1. Introduction
Marine invertebrates, particularly octocorals have been well recognized as a rich source of interesting steroid metabolites [
1]. In continuation of research into new substances from marine invertebrates collected off the waters of Taiwan, a series of steroid derivatives have been isolated from the octocorals belonging to the genus
Dendronephthya (phylum Cnidaria, class Anthozoa, order Alcyonacea, family Nephtheidae), octocorals distributed in the tropical and subtropical waters of the Indo-Pacific Ocean, and some of these metabolites were found to possess interesting bioactivities, such as cytotoxic [
2] and anti-inflammatory activity [
3,
4]. Recently, chemical examination of an octocoral identified as
Dendronephthya sp. resulted in the isolation of two new marine steroids, dendronesterones D (
1) and E (
2) (
Figure 1), along with three known steroids, including an antifouling compound, methyl 3-oxochola-4,22-dien-24-oate (
3), which was first isolated from a Japanese soft coral
Dendronephthya sp. [
5], and two cytotoxic metabolites, 5α,8α-epidioxy-24(
S)-methylcholesta-6,22-dien-3β-ol (
4) and 5α,8α-epidioxy-24(
S)-methylcholesta- 6,9(11),22-trien-3β-ol (
5) [
6] (
Figure 1), which were obtained from various marine invertebrates, such as sea squirts
Trididemnum inarmatum [
6] and
Ascidia nigra [
7], a hard coral
Dendrogyra cylindrus [
7], and a sponge
Thalysias juniperina [
7]. We reported herein the isolation and structural determination of steroids
1–
5. The ability of
1–
5 to reduce the expression of the pro-inflammatory iNOS (inducible nitric oxide synthase) and COX-2 (cyclooxygenase-2) proteins in LPS (lipopolysaccharides)-stimulated RAW264.7 macrophage cells was determined.
2. Results
The new metabolite dendronesterone D (
1) was isolated as a colorless oil, and its molecular formula was established as C
27H
36O
5 (unsaturation degrees = 10) from a sodium adduct at
m/z 463 in the (+)-ESIMS and further supported by the (+)-HRESIMS at
m/z 463.24530 (calculated for C
27H
36O
5 + Na, 463.24550). The
13C and DEPT spectroscopic data showed that this compound has 27 carbons (
Table 1), including five methyls, five sp
3 methylenes, six sp
3 methines, two sp
3 quaternary carbons, five sp
2 methines, an sp
2 quaternary carbon, two ester carbonyls, and a ketonic carbonyl. The IR spectrum revealed the presence of ester carbonyl (1724 cm
−1) and α,β-unsaturated ketonic (1663 cm
−1) groups. The
1H NMR spectra (
Table 1) showed the presence of five olefinic methine protons (δ
H 6.78, d,
J = 10.8 Hz; 6.74, dd,
J = 15.6, 10.0 Hz; 6.13, dd,
J = 10.8, 2.0 Hz; 6.10, dd,
J = 2.0, 1.6 Hz; 5.79, d,
J = 15.6 Hz) and an oxymethine proton (δ
H 5.17, ddd,
J = 10.8, 10.8, 5.6 Hz). In addition, a carbonyl resonance at δ
C 169.7 further confirmed the existence of an ester group. The result of
1H NMR spectrum analysis indicated an acetate methyl (δ
H 2.01, 3H, s). The carbon signals at δ
C 156.2 (CH), 125.7 (CH), 186.2 (C), 124.6 (CH), and 167.1 (C) as well as the proton at δ
H 6.78 (1H, d,
J = 10.8 Hz), 6.13 (1H, dd,
J = 10.8, 2.0 Hz), and 6.10 (1H, dd,
J = 2.0, 1.6 Hz) were characteristic signals of steroids with a 1,4-dien-3-one moiety in ring A [
8,
9,
10,
11,
12].
1H NMR coupling information in the COSY spectrum of
1 enabled identification of H-1/H-2, H-2/H-4 (by a long range
W-coupling), H
2-6/H
2-7/H-8/H-9/H-11/H
2-12, H-8/H-14/H
2-15/H
2-16/H-17/ H-20/H-22/H-23, and H-20/H
3-21 (
Figure 2). These data, together with the key heteronuclear multiple bond correlation (HMBC) between protons and quaternary carbons, such as H-1/C-3; H-1, H
2-6, H
3-19/C-5; H-1, H-4, H-9, H-11, H
3-19/C-10; H
2-12, H
3-18/C-13; and H-22, H-23/C-24, allowed us to establish the molecular skeleton of
1. H-11 (δ
H 5.17) showed HMBC to C-10 and acetate carbonyl carbon at δ
C 169.7, demonstrating the acetoxy group at C-11. The methoxy group at C-24 was confirmed by the HMBC between the methyl protons of methoxy group (δ
H 3.72) and C-24 (δ
C 166.8).
The relative configuration of
1 was elucidated by the NOE correlations observed in a NOESY experiment. H-8 showed correlations with both H
3-18 and H
3-19, and H
3-18 exhibited correlations with H-11 and H-20; therefore, due to the β-orientation of Me-18 at C-13, all of H-8, H-11, H
3-19, and H-20 should be positioned on the β-face. Furthermore, NOE responses between H-14 and H-9, and H-14 and H-17, were observed on the α-orientation of H-9, H-14, and H-17 (
Figure 3) (
Supplementary Materials, Figures S1–S10).
Steroid
2 (dendronesterone E) was isolated a colorless oil and was found to possess a molecular formula C
25H
34O
4, as determined by (+)-HRESIMS at
m/z 421.23502 (calculated for C
25H
34O
4 + Na, 421.23493). IR absorptions at 3395, 1720, and 1657 cm
–1 revealed the presence of hydroxy, ester, and α,β-unsaturated ketonic groups. Comparison of the
1H and
13C NMR data of
2 with those of
1 (
Table 1) suggested that
2 is the 11-
O-deacetyl analogue of
1. This was further confirmed by the upfield shifts observed for H-11 (δ
H 3.99) and C-11 (δ
C 67.9) relative to those of
1 (δ
H 5.17; δ
C 69.8). The planar structure of
2, including the positions of hydroxy group, carboxylate, and the olefinic double bonds, could be deduced from analysis of 2D NMR spectrum, including COSY and HMBC (
Figure 2). The relative stereochemistry of
2 was established by the analysis of the NOE correlations in NOESY spectrum of
2, as illustrated in
Figure 4 (
Supplementary Materials, Figures S11–S20).
The known steroids
3–
5 were identified as methyl 3-oxochola-4,22-dien-24-oate [
5], 5α,8α- epidioxy-24(
S)-methylcholesta-6,22-dien-3β-ol [
6,
7], and 5α,8α-epidioxy-24(
S)-methylcholesta-6,9 (11),22-trien-3β-ol [
6,
7], respectively, according to a comparison of their spectroscopic data analysis with the information described in the literature.
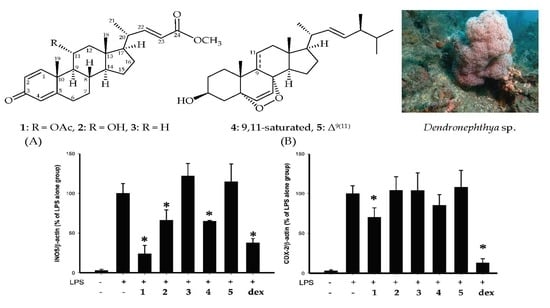
Using an in vitro pro-inflammatory suppression assay, the effects of steroids
1–
5 on the release of iNOS and COX-2 protein from LPS-stimulated RAW264.7 macrophage cells were assessed. The results of the in vitro pro-inflammatory suppression assay showed that steroid
1 at 10 μM suppressed the expression of iNOS/β-actin and COX-2/β-actin to 24.2 ± 10.6 and 70.4 ± 11.9%, as compared with LPS alone group (
Figure 5). Compounds
1–
5 did not significantly affect the viability of macrophage cells 16 h after treatments.
3. Discussion
Dendronephthya spp. have been demonstrated to have a wide structural diversity of interesting steroids that possess various pharmacological properties, specifically in anti-inflammatory activities [
13,
14]. In our study of
Dendronephthya sp., two previously unreported steroids, dendronesterones D (
1) and E (
2), were isolated together with the previously described marine steroids, methyl 3-oxochola-4,22-dien-24-oate (
3), 5α,8α-epidioxy-24(
S)-methylcholesta-6,22-dien-3β-ol (
4), and 5α, 8α-epidioxy-24(
S)-methylcholesta-6,9(11),22-trien-3β-ol (
5). In the present study, the structures of new metabolites
1 and
2 were elucidated by spectroscopic methods and anti-inflammatory activities of steroids
1–
5 were assessed using inhibition of pro-inflammatory iNOS and COX-2 release from macrophages. The results indicated that dendronesterone D (
1) showed the most potent suppressive effects on iNOS release and steroids
2 and
3 showed more weak suppressive effects on iNOS/β-actin and COX-2/β-actin expression than those of
1. The results suggested that the anti- inflammatory activities of steroids
1–
3 were mainly reliant on the functional group at C-11. Furthermore, steroid
5 was found to be inactive in terms of reducing the expression of iNOS/β-actin, indicating that the anti-inflammatory activities of steroids
4 and
5 are dependent on the existence of the carbon–carbon double bond between C-9/11.
4. Experimental Section
4.1. General Experimental Procedures
Optical rotations were measured on a Jasco P-1010 digital polarimeter (Japan Spectroscopic Corporation, Tokyo, Japan); infrared spectra were recorded on a Thermo, Nicolet iS5 FT-IR (Thermo Scientific Nicolet, Waltham, MA, USA); peaks are reported in cm–1. The NMR spectra were recorded on a Jeol FT-NMR (model ECZ400S, Tokyo, Japan) spectrometer operating at 400 MHz for 1H and 100 MHz for 13C, using the residual CHCl3 signal (δH 7.26 ppm) as an internal standard for 1H NMR and CDCl3 (δC 77.1 ppm) for 13C NMR; coupling constants (J) are given in Hz. ESIMS and HRESIMS were recorded using a Bruker 7 Tesla solariX FTMS system (Bremen, Germany). Column chromatography was performed on silica gel (230–400 mesh, Merck). TLC was carried out on precoated Kieselgel 60 F254 (0.25 mm, Merck); spots were visualized by spraying with 10% H2SO4 solution followed by heating. Normal-phase HPLC (NP-HPLC) was performed using a system comprised of a Hitachi 5110 pump (Hitachi, Tokyo, Japan) and a Rheodyne 7725 injection port (Rheodyne, Rohnert Park, CA, USA). A normal-phase column (Luna, 5 μm, Silica (2) 100Å, 250 × 10 mm) was used for NP-HPLC. Reversed-phase HPLC (RP-HPLC) was performed using a system comprised of a Hitachi L-2130 pump, a Hitachi L-2455 photodiode array detector, and a Rheodyne 7725 injection port. A reverse phase column (Luna, 5 μm C18(2) 100Å, 250 × 21.2 mm) was used for RP-HPLC.
4.2. Animal Material
Specimens of the octocoral
Dendronephthya sp. were collected by hand using self-contained underwater breathing apparatus (SCUBA) diving off the northeast coast of Taiwan in August 30th, 2018, and stored in a –20 °C freezer until extraction. A voucher specimen (NMMBA-TW-SC-2018-018) was deposited in the National Museum of Marine Biology and Aquarium (NMMBA), Taiwan. This organism was identified by comparison with previous descriptions [
15].
4.3. Extraction and Separation
Sliced bodies of Dendronephthya sp. (wet weight 748.7 g; dry weight 186.8 g) were extracted with a 1:1 mixture of methanol (MeOH) and dichloromethane to give 12.2 g of crude extract which was partitioned between ethyl acetate (EtOAc) and H2O. The EtOAc extract (2.4 g) was applied on silica gel column chromatography and eluted with gradients of n-hexane/EtOAc (100:1—pure EtOAc, stepwise), to furnish 14 fractions (fractions: A–N). Fractions I, L, and M were purified by NP-HPLC using a mixture of n-hexane/acetone, 4:1 for fractions I and M, and 6:1 for fraction L, to afford 3 (8.5 mg), 1 (6.6 mg), and 2 (3.0 mg), respectively. Fraction J was purified by NP-HPLC using a mixture of n-hexane/acetone (5:1) to yield nine fractions J1–J9. Fraction J6 was separated by RP-HPLC using a mixture of MeOH/H2O (95:5) to afford 5 (0.6 mg) and 4 (1.5 mg), respectively.
Dendronesterone D (
1): Colorless oil:
+79 (
c 0.3, CHCl
3); IR (ATR) ν
max 1724, 1663 cm
−1;
1H (400 MHz, CDCl
3) and
13C (100 MHz, CDCl
3) NMR data, see
Table 1; ESIMS
m/z 463 [M + Na]
+; HRESIMS
m/z 463.24530 (calculated for C
27H
36O
5 + Na, 463.24550).
Dendronesterone E (
2): Colorless oil:
+57 (
c 0.08, CHCl
3); IR (ATR) ν
max 3395, 1720, 1657 cm
−1;
1H (400 MHz, CDCl
3) and
13C (100 MHz, CDCl
3) NMR data, see
Table 1; ESIMS
m/z 421 [M + Na]
+; HRESIMS
m/z 421.23502 (calculated for C
25H
34O
4 + Na, 421.23493).
Methyl 3-oxochola-4,22-dien-24-oate (
3): Colorless oil:
+52 (
c 0.2, CHCl
3) (ref. [
5],
+53.6 (
c 0.28, CHCl
3)); IR (ATR) ν
max 1721, 1662 cm
−1;
1H (400 MHz, CDCl
3) and
13C (100 MHz, CDCl
3) NMR data were found to be in full agreement with those reported previously [
5]; ESIMS
m/
z 405 [M + Na]
+.
5α,8α-Epidioxy-24(
S)-methylcholesta-6,22-dien-3β-ol (
4): Amorphous powder:
−6 (
c 0.07, CHCl
3); IR (ATR) ν
max 3375 cm
−1;
1H (400 MHz, CDCl
3) and
13C (100 MHz, CDCl
3) NMR data were found to be in full agreement with those reported previously [
7]; ESIMS
m/
z 451 [M + Na]
+.
5α,8α-Epidioxy-24(
S)-methylcholesta-6,9(11)22-trien-3β-ol (
5): Amorphous powder:
+214 (
c 0.2, CHCl
3); IR (ATR) ν
max 3391 cm
−1;
1H (400 MHz, CDCl
3) and
13C (100 MHz, CDCl
3) NMR data were found to be in full agreement with those reported previously [
7]; ESIMS
m/
z 449 [M + Na]
+.
4.4. In Vitro Anti-Inflammatory Assay
The anti-inflammatory activity method used was modified from our previous studies [
16,
17,
18]. We examined the effects of steroids
1–
5 on pro-inflammatory iNOS and COX-2 protein expressions in LPS-stimulated RAW264.7 cells by Western blotting analysis. RAW264.7 were obtained from the American Type Culture Collection (ATCC TIB-71, Mannassas, VA, USA). The cells was seeded in 10-cm dishes at a density of 1 × 10
6 cells. The inflammatory response was induced by incubation of LPS (0.01 μg/mL) for 16h. For the anti-inflammatory activity assay, steroids
1–
5 and dexamethasone (as positive control) at 10 μM were added to the cells 10 min before LPS challenge. After 16 h, the cells were then washed with ice-cold phosphate-buffered saline, lysed in lysis buffer (50 mM Tris, pH 7.5, 150 mM NaCl, 1% Triton X-100, 100 μg/mL phenylmethylsulfonyl fluoride and 1 μg/mL aprotinin), and centrifuged at 20,000 g for 30 min at 4 °C. The supernatants were reserved for western blotting. Protein concentrations were measured by the DC protein assay kit (Bio-Rad, Hercules, CA, USA). An equal volume of sample buffer (2% 2-mercaptoethanol, 2% sodium dodecyl suflate (SDS), 0.1% bromophenol blue, 10% glycerol, and 50 mM Tris-HCl (pH 7.2)) was added to the samples, and the protein lysates (50 μg) loaded onto tricine SDS-polyacrylamide (7% or 10%) gel. After electrophoresis, proteins were transferred to polyvinylidene difluoride (PVDF) membranes (Immobilon-P; pore size, 0.45 μM; Millipore, Bedford, MA, USA) at 135 mA overnight at 4 °C in transfer buffer (50 mM Tris-HCl, 380 mM glycine, 1% SDS, 20% methanol). The PVDF was incubated overnight at 4 °C with the anti-iNOS, anti-COX-2, or anti-β-actin antibodies. A horseradish peroxidase-conjugated secondary antibody was used for detection. Anti-iNOS (catalog no. 160862) and anti-COX-2 (catalog no. 160106) antibodies were purchased from Cayman Chemical Company (Ann Arbor, MI, USA). The β-actin antibody (catalog no. Actin sigma A5441) was purchased from Sigma-Aldrich (St. Louis, MO, USA). Immunoreactive bands were visualized by enhanced chemiluminescence (ECL kit; Millipore) and the BioChemi Imaging System and relative densitometric quantification was performed using LabWorks v6.2 (UVP, Upland, CA, USA). Bands for iNOS, COX-2, and β-actin antibodies were recognized at ~135, ~72, and ~45 kDa, respectively. The experiment was repeated 3−4 times and data presented as the mean ± standard error of the mean (SEM). For statistical analysis of immunoblot, the integrated optical density of the LPS group was set to 100%, and β-actin was used to verify that equivalent amounts of protein were loaded in each lane. The data was analyzed by analysis of variance (ANOVA) with the Student–Newman– Keuls post hoc test for multiple comparisons. The difference was significant when
p was less than 0.05.
4.5. Cell Viability
The RAW264.7 macrophage cell viability was determined after treatment with alamar blue (invitrogen, Carlsbad, CA, USA) [
18], a tetrazolium dye that is reduced by living cells to fluorescent products. This assay is similar in principle to the cell viability assay using 3-(4,5-dimethyldiazol-2- yl)-2,5-diphenyltetrazolium bromide and has been validated as an accurate measure of the survival of RAW264.7 macrophage cells.

 ) correlations and selective HMBC (
) correlations and selective HMBC (  ) of steroids 1 and 2.
) of steroids 1 and 2.
 ) of 1.
) of 1.
 ) of 2.
) of 2.