Effects of Chronic Exposure to Microcystin-LR on Kidney in Mice
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Animals and Experimental Design
2.3. Determination of Renal Function Indicators
2.4. Histological Observations
2.5. Statistical Analysis
3. Results
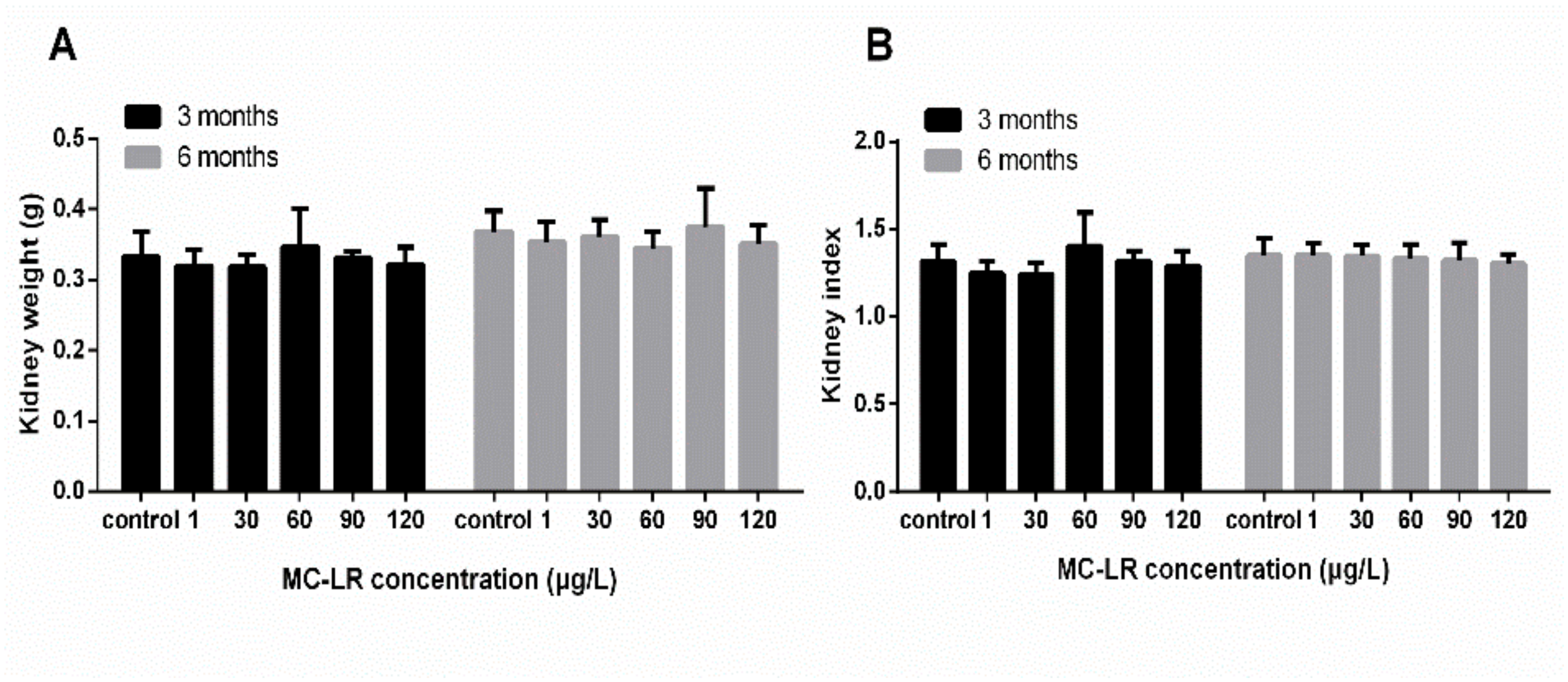
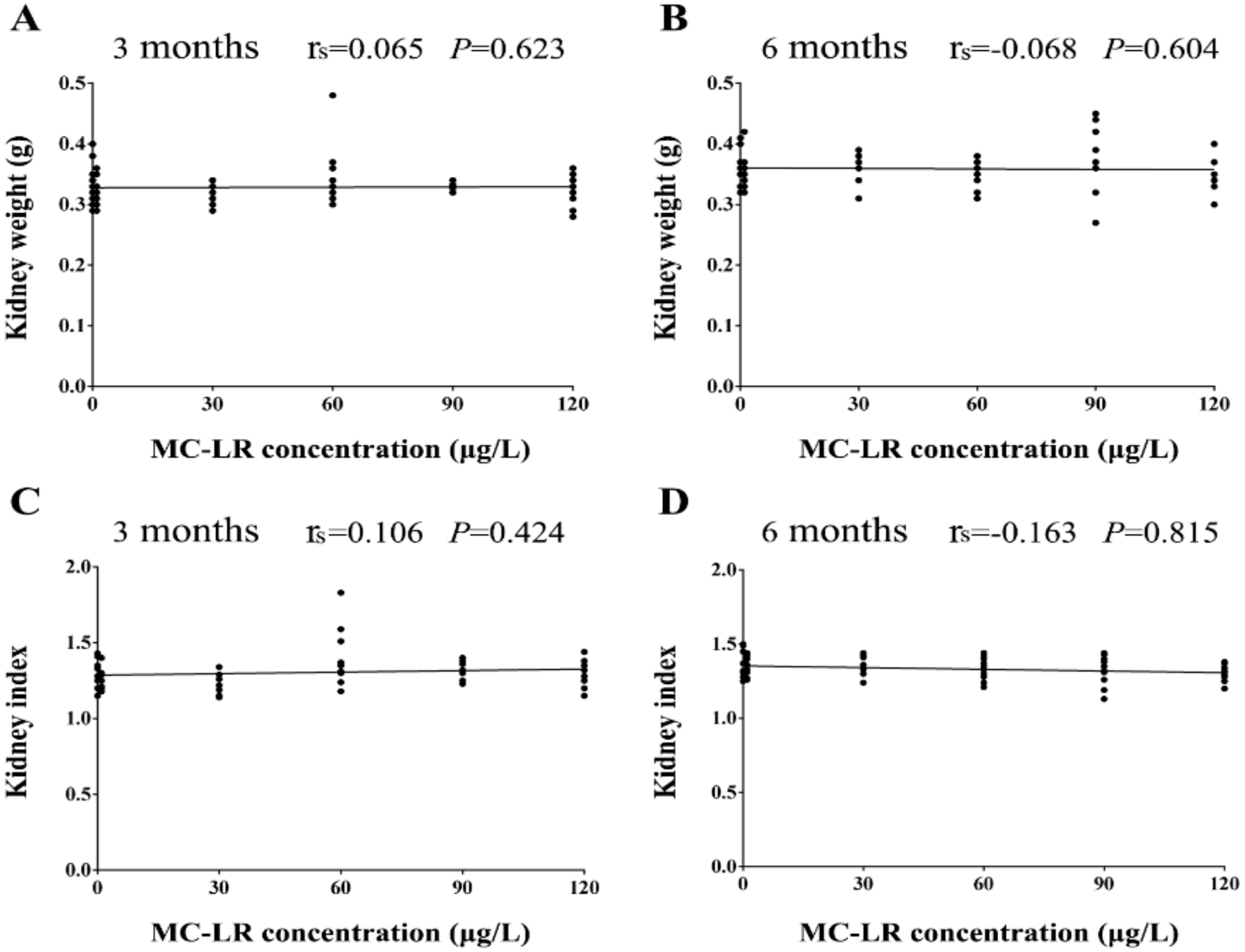
3.1. Body Weight and Kidney Index
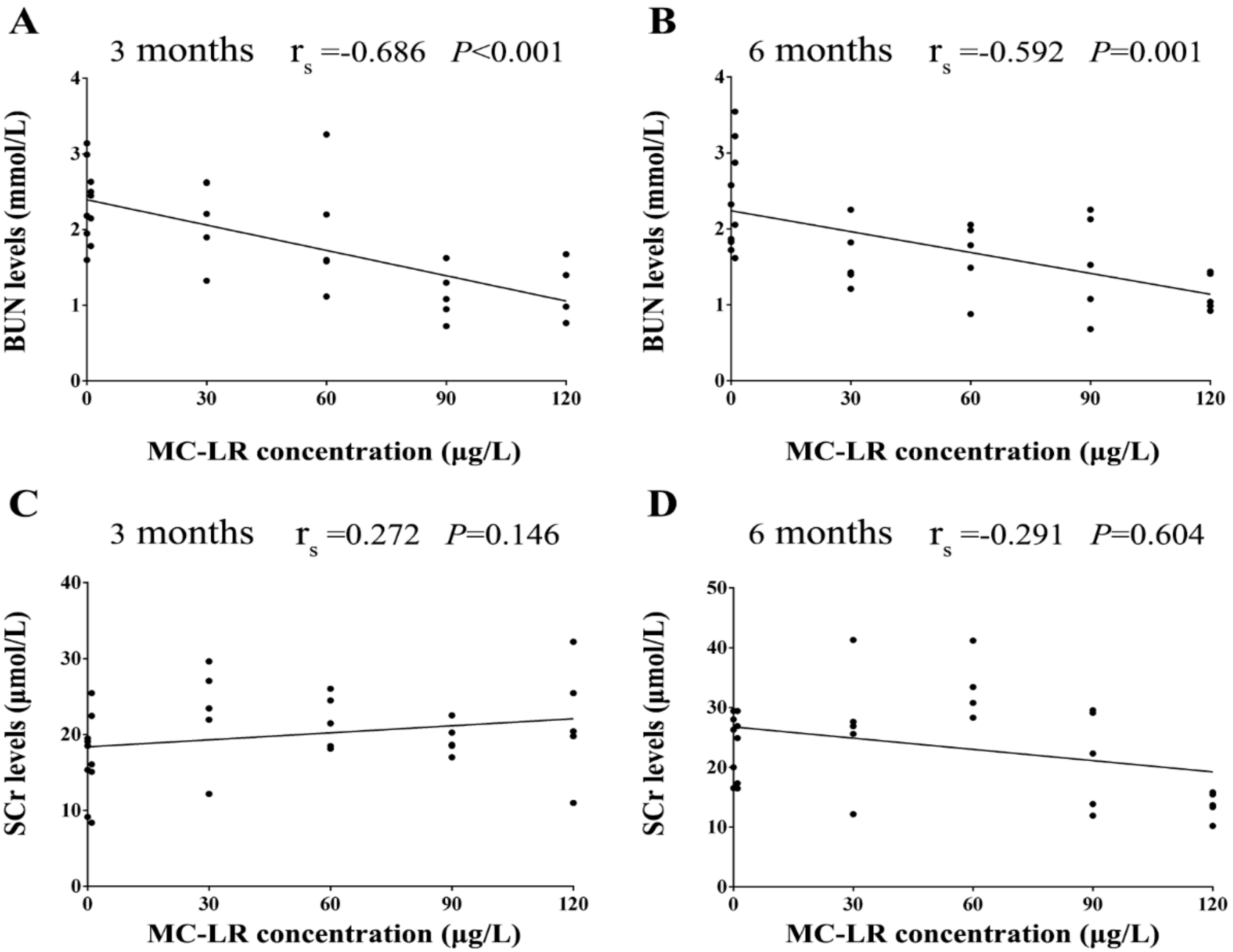
3.2. Renal Function Indicators
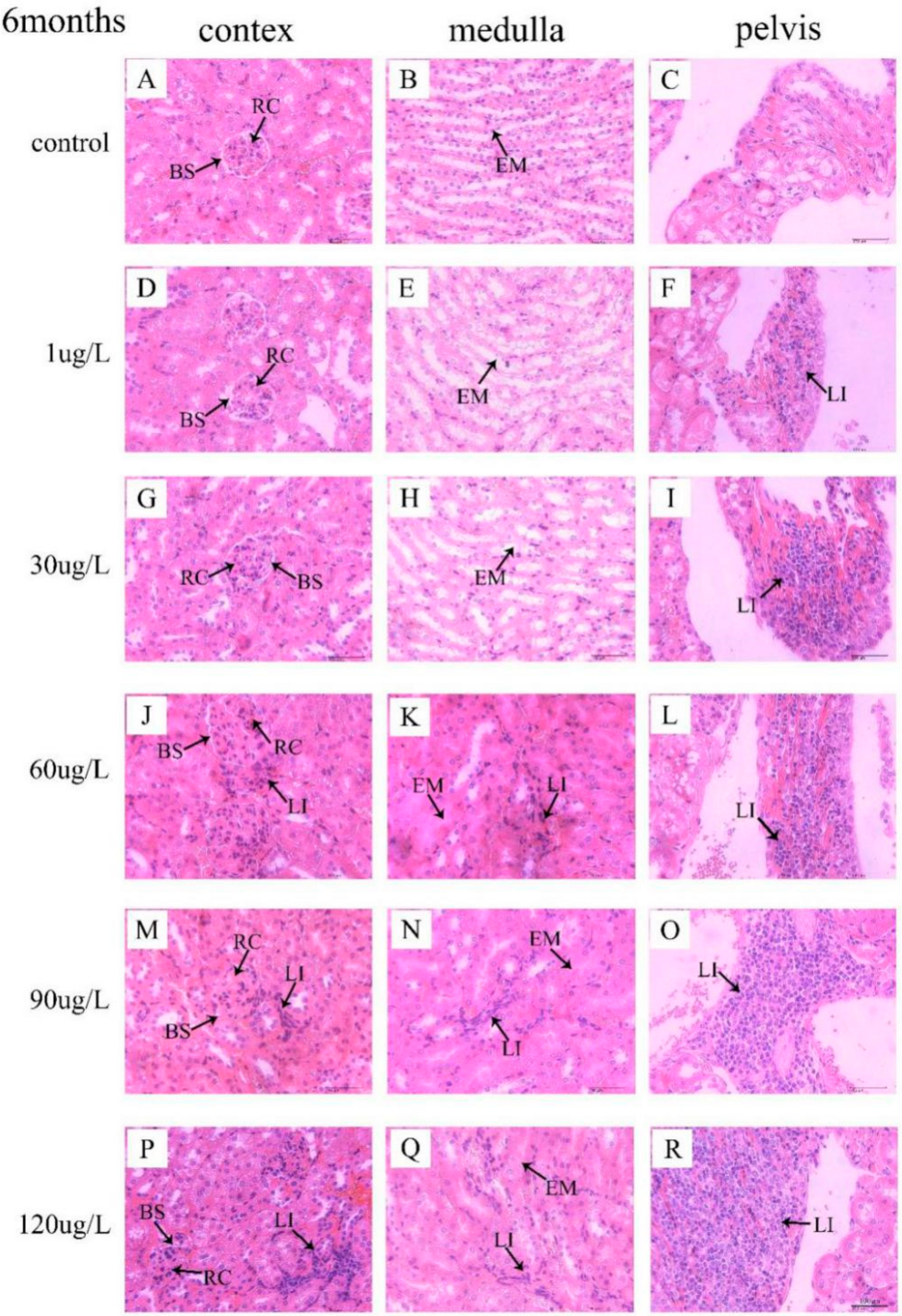
3.3. Histological Observation of Kidneys
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Svirčev, Z.; Drobac, D.; Tokodi, N.; Mijovic, B.; Codd, G.A.; Meriluoto, J. Toxicology of microcystins with reference to cases of human intoxications and epidemiological investigations of exposures to cyanobacteria and cyanotoxins. Arch. Toxicol. 2017, 91, 621–650. [Google Scholar] [CrossRef] [PubMed]
- Svirčev, Z.; Lujić, J.; Marinović, Z.; Drobac, D.; Tokodi, N.; Stojiljković, B.; Meriluoto, J. Toxicopathology Induced by Microcystins and Nodularin: A Histopathological Review. J. Environ. Sci. Health C Environ. Carcinog. Ecotoxicol. Rev. 2015, 33, 125–167. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Chen, L.; Wen, C.; Zhang, X.; Feng, X.; Yang, F. MicroRNA expression profiling involved in MC-LR-induced hepatotoxicity using high-throughput sequencing analysis. J. Toxicol. Environ. Health Part A 2017, 81, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Huang, F.; Massey, I.Y.; Wen, C.; Zheng, S.; Xu, S.; Yang, F. Effects of Microcystin-LR on the Microstructure and Inflammation-Related Factors of Jejunum in Mice. Toxins (Basel) 2019, 11, 482. [Google Scholar] [CrossRef] [Green Version]
- Massey, I.Y.; Yang, F.; Ding, Z.; Yang, S.; Guo, J.; Tezi, C.; Al-Osman, M.; Kamegni, R.B.; Zeng, W. Exposure routes and health effects of microcystins on animals and humans: A mini-review. Toxicon 2018, 151, 156–162. [Google Scholar] [CrossRef]
- Cao, L.; Massey, I.Y.; Feng, H.; Yang, F. A Review of Cardiovascular Toxicity of Microcystins. Toxins (Basel) 2019, 11, 507. [Google Scholar] [CrossRef] [Green Version]
- Wei, J.; Xie, X.; Huang, F.; Xiang, L.; Wang, Y.; Han, T.; Massey, I.Y.; Liang, G.; Pu, Y.; Yang, F. Simultaneous Microcystis algicidal and microcystin synthesis inhibition by a red pigment prodigiosin. Environ. Pollut. 2019, 113444. [Google Scholar] [CrossRef]
- Meriluoto, J.; Spoof, L.; Codd, G.A. Appendix 3: Tables of Microcystins and Nodularins; John Wiley & Sons: New York, NY, USA, 2017. [Google Scholar]
- Du, X.; Liu, H.; Yuan, L.; Wang, Y.; Ma, Y.; Wang, R.; Chen, X.; Losiewicz, M.D.; Guo, H.; Zhang, H. The Diversity of Cyanobacterial Toxins on Structural Characterization, Distribution and Identification: A Systematic Review. Toxins (Basel) 2019, 11, 530. [Google Scholar] [CrossRef] [Green Version]
- Fujiki, H.; Suganuma, M. Tumor promoters--microcystin-LR, nodularin and TNF-alpha and human cancer development. Anticancer Agents Med. Chem. 2011, 11, 4–18. [Google Scholar] [CrossRef]
- Hagenbuch, B.; Meier, P.J. The superfamily of organic anion transporting polypeptides. Biochim. Biophys. Acta 2003, 1609, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Clark, S.P.; Davis, M.A.; Ryan, T.P.; Searfoss, G.H.; Hooser, S.B. Hepatic Gene Expression Changes in Mice Associated with Prolonged Sublethal Microcystin Exposure. Toxicol. Pathol. 2016, 35, 594–605. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Su, Y.; Lin, W.; Guo, H.; Xie, P.; Chen, J.; Gu, Z.; Li, L. Microcystin-LR retards gonadal maturation through disrupting the growth hormone/insulin-like growth factors system in zebrafish. Ecotoxicol. Environ. Saf. 2017, 139, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Martins, N.D.; Colvara, W.A.; Rantin, F.T.; Kalinin, A.L. Microcystin-LR: How it affects the cardio-respiratory responses to hypoxia in Nile tilapia, Oreochromis niloticus. Chemosphere 2011, 84, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Gu, S.; Yin, X.; Yuan, M.; Xiang, Z.; Li, Z.; Cao, H.; Meng, X.; Hu, K.; Han, X. The toxic effects of microcystin-LR on mouse lungs and alveolar type II epithelial cells. Toxicon 2016, 115, 81–88. [Google Scholar] [CrossRef] [Green Version]
- Yan, W.; Li, L.; Li, G.; Zhao, S. Microcystin-LR induces changes in the GABA neurotransmitter system of zebrafish. Aquat. Toxicol. 2017, 188, 170–176. [Google Scholar] [CrossRef]
- Botha, N.; Venter, M.V.D.; Downing, T.G.; Shephard, E.G.; Gehringer, M.M. The effect of intraperitoneally administered microcystin-LR on the gastrointestinal tract of Balb/c mice. Toxicon 2004, 43, 251–254. [Google Scholar] [CrossRef]
- Jia, J.; Luo, W.; Lu, Y.; Giesy, J.P. Bioaccumulation of microcystins (MCs) in four fish species from Lake Taihu, China: Assessment of risks to humans. Sci. Total Environ. 2014, 487, 224–232. [Google Scholar] [CrossRef]
- Menezes, C.; Elsa, E.V.A. The Kidney Vero-E6 Cell Line: A Suitable Model to Study the Toxicity of Microcystins. Tech 2013, 29–48. [Google Scholar]
- Piyathilaka, M.A.; Pathmalal, M.M.; Tennekoon, K.H.; De Silva, B.G.; Samarakoon, S.R.; Chanthirika, S. Microcystin-LR-induced cytotoxicity and apoptosis in human embryonic kidney and human kidney adenocarcinoma cell lines. Microbiology 2015, 161, 819–828. [Google Scholar] [CrossRef]
- Kotak, B.G.; Semalulu, S.; Fritz, D.L.; Prepas, E.E.; Hrudey, S.E.; Coppock, R.W. Hepatic and renal pathology of intraperitoneally administered microcystin-LR in rainbow trout (Oncorhynchus mykiss). Toxicon 1996, 34, 517–525. [Google Scholar] [CrossRef]
- Atencio, L.; Moreno, I.; Jos, A.; Pichardo, S.; Moyano, R.; Blanco, A.; Camean, A.M. Dose-dependent antioxidant responses and pathological changes in tenca (Tinca tinca) after acute oral exposure to Microcystis under laboratory conditions. Toxicon 2008, 52, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Xie, P.; Lei, H.; Zhang, X. Renal accumulation and effects of intraperitoneal injection of extracted microcystins in omnivorous crucian carp (Carassius auratus). Toxicon 2013, 70, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Molina, R.; Moreno, I.; Pichardo, S.; Jos, A.; Moyano, R.; Monterde, J.G.; Camean, A. Acid and alkaline phosphatase activities and pathological changes induced in Tilapia fish (Oreochromis sp.) exposed subchronically to microcystins from toxic cyanobacterial blooms under laboratory conditions. Toxicon 2005, 46, 725–735. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Yang, L.; Zhang, X.; Li, Y.; Wang, J.; Zhang, S.; Liu, H.; Huang, H.; Wang, Y.; Yuan, L.; et al. MC-LR induces dysregulation of iron homeostasis by inhibiting hepcidin expression: A preliminary study. Chemosphere 2018, 212, 572–584. [Google Scholar] [CrossRef] [PubMed]
- Moreno, I.; Pichardo, S.; Jos, A.; Gomez-Amores, L.; Mate, A.; Vazquez, C.M.; Camean, A.M. Antioxidant enzyme activity and lipid peroxidation in liver and kidney of rats exposed to microcystin-LR administered intraperitoneally. Toxicon 2005, 45, 395–402. [Google Scholar] [CrossRef]
- Nobre, A.C.; Coelho, G.R.; Coutinho, M.C.; Silva, M.M.; Angelim, E.V.; Menezes, D.B.; Fonteles, M.C.; Monteiro, H.S. The role of phospholipase A(2) and cyclooxygenase in renal toxicity induced by microcystin-LR. Toxicon 2001, 39, 721–724. [Google Scholar] [CrossRef]
- Milutinovic, A.; Zivin, M.; Zorc-Pleskovic, R.; Sedmak, B.; Suput, D. Nephrotoxic effects of chronic administration of microcystins -LR and -YR. Toxicon 2003, 42, 281–288. [Google Scholar] [CrossRef]
- Florczyk, M.; Łakomiak, A.; Woźny, M.; Brzuzan, P. Neurotoxicity of cyanobacterial toxins. Environ. Biotechnol. 2014, 10, 26–43. [Google Scholar] [CrossRef] [Green Version]
- Lone, Y.; Koiri, R.K.; Bhide, M. An overview of the toxic effect of potential human carcinogen Microcystin-LR on testis. Toxicol. Rep. 2015, 2, 289–296. [Google Scholar] [CrossRef] [Green Version]
- Wu, J.X.; Huang, H.; Yang, L.; Zhang, X.F.; Zhang, S.S.; Liu, H.H.; Wang, Y.Q.; Yuan, L.; Cheng, X.M.; Zhuang, D.G.; et al. Gastrointestinal toxicity induced by microcystins. World J. Clin. Cases 2018, 6, 344–354. [Google Scholar] [CrossRef]
- Woolbright, B.L.; Williams, C.D.; Ni, H.; Kumer, S.C.; Schmitt, T.; Kane, B.; Jaeschke, H. Microcystin-LR induced liver injury in mice and in primary human hepatocytes is caused by oncotic necrosis. Toxicon 2017, 125, 99–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heinze, R. Toxicity of the cyanobacterial toxin microcystin-LR to rats after 28 days intake with the drinking water. Environ. Toxicol. 1999, 14, 57–60. [Google Scholar] [CrossRef]
- Ueno, Y.; Makita, Y.; Nagata, S.; Tsutsumi, T.; Yoshida, F.; Tamura, S.; Sekijima, M.; Tashiro, F.; Harada, T.; Yoshida, T. No chronic oral toxicity of a low dose of microcystin-LR, a cyanobacterial hepatotoxin, in female BALB/c mice. Environ. Toxicol. 1999, 14, 45–55. [Google Scholar] [CrossRef]
- Fawell, J.K.; Mitchell, R.E.; Everett, D.J.; Hill, R.E. The toxicity of cyanobacterial toxins in the mouse: I microcystin-LR. Hum. Exp. Toxicol. 1999, 18, 162–167. [Google Scholar] [CrossRef]
- Adamovsky, O.; Palikova, M.; Ondrackova, P.; Zikova, A.; Kopp, R.; Mares, J.; Pikula, J.; Paskerova, H.; Kohoutek, J.; Blaha, L. Biochemical and histopathological responses of Wistar rats to oral intake of microcystins and cyanobacterial biomass. Neuroendocrinol. Lett. 2013, 34 (Suppl. 2), 11–20. [Google Scholar]
- Xiangyan, L.; Fanyun, K.; Haiqing, Z.; Renxian, T.; Kuiyang, Z. The duplication and identification of anti-glomerular basement membrane (GBM) nephritis model in mice. Acta Acad. Med. Xuzhou 2011, 31, 527–530. [Google Scholar]
- Palikova, M.; Navratil, S.; Krejci, R.; Sterba, F.; Tichy, F.; Kubala, L.; Marsalek, B.; Blaha, L. Outcomesof repeated exposure of the carp (Cyprinus carpio L.) to cyanobacteria extract. ACTA Vet. Brno 2004, 73, 259–265. [Google Scholar] [CrossRef] [Green Version]
- Solter, P.; Liu, Z.L.; Guzman, R. Decreased hepatic ALT synthesis is an outcome of subchronic microcystin-LR toxicity. Toxicol. Appl. Pharmacol. 2000, 164, 216–220. [Google Scholar] [CrossRef]
- Milutinovic, A.; Zorc-Pleskovic, R.; Zivin, M.; Vovk, A.; Sersa, I.; Suput, D. Magnetic resonance imaging for rapid screening for the nephrotoxic and hepatotoxic effects of microcystins. Mar. Drugs 2013, 11, 2785–2798. [Google Scholar] [CrossRef]
- Lin, H.; Liu, W.; Zeng, H.; Pu, C.; Zhang, R.; Qiu, Z.; Chen, J.; Wang, L.; Tan, Y.; Zheng, C.; et al. Determination of Environmental Exposure to Microcystin and Aflatoxin as a Risk for Renal Function Based on 5493 Rural People in Southwest China. Environ. Sci. Technol. 2016, 50, 5346–5356. [Google Scholar] [CrossRef]
- Estrella, M.M.; Li, M.; Tin, A.; Abraham, A.G.; Shlipak, M.G.; Penugonda, S.; Hussain, S.K.; Palella, F.J.; Wolinsky, S.M.; Martinson, J.J.; et al. The Association Between APOL1 Risk Alleles and Longitudinal Kidney Function Differs by HIV Viral Suppression Status. Clin. Infect. Dis. 2015, 60, 646–652. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Levey, A.S.; Atkins, R.; Coresh, J.; Cohen, E.P.; Collins, A.J.; Eckardt, K.U.; Nahas, M.E.; Jaber, B.L.; Jadoul, M.; Levin, A.; et al. Chronic kidney disease as a global public health problem: Approaches and initiatives-a position statement from Kidney Disease Improving Global Outcomes. Kidney Int. 2007, 72, 247–259. [Google Scholar] [CrossRef] [PubMed] [Green Version]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yi, X.; Xu, S.; Huang, F.; Wen, C.; Zheng, S.; Feng, H.; Guo, J.; Chen, J.; Feng, X.; Yang, F. Effects of Chronic Exposure to Microcystin-LR on Kidney in Mice. Int. J. Environ. Res. Public Health 2019, 16, 5030. https://doi.org/10.3390/ijerph16245030
Yi X, Xu S, Huang F, Wen C, Zheng S, Feng H, Guo J, Chen J, Feng X, Yang F. Effects of Chronic Exposure to Microcystin-LR on Kidney in Mice. International Journal of Environmental Research and Public Health. 2019; 16(24):5030. https://doi.org/10.3390/ijerph16245030
Chicago/Turabian StyleYi, Xiping, Shuaishuai Xu, Feiyu Huang, Cong Wen, Shuilin Zheng, Hai Feng, Jian Guo, Jihua Chen, Xiangling Feng, and Fei Yang. 2019. "Effects of Chronic Exposure to Microcystin-LR on Kidney in Mice" International Journal of Environmental Research and Public Health 16, no. 24: 5030. https://doi.org/10.3390/ijerph16245030




