried crickets than in deep-fried and extruded crickets [
123].
Bacillus sp., a spore-forming pathogen, is frequently observed in cricket-based foods. The bacilli are strongly related to human diseases including diarrhea and emesis when the value is >6.0 log CFU/g [
124]. Some previous studies reported a positive result for
Bacillus sp. in processed crickets. A study on commercial cricket flour highlighted the presumptive sample load of
Bacillus cereus, with
Bacillus count between 1.0 and 3.5 log CFU/g [
122]. High prevalence of
Bacillus cereus in smoked and dried crickets and extruded cricket products has also been reported [
45,
125].
Thermal treatments such as blanching, boiling, pre-frying, drying and extrusion methods are all used in cricket production. Klunder et al. (2016) reported that both boiling and stir-frying effectively decreased Enterobacteriaceae and bacterial spores [
121]. Gatheru et al. (2019) highlighted the effectiveness of freeze-drying and deep-frying methods to eliminate microbes and fungi, while boiling and sun-drying methods only decreased microbial and fungal counts [
126]. Vandeweyer et al. (2018) successfully reduced the numbers of microorganisms and spores using various thermal treatments such as freezing, boiling, oven-drying and smoke-drying. They highlighted freezing and boiling as the most effective treatments [
125]. Grabowski et al. (2017) varied the cooking time, drying temperature and drying time. They reported a decrease in microbial counts after sequential drying at 80 and 100 °C for 12 h after 30 min of cooking [
124].
Microbial counts after 6 h storage at ambient temperature for dried cricket products and frozen cricket products at −25 °C remained the same [
125]. By contrast, boiled cricket products at ambient temperature were more susceptible to biological hazards than at chilling temperature (5 to 7 °C) over six months of storage [
121]. Results showed a correlation between heat treatment and storage conditions to maintain the safety of edible cricket products. Post-contamination was also related to packaging, labeling and storing processes. Thus, appropriate conditions including the packaging materials, labeling procedures and storage conditions should be monitored to avoid recontamination of fungi and pathogenic bacteria such as
Aspergillus sp.,
Penicillium,
Fusarium,
Listeria monocytogenes and
Salmonella [
120].
Environmental contamination from heavy metals, flame retardants, dioxins and pesticides can also be potential chemical hazards in edible insects. The ‘Guidance on sustainable cricket farming’ highlights the risk of chemical contaminants during the rearing processes. Chemical hazards pertaining to raw and processed crickets include heavy metals (cadmium, arsenic, chromium, lead and tin) [
103], allergens (tropomyosin, arginine kinase and the specific allergen in
G. bimaculatus, hexamerin B1) [
103,
127,
128], polybrominated diphenyl ether (PBDE) flame retardants [
129], dioxins (polychlorinated dibenzodioxins and dioxin-like polychlorinated biphenyls) [
15] and high pesticide residues (carbamate and organophosphate) [
130]. Whole crickets, cricket powder and cricket protein hydrolysates contain tropomyosin; thus, individuals with prawn allergy may experience an allergic reaction after consuming crickets [
128]. Cassi et al. (2018) stated that the chemical hazards in crickets, especially allergens and heavy metals, were medium risk because allergen modulation relies on the processing stage, while cadmium also showed high chemical activity [
103].
Past decades have seen increasing numbers of reports on microplastic contamination in the soil ecosystem as the living environment of crickets [
131,
132,
133]. Thus, microplastic contamination could be considered a potential physical hazard for crickets. Chemical hazards found in crickets included PBDE flame retardant, which is generally used in consumer plastic products. Gaylor et al. (2011) reported that the accumulation of PBDE in crickets might be sourced from furniture and the automotive sector, where these plastic-based materials commonly accumulate in the terrestrial area [
129]. However, the only study on microplastic analysis of natural living insects was conducted in freshwater insects [
134].
The occurrence of biological and chemical hazards in crickets, excluding allergens, is mostly caused by unhygienic rearing and production processes [
15,
103,
120]. Therefore, preventive measures should be applied to maintain the safety and quality of cricket products [
15] such as Good Farming Practice (GFP), Good Manufacturing Practice (GMP) and Hazard Analysis Critical Control Points (HACCP). Hazard preventive control should include the use of hygiene equipment and safe nourishment for feeding, application of personal hygiene practice, good waste management and good hygiene throughout the food supply chain [
120].
Global interest in entomophagy has recently increased. National and regional bodies concerned with food safety in some countries have announced standards for farming and marketing edible crickets. The relevant authorities in some countries now guarantee the quality of edible crickets such as the European Commission (EC) and European Food Safety Authority (EFSA), the Kenya Bureau of Standards (KEBS), the United States Food and Drug Administration (USFDA), the Canadian Food Inspection Agency (CFIA), Food Standards Australia New Zealand (FSANZ) and the Committee on Novel Foods (ACNF) in Australia and New Zealand, the Guangxi Zhuang Autonomous Region in China and the Thai Food and Drug Administration (Thai FDA) and Agricultural Commodity and Food Standards (ACFS) in Thailand [
15].
8. Future Perspectives
Globally, insects are processed using various methods before consumption. In Asia, Africa and Latin America entomophagy has been practiced since ancient times and people are familiar with consuming the whole edible insects [
161]. Product processing methods include boiling and drying, toasting, frying, steaming, roasting, smoking, stewing and curing [
115,
161]. In Western countries, North America and Europe, people are not familiar with eating whole insects [
161]. Processing methods include pulverizing and grinding to deform the insects into granulated and paste forms [
162]. The strategy for promoting entomophagy in Western countries is through granulated and paste forms of insects that are more suitable to produce snack products [
161,
163]. The EFSA announced the additional step of removing the legs and wings, especially on crickets before consumption; however, this is still debated due to the adverse effect on nutritional properties and lack of scientific evidence [
148].
Apart from conventional methods, advanced technology is being developed by scientists to diversify the market variety of insect products as pastes, sauces, powders, flour, snacks and pasta [
11,
161,
164]. In China, the population consumes fried insects [
161]. Similarly, in Africa, insects are commonly processed as smoked, toasted, roasted and fried before consumption [
161,
165]. Nowadays, a wide range of cricket food applications has become a new food industry as extruded food products (cricket biscuit, cricket protein energy bars, cricket chips, cricket crackers and cricket pasta), diet supplements and food preservatives.
Consumer surveys have progressed in parallel with insect production. A study on insect consumption by children from 6 to 11 years old reported that children are not averse to trying insect products [
30]. To increase customer interest in entomophagy, knowledge of the nutritional value of insects should be emphasized [
142,
166] and instilled from childhood. An analysis of consumer perception on edible insects also reported a positive response by most participants with ages ranging from 18 to over 55 years old, and 77% were willing to try entomophagy [
159], while 22% preferred to consume insect-based snacks [
159]. Thus, the food industry needs to diversify edible insect products or to incorporate the insects with native ingredients. These could be an effective introduction to increase consumer adoption to edible insect foods. For example, a study on cricket-based biscuits confirmed a high sensory result and revealed the just-about-right color, tasty flavor, pleasant odor and soft texture to suggest market demand [
163].
Crickets are a high source of protein and can be used to treat protein-energy malnutrition diseases such as marasmus and kwashiorkor [
167]. Moreover, the derived product of crickets as glycosaminoglycan has anti-oxidative and anti-inflammatory properties to treat diabetes [
88] and arthritis [
83], respectively. A mixture of wheat flour and cricket protein was shown to increase iron solubility and the availability of minerals more than the use of conventional livestock protein (chicken, pig and cattle) [
168]. Thus, the addition of crickets into a starch-based diet can positively enhance micronutrients [
168]. In addition, hydrolysis of cricket protein can produce novel peptides with bioavailability, for instance antihypertension and antidiabetic or antioxidation which can be used for food and pharmaceutical applications [
169,
170,
171].
The application of cricket lipids is limited to the application in food, as they contain high unsaturated fatty acids [
43,
44,
172]. The cricket lipids can also be potentially used as an alternative source of lecithin for food and pharmaceutical uses since their phospholipid patterns are similar to those of lecithin [
172]. In addition to its use as a promising food ingredient, the functionality (i.e., antioxidation) of cricket lipids also attracts interest in scientific point of view [
69].
Apart from consumption, insect extract can also be used as an effective food preservative. Chitosan, produced from the deacetylation of chitin in insects is a promising polysaccharide that enhances food safety, shelf life and quality control of food products through its antioxidant and antibacterial effects [
173]. Comprehensive research has successfully developed chitosan as a bio-based polymer for food packaging [
14]. Chitosan prolongs food shelf life. In the food industry, various methods of food preservation using chitosan are direct coating (dipping, brushing and spraying methods), layer-by-layer assembly and bioactive film packaging [
174].
While the future looks bright for edible cricket market, manufacturing process development is still a major challenge in the production and industrialization of insect-based products. Emphasis should also be placed on research highlighting the implementation of technology and process innovation for cricket farming and processing. The process of technology and scientific knowledge transfer will help to facilitate commercial exploitation and the social impact of the use of edible cricket.