Abstract
Fucoxanthin, a carotenoid with remarkable antioxidant properties, has considerable potential for high-value biotechnological applications in the pharmaceutical, nutraceutical, and cosmeceutical fields. However, conventional extraction methods of this molecule from microalgae are limited in terms of cost-effectiveness. This study focused on optimizing biomass and fucoxanthin production from Isochrysis galbana, isolated from the coast of Tadjoura (Djibouti), by testing various culture media. The antioxidant potential of the cultures was evaluated based on the concentrations of fucoxanthin, carotenoids, and total phenols. Different nutrient formulations were tested to determine the optimal combination for a maximum biomass yield. Using the statistical methodology of principal component analysis, Walne and Guillard F/2 media were identified as the most promising, reaching a maximum fucoxanthin yield of 7.8 mg/g. Multiple regression models showed a strong correlation between antioxidant activity and the concentration of fucoxanthin produced. A thorough study of the optimization of I. galbana growth conditions, using a design of experiments, revealed that air flow rate and CO2 flow rate were the most influential factors on fucoxanthin production, reaching a value of 13.4 mg/g. Finally, to validate the antioxidant potential of fucoxanthin, an in silico analysis based on molecular docking was performed, showing that fucoxanthin interacts with antioxidant proteins (3FS1, 3L2C, and 8BBK). This research not only confirmed the positive results of I. galbana cultivation in terms of antioxidant activity, but also provided essential information for the optimization of fucoxanthin production, opening up promising prospects for industrial applications and future research.
1. Introduction
Microalgae are emerging as a game-changer in the realm of natural ingredients [1]. These microscopic organisms hold immense potential for various industrial applications, particularly in the food and pharmaceutical sectors [2,3,4]. Their remarkable biodiversity translates into producing a diverse range of valuable intracellular metabolites. These include proteins, carbohydrates, lipids, and most importantly, carotenoids—a class of compounds renowned for their potent antioxidant properties that significantly contribute to human health [5,6].
Carotenoids are a diverse group of pigments found in all photosynthetic organisms and some non-photosynthetic ones, including microalgae [7]. These vibrant molecules, responsible for the yellow, orange, and red hues in fruits, flowers, and even our skin, are built on a C40 isoprenoid backbone. Depending on the specific structure, this backbone can be acyclic or cyclic, with various modifications at its ends [8,9,10]. Common and well-studied carotenoids include lycopene, β-carotene, α-carotene, lutein, zeaxanthin, and β-cryptoxanthin [11,12]. These compounds are not just visually striking; they play an essential role in human health by acting as precursors to vitamin A and providing essential antioxidant protection against cellular damage [13,14]. In recent years, fucoxanthin has garnered significant interest due to its therapeutic potential and ability to address nutritional deficiencies. However, traditional methods for extracting fucoxanthin from brown algae (macroalgae) face limitations. These methods often involve complex and lengthy growth cycles, making large-scale production impractical and driving up costs [15,16].
Microalgae, on the other hand, offer a promising solution for the commercial production of fucoxanthin. These diverse photoautotrophs have the advantage of efficiently accumulating fucoxanthin and achieving high biomass productivity [17]. Unlike macroalgae, their cultivation cycles are significantly shorter and more manageable, paving the way for sustainable and cost-effective production [18]. Among the various fucoxanthin-producing microalgae, I. galbana, a member of the Prymnesiophyceae class, takes center stage. Its small size, high digestibility, and rich content of essential nutrients make it a valuable food source for the larvae of shellfish like mussels and clams [19,20,21]. Furthermore, I. galbana demonstrates significant potential for successful industrialization due to its ease of cultivation and rapid growth. These advantages have propelled this microalga to the forefront of research and development efforts focused on commercial applications [22,23,24].
Several previous works have explored the optimization of fucoxanthin production from various microalgae, including Isochrysis galbana, Phaeodactylum tricornutum, and Tisochrysis lutea [25,26]. These studies analyzed the impact of different culture media, abiotic parameters (such as temperature, irradiance, and pH), and nutrient sources on fucoxanthin productivity. Other studies have also used sophisticated approaches [27,28], such as response surface methodology and machine learning algorithms, to optimize fucoxanthin production. However, there are still gaps in understanding the complex interactions between the various factors influencing fucoxanthin production and the underlying molecular mechanisms. This research study aims to lay the groundwork for enhancing the cultivation efficiency of I. galbana and explore its potential in the pharmaceutical and nutraceutical industries. The primary objective is to optimize fucoxanthin production and the biomass’s overall antioxidant activity. This will be achieved through controlled cultivation experiments examining the influence of different enrichment solutions in the culture media. We will explore the relationships between these factors and their impact on the microalgae.
The first step of our investigation focused on analyzing the correlations between antioxidant activity, fucoxanthin production, carotenoid content, and total phenol levels. Biostatistical tools, such as principal component analysis (PCA) and multiple regression, were used to identify these connections within different culture media formulations designed explicitly for I. galbana. Building on these results, the second step consisted of implementing experimental models to optimize fucoxanthin production. The influence of modifiable growth conditions, including choice of growing medium, temperature, pH, lighting intensity, air flow rate, and CO2 flow rate, was explored. These parameters were meticulously adjusted to maximize fucoxanthin production while simultaneously assessing their impact on the antioxidant properties of the microalgae. Finally, the last step consisted of an in silico study based on molecular docking to study the molecular interaction between fucoxanthin and proteins with renowned antioxidant potentials in cellular processes.
2. Results
2.1. Optimization of Growth Parameters
The optimal enriched culture medium for maximizing the biomass production of I. galbana, as well as the content of fucoxanthin, carotenoids, total phenols, and antioxidant activity, was selected from several media: 3N-BBM+V medium, ASP-M medium, CHU-10 medium, Conway medium, Erdschreiber medium, Guillard F/2 medium, PM medium, Walne medium, and WC aquatic culture medium. Natural seawater served as a negative control due to its lack of nutrient supplements compared with enriched solutions.
Under standard operating conditions (temperature T = 25 °C, pH = 6.5, light intensity LI = 100 µmol/m2/s, air flow AFR = 0.5 L/min, and CO2 flow CO2FR from 0.1 to 0.2 L/min) over a culture period of 15 days, the results are summarized in Table 1. This table presents data on biomass, antioxidant activity of each extract, and biomolecule contents of each culture medium.

Table 1.
Dry biomass, antioxidant activity, carotenoid content, total phenolic content, and fucoxanthin content of cultures of the microalga I. galbana.
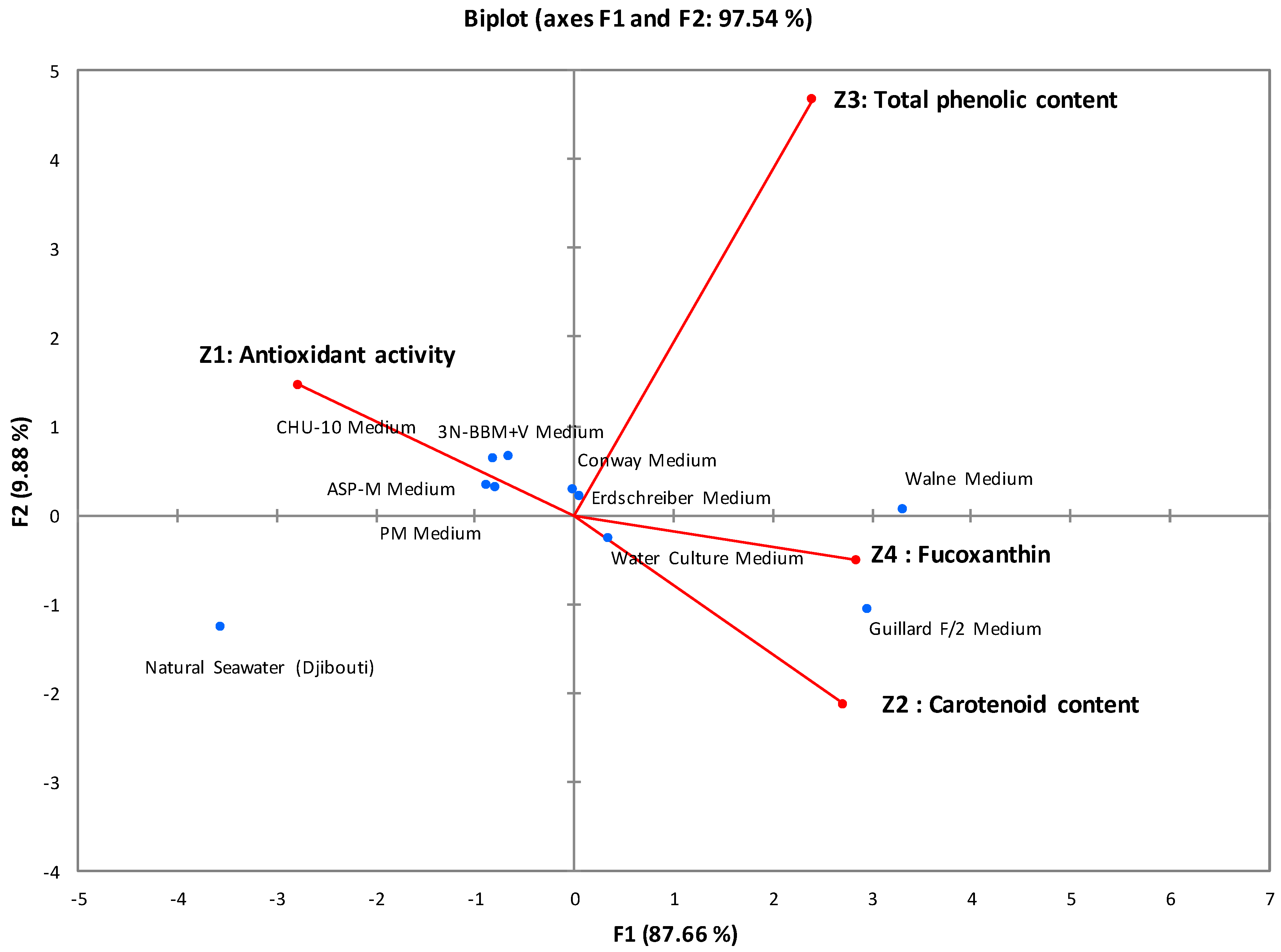
Principal component analysis (PCA) was used to determine the correlations between the studied parameters. Figure 1 graphically illustrates all the correlations obtained, with 97.54% of the variance explained by the first two axes (F1 = 87.66% and F2 = 9.88%). This statistical analysis classified the culture media into three distinct groups:
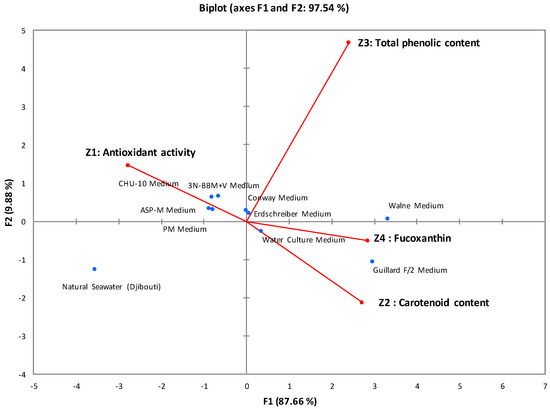
Figure 1.
Correlations between antioxidant activity, carotenoid content, total phenolic content, and fucoxanthin content of I. galbana cultures. The various circles, represented by distinct colors, symbolize groups sharing common characteristics.
First group: Comprising Walne medium and Guillard F/2 medium, which showed promising results in terms of biomass, antioxidant activity, and active biomolecule content. The IC50 of antioxidant activity ranged from 285 μg/mL to 304 μg/mL, and the fucoxanthin content reached 7.8 mg/g.
Second group: Including 3N-BBM+V, ASP-M, CHU-10, Conway, PM, WC aquatic culture, and Erdschreiber media, this group showed moderate results in terms of biomass, antioxidant activity, and active biomolecule content. The IC50 of antioxidant activity ranged from 405 μg/mL to 440 ± 2.5 μg/mL, and the fucoxanthin content ranged from 4.6 mg/g to 5.5 mg/g.
Third group: Containing only natural seawater, which served as a negative control with inferior results compared with enriched media. The measured antioxidant activity was 481 μg/mL, and the fucoxanthin content was 2.8 mg/g.
The experimental and statistical results led to the selection of Walne and Guillard F/2 media as the most effective due to their antioxidant power and high fucoxanthin content. These media share essential macronutrients, micronutrients, and vitamins, with Guillard F/2 medium distinguished by including Na2EDTA, a chelating agent absent from Walne medium. The differences between these environments are mainly based on the specific proportions of the shared compounds, distinctly influencing the growth and development of microalgae [29,30]. Finally, Walne medium was favored for subsequent studies because of its high fucoxanthin content (7.8 mg/g).
2.2. Correlation between Antioxidant Activity and Compound Content
The correlation between antioxidant activity (Z1) and the contents of carotenoids (Z2), total phenolic compounds (Z3), and fucoxanthin (Z4) in cultures of the microalga I. galbana was studied using two statistical tools: the matrix of correlation and multiple regression modeling, including multiple linear regression (MLR) and nonlinear multiple regression (MNLR).
The correlation matrix in Table 2 shows the correlation coefficients between each pair of variables. It reveals a strong positive correlation between carotenoid content (Z2) and fucoxanthin (Z4), indicating that fucoxanthin is a significant component of carotenoids. In contrast, antioxidant activity (Z1) showed negative correlations with carotenoid and fucoxanthin content, suggesting that lower values correspond to higher antioxidant activity.

Table 2.
Correlation matrix.
The results of multiple regression modeling (Table S1) show that the two models, MLR and MNLR, perform very well with high coefficients of determination (R2) of 0.954 and 0.986, respectively. However, the nonlinear model (MNLR) shows greater explanatory capacity, indicating a more complex relationship between variables, which is better captured by this model. In terms of predictive accuracy, MNLR has a mean square error (MCE) of 156.573, lower than that of MLR (262.950), confirming better accuracy of the nonlinear model. Similarly, the root mean square error (RMCE) is lower for MNLR (12.513) than for MLR (16.216), further emphasizing the superior fit of the nonlinear model to the observed data.
Figure S1 illustrates the linearity of the predictive results compared with the experimental results, demonstrating that MNLR aligns better with the experimental data than MLR. In conclusion, the results indicate that the relationships between antioxidant activity (Z1) and the contents of carotenoids (Z2), total phenolic compounds (Z3), and fucoxanthin (Z4) are well correlated, and nonlinear models are more appropriate for capturing the complexity of these relationships in I. galbana cultures.
2.3. Optimization of Experimental Conditions by Experimental Design
The experiments were planned to optimize the experimental conditions aimed at maximizing the production of fucoxanthin following the statistical tool of the experimental plan, taking into consideration five operational parameters: temperature, pH, light intensity, air flow rate, and CO2 flow rate. Table S2 presents the results obtained for the fucoxanthin content for each of the 32 tests according to the polynomial model of full factorial designs at two levels (−1 and +1).
A first-order polynomial mathematical model was developed from the results obtained, including the main factors and higher-order interactions up to the fourth order between these five parameters. The coefficients of the fucoxanthin production model equation are listed in Table S3. The average yield achieved by the optimization of conditions was 7.93 mg/g with a coefficient of variation of 1.11%.
The statistical model derived from the experimental design is extremely accurate and well fitted to the experimental data. The statistical parameters presented in Table S4 (coefficient of determination: R2 = 0.99, adjusted R2: R2adj = 0.99, predicted R2: R2pre = 0.91, and adequate precision: P = 87.36%) indicate that the model explains a large part of the observed variation and has good predictive ability for new observations.
Table S5 presents the analysis of variance (ANOVA), which is used to assess the significance of the effects of different factors in the experimental design. The model is considered significant with an F value of 386.15, with only a 4.02% probability that such a high F value is due to chance. p-Values of less than 0.05 indicate that some model terms are substantial—D, E, AE, CE, DE, ACE, CDE, and BCDE—while others are not.
Finally, Figure S2 graphically illustrates the main interactions, which are significant contributors to the variation in fucoxanthin concentration, by showing interactions in one dimension (D and E), two dimensions (AE, CE, and DE), and three dimensions (ACE and CDE).
2.4. In Silico Study
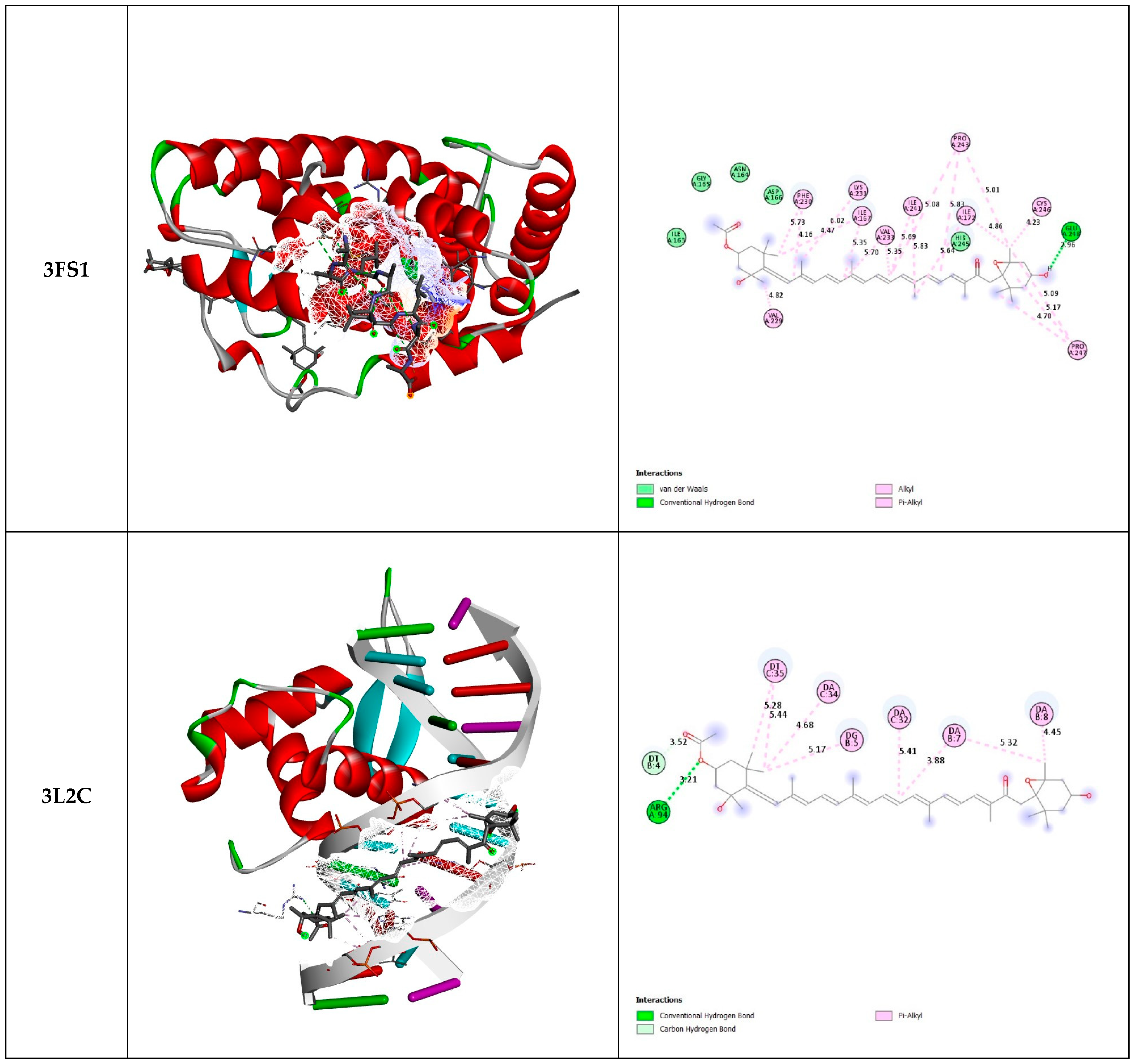
The results of molecular docking of fucoxanthin with the three selected proteins (3FS1, 3L2C, and 8BBK) are shown in Figure 2 and Table 3.
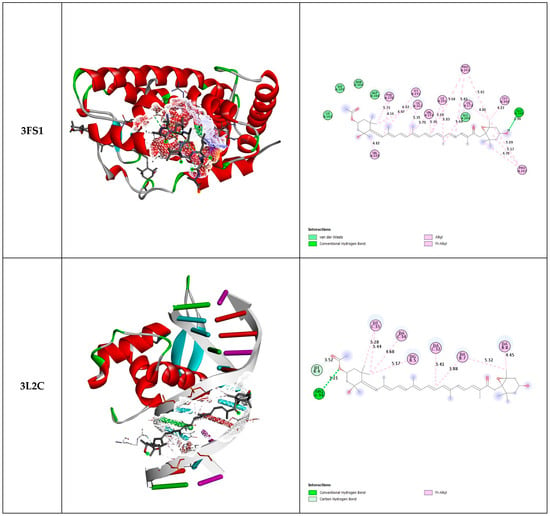

Figure 2.
Three-dimensional and two-dimensional docked views of fucoxanthin with 3FS1, 3L2C, and 8BBK proteins, respectively.

Table 3.
Qualitative and energetic characteristics of molecular docking of fucoxanthin with antioxidant activity proteins.
The three proteins studied play an important role in antioxidant processes, as follows:
The 3FS1 protein, corresponding to HNF4a (hepatocyte nuclear factor 4 alpha), plays an essential role in the cellular antioxidant process by modulating the expression of genes associated with detoxification and the response to oxidative stress. As a transcription factor, HNF4a induces the expression of genes encoding antioxidant enzymes such as superoxide dismutase (SOD), catalase, and glutathione-S-transferases. These enzymes neutralize reactive oxygen species (ROS) and attenuate oxidative damage in liver cells and other tissues where HNF4a is expressed [31,32].
The 3L2C protein, identified as FOXO4 (forkhead box O4), plays a role in the antioxidant response by regulating the expression of genes encoding antioxidant enzymes and DNA repair proteins. In response to various cellular signals, including oxidative stress, FOXO4 migrates to the cell nucleus, where it binds to specific DNA sequences and activates the transcription of genes such as SOD, glutathione peroxidase, and other enzymes and antioxidants. This action helps neutralize ROS and restore redox balance in cells, thus reducing oxidative damage [33,34,35].
As for the 8BBK protein, corresponding to Sirt3 (Sirtuin 3), Sirt3 participates in the antioxidant process by deacetylating and activating key enzymes involved in defense against oxidative stress. Notably, Sirt3 deacetylates and activates SOD2 (MnSOD) localized in mitochondria, which increases the activity of this critical antioxidant enzyme. SOD2 converts superoxide to hydrogen peroxide, which is then broken down into water and oxygen by other enzymes. In addition, Sirt3 regulates other proteins involved in the mitochondrial antioxidant response, thereby preserving the functional integrity of mitochondria and reducing cellular oxidative stress [36,37].
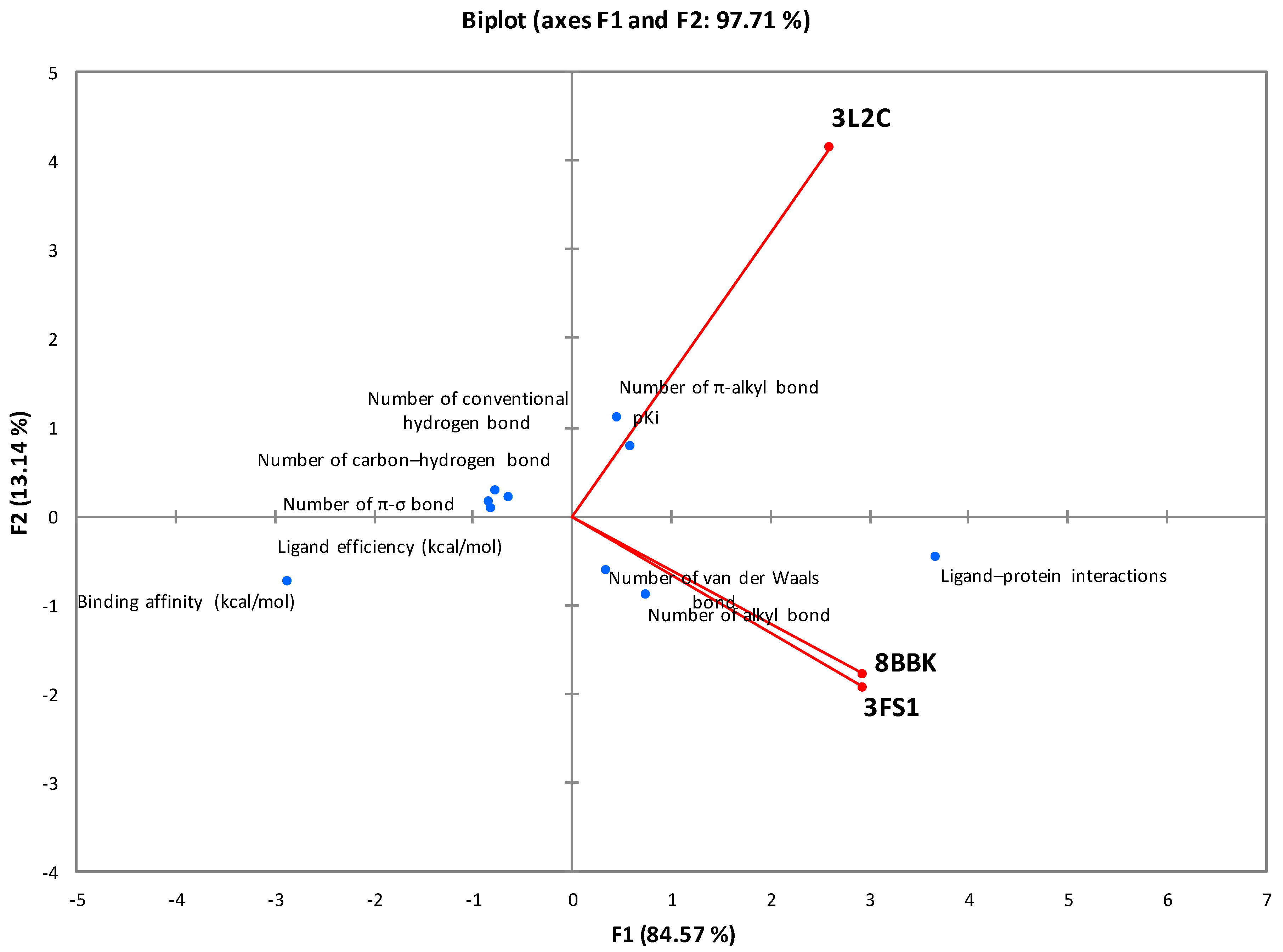
Fucoxanthin exhibits a stronger interaction with 3L2C and 8BBK proteins compared with 3FS1, indicating a higher affinity of fucoxanthin for FOXO4 (3L2C) and Sirt3 (8BBK) compared with HNF4a (3FS1). This higher affinity suggests that fucoxanthin has an increased propensity to bind FOXO4 and Sirt3. Additionally, higher ligand efficiency implies that the ligand performs better per unit size in binding to the protein. In this context, fucoxanthin demonstrates slightly higher efficacy when interacting with FOXO4 (3L2C) and Sirt3 (8BBK) compared with HNF4a (3FS1).
The correlation with the principal component analysis (PCA) of all thermodynamic parameters and interaction bonds of the different types of molecular docking is presented in Figure 3. The analysis of this representation, based on the F1 and F2 axes (97.71%), proves that the specific interactions between fucoxanthin and the HNF4a (3FS1) and Sirt3 (8BBK) proteins are well correlated. On the other hand, the interactions of these last two proteins are not correlated with FOXO4, which suggests that fucoxanthin adopts two different mechanisms depending on the proteins studied.
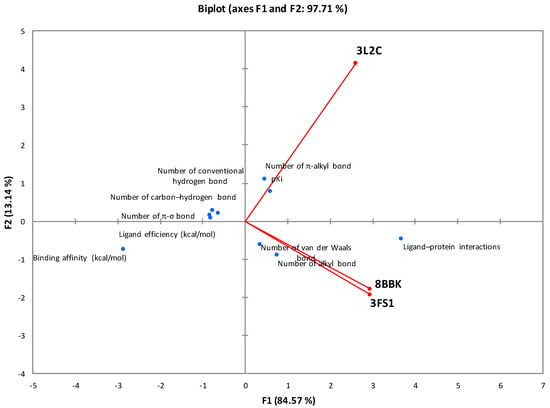
Figure 3.
Correlation between the molecular docking parameters between fucoxanthin and the proteins studied.
3. Discussion
Fucoxanthin is widely recognized for its bioactive potential and substantial antioxidant activity, making it a compound with many industrial applications. These advantages are enhanced by continued advances in extraction and quantification techniques and a better understanding of its biological properties [38].
The quantification of fucoxanthin, an abundant carotenoid in microalgal biomass, particularly in I. galbana, is essential for its industrial and therapeutic applications [39]. In the pharmaceutical sector, it is used to develop food supplements and drugs due to its beneficial antioxidant, anticancer, anti-diabetes, anti-inflammatory effects, etc. [40,41,42,43]. Nutraceuticals incorporate it into functional food products for its health properties. In the cosmetic field, it is incorporated into skin care formulations for its anti-ageing effects and ability to protect against UV damage. In agriculture, its bioactive properties make it a potential candidate as a biofertilizer and plant protective agent [44].
The main objective of this study was to quantify fucoxanthin and valorize its antioxidant properties as an abundant carotenoid in I. galbana while maximizing its extraction and use in various industrial sectors. This included optimizing production and standardizing and establishing accurate quantification methods to ensure the quality and consistency of fucoxanthin products, facilitating their introduction to the market. Furthermore, the exploitation of the antioxidant properties of fucoxanthin has been explored for the development of new products while promoting the use of natural and renewable sources to support sustainable industrial practices.
The significant results of this study showed that Walne and Guillard F/2 culture media were the most effective in producing fucoxanthin in I. galbana culture due to their high antioxidant potential and increased fucoxanthin content. Correlation analysis revealed a strong association between antioxidant activity and fucoxanthin and carotenoid content and a moderate correlation with total phenols. Regression models, whether linear or nonlinear, have been used to model these relationships, with the nonlinear model showing a better ability to explain and predict antioxidant activity, suggesting a more accurate consideration of complex interactions. Using experimental designs, optimal cultivation conditions were identified, including temperature, pH, light intensity, air flow rate, and CO2 flow rate, with particular attention paid to the last two parameters to influence the production of fucoxanthin significantly. The developed mathematical models demonstrated robust predictive ability and offer promising prospects for future optimization of fucoxanthin production in a sustainable industrial setting.
Recent works (Table 4) that have received good recognition in the literature concerning optimizing fucoxanthin production are numerous, among which the most important over the last 5 years are presented below.

Table 4.
Main works of optimization of fucoxanthin production.
In our study on fucoxanthin production from I. galbana, we explored various culture media and advanced analytical approaches to optimize the production of this bioactive pigment. Compared with previous studies, our analysis reveals several important and divergent aspects.
Medina et al. (2019) [45] demonstrated that methanol and ethanol are the most effective solvents to extract fucoxanthin from I. galbana, with significantly higher yields than petroleum ether and n-hexane. Their optimization of extraction time also highlighted the potential of I. galbana as a natural source of fucoxanthin. In contrast, our study shows that optimization of culture conditions and media, such as Walne and Guillard media, also plays a critical role in enhancing fucoxanthin production.
Gao et al. (2020) [46] optimized fucoxanthin production in Tisochrysis lutea by adjusting culture and irradiance parameters. They observed that higher dilution rates and specific irradiance maximized production. Our results support these observations to some extent, but our multidimensional approach allowed for further optimization by combining culture, statistical analysis, and bioinformatics validation techniques, which could provide additional insights for large-scale production.
Pereira et al. (2021) [47] illustrated seasonal variations in fucoxanthin production, showing that Phaeodactylum tricornutum performs better in autumn/winter, while T. lutea performs better in spring/summer. While their study highlights the feasibility of continuous production, our study highlights the impact of specific growth conditions and analytical approaches on maximizing production.
The works of Mohamadnia et al. (2021) [48] and Mohamadnia et al. (2022) [49] on optimizing fucoxanthin production using response surface methodology confirm the importance of precise culture conditions. Their approach identified optimal parameters for T. lutea, which is consistent with our observation that growth conditions play an essential role. However, our use of in silico analyses to study the molecular interactions of fucoxanthin added an additional dimension to understanding its bioactive properties.
McElroy et al. (2023) [50] implemented an integrated biorefinery approach to extract fucoxanthin and other compounds from Saccharina latissima. Their process integration to reduce the ecological footprint is particularly relevant, although our study mainly focused on optimizing culture conditions and validating the results using bioinformatics approaches.
A study conducted by Xia et al. (2023) [51] showed that intermittent CO2 supply promotes better fucoxanthin accumulation in I. galbana, a result in agreement with our observation that optimization of culture conditions is crucial for increased production. Furthermore, a study conducted by Bo et al. (2023) [52] revealed that spermidine positively influences fucoxanthin biosynthesis, highlighting the importance of biological factors in the production of this pigment.
Finally, Garcia-García et al. (2024) [53] explored the use of green solvents for fucoxanthin extraction, highlighting the advantages of modern techniques over traditional methods. This reinforces our conclusion that the use of advanced methods and solvents is essential to optimize extraction while preserving the bioactivity of the extracts.
In conclusion of this discussion, our study, through its multidimensional approach integrating the optimization of growth conditions, advanced statistical analysis, and bioinformatics validation, provides a more comprehensive and innovative overview for fucoxanthin production. These results contribute to a better understanding of the parameters influencing the production of this pigment and offer promising prospects for its industrial application.
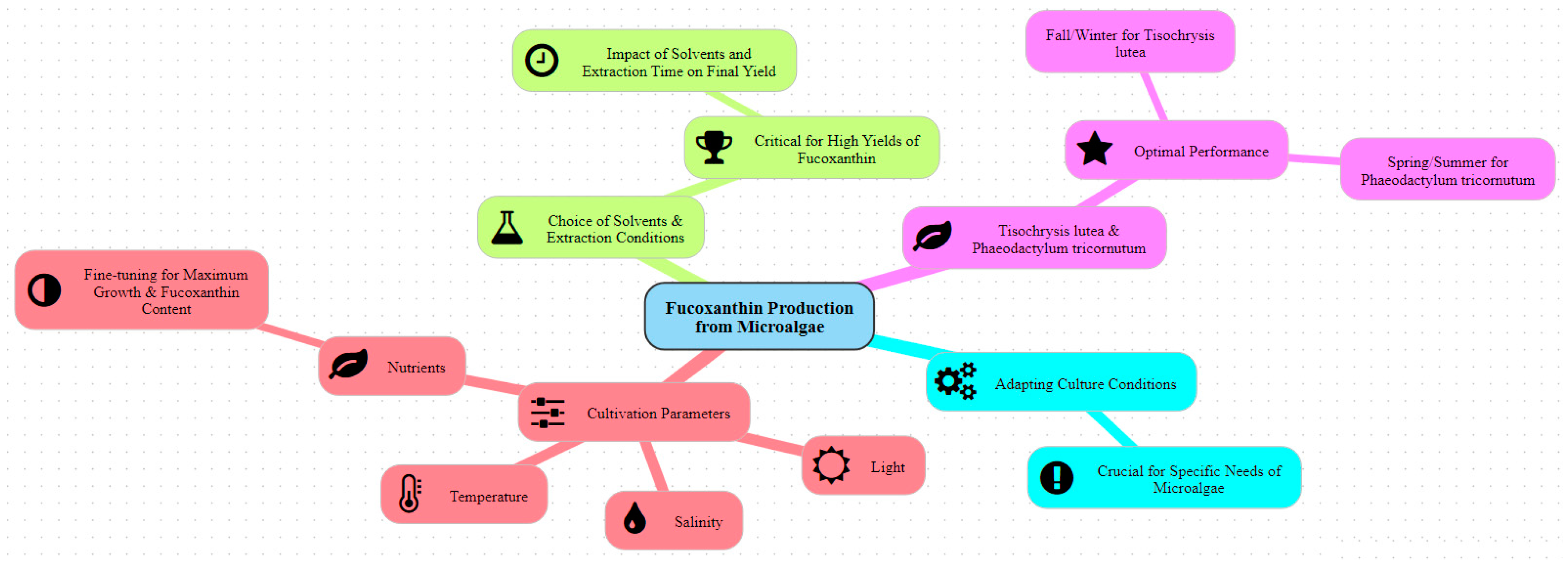
The mentioned research on the production of fucoxanthin from microalgae has focused its attention on three key aspects: optimization of environmental variations; precision in the adjustment of culture parameters such as temperature, lighting, pH, and salinity and the sources of nutrients, O2 and CO2; and the improvement of extraction processes using specific solvents and optimized conditions (Figure 4) [54,55,56,57,58]. These studies highlight the remarkable importance of adapting culture conditions to maximize productivity and fucoxanthin concentration in microalgae cultures, considering seasonal variations, while judiciously choosing extraction methods to ensure high yields of this bioactive compound.
Figure 4.
The main factors in the production of fucoxanthin from microalgae (created with www.map-this.com, accessed on 3 June 2024).
Molecular docking results of fucoxanthin with 3FS1 (HNF4a), 3L2C (FOXO4), and 8BBK (Sirt3) proteins revealed distinct interactions, highlighting a marked preference of fucoxanthin for FOXO4 and Sirt3 over HNF4a. These proteins have remarkable biochemical processes in the fine regulation of antioxidant responses and defense against oxidative stress, as evidenced by their ability to influence the expression of enzymes such as superoxide dismutase and glutathione peroxidase [59,60,61]. The analysis highlighted the specificity of these interactions, revealing that fucoxanthin adopts diverse mechanisms depending on the target proteins. This functional diversity reinforces the therapeutic potential of fucoxanthin as an antioxidant. By optimizing the production of fucoxanthin from I. galbana under specified conditions, this study laid a solid foundation for future biotechnological applications aimed at exploiting the promising antioxidant properties of this natural compound.
4. Material and Methods
4.1. Cultivation of the Microalgae I. galbana
The marine microalgae strain I. galbana was isolated from the coast of Tadjoura in Djibouti (1°46′58.084″ N, 42°53′1.667″ E). Initial isolation was conducted using Falcon conical tubes and cell isolation through micropipettes under a microscope after successive dilutions. The microalgae were then cultured in a liquid medium and on agar plates. Cultivation was carried out in seawater collected from the same location, which was sterilized and enriched with various culture media, including 3N-BBM+V medium, ASP-M medium, CHU-10 medium, Conway medium, Erdschreiber medium, Guillard F/2 medium, PM medium, Walne medium, and WC aquatic culture medium. Some of these media were supplemented with silicate (sodium metasilicate, Sigma-Aldrich 307815, Missouri, United States), particularly CHU-10 medium, Conway medium, and PM medium.
Initially, I. galbana was cultured at 25 °C in shakers using natural seawater under continuous illumination of 150 μmol m−2 s−1. Growth was monitored by measuring the optical density at 680 nm (Shimadzu UV-1601 spectrophotometer, Kyoto, Japan) every 2 days over a growth period of approximately 10 days. In the subsequent phase, cultures were sub-cultured according to the specific experimental conditions (selected medium, temperature, pH, lighting, flow rate, and CO2 flow rate) until the biomass reached the stationary phase (15th day of culture). Biomass was then harvested by centrifugation at 2000 rpm (Thermo Scientific™ Megafuge™ 8, Waltham, MA, USA) for 10 min, freeze-dried (Christ Alpha-2–4 LDPlus, Osterode am Harz, Germany), and stored at −20 °C.
4.2. Procedure for Obtaining Extracts for Analyses
The extraction process was performed using the maceration method. For this, 1 g of I. galbana biomass was extracted with 100 mL of a methanol/chloroform mixture (1:1, v/v) for 12 h at room temperature in complete darkness. This extraction was repeated three times, and all extracts were combined. The pooled extracts were then filtered through Whatman No. 4 filter paper and concentrated under reduced pressure using a rotary evaporator. The final extracts were stored in amber glass vials at −20 °C until further use [62].
4.3. Determination of Antioxidant Activity by the DPPH Method
The antioxidant activity by scavenging DPPH free radicals was evaluated following the method of Flieger et al. (2020) [63]. A stock solution of each extract was prepared in methanol at a concentration of 20 mg/mL. Then, 20 µL of each extract was added to 180 µL of DPPH radical solution (60 µM) in 96-well plates, resulting in final concentrations of 50, 100, 200, 500, and 1000 µg/mL. The samples were shaken to ensure thorough mixing. Since some colored extracts can absorb at 520 nm, a control (blank sample) was prepared by adding 20 μL of each sample solution to 180 μL of methanol. The absorbance of the samples was measured at 30, 60, and 120 min at 520 nm using methanol as a blank in a Shimadzu UV-1601 spectrophotometer. The IC50 (50% inhibitory concentration) was calculated to compare the free radical scavenging efficiency. DPPH radical scavenging activity was calculated using the following equation:
AS: Sample absorbance is the absorbance of the methanolic solution of DPPH in the presence of all the extracts and the standard.
AB: Blank absorbance is the absorbance of the sample of extracts in methanol (without DPPH to subtract the absorbance of the colored extracts).
AC: Control absorbance is the absorbance of the methanolic solution of DPPH.
4.4. Estimation of Carotenoid Content
A method described by Zhou et al. (2020) [64] was employed to determine the carotenoid content of I. galbana samples. Approximately 2 g of each sample was mixed with 25 mL of methanol, vortexed for 10 min, and filtered through Whatman No. 1 filter paper. The filtrate was fractionated with 20 mL of petroleum ether and subsequently washed with 100 mL of distilled water. Any residual water was removed using Whatman No. 1 filter paper coated with 5 g of anhydrous sodium sulfate. The extract volume was adjusted to 25 mL with ethanol, and the absorbance was measured at 450 nm using a spectrophotometer.
4.5. Total Phenolic Content
Total phenolic content was determined using the Folin–Ciocalteu reagent, according to the procedure described by Pauliuc (2020) [65]. Each sample (1 mg/mL) was mixed with 5 mL of reagent, diluted (1:10 v/v) with water, and mixed with 4 mL of 7.5% sodium carbonate. The total phenolic content was measured at 750 nm using a spectrophotometer. Gallic acid was used as a standard (0 to 200 mg/mL). The result was expressed as mg of gallic acid equivalent (GAE) per 100 g of sample.
4.6. Quantification of Fucoxanthin
Fucoxanthin quantification was carried out according to the method described by Li et al. (2021) [66] using high performance liquid chromatography (HPLC) coupled with UV detection. The analyses were carried out on an Agilent 1200 HPLC System (Agilent Technologies), including a quaternary pump and a diode array detector (DAD). Chromatographic separation was carried out using a YMC-Pack ODS-A, C18 column (250 mm × 4.6 mm, 5 µm). The column temperature was regulated at 30 °C to optimize the separation of the analytes. The mobile phase, consisting of a methanol–water mixture (80:20, v/v), was supplied at a constant flow rate of 1 mL/min. Samples were injected at a volume of 20 µL. The detection of fucoxanthin was performed at a wavelength of 450 nm, specifically chosen for this analysis. Standard solutions of fucoxanthin were prepared by dissolving the compound in methanol to obtain a stock solution, which was then diluted to prepare standard solutions with concentrations ranging from 0.5 to 12 ppb; hence, Figure S3 presents the calibration curve made for this quantification. The microalgae extracts were also diluted in methanol and injected to a volume of 20 μL under the same operating conditions to quantify fucoxanthin. Before being injected into the HPLC system, all solutions were degassed and filtered through a 0.22 µm membrane to remove particles that could damage the column or disrupt the analysis.
4.7. Statistical Studies
Numerical data were collected from three replicates for each test to ensure the accuracy of the results. Type A uncertainty assessment was performed for the statistical analysis of numerical data. The tests were then subjected to Student’s t-test (p < 0.05) to assess their significance. To detect significant differences between groups of samples, analysis of variance (ANOVA) was performed, followed by Tukey’s test for multiple comparisons. This test was used to identify significant variations between groups and provide a detailed analysis of the observed differences.
For correlation studies, data modeling was conducted using XLSTAT version 2016, employing several statistical correlation methods to determine relationships between parameters. Principal component analysis (PCA) was utilized to transform a set of correlated variables into a reduced number of uncorrelated principal components [67]. This analysis evaluated the correlation between the enrichment of culture media of the microalga I. galbana and antioxidant activity, as well as the contents of carotenoids, total phenols, and fucoxanthin, classifying the environments according to these studied parameters. Additionally, the correlation between antioxidant activity, carotenoids, total phenols, and fucoxanthin was analyzed using simple linear regressions between each pair of parameters and multiple linear regression (MLR) and multiple nonlinear regression (MNLR) [68]. These statistical approaches aimed to establish the relationships between antioxidant activity and the contents of carotenoids, total phenols, and fucoxanthin. Design-Expert 13 was used for all calculations and graphical representations for optimization studies utilizing experimental designs. The methodology aimed to minimize the experimental conditions for producing fucoxanthin from the microalga I. galbana.
The experimental design used was a full factorial design based on the following five Xi factors:
- Factor 1 = T: temperature (25 °C and 30 °C);
- Factor 2 = pH: pH (6.5 and 7.5);
- Factor 3 = light intensity: LI (100 µmol/m2/s and 500 µmol/m2/s);
- Factor 4 = air flow rate: AFR (0.5 L/min and 1.0 L/min);
- Factor 5 = CO2 flow rate: CO2FR (0.1–0.2 L/min).
Each factor was studied at two levels—high (+1) and low (−1)—resulting in 25 = 32 trials in total. The polynomial model included the following:
- A mean: a0;
- 5 main effects for each factor: ai;
- 10 interactions of order 2: aij;
- 10 interactions of order 3: aijk;
- 5 interactions of order 4: aijkl;
- 1 interaction of order 5: aijklm.
All values obtained are presented as mean ± uncertainty, with a significance level of 5% for each experiment, as determined by statistical analysis using the Student’s t-test. Each experimental variant was tested in triplicate.
4.8. In Silico Study
This in silico study used molecular docking methodology to perform a virtual screening of the crystal structures of a few antioxidant proteins available in the RCSB database, including PDB IDs (3FS1, 3L2C, and 8BBK). The choice of these crystal structures for molecular docking was guided by their relevance to the study objectives aimed at evaluating the antioxidant activity of fucoxanthin. These proteins were selected based on their potential involvement in antioxidant processes, as confirmed by previous works, as well as their structural availability in the RCSB database, ensuring their validity and feasibility for comparative computational analysis [69,70,71]. This approach allowed the investigation of specific interactions with proteins relevant to this study, thereby contributing to a cross-sectional understanding of the potential effects of fucoxanthin. To carry out this analysis, several software tools were employed, including MGLtools 1.4.6, Autodock 4.0, Autogrid 4.0, BIOVIA Discovery Studio Visualizer 2.5, ChemBiodraw Ultra 12.0, and Chemdraw 3D 10.0. Initially, protein structures were prepared using the BIOVIA Discovery Studio Visualizer to remove heteroatoms, co-crystallized ligands, and solvents to optimize conditions for docking. Autodock tools were then used to assign appropriate polar charges and generate optimized pdbqt files for each protein structure. For fucoxanthin, the structure was drawn with ChemDraw Ultra, then energetically minimized with Chem 3D Pro before being converted to pdbqt format via OpenBabel GUI. Structure-based virtual screening was performed with Autodock 4, docking fucoxanthin independently into the active site of each target protein. Ligand–protein interactions were visualized and analyzed with the BIOVIA Discovery Studio Visualizer. To validate the results, root mean square deviation (RMSD) values were calculated, ensuring that accepted poses had RMSD values of less than 2.0 for ligands redocked by co-crystallization.
5. Conclusions
This study successfully optimized the production of fucoxanthin from cultures of I. galbana. F/2 media were identified as the most effective, promoting high fucoxanthin content and improving the overall antioxidant potential of the alga. Analyses demonstrated close correlations between fucoxanthin and total carotenoids, linked to antioxidant activity. A specific statistical model (MNLR) effectively captured the complex interactions between various factors influencing fucoxanthin production. Furthermore, the experiments identified air flow rate and CO2 flow rate as crucial factors for maximizing fucoxanthin yields. Computational molecular docking analyses showed that fucoxanthin binds most efficiently to FOXO4 and Sirt3, two proteins playing critical roles in the antioxidant response and regulation of key enzymes against oxidative stress. Furthermore, the results confirmed these specific interactions, highlighting the propensity of fucoxanthin to adopt different mechanisms depending on the targeted proteins. These findings suggest the significant potential of fucoxanthin in antioxidant and therapeutic applications. In conclusion, all the data from this work validate the robustness of the developed model, which is very promising for the future optimization of fucoxanthin production in biotechnological applications.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/md22080358/s1, Figure S1. displays a validation graph depicting the predicted antioxidant activity of the marine microalga I. galbana; Figure S2. Illustrates the representations of fucoxanthin across one-dimensional, two-dimensional, and three-dimensional formats; Figure S3. Calibration curve of fucoxanthin by HPLC-UV; Table S1. Statistical parameters of the multiple regression of antioxidant activity as a function of the content of carotenoids, total phenolic compounds and fucoxanthin in cultures of the microalga I. galbana; Table S2. Fucoxanthin is used for each test in all experiments according to the design of the experiments; Table S3. Coefficients of optimized models for fucoxanthin production; Table S4. Statistical studies of the model; Table S5. ANOVA for the selected factorial model.
Author Contributions
Conceptualization: F.M.A.-L. and T.A.; Data curation: A.A. (Ayoub Ainane) and F.M.A.-L.; Formal analysis: H.M. and T.A.; Funding acquisition: F.M.A.-L.; Investigation: F.M.A.-L. and L.A.; Methodology: A.A. (Ayoub Ainane), F.M.A.-L. and L.A.; Project administration: F.M.A.-L.; Resources: P.P.J., A.M.A. and A.A. (Ahmad Ali); Software: H.M., A.M.A. and A.A. (Ayoub Ainane); Supervision: F.M.A.-L.; Validation: F.M.A.-L. and T.A.; Visualization: A.A. (Ayoub Ainane), F.M.A.-L. and T.A.; Roles/Writing—original draft: F.M.A.-L. and T.A.; Writing—review & editing: F.M.A.-L., A.A. (Ahmad Ali), P.P.J. and T.A. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
Data are contained within the article.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Bürck, M.; Ramos, S.D.P.; Braga, A.R.C. Enhancing the Biological Effects of Bioactive Compounds from Microalgae through Advanced Processing Techniques: Pioneering Ingredients for Next-Generation Food Production. Foods 2024, 13, 1811. [Google Scholar] [CrossRef] [PubMed]
- Maghzian, A.; Aslani, A.; Zahedi, R. A comprehensive review on effective parameters on microalgae productivity and carbon capture rate. J. Environ. Manag. 2024, 355, 120539. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Liang, H.; Du, H.; Jesumani, V.; He, W.; Cheong, K.-L.; Li, T.; Hong, T. Industry chain and challenges of microalgal food industry—A review. Crit. Rev. Food Sci. Nutr. 2024, 64, 4789–4816. [Google Scholar] [CrossRef]
- Ainane, T.; Abdoul-Latif, F.M.; El Yaagoubi, B.; Boujaber, N.; Oumaskour, K.; Ainane, A. Nannochloropsis oculata Microalga for Fertilization: Bioguided Fractionation of N-Hexane Extract by Stimulating Growth Activity for Cucurbita moschata. Pharmacologyonline 2021, 3, 1803–1809. [Google Scholar]
- Dhandwal, A.; Bashir, O.; Malik, T.; Salve, R.V.; Dash, K.K.; Amin, T.; Shams, R.; Wani, A.W.; Shah, Y.A. Sustainable microalgal biomass as a potential functional food and its applications in food industry: A comprehensive review. Environ. Sci. Pollut. Res. 2024, 1–19. [Google Scholar] [CrossRef]
- Guehaz, K.; Boual, Z.; Abdou, I.; Telli, A.; Belkhalfa, H. Microalgae’s polysaccharides, are they potent antioxidants? Critical review. Arch. Microbiol. 2023, 206, 14. [Google Scholar] [CrossRef]
- Duan, X.; Xie, C.; Hill, D.R.A.; Barrow, C.J.; Dunshea, F.R.; Martin, G.J.O.; Suleria, H.A. Bioaccessibility, bioavailability and bioactivities of carotenoids in microalgae: A review. Food Rev. Int. 2023, 40, 230–259. [Google Scholar] [CrossRef]
- Chandrika, U.G. Carotenoid Dyes—Properties and Production. Handb. Nat. Color. 2023, 351–369. [Google Scholar]
- Meléndez-Martínez, A.J.; Esquivel, P.; Rodriguez-Amaya, D.B. Comprehensive review on carotenoid composition: Transformations during processing and storage of foods. Food Res. Int. 2023, 169, 112773. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Sagasti, M.T.; López-Pozo, M.; Artetxe, U.; Becerril, J.M.; Hernández, A.; García-Plazaola, J.I.; Esteban, R. Carotenoids and their derivatives: A “Swiss Army knife-like” multifunctional tool for fine-tuning plant-environment interactions. Environ. Exp. Bot. 2023, 207, 105229. [Google Scholar] [CrossRef]
- Xu, J.; Lin, J.; Peng, S.; Zhao, H.; Wang, Y.; Rao, L.; Liao, X.; Zhao, L. Development of an HPLC-PDA Method for the Determination of Capsanthin, Zeaxanthin, Lutein, β-Cryptoxanthin and β-Carotene Simultaneously in Chili Peppers and Products. Molecules 2023, 28, 2362. [Google Scholar] [CrossRef]
- González-Peña, M.A.; Ortega-Regules, A.E.; Anaya de Parrodi, C.; Lozada-Ramírez, J.D. Chemistry, occurrence, properties, applications, and encapsulation of carotenoids—A review. Plants 2023, 12, 313. [Google Scholar] [CrossRef]
- Bohn, T.; de Lera, A.R.; Landrier, J.-F.; Rühl, R. Carotenoid metabolites, their tissue and blood concentrations in humans and further bioactivity via retinoid receptor-mediated signalling. Nutr. Res. Rev. 2023, 36, 498–511. [Google Scholar] [CrossRef]
- Jiří, B.; Lenka, V.; Josef, S.; Věra, K. Exploring carotenoids: Metabolism, antioxidants, and impacts on human health. J. Funct. Foods 2024, 118, 106284. [Google Scholar]
- Zhang, Z.; Wei, Z.; Xue, C. Delivery systems for fucoxanthin: Research progress, applications and future prospects. Crit. Rev. Food Sci. Nutr. 2024, 64, 4643–4659. [Google Scholar] [CrossRef]
- Abdoul-Latif, F.M.; Ainane, A.; Aboubaker, I.H.; Ali, A.M.; Mohamed, H.; Jutur, P.P.; Ainane, T. Unlocking the Green Gold: Exploring the Cancer Treatment and the Other Therapeutic Potential of Fucoxanthin Derivatives from Microalgae. Pharmaceuticals 2024, 17, 960. [Google Scholar] [CrossRef]
- Sun, H.; Yang, S.; Zhao, W.; Kong, Q.; Zhu, C.; Fu, X.; Zhang, F.; Liu, Z.; Zhan, Y.; Mou, H.; et al. Fucoxanthin from marine microalgae: A promising bioactive compound for industrial production and food application. Crit. Rev. Food Sci. Nutr. 2023, 63, 7996–8012. [Google Scholar] [CrossRef] [PubMed]
- Mapelli-Brahm, P.; Gómez-Villegas, P.; Gonda, M.L.; León-Vaz, A.; León, R.; Mildenberger, J.; Rebours, C.; Saravia, V.; Vero, S.; Vila, E.; et al. Microalgae, seaweeds and aquatic bacteria, archaea, and yeasts: Sources of carotenoids with potential antioxidant and anti-inflammatory health-promoting actions in the sustainability era. Mar. Drugs 2023, 21, 340. [Google Scholar] [CrossRef]
- Gonçalves, C.d.A.; Figueredo, C.C. What we really know about the composition and function of microalgae cell coverings? — An overview. Acta Bot. Bras. 2020, 34, 599–614. [Google Scholar] [CrossRef]
- Zarrinmehr, M.J.; Farhadian, O.; Heyrati, F.P.; Keramat, J.; Koutra, E.; Kornaros, M.; Daneshvar, E. Effect of ni-trogen concentration on the growth rate and biochemical composition of the microalga, Isochrysis galbana. Egypt. J. Aquat. Res. 2020, 46, 153–158. [Google Scholar] [CrossRef]
- Matos, J.; Afonso, C.; Cardoso, C.; Serralheiro, M.L.; Bandarra, N.M. Yogurt Enriched with Isochrysis galbana: An Innovative Functional Food. Foods 2021, 10, 1458. [Google Scholar] [CrossRef]
- Solomonova, E.S.; Shoman, N.Y.; Akimov, A.I. Hormesis effect of the herbicide glyphosate on growth and lipid synthesis in the microalga Isochrysis galbana, an object of industrial cultivation. Aquac. Int. 2024, 1–9. [Google Scholar] [CrossRef]
- Wang, Y.-Y.; Xu, S.-M.; Cao, J.-Y.; Wu, M.-N.; Lin, J.-H.; Zhou, C.-X.; Zhang, L.; Zhou, H.-B.; Li, Y.-R.; Xu, J.-L.; et al. Co-cultivation of Isochrysis galbana and Marinobacter sp. can enhance algal growth and docosahexaenoic acid production. Aquaculture 2022, 556, 738248. [Google Scholar] [CrossRef]
- Abdoul-Latif, F.M.; Oumaskour, K.; Boujaber, N.; Ainane, A.; Mohamed, J.; Ainane, T. Formulations of a cosmetic product for hair care based on extract of the microalga Isochrysis galbana: In vivo and in vitro activities. J. Anal. Sci. Appl. Biotechnol. 2021, 3, 15–19. [Google Scholar]
- Pocha, C.K.R.; Chia, W.Y.; Chew, K.W.; Munawaroh, H.S.H.; Show, P.L. Current advances in recovery and biorefinery of fucoxanthin from Phaeodactylum tricornutum. Algal Res. 2022, 65, 102735. [Google Scholar] [CrossRef]
- Kim, S.M.; Kang, S.-W.; Kwon, O.-N.; Chung, D.; Pan, C.-H. Fucoxanthin as a major carotenoid in Isochrysis aff. galbana: Characterization of extraction for commercial application. J. Korean Soc. Appl. Biol. Chem. 2012, 55, 477–483. [Google Scholar] [CrossRef]
- Stock, A.; Subramaniam, A. Accuracy of empirical satellite algorithms for mapping phytoplankton diagnostic pigments in the open ocean: A supervised learning perspective. Front. Mar. Sci. 2020, 7, 599. [Google Scholar] [CrossRef]
- Shannon, E.; Abu-Ghannam, N. Optimisation of fucoxanthin extraction from Irish seaweeds by response surface methodology. J. Appl. Phycol. 2017, 29, 1027–1036. [Google Scholar] [CrossRef]
- Grubišić, M.; Šantek, B.; Kuzmić, M.; Čož-Rakovac, R.; Šantek, M.I. Enhancement of Biomass Production of Diatom Nitzschia sp. S5 through Optimisation of Growth Medium Composition and Fed-Batch Cultivation. Mar. Drugs 2024, 22, 46. [Google Scholar] [CrossRef]
- Trinh, K.S. Comparison of Growth of Chlorella vulgaris in Flat-Plate Photobioreactor Using Batch, Fed-Batch, and Repeated Fed-Batch Techniques with Various Concentrations of Walne Medium. J. Tech. Educ. Sci. 2023, 18, 9–15. [Google Scholar] [CrossRef]
- Somade, O.T.; Ajayi, B.O.; Adeyi, O.E.; Dada, T.A.; Ayofe, M.A.; Inalu, D.C.; Ajiboye, O.I.; Shonoiki, O.M.; Adelabu, A.O.; Onikola, R.T.; et al. Ferulic acid inter-ventions ameliorate NDEA-CCl4-induced hepatocellular carcinoma via Nrf2 and p53 upregulation and Akt/PKB-NF-κB-TNF-α pathway downregulation in male Wistar rats. Toxicol. Rep. 2024, 12, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Alqrad, M.A.; El-Agamy, D.S.; Ibrahim, S.R.; Sirwi, A.; Abdallah, H.M.; Abdel-Sattar, E.; El-Halawany, A.M.; Elsaed, W.M.; Mohamed, G.A. SIRT1/Nrf2/NF-κB Signaling Mediates Anti-Inflammatory and Anti-Apoptotic Activities of Oleanolic Acid in a Mouse Model of Acute Hepatorenal Damage. Medicina 2023, 59, 1351. [Google Scholar] [CrossRef] [PubMed]
- Jeung, D.; Lee, G.-E.; Chen, W.; Byun, J.; Nam, S.-B.; Park, Y.-M.; Lee, H.S.; Kang, H.C.; Lee, J.Y.; Kim, K.D.; et al. Ribosomal S6 kinase 2-forkhead box protein O4 signaling pathway plays an essential role in melanogenesis. Sci. Rep. 2024, 14, 9440. [Google Scholar] [CrossRef] [PubMed]
- Salama, A.; Elgohary, R.; Amin, M.M.; Elwahab, S.A. Impact of protocatechuic acid on alleviation of pulmonary damage induced by cyclophosphamide targeting peroxisome proliferator activator receptor, silent information regulator type-1, and fork head box protein in rats. Inflammopharmacology 2023, 31, 1361–1372. [Google Scholar] [CrossRef] [PubMed]
- Omorou, M.; Huang, Y.; Gao, M.; Mu, C.; Xu, W.; Han, Y.; Xu, H. The forkhead box O3 (FOXO3): A key player in the regulation of ischemia and reperfusion injury. Cell. Mol. Life Sci. 2023, 80, 102. [Google Scholar] [CrossRef] [PubMed]
- Dhillon, V.S.; Shahid, M.; Deo, P.; Fenech, M. Reduced SIRT1 and SIRT3 and Lower Antioxidant Capacity of Seminal Plasma Is Associated with Shorter Sperm Telomere Length in Oligospermic Men. Int. J. Mol. Sci. 2024, 25, 718. [Google Scholar] [CrossRef] [PubMed]
- Mishra, Y.; Kaundal, R.K. Role of SIRT3 in mitochondrial biology and its therapeutic implications in neuro-degenerative disorders. Drug Discov. Today 2023, 28, 103583. [Google Scholar] [CrossRef]
- Miyashita, K.; Beppu, F.; Hosokawa, M.; Liu, X.; Wang, S. Bioactive significance of fucoxanthin and its effective extraction. Biocatal. Agric. Biotechnol. 2020, 26, 101639. [Google Scholar] [CrossRef]
- Wu, S.; Xie, X.; Huan, L.; Zheng, Z.; Zhao, P.; Kuang, J.; Liu, X.; Wang, G. Selection of optimal flocculant for effective harvesting of the fucoxanthin-rich marine microalga Isochrysis galbana. J. Appl. Phycol. 2016, 28, 1579–1588. [Google Scholar] [CrossRef]
- Bigagli, E.; D’ambrosio, M.; Cinci, L.; Niccolai, A.; Biondi, N.; Rodolfi, L.; Nascimiento, L.B.D.S.; Tredici, M.R.; Luceri, C. A comparative in vitro evaluation of the anti-inflammatory effects of a Tisochrysis lutea extract and fucoxanthin. Mar. Drugs 2021, 19, 334. [Google Scholar] [CrossRef]
- Méresse, S.; Fodil, M.; Fleury, F.; Chénais, B. Fucoxanthin, a marine-derived carotenoid from brown seaweeds and microalgae: A promising bioactive compound for cancer therapy. Int. J. Mol. Sci. 2020, 21, 9273. [Google Scholar] [CrossRef] [PubMed]
- Kawee-Ai, A.; Kim, A.T.; Kim, S.M. Inhibitory activities of microalgal fucoxanthin against α-amylase, α-glucosidase, and glucose oxidase in 3T3-L1 cells linked to type 2 diabetes. J. Oceanol. Limnol. 2019, 37, 928–937. [Google Scholar] [CrossRef]
- Foo, S.C.; Yusoff, F.M.; Ismail, M.; Basri, M.; Yau, S.K.; Khong, N.M.; Chan, K.W.; Ebrahimi, M. Antioxidant capacities of fucoxanthin-producing algae as influenced by their carotenoid and phenolic contents. J. Biotechnol. 2017, 241, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Sandhya, S.V.; Vijayan, K.K. Symbiotic association among marine microalgae and bacterial flora: A study with special reference to commercially important Isochrysis galbana culture. J. Appl. Phycol. 2019, 31, 2259–2266. [Google Scholar] [CrossRef]
- Medina, E.; Cerezal, P.; Morales, J.; Ruiz-Domínguez, M.C. Fucoxanthin from marine microalga Isochrysis galbana: Optimization of extraction methods with organic solvents. Dyna 2019, 86, 174–178. [Google Scholar] [CrossRef]
- Gao, F.; (Cabanelas, I.I.T.; Wijffels, R.H.; Barbosa, M.J. Process optimization of fucoxanthin production with Tisochrysis lutea. Bioresour. Technol. 2020, 315, 123894. [Google Scholar] [CrossRef]
- Pereira, H.; Sá, M.; Maia, I.; Rodrigues, A.; Teles, I.; Wijffels, R.H.; Navalho, J.; Barbosa, M. Fucoxanthin production from Tisochrysis lutea and Phaeodactylum tricornutum at industrial scale. Algal Res. 2021, 56, 102322. [Google Scholar] [CrossRef]
- Mohamadnia, S.; Tavakoli, O.; Faramarzi, M.A. Enhancing production of fucoxanthin by the optimization of culture media of the microalga Tisochrysis lutea. Aquaculture 2021, 533, 736074. [Google Scholar] [CrossRef]
- Mohamadnia, S.; Tavakoli, O.; Faramarzi, M.A. Optimization of metabolic intermediates to enhance the production of fucoxanthin from Tisochrysis lutea. J. Appl. Phycol. 2022, 34, 1269–1279. [Google Scholar] [CrossRef]
- McElroy, C.R.; Kopanitsa, L.; Helmes, R.; Fan, J.; Attard, T.M.; Simister, R.; van den Burg, S.; Ladds, G.; Bailey, D.S.; Gomez, L.D. Integrated biorefinery approach to valorise Saccharina latissima biomass: Combined sustainable processing to produce biologically active fuco-xanthin, mannitol, fucoidans and alginates. Environ. Technol. Innov. 2023, 29, 103014. [Google Scholar] [CrossRef]
- Xia, Y.; Sekine, M.; Hirahara, M.; Kishinami, H.; Yusoff, F.M.; Toda, T. Effects of concentration and frequency of CO2 supply on productivity of marine microalga Isochrysis galbana. Algal Res. 2023, 70, 102985. [Google Scholar] [CrossRef]
- Bo, Y.; Wang, S.; Ma, F.; Manyakhin, A.Y.; Zhang, G.; Li, X.; Zhou, C.; Ge, B.; Yan, X.; Ruan, R.; et al. The influence of spermidine on the build-up of fucoxanthin in Isochrysis sp. Acclimated to varying light intensities. Bioresour. Technol. 2023, 387, 129688. [Google Scholar] [CrossRef] [PubMed]
- García-García, P.; Ospina, M.; Señoráns, F.J. Tisochrysis lutea as a source of omega-3 polar lipids and fucoxanthin: Extraction and characterization using green solvents and advanced techniques. J. Appl. Phycol. 2024, 36, 1697–1708. [Google Scholar] [CrossRef]
- Manochkumar, J.; Jonnalagadda, A.; Cherukuri, A.K.; Vannier, B.; Janjaroen, D.; Ramamoorthy, S. Enhancing cellular production of fucoxanthin through machine learning assisted predictive approach in Isochrysis galbana. bioRxiv 2024, 26, 591373. [Google Scholar]
- Khaw, Y.S.; Yusoff, F.M.; Tan, H.T.; Mazli, N.A.I.N.; Nazarudin, M.F.; Shaharuddin, N.A.; Omar, A.R.; Takahashi, K. Fucoxanthin production of microalgae under different culture factors: A systematic review. Mar. Drugs 2022, 20, 592. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Zhou, C.; Cheng, P.; Zhu, J.; Hou, Y.; Li, Y.; Zhang, J.; Yan, X. A simple and efficient strategy for fucoxanthin extraction from the microalga Phaeodactylum tricornutum. Algal Res. 2022, 61, 102610. [Google Scholar] [CrossRef]
- Mohamadnia, S.; Tavakoli, O.; Faramarzi, M.A.; Shamsollahi, Z. Production of fucoxanthin by the microalga Ti-sochrysis lutea: A review of recent developments. Aquaculture 2020, 516, 734637. [Google Scholar] [CrossRef]
- Sun, Z.; Wang, X.; Liu, J. Screening of Isochrysis strains for simultaneous production of docosahexaenoic acid and fucoxanthin. Algal Res. 2019, 41, 101545. [Google Scholar] [CrossRef]
- Ranjith, H.V.; Sagar, D.; Kalia, V.K.; Dahuja, A.; Subramanian, S. Differential activities of antioxidant enzymes, superoxide dismutase, peroxidase, and catalase vis-à-vis phosphine resistance in field populations of lesser grain borer (Rhyzopertha dominica) from India. Antioxidants 2023, 12, 270. [Google Scholar] [CrossRef]
- Vašková, J.; Kočan, L.; Vaško, L.; Perjési, P. Glutathione-related enzymes and proteins: A review. Molecules 2023, 28, 1447. [Google Scholar] [CrossRef]
- Halliwell, B. Understanding mechanisms of antioxidant action in health and disease. Nat. Rev. Mol. Cell Biol. 2024, 25, 13–33. [Google Scholar] [CrossRef] [PubMed]
- Lfitat, A.; Ed-dahmani, I.; Bousraf, F.Z.; Belhaj, A.; Ainane, T.; Taleb, M.; Gourch, A.; Abdellaoui, A. Antifungal Activity of the Argan Kernels Extracts and the Heat Treatment Effect on the Activity Variation. Indones. Food Nutr. Prog. 2023, 20, 10–23. [Google Scholar] [CrossRef]
- Flieger, J.; Flieger, M. The [DPPH●/DPPH-H]-HPLC-DAD method on tracking the antioxidant activity of pure antioxidants and goutweed (Aegopodium podagraria L.) hydroalcoholic extracts. Molecules 2020, 25, 6005. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Niu, Y.; Ding, X.; Zhao, S.; Li, Y.; Fan, G.; Zhang, S.; Liao, K. Analysis of carotenoid content and diversity in apricots (Prunus armeniaca L.) grown in China. Food Chem. 2020, 330, 127223. [Google Scholar] [CrossRef]
- Pauliuc, D.; Dranca, F.; Oroian, M. Antioxidant activity, total phenolic content, individual phenolics and physico-chemical parameters suitability for Romanian honey authentication. Foods 2020, 9, 306. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Feng, H.; Ouyang, X.; Ling, J. Determination of Fucoxanthin in Bloom-Forming Macroalgae by HPLC–UV. J. Chromatogr. Sci. 2021, 59, 978–982. [Google Scholar] [CrossRef] [PubMed]
- Abdoul-Latif, F.M.; Elmi, A.; Merito, A.; Nour, M.; Risler, A.; Ainane, A.; Bignon, J.; Ainane, T. Essential oil of Ruta chalepensis L. from Djibouti: Chemical Analysis and Modeling of In Vitro Anticancer Profiling. Separations 2022, 9, 387. [Google Scholar] [CrossRef]
- Aamir, H.; Aamir, K.; Javed, M.F. Linear and Non-Linear Regression Analysis on the Prediction of Compressive Strength of Sodium Hydroxide Pre-Treated Crumb Rubber Concrete. Eng. Proc. 2023, 44, 5. [Google Scholar] [CrossRef]
- Muhammed, M.T.; Aki-Yalcin, E. Molecular docking: Principles, advances, and its applications in drug discovery. Lett. Drug Des. Discov. 2024, 21, 480–495. [Google Scholar] [CrossRef]
- Mohamed Abdoul-Latif, F.; Ainane, A.; Merito, A.; Aboubaker, I.H.; Mohamed, H.; Cherroud, S.; Ainane, T. The Effects of Khat Chewing among Djiboutians: Dental Chemical Studies, Gingival Histopathological Analyses and Bioinformatics Approaches. Bioengineering 2024, 11, 716. [Google Scholar] [CrossRef]
- Chen, G.; Seukep, A.J.; Guo, M. Recent advances in molecular docking for the research and discovery of potential marine drugs. Mar. Drugs 2020, 18, 545. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).