In Vivo Comparison of the Phenotypic Aspects and Molecular Mechanisms of Two Nephrotoxic Agents, Sodium Fluoride and Uranyl Nitrate
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Intraperitoneal Injection of Animals with U or F and Urine Collection
2.3. Euthanasia and Kidney Collection
2.4. U and F Measurements in Urine and Kidney Tissue
2.5. Urinary Biochemical Measurements
2.6. Histopathology and Immunostaining
2.7. KIM-1 and CLU Detection in Urine
2.8. Real-Time RT-PCR
2.9. Caspase 3/7 Activity
2.10. Statistical Analyses
3. Results
3.1. Dose-Dependent Increase of F and U in Urine
3.2. Traditional Nephrotoxicity Biomarkers
3.3. Both U and F Induce Structural Lesions in the Kidney
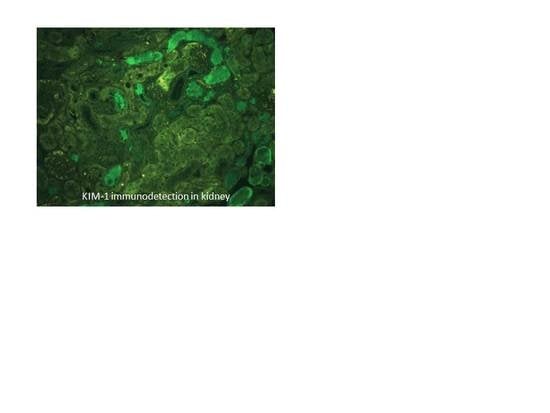
3.4. Biomarkers of Early Kidney Injury Increase after F and U Treatments
3.5. ICAM and VCAM Increase with F and U Treatments
3.6. Caspase 3/7 Activities in the Kidneys of Mice Treated with F or U
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Guéguen, Y.; Roy, L.; Hornhardt, S.; Badie, C.; Hall, J.; Baatout, S.; Pernot, E.; Tomasek, L.; Laurent, O.; Ebrahimian, T.; et al. Biomarkers for uranium risk assessment for the development of the CURE (Concerted Uranium Research in Europe) molecular epidemiological protocol. Radiat. Res. 2017, 187, 107–127. [Google Scholar] [CrossRef] [PubMed]
- Browne, D.; Whelton, H.; O’Mullane, D. Fluoride metabolism and fluorosis. J. Dent. 2005, 33, 177–186. [Google Scholar] [CrossRef]
- Xiong, X.; Liu, J.; He, W.; Xia, T.; He, P.; Chen, X.; Yang, K.; Wang, A. Dose-effect relationship between drinking water fluoride levels and damage to liver and kidney functions in children. Environ. Res. 2007, 103, 112–116. [Google Scholar] [CrossRef] [PubMed]
- UNSCEAR. Sources, effects and risks of ionizing radiation. In UNSCEAR 2016 Report; United Nations: New York, NY, USA, 2016. [Google Scholar]
- Barbier, O.; Arreola-Mendoza, L.; Del Razo, L.M. Molecular mechanisms of fluoride toxicity. Chem. Biol. Interact. 2010, 188, 319–333. [Google Scholar] [CrossRef]
- Vicente-Vicente, L.; Quiros, Y.; Perez-Barriocanal, F.; Lopez-Novoa, J.M.; Lopez-Hernandez, F.J.; Morales, AI. Nephrotoxicity of uranium: Pathophysiological, diagnostic and therapeutic perspectives. Toxicol. Sci. 2010, 118, 324–347. [Google Scholar] [CrossRef]
- Katz, S.A. The Chemistry and Toxicology of Depleted Uranium. Toxics 2014, 2, 50–78. [Google Scholar] [CrossRef] [Green Version]
- Miller, A.C.; Stewart, M.; Rivas, R.; Marino, S.; Randers-Pehrson, G.; Shi, L. Observation of radiation-specific damage in cells exposed to depleted uranium: Hprt gene mutation frequency. Radiat. Meas. 2007, 42, 1029–1032. [Google Scholar] [CrossRef]
- Toxicological Profile for Fluorides, Hydrogen Fluoride, and Fluorine; U.S. Agency for Toxic Substances and Disease Registry: Atlanta, GA, USA, 2003.
- Paquet, F.; Houpert, P.; Blanchardon, E.; Delissen, O.; Maubert, C.; Dhieux, B.; Moreels, A.M.; Frelon, S.; Gourmelon, P. Accumulation and distribution of uranium in rats after chronic exposure by ingestion. Health Phys. 2006, 90, 139–147. [Google Scholar] [CrossRef]
- Poisson, C.; Stefani, J.; Manens, L.; Delissen, O.; Suhard, D.; Tessier, C.; Dublineau, I.; Gueguen, Y. Chronic uranium exposure dose-dependently induces glutathione in rats without any nephrotoxicity. Free Radic. Res. 2014, 48, 1218–1231. [Google Scholar] [CrossRef] [PubMed]
- Usuda, K.; Kono, K.; Dote, T.; Nishiura, K.; Miyata, K.; Nishiura, H.; Shimahara, M. Urinary biomarkers monitoring for experimental fluoride nephrotoxicity. Arch. Toxicol. 1998, 72, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Manocha, S.L.; Warner, H.; Olkowski, Z.L. Cytochemical response of kidney, liver and nervous system of fluoride ions in drinking water. Histochem. J. 1975, 7, 343–355. [Google Scholar] [CrossRef]
- Kathren, R.L.; Burklin, R.K. Acute chemical toxicity of uranium. Health Phys. 2008, 94, 170–179. [Google Scholar] [CrossRef]
- Zamora, M.L.; Tracy, B.L.; Zielinski, J.M.; Meyerhof, D.P.; Moss, M.A. Chronic ingestion of uranium in drinking water: A study of kidney bioeffects in humans. Toxicol. Sci. 1998, 43, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Gueguen, Y.; Rouas, C. Données nouvelles sur la néphrotoxicité de l’uranium. Radioprotection 2012, 47, 345–359. [Google Scholar] [CrossRef] [Green Version]
- Zimmerman, K.L.; Barber, D.S.; Ehrich, M.F.; Tobias, L.; Hancock, S.; Hinckley, J.; Binder, E.M.; Jortner, B.S. Temporal clinical chemistry and microscopic renal effects following acute uranyl acetate exposure. Toxicol. Pathol. 2007, 35, 1000–1009. [Google Scholar] [CrossRef] [PubMed]
- Sangeetha Vijayan, P.; Rekha, P.D.; Dinesh, U.; Arun, A.B. Biochemical and histopathological responses of the Swiss albino mice treated with uranyl nitrate and its recovery. Ren. Fail. 2016, 38, 770–775. [Google Scholar] [CrossRef]
- Dote, T.; Kono, K.; Usuda, K.; Nishiura, H.; Tagawa, T.; Miyata, K.; Shimahara, M.; Hashiguchi, N.; Senda, J.; Tanaka, Y. Toxicokinetics of intravenous fluoride in rats with renal damage caused by high-dose fluoride exposure. Int. Arch. Occup. Environ. Health 2000, 73, S90–S92. [Google Scholar] [CrossRef]
- Santoyo-Sanchez, M.P.; del Carmen Silva-Lucero, M.; Arreola-Mendoza, L.; Barbier, O.C. Effects of acute sodium fluoride exposure on kidney function, water homeostasis, and renal handling of calcium and inorganic phosphate. Biol. Trace. Elem. Res. 2013, 152, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Pierrefite-Carle, V.; Santucci-Darmanin, S.; Breuil, V.; Gritsaenko, T.; Vidaud, C.; Creff, G.; Solari, P.L.; Pagnotta, S.; Al-Sahlanee, R.; Den Auwer, C.; et al. Effect of natural uranium on the UMR-106 osteoblastic cell line: Impairment of the autophagic process as an underlying mechanism of uranium toxicity. Arch. Toxicol. 2017, 4, 1903–1914. [Google Scholar] [CrossRef]
- Thiebault, C.; Carriere, M.; Milgram, S.; Simon, A.; Avoscan, L.; Gouget, B. Uranium induces apoptosis and is genotoxic to normal rat kidney (NRK-52E) proximal cells. Toxicol. Sci. 2007, 98, 479–487. [Google Scholar] [CrossRef]
- Hao, Y.; Ren, J.; Liu, C.; Li, H.; Liu, J.; Yang, Z.; Li, R.; Su, Y. Zinc protects human kidney cells from depleted uranium-induced apoptosis. Basic Clin. Pharmacol. Toxicol. 2014, 114, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.; Xu, Z.; Xia, T.; He, P.; Gao, P.; He, W.; Zhang, M.; Guo, L.; Niu, Q.; Wang, A. Effects of the Fas/Fas-L pathway on fluoride-induced apoptosis in SH-SY5Y cells. Environ. Toxicol. 2011, 26, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Jung, J.Y.; Jeong, Y.J.; Park, J.H.; Yang, K.H.; Choi, N.K.; Kim, S.H.; Kim, W.J. Involvement of both mitochondrial- and death receptor-dependent apoptotic pathways regulated by Bcl-2 family in sodium fluoride-induced apoptosis of the human gingival fibroblasts. Toxicology 2008, 243, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Li, Y.; Wang, J.; Manthari, R.K. Fluoride induces apoptosis and autophagy through the IL-17 signaling pathway in mice hepatocytes. Arch. Toxicol. 2018, 92, 3277–3289. [Google Scholar] [CrossRef]
- Hao, Y.; Huang, J.; Gu, Y.; Liu, C.; Li, H.; Liu, J.; Ren, J.; Yang, Z.; Peng, S.; Wang, W.; et al. Metallothionein deficiency aggravates depleted uranium-induced nephrotoxicity. Toxicol. Appl. Pharmacol. 2015, 287, 306–315. [Google Scholar] [CrossRef] [PubMed]
- Linares, V.; Belles, M.; Albina, M.L.; Sirvent, J.J.; Sanchez, D.J.; Domingo, J.L. Assessment of the pro-oxidant activity of uranium in kidney and testis of rats. Toxicol. Lett. 2006, 167, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, D.A.; Cardoso, C.M.; Yujra, V.Q.; Viana, M.B.; Aguiar, O., Jr.; Pisani, L.P.; Oshima, C.T.F. Fluoride Induces Apoptosis in Mammalian Cells: In Vitro and In Vivo Studies. Anticancer Res. 2017, 37, 4767–4777. [Google Scholar]
- Sun, Z.; Niu, R.; Wang, B.; Jiao, Z.; Wang, J.; Zhang, J.; Wang, S.; Wang, J. Fluoride-induced apoptosis and gene expression profiling in mice sperm in vivo. Arch. Toxicol. 2011, 85, 1441–1452. [Google Scholar] [CrossRef]
- Singh, R.; Khatri, P.; Srivastava, N.; Jain, S.; Brahmachari, V.; Mukhopadhyay, A.; Mazumder, S. Fluoride exposure abates pro-inflammatory response and induces in vivo apoptosis rendering zebrafish (Danio rerio) susceptible to bacterial infections. Fish Shellfish Immunol. 2017, 63, 314–321. [Google Scholar] [CrossRef]
- Dublineau, I.; Grandcolas, L.; Grison, S.; Baudelin, C.; Paquet, F.; Voisin, P.; Aigueperse, J.; Gourmelon, P. Modifications of inflammatory pathways in rat intestine following chronic ingestion of depleted uranium. Toxicol. Sci. 2007, 98, 458–468. [Google Scholar] [CrossRef]
- Inkielewicz-Stepniak, I.; Czarnowski, W.; Krechniak, J. Determination of Fluoride in Soft Tissues. Fluoride 2003, 36, 16–20. [Google Scholar]
- Rouas, C.; Stefani, J.; Grison, S.; Grandcolas, L.; Baudelin, C.; Dublineau, I.; Pallardy, M.; Gueguen, Y. Effect of nephrotoxic treatment with gentamicin on rats chronically exposed to uranium. Toxicology 2011, 279, 27–35. [Google Scholar] [CrossRef]
- Cardenas-Gonzalez, M.C.; Del Razo, L.M.; Barrera-Chimal, J.; Jacobo-Estrada, T.; Lopez-Bayghen, E.; Bobadilla, N.A.; Barbier, O. Proximal renal tubular injury in rats sub-chronically exposed to low fluoride concentrations. Toxicol. Appl. Pharmacol. 2013, 272, 888–894. [Google Scholar] [CrossRef]
- Homma-Takeda, S.; Terada, Y.; Nakata, A.; Sahoo, S.K.; Yoshida, S.; Ueno, S.; Inoue, M.; Iso, H.; Ishikawa, T.; Konishi, T.; et al. Elemental imaging of kidneys of adult rats exposed to uranyl acetate. Nucl. Instrum. Methods Phys. Res. B 2009, 267, 2167–2170. [Google Scholar] [CrossRef]
- Shashi, A.; Singh, J.; Thapar, S. Toxic effects of fluoride on rabbit kidney. Fluoride 2002, 35, 38–50. [Google Scholar]
- Haley, D.P.; Bulger, R.E.; Dobyan, D.C. The long-term effects of uranyl nitrate on the structure and function of the rat kidney. Virchows Arch. B Cell Pathol. Incl. Mol. Pathol. 1982, 41, 181–192. [Google Scholar] [CrossRef]
- Kour, K.; Singh, J. Histological findings in kidneys of mice following sodium fluoride administration. Fluoride 1980, 13, 163–167. [Google Scholar]
- McCay, C.M.; Ramseyer, W.F.; Smith, C.A. Effect of sodium fluoride administration on body changes in old rats. J. Gerontol. 1957, 12, 14–19. [Google Scholar]
- Sanchez, D.J.; Belles, M.; Albina, M.L.; Sirvent, J.J.; Domingo, J.L. Nephrotoxicity of simultaneous exposure to mercury and uranium in comparison to individual effects of these metals in rats. Biol. Trace Elem. Res. 2001, 84, 139–154. [Google Scholar] [CrossRef]
- Chattopadhyay, A.; Podder, S.; Agarwal, S.; Bhattacharya, S. Fluoride-induced histopathology and synthesis of stress protein in liver and kidney of mice. Arch. Toxicol. 2011, 85, 327–335. [Google Scholar] [CrossRef]
- Machalinska, A.; Wiszniewska, B.; Tarasiuk, J.; Machalinski, B. Morphological effects of sodium fluoride on hematopoietic organs in mice. Fluoride 2002, 35, 231–238. [Google Scholar]
- Fujigaki, Y. Different modes of renal proximal tubule regeneration in health and disease. World J. Nephrol. 2012, 1, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, D. How much can the tubule regenerate and who does it? An open question. Nephrol. Dial. Transplant. 2016, 31, 1243–1250. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, D.N.; Houghton, D.C.; Bennett, W.M.; Plamp, C.E.; Reger, K.; Porter, G.A. Reversibility of gentamicin nephrotoxicity in rats: Recovery during continuous drug administration. Proc. Soc. Exp. Biol. Med. 1979, 160, 99–103. [Google Scholar] [CrossRef]
- Xu, H.; Hu, L.S.; Chang, M.; Jing, L.; Zhang, X.Y.; Li, G.S. Proteomic analysis of kidney in fluoride-treated rat. Toxicol. Lett. 2005, 160, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Bonventre, J.V. Kidney injury molecule-1 (KIM-1): A urinary biomarker and much more. Nephrol. Dial. Transplant. 2009, 24, 3265–3268. [Google Scholar] [CrossRef]
- Ko, G.J.; Grigoryev, D.N.; Linfert, D.; Jang, H.R.; Watkins, T.; Cheadle, C.; Racusen, L.; Rabb, H. Transcriptional analysis of kidneys during repair from AKI reveals possible roles for NGAL and KIM-1 as biomarkers of AKI-to-CKD transition. Am. J. Physiol. Renal. Physiol. 2010, 298, F1472–F1483. [Google Scholar] [CrossRef]
- Vaidya, V.S.; Ozer, J.S.; Dieterle, F.; Collings, F.B.; Ramirez, V.; Troth, S.; Muniappa, N.; Thudium, D.; Gerhold, D.; Holder, D.J.; et al. Kidney injury molecule-1 outperforms traditional biomarkers of kidney injury in preclinical biomarker qualification studies. Nat. Biotechnol. 2010, 28, 478–485. [Google Scholar] [CrossRef]
- Iwakura, T.; Fujigaki, Y.; Fujikura, T.; Ohashi, N.; Kato, A.; Yasuda, H. Acquired resistance to rechallenge injury after acute kidney injury in rats is associated with cell cycle arrest in proximal tubule cells. Am. J. Physiol. Renal. Physiol. 2016, 310, F872–F884. [Google Scholar] [CrossRef] [Green Version]
- Fujigaki, Y.; Sun, D.F.; Goto, T.; Hishida, A. Temporary changes in macrophages and MHC class-II molecule-expressing cells in the tubulointerstitium in response to uranyl acetate-induced acute renal failure in rats. Virchows Arch. 2003, 443, 206–216. [Google Scholar] [CrossRef]
- Correa-Rotter, R.; Ibarra-Rubio, M.E.; Schwochau, G.; Cruz, C.; Silkensen, J.R.; Pedraza-Chaverri, J.; Chmielewski, d.; Rosenberg, M.E. Induction of clusterin in tubules of nephrotic rats. J. Am. Soc. Nephrol. 1998, 9, 33–37. [Google Scholar] [PubMed]
- Aulitzky, W.K.; Schlegel, P.N.; Wu, D.F.; Cheng, C.Y.; Chen, C.L.; Li, P.S.; Goldstein, M.; Reidenberg, M.; Bardin, C.W. Measurement of urinary clusterin as an index of nephrotoxicity. Proc. Soc. Exp. Biol. Med. 1992, 199, 93–96. [Google Scholar] [CrossRef]
- Brooks, C.R.; Yeung, M.Y.; Brooks, Y.S.; Chen, H.; Ichimura, T.; Henderson, J.M.; Bonventre, J.V. KIM-1-/TIM-1-mediated phagocytosis links ATG5-/ULK1-dependent clearance of apoptotic cells to antigen presentation. EMBO J. 2015, 34, 2441–2464. [Google Scholar] [CrossRef] [Green Version]
- Ichimura, T.; Asseldonk, E.J.; Humphreys, B.D.; Gunaratnam, L.; Duffield, J.S.; Bonventre, J.V. Kidney injury molecule-1 is a phosphatidylserine receptor that confers a phagocytic phenotype on epithelial cells. J. Clin. Invest. 2008, 118, 1657–1668. [Google Scholar] [CrossRef]
- Taulan, M.; Paquet, F.; Argiles, A.; Demaille, J.; Romey, M.C. Comprehensive analysis of the renal transcriptional response to acute uranyl nitrate exposure. BMC Genomics 2006, 7, 2. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Brooks, C.R.; Xiao, S.; Sabbisetti, V.; Yeung, M.Y.; Hsiao, L.L.; Ichimura, T.; Kuchroo, V.; Bonventre, J.V. KIM-1-mediated phagocytosis reduces acute injury to the kidney. J. Clin. Invest. 2015, 125, 1620–1636. [Google Scholar] [CrossRef] [PubMed]
- Monleau, M.; De Meo, M.; Paquet, F.; Chazel, V.; Dumenil, G.; Donnadieu-Claraz, M. Genotoxic and inflammatory effects of depleted uranium particles inhaled by rats. Toxicol. Sci. 2006, 89, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Aydin, G.; Cicek, E.; Akdogan, M.; Gokalp, O. Histopathological and biochemical changes in lung tissues of rats following administration of fluoride over several generations. J. Appl. Toxicol. 2003, 23, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Miranda, L.C.; Viaro, F.; Ceneviva, R.; Evora, P.R. Endothelium-dependent and -independent hepatic artery vasodilatation is not impaired in a canine model of liver ischemia-reperfusion injury. Braz. J. Med. Biol. Res. 2007, 40, 857–865. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, P.; Verin, A.D.; Birukova, A.; Gilbert-McClain, L.I.; Jacobs, K.; Garcia, J.G. Mechanisms of sodium fluoride-induced endothelial cell barrier dysfunction: Role of MLC phosphorylation. Am. J. Physiol. Lung Cell Mol. Physiol. 2001, 281, L1472–L1483. [Google Scholar] [CrossRef]
- Akcay, A.; Nguyen, Q.; Edelstein, C.L. Mediators of inflammation in acute kidney injury. Mediators Inflamm. 2009, 2009, 137072. [Google Scholar] [CrossRef] [PubMed]
- Guerrero-Beltran, C.E.; Mukhopadhyay, P.; Horvath, B.; Rajesh, M.; Tapia, E.; Garcia-Torres, I.; Pedraza-Chaverri, J.; Pacher, P. Sulforaphane, a natural constituent of broccoli, prevents cell death and inflammation in nephropathy. J. Nutr. Biochem. 2012, 23, 494–500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, J.J.; Lee, S.H.; Kim, D.K.; Jin, R.; Jung, D.S.; Kwak, S.J.; Kim, S.H.; Han, S.H.; Lee, J.E.; Moon, S.J.; et al. Colchicine attenuates inflammatory cell infiltration and extracellular matrix accumulation in diabetic nephropathy. Am. J. Physiol. Renal. Physiol. 2009, 297, F200–F209. [Google Scholar] [CrossRef] [PubMed] [Green Version]








| Gene | Accession Number | Forward | Reverse |
|---|---|---|---|
| CLU | AF182509 | TCGGGCATCTGGCATCA | AAGCTCACGGGCGAAGAAC |
| GAPDH | GU214026 | TCCACTCACGGCAAATTCAACG | TAGACTCCACGACATACTCAGC |
| HPRT | NM_013556 | TGCTGCGTCCCCAGACTTTTG | AGATAAGCGACAATCTACCAGAGG |
| ICAM-1 | NM_010493 | CACCCCAAGGACCCCAAGGAGAT | CGACGCCGCTCAGAAGAACCAC |
| KIM-1 | BC053400 | TTTCAGGCCTCATACTGCTTCTC | TGACCCACCACCCCCTTT |
| OPN | AF515708 | CCCTCGATGTCATCCCTGTT | TTCCGTTGTTGTCCTGATCAGA |
| VCAM-1 | BC029823 | TCGCGGTCTTGGGAGCCTCA | TGACCGTGACCGGCTTCCCA |
| Fluoride | Mean ± SEM | |
|---|---|---|
| µg/24 h | ||
| Before injection | Control | 2.8 ± 0.4 |
| 2 mg/kg | 2.8 ± 0.4 | |
| 5 mg/kg | 2.5 ± 0.3 | |
| 7.5 mg/kg | 2.7 ± 0.3 | |
| 10 mg/kg | 2.4 ± 0.2 | |
| After injection | Control | 3.0 ± 0.5 |
| 2 mg/kg | 15.1 ± 1.2 * | |
| 5 mg/kg | 35.5 ± 3.2 *** | |
| 7.5 mg/kg | 43.0 ± 5.5 *** | |
| 10 mg/kg | 60.7 ± 5.4 *** | |
| Uranyl Nitrate | Mean ± SEM | |
|---|---|---|
| ng/24 h | ||
| Before injection | Control | 17 ± 7 |
| 2 mg/kg | 32 ± 8 | |
| 4 mg/kg | 49 ± 21 | |
| 5 mg/kg | 40 ± 14 | |
| After injection | Control | 22 ± 9 |
| 2 mg/kg | 3 870 ± 497 * | |
| 4 mg/kg | 8 323 ± 2 009 *** | |
| 5 mg/kg | 8 599 ± 2 442 *** | |
| Fluoride | Creatinine | Glucose | Potassium | Sodium | Phosphorus | Total Protein | Urea | Calcium | |
|---|---|---|---|---|---|---|---|---|---|
| µmol/24 h | µmol/24 h | mmol/24 h | mmol/24 h | mmol/24 h | mg/24 h | mmol/24 h | µmol/24 h | ||
| Before injection | Control | 4.07 ± 0.60 | 2.94 ± 0.45 | 0.14 ± 0.01 | 0.13 ± 0.02 | 0.14 ± 0.03 | 9.03 ± 1.86 | 1.67 ± 0.23 | 4.02 ± 0.66 |
| 2 mg/kg | 4.31 ± 0.28 | 4.98 ± 0.30 | 0.15 ± 0.01 | 0.13 ± 0.02 | 0.11 ± 0.03 | 8.15 ± 4.03 | 2.26 ± 0.32 | 4.74 ± 0.89 | |
| 5 mg/kg | 4.12 ± 0.27 | 4.32 ± 0.35 | 0.14 ± 0.01 | 0.12 ± 0.01 | 0.12 ± 0.01 | 14.73 ± 3.47 | 2.20 ± 0.19 | 3.22 ± 0.28 | |
| 7.5 mg/kg | 3.82 ± 0.44 | 3.03 ± 0.05 | 0.15 ± 0.05 | 0.16 ± 0.01 | 0.15 ± 0.00 | 7.80 ± 0.28 | 1.39 ± 0.27 | 4.88 ± 0.56 | |
| 10 mg/kg | 3.53 ± 0.52 | 3.29 ± 0.56 | 0.13 ± 0.01 | 0.10 ± 0.01 | 0.14 ± 0.02 | 8.50 ± 1.83 | 1.61 ± 0.26 | 5.04 ± 1.32 | |
| After injection | Control | 3.61 ± 0.51 | 3.41 ± 0.58 | 0.13 ± 0.01 | 0.12 ± 0.01 | 0.13 ± 0.01 | 9.15 ± 2.47 | 1.93 ± 0.39 | 2.58 ± 0.41 |
| 2 mg/kg | 4.19 ± 0.38 | 4.32 ± 0.86 | 0.11 ± 0.01 | 0.11 ± 0.01 | 0.11 ± 0.01 | 10.66 ± 3.54 | 2.06 ± 0.39 | 3.57 ± 0.84 | |
| 5 mg/kg | 4.56 ± 0.25 | 4.43 ± 0.43 | 0.15 ± 0.01 | 0.15 ± 0.02 | 0.13 ± 0.01 | 17.89 ± 2.30 | 2.55 ± 0.23 | 3.16 ± 0.35 | |
| 7.5 mg/kg | 2.11 ± 0.49 | 17.52 ± 10.50 | 0.09 ± 0.02 | 0.13 ± 0.03 | 0.11 ± 0.02 | 3.82 ± 0.71 | 0.91 ± 0.21 | 2.79 ± 1.25 | |
| 10 mg/kg | 4.72 ± 0.59 | 157.4 ± 46.08 *** | 0.23 ± 0.03 *** | 0.47 ± 0.09 *** | 0.15 ± 0.02 | 16.88 ± 3.76 | 1.24 ± 0.17 | 3.89 ± 0.69 | |
| Uranyl Nitrate | Creatinine | Glucose | Potassium | Sodium | Phosphorus | Total Protein | Urea | Calcium | |
|---|---|---|---|---|---|---|---|---|---|
| µmol/24 h | µmol/24 h | mmol/24 h | mmol/24 h | mmol/24 h | mg/24 h | mmol/24 h | µmol/24 h | ||
| Before injection | Control | 2.88 ± 0.52 | 4.31 ± 0.37 | 0.14 ± 0.01 | 0.21 ± 0.05 | 0.20 ± 0.02 | 10.05 ± 2.75 | 1.76 ± 0.27 | 4.03 ± 0.63 |
| 2 mg/kg | 3.11 ± 0.52 | 4.59 ± 0.39 | 0.14 ± 0.01 | 0.20 ± 0.02 | 0.16 ± 0.01 | 15.90 ± 3.36 | 1.48 ± 0.22 | 3.78 ± 0.25 | |
| 4 mg/kg | 2.66 ± 0.50 | 3.88 ± 0.44 | 0.14 ± 0.01 | 0.23 ± 0.02 | 0.18 ± 0.02 | 10.67 ± 2.11 | 1.45 ± 0.24 | 4.13 ± 0.63 | |
| 5 mg/kg | 3.35 ± 0.43 | 3.51 ± 0.59 | 0.15 ± 0.01 | 0.24 ± 0.03 | 0.18 ± 0.02 | 16.65 ± 2.73 | 1.80 ± 0.28 | 4.16 ± 0.47 | |
| After injection | Control | 3.09 ± 0.55 | 4.50 ± 0.34 | 0.15 ± 0.01 | 0.21 ± 0.03 | 0.22 ± 0.01 | 16.33 ± 3.84 | 1.70 ± 0.29 | 3.71 ± 0.42 |
| 2 mg/kg | 2.99 ± 0.38 | 3.22 ± 0.19 | 0.13 ± 0.01 | 0.23 ± 0.02 | 0.17 ± 0.01 | 12.20 ± 2.00 | 1.37 ± 0.22 | 5.70 ± 0.65 * | |
| 4 mg/kg | 3.44 ± 0.35 | 7.06 ± 2.72 | 0.14 ± 0.02 | 0.29 ± 0.07 | 0.16 ± 0.02 | 11.31 ± 2.36 | 1.27 ± 0.29 | 6.82 ± 1.31 * | |
| 5 mg/kg | 3.61 ± 0.25 | 15.76 ± 3.71 *** | 0.14 ± 0.01 | 0.28 ± 0.08 | 0.18 ± 0.01 | 19.36 ± 2.90 | 1.52 ± 0.21 | 6.23 ± 0.74 * | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bontemps, A.; Conquet, L.; Elie, C.; Magneron, V.; Gloaguen, C.; Kereselidze, D.; Tack, K.; Barbier, O.C.; Guéguen, Y. In Vivo Comparison of the Phenotypic Aspects and Molecular Mechanisms of Two Nephrotoxic Agents, Sodium Fluoride and Uranyl Nitrate. Int. J. Environ. Res. Public Health 2019, 16, 1136. https://doi.org/10.3390/ijerph16071136
Bontemps A, Conquet L, Elie C, Magneron V, Gloaguen C, Kereselidze D, Tack K, Barbier OC, Guéguen Y. In Vivo Comparison of the Phenotypic Aspects and Molecular Mechanisms of Two Nephrotoxic Agents, Sodium Fluoride and Uranyl Nitrate. International Journal of Environmental Research and Public Health. 2019; 16(7):1136. https://doi.org/10.3390/ijerph16071136
Chicago/Turabian StyleBontemps, Alice, Laurine Conquet, Christelle Elie, Victor Magneron, Céline Gloaguen, Dimitri Kereselidze, Karine Tack, Olivier C. Barbier, and Yann Guéguen. 2019. "In Vivo Comparison of the Phenotypic Aspects and Molecular Mechanisms of Two Nephrotoxic Agents, Sodium Fluoride and Uranyl Nitrate" International Journal of Environmental Research and Public Health 16, no. 7: 1136. https://doi.org/10.3390/ijerph16071136