Hepatitis C Virus Treatment Status and Barriers among Patients in Methadone Maintenance Treatment Clinics in Guangdong Province, China: A Cross-Sectional, Observational Study
Abstract
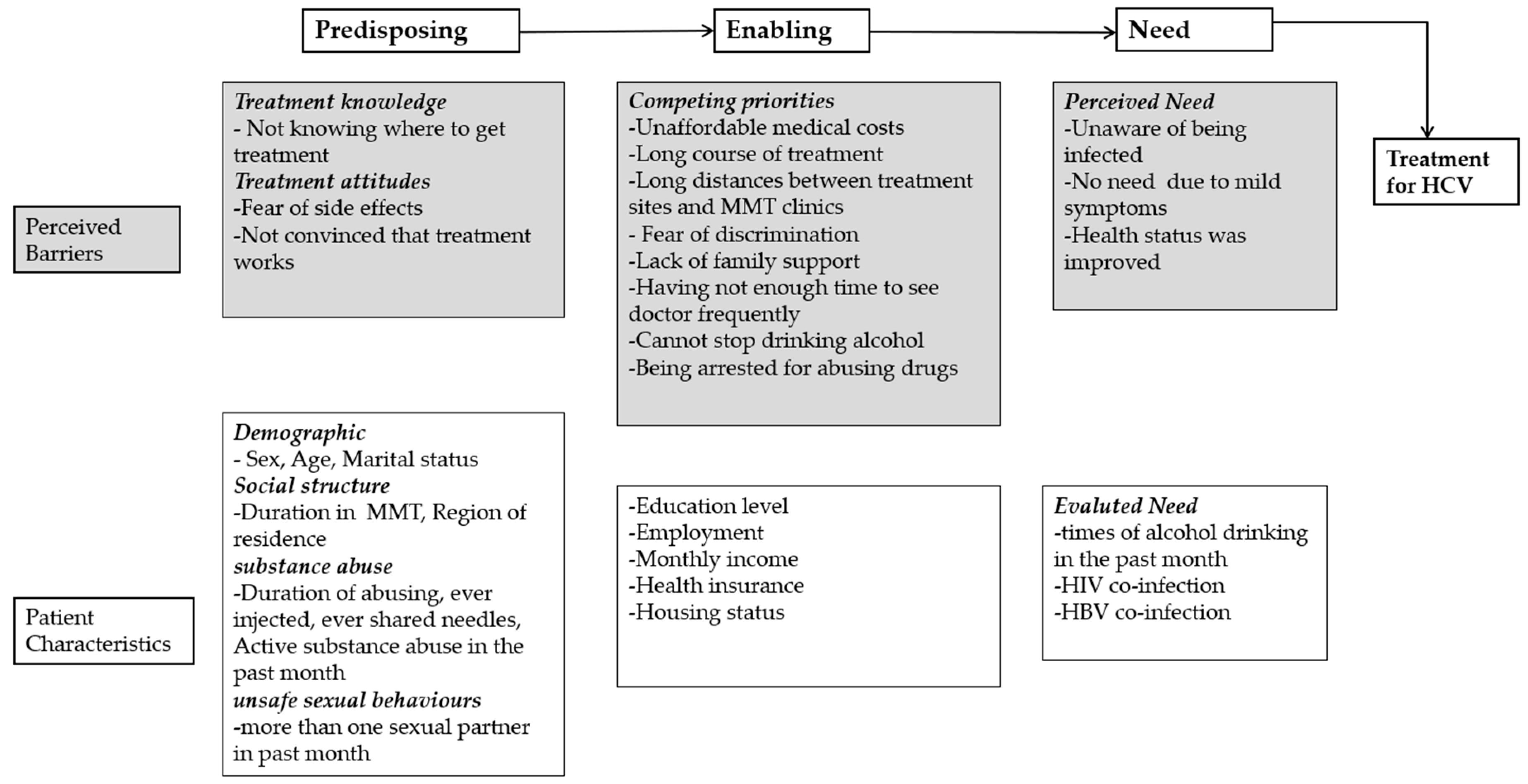
:1. Introduction
2. Materials and Methods
2.1. Design and Setting
2.2. Participants
2.3. Data Collection
2.4. Measures
2.4.1. Self-Reported HCV Infection Status
2.4.2. HCV Treatment Status
2.4.3. Patients Characteristics
2.4.4. Willingness to Accept DOT and Barriers Against It
2.4.5. Willingness to Use DAAs and Barriers against Using Them
2.5. Statistical Analysis
3. Results
3.1. Patient Characteristics According to Self-Reported HCV Status
3.2. HCV Treatment Status among Those in the MMT Program who Reported HCV Infection
3.3. Reasons for Not Initiating Treatment among Those in the MMT Program Who Reported HCV Infection
3.4. Reasons for Discontinuing Treatment among Those Initiating Treatment for HCV in the MMT Program
3.5. Factors for Not Initiating Treatment among Those in the MMT Program Reporting HCV Infection
3.6. Willingness to Use DOT in MMT Clinics among Those in the MMT Program Reporting HCV Infection
3.7. Willingness to use DAAs in MMT Clinics among Those in the MMT Program Reporting HCV Infection
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. Global Hepatitis Report; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Su, Y.Y.; Wang, N. Primary risk factors of hepatitis C virus infection: A Meta-analysis. Zhonghua Liu Xing Bing Xue Za Zhi = Zhonghua Liuxingbingxue Zazhi 2011, 32, 940–945. [Google Scholar] [PubMed]
- Grebely, J.; Hajarizadeh, B.; Dore, G.J. Direct-acting antiviral agents for HCV infection affecting people who inject drugs. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 641–651. [Google Scholar] [CrossRef] [PubMed]
- Office of China National Narcotics Control Commission. Annual Report on Drug Control in China, 2017; Ministry of Public Security: Bejing, China, 2017. [Google Scholar]
- Lin, G.; Dongmin, L.; Peilong, L.; Wei, G.; Yan, C. Population specific sentinel surveillance for HIV infection, syphilis and HCV infection in China, during 2010–2015. Dis. Surveill. 2017, 32. [Google Scholar] [CrossRef]
- WHO. Global Health Sector Strategy on Viral Hepatitis, 2016–2021; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Litwin, A.H.; Soloway, I.; Gourevitch, M.N. Integrating services for injection drug users infected with hepatitis C virus with methadone maintenance treatment: Challenges and opportunities. Clin. Infect. Dis. 2005, 40 (Suppl. 5), S339–S345. [Google Scholar] [CrossRef]
- Li, J.; Gordon, S.C.; Rupp, L.B.; Zhang, T.; Trudeau, S.; Holmberg, S.D.; Moorman, A.C.; Spradling, P.R.; Teshale, E.H.; Boscarino, J.A.; et al. Sustained virological response to hepatitis C treatment decreases the incidence of complications associated with type 2 diabetes. Aliment. Pharmacol. Ther. 2019, 49, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Zou, X.; Ling, L.; Zhang, L. Trends and risk factors for HIV, HCV and syphilis seroconversion among drug users in a methadone maintenance treatment programme in China: A 7-year retrospective cohort study. BMJ Open 2015, 5, e008162. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Mao, W.; Zhang, L.; Jiang, B.; Xiao, Y.; Jia, Y.; Wu, P.; Cassell, H.; Vermund, S. Methadone maintenance therapy and HIV counseling and testing are associated with lower frequency of risky behaviors among injection drug users in China. Subst. Use Misuse 2015, 50, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, X.; Liang, Y.; Chow, E.P.; Wang, Y.; Wilson, D.P.; Zhang, L. HIV and HCV prevalence among entrants to methadone maintenance treatment clinics in China: A systematic review and meta-analysis. BMC Infect. Dis. 2012, 12, 130. [Google Scholar] [CrossRef] [PubMed]
- Waizmann, M.; Ackermann, G. High rates of sustained virological response in hepatitis C virus-infected injection drug users receiving directly observed therapy with peginterferon alpha-2a (40KD) (PEGASYS) and once-daily ribavirin. J. Subst. Abus. Treat. 2010, 38, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Harris, K.A., Jr.; Arnsten, J.H.; Litwin, A.H. Successful integration of hepatitis C evaluation and treatment services with methadone maintenance. J. Addict. Med. 2010, 4, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Grebely, J.; Genoway, K.; Khara, M.; Duncan, F.; Viljoen, M.; Elliott, D.; Raffa, J.D.; De Vlaming, S.; Conway, B. Treatment uptake and outcomes among current and former injection drug users receiving directly observed therapy within a multidisciplinary group model for the treatment of hepatitis C virus infection. Int. J. Drug Policy 2007, 18, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.; Hou, J.L. The guideline of prevention and treatment for hepatitis C: A 2015 update. Zhonghua Gan Zang Bing Za Zhi = Zhonghua Ganzangbing Zazhi = Chin. J. Hepatol. 2015, 23, 906–923. [Google Scholar] [CrossRef]
- WHO. Guidelines for the Screening, Care and Treatment of Persons with Chronic Hepatitis C Infection; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Du, J.; Wang, Z.; Xie, B.; Zhao, M. Hepatitis C knowledge and alcohol consumption among patients receiving methadone maintenance treatment in Shanghai, China. Am. J. Drug Alcohol Abuse. 2012, 38, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.B.; Zhang, L.; Wang, J.; Huang, L.P.; Zhou, Z.R.; Cao, Y.N.; Zhao, M.; Du, J. Hepatitis C infection, related services, and barriers to HCV treatment among drug users in methadone maintenance treatment (MMT) clinics in Shanghai, China. Harm Reduct. J. 2017, 14, 71. [Google Scholar] [CrossRef] [PubMed]
- Mravcik, V.; Strada, L.; Stolfa, J.; Bencko, V.; Groshkova, T.; Reimer, J.; Schulte, B. Factors associated with uptake, adherence, and efficacy of hepatitis C treatment in people who inject drugs: A literature review. Patient Prefer. Adherence 2013, 7, 1067–1075. [Google Scholar] [CrossRef] [PubMed]
- Taherkhani, R.; Farshadpour, F. Global elimination of hepatitis C virus infection: Progresses and the remaining challenges. World J. Hepatol. 2017, 9, 1239–1252. [Google Scholar] [CrossRef] [PubMed]
- The Directorate-General Approves the Listing of Dalapitavir Hydrochloride Tablets and Aspirin Soft Capsules. Available online: http://www.sda.gov.cn/WS01/CL0051/172150.html (accessed on 27 March 2019).
- Pol, S.; Parlati, L.; Jadoul, M. Hepatitis C virus and the kidney. J. Int. AIDS Soc. 2019, 15, 73–86. [Google Scholar] [CrossRef] [PubMed]
- China National Narcotic Control Committee. China Drug Report; China National Narcotic Control Committee: Beijing, China, 2017. [Google Scholar]
- Marciniewicz, E.; Podgorski, P.; Pawlowski, T.; Malyszczak, K.; Fleischer-Stepniewska, K.; Knysz, B.; Waliszewska-Prosol, M.; Zelwetro, A.; Rymer, W.; Inglot, M.; et al. Evaluation of brain volume alterations in HCV-infected patients after interferon-free therapy: A pilot study. J. Neurol. Sci. 2019, 399, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhang, D.; Chen, W.; Zou, X.; Ling, L. High prevalence of HIV, HCV and tuberculosis and associated risk behaviours among new entrants of methadone maintenance treatment clinics in Guangdong Province, China. PLoS ONE 2013, 8, e76931. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Shi, C.X.; Rou, K.; Zhao, Y.; Cao, X.; Luo, W.; Liu, E.; Wu, Z. Baseline HCV Antibody Prevalence and Risk Factors among Drug Users in China’s National Methadone Maintenance Treatment Program. PLoS ONE 2016, 11, e0147922. [Google Scholar] [CrossRef] [PubMed]
- Aspinall, E.J.; Corson, S.; Doyle, J.S.; Grebely, J.; Hutchinson, S.J.; Dore, G.J.; Goldberg, D.J.; Hellard, M.E. Treatment of hepatitis C virus infection among people who are actively injecting drugs: A systematic review and meta-analysis. Clin. Infect. Dis. 2013, 57 (Suppl. 2), S80–S89. [Google Scholar] [CrossRef] [PubMed]
- Trooskin, S.B.; Poceta, J.; Towey, C.M.; Yolken, A.; Rose, J.S.; Luqman, N.L.; Preston, T.W.; Chan, P.A.; Beckwith, C.; Feller, S.C.; et al. Results from a Geographically Focused, Community-Based HCV Screening, Linkage-to-Care and Patient Navigation Program. J. Gen. Intern. Med. 2015, 30, 950–957. [Google Scholar] [CrossRef] [PubMed]
- Falade-Nwulia, O.; Mehta, S.H.; Lasola, J.; Latkin, C.; Niculescu, A.; O’Connor, C.; Chaulk, P.; Ghanem, K.; Page, K.R.; Sulkowski, M.S.; et al. Public health clinic-based hepatitis C testing and linkage to care in Baltimore. J. Viral Hepat. 2016, 23, 366–374. [Google Scholar] [CrossRef] [PubMed]
- Havens, J.R.; Lofwall, M.R.; Frost, S.D.; Oser, C.B.; Leukefeld, C.G.; Crosby, R.A. Individual and network factors associated with prevalent hepatitis C infection among rural Appalachian injection drug users. Am. J. Public Health 2013, 103, e44–e52. [Google Scholar] [CrossRef] [PubMed]
- Andersen, R.M. Revisiting the behavioral model and access to medical care: Does it matter? J. Health Soc. Behav. 1995, 36, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Notice on Printing and Distributing Methods for the Administration of Drug Maintenance Treatment. Available online: http://www.nhfpc.gov.cn/jkj/s3584/201701/0a97d574cf4744e1bbc335ed5de315b9.shtml (accessed on 30 March 2019).
- Wu, B.; Wang, Z.; Xie, Q. Cost-effectiveness of novel regimens for Chinese patients with chronic hepatitis C. Curr. Med Res. Opin. 2018, 35, 847–857. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Jin, X.; Duan, C.A.; Chang, F. Cost-effectiveness of daclatasvir plus asunaprevir for chronic hepatitis C genotype 1b treatment-naive patients in China. Clin. Drug Investig. 2018, 13, e0195117. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Ma, A.; Liu, Q. Cost-Effectiveness of Elbasvir/Grazoprevir Versus Daclatasvir Plus Asunaprevir in Patients with Chronic Hepatitis C Virus Genotype 1b Infection in China. Clin. Drug Investig. 2018, 38, 1031–1039. [Google Scholar] [CrossRef] [PubMed]
- Notice of the Ministry of Human Resources and Social Security of the National Healthcare Security Administration on Printing and Distributing the Catalogue of National Basic Medical Insurance, Work Injury Insurance and Maternity Insurance Drugs. Available online: http://www.nhsa.gov.cn/art/2019/8/20/art_37_1666.html (accessed on 05 September 2019).
- Lang, H.; Wen, J.; Hou, M.; Hei, F.; Wang, X. HCV infection and liver fibrosis among drug users in methadone maintenance treatment clinics. China J. AIDS STD 2017, 23, 512–516. [Google Scholar] [CrossRef]
- Grebely, J.; Knight, E.; Ngai, T.; Genoway, K.A.; Raffa, J.D.; Storms, M.; Gallagher, L.; Krajden, M.; Dore, G.J.; Duncan, F.; et al. Reinfection with hepatitis C virus following sustained virological response in injection drug users. J. Gastroenterol. Hepatol. 2010, 25, 1281–1284. [Google Scholar] [CrossRef] [PubMed]
- Islam, N.; Krajden, M.; Shoveller, J.; Gustafson, P.; Gilbert, M.; Buxton, J.A.; Wong, J.; Tyndall, M.W.; Janjua, N.Z. Incidence, risk factors, and prevention of hepatitis C reinfection: A population-based cohort study. Lancet Gastroenterol. Hepatol. 2017, 2, 200–210. [Google Scholar] [CrossRef]
- Cipriano, L.E.; Goldhaber-Fiebert, J.D. Population Health and Cost-Effectiveness Implications of a “Treat All” Recommendation for HCV: A Review of the Model-Based Evidence. MDM Policy Pract. 2018, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Obach, D.; Deuffic-Burban, S.; Esmat, G.; Anwar, W.A.; Dewedar, S.; Canva, V.; Cousien, A.; Doss, W.; Mostafa, A.; Pol, S.; et al. Effectiveness and Cost-effectiveness of Immediate Versus Delayed Treatment of Hepatitis C Virus–Infected Patients in a Country With Limited Resources: The Case of Egypt. Clin. Infect. Dis. 2014, 58, 1064–1071. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bruggmann, P. Accessing Hepatitis C patients who are difficult to reach: It is time to overcome barriers. J. Viral Hepat. 2012, 19, 829–835. [Google Scholar] [CrossRef] [PubMed]
- Crowley, D.; Cullen, W.; Laird, E.; Lambert, J.S.; Mc Hugh, T.; Murphy, C.; Van Hout, M.C. Exploring Patient Characteristics and Barriers to Hepatitis C Treatment in Patients on Opioid Substitution Treatment Attending a Community Based Fibro-scanning Clinic. J. Transl. Int. Med. 2017, 5, 112–119. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zickmund, S.; Ho, E.Y.; Masuda, M.; Ippolito, L.; LaBrecque, D.R. “They treated me like a leper”. Stigmatization and the quality of life of patients with hepatitis C. J. Gen. Intern. Med. 2003, 18, 835–844. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reid, K.W.; Vittinghoff, E.; Kushel, M.B. Association between the level of housing instability, economic standing and health care access: A meta-regression. J. Health Care Poor Underserved 2008, 19, 1212–1228. [Google Scholar] [CrossRef] [PubMed]
- Charlebois, A.; Lee, L.; Cooper, E.; Mason, K.; Powis, J. Factors associated with HCV antiviral treatment uptake among participants of a community-based HCV programme for marginalized patients. J. Viral Hepat. 2012, 19, 836–842. [Google Scholar] [CrossRef] [PubMed]
- Richmond, J.A.; Dunning, T.L.; Desmond, P.V. Health professionals’ attitudes toward caring for people with hepatitis C. J. Viral Hepat. 2007, 14, 624–632. [Google Scholar] [CrossRef] [PubMed]
- Frazer, K.; Glacken, M.; Coughlan, B.; Staines, A.; Daly, L. Hepatitis C virus in primary care: Survey of nurses’ attitudes to caring. J. Adv. Nurs. 2011, 67, 598–608. [Google Scholar] [CrossRef] [PubMed]
- Grebely, J.; Genoway, K.A.; Raffa, J.D.; Dhadwal, G.; Rajan, T.; Showler, G.; Kalousek, K.; Duncan, F.; Tyndall, M.W.; Fraser, C.; et al. Barriers associated with the treatment of hepatitis C virus infection among illicit drug users. Drug Alcohol Depend. 2008, 93, 141–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mehta, S.H.; Lucas, G.M.; Mirel, L.B.; Torbenson, M.; Higgins, Y.; Moore, R.D.; Thomas, D.L.; Sulkowski, M.S. Limited effectiveness of antiviral treatment for hepatitis C in an urban HIV clinic. AIDS 2006, 20, 2361–2369. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.J. Treatment of patients with dual hepatitis C virus and hepatitis B virus infection: Resolved and unresolved issues. J. Gastroenterol. Hepatol. 2014, 29, 26–30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.; Swann, R.; Thomas, E.; Innes, H.A.; Valerio, H.; Hayes, P.C.; Allen, S.; Barclay, S.T.; Wilks, D.; Fox, R.; et al. Impact of previous hepatitis B infection on the clinical outcomes from chronic hepatitis C? A population-level analysis. J. Viral Hepat. 2018. [Google Scholar] [CrossRef] [PubMed]
- Yin, W.; Hao, Y.; Sun, X.; Gong, X.; Li, F.; Li, J.; Rou, K.; Sullivan, S.G.; Wang, C.; Cao, X.; et al. Scaling up the national methadone maintenance treatment program in China: Achievements and challenges. Int. J. Epidemiol. 2010, 39 (Suppl. 2), ii29–ii37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hua, L.; Andersen, J.W.; Daar, E.S.; Glesby, M.J.; Hollabaugh, K.; Tierney, C. Hepatitis C virus/HIV coinfection and responses to initial antiretroviral treatment. AIDS 2013, 27, 2725–2734. [Google Scholar] [CrossRef] [PubMed]
- Pol, S.; Parlati, L. Treatment of hepatitis C: The use of the new pangenotypic direct-acting antivirals in “special populations”. Liver Int. 2018, 38 (Suppl. 1), 28–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Micallef, J.M.; Kaldor, J.M.; Dore, G.J. Spontaneous viral clearance following acute hepatitis C infection: A systematic review of longitudinal studies. J. Viral Hepat. 2006, 13, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Martinello, M.; Dore, G.J.; Matthews, G.V.; Grebely, J. Strategies to Reduce Hepatitis C Virus Reinfection in People Who Inject Drugs. Infect. Dis. Clin. N. Am. 2018, 32, 371–393. [Google Scholar] [CrossRef] [PubMed]




| Variables | Initiating Treatment (%) | COR (95% CI) | p | AOR (95% CI) | p | |
|---|---|---|---|---|---|---|
| No (n = 222) | Yes (n = 144) | |||||
| Predisposing factors | ||||||
| Gender | 0.016 | 0.019 | ||||
| male | 188 (58) | 134 (42) | Reference | Reference | ||
| female | 34 (77) | 10 (23) | 0.41 (0.20–0.86) | 0.40 (0.18–0.86) | ||
| Age | 0.663 | |||||
| 18–34 | 20 (63) | 12 (37) | Reference | |||
| 34–44 | 126 (62) | 76 (38) | 1.01 (0.47–2.17) | |||
| ≥45 | 76 (58) | 56 (42.) | 1.23 (0.56–2.72) | |||
| 42.5 ± 6.25 | 42.9 ± 6.08 | 1.01 (0.98–1.05) | ||||
| Marital Status | 0.278 | |||||
| Married | 128 (58) | 91 (42) | Reference | |||
| Single | 58 (60) | 38 (40) | 0.92 (0.57–1.50) | |||
| Divorced or windowed | 36 (71) | 15 (29) | 0.59 (0.30–1.13) | |||
| Duration in MMT (years) | 0.083 | 0.173 | ||||
| <5 | 64 (54) | 54 (46) | Reference | Reference | ||
| ≥5 | 158 (64) | 90 (36) | 0.68 (0.43–1.05) | 0.72 (0.45–1.16) | ||
| Region of residence | 0.994 | |||||
| urban | 151 (61) | 98 (39) | Reference | |||
| rural | 71 (61) | 46 (39) | 1.00 (0.64, 1.57) | |||
| Duration of abusing drugs(years) # | 0.369 | |||||
| <10 | 44 (59) | 31 (41) | Reference | |||
| 10–19 | 144 (63) | 86 (37) | 0.85 (0.50–1.44) | |||
| ≥20 | 30 (53) | 27 (47) | 1.28 (0.64–2.56) | |||
| 14.0 ± 5.47 | 14.3 ± 6.03 | 0.99 (0.96–1.03) | ||||
| Ever injected drugs | 0.048 | 0.008 | ||||
| No | 29 (49) | 30 (51) | Reference | Reference | ||
| Yes | 193 (63) | 114 (37) | 0.57 (0.33–1.00) | 0.44 (0.24–0.81) | ||
| Ever shared needles | 0.336 | |||||
| No | 160 (62) | 97 (38) | Reference | |||
| Yes | 62 (57) | 47 (43) | 1.25 (0.79–1.97) | |||
| Abusing drugs in the past month | 0.397 | |||||
| No | 194 (56) | 130 (40) | Reference | |||
| Yes | 28 (67) | 14 (33) | 0.75 (0.38–1.47) | |||
| More than one sexual partner in the past month | 0.343 | |||||
| No | 213 (61) | 135 (39) | Reference | |||
| Yes | 9 (50) | 9 (50) | 1.58 (0.61–4.07) | |||
| Enabling factors | ||||||
| Education level | 0.386 | |||||
| ≤Primary school | 56 (66) | 29 (34) | Reference | |||
| Junior high school | 124 (61) | 81 (39) | 1.26 (0.74–2.14) | |||
| ≥Senior high school | 42 (55) | 34 (45) | 1.56 (0.83–2.96) | |||
| Employment | 0.618 | |||||
| Unemployed | 102 (59) | 70 (41) | Reference | |||
| Employed | 120 (62) | 74 (38) | 0.90 (0.59–1.37) | |||
| Monthly income | 0.436 | |||||
| <3000 | 132 (61) | 85 (39) | Reference | |||
| 3000–5000 | 71 (64) | 40 (36) | 0.88 (0.55–1.41) | |||
| ≥5000 | 17 (51) | 16 (49) | 1.46 (0.70–3.05) | |||
| Have health insurance | 0.616 | |||||
| Yes | 163 (60) | 109 (40) | Reference | |||
| No | 56 (63) | 33 (37) | 0.88 (0.54–1.44) | |||
| Have stable residence | 0.074 | 0.049 | ||||
| Yes | 181 (59) | 127 (41) | Reference | Reference | ||
| No | 40 (71) | 16 (29) | 0.57 (0.31–1.06) | 0.26 (0.25–0.99) | ||
| Need factors | ||||||
| Drinking times in the past month | 0.484 | |||||
| Never | 160 (62) | 98 (38) | Reference | |||
| 1–3 times per month | 36 (63) | 21 (37) | 0.95 (0.52–1.73) | |||
| ≥1 times per week | 13 (52) | 12 (48) | 1.51 (0.66–3.44) | |||
| ≥1 times per day | 13 (52) | 12 (48) | 1.51 (0.66–3.44) | |||
| HIV | 0.001 | 0.001 | ||||
| No | 201 (64) | 112 (36) | Reference | Reference | ||
| Yes | 21 (40) | 32 (60) | 2.73 (1.51–4.97) | 2.83 (1.50–5.33) | ||
| HBV | 0.015 | 0.024 | ||||
| No | 209 (63) | 125 (37) | Reference | Reference | ||
| Yes | 13 (41) | 19 (59) | 2.44 (1.17–5.12) | 2.46 (1.12–5.39) | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Zou, X.; Chen, W.; Gong, C.; Ling, L. Hepatitis C Virus Treatment Status and Barriers among Patients in Methadone Maintenance Treatment Clinics in Guangdong Province, China: A Cross-Sectional, Observational Study. Int. J. Environ. Res. Public Health 2019, 16, 4436. https://doi.org/10.3390/ijerph16224436
Liu Y, Zou X, Chen W, Gong C, Ling L. Hepatitis C Virus Treatment Status and Barriers among Patients in Methadone Maintenance Treatment Clinics in Guangdong Province, China: A Cross-Sectional, Observational Study. International Journal of Environmental Research and Public Health. 2019; 16(22):4436. https://doi.org/10.3390/ijerph16224436
Chicago/Turabian StyleLiu, Yin, Xia Zou, Wen Chen, Cheng Gong, and Li Ling. 2019. "Hepatitis C Virus Treatment Status and Barriers among Patients in Methadone Maintenance Treatment Clinics in Guangdong Province, China: A Cross-Sectional, Observational Study" International Journal of Environmental Research and Public Health 16, no. 22: 4436. https://doi.org/10.3390/ijerph16224436





