Distribution, Sources and Water Quality Evaluation of the Riverine Solutes: A Case Study in the Lancangjiang River Basin, Tibetan Plateau
Abstract
:1. Introduction
2. Materials and Methods
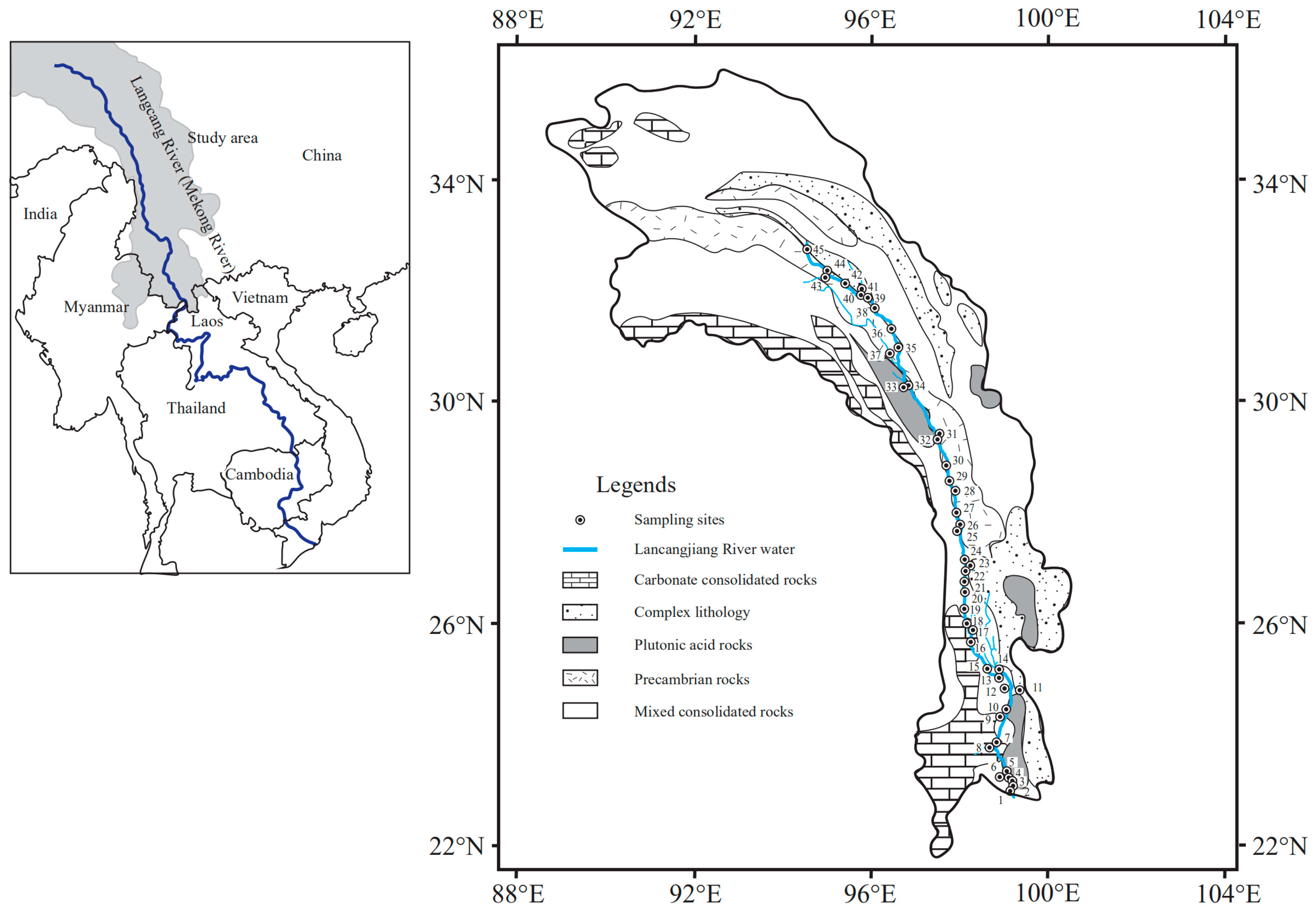
2.1. Study Area Description and Sample Collection
2.2. Sample Collection and Analysis
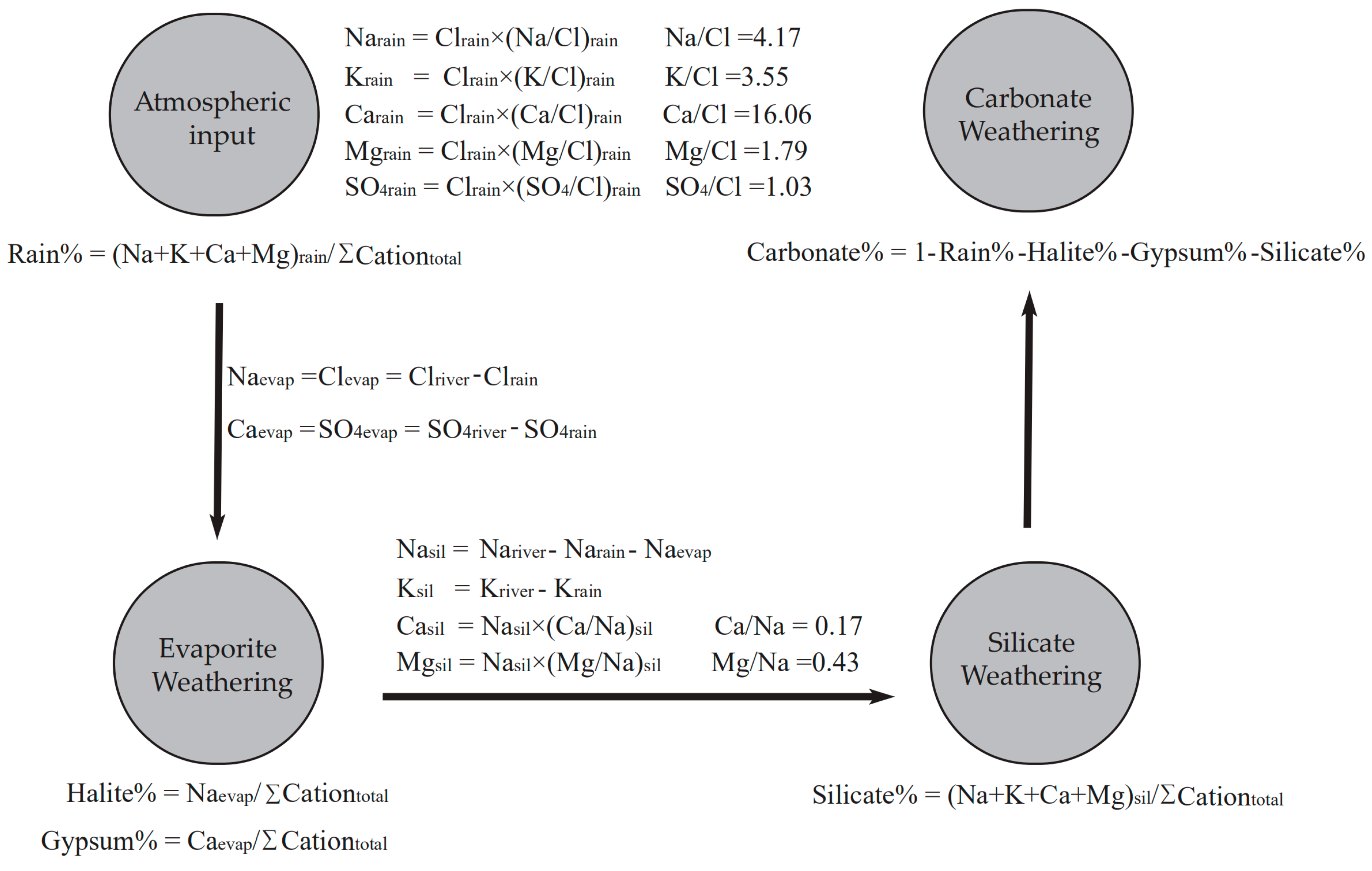
2.3. Mass Balance Model Calculation
2.4. Water Quality Evaluation
3. Results and Discussion
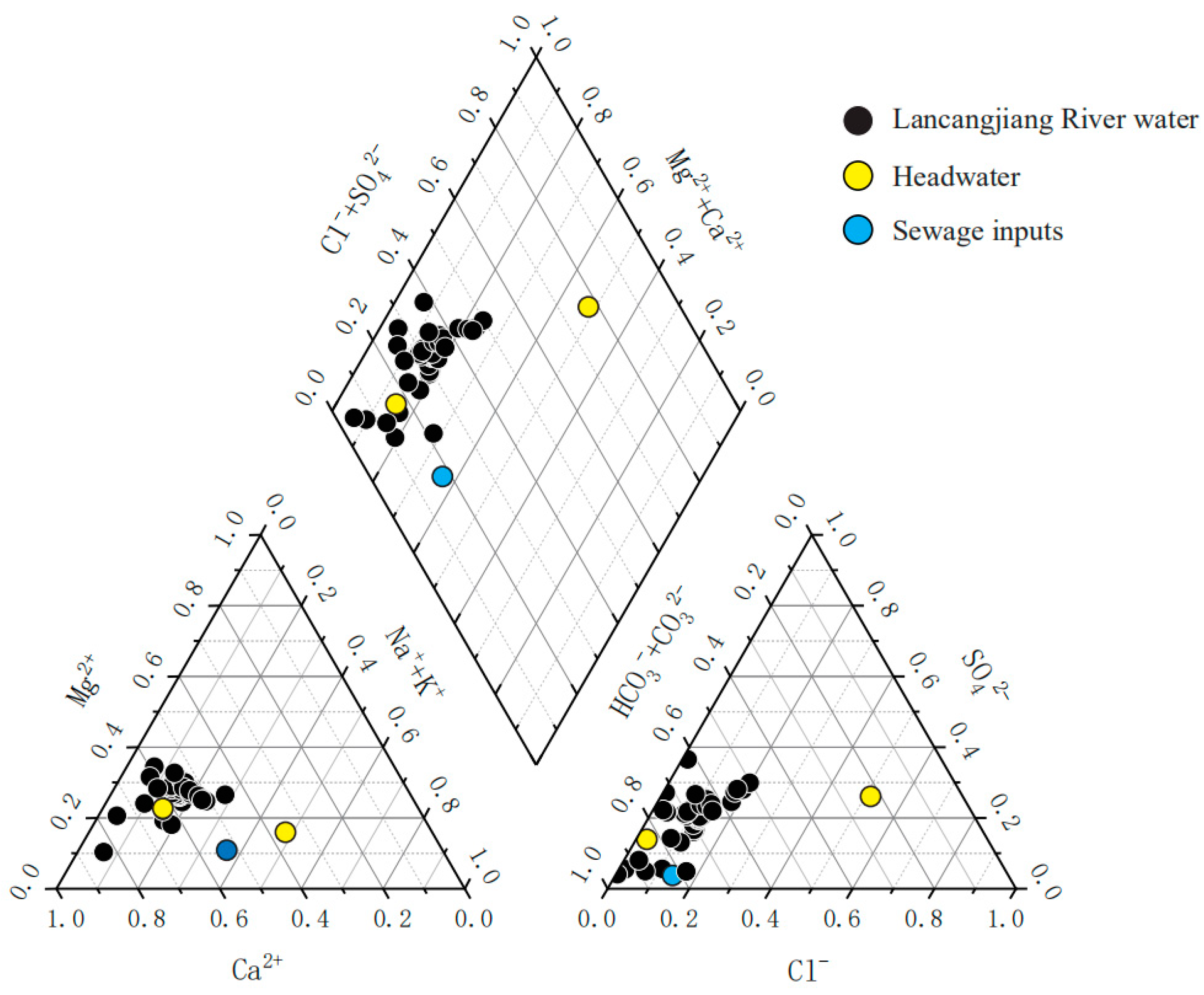
3.1. Hydrochemical Characteristics of the River Water
3.2. Relationship between the Major Ions
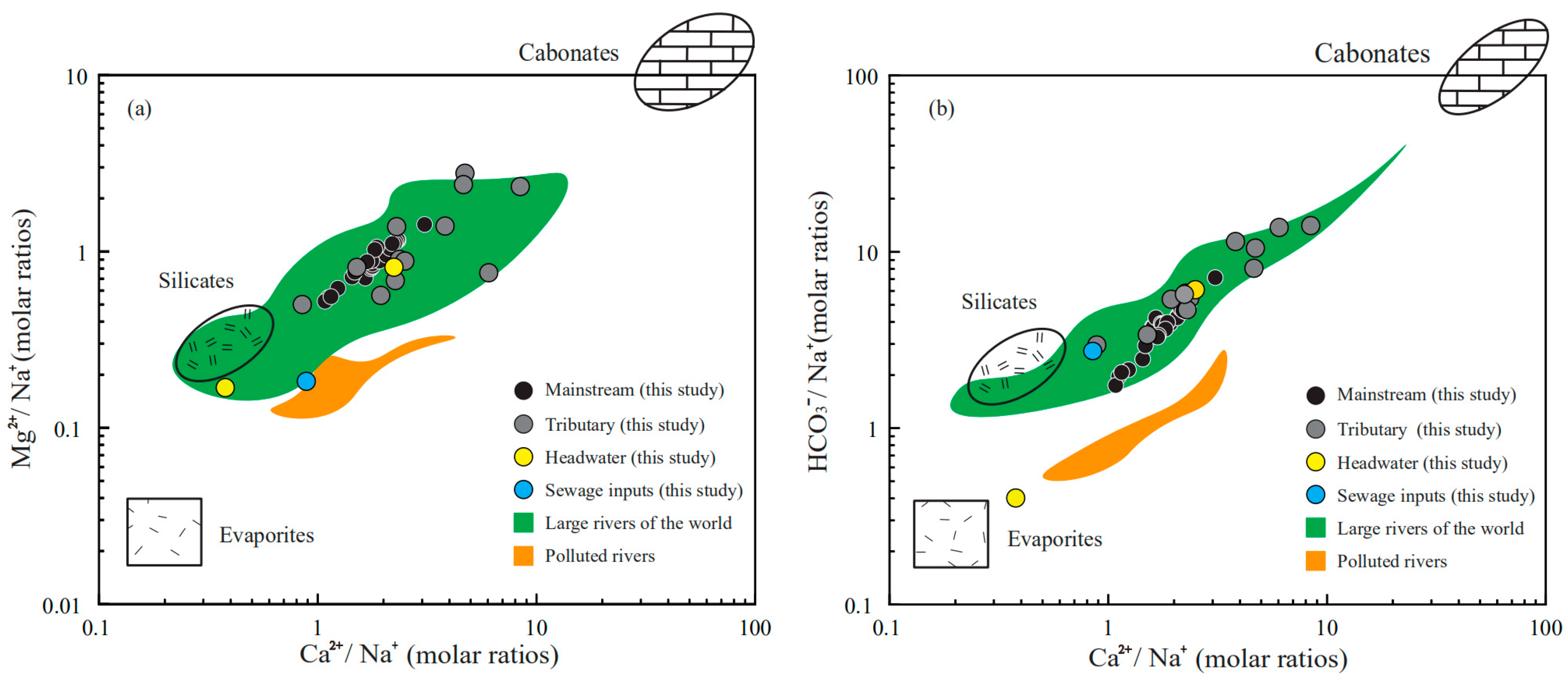
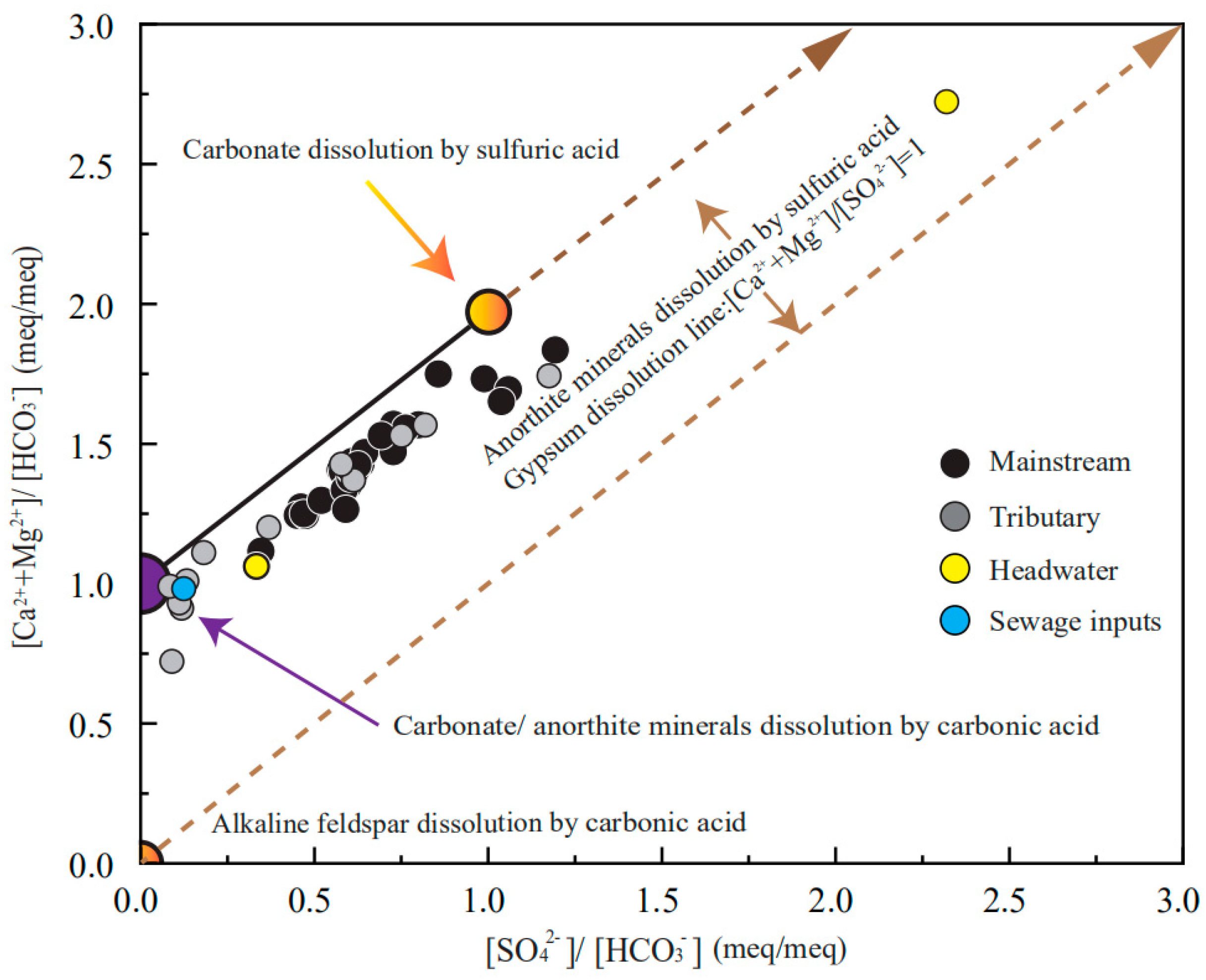
3.3. Stoichiometry of Weathering Processes
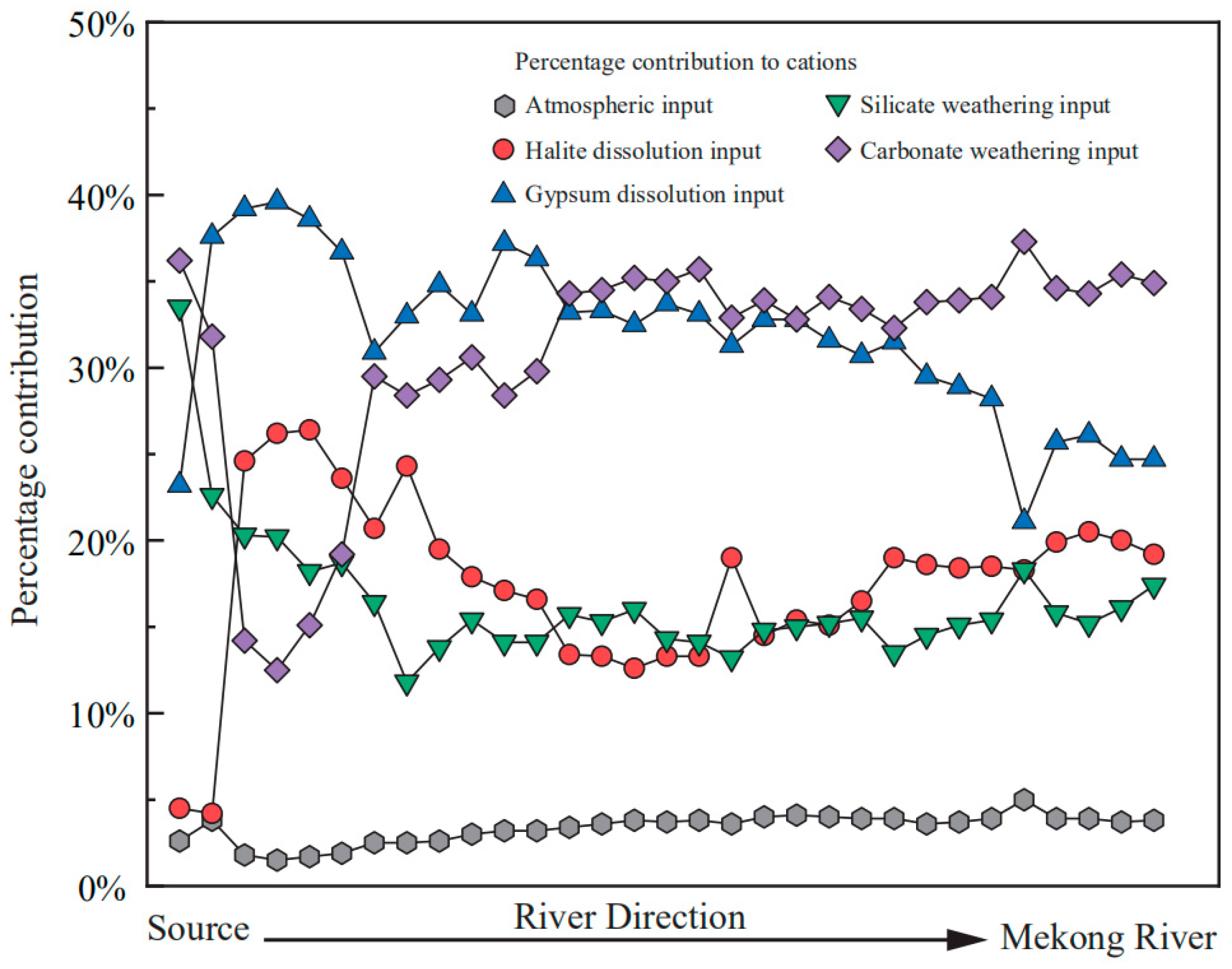
3.4. The Mixing Proportions
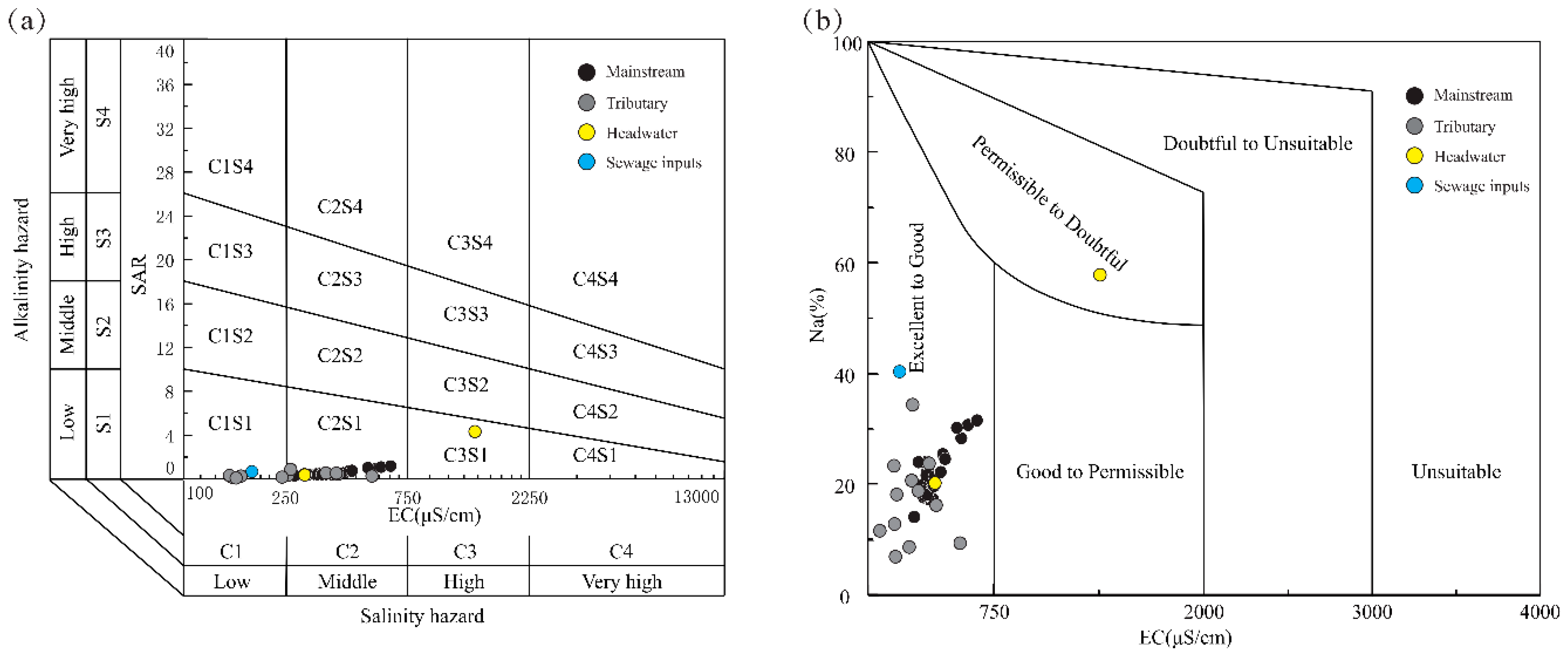
3.5. Drinking and Irrigation Water Quality Assessments
4. Conclusions
- (1)
- The river water chemistry is controlled by mineral weathering rather than anthropogenic inputs. The concentration order of the anions is HCO3− > SO42− > Cl−, while the order of cations is Ca2+ > Mg2+ > Na+ + K+. Most samples are of the HCO3−-Ca·Mg type, the river waters are slightly alkaline, and water temperatures decrease as the elevation increases.
- (2)
- The employment of the elemental ratios indicates the mixing relationships between different rocks. The chemical composition of the river water is the mixture of carbonate weathering inputs, evaporite dissolution input and silicate weathering input. In the source area of Lancangjiang River, the complex lithology lead to a relatively large difference in the chemical composition of each tributary.
- (3)
- The forward method indicates that on the watershed scale, the contributions of the atmospheric inputs are limited (<5%), the gypsum dissolution and carbonate minerals dissolution make up ~60% of the total cation budgets (34.9% for carbonate weathering, 24.7% for gypsum dissolution, respectively), the silicate weathering constitutes 17.4% while the halite dissolution makes up 19.2% of the total cations. This result reflects the dissolution rates of different minerals and lithologic distributions, the predominance of the Ca2+ and HCO3− in river water results from both the wide distribution, and the fast dissolution rate of carbonate minerals
- (4)
- The water quality is generally suitable for irrigation and drinking purposes in terms of major ions. In the upper reaches of Lancangjiang River, the dissolution of gypsum and halite transport abundant Na+ and SO42− into river waters, and these contents exceed the recommended values, which may cause health and soil problems, such as soil compaction and salinization. This result suggests even in the pristine areas with little human disturbance, the water quality requires monitoring and assessment due to chemical weathering processes.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Meybeck, M. Global geochemical weathering of surficial rocks estimates from river dissolved loads. Am. J. Sci. 1987, 287, 401–428. [Google Scholar] [CrossRef]
- Suchet, P.A.; Probst, J.L.; Ludwig, W. Worldwide distribution of continental rock lithology: Implicationsfor the atmospheric/soil CO2 uptake for continental weathering and alkalinity river transport to the oceans. Glob. Biogeochem. Cycles 2003, 17. [Google Scholar] [CrossRef]
- Han, G.L.; Song, Z.L.; Tang, Y.; Wu, Q.X.; Wang, Z.R. Ca and Sr isotope compositions of rainwater from Guiyang city, Southwest China: Implication for the sources of atmospheric aerosols and their seasonal variations. Atmos. Environ. 2019, 214. [Google Scholar] [CrossRef]
- Ahearn, D.S.; Sheibley, R.W.; Dahlgren, R.A.; Anderson, M.; Johnson, J.; Tate, K.W. Land use and land cover influence on water quality in the last free-flowing river draining the western Sierra Nevada, California. J. Hydrol. 2005, 313, 234–247. [Google Scholar] [CrossRef]
- Liu, J.; Han, G.; Liu, X.; Liu, M.; Song, C.; Zhang, Q.; Yang, K.; Li, X. Impacts of Anthropogenic Changes on the Mun River Water: Insight from Spatio-Distributions and Relationship of C and N Species in Northeast Thailand. Int. J. Environ. Res. Public Health 2019, 16, 659. [Google Scholar] [CrossRef]
- Liu, J.; Han, G.; Liu, X.; Liu, M.; Song, C.; Yang, K.; Li, X.; Zhang, Q. Distributive Characteristics of Riverine Nutrients in the Mun River, Northeast Thailand: Implications for Anthropogenic Inputs. Water 2019, 11, 954. [Google Scholar] [CrossRef]
- Gaillardet, J.; Dupre, B.; Louvat, P.; Allegre, C.J. Global silicate weathering and CO2 consumption rates deduced from the chemistry of large rivers. Chem. Geol. 1999, 159, 3–30. [Google Scholar] [CrossRef]
- Moon, S.; Huh, Y.; Qin, J.; van Pho, N. Chemical weathering in the Hong (Red) River basin: Rates of silicate weathering and their controlling factors. Geochim. Cosmochim. Acta 2007, 71, 1411–1430. [Google Scholar] [CrossRef]
- Liu, J.K.; Han, G.L.; Liu, X.L.; Yang, K.H.; Li, X.Q.; Liu, M. Examining the Distribution and Variation of Dissolved Carbon Species and Seasonal Carbon Exports within the Jiulongjiang River Basin (Southeast China). J. Coast. Res. 2019, 35, 784–793. [Google Scholar] [CrossRef]
- Liu, Z.; Macpherson, G.L.; Groves, C.; Martin, J.B.; Yuan, D.; Zeng, S. Large and active CO2 uptake by coupled carbonate weathering. Earth-Sci. Rev. 2018, 182, 42–49. [Google Scholar] [CrossRef]
- Galy, A.; France-Lanord, C. Weathering processes in the Ganges-Brahmaputra basin and the riverine alkalinity budget. Chem. Geol. 1999, 159, 31–60. [Google Scholar] [CrossRef]
- Chetelat, B.; Liu, C.Q.; Zhao, Z.Q.; Wang, Q.L.; Li, S.L.; Li, J.; Wang, B.L. Geochemistry of the dissolved load of the Changjiang Basin rivers: Anthropogenic impacts and chemical weathering. Geochim. Cosmochim. Acta 2008, 72, 4254–4277. [Google Scholar] [CrossRef]
- Noh, H.; Huh, Y.; Qin, J.; Ellis, A. Chemical weathering in the Three Rivers region of Eastern Tibet. Geochim. Cosmochim. Acta 2009, 73, 1857–1877. [Google Scholar] [CrossRef]
- Li, P.; Wu, J. Drinking Water Quality and Public Health. Expo. Health 2019, 11, 73–79. [Google Scholar] [CrossRef]
- Wang, J.; Liu, G.; Liu, H.; Lam, P.K.S. Multivariate statistical evaluation of dissolved trace elements and a water quality assessment in the middle reaches of Huaihe River, Anhui, China. Sci. Total Environ. 2017, 583, 421–431. [Google Scholar] [CrossRef]
- Jaiswal, D.; Pandey, J. An ecological response index for simultaneous prediction of eutrophication and metal pollution in large rivers. Water Res. 2019, 161, 423–438. [Google Scholar] [CrossRef]
- Zhao, M.M.; Chen, Y.P.; Xue, L.G.; Fan, T.T.; Emaneghemi, B. Greater health risk in wet season than in dry season in the Yellow River of the Lanzhou region. Sci. Total Environ. 2018, 644, 873–883. [Google Scholar] [CrossRef]
- Zaric, N.M.; Deljanin, I.; Ilijevic, K.; Stanisavljevic, L.; Ristic, M.; Grzetic, I. Assessment of spatial and temporal variations in trace element concentrations using honeybees (Apis mellifera) as bioindicators. PeerJ 2018, 6. [Google Scholar] [CrossRef]
- Liu, M.; Han, G.; Zhang, Q. Effects of agricultural abandonment on soil aggregation, soil organic carbon storage and stabilization: Results from observation in a small karst catchment, Southwest China. Agric. Ecosyst. Environ. 2019, 288, 106719. [Google Scholar] [CrossRef]
- Li, P.; Tian, R.; Liu, R. Solute Geochemistry and Multivariate Analysis of Water Quality in the Guohua Phosphorite Mine, Guizhou Province, China. Expo. Health 2018, 11, 81–94. [Google Scholar] [CrossRef]
- Santacruz de León, G.; Ramos Leal, J.A.; Moran Ramirez, J.; Lopez Alvarez, B.; Santacruz De Leon, E.E. Quality indices of groundwater for agricultural use in the Soconusco, Chiapas, Mexico. Earth Sci. Res. J. 2017, 21, 117–127. [Google Scholar] [CrossRef]
- Wu, J.H.; Wang, L.; Wang, S.T.; Tian, R.; Xue, C.Y.; Feng, W.; Li, Y.H. Spatiotemporal variation of groundwater quality in an arid area experiencing long-term paper wastewater irrigation, northwest China. Environ. Earth Sci. 2017, 76. [Google Scholar] [CrossRef]
- Li, P.Y.; Wu, J.H.; Qian, H. Hydrochemical appraisal of groundwater quality for drinking and irrigation purposes and the major influencing factors: A case study in and around Hua County, China. Arab. J. Geosci. 2016, 9. [Google Scholar] [CrossRef]
- Zeng, J.; Yue, F.J.; Wang, Z.J.; Wu, Q.; Qin, C.Q.; Li, S.L. Quantifying depression trapping effect on rainwater chemical composition during the rainy season in karst agricultural area, southwestern China. Atmos. Environ. 2019, 218, 116998. [Google Scholar] [CrossRef]
- Xu, Z.; Liu, C.-Q. Chemical weathering in the upper reaches of Xijiang River draining the Yunnan–Guizhou Plateau, Southwest China. Chem. Geol. 2007, 239, 83–95. [Google Scholar] [CrossRef]
- Han., G.; Tang., Y.; Wu., Q.; Liu., M.; Wang, Z. Assessing Contamination Sources by Using Sulfur and Oxygen Isotopes of Sulfate Ions in Xijiang River Basin, Southwest China. J. Environ. Qual. 2019, 48. [Google Scholar] [CrossRef]
- Huang, X.; Sillanpaa, M.; Gjessing, E.T.; Vogt, R.D. Water quality in the Tibetan Plateau: Major ions and trace elements in the headwaters of four major Asian rivers. Sci. Total Environ. 2009, 407, 6242–6254. [Google Scholar] [CrossRef]
- Zhang, L.-L.; Zhao, Z.-Q.; Zhang, W.; Tao, Z.-H.; Huang, L.; Yang, J.-X.; Wu, Q.-X.; Liu, C.-Q. Characteristics of water chemistry and its indication of chemical weathering in Jinshajiang, Lancangjiang and Nujiang drainage basins. Environ. Earth Sci. 2016, 75, 506. [Google Scholar] [CrossRef]
- Wu, W.; Xu, S.; Yang, J.; Yin, H. Silicate weathering and CO2 consumption deduced from the seven Chinese rivers originating in the Qinghai-Tibet Plateau. Chem. Geol. 2008, 249, 307–320. [Google Scholar] [CrossRef]
- Qin, J.; Huh, Y.; Edmond, J.M.; Du, G.; Ran, J. Chemical and physical weathering in the Min Jiang, a headwater tributary of the Yangtze River. Chem. Geol. 2006, 227, 53–69. [Google Scholar] [CrossRef]
- Jiang, L.G.; Yao, Z.J.; Wang, R.; Liu, Z.F.; Wang, L.; Wu, S.S. Hydrochemistry of the middle and upper reaches of the Yarlung Tsangpo River system: Weathering processes and CO2 consumption. Environ. Earth Sci. 2015, 74, 2369–2379. [Google Scholar] [CrossRef]
- Raymo, M.E.; Ruddiman, W.F. Tectonic Forcing of Late Cenozoic Climate. Nature 1992, 359, 117–122. [Google Scholar] [CrossRef]
- Hallet, B.; Molnar, P. Distorted drainage basins as markers of crustal strain east of the Himalaya. J. Geophys. Res.-Sol. Earth 2001, 106, 13697–13709. [Google Scholar] [CrossRef]
- Wang, X.F.; Metcalfe, I.; Jian, P.; He, L.Q.; Wang, C.S. The Jinshajiang-Ailaoshan Suture Zone, China: Tectonostratigraphy, age and evolution. J. Asian Earth Sci. 2000, 18, 675–690. [Google Scholar] [CrossRef]
- Durr, H.H.; Meybeck, M.; Durr, S.H. Lithologic composition of the Earth’s continental surfaces derived from a new digital map emphasizing riverine material transfer. Glob. Biogeochem. Cycles 2005, 19. [Google Scholar] [CrossRef]
- CO2SYS, Program. Available online: https://ecology.wa.gov/Research-Data/Data-resources/Models-spreadsheets/ Modeling -the-environment/Models-tools-for-TMDLs (accessed on 23 November 2019).
- Zhang, D.D.; Peart, M.; Jim, C.Y.; He, Y.Q.; Li, B.S.; Chen, J.A. Precipitation chemistry of Lhasa and other remote towns, Tibet. Atmos. Environ. 2003, 37, 231–240. [Google Scholar] [CrossRef] [Green Version]
- Stumm, W.; Morgan, J.J. Aquatic Chemistry: An Introduction Emphasizing Chemical Equilibria in Natural Waters; Wiley: New York, NY, USA, 1981. [Google Scholar]
- Barnes, R.T.; Raymond, P.A. The contribution of agricultural and urban activities to inorganic carbon fluxes within temperate watersheds. Chem. Geol. 2009, 266, 318–327. [Google Scholar] [CrossRef]
- Roy, S.; Gaillardet, J.; Allegre, C.J. Geochemistry of dissolved and suspended loads of the Seine river, France: Anthropogenic impact, carbonate and silicate weathering. Geochim. Cosmochim. Acta 1999, 63, 1277–1292. [Google Scholar] [CrossRef]
- Ryu, J.-S.; Lee, K.-S.; Chang, H.-W.; Shin, H.S. Chemical weathering of carbonates and silicates in the Han River basin, South Korea. Chem. Geol. 2008, 247, 66–80. [Google Scholar] [CrossRef]
- Li, S.-L.; Calmels, D.; Han, G.; Gaillardet, J.; Liu, C.-Q. Sulfuric acid as an agent of carbonate weathering constrained by δ13CDIC: Examples from Southwest China. Earth Planet. Sci. Lett. 2008, 270, 189–199. [Google Scholar] [CrossRef]
- Li, S.-L.; Chetelat, B.; Yue, F.; Zhao, Z.; Liu, C.-Q. Chemical weathering processes in the Yalong River draining the eastern Tibetan Plateau, China. J. Asian Earth Sci. 2014, 88, 74–84. [Google Scholar] [CrossRef]
- Brantley, S. Kinetics of Mineral Dissolution. In Kinetics of Water-Rock Interaction; Brantley, S., Kubicki, J., White, A., Eds.; Springer: New York, NY, USA, 2008. [Google Scholar]
- Bluth, G.J.S.; Kump, L.R. Lithologic and Climatologic Controls of River Chemistry. Geochim. Cosmochim. Acta 1994, 58, 2341–2359. [Google Scholar] [CrossRef]
- White, A.F.; Bullen, T.D.; Vivit, D.V.; Schulz, M.S.; Clow, D.W. The role of disseminated calcite in the chemical weathering of granitoid rocks. Geochim. Cosmochim. Acta 1999, 63, 1939–1953. [Google Scholar] [CrossRef]
- Welch, S.A.; Ullman, W.J. The temperature dependence of bytownite feldspar dissolution in neutral aqueous solutions of inorganic and organic ligands at low temperature (5–35 °C). Chem. Geol. 2000, 167, 337–354. [Google Scholar] [CrossRef]
- Nightingale, M.; Mayer, B. Identifying sources and processes controlling the sulphur cycle in the Canyon Creek watershed, Alberta, Canada. Isot. Environ. Heath Stud. 2012, 48, 89–104. [Google Scholar] [CrossRef]
- Han, G.; Liu, C.-Q. Water geochemistry controlled by carbonate dissolution: A study of the river waters draining karst-dominated terrain, Guizhou Province, China. Chem. Geol. 2004, 204, 1–21. [Google Scholar] [CrossRef]
- China EPA. Environmental Quality Standards for Drinking Water Quality (GB 5749-2006). 2006. Available online: http://www.nhfpc.gov.cn/wjw/pgw/201212/33644.shtml (accessed on 1 July 2007).
- WHO. Guidelines for Drinking-Water Quality: Fourth Edition Incorporating the First Addendum; World Health Organization: Geneva, Switzerland, 2017; 631p. [Google Scholar]
- Li, P.Y.; Wu, J.H.; Qian, H. Assessment of groundwater quality for irrigation purposes and identification of hydro-geochemical evolution mechanisms in Pengyang County, China. Environ. Earth Sci. 2013, 69, 2211–2225. [Google Scholar] [CrossRef]
| Parameters | DIC | F− | Cl− | NO3− | SO42− | Ca2+ | K+ | Mg2+ | Na+ | EC |
|---|---|---|---|---|---|---|---|---|---|---|
| DIC | 1 | |||||||||
| F− | 0.643 ** | 1 | ||||||||
| Cl− | 0.349 * | 0.306 * | 1 | |||||||
| NO3− | 0.147 | 0.175 | 0.142 | 1 | ||||||
| SO42− | 0.664 ** | 0.621 *** | 0.817 ** | −0.006 | 1 | |||||
| Ca2+ | 0.839 ** | 0.731 ** | 0.731 ** | 0.055 | 0.958 ** | 1 | ||||
| K+ | 0.337 * | 0.391 ** | 0.693 ** | 0.683 ** | 0.531 ** | 0.507 ** | 1 | |||
| Mg2+ | 0.805 ** | 0.657 ** | 0.651 ** | 0.087 | 0.939 ** | 0.959 ** | 0.414 ** | 1 | ||
| Na+ | 0.428 ** | 0.365 * | 0.995 ** | 0.172 | 0.846 ** | 0.780 ** | 0.711 ** | 0.694 ** | 1 | |
| EC | 0.669 ** | 0.552 ** | 0.909 ** | 0.105 | 0.967 ** | 0.946 ** | 0.643 ** | 0.894 ** | 0.936 ** | 1 |
| Parameters | Min | Max | Mean | SD | Chinese Guidelines | WHO Guideline |
|---|---|---|---|---|---|---|
| pH | 7.76 | 8.77 | 8.32 | 0.20 | 6.5–8.5 | 6.5–8.5 |
| TDS (mg/L) | 52.7 | 1313 | 286 | 189 | 1000 | 1000 |
| F− (mg/L) | 0.01 | 0.33 | 0.16 | 0.06 | 1 | 1.5 |
| Cl− (mg/L) | 0.13 | 331 | 22.8 | 48.1 | 250 | 250 |
| NO3−-N (mg/L) | 0.05 | 2.84 | 0.64 | 0.44 | 20 mg/L as N | 50 |
| SO42− (mg/L) | 3.37 | 455 | 88.3 | 77.4 | 250 | 250 |
| SAR | 0.08 | 4.31 | 0.58 | 0.61 | / | / |
| Na (%) | 7.10 | 57.9 | 22.1 | 8.4 | / | / |
| RSC | −7.04 | 0.39 | −1.18 | 1.22 | / | / |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.; Han, G.; Liu, M.; Zeng, J.; Liang, B.; Qu, R. Distribution, Sources and Water Quality Evaluation of the Riverine Solutes: A Case Study in the Lancangjiang River Basin, Tibetan Plateau. Int. J. Environ. Res. Public Health 2019, 16, 4670. https://doi.org/10.3390/ijerph16234670
Liu J, Han G, Liu M, Zeng J, Liang B, Qu R. Distribution, Sources and Water Quality Evaluation of the Riverine Solutes: A Case Study in the Lancangjiang River Basin, Tibetan Plateau. International Journal of Environmental Research and Public Health. 2019; 16(23):4670. https://doi.org/10.3390/ijerph16234670
Chicago/Turabian StyleLiu, Jinke, Guilin Han, Man Liu, Jie Zeng, Bin Liang, and Rui Qu. 2019. "Distribution, Sources and Water Quality Evaluation of the Riverine Solutes: A Case Study in the Lancangjiang River Basin, Tibetan Plateau" International Journal of Environmental Research and Public Health 16, no. 23: 4670. https://doi.org/10.3390/ijerph16234670
APA StyleLiu, J., Han, G., Liu, M., Zeng, J., Liang, B., & Qu, R. (2019). Distribution, Sources and Water Quality Evaluation of the Riverine Solutes: A Case Study in the Lancangjiang River Basin, Tibetan Plateau. International Journal of Environmental Research and Public Health, 16(23), 4670. https://doi.org/10.3390/ijerph16234670







