Fluoride Exposure Induces Inhibition of Sodium-and Potassium-Activated Adenosine Triphosphatase (Na+, K+-ATPase) Enzyme Activity: Molecular Mechanisms and Implications for Public Health
Abstract
:1. Introduction
2. The Role of Fluoride in Oral Health and Sources of Fluoride Exposure
| Non Fluoridated Region mg/L | Fluoridated Region mg/L | Reference | |
|---|---|---|---|
| Human milk | 0.004 | 0.009 | [154,161] |
| Cow’s Milk | 0.016 | 0.074–0.18 | [156] |
| Cow’s milk based powdered | 0.02–0.18 | 0.49–1.40 | [159] |
| infant formula reconstituted | |||
| with tap water | |||
| Non-Fluoridated | Fluoridated | Reference | |
| Ionic F levels | Ionic F levels | ||
| µM | µM | ||
| Fully Breast-fed infants | |||
| 1–6 months | = 0.22 | [163] | |
| Formula fed | |||
| 1–6 months | = 0.29 | [163] | |
| Breast fed with semi solids | |||
| 6–12 months | = 0.35 | [163] | |
| (0.10–0.67) | |||
| Aged 1 month | 0.89 | [169] | |
| Aged 7 months | 0.53 | [169] | |
| Breast and formula fed | |||
| Aged 4–6 months | 4.33 | [164] | |
| (0.52–8.0) | |||
| Aged 7–12 months | 1.56 | [164] | |
| with semi solids | (1.03–2.1) | ||
| Aged 4–18 months |
3. Na+, K+-ATPase Regulation by Phosphorylation/Dephosphorylation
4. Molecular Mechanisms by which Fluoride Inhibits Na+, K+-ATPase Activity
4.1. The Role of Protein Kinase RNA-like ER Kinase (PERK) in Regulating Na+, K+-ATPase Activity and the Influence of Fluoride on PERK Activity
4.2. The role of Protein Kinase C (PKC) in Regulating Na+, K+-ATPase Activity and the Influence of Fluoride in Regulating PKC Activity
4.3. The Role of Cyclic Adenosine-Monophosphate (cAMP) in Regulating Na+, K+-ATPase Activity and the Influence of Fluoride in Regulating cAMP
4.4. The Influence of Magnesium in Regulating Na+, K+-ATPase Activity and the Influence of Fluoride on Magnesium Homeostasis
4.5. The Influence of Calcineurin, Calmodulin and Manganese in Regulating Na+, K+-ATPase Activity and the Influence of Fluoride on Calcineurin, Calmodulin and Manganese Homeostasis
4.6. The Role of Cyclic Guanosine Monophosphate (cGMP) and Nitric Oxide in Regulating Na+, K+-ATPase Activity and the Contribution of Fluoride to Regulating cGMP and Nitric Oxide
4.7. Cytokine TGF-β1 Inhibits NKA Activity
4.8. The Role of Inorganic Phosphate in Regulating Na+, K+-ATPase Activity
4.9. Dopamine Inhibits Na+-K+-ATPase Activity
4.10. Parathyroid Hormone Inhibits Na(+)-K(+)-ATPase Activity
4.11. Hyperglycaemia Inhibits Na+ K+ ATPase Activity via Activation of PGE2 Production
4.12. Advanced Glycation end Products Inhibit Na+ K+ ATPase
5. Discussion
Additional Perspectives
6. Conclusions
Funding
Conflicts of Interest
Abbreviations
| AA | Arachidonic Acid |
| ATP | Adenosine-triphosphate |
| ALP | Alkaline phosphatase |
| cAMP | cyclic adenosine-monophosphate monophosphate |
| CaM | Calmodulin |
| cGMP | Cyclic guanosine monophosphate |
| Cn | Calcineurin |
| COPD | Chronic obstructive pulmonary disease |
| CT | Calcitonin |
| F | Fluoride |
| Mn | Magnesium |
| NKA: | Na+, K+-ATPase |
| NO | Nitric oxide |
| PKC | Protein kinase C |
| Pi | Inorganic phosphate |
| PGE2: | Prostaglandin E2 |
| PTH | Parathyroid hormone |
| PLA2 | Phospholipase A2 |
| RAGE | Receptors for advanced glycation end products |
| TGF-β1 | Transforming growth factor β 1 |
| TSH | Thyroid-stimulating-hormone, also called Thyrotropin |
| α | Alpha |
| β | Beta |
| U.S.A | United States of America |
References
- Skou, J.C. The influence of some cations on an adenosine triphosphatase from peripheral nerves. Biochim. Biophys. Acta 1957, 23, 394–401. [Google Scholar] [CrossRef]
- Jorgensen, P.L.; Hakansson, K.O.; Karlish, S.J. Structure and mechanism of Na, K-ATPase: Functional sites and their interactions. Annu. Rev. Physiol. 2003, 65, 817–849. [Google Scholar] [CrossRef]
- De Lores Arnaiz, G.R.; López Ordieres, M.G. Brain Na+, K+-ATPase Activity in Aging and Disease. Int. J. Biomed. Sci. 2014, 10, 85–102. [Google Scholar]
- Kristensen, A.S.; Andersen, J.; Jørgensen, T.N.; Sørensen, L.; Eriksen, J.; Loland, C.J.; Strømgaard, K.; Gether, U. SLC6 neurotransmitter transporters: Structure, function, and regulation. Pharmacol. Rev. 2011, 63, 585–640. [Google Scholar] [CrossRef] [PubMed]
- Mobasheri, A.; Avila, J.; Cózar-Castellano, I.; Brownleader, M.D.; Trevan, M.; Francis, M.J.; Lamb, J.F.; Martín-Vasallo, P. Na+, K+-ATPase isozyme diversity: Comparative biochemistry and physiological implications of novel functional interactions. Biosci. Rep. 2000, 20, 51–91. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, D.E.; Miller, R.B.; Thiesfeldt, S.; Lakhani, H.V.; Shapiro, J.I.; Sodhi, K. The Role of Na/K-ATPase Signaling in Oxidative Stress Related to Aging: Implications in Obesity and Cardiovascular Disease. Int. J. Mol. Sci. 2018, 19, 2139. [Google Scholar] [CrossRef] [PubMed]
- Clausen, M.V.; Hilbers, F.; Poulsen, H. The Structure and Function of the Na, K-ATPase Isoforms in Health and Disease. Front. Physiol. 2017, 8, 371. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Shapiro, J.I. The physiological and clinical importance of sodium potassium ATPase in cardiovascular diseases. Curr. Opin. Pharmacol. 2016, 27, 43–49. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Srikanthan, K.; Shapiro, J.I.; Sodhi, K. The Role of Na/K-ATPase Signaling in Oxidative Stress Related to Obesity and Cardiovascular Disease. Molecules 2016, 21, 1172. [Google Scholar] [CrossRef]
- Kinoshita, P.F.; Leite, J.A.; Orellana, A.M.; Vasconcelos, A.R.; Quintas, L.E.; Kawamoto, E.M.; Scavone, C. The Influence of Na(+), K(+)-ATPase on Glutamate Signaling in Neurodegenerative Diseases and Senescence. Front. Physiol. 2016, 7, 195. [Google Scholar] [CrossRef] [PubMed]
- Iannello, S.; Milazzo, P.; Belfiore, F. Animal and human tissue Na,K-ATPase in normal and insulin-resistant states: Regulation, behaviour and interpretative hypothesis on NEFA effects. Obes. Rev. 2007, 8, 231–251. [Google Scholar] [CrossRef]
- Suhail, M. Na+, K+-ATPase: Ubiquitous Multifunctional Transmembrane Protein and its Relevance to Various Pathophysiological Conditions. J. Clin. Med. Res. 2010, 2, 1–17. [Google Scholar] [CrossRef]
- Rose, A.M.; Valdes, R., Jr. Understanding the sodium pump and its relevance to disease. Clin. Chem. 1994, 40, 1674–1685. [Google Scholar] [PubMed]
- Lichtstein, D.; Ilani, A.; Rosen, H.; Horesh, N.; Singh, S.V.; Buzaglo, N.; Hodes, A. Na+, K+-ATPase Signaling and Bipolar Disorder. Int. J. Mol. Sci. 2018, 19, 2314. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Pogwizd, S.M.; Prabhu, S.D.; Zhou, L. Inhibiting Na+/K+ ATPase can impair mitochondrial energetics and induce abnormal Ca2+ cycling and automaticity in guinea pig cardiomyocytes. PLoS ONE 2014, 9, e93928. [Google Scholar] [CrossRef]
- Roy, S.; Dasgupta, A.; Banerjee, U.; Chowdhury, P.; Mukhopadhyay, A.; Saha, G.; Singh, O. Role of membrane cholesterol and lipid peroxidation in regulating the Na+/K+-ATPase activity in schizophrenia. Indian J. Psychiatry 2016, 58, 317–325. [Google Scholar] [PubMed]
- Yan, Y.; Haller, S.; Shapiro, A.; Malhotra, N.; Tian, J.; Xie, Z.; Malhotra, D.; Shapiro, J.I.; Liu, J. Ouabain-stimulated trafficking regulation of the Na/K-ATPase and NHE3 in renal proximal tubule cells. Mol. Cell. Biochem. 2012, 367, 175–183. [Google Scholar] [CrossRef]
- Gentile, D.A.; Skoner, D.P. The relationship between airway hyperreactivity (AHR) and sodium, potassium adenosine triphosphatase (Na+,K+ATPase) enzyme inhibition. J. Allergy Clin. Immunol. 1997, 99, 367–373. [Google Scholar] [CrossRef]
- Chhabra, S.K.; Khanduja, A.; Jain, D. Decreased sodium-potassium and calcium adenosine triphosphatase activity in asthma: Modulation by inhaled and oral corticosteroids. Indian J. Chest Dis. Allied Sci. 1999, 41, 15–26. [Google Scholar]
- Chhabra, S.K.; Khanduja, A.; Jain, D. Increased intracellular calcium and decreased activities of leucocyte Na+, K+-ATPase and Ca2+-ATPase in asthma. Clin. Sci. 1999, 97, 595–601. [Google Scholar] [CrossRef] [PubMed]
- Bukowska, B.; Sicińska, P.; Pająk, A.; Koceva-Chyla, A.; Pietras, T.; Pszczółkowska, A.; Górski, P.; Koter-Michalak, M. Oxidative stress and damage to erythrocytes in patients with chronic obstructive pulmonary disease—Changes in ATPase and acetylcholinesterase activity. Biochem. Cell. Biol. 2015, 93, 574–580. [Google Scholar] [CrossRef] [PubMed]
- Skoner, D.P.; Gentile, D.; Evans, R. Decreased activity of the platelet Na+, K(+)-adenosine triphosphatase enzyme in allergic subjects. J. Lab. Clin. Med. 1990, 115, 535–540. [Google Scholar] [PubMed]
- Skoner, D.P.; Gentile, D.; Evans, R.W. A circulating inhibitor of the platelet Na+, K+ adenosine triphosphatase (ATPase) enzyme in allergy. J. Allergy Clin. Immunol. 1991, 87, 476–482. [Google Scholar] [CrossRef]
- Van Deusen, M.A.; Gentile, D.A.; Skoner, D.P. Inhibition of the sodium, potassium adenosine triphosphatase enzyme in peripheral blood mononuclear cells of subjects with allergic rhinitis. Ann. Allergy Asthma Immunol. 1997, 78, 259–264. [Google Scholar] [CrossRef]
- Omar, A.K.; Ahmed, K.A.; Helmi, N.M.; Abdullah, K.T.; Qarii, M.H.; Hasan, H.E.; Ashwag, A.; Nabil, A.M.; Abdu, A.L.G.M.; Salama, M.S. The sensitivity of Na+, K+ ATPase as an indicator of blood diseases. Afr. Health Sci. 2017, 17, 262–269. [Google Scholar] [CrossRef]
- Woo, A.L.; James, P.F.; Lingrel, J.B. Sperm Motility Is Dependent on a Unique Isoform of the Na, K-ATPase. J. Biol. Chem. 2000, 275, 20693–20699. [Google Scholar] [CrossRef] [PubMed]
- Newton, L.D.; Krishnakumar, S.; Menon, A.G.; Kastelic, J.P.; van der Thundathil, J.C. Na+/K+ATPase regulates sperm capacitation through a mechanism involving kinases and redistribution of its testis-specific isoform. Mol. Reprod. Dev. 2009, 77, 136–148. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, T.; McDermott, J.P.; Sánchez, G.; Blanco, G. Na, K-ATPase α4 isoform is essential for sperm fertility. Proc. Natl. Acad. Sci. USA 2011, 108, 644–649. [Google Scholar] [CrossRef]
- Meskalo, О.I.; Fafula, R.V.; Lychkovskyj, E.I.; Vorobets, Z.D. Na+, K+-ATPase and Ca2+, Mg2+-ATPase Activity in Spermatozoa of Infertile Men with Different Forms of Pathospermia. Biol. Stud. 2017, 11, 5–12. [Google Scholar] [CrossRef]
- Testa, I.; Rabini, R.A.; Corvetta, A.; Danieli, G. Decreased NA+, K+-ATPase activity in erythrocyte membrane from rheumatoid arthritis patients. Scand. J. Rheumatol. 1987, 16, 301–305. [Google Scholar] [CrossRef]
- Kiziltunc, A.; Cogalgil, S.; Ugur, M.; Avci, B.; Akcay, F. Sialic acid, transketolase and Na+, K+, ATPase in patients with rheumatoid arthritis. Clin. Chem. Lab. Med. 1998, 36, 289–293. [Google Scholar] [CrossRef] [PubMed]
- Chibalin, A.V. Regulation of the Na, K-ATPase: Special implications for cardiovascular complications of metabolic syndrome. Pathophysiology 2007, 14, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Vásárhelyi, B.; Sallay, P.; Balog, E.; Reusz, G.; Tulassay, T. Altered Na(+)-K+ ATPase activity in uraemic adolescents. Acta Paediatr. 1996, 85, 919–922. [Google Scholar] [CrossRef]
- Kaji, D.; Thomas, K. Na+-K+ pump in chronic renal failure. Am. J. Physiol. 1987, 252, F785–F793. [Google Scholar] [CrossRef]
- Welt, L.G.; Sachs, J.R.; McManus, T.J. An ion transport defect in erythrocytes from uremic patients. Trans. Assoc. Am. Phys. 1964, 77, 169–181. [Google Scholar] [PubMed]
- Finotti, P.; Palatini, P. Reduction of erythrocyte (Na-K1)ATPase activity in typeI (insulin dependent) diabetic subjects and its activation by homologous plasma. Diabetologia 1986, 29, 623–628. [Google Scholar] [CrossRef] [PubMed]
- Tsimarato, M.; Coste, T.C.; Djemli-Shipkolye, A.; Daniel, L.; Shipkolye, F.; Vague, P.; Raccah, D. Evidence of time-dependent changes in renal medullary Na,K-ATPase activity and expression in diabetic rats. Cell. Mol. Biol. 2001, 47, 239–245. [Google Scholar]
- Vague, P.; Coste, T.C.; Jannot, M.F.; Raccah, D.; Tsimaratos, M. C-peptide, Na+,K(+)-ATPase, and diabetes. Exp. Diabesity Res. 2004, 5, 37–50. [Google Scholar] [CrossRef]
- Djemli-Shipkolye, A.; Gallice, P.; Coste, T.; Jannot, M.F.; Tsimaratos, M.; Raccah, D.; Vague, P. The effects ex vivo and in vitro of insulin and C-peptide on Na/K adenosine triphosphatase activity in red blood cell membranes of type 1 diabetic patients. Metabolism 2000, 49, 868–872. [Google Scholar] [CrossRef]
- Mimura, M.; Makino, H.; Kanatsuka, A.; Asai, T.; Yoshida, S. Reduction of erythrocyte (Na(+)-K+)ATPase activity in type 2 (non-insulin-dependent) diabetic patients with microalbuminuria. Horm. Metab. Res. 1994, 26, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Schwinger, R.H.; Bundgaard, H.; Muller-Ehmsen, J.; Kjeldsen, K. The Na, K-ATPase in the failing human heart. Cardiovasc. Res. 2003, 57, 913–920. [Google Scholar] [CrossRef] [Green Version]
- Muller-Ehmsen, J.; McDonough, A.A.; Farley, R.A.; Schwinger, R.H. Sodium pump isoform expression in heart failure: Implication for treatment. Basic Res. Cardiol. 2002, 97, I25–I30. [Google Scholar] [CrossRef]
- Stefanon, I.; Cade, J.R.; Fernandes, A.A.; Ribeiro Junior, R.F.; Targueta, G.P.; Mill, J.G.; Vassallo, D.V. Ventricular performance and Na+-K+ ATPase activity are reduced early and late after myocardial infarction in rats. Braz. J. Med. Biol. Res. 2009, 42, 902–911. [Google Scholar] [CrossRef] [PubMed]
- Ringel, R.E.; Hamlyn, J.M.; Hamilton, B.P.; Pinkas, G.A.; Chalew, S.A.; Berman, M.A. Red blood cell Na1, K1-ATPase in men with newly diagnosed or previously treated essential hypertension. Hypertension 1987, 9, 437–443. [Google Scholar] [CrossRef]
- Weiler, E.W.J.; Tuck, M.; Gonick, H.C. Observations on the “cascade” of Na-K-ATPase inhibitory and digoxin-like immunoreactive material in human urine: Possible relevance to essential hypertension. Clin. Exp. Hypertens. Theory Prac. 1985, A7, 809–836. [Google Scholar] [CrossRef]
- Tranquilli, A.L.; Mazzanti, L.; Ancona, A.; Brandi, S.; Bertoli, E.; Romanini, C. Inhibition of Na/K ATPase Activity in Maternal and Neonatal Erythrocyte Ghosts in Pregnancy-Induced Hypertension. Clin. Exp. Hypertens. Part B Hypertens. Pregnancy 1987, 6, 321–326. [Google Scholar] [CrossRef]
- Gonick, H.C.; Weiler, E.; Khalil-Manesh, F. Pattern of Na-K-ATPase inhibitors in plasma and urine of hypertensive patients: A preliminary report. Klin. Wochenschr. 1987, 65, 139–145. [Google Scholar] [PubMed]
- Vasdev, S.; Fernandez, P.G.; Longerich, L.; Gault, H. Higher plasma Na+, K+-ATPase inhibitory activity in essential hypertensive patients. Can. J. Cardiol. 1989, 5, 249–254. [Google Scholar]
- Miyagi, H.; Higuchi, M.; Nakayama, M.; Moromizato, H.; Sakanashi, M. Ouabain-like Na+, K(+)-ATPase inhibitory activity of a plasma extract in normal pregnancy and pregnancy induced hypertension. Jpn. J. Pharmacol. 1991, 57, 571–581. [Google Scholar] [CrossRef]
- Gonick, H.C.; Weiler, E.W.; Khalil-Manesh, F.; Weber, M.A. Predominance of high molecular weight plasma Na(+)-K(+)-ATPase inhibitor in essential hypertension. Am. J. Hypertens. 1993, 6, 680–687. [Google Scholar] [CrossRef]
- Kaplan, J.H. The sodium pump and hypertension: A physiological role for the cardiac glycoside binding site of the Na,K-ATPase. Proc. Natl. Acad. Sci. USA 2005, 102, 15723–15724. [Google Scholar] [CrossRef] [Green Version]
- Jaitovich, A.; Bertorello, A.M. Salt, Na+, K+-ATPase and hypertension. Life Sci. 2010, 86, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Waugh, D.T. The Contribution of Fluoride to the Pathogenesis of Eye Diseases: Molecular Mechanisms and Implications for Public Health. Int. J. Environ. Res. Public Health 2019, 16, 856. [Google Scholar] [CrossRef]
- Rajasekaran, S.A.; Huynh, T.P.; Wolle, D.G.; Espineda, C.E.; Inge, L.J.; Skay, A.; Lassman, C.; Nicholas, S.B.; Harper, J.F.; Reeves, A.E.; et al. Na, K-ATPase subunits as markers for epithelial-mesenchymal transition in cancer and fibrosis. Mol. Cancer Ther. 2010, 9, 1515–1524. [Google Scholar] [CrossRef] [Green Version]
- Blok, L.J.; Chang, G.T.; Steenbeek-Slotboom, M.; van Weerden, W.M.; Swarts, H.G.; De Pont, J.J.; van Steenbrugge, G.J.; Brinkmann, A.O. Regulation of expression of Na+, K+-ATPase in androgen-dependent and androgen-independent prostate cancer. Br. J. Cancer 1999, 81, 28–36. [Google Scholar] [CrossRef]
- Espineda, C.; Seligson, D.B.; James Ball, W., Jr.; Rao, J.; Palotie, A.; Horvath, S.; Huang, Y.; Shi, T.; Rajasekaran, A.K. Analysis of the Na, K-ATPase alpha- and beta-subunit expression profiles of bladder cancer using tissue microarrays. Cancer 2003, 97, 1859–1868. [Google Scholar] [CrossRef] [PubMed]
- Espineda, C.E.; Chang, J.H.; Twiss, J.; Rajasekaran, S.A.; Rajasekaran, A.K. Repression of Na,K-ATPase beta1-subunit by the transcription factor snail in carcinoma. Mol. Biol. Cell 2004, 15, 1364–1373. [Google Scholar] [CrossRef]
- Inge, L.J.; Rajasekaran, S.A.; Yoshimoto, K.; Mischel, P.S.; McBride, W.; Landaw, E.; Rajasekaran, A.K. Evidence for a potential tumor suppressor role for the Na, K-ATPase beta1-subunit. Histol. Histopathol. 2008, 23, 459–467. [Google Scholar]
- Rajasekaran, S.A.; Ball, W.J., Jr.; Bander, N.H.; Liu, H.; Pardee, J.D.; Rajasekaran, A.K. (Reduced expression of beta-subunit of Na,K-ATPase in human clear-cell renal cell carcinoma. J. Urol. 1999, 162, 574–580. [Google Scholar] [CrossRef]
- Sun, M.Z.; Kim, J.M.; Oh, M.C.; Safaee, M.; Kaur, G.; Clark, A.J.; Bloch, O.; Ivan, M.E.; Kaur, R.; Oh, T.; et al. Na+/K+-ATPase β2-subunit (AMOG) expression abrogates invasion of glioblastoma-derived brain tumor-initiating cells. Neuro Oncol. 2013, 15, 1518–1531. [Google Scholar] [CrossRef]
- Sakai, H.; Suzuki, T.; Maeda, M.; Takahashi, Y.; Horikawa, N.; Minamimura, T.; Tsukada, K.; Takeguchi, N. Up-regulation of Na(+), K(+)-ATPase alpha 3-isoform and down-regulation of the alpha1-isoform in human colorectal cancer. FEBS Lett. 2004, 563, 151–154. [Google Scholar] [CrossRef]
- Lees, G.J. Contributory mechanisms in the causation of neurodegenerative disorders. Neuroscience 1993, 54, 287–322. [Google Scholar] [CrossRef]
- Lees, G.J. Inhibition of sodium-potassium-ATPase: A potentially ubiquitous mechanism contributing to central nervous system neuropathology. Brain Res. Rev. 1991, 16, 283–300. [Google Scholar] [CrossRef]
- Bolotta, A.; Visconti, P.; Fedrizzi, G.; Ghezzo, A.; Marini, M.; Manunta, P.; Messaggio, E.; Posar, A.; Vignini, A.; Abruzzo, P.M. Na+, K+ -ATPase activity in children with autism spectrum disorder: Searching for the reason(s) of its decrease in blood cells. Autism Res. 2018, 11, 1388–1403. [Google Scholar] [CrossRef] [PubMed]
- Ghezzo, A.; Visconti, P.; Abruzzo, P.M.; Bolotta, A.; Ferreri, C.; Gobbi, G.; Mazzanti, L. Oxidative stress and erythrocyte membrane alterations in children with autism: Correlation with clinical features. PLoS ONE 2013, 8, e66418. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.N.; Sun, Y.J.; Pan, S.; Li, J.X.; Qu, Y.E.; Li, Y.; Wang, Y.L.; Gao, Z.B. Na+, K+-ATPase, a potent neuroprotective modulator against Alzheimer disease. Fundam. Clin. Pharmacol. 2013, 27, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Abruzzo, P.M.; Ghezzo, A.; Bolotta, A.; Ferreri, C.; Minguzzi, R.; Vignini, A.; Marini, M. Perspective biological markers for autism spectrum disorders: Advantages of the use of receiver operating characteristic curves in evaluating marker sensitivity and specificity. Dis. Markers 2015, 2015, 329607. [Google Scholar] [CrossRef]
- Kurup, R.K.; Kurup, P.A. A hypothalamic digoxin-mediated model for autism. Int. J. Neurosci. 2003, 113, 1537–1559. [Google Scholar] [CrossRef]
- Kumar, A.R.; Kurup, P.A. Endogenous sodium-potassium ATPase inhibition related biochemical cascade in trisomy 21 and Huntington’s disease: Neural regulation of genomic function. Neurol. India 2002, 50, 174–180. [Google Scholar]
- Ellis, D.Z.; Rabe, J.; Sweadner, K.J. Global loss of Na,K-ATPase and its nitric oxide-mediated regulation in a transgenic mouse model of amyotrophic lateral sclerosis. J. Neurosci. 2003, 23, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Naylor, G.J.; Smith, A.H.; Dick, E.G.; Dick, D.A.; McHarg, A.M.; Chambers, C.A. Erythrocyte membrane cation carrier in manic-depressive psychosis. Psycol. Med. 1980, 10, 521–525. [Google Scholar] [CrossRef]
- Hokin-Neaverson, M.; Jefferson, J.W. Deficient erythrocyte NaK-ATPase activity in different affective states in bipolar affective disorder and normalization by lithium therapy. Neuropsychobiology 1989, 22, 18–25. [Google Scholar] [CrossRef]
- Wood, A.J.; Smith, C.E.; Clarke, E.E.; Cowen, P.J.; Aronson, J.K.; Grahame-Smith, D.G. Altered in vitro adaptive responses of lymphocyte Na(+), K(+)-ATPase in patients with manic depressive psychosis. J. Affect. Disord. 1991, 21, 199–206. [Google Scholar] [CrossRef]
- Tochigi, M.; Iwamoto, K.; Bundo, M.; Sasaki, T.; Kato, N.; Kato, T. Gene expression profiling of major depression and suicide in the prefrontal cortex of postmortem brains. Neurosci. Res. 2008, 60, 184–191. [Google Scholar] [CrossRef] [PubMed]
- El-Mallakh, R.S.; Wyatt, R.J. The Na, K-ATPase hypothesis for bipolar illness. BiolPsychiatry 1995, 37, 235–244. [Google Scholar] [CrossRef]
- Bagrov, A.Y.; Bagrov, Y.Y.; Fedorova, O.V.; Kashkin, V.A.; Patkina, N.A.; Zvartau, E.E. Endogenous digitalis-like ligands of the sodium pump: Possible involvement in mood control and ethanol addiction. Eur. Neuropsychopharmacol. 2002, 12, 1–12. [Google Scholar] [CrossRef]
- Yao, J.K.; van Kammen, D.P. Red blood cell membrane dynamics in schizophrenia. I. Membrane fluidity. Schizophr. Res. 1994, 11, 209–216. [Google Scholar] [CrossRef]
- Kurup, R.K.; Kurup, P.A. Schizoid neurochemical pathology-induced membrane Na(+)-K+ ATPase inhibition in relation to neurological disorders. Int. J. Neurosci. 2003, 113, 1705–1717. [Google Scholar] [CrossRef]
- Corti, C.; Xuereb, J.H.; Crepaldi, L.; Corsi, M.; Michielin, F.; Ferraguti, F. Altered levels of glutamatergic receptors and Na+/K+ ATPase-alpha1 in the prefrontal cortex of subjects with schizophrenia. Schizophr. Res. 2011, 128, 7–14. [Google Scholar] [CrossRef]
- Chauhan, N.B.; Lee, J.M.; Siegel, G.J. Na, K-ATPase mRNA levels and plaque load in Alzheimer’s disease. J. Mol. Neurosci. 1997, 9, 151–166. [Google Scholar] [CrossRef]
- Hattori, N.; Kitagawa, K.; Higashida, T.; Yagyu, K.; Shimohama, S.; Wataya, T.; Perry, G.; Smith, M.A.; Inagaki, C. CI-ATPase and Na+/K(+)-ATPase activities in Alzheimer’s disease brains. Neurosci. Lett. 1998, 254, 141–144. [Google Scholar] [CrossRef]
- Sadanand, A.; Janardhanan, A.; Sankaradoss, A.; Vanisree, A.J.; Arulnambi, T.; Bhanu, K. Erythrocyte membrane in the evaluation of neurodegenerative disorders. Degener. Neurol. Neuromuscul. Dis. 2017, 7, 127–134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gamaro, G.D.; Streck, E.L.; Matte, C.; Prediger, M.E.; Wyse, A.T. Reduction of hippocampal Na+, K+-ATPase activity in rats subjected to an experimental model of depression. Neurochem. Res. 2003, 28, 1339–1344. [Google Scholar] [CrossRef] [PubMed]
- De Vasconcellos, A.P.; Zugno, A.I.; Dos Santos, A.H.; Nietto, F.B.; Crema, L.M.; Gonçalves, M.; Franzon, R.; de Souza Wyse, A.T.; da Rocha, E.R.; Dalmaz, C. Na+, K+-ATPase activity is reduced in hippocampus of rats submitted to an experimental model of depression: Effect of chronic lithium treatment and possible involvement in learning deficits. Neurobiol. Learn. Mem. 2005, 84, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Bedin, M.; Estrella, C.H.; Ponzi, D.; Duarte, D.V.; Dutra-Filho, C.S.; Wyse, A.T.; Wajner, M.; Wannmacher, C.M. Reduced Na(+), K(+)-ATPase activity in erythrocyte membranes from patients with phenylketonuria. Pediatr. Res. 2001, 50, 56–60. [Google Scholar] [CrossRef] [PubMed]
- Renkawek, K.; Renier, W.O.; de Pont, J.J.; Vogels, O.J.; Gabreels, F.J. Neonatal status convulsivus, spongiform encephalopathy, and low activity of Na+, K+-ATPase in the brain. Epilepsia 1992, 33, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Rapport, R.L.; Harris, A.B.; Friel, P.N.; Ojemann, G.A. Human epileptic brain Na, K ATPase activity and phenytoin concentrations. Arch. Neurol. 1975, 32, 549–554. [Google Scholar] [CrossRef] [PubMed]
- Wyse, A.T.S.; Streck, E.L.; Worm, P.; Wajner, A.; Ritter, F.; Netto, C.A. Preconditioning prevents the inhibition of Na+, K+-ATPase activity after brain ischemia. Neurochem. Res. 2000, 25, 971–975. [Google Scholar] [CrossRef]
- Carletti, J.V.; Deniz, B.F.; Miguel, P.M.; Rojas, J.J.; Kolling, J.; Scherer, E.B.; de Souza Wyse, A.T.; Netto, C.A.; Pereira, L.O. Folic Acid Prevents Behavioral Impairment and Na+, K+-ATPase Inhibition Caused by Neonatal Hypoxia–Ischemia. Neurochem. Res. 2012, 37, 1624–1630. [Google Scholar] [CrossRef] [PubMed]
- Desfrere, L.; Karlsson, M.; Hiyoshi, H.; Malmerjö, S.; Nanou, E.; Estrada, M.; Miyakawa, A.; Lagercrantz, H.; El Manira, A.; Lal, M.; et al. Na, K-ATPase signal transduction triggers CREB activation and dendritic growth. Proc. Natl. Acad. Sci. USA 2009, 106, 2212–2217. [Google Scholar] [CrossRef]
- Lees, G.J.; Leong, W. The sodium-potassium-ATPase inhibitor ouabain is neurotoxic in the rat substantia nigra and striatum. Neurosci. Lett. 1995, 188, 113–116. [Google Scholar] [CrossRef]
- Lees, G.J.; Leong, W. Brain lesions induced by specific and non-specific inhibitors of sodium-potassium ATPase. Brain Res. 1994, 649, 225–233. [Google Scholar] [CrossRef]
- Clapcote, S.J.; Duffy, S.; Xie, G.; Kirshenbaum, G.; Bechard, A.R.; Rodacker Schack, V.; Petersen, J.; Sinai, L.; Saab, B.J.; Lerch, J.P.; et al. Mutation I810N in the alpha3 isoform of Na+, K+-ATPase causes impairments in the sodium pump and hyperexcitability in the CNS. Proc. Natl. Acad. Sci. USA 2009, 106, 14085–14090. [Google Scholar] [CrossRef]
- Kirshenbaum, G.S.; Dawson, N.; Mullins, J.G.; Johnston, T.H.; Drinkhill, M.J.; Edwards, I.J.; Fox, S.H.; Pratt, J.A.; Brotchie, J.M.; Order, J.C.; et al. Alternating hemiplegia of childhood-related neural and behavioural phenotypes in Na+, K+-ATPase α3 missense mutant mice. PLoS ONE 2013, 8, e60141. [Google Scholar] [CrossRef] [PubMed]
- Lees, G.J.; Lehmann, A.; Sandberg, M.; Hamberger, A. The neurotoxicity of ouabain, a sodium-potassium ATPase inhibitor, in the rat hippocampus. Neurosci. Lett. 1990, 120, 159–162. [Google Scholar] [CrossRef]
- Brines, M.L.; Robbins, R.J. Inhibition of alpha 2/alpha 3 sodium pump isoforms potentiates glutamate neurotoxicity. Brain Res. 1992, 591, 94–102. [Google Scholar] [CrossRef]
- Zhang, D.; Hou, Q.; Wang, M.; Lin, A.; Jarzylo, L.; Navis, A.; Raissi, A.; Liu, F.; Man, H.Y. Na, K-ATPase activity regulates AMPA receptor turnover through proteasome-mediated proteolysis. J. Neurosci. 2009, 29, 4498–4511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murrough, J.W.; Abdallah, C.G.; Mathew, S.J. Targeting glutamate signalling in depression: Progress and prospects. Nat. Rev. Drug Discov. 2017, 16, 472–486. [Google Scholar] [CrossRef]
- De Berardis, D.; Fornaro, M.; Valchera, A.; Cavuto, M.; Perna, G.; Di Nicola, M.; Serafini, G.; Carano, A.; Pompili, M.; Vellante, F.; et al. Eradicating Suicide at Its Roots: Preclinical Bases and Clinical Evidence of the Efficacy of Ketamine in the Treatment of Suicidal Behaviors. Int. J. Mol. Sci. 2018, 19, 2888. [Google Scholar] [CrossRef]
- Tomasetti, C.; Iasevoli, F.; Buonaguro, E.F.; De Berardis, D.; Fornaro, M.; Fiengo, A.L.; Martinotti, G.; Orsolini, L.; Valchera, A.; Di Giannantonio, M.; et al. Treating the Synapse in Major Psychiatric Disorders: The Role of Postsynaptic Density Network in Dopamine-Glutamate Interplay and Psychopharmacologic Drugs Molecular Actions. Int. J. Mol. Sci. 2017, 18, 135. [Google Scholar] [CrossRef] [PubMed]
- Plitman, E.; Nakajima, S.; de la Fuente-Sandoval, C.; Gerretsen, P.; Chakravarty, M.M.; Kobylianskii, J.; Chung, J.K.; Caravaggio, F.; Iwata, Y.; Remington, G.; et al. Glutamate-mediated excitotoxicity in schizophrenia: A review. Eur. Neuropsychopharmacol. 2014, 24, 1591–1605. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lewerenz, J.; Maher, P. Chronic Glutamate Toxicity in Neurodegenerative Diseases-What is the Evidence? Front. Neurosci. 2015, 9, 469. [Google Scholar] [CrossRef]
- Pastural, E.; Ritchie, S.; Lu, Y.; Jin, W.; Kavianpour, A.; Khine Su-Myat, K.; Heath, D.; Wood, P.L.; Fisk, M.; Goodenowe, D.B. Novel plasma phospholipid biomarkers of autism: Mitochondrial dysfunction as a putative causative mechanism. Prostaglandins Leukot Essent Fat. Acids 2009, 81, 253–264. [Google Scholar] [CrossRef]
- Ghanizadeh, A. Targeting of glycine site on NMDA receptor as a possible new strategy for autism treatment. Neurochem. Res. 2012, 36, 922–923. [Google Scholar] [CrossRef] [PubMed]
- Stewart, M.; Lau, P.; Banks, G.; Bains, R.S.; Castroflorio, E.; Oliver, P.L.; Dixon, C.L.; Kruer, M.C.; Kullmann, D.M.; Acevedo-Arozena, A.; et al. Loss of Frrs1l disrupts synaptic AMPA receptor function, and results in neurodevelopmental, motor, cognitive and electrographical abnormalities. Dis. Models Mech. 2019, 12. [Google Scholar] [CrossRef]
- Dalsgaard, S.; Mortensen, P.B.; Frydenberg, M.; Maibing, C.M.; Nordentoft, M.; Thomsen, P.H. Association between Attention-Deficit Hyperactivity Disorder in childhood and schizophrenia later in adulthood. Eur. Psychiatry 2014, 29, 259–263. [Google Scholar] [CrossRef]
- Adisetiyo, V.; Tabesh, A.; Di Martino, A.; Falangola, M.F.; Castellanos, F.X.; Jensen, J.H.; Helpern, J.A. Attention-deficit/hyperactivity disorder without comorbidity is associated with distinct atypical patterns of cerebral microstructural development. Hum. Brain Mapp. 2014, 35, 2148–2162. [Google Scholar] [CrossRef] [PubMed]
- Paire, A.; Bernier-Valentin, F.; Rabilloud, R.; Watrin, C.; Selmi-Ruby, S.; Rousset, B. Expression of alpha- and beta-subunits and activity of Na+K+ ATPase in pig thyroid cells in primary culture: Modulation by thyrotropin and thyroid hormones. Mol. Cell. Endocrinol. 1998, 146, 93–101. [Google Scholar] [CrossRef]
- Hingorani, M.; Spitzweg, C.; Vassaux, G.; Newbold, K.; Melcher, A.; Pandha, H.; Vile, R.; Harrington, K. The Biology of the Sodium Iodide Symporter and its Potential for Targeted Gene Delivery. Curr. Cancer Drug Targets 2010, 10, 242–267. [Google Scholar] [CrossRef]
- Waugh, D.T. Fluoride Exposure Induces Inhibition of Sodium/Iodide Symporter (NIS) Contributing to Impaired Iodine Absorption and Iodine Deficiency: Molecular Mechanisms of Inhibition and Implications for Public Health. Int. J. Environ. Res. Public Health 2019, 16, 1086. [Google Scholar] [CrossRef]
- Zimmermann, M.B. Iodine deficiency. Endocr. Rev. 2009, 30, 376–408. [Google Scholar] [CrossRef] [PubMed]
- Delange, F. The disorders induced by iodine deficiency. Thyroid 1994, 4, 107–128. [Google Scholar] [CrossRef] [PubMed]
- Valcana, T.; Timiras, P.S. Effect of hypothyroidism on ionic metabolism and Na-K activated ATP phosphohydrolase activity in the developing rat brain. J. Neurochem. 1969, 16, 935–943. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, C.A.; MacDonough, A.A. Thyroid hormone regulates and isoforms of Na+, K+-ATPase during development in neonatal rat brain. J. Biol. Chem. 1988, 263, 17643–17649. [Google Scholar]
- Ahmed, O.M.; Abd El-Tawab, S.M.; Ahmed, R.G. Effects of experimentally induced maternal hypothyroidism and hyperthyroidism on the development of rat offspring: I. The development of the thyroid hormones-neurotransmitters and adenosinergic system interactions. Int. J. Dev. Neurosci. 2010, 28, 437–454. [Google Scholar] [CrossRef]
- Eayrs, J.T. The development of cerebral cortex in hypothyroid and starved rats. Anat. Rec. 1955, 121, 53–61. [Google Scholar] [CrossRef]
- Opit, L.J.; Potter, H.; Charnock, J.S. The effect of anions on (Na+ + K+)-activated. ATPase. Biochim. Biophys. Acta 1966, 120, 159–161. [Google Scholar] [CrossRef]
- Yoshida, H.; Nagai, K.; Kamei, M.; Nakagawa, Y. Irreversible inactivation of (Na+-K+)-dependent ATPase and K+-dependent phosphatase by fluoride. Biochim. Biophys. Acta 1968, 150, 162–164. [Google Scholar] [CrossRef]
- Millman, M.S.; Omachi, A. The Role of Oxidized Nicotinamide Adenine Dinucleotide in Fluoride Inhibition of Active Sodium Transport in Human Erythrocytes. J. Gen. Physiol. 1972, 60, 337–350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iukhnovets, R.A.; Bachinskiĭ, P.P. Effect of fluoride and insulin on cation-dependent ATPase activity of the enterocytes during threonine absorption. Vopr. Med. Khim. 1982, 28, 46–50. [Google Scholar] [PubMed]
- Robinson, J.D.; Davis, R.L.; Steinberg, M. Fluoride and beryllium interact with the (Na + Kbdependent ATPase as analogs of phosphate. J. Bioenerg. Biomembr. 1986, 18, 521–531. [Google Scholar] [CrossRef] [PubMed]
- Swann, A.C. Inhibition of (Na+, K+)-ATPase by fluoride: Evidence for a membrane adaptation to ethanol. Alcohol 1990, 7, 91–95. [Google Scholar] [CrossRef]
- Murphy, A.J.; Hoover, J.C. Inhibition of the Na/K-ATPase by fluoride. Parallels with its inhibition of the sarcoplasmic reticulum CaATPase. J. Biol. Chem. 1992, 267, 16995–17000. [Google Scholar]
- Façanha, A.R.; de Meis, L. Inhibition of Maize Root H+-ATPase by Fluoride and Fluoroaluminate Complexes. Plant. Physiol. 1995, 108, 241–246. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suketa, Y.; Suzuki, K.; Taki, T.; Itoh, Y.; Yamaguchi, M.; Sakurai, T.; Tanishita, Y. Effect of fluoride on the activities of the Na+/glucose cotransporter and Na+/K(+)-ATPase in brush border and basolateral membranes of rat kidney (in vitro and in vivo). Biol. Pharm. Bull. 1995, 18, 273–278. [Google Scholar] [CrossRef]
- Liu, G.Y.; Chai, C.Y.; Kang, S.L. Effects of Fluoride on the Activity of ATPase on Erythrocytic membrane in Chicks. Heilongjinag J. Anim Sci. Vet. Med. 2002, 8. Available online: http://en.cnki.com.cn/Article_en/CJFDTOTAL-HLJX200208001.htm (accessed on 28 February 2019).
- Ekambaram, P.; Paul, V. Modulation of fluoride toxicity in rats by calcium carbonate and by withdrawal of fluoride exposure. Pharmacol. Toxicol. 2002, 90, 53–58. [Google Scholar] [CrossRef]
- Cittanova, M.L.; Estepa, L.; Bourbouze, R.; Blanc, O.; Verpont, M.C.; Wahbe, E.; Coriat, P.; Daudon, M.; Ronco, P.M. Fluoride ion toxicity in rabbit kidney thick ascending limb cells. Eur. J. Anaesthesiol. 2002, 19, 341–349. [Google Scholar] [CrossRef]
- Kravtsova, V.V.; Kravtsov, O.V. Inactivation of Na+, K+-ATPase from cattle brain by sodium fluoride. Ukr. Biokhim. Zhurnal 2004, 76, 39–47. [Google Scholar]
- Zhan, X.A.; Li, J.X.; Wang, M.; Xu, Z.R. Effects of Fluoride on Growth and Thyroid Function in Young Pigs. Fluoride 2006, 39, 95–100. [Google Scholar]
- Agalakova, N.I.; Gusev, G.P. Diverse effects of fluoride on Na+ and K+ transport across the rat erythrocyte membrane. Fluoride 2008, 41, 28–39. [Google Scholar]
- Agalakova, N.I.; Gusev, G.P. Fluoride-induced death of rat erythrocytes in vitro. Toxicol. In Vitro 2011, 25, 1609–1618. [Google Scholar] [CrossRef]
- Agalakova, N.I.; Gusev, G.P. Molecular Mechanisms of Cytotoxicity and Apoptosis Induced by Inorganic Fluoride. ISRN Cell Biol. 2012, 2012, 403835. [Google Scholar] [CrossRef]
- Sarkar, C.; Pal, S. Ameliorative effect of resveratrol against fluoride-induced alteration of thyroid function in male wistar rats. Biol. Trace Elem. Res. 2014, 162, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, C.; Pal, S. Effects of sub-acute fluoride exposure on discrete regions of rat brain associated with thyroid dysfunction: A comparative study. Int. J. Biomed. Res. 2015, 6, 647–660. [Google Scholar] [CrossRef]
- Mondragão, M.A.; Schmidt, H.; Kleinhans, C.; Langer, J.; Kafitz, K.W.; Rose, C.R. Extrusion versus diffusion: Mechanisms for recovery from sodium loads in mouse CA1 pyramidal neurons. J. Physiol. 2016, 594, 5507–5527. [Google Scholar] [CrossRef]
- Arulkumar, M.; Vijayan, R.; Penislusshiyan, S.; Sathishkumar, P.; Angayarkanni, J.; Palvannan, T. Alteration of paraoxonase, arylesterase and lactonase activities in people around fluoride endemic area of Tamil Nadu, India. Clin. Chim. Acta 2017, 471, 206–215. [Google Scholar] [CrossRef]
- Shashi, A.; Meenakshi, G. Inhibitory Effect of Fluoride on Na+, K+ ATPase Activity in Human Erythrocyte Membrane. Biol. Trace Elem. Res. 2015, 168, 340–348. [Google Scholar]
- Peckham, S.; Lowery, D.; Spencer, S. Are fluoride levels in drinking water associated with hypothyroidism prevalence in England? A large observational study of GP practice data and fluoride levels in drinking water. J. Epidemiol. Community Health 2015, 69, 619–624. [Google Scholar] [CrossRef] [Green Version]
- Kheradpisheh, Z.; Mirzaei, M.; Mahvi, A.H.; Mokhtari, M.; Azizi, R.; Fallahzadeh, H.; Ehrampoush, M.H. Impact of Drinking Water Fluoride on Human Thyroid Hormones: A Case—Control Study. Sci. Rep. 2018, 8, 2674. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA). Scientific Opinion on Dietary Reference Values for fluoride. EFSA J. 2013, 11, 3332. [Google Scholar] [CrossRef] [Green Version]
- Recommendations for Using Fluoride to Prevent and Control Dental Caries in the United States; MMWR; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2001; Volume 50, pp. 1–42.
- Ten Cate, J.M. In vitro studies on the effects of fluoride on de- and remineralization. J. Dent. Res. 1990, 69, 614–619. [Google Scholar] [CrossRef]
- Water Fluoridation for the Prevention of Dental Caries (Review); The Cochrane Library: Hoboken, NJ, USA, 2015; Available online: https://www.cochrane.org/CD010856/ORAL_water-fluoridation-prevent-tooth-decay (accessed on 15 February 2019).
- Opinion of the Scientific Committee on Cosmetic Products and Non-Food Products Intended for Consumers Concerning the Safety of Fluorine Compounds in Oral Hygiene Products for Children under the Age of 6 Years. June 2003. SCCNFP/0653/03: Final. Available online: https://ec.europa.eu/health/phrisk/committees/04sccp/docs/sccpo024.pdf (accessed on 15 February 2019).
- Sutton, S.V.; Bender, G.R.; Marquis, R.E. Fluoride Inhibition of Proton-Translocating ATPases of Oral Bacteria. Infect. Immun. 1987, 55, 2597–2603. [Google Scholar] [PubMed]
- Sturr, M.G.; Marquis, R.E. Inhibition of proton-translocating ATPases of Streptococcus mutans and Lactobacillus casei by fluoride and aluminum. Arch. Microbiol. 1990, 155, 22–27. [Google Scholar] [CrossRef]
- Marquis, R.E. Diminished acid tolerance of plaque bacteria caused by fluoride. J. Dent. Res. 1990, 69, 672–675. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, I.R. Biochemical effects of fluoride on oral bacteria. J. Dent. Res. 1990, 69, 660–667. [Google Scholar] [CrossRef]
- National Research Council. Fluoride in Drinking Water: A Scientific Review of EPA’s Standards; The National Academies Press: Washington, DC, USA, 2006. [Google Scholar]
- Chan, L.; Mehra, A.; Saikat, S.; Lynch, P. Human exposure assessment of fluoride from tea (Camellia sinensis L.). Food Res. Int. 2013, 51, 564–570. [Google Scholar] [CrossRef]
- Waugh, D.T.; Potter, W.; Limeback, H.; Godfrey, M. Risk assessment of fluoride intake from tea in the republic of Ireland and its implications for public health and water fluoridation. Int. J. Environ. Res. Public Health 2016, 13, 259. [Google Scholar] [CrossRef] [PubMed]
- Waugh, D.T.; Godfrey, M.; Limeback, H.; Potter, W. Black Tea Source, Production, and Consumption: Assessment of Health Risks of Fluoride Intake in New Zealand. J. Environ. Public Health 2017, 2017, 5120504. [Google Scholar] [CrossRef] [PubMed]
- Dabeka, R.W.; Karpinski, K.; McKenzie, A.; Bajdik, C. Survey of lead, cadmium and fluoride in human milk and correlation of levels with environmental and food factors. Food Chem. Toxicol. 1986, 24, 913–921. [Google Scholar] [CrossRef]
- Fomon, S.J.; Ekstrand, J. Fluoride Intake by Infants. J. Public Health Dentristry 1999, 59, 229–234. [Google Scholar] [CrossRef]
- Gupta, P.; Gupta, N.; Meena, K.; Moon, N.J.; Kumar, P.; Kaur, R. Concentration of Fluoride in Cow’s and Buffalo’s Milk in Relation to Varying Levels of Fluoride Concentration in Drinking Water of Mathura City in India—A Pilot Study. J. Clin. Diagn Res. 2015, 9, LC05–LC07. [Google Scholar] [CrossRef]
- Zohoori, V.; Maguire, A. Database of the Fluoride (F)content of Selected Drinks and Foods in the UK; Newscastle University: Newcastle upon Tyne, UK; Teeside University: Middlesbrough, UK, 2015; Available online: https://www.tees.ac.uk/docs/docrepo/research/finalfluoridedatabase.pdf (accessed on 15 March 2019).
- Suttie, J.W.; Miller, R.F.; Phillips, P.H. Effects of dietary sodium fluoride on dairy cows. 1. The physiological effects and the development symptoms of fluorosis. J. Nutr. 1957, 63, 211–224. [Google Scholar] [CrossRef]
- Zohoori, F.V.; Moynihan, P.J.; Omid, N.; Abuhaloob, L.; Maguire, A. Impact of water fluoride concentration on the fluoride content of infant foods and drinks requiring preparation with liquids before feeding. Community Dent. Oral Epidemiol. 2012, 40, 432–440. [Google Scholar] [CrossRef]
- Johnson, J., Jr.; Bawden, J.W. The fluoride content of infant formulas available in 1985. Pediatr. Dent. 1987, 9, 33–37. [Google Scholar]
- Siew, C.; Strock, S.; Ristic, H.; Kang, P.; Chou, H.; Chen, J.; Frantsve-Hawley, J.; Meyer, D.M. Assessing a potential risk factor for enamel fluorosis: A preliminary evaluation of fluoride content in infant formulas. J. Am. Dent. Assoc. 2009, 140, 1228–1236. [Google Scholar] [CrossRef]
- McKnight-Hanes, M.C.; Leverett, D.H.; Adair, S.M.; Shields, C.P. Fluoride content of infant formulas: Soy-based formulas as a potential factor in dental fluorosis. Fluoride content of infant formulas: Soy-based formulas as a potential factor in dental fluorosis. Pediatr. Dent. 1988, 10, 189–194. [Google Scholar]
- Hossny, E.; Reda, S.; Marzouk, S.; Diab, D.; Fahmy, H. Serum fluoride levels in a group of Egyptian infants and children from Cairo city. Arch. Environ. Health. 2003, 58, 306–315. [Google Scholar] [CrossRef]
- Warady, B.A.; Koch, M.; O’Neal, D.W.; Higginbotham, M.; Harris, D.J.; Hellerstein, S. Plasma fluoride concentration in infants receiving long-term peritoneal dialysis. J. Pediatr. 1989, 115, 436–439. [Google Scholar] [CrossRef]
- Anderson, W.A.; Pratt, I.; Ryan, M.R.; Flynn, A. A probabilistic estimation of fluoride intake by infants up to the age of 4 months from infant formula reconstituted with tap water in the fluoridated regions of Ireland. Caries Res. 2004, 38, 421–429. [Google Scholar] [CrossRef]
- Harriehausen, C.X.; Dosani, F.Z.; Chiquet, B.T.; Barratt, M.S.; Quock, R.L. Fluoride Intake of Infants from Formula. J. Clin. Pediatr. Dent. 2019, 43, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Aperia, A.; Broberger, O.; Herin, P.; Thodenius, K.; Zetterström, R. Postnatal control of water and electrolyte homeostasis in pre-term and full-term infants. Acta Paediatr. Scand. Suppl. 1983, 305, 61–65. [Google Scholar] [CrossRef]
- Arant, B.S., Jr. Postnatal development of renal function during the first year of life. Pediatr. Nephrol. 1987, 1, 308–313. [Google Scholar] [CrossRef]
- Ekstrand, J.; Fomon, S.J.; Ziegler, E.E.; Nelson, S.E. Fluoride Pharmacokinetics in Infancy. Pediatr. Res. 1994, 35, 157–163. [Google Scholar] [CrossRef] [Green Version]
- Ehrnebo, M.; Ekstrand, J. Occupational fluoride exposure and plasma fluoride levels in man. Int. Arch. Occup. Environ. Health 1986, 58, 179–190. [Google Scholar] [CrossRef]
- Kono, K.; Yoshisda, Y.; Watanabe, M.; Usada, K.; Nagaie, H.; Takahashi, Y. Serum and urine monitoring of fluoride exposed workers in aluminium smelting industry. J. Environ. Sci. 1996, 8, 242–248. [Google Scholar]
- Lobo, J.G.V.M.; Leite, A.L.; Pereira, H.A.B.S.; Fernandes, M.S.; Peres-Buzalaf, C.; Sumida, D.H.; Rigalli, A.; Buzalaf, M.A.R. Low-Level Fluoride Exposure Increases Insulin Sensitivity in Experimental Diabetes. J. Dent. Res. 2015, 94, 990–997. [Google Scholar] [CrossRef] [PubMed]
- Da Silva Pereira, H.A.; de Lima Leite, A.; Charone, S.; Lobo, J.G.; Cestari, T.M.; Peres-Buzalaf, C.; Buzalaf, M.A. Proteomic Analysis of Liver in Rats Chronically Exposed to Fluoride. PLoS ONE 2013, 8, e75343. [Google Scholar]
- Amaral, S.L.; Azevedo, L.B.; Buzalaf, M.A.; Fabricio, M.F.; Fernandes, M.S.; Valentine, R.A.; Maguire, A.; Zohoori, F.V. Effect of chronic exercise on fluoride metabolism in fluorosis-susceptible mice exposed to high fluoride. Sci. Rep. 2018, 8, 3211. [Google Scholar] [CrossRef]
- Trautner, K.; Siebert, G. An experimental study of bio-availability of fluoride from dietary sources in man. Arch. Oral Biol. 1986, 31, 223–228. [Google Scholar] [CrossRef]
- Toyota, S. Fluorine content in the urine and in the serum of hydrofluoric acid workers as an index of health administration. Sangyo Igaku 1979, 21, 335–348. [Google Scholar] [CrossRef] [PubMed]
- Ekstrand, J.; Alvan, G.; Boreus, L.; Norlin, A. Pharmacokinetics of fluoride in man after single and multiple oral doses. Eur. J. Clin. Pharmacol. 1977, 12, 311–317. [Google Scholar] [CrossRef]
- Ekstrand, J.; Ehrnebo, M.; Boreus, L. Fluoride bioavailability after intravenous and oral administration: Importance of renal clearance and urine flow. Clin. Pharmacol. Ther. 1978, 23, 329–371. [Google Scholar] [CrossRef] [PubMed]
- Ekstrand, J. Relationship between fluoride in the drinking water and the plasma fluoride concentration in man. Caries Res. 1978, 12, 123–127. [Google Scholar] [CrossRef]
- Reddy, P.V. Organofluorine Compounds in Biology and Medicine; Elsevier: Waltham, MA, USA, 2015; ISBN 978-0-444-53748-5. [Google Scholar]
- Skiles, J.L.; Imel, E.A.; Christenson, J.C.; Bell, J.E.; Hulbert, M.L. Fluorosis because of prolonged voriconazole therapy in a teenager with acute myelogenous leukemia. J. Clin. Oncol. 2011, 29, e779–e782. [Google Scholar] [CrossRef] [PubMed]
- Wermers, R.A.; Cooper, K.; Razonable, R.R.; Deziel, P.J.; Whitford, G.M.; Kremers, W.K.; Moyer, T.P. Fluoride Excess and Periostitis in Transplant Patients Receiving Long-Term Voriconazole Therapy. Clin. Infect. Dis. 2011, 52, 604–611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perbet, S.; Salavert, M.; Amarger, S.; Constantin, J.M.; D’ Incan, M.; Bazin, J.E. Fluoroderma after exposure to sevoflurane. Br. J. Anaesth. 2011, 107, 106–107. [Google Scholar] [CrossRef] [Green Version]
- Goldberg, M.E.; Cantillo, J.; Larijani, G.E.; Torjman, M.; Vekeman, D.; Schieren, H. Sevoflurane versus isoflurane for maintenance of anesthesia: Are serum inorganic fluoride ion concentrations of concern? Anesth. Analg. 1996, 82, 1268–1272. [Google Scholar]
- Ekstrand, J.; Koch, G. Systemic fluoride absorption following fluoride gel application. J. Dent. Res. 1980, 59, 1067. [Google Scholar] [CrossRef]
- Ekstrand, J.; Koch, G.; Lindgren, L.E.; Petersson, L.G. Pharmacokinetics of fluoride gels in children and adults. Caries Res. 1981, 15, 213–220. [Google Scholar] [CrossRef]
- Ekstrand, J.; Ehrnebo, M. Absorption of fluoride from fluoride dentifrices. Caries Res. 1980, 14, 96–102. [Google Scholar] [CrossRef]
- Zohoori, F.V.; Innerd, A.; Azevedo, L.B.; Whitford, G.M.; Maguire, A. Effect of exercise on fluoride metabolism in adult humans: A pilot study. Sci. Rep. 2015, 5, 16905. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, A.P.M. Low Fluoride Concentrations: Their Relevance to the Inhibition of Dental Caries. Ph.D. Thesis, Faculty of Medicine, The University of Glasgow, Glasgow, UK, 1995. Available online: http://theses.gla.ac.uk/2479/1/1995jacobsonphd.pdf (accessed on 12 December 2018).
- Kurland, E.S.; Schulman, R.C.; Zerwekh, J.E.; Reinus, W.R.; Dempster, D.W.; Whyte, M.P. Recovery from skeletal fluorosis (an enigmatic, American case). J. Bone Min. Res. 2007, 22, 163–170. [Google Scholar] [CrossRef] [PubMed]
- European Academy of Paediatric Dentistry. Guidelines on the use of fluoride in children: An EAPD policy document. Eur. Arch. Paediatr. Dent. 2009, 10, 129–135. [Google Scholar] [CrossRef]
- Irish Oral Health Services Guideline Initiative. Topical Fluorides: Evidence-Based Guidance on the Use of Topical Fluorides for Caries Prevention in Children and Adolescents in Ireland. 2008. Available online: https://www.ucc.ie/en/media/research/ohsrc/TopicalFluoridesFull.pdf (accessed on 12 January 2019).
- American Academy of Pediatric Dentistry. Policy on early childhood caries (ECC): Classifications, consequences, and preventive strategies. Pediatr. Dent. 2008, 30, 40–42. [Google Scholar]
- Levy, S.M.; McGrady, J.A.; Bhuridej, P.; Warren, J.J.; Heilman, J.R.; Wefel, J.S. Factors affecting dentifrice use and ingestion among a sample of U.S. preschoolers. Pediatr. Dent. 2000, 22, 389–394. [Google Scholar]
- Hargreaves, J.A.; Ingram, G.S.; Wagg, B.J. A Gravimetric Study of the Ingestion of Toothpaste by Children. Caries Res. 1972, 6, 237–243. [Google Scholar] [CrossRef]
- Omid, N.; Maguire, A.; O’Hare, W.T.; Zohoori, F.V. Total daily fluoride intake and fractional urinary fluoride excretion in 4- to 6-year-old children living in a fluoridated area: Weekly variation? Community Dent. Oral Epidemiol. 2017, 45, 12–19. [Google Scholar] [CrossRef]
- Kaplan, J.H. Biochemistry of Na,K-ATPase. Annu. Rev. Biochem. 2002, 71, 511–535. [Google Scholar] [CrossRef] [PubMed]
- Sweadner, K.J. Isozymes of the Na+/K+-ATPase. Biochim. Biophys. Acta 1989, 988, 185–220. [Google Scholar] [CrossRef]
- Blanco, G.; Mercer, R.W. Isozymes of the Na-K-ATPase: Heterogeneity in structure, diversity in function. Am. J. Physiol. 1998, 275 Pt 2, F633–F650. [Google Scholar] [CrossRef]
- Skou, J.C.; Esmann, M. The Na, K-ATPase. J. Bioenerg. Biomembr. 1992, 24, 249–261. [Google Scholar] [PubMed]
- McDonough, A.A.; Geering, K.; Farley, R.A. The sodium pump needs its beta subunit. FASEB J. 1990, 4, 1598–1605. [Google Scholar] [CrossRef] [PubMed]
- Mercer, R.W. Structure of the Na, K-ATPase. Int. Rev. Cytol. 1993, 137C, 139–168. [Google Scholar] [PubMed]
- Scheiner-Bobis, G. The sodium pump. Its molecular properties and mechanics of ion transport. Eur. J. Biochem. 2002, 269, 2424–2433. [Google Scholar] [CrossRef] [PubMed]
- Bertorello, A.M.; Aperia, A.; Walaas, S.I.; Nairn, A.C.; Greengard, P. Phosphorylation of the catalytic subunit of Na+, K+-ATPase inhibits the activity of the enzyme. Proc. Natl. Acad. Sci. USA 1991, 88, 11359–11362. [Google Scholar] [CrossRef]
- Bertorello, A.; Aperia, A. Na+-K+-ATPase is an effector protein for protein kinase C in renal proximal tubule cells. Am. J. Physiol. 1989, 256 Pt 2, F370–F373. [Google Scholar] [CrossRef]
- Fukuda, Y.; Bertorello, A.; Aperia, A. Ontogeny of the regulation of Na+, K(+)-ATPase activity in the renal proximal tubule cell. Pediatr. Res. 1991, 30, 131–134. [Google Scholar] [CrossRef] [PubMed]
- Beguin, P.; Beggah, A.T.; Chibalin, A.V.; Burgener-Kairuz, P.; Jaisser, F.; Mathews, P.M.; Rossier, B.C.; Cotecchia, S.; Geering, K. Phosphorylation of the Na,K-ATPase alpha-subunit by protein kinase A and C in vitro and in intact cells. Identification of a novel motif for PKC-mediated phosphorylation. J. Biol. Chem. 1994, 269, 24437–24445. [Google Scholar]
- Marcaida, G.; Kosenko, E.; Miñana, M.D.; Grisolía, S.; Felipo, V. Glutamate induces a calcineurin-mediated dephosphorylation of Na+,K(+)-ATPase that results in its activation in cerebellar neurons in culture. J. Neurochem. 1996, 66, 99–104. [Google Scholar] [CrossRef]
- Cheng, X.J.; Höög, J.O.; Nairn, A.C.; Greengard, P.; Aperia, A. Regulation of rat Na(+)-K(+)-ATPase activity by PKC is modulated by state of phosphorylation of Ser-943 by PKA. Am. J. Physiol. 1997, 273 Pt 1, C1981–C1986. [Google Scholar] [CrossRef]
- Chibalin, A.V.; Pedemonte, C.H.; Katz, A.I.; Féraille, E.; Berggren, P.O.; Bertorello, A.M. Phosphorylation of the catalyic alpha-subunit constitutes a triggering signal for Na+, K+-ATPase endocytosis. J. Biol. Chem. 1998, 273, 8814–8819. [Google Scholar] [CrossRef] [PubMed]
- Nishi, A.; Fisone, G.; Snyder, G.L.; Dulubova, I.; Aperia, A.; Nairn, A.C.; Greengard, P. Regulation of Na+, K+-ATPase isoforms in rat neostriatum by dopamine and protein kinase C. J. Neurochem. 1999, 73, 1492–1501. [Google Scholar] [CrossRef]
- Taub, M.; Springate, J.E.; Cutuli, F. Targeting of renal proximal tubule Na,K-ATPase by salt-inducible kinase. Biochem. Biophys. Res. Commun. 2010, 393, 339–344. [Google Scholar] [CrossRef]
- Liu, T.; Konkalmatt, P.R.; Yang, Y.; Jose, P.A. Gastrin decreases Na+, K+-ATPase activity via a PI3 kinase- and PKC-dependent pathway in human renal proximal tubule cells. Am. J. Physiol. Endocrinol. Metab. 2016, 310, E565–E571. [Google Scholar] [CrossRef]
- Saha, P.; Manoharan, P.; Arthur, S.; Sundaram, S.; Kekuda, R.; Sundaram, U. Molecular mechanism of regulation of villus cell Na-K-ATPase in the chronically inflamed mammalian small intestine. Biochim. Biophys. Acta 2015, 1848, 702–711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zatyka, M.; Ricketts, C.; da Silva Xavier, G.; Minton, J.; Fenton, S.; Hofmann-Thiel, S.; Rutter, G.A.; Barrett, T.G. Sodium-potassium ATPase 1 subunit is a molecular partner of Wolframin, an endoplasmic reticulum protein involved in ER stress. Hum. Mol. Genet. 2008, 17, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, X.; Xu, Z. Endoplasmic Reticulum Stress in Hearing Loss. J. Otorhinolaryngol. Hear. Balance Med. 2018, 1, 3. [Google Scholar] [CrossRef]
- Kobayashi, C.A.; Leite, A.L.; Peres-Buzalaf, C.; Carvalho, J.G.; Whitford, G.M.; Everett, E.T.; Siqueira, W.L.; Buzalaf, M.A. Bone response to fluoride exposure is influenced by genetics. PLoS ONE 2014, 11, e114343. [Google Scholar] [CrossRef]
- Poulsen, H.; Morth, P.; Egebjerg, J.; Nissen, P. Phosphorylation of the Na+, K+-ATPase and the H+, K+-ATPase. FEBS Lett. 2010, 584, 2589–2595. [Google Scholar] [CrossRef] [PubMed]
- Krasilnikova, O.A.; Kavok, N.S.; Babenko, N.A. Drug-induced and postnatal hypothyroidism impairs the accumulation of diacylglycerol in liver and liver cell plasma membranes. BMC Physiol. 2002, 2, 12. [Google Scholar] [CrossRef]
- Hermenegildo, C.; Felipo, V.; Miñana, M.D.; Grisolía, S. Inhibition of protein kinase C restores Na+, K+-ATPase activity in sciatic nerve of diabetic mice. J. Neurochem. 1992, 58, 1246–1249. [Google Scholar] [CrossRef] [PubMed]
- Hermenegildo, C.; Felipo, V.; Miñana, M.D.; Romero, F.J.; Grisolía, S. Sustained recovery of Na(+)-K(+)-ATPase activity in the sciatic nerve of diabetic mice by administration of H7 or calphostin C, inhibitors of PKC. Diabetes 1993, 42, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Bocanera, L.V.; Krawiec, L.; Nocetti, G.; Juvenal, G.J.; Silberschmidt, D.; Pisarev, M.A. The protein kinase C pathway inhibits iodide uptake by calf thyroid cells via sodium potassium-adenosine triphosphatase. Thyroid 2001, 11, 813–817. [Google Scholar] [CrossRef] [PubMed]
- Rybin, V.; Steinberg, S.F. Thyroid hormone represses protein kinase C isoform expression and activity in rat cardiac myocytes. Circ. Res. 1996, 79, 388–398. [Google Scholar] [CrossRef] [PubMed]
- Meier, C.A.; Fabbro, D.; Meyhack, I.; Hemmings, B.; Olbrecht, U.; Jakob, A.; Walter, P. Effect of hypothyroidism and thyroid hormone replacement on the level of protein kinase C and protein kinase A in rat liver. FEBS Lett. 1991, 282, 397–400. [Google Scholar] [CrossRef] [Green Version]
- Agalakova, N.I.; Gusev, G.P. Transient activation of protein kinase C contributes to fluoride-induced apoptosis of rat erythrocytes. Toxicol. In Vitro 2013, 27, 2335–2341. [Google Scholar] [CrossRef]
- Refsnes, M.; Kersten, H.; Schwarze, P.E.; Lag, M. Involvement of protein kinase C in fluoride-induced apoptosis in different types of lung cells. Ann. N. Y. Acad. Sci. 2002, 973, 218–220. [Google Scholar] [CrossRef] [PubMed]
- Chirumari, K.; Reddy, P.K. Dose-Dependent Effects of Fluoride on Neurochemical Milieu in the Hippocampus and Neocortex of Rat Brain. Fluoride 2007, 40, 101–110. [Google Scholar]
- Refsnes, M.; Schwarze, P.E.; Holme, J.A.; Låg, M. Fluoride-induced apoptosis in human epithelial lung cells (A549 cells): Role of different G protein-linked signal systems. Hum. Exp. Toxicol. 2003, 22, 111–123. [Google Scholar] [CrossRef]
- Hauschildt, S.; Hirt, W.; Bessler, W. Modulation of protein kinase C activity by NaF in bone marrow derived macrophages. FEBS Lett. 1988, 230, 121–124. [Google Scholar] [CrossRef] [Green Version]
- Murthy, K.S.; Makhlouf, G.M. Fluoride activates G protein-dependent and -independent pathways in dispersed intestinal smooth muscle cells. Biochem. Biophys. Res. Commun. 1994, 202, 1681–1687. [Google Scholar] [CrossRef] [PubMed]
- Bertorello, A.; Aperia, A. Short-term regulation of Na+, K+-ATPase activity by dopamine. Am. J. Hypertension. 1990, 3, 51S–54S. [Google Scholar] [CrossRef]
- Aperia, A.; Holtback, U.; Syren, M.L.; Svensson, L.B.; Fryckstedt, J.; Greengard, P. Activation/deactivation of renal Na-K ATPase: A final common pathway for regulation of natriuresis. FASEB J. 1994, 8, 436–439. [Google Scholar] [CrossRef] [PubMed]
- Delamere, N.A.; King, K.L. The Influence of Cyclic AMP Upon Na,K-ATPase Activity in Rabbit Ciliary Epithelium. Investig. Ophthalmol. Vis. Sci. 1992, 33, 430–435. [Google Scholar]
- Tipsmark, C.K.; Madsen, S.S. Rapid Modulation of Na+/K+-ATPase Activity in Osmoregulatory Tissues of a Salmonid Fish. J. Exp. Biol. 2001, 204, 701–709. [Google Scholar] [PubMed]
- Chiba, F.Y.; Garbin, C.A.S.; Sumida, D.H. Effect of Fluoride intake on Carbohydrate Metabolism, Glucose Tolerance, and Insulin Signaling. Fluoride 2012, 45, 236–241. [Google Scholar]
- Preedy, V.R. Fluorine: Chemistry, Analysis, Function and Effects; The Royal Society of Chemistry: Cambridge, UK, 2015. [Google Scholar]
- Mornstad, H.; Sundstrom, B.; Hedner, R. Increased Urinary Excretion of cAMP Following Administration of Sodium Fluoride. J. Dent. Res. 1975, 54. AbstractL39. [Google Scholar]
- Kornegay, D.; Pennington, S. A Review of the Effect of Fluoride ion on Adenyl Cyclase. Fluoride 1973, 6, 19–32. [Google Scholar]
- Isenberg, K.; Allmann, D.W. Effect of Inorganic Fluoride on Urinary Excretion of 35-Cyclic AMP. Clin. Res. 1976, 24, 601. [Google Scholar]
- Edgar, W.; Jenkins, G.N.; Prudhoe, P. Urinary cAMP Excretion in Human Subjects Following Single and Divided Doses of Sodium Fluoride. J. Dent. Res. 1979, 58, 1229. [Google Scholar]
- Mornstad, H.; Van Dijken, J. Caries Preventive Doses of Fluoride and Cyclic AMP Levels in Human Plasma. Caries Res. 1982, 16, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Shashi, A.; Bhardwaj, M. Study on blood biochemical diagnostic indices for hepatic function biomarkers in endemic skeletal fluorosis. Biol. Trace Elem. Res. 2011, 143, 803–814. [Google Scholar] [CrossRef]
- Mörnstad, H.; van Dijken, J. The Effect of Low Doses of Fluoride on the Content of Cyclic AMP and Amylase in Human Parotid Saliva. Caries Res. 1985, 19, 433–438. [Google Scholar] [CrossRef]
- Kleiner, H.S.; Allmann, D.W. The effects of fluoridated water on rat urine and tissue cAMP levels. Arch. Oral Biol. 1982, 27, 107–112. [Google Scholar] [CrossRef]
- Allmann, D.W.; Dunipace, A.; Curro, F.A. Stimulation of cAMP production by NaF in isolated rat aorta. J. Dent. Res. 1982, 61, 321. [Google Scholar]
- Kleiner, H.S.; Miller, A.; Allmann, D.W. Effect of dietary fluoride on rat tissue 3′,5′ cyclic AMP levels. J. Dent. Res. 1979, 58, 1920. [Google Scholar] [CrossRef] [PubMed]
- Susheela, A.K.; Singh, M. Adenyl cyclase activity following fluoride ingestion. Toxicol. Lett. 1982, 10, 209–212. [Google Scholar] [CrossRef]
- Singh, M.; Sushella, A.K. Adenyl Cyclase activity and cyclic AMP levels following F-ingestion in rabbits and human subjects. Fluoride 1982, 15, 202–208. [Google Scholar]
- Gutowska, I.; Baranowska-Bosiacka, I.; Siennicka, A.; Telesiński, A.; Stańczyk-Dunaj, M.; Wesołowska, T.; Gąssowska, M.; Kłos, P.; Zakrzewska, H.; Machaliński, B.; et al. Activation of phospholipase A2 by low levels of fluoride in THP1 macrophages via altered Ca2+ and cAMP concentration. Prostaglandins Leukot Essent Fatty Acids 2012, 86, 99–105. [Google Scholar] [CrossRef]
- Bidey, S.B.; Marshall, M.J.; Ekins, R.P. Cyclic AMP release from normal human thyroid slices in response to thyrotrophin. Acta Endocrinol. 1980, 95, 335–340. [Google Scholar] [CrossRef] [PubMed]
- DeRubertis, F.; Yamashita, K.; Dekker, A.; Larsen, P.R.; Field, J.B. Effects of thyroid-stimulating hormone on adenyl cyclase activity and intermediary metabolism of “cold” thyroid nodules and normal human thyroid tissue. J. Clin. Investig. 1972, 51, 1109–1117. [Google Scholar] [CrossRef] [Green Version]
- Wolff, J.; Jones, A.B. The Purification of Bovine Thyroid Plasma Membranes and the Properties of Membrane-bound Adenyl Cyclase. J. Biol. Chem. 1971, 246, 3939–3947. [Google Scholar] [PubMed]
- Burke, G. Comparison of thyrotropin and sodium fluoride effects on thyroid adenyl cyclase. Endocrinology 1970, 86, 346–352. [Google Scholar] [CrossRef]
- Bech, K.; Madsen, S.N. Human thyroid adenylate cyclase in non-toxic goitre: Sensitivity to TSH, fluoride and thyroid stimulating immunoglobulins. Clin. Endocrinol. 1978, 8, 457–466. [Google Scholar] [CrossRef]
- Zhang, S.; Zhang, X.; Liu, H.; Qu, W.; Guan, Z.; Zeng, Q.; Jiang, C.; Gao, H.; Zhang, C.; Lei, R.; et al. Modifying Effect of COMT Gene Polymorphism and a Predictive Role for Proteomics Analysis in Children’s Intelligence in Endemic Fluorosis Area in Tianjin, China. Toxicol. Sci. 2015, 144, 238–245. [Google Scholar] [CrossRef] [Green Version]
- Singh, N.; Verma, K.G.; Verma, P.; Sidhu, G.K.; Sachdeva, S. A comparative study of fluoride ingestion levels, serum thyroid hormone & TSH level derangements, dental fluorosis status among school children from endemic and non-endemic fluorosis areas. Springerplus 2014, 3, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yasmin, S.; Ranjan, S.; D’Souza, D.; D’Souza, H. Effect of excess fluoride ingestion on human thyroid function in Gaya region, Bihar, India. Toxicol. Environ. Chem. 2013, 95, 1235–1243. [Google Scholar] [CrossRef]
- Shashi, A.; Singla, S. Syndrome of Low Triiodothyronine in Chronic Fluorosis. Int. J. Appl. Basic Med. Res. 2013, 3, 152–160. [Google Scholar]
- Hosur, M.B.; Puranik, R.S.; Vanaki, S.; Puranik, S. Study of thyroid hormones free triiodothyronine (FT3), free thyroxine (FT4) and thyroid stimulating hormone (TSH) in subjects with dental fluorosis. Eur. J. Dent. 2012, 6, 184–190. [Google Scholar]
- Karademir, S.; Mustafa, A.; Kuybulu, A.E.; Olgar, S.; Öktem, F. Effects of fluorosis on QT dispersion, heart rate variability and echocardiographic parameters in children. Anadolu Kardiyol. Derg. 2011, 1, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Q.; Chen, L.; Miang, Y.; Wu, M.; Chen, B. Fluoride and thyroid function in children in two villages in China. J. Toxicol. Environ. Health Sci. 2009, 1, 54–59. [Google Scholar]
- Bahijri, S.M.; Al-Fares, A.; Al-Khateeb, T.; Mufti, A.B. Hyperparathyroidism and Hypothyroidism in Individuals Consuming High Fluoride Intake in Jeddah-Saudi Arabia. Syr. Clin. Lab. Assoc. 2008, 4, 1428–1436. [Google Scholar]
- Ruiz-Payan, A. Chronic Effects of Fluoride on Growth, Blood Chemistry and Thyroid Hormones in Adolescents Residing in Three Communities in Northern Mexico; AAI3214004; ETD Collection for University of Texas: El Paso, TX, USA, 2006. [Google Scholar]
- Suskeela, A.K.; Bhatnagar, M.; Vig, K.; Monda, N.K. Excess fluoride ingestion and thyroid hormone derangements in children living in Delhi, India. Fluoride 2005, 38, 98–108. [Google Scholar]
- Wang, X.; Wang, L.; Hu, P.; Guo, X.; Luo, X. Effects of high iodine and high fluorine on children’s intelligence and thyroid function. Chin. J. Endemiol. 2001, 20, 288–290. [Google Scholar]
- Xiaoli, L.; Zhongxue, F.; Jili, H.; Qinlan, W.; Hongyin, W. The Detection of Children’s T3, T4 and TSH Contents in Endemic Fluorosis Area. Endem. Dis. Bull. 1999, 14, 16–17. [Google Scholar]
- Yao, Y.; Qicheng, C.; Fengyan, L. Analysis on TSH and intelligence level of children with dental Fluorosis in a high fluoride area. Lit. Inf. Prev. Med. 1996, 2, 26–27. [Google Scholar]
- Lin, F.; Aihaiti, H.X.; Zhao, H.X.; Jin, L.; Ji-Yong, J.; Maimaiti, A. The relationship of a low-iodine and high-fluoride environment to subclinical cretinism in Xinjiang. Endem. Dis. Bull. 1991, 6, 62–67. [Google Scholar]
- Bachinskii, P.P.; Gutsalenko, O.A.; Naryzhniuk, N.D.; Sidora, V.D.; Shliakhta, A.I. Action of the body fluorine of healthy persons and thyroidopathy patients on the function of hypophyseal-thyroid the system. Probl. Endokrinol. 1985, 31, 25–29. [Google Scholar]
- Gutowska, I.; Baranowska-Bosiacka, I.; Baśkiewicz, M.; Milo, B.; Siennicka, A.; Marchlewicz, M.; Wiszniewska, B.; Machaliński, B.; Stachowska, E. Fluoride as a pro-inflammatory factor and inhibitor of ATP bioavailability in differentiated human THP1 monocytic cells. Toxicol. Lett. 2010, 196, 74–79. [Google Scholar] [CrossRef]
- Agalakova, N.I.; Gusev, G.P. Fluoride induces oxidative stress and ATP depletion in the rat erythrocytes in vitro. Environ. Toxicol. Pharmacol. 2012, 34, 334–337. [Google Scholar] [CrossRef] [PubMed]
- Cooper, G.M. The Cell: A Molecular Approach, 2nd ed.; Sinauer Associates: Sunderland, MA, USA, 2000. Available online: https://www.ncbi.nlm.nih.gov/books/NBK9903/ (accessed on 20 February 2019).
- Engelking, L.R. Intermediate Reactions in Anaerobic Glycolysis. In Textbook of Veterinary Physiological Chemistry, 3rd ed.; Academic Press, Elsevier: Waltham, MA, USA, 2015; Chapter 26; pp. 164–168. [Google Scholar]
- Shephard, E.G.; Anderson, R.; Rosen, O.; Fridkin, M. C-reactive protein (CRP) peptides inactivate enolase in human neutrophils leading to depletion of intracellular ATP and inhibition of superoxide generation. Immunology 1992, 76, 79–85. [Google Scholar] [PubMed]
- Warburg, O.; Christian, W. Isolation and crystallization of enolase. Biochem. Z. 1942, 310, 384–421. [Google Scholar]
- Cimasoni, G. The Inhibition of Enolase by Fluoride in vitro. Caries Res. 1972, 6, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Chai, G.; Brewer, J.M.; Lovelace, L.L.; Lebioda, L. Fluoride inhibition of enolase: Crystal structure and thermodynamics. Biochemistry 2006, 45, 793–800. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Himoe, A. Kinetics of the rabbit muscle enolase-catalyzed dehydration of 2-phosphoglycerate. J. Biol. Chem. 1974, 249, 3895–3902. [Google Scholar] [PubMed]
- Kiroytcheva, M.; Cheval, L.; Carranza, M.L.; Martin, P.Y.; Favre, H.; Doucet, A.; Féraille, E. Effect of cAMP on the activity and the phosphorylation of Na+,K(+)-ATPase in rat thick ascending limb of Henle. Kidney Int. 1999, 55, 1819–1831. [Google Scholar] [CrossRef] [PubMed]
- Wacker, W.E.; Parisi, A.F. Magnesium metabolism. N. Engl. J. Med. 1968, 278, 658–663. [Google Scholar] [CrossRef] [PubMed]
- Martyka, Z.; Kotela, I.; Blady-Kotela, A. Clinical use of magnesium. Przegl. Lek. 1996, 53, 155–158. [Google Scholar]
- Vormann, J. Magnesium: Nutrition and metabolism. Mol. Asp. Med. 2003, 24, 27–37. [Google Scholar] [CrossRef]
- Cerklewski, F.L. Influence of dietary magnesium on fluoride bioavailability in the rat. J. Nutr. 1987, 117, 496–500. [Google Scholar] [CrossRef]
- Ophaug, R.H.; Singer, L. Effect of fluoride on the mobilization of skeletal magnesium and soft-tissue calcinosis during acute magnesium deficiency in the rat. J. Nutr. 1976, 106, 771–777. [Google Scholar] [CrossRef]
- Chen, P.; Yun, Z.; Li, T.; Gao, H.; Hao, J.; Qin, Y. Relations between endemic fluorosis and chemicalelements in environment. Zhongguo Gonggong Weisheng Xuebo 2002, 18, 433–434. [Google Scholar]
- Meral, I.; Demir, H.; Gunduz, H.; Mert, N.; Dogan, I.; Turkey, V. Serum copper, zinc, manganese, and magnesium status of subjects with chronic fluorosis. Fluoride 2004, 37, 102–106. [Google Scholar]
- Ersoy, I.H.; Koroglu, B.K.; Varol, S.; Ersoy, S.; Varol, E.; Aylak, F.; Tamer, M.N. Serum copper, zinc, and magnesium levels in patients with chronic fluorosis. Biol. Trace Elem. Res. 2011, 143, 619–624. [Google Scholar] [CrossRef] [PubMed]
- Kessabi, M.; Khouzaimi, M.; Braun, J.P.; Hamliri, A. Serum Biochemical Effects of Fluoride on Cattle in the Darmous Area. Vet. Hum. Toxicol. 1983, 25, 403–406. [Google Scholar] [PubMed]
- Rajendraprasad, U.S. Bovine Fluorosis in Central East India: Monitoring and Treatment Strategies. Master’s Thesis, Indira Gandhi Agriculture University, Raipur, India, 2002. [Google Scholar]
- Singh, J.L.; Swarup, D. Biochemical changes in serum and urine bovine fluorosis. Indian J. Anim. Sci. 1999, 69, 776–778. [Google Scholar]
- Wu, Z.J.; Ding, J.Y.; Qi, D.S.; Yu, Y.H. Biochemical indexes of buffalo with fluorosis and their significance for diagnosis. J. Huazhong Agric. Univ. 1995, 14, 369–373. [Google Scholar]
- Altuğ, N.; Arslan, S.; Yüksek, N.; Keles, I.; Yörük, H.I.; Başbuğan, Y.; Aytekin, I. The levels of trace elements and selected vitamins in goats with chronic fluorosis. Turk. J. Vet. Anim. Sci. 2013, 37, 529–534. [Google Scholar] [CrossRef]
- Aperia, A.; Ibarra, F.; Svensson, L.B.; Klee, C.; Greengard, P. Calcineurin mediates alpha-adrenergic stimulation of Na+, K(+)-ATPase activity in renal tubule cells. Proc. Natl. Acad. Sci. USA 1992, 89, 7394–7397. [Google Scholar] [CrossRef]
- Pallen, C.J.; Valentine, K.A.; Wang, J.H.; Hollenberg, M.D. Calcineurin-Mediated Dephosphorylation of the Human Placental Membrane Receptor for Epidermal Growth Factor Urogastrone1. Biochemistry 1985, 24, 4727–4730. [Google Scholar] [CrossRef] [PubMed]
- Tallant, E.A.; Cheung, W.Y. Characterization of bovine brain calmodulin-dependent protein phosphatase. Arch. Biochem. Biophys. 1984, 232, 260–279. [Google Scholar] [CrossRef]
- Klee, C.; Wang, X.; Ren, H. Calcium-Regulated Protein Dephosphorylation. In Calcium as a Cellular Regulator; Oxford University Press: Oxford, UK, 1999; pp. 344–370. [Google Scholar]
- King, M.M.; Huang, C.Y. The Calmodulin-dependent Activation and Deactivation of the Phosphoprotein Phosphatase, Calcineurin, and the Effect of Nucleotides, Pyrophosphate, and Divalent Metal Ions. J. Biol. Chem. 1984, 259, 8847–8856. [Google Scholar]
- Gupta, R.C.; Khandelwal, R.L.; Sulakhe, P.V. Divalent cation effects on calcineurin phosphatase: Differential involvement of hydrophobic and metal binding domains in the regulation of the enzyme activity. Mol. Cell. Biochem. 1990, 97, 53–66. [Google Scholar] [CrossRef] [PubMed]
- Ping, L.; Ke, Z.; Benqiong, X.; Qun, W. Effect of metal ions on the activity of the catalytic domain of calcineurin. Biometals 2004, 17, 157–165. [Google Scholar] [CrossRef]
- Amiranoff, B.M.; Laburthe, M.C.; Rouyer-Fessard, C.M.; Demaille, J.G.; Rosselin, G.E. Calmodulin Stimulation of Adenylate Cyclase of Intestinal Epithelium. Eur. J. Biochem. 1983, 130, 33–37. [Google Scholar] [CrossRef]
- Margaret, L.R.; Israels, S.J. Molecular Basis of Platelet Function, 7th ed.; Hematology: Fort Worth, TX, USA, 2018; Chapter 125; pp. 1870–1884.e2. [Google Scholar]
- Yingst, D.R.; Ye-Hu, J.; Chen, H.; Barrett, V. Calmodulin increases Ca-dependent inhibition of the Na,K-ATPase in human red blood cells. Arch. Biochem. Biophys. 1992, 295, 49–54. [Google Scholar] [CrossRef]
- Smilowitz, H.; Hadjian, R.A.; Dwyer, J.; Feinstein, M.B. Regulation of acetylcholine receptor phosphorylation by calcium and calmodulin. Proc. Natl. Acad. Sci. USA 1981, 78, 4708–4712. [Google Scholar] [CrossRef]
- Yorio, T.; Sinclair, R.; Henry, S. Fluoride inhibition of the hydro-osmotic response of the toad urinary bladder to antidiuretic hormone. J. Pharmacol. Exp. Ther. 1981, 219, 459–463. [Google Scholar]
- Feneck, R. Phosphodiesterase inhibitors and the cardiovascular system. Contin. Educ. Anaesth. Crit. Care Pain 2007, 6, 203–207. [Google Scholar] [CrossRef]
- Wang, Z.; Li, X.D.; Li, M.Q.; Wang, Q.P. Changes in basic metabolic elements associated with the degeneration and ossification of ligamenta flava. J. Spinal Med. 2008, 31, 279–284. [Google Scholar] [CrossRef]
- Tao, X.; Xu, Z.R.; Wang, Y.Z. Effect of excessive dietary fluoride on nutrient digestibility and retention of iron, copper, zinc and manganese in growing pigs. Biol. Trace Elem. Res. 2005, 107, 141–151. [Google Scholar] [CrossRef]
- Karademir, B. Effect of fluoride ingestion on serum levels of the trace minerals Co, Mo, Cr, Mn and Li in adult male mice. Fluoride 2010, 43, 174–178. [Google Scholar]
- Singh, M. Biochemical and cytochemical alterations in liver and kidney following experimental fluorosis. Fluoride 1984, 17, 81–93. [Google Scholar]
- Ranjan, R.; Swarup, D.; Patra, R.C. Changes in levels of zinc, copper, cobalt, and manganese in soft tissues of fluoride-exposed rabbits. Fluoride 2011, 44, 83–88. [Google Scholar]
- Birkner, E.; Grucka-Mamczar, E.; Machoy, Z.; Tamawski, R.; Polaniak, R. Disturbance of protein metabolism in rats after acute poisoning with sodium fluoride. Fluoride 2000, 33, 182–186. [Google Scholar]
- Smita, P.B.; Pawar, S.S. Effect of fluoride ingestion on trace elements on brain and liver of Rat Rattus rattus (Wistar). Int. J. Life Sci. 2017, A8, 125–128. [Google Scholar]
- McKee, M.; Scavone, C.; Nathanson, J.A. Nitric oxide, cGMP, and hormone regulation of active sodium transport. Proc. Natl. Acad. Sci. USA 1994, 91, 12056–12060. [Google Scholar] [CrossRef]
- Scavone, C.; Scanlon, C.; McKee, M.; Nathanson, J.A. Atrial natriuretic peptide modulates sodium and potassium-activated adenosine triphosphatase through a mechanism involving cyclic GMP and cyclic GMP-dependent protein kinase. J. Pharmacol. Exp. Ther. 1995, 272, 1036–1043. [Google Scholar]
- De Oliveira Elias, M.; Tavares de Lima, W.; Vannuchi, Y.B.; Marcourakis, T.; da Silva, Z.L.; Trezena, A.G.; Scavone, C. Nitric oxide modulates Na+, K+-ATPase activity through cyclic GMP pathway in proximal rat trachea. Eur. J. Pharmacol. 1999, 367, 307–314. [Google Scholar] [CrossRef]
- Liang, M.; Knox, F.G. Nitric oxide reduces the molecular activity of Na+, K+-ATPase in opossum kidney cells. Kidney Int. 1999, 56, 627–634. [Google Scholar] [CrossRef]
- Liang, M.; Knox, F.G. Nitric oxide activates PKCalpha and inhibits Na+-K+-ATPase in opossum kidney cells. Am. J. Physiol. 1999, 277, F859–F865. [Google Scholar] [PubMed]
- Linas, S.L.; Repine, J.E. Endothelial cells regulate proximal tubule epithelial cell sodium transport. Kidney Int. 1999, 55, 1251–1258. [Google Scholar] [CrossRef] [Green Version]
- Bełtowski, J.; Marciniak, A.; Wójcicka, G.; Górny, D. Nitric oxide decreases renal medullary Na+, K+-ATPase activity through cyclic GMP-protein kinase G dependent mechanism. J. Physiol. Pharmacol. 2003, 54, 191–210. [Google Scholar] [PubMed]
- Bhatnagar, M.; Sukhwal, P.; Suhalka, P.; Jain, A.; Joshi, C.; Sharma, D. Effects of Fluoride in Drinking Water on Nadphdiaphorase Neurons in the Forebrain of Mice: A possible mechanism of fluoride neurotoxicity. Fluoride 2011, 44, 195–209. [Google Scholar]
- Shanmugam, T.; Abdulla, S.; Yakulasamy, V.; Selvaraj, M.; Mathan, R. A mechanism underlying the neurotoxicity induced by sodium fluoride and its reversal by epigallocatechin gallate in the rat hippocampus: Involvement of NrF2/Keap-1 signaling pathway. J. Basic Appl. Zool. 2018, 79, 17. [Google Scholar] [CrossRef]
- Liu, G.; Chai, C.; Cui, L. Fluoride causing abnormally elevated serum nitric oxide levels in chicks. Environ. Toxicol. Pharmacol. 2003, 13, 199–204. [Google Scholar] [CrossRef]
- Guoyan, L.; Chunyan, C.; Shiliang, K. Functions of nitric oxide in the mechanism of chick fluorosis. Chin. J. Vet. Sci. 2000, 20, 588–590. [Google Scholar]
- Zhan, X.; Xu, Z.; Li, J.; Wang, M. Effects of fluorosis on lipid peroxidation and antioxidant systems in young pigs. Fluoride 2005, 38, 157–161. [Google Scholar]
- Panneerselvam, L.; Subbiah, K.; Arumugum, A.; Senapathy, J.G. Ferulic acid modulates fluoride induced oxidative hepatotoxicity in male Wistar rats. Biol. Trace Elem. Res. 2013, 151, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Hassan, H.A.; Yousef, M.I. Mitigating effects of antioxidant properties of black berry juice on sodium fluoride induced hepatotoxicity and oxidative stress in rats. Food Chem. Toxicol. 2009, 47, 2332–2337. [Google Scholar] [CrossRef] [PubMed]
- White, A.A.; Aurbach, G.D. Detection of guanyl cyclase in mammalian tissues. Biochim. Biophys. Acta 1969, 191, 686–697. [Google Scholar] [CrossRef]
- Yamashita, K.; Field, J.B. Elevation of Cyclic Guanosine 3′,5′-Monophosphate Levels in Dog Thyroid Slices Caused by Acetylcholine and Sodium Fluoride. J. Biol. Chem. 1972, 247, 7062–7066. [Google Scholar] [PubMed]
- Zhao, Y.; Li, Y.; Gao, Y.; Yuan, M.; Manthari, R.K.; Wang, J.; Wang, J. TGF-β1 acts as mediator in fluoride-induced autophagy in the mouse osteoblast cells. Food Chem. Toxicol. 2018, 115, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Sakallioğlu, E.E.; Muğlali, M.; Baş, B.; Gulbahar, M.Y.; Lütfioğlu, M.; Aksoy, A. Effects of Excessive Fluoride intake on Bone Turnover in Mandible: An Immunohistochemical Study in Rabbits. Fluoride 2014, 47, 23–30. [Google Scholar]
- Gao, Y.H.; Fu, S.B.; Sun, H.; Zhou, L.W.; Yu, J.; Li, Y.; Wang, Y.; Sun, D.J. Expression of the transforming growth factor-β superfamily in bone turnover of fluorosis. Chin. J. Endemiol. 2006, 25, 374–378. [Google Scholar]
- Yang, C.; Wang, Y.; Xu, H. Fluoride Regulate Osteoblastic Transforming Growth Factor-β1 Signaling by Mediating Recycling of the Type I Receptor ALK5. PLoS ONE 2017, 12, e0170674. [Google Scholar]
- Liu, X.L.; Song, J.; Liu, K.J.; Wang, W.P.; Xu, C.; Zhang, Y.Z.; Liu, Y. Role of inhibition of osteogenesis function by Sema4D/Plexin-B1 signaling pathway in skeletal fluorosis in vitro. J. Huazhong Univ. Sci. Technol. Med. Sci. 2015, 35, 712–715. [Google Scholar] [CrossRef]
- Wang, Y.Q.; Yuan, R.; Sun, Y.P.; Lee, T.J.; Shah, G.V. Antiproliferative action of calcitonin on lactotrophs of the rat anterior pituitary gland: Evidence for the involvement of transforming growth factor beta 1 in calcitonin action. Endocrinology 2003, 144, 2164–2171. [Google Scholar] [CrossRef] [PubMed]
- Krishnamachari, K.A.V.R.; Sivakumar, B. Endemic genu valgum. A new dimension to the fluorosis problem in India. Fluoride 1976, 9, 185–200. [Google Scholar]
- Teotia, S.P.S.; Teotia, M.; Singh, R.K.; Teotia, N.P.S.; Taves, D.R.; Heels, S. Plasma fluoride, 25-hydroxycholecalciferol, immunoreactive parathyroid Hormone and calcitonin in patients with endemic skeletal fluorosis. Fluoride 1978, 11, 115–119. [Google Scholar]
- Ma, J.; Li, M.; Song, Y.; Tu, J.; Liu, F.; Liu, K. Serum Osteocalcin and Calcitonin in adult males with different fluoride exposures. Fluoride 2009, 42, 133–136. [Google Scholar]
- Chen, S.; Li, B.; Lin, S.; Huang, Y.; Zhao, X.; Zhang, M.; Xia, Y.; Fang, X.; Wang, J.; Hwang, S.A.; et al. Change of urinary fluoride and bone metabolism indicators in the endemic fluorosis areas of southern china after supplying low fluoride public water. BMC Public Health 2013, 13, 156. [Google Scholar] [CrossRef]
- Shashi, A.; Singla, S. Parathyroid Function in Osteofluorosis. World J. Med. Sci. 2013, 8, 67–73. [Google Scholar]
- Ba, Y.; Zhu, J.; Yang, Y.; Yu, B.; Huang, H.; Wang, G.; Ren, L.; Cheng, X.; Cui, L.; Zhang, Y. Serum calciotropic hormone levels, and dental fluorosis in children exposed to different concentrations of fluoride and iodine in drinking water. Chin. Med. J. 2010, 123, 675–679. [Google Scholar]
- Apell, H.J.; Roudna, M.; Corrie, J.E.; Trentham, D.R. Kinetics of the phosphorylation of Na,K-ATPase by inorganic phosphate detected by a fluorescence method. Biochemistry 1996, 35, 10922–10930. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.H.; Askari, A. Regulation of (Na++K+)-ATPase by inorganic phosphate: pH dependence and physiological implications. Biochem. Biophys. Res. Commun. 1984, 123, 438–443. [Google Scholar] [CrossRef]
- Martin, B.L.; Graves, D.J. Mechanistic aspects of the low-molecular-weight phosphatase activity of the calmodulin-activated phosphatase, calcineurin. J. Biol. Chem. 1986, 261, 14545–14550. [Google Scholar] [PubMed]
- Li, J.W.; Xu, C.; Fan, Y.; Wang, Y.; Xiao, Y.B. Can serum levels of alkaline phosphatase and phosphate predict cardiovascular diseases and total mortality in individuals with preserved renal function? A systemic review and meta-analysis. PLoS ONE 2014, 9, e102276. [Google Scholar] [CrossRef] [PubMed]
- Penido, M.G.M.G.; Alon, U.S. Phosphate homeostasis and its role in bone health. Pediatr. Nephrol. 2012, 27, 2039–2048. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.L.; Cheng, X.M.; Fan, Q.T.Q. Water fluoride concentration and human health effect. Chin. J. Endemiol. 1993, 12, 21–23. [Google Scholar]
- Ravula, S.; Harinarayan, C.V.; Prasad, U.V.; Ramalakshmi, T.; Rupungudi, A.; Madrol, V. Effect of Fluoride on Reactive Oxygen Species and Bone Metabolism in Postmenopausal Women. Fluoride 2012, 45, 108–115. [Google Scholar]
- Lavanya, R.; Devadoss, Y. Study of bone markers and fluoride levels in adult men with skeletal fluorosis. IOSR J. Dent. Med. Sci. 2018, 17, 78–80. [Google Scholar]
- Raghuramula, N.; Krishnamachari, K.A.V.R.; Rao, B.S. Serum 25-Hydroxy Vitamin D3 in Endemic fluorosis Genu ValGum and Fluorosis. Fluoride 1997, 30, 147–152. [Google Scholar]
- Jackson, R.; Kelly, S.; Noblitt, T.; Zhang, W.; Dunipace, A.; Li, Y.; Stookey, G.; Katz, B.; Brizendine, E.; Farley, S.; et al. The effect of fluoride therapy on blood chemistry parameters in osteoporotic females. Bone Miner. 1994, 27, 13–23. [Google Scholar] [CrossRef]
- Murugan, A.; Subramanian, A. Studies on the Biological Effects of Fluoride Intoxication in Dental Fluorosis Cases. Aust. J. Basic Appl. Sci. 2011, 5, 1362–1367. [Google Scholar]
- Pan, L.; Shib, X.; Liua, S.; Guo, X.; Zhao, M.; Cai, R.; Sun, G. Fluoride promotes osteoblastic differentiation through canonicalWnt/β-catenin signaling pathway. Toxicol. Lett. 2014, 225, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Farley, J.R.; Wergedal, J.E.; Hall, S.L.; Herring, S.; Tarbaux, N.M. Calcitonin has direct effects on 3[H]-thymidine incorporation and alkaline phosphatase activity in human osteoblast-line cells. Calcif. Tissue Int. 1991, 48, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Potts, J.T. Parathyroid hormone: Past and present. J. Endocrinol. 2005, 187, 311–325. [Google Scholar] [CrossRef]
- Torres, P.A.; De Brauwere, D.P. Three feedback loops precisely regulating serum phosphate concentration. Kidney Int. 2011, 80, 443–445. [Google Scholar] [CrossRef] [Green Version]
- Gupta, A.; Guo, X.L.; Alvarez, U.M.; Hruska, K.A. Regulation of sodium-dependent phosphate transport in osteoclasts. J. Clin. Investig. 1997, 100, 538–549. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Liu, H.; Yu, X.; Wang, Y.; Yang, C.; Xu, H. Analysis of the Role of Insulin Signaling in Bone Turnover Induced by Fluoride. Biol. Trace Elem. Res. 2016, 171, 380–390. [Google Scholar] [CrossRef]
- Santoyo-Sanchez, M.P.; del Carmen Silva-Lucero, M.; Arreola-Mendoza, L.; Barbier, O.C. Effects of acute sodium fluoride exposure on kidney function, water homeostasis, and renal handling of calcium and inorganic phosphate. Biol. Trace Elem. Res. 2013, 152, 367–372. [Google Scholar] [CrossRef]
- Monsour, P.A.; Kruger, B.J.; Smid, J.R. Effects of a single intravenous dose of sodium fluoride on plasma electrolytes and metabolites in rats, rabbits, and cockerels. J. Dent. Res. 1985, 64, 1281–1285. [Google Scholar] [CrossRef]
- Di Loreto, V.; Rigalli, A.; Puche, R. Effect of sodium fluoride administration to rats on bone phosphorous content and phosphatemia. Arzneimittelforschung 2006, 56, 760–766. [Google Scholar] [CrossRef]
- Dandona, P.; Coumar, A.; Gill, D.S.; Bell, J.; Thomas, M. Sodium Fluoride Stimulates Osteocalcin in Normal Subjects. Clin. Endocrinol. 1988, 29, 437–441. [Google Scholar] [CrossRef]
- Nabavi, S.M.; Habtemariam, S.; Nabavi, S.F.; Sureda, A.; Daglia, M.; Moghaddam, A.H.; Amani, M.A. Protective effect of gallic acid isolated from Peltiphyllum peltatum against sodium fluoride-induced oxidative stress in rat’s kidney. Mol. Cell. Biochem. 2013, 372, 233–239. [Google Scholar] [CrossRef]
- Shanthakumari, D.; Subramanian, S. Effect of Fluoride Intoxication on Bone Tissue of Experimental Rats. Res. J. Environ. Sci. 2007, 1, 82–92. [Google Scholar]
- Gupta, A.R.; Dey, S.; Saini, M.; Swarup, D. Toxic effect of sodium fluoride on hydroxyproline level and expression of collagen-1 gene in rat bone and its amelioration by Tamrindus indica L. fruit pulp extract. Interdiscip. Toxicol. 2017, 9, 12–16. [Google Scholar] [CrossRef] [PubMed]
- Ranjan, R.; Swarup, D.; Patra, R.C.; Chandra, V. Tamarindus indica L. and Moringa oleifera M. extract administration ameliorate fluoride toxicity in rabbits. Indian J. Exp. Biol. 2009, 47, 900–905. [Google Scholar]
- Gupta, A.R.; Dey, S.; Swarup, D.; Saini, M.; Saxena, A.; Dan, A. Ameliorative effect of Tamarindus indica L. on biochemical parameters of serum and urine in cattle from fluoride endemic area. Vet. Arhiv. 2013, 83, 487–496. [Google Scholar]
- Gupta, A.R.; Dey, S.; Saini, M.; Swarup, D. Toxic effect of fluoride on biochemical parameters and collagen metabolism in osseous and non-osseous tissues of rats. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2015, 85, 719–724. [Google Scholar] [CrossRef]
- de Lores, R.; Arnaiz, G. Neuronal Na+-K+-ATPase and its regulation by catecholamines. Intern. Brain Res. Org. Monogr. Ser. 1983, 10, 147–158. [Google Scholar]
- Seri, I.; Kone, B.C.; Gullans, S.R.; Aperia, A.; Brenner, B.M.; Ballermann, B.J. Locally formed dopamine inhibits Na+-K+-ATPase activity in rat renal cortical tubule cells. Am. J. Physiol. 1988, 255, F666–F673. [Google Scholar] [CrossRef]
- Seri, I.; Kone, B.C.; Gullans, S.R.; Aperia, A.; Brenner, B.M.; Ballermann, B.J. Influence of Na+ intake on dopamine-induced inhibition of renal cortical Na(+)-K(+)-ATPase. Am. J. Physiol. 1990, 258, F52–F60. [Google Scholar] [CrossRef]
- Satoh, T.; Cohen, H.T.; Katz, A.I. Intracellular signaling in the regulation of renal Na-K-ATPase. I. Role of cyclic AMP and phospholipase A2. J. Clin. Investig. 1992, 89, 1496–1500. [Google Scholar] [CrossRef] [PubMed]
- Shahedi, M.; Laborde, K.; Azimi, S.; Hamdani, S.; Sachs, C. Mechanisms of dopamine effects on Na-K-ATPase activity in Madin-Darby canine kidney (MDCK) epithelial cells. Pflug. Arch. 1995, 429, 832–840. [Google Scholar] [CrossRef]
- Pinto-do-O, P.C.; Chibalin, A.V.; Katz, A.I.; Soares-da-Silva, P.; Bertorello, A.M. Short term vs. sustained inhibition of proximal tubule Na-K ATPase activity by dopamine: Cellular mechanisms. Clin. Exp. Hypertens. 1997, 19, 73–86. [Google Scholar] [CrossRef]
- Khundmiri, S.J.; Lederer, E. PTH and DA regulate Na-K ATPase through divergent pathways. Am. J. Physiol. Ren. Physiol. 2002, 282, F512–F522. [Google Scholar] [CrossRef] [PubMed]
- Khan, F.H.; Sen, T.; Chakrabarti, S. Dopamine oxidation products inhibit Na+, K+-ATPase activity in crude synaptosomal-mitochondrial fraction from rat brain. Free Radic. Res. 2003, 37, 597–601. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, F.; Hamet, P.; Orlov, S.N. Na+, K+ pump and Na+-coupled ion carriers in isolated mammalian kidney epithelial cells: Regulation by protein kinase C. Can. J. Physiol. Pharmacol. 1999, 77, 305–319. [Google Scholar] [CrossRef]
- Hatta, S.; Amemiya, N.; Takemura, H.; Ohshika, H. Effects of Dopamine on Adenylyl Cyclase Activity and Amylase Secretion in Rat Parotid Tissue. J. Dent. Res. 1995, 74, 1289–1294. [Google Scholar] [CrossRef] [PubMed]
- Vortherms, T.A.; Nguyen, C.H.; Bastepe, M.; Jüppner, H.; Watts, V.J. D2 dopamine receptor-induced sensitization of adenylyl cyclase type 1 is G alpha(s) independent. Neuropharmacology 2006, 50, 576–584. [Google Scholar] [CrossRef] [PubMed]
- Osborne, N.N. Effects of GTP, forskolin, sodium fluoride, serotonin, dopamine, and carbachol on adenylate cyclase in Teleost retina. Neurochem. Res. 1990, 15, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Battaglia, G.; Norman, A.B.; Hess, E.J.; Creese, I. Forskolin potentiates the stimulation of rat striatal adenylate cyclase mediated by D-1 dopamine receptors, guanine nucleotides, and sodium fluoride. J. Neurochem. 1986, 46, 1180–1185. [Google Scholar] [CrossRef]
- Woodward, J.J.; Harms, J. Potentiation of N-methyl-D-aspartate-stimulated dopamine release from rat brain slices by aluminum fluoride and carbachol. J. Neurochem. 1992, 58, 1547–1554. [Google Scholar] [CrossRef]
- Tsunoda, M.; Aizawa, Y.; Nakano, K.; Liu, Y.; Horiuchi, T.; Itai, K.; Tsunoda, H. Changes in Fluoride Levels in the Liver, Kidney, and Brain and in Neurotransmitters of Mice after Subacute Administration of Fluoride. Fluoride 2005, 38, 284–292. [Google Scholar]
- Pereira, M.; Dombrowski, P.A.; Losso, E.M.; Chioca, L.R.; Da Cunha, C.; Andreatini, R. Memory impairment induced by sodium fluoride is associated with changes in brain monoamine levels. Neurotox. Res. 2011, 19, 55–62. [Google Scholar] [CrossRef]
- Hall, R.; Amos, J.; Garry, R.; Buxton, R.L. Thyroid-stimulating hormone response to synthetic thyrotropin releasing hormone in man. Br. Med. J. 1970, 2, 274–277. [Google Scholar] [CrossRef] [PubMed]
- Sergev, O.; Rácz, K.; Varga, I.; Kiss, R.; Futo, L.; Mohari, K.; Gláz, E. Thyrotropin-releasing hormone increases plasma atrial natriuretic peptide levels in human. J. Endocrinol. Investig. 1990, 13, 649–652. [Google Scholar] [CrossRef]
- Ikegami, H.; Spahn, S.A.; Prasad, C. Neuropeptide-dopamine interactions. IV. Effect of thyrotropin-releasing hormone on striatal dopaminergic neurons. Peptides 1989, 10, 681–685. [Google Scholar] [CrossRef]
- Prasad, C. Modulation of striatal dopamine system by thyrotropin-releasing hormone and cyclo(His-Pro). In Basal Ganglia: Structure and Function II; Carpenter, M.B., Jayara-Man, A., Eds.; Plenum Publishing Co.: New York, NY, USA, 1987; pp. 155–168. [Google Scholar]
- Narumi, S.; Nagai, Y.; Saji, Y.; Nagawa, Y. A possible mechanism of action of thyrotropin-releasing hormone (TRH) and its analog DN-1417 on the release of dopamine from the nucleus accumbens and striatum in rats. Jpn. J. Pharmacol. 1985, 39, 425–435. [Google Scholar] [CrossRef]
- Crocker, A.D.; Overstreet, D.H.; Crocker, J.M. Hypothyroidism leads to increased dopamine receptor sensitivity and concentration. Pharmacol. Biochem. Behav. 1986, 24, 1593–1597. [Google Scholar] [CrossRef]
- Bunney, B.S.; Achajanian, G.K. d-Amphetamine-induced inhibition of central dopaminergic neurons: Mediation by a striato-nigral feedback pathway. Science 1976, 192, 391–393. [Google Scholar] [CrossRef] [PubMed]
- Mitnick, M.; Reichlin, S. Enzymatic Synthesis of Thyrotropin-Releasing Hormone (TRH) by Hypothalamic “TRH Synthetase”. Endocrinology 1972, 91, 1145–1153. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Yang, W.; Dong, Z.; Wan, C.; Zhang, J.; Liu, J.; Xiao, K.; Huang, Y. Neurotransmitter and Receptor Changes in the Brains of Fetuses from Areas of Endemic Fluorosis. Chin. J. Endemiol. 1996, 15, 257–259. [Google Scholar]
- Ribeiro, C.P.; Mandel, L.J. Parathyroid hormone inhibits proximal tubule Na(+)-K(+)-ATPase activity. Am. J. Physiol. 1992, 262, F209–F216. [Google Scholar] [CrossRef]
- Zhang, Y.; Norian, J.M.; Magyar, C.E.; Holstein-Rathlou, N.H.; Mircheff, A.K.; McDonough, A.A. In vivo PTH provokes apical NHE3 and NaPi-2 redistribution and Na-K ATPase inhibition. Am. J. Physiol. 1999, 276, F711–F719. [Google Scholar] [PubMed]
- Khundmiri, S.J.; Dean, W.L.; McLeish, K.R.; Lederer, E.D. Parathyroid hormone-mediated regulation of Na+-K+-ATPase requires ERK-dependent translocation of protein kinase Calpha. J. Biol. Chem. 2005, 280, 8705–8713. [Google Scholar] [CrossRef]
- Khundmiri, S.J.; Weinman, E.J.; Steplock, D.; Cole, J.; Ahmad, A.; Baumann, P.D.; Barati, M.; Rane, M.J.; Lederer, E. Parathyroid hormone regulation of NA+, K+-ATPase requires the PDZ 1 domain of sodium hydrogen exchanger regulatory factor-1 in opossum kidney cells. J. Am. Soc. Nephrol. 2005, 16, 2598–2607. [Google Scholar] [CrossRef]
- Derrickson, B.H.; Mandel, L.J. Parathyroid hormone inhibits Na-K ATPase through Gq/G11and the calcium independent phospholipase A2. Am. J. Physiol. Ren. Physiol. 1997, 272, F781–F788. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, C.P.; Dubay, G.R.; Falck, J.R.; Mandel, L.J. Parathyroid hormone inhibits Na-K-ATPase through a cytochrome P-450 pathway. Am. J. Physiol. Ren. Fluid Electrolyte Physiol. 1994, 266, F497–F505. [Google Scholar] [CrossRef]
- Koroglu, B.K.; Ersoy, I.H.; Koroglu, M.; Balkarli, A.; Ersoy, S.; Varol, S.; Tamer, M.N. Serum parathyroid hormone levels in chronic endemic fluorosis. Biol. Trace Elem. Res. 2011, 143, 79–86. [Google Scholar] [CrossRef]
- Schwartz, P.; Madsen, J.C.; Rasmussen, A.Q.; Transbol, I.B.; Brown, E.M. Evidence for a role of intracellular stored parathyroid hormone in producing hysteresis of the PTH-Calcium relationship in normal humans. Clin. Endo Crinol. 1998, 48, 725–732. [Google Scholar] [CrossRef]
- Greene, D.A.; Lattimer, S.A.; Sima, A.F. Sorbitol, phosphoinositides, and sodium-potassium-ATPase in the pathogenesis of diabetes complications. N. Engl. J. Med. 1987, 316, 599–606. [Google Scholar]
- Winegrad, A.I. Does a common mechanism induce the diverse complications of diabetes? Diabetes 1987, 36, 396–406. [Google Scholar] [CrossRef] [PubMed]
- MacGregor, L.C.; Matschinsky, F.M. Altered retinal metabolism in diabetes: II. measurement of sodium-potassium ATPase and total sodium and potassium in individual retinal layers. J. Biol. Chem. 1986, 261, 4052–4058. [Google Scholar]
- Owada, S.; Larsson, O.; Arkhammar, P.; Katz, A.I.; Chibalin, A.V.; Berggren, P.O.; Bertorello, A.M. Glucose decreases Na+, K+-ATPase activity in pancreatic beta-cells. An effect mediated via Ca2+-independent phospholipase A2 and protein kinase C-dependent phosphorylation of the alpha-subunit. J. Biol. Chem. 1999, 274, 2000–2008. [Google Scholar] [CrossRef] [PubMed]
- Rivelli, J.F.; Amaiden, M.R.; Monesterolo, N.E.; Previtali, G.; Santander, V.S.; Fernandez, A.; Arce, C.A.; Casale, C.H. High glucose levels induce inhibition of Na, K-ATPase via stimulation of aldose reductase, formation of microtubules and formation of an acetylated tubulin/Na,K-ATPase complex. Int. J. Biochem. Cell Biol. 2012, 44, 1203–1213. [Google Scholar] [CrossRef]
- Xia, P.; Kramer, R.M.; King, G.L. Identification of the mechanism for the inhibition of Na+, K(+)-adenosine triphosphatase by hyperglycemia involving activation of protein kinase C and cytosolic phospholipase A2. J. Clin. Investig. 1995, 96, 733–740. [Google Scholar] [CrossRef]
- Trivedi, N.; Mithal, A.; Gupta, S.K.; Godbole, M.M. Reversible impairment of glucose tolerance in patients with endemic fluorosis. Diabetologia 1993, 36, 826–828. [Google Scholar] [CrossRef] [PubMed]
- Guan, Z.; Yang, P.; Yu, N.; Zhuang, Z. An Experimental study of blood biochemical diagnostic indices for chronic fluorosis. Fluoride 1989, 22, 108–111. [Google Scholar]
- Suketa, Y.; Asao, Y.; Kanamoto, Y.; Shakashita, T.; Okada, S. Changes in adrenal function as a possible mechanism for elevation of serum glucose by single large dose of fluoride. Toxicol. Appl. Pharmacol. 1985, 80, 199–205. [Google Scholar] [CrossRef]
- Rigalli, A.; Ballina, J.C.; Roveri, E.; Puche, R.C. Inhibitory effect of fluoride on the secretion of Insulin. Calcif. Tissue Int. 1990, 46, 333–338. [Google Scholar] [CrossRef]
- Grucka-Mamczar, E.; Birkner, E.; Kasperczyk, S.; Kasperczyk, A.; Chlubek, D.; Samujto, D.; Cegłowska, A. Lipid Balance in rats with fluoride-induced hyperglycemia. Fluoride 2004, 37, 195–200. [Google Scholar]
- McGown, E.L.; Suttie, J.W. Mechanism of fluoride-induced hyperglycemia in the Rat. Toxicol. Appl. Pharmacol. 1977, 40, 83–90. [Google Scholar] [CrossRef]
- Allmann, D.W.; Kleiner, H.S. Effect of NaF on Rat Tissue cAMP levels in vivo. Pharmacol. Ther. Dent. 1980, 5, 73–78. [Google Scholar] [PubMed]
- Grucka-Mamczar, E.; Birkner, E.; Zalejska-Fiolka, J.; Machoy, Z. Disturbances of kidney function in rats with fluoride-induced hyperglycemia after acute poisoning by fluoride. Fluoride 2005, 38, 48–51. [Google Scholar]
- Szymafiska, H.; Mandat, A.; Jaroszewicz-Heigelmann, H.; Szymadski, Z.; Holicki, M.; Neuman, Z.; Ruszkowska, A. The results of assorted investigations carried out in workers exposed to Fluorine compounds. Metabolism of Fluorine. Soc. Sci. Stetin. PWN Warszawa Poznafi 1982, 96–102. [Google Scholar]
- Chlubek, D.; Grucka-Mamczar, E.; Birkner, E.; Polaniak, R.; Stawiarska-Pieta, B.; Duliban, H. Activity of pancreatic antioxidative enzymes and malondialdehyde concentrations in rats with hyperglycemia caused by fluoride intoxication. J. Trace Elem. Med. Biol. 2003, 17, 57–60. [Google Scholar] [CrossRef]
- Rupal, A.V.; Dhrutigna, R.K.; Krutika, L.B.; Narasimhacharya, A.V.R.L. Therapeutic benefits of glibenclamide in fluoride intoxicated diabetic rats. Fluoride 2010, 43, 141–149. [Google Scholar]
- García-Montalvo, E.A.; Reyes-Pérez, H.; Del Razo, L.M. Fluoride exposure impairs glucose tolerance via decreased insulin expression and oxidative stress. Toxicology 2009, 263, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Grucka-Mamczar, E.; Birkner, E.; Zalejska-Fiolka, J.; Machoy, Z.; Kasperczyk, S.; Blaszczyk, I. Influence of extended exposure to sodium fluoride and caffeine on the activity of carbohydrate metabolism enzymes in rat blood serum and liver. Fluoride 2007, 40, 62–66. [Google Scholar]
- Sakurai, T.; Suzuki, K.; Taki, T.; Suketa, V.T. The mechanism of changes in metabolism and transport of glucose caused by fluoride administration to rats. Fluoride 1993, 26, 210. [Google Scholar]
- Al Alam, N.; Kreydiyyeh, S.I. FTY720P inhibits hepatic Na+–K+ ATPase via S1PR2 and PGE2. Biochem. Cell Biol. 2016, 94, 371–377. [Google Scholar] [CrossRef]
- Al Alam, N.; Kreydiyyeh, S.I. Signaling pathway involved in the inhibitory effect of FTY720P on the Na+/K+ ATPase in HepG2 cells. J. Cell Commun. Signal. 2017, 11, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Gutowska, I.; Baranowska-Bosiacka, I.; Siennicka, A.; Bakiewicz, M.; Machaliński, B.; Stachowska, E.; Chlube, D. Fluoride and generation of pro-inflammatory factors in human macrophages. Fluoride 2011, 44, 125–134. [Google Scholar]
- Gutowska, I.; Baranowska-Bosiacka, I.; Goschorska, M.; Kolasa, A.; Łukomska, A.; Jakubczyk, K.; Dec, K.; Chlubek, D. Fluoride as a factor initiating and potentiating inflammation in THP1 differentiated monocytes/macrophages. Toxicol. In Vitro 2015, 29, 1661–1668. [Google Scholar] [CrossRef] [PubMed]
- Kitazawa, M.; Shibata, Y.; Hashimoto, S.; Ohizumi, Y.; Yamakuni, T. Proinsulin C-peptide stimulates a PKC/IkappaB/NF-kappaB signaling pathwayto activate COX-2 gene transcription in Swiss 3T3 fibroblasts. J. Biochem. 2006, 139, 1083–1088. [Google Scholar] [CrossRef] [PubMed]
- Ke, J.; Long, X.; Liu, Y.; Zhang, Y.F.; Li, J.; Fang, W.; Meng, Q.G. Role of NF-kappaB in TNF-alpha-induced COX-2 expression in synovial fibroblasts from human TMJ. J. Dent. Res. 2007, 86, 363–367. [Google Scholar] [CrossRef]
- Stachowska, E.; Baśkiewicz-Masiuk, M.; Machaliński, B.; Rybicka, M.; Gutowska, I.; Bober, J.; Grymula, K.; Dziedziejko, V.; Chlubek, D. Sodium fluoride enhancement of monocyte differentiation via nuclear factor κB mechanism. Fluoride 2005, 38, 297–306. [Google Scholar]
- Tian, Y.; Huo, M.; Li, G.; Wang, J. Regulation of LPS-induced mRNA expression of pro-inflammatory cytokines via alteration of NF-κB activity in mouse peritoneal macrophages exposed to fluoride. Chemosphere 2016, 161, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Refsnes, M.; Skuland, T.; Låg, M.; Schwarze, P.E.; Øvrevik, J. Differential NF-κB and MAPK activation underlies fluoride- and TPA-mediated CXCL8 (IL-8) induction in lung epithelial cells. J. Inflamm. Res. 2014, 7, 169–185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, Q.; Cui, H.; Deng, H.; Kuang, P.; Liu, H.; Lu, Y.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; et al. Sodium fluoride induces renal inflammatory responses by activating NF-κB signaling pathway and reducing anti-inflammatory cytokine expression in mice. Oncotarget 2017, 8, 80192–80207. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thangapandiyan, S.; Miltonprabu, S. Epigallocatechin gallate supplementation protects against renal injury induced by fluoride intoxication in rats: Role of Nrf2/HO-1 signaling. Toxicol. Rep. 2014, 1, 12–30. [Google Scholar] [CrossRef] [Green Version]
- Deng, H.; Kuang, P.; Cui, H.; Luo, Q.; Liu, H.; Lu, Y.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; et al. Sodium fluoride induces apoptosis in mouse splenocytes by activating ROS-dependent NF-κB signalling. Oncotarget 2017, 8, 114428–114441. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Wang, A.; Xia, T.; He, P. Effects of fluoride on DNA damage, S-phase cell-cycle arrest and the expression of NF-kappaB in primary cultured rat hippocampal neurons. Toxicol. Lett. 2008, 179, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhu, W.J.; Xu, X.H.; Zhang, Z.G. Effect of fluoride on calcium ion concentration and expression of nuclear transcription factor kappa-B ρ65 in rat hippocampus. Exp. Toxicol. Pathol. 2011, 63, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Misra, U.K.; Gawdi, G.; Pizzo, S.V. Beryllium fluoride-induced cell proliferation: A process requiring P21(ras)-dependent activated signal transduction and NF-kappaB-dependent gene regulation. J. Leukoc. Biol. 2002, 71, 487–494. [Google Scholar]
- Abdulrazzaq, Y.M.; Osman, N.; Nagelkerke, N.; Kosanovic, M.; Adem, A. Trace element composition of plasma and breast milk of well-nourished women. J. Environ. Sci. Health A Toxicol. Hazard. Subst. Environ. Eng. 2008, 43, 329–334. [Google Scholar] [CrossRef]
- Ikem, A.; Nwankwoala, A.; Odueyungbo, S.; Nyavora, K.; Egiebora, N. Levels of 26 elements in infant formula from USA, UK, and Nigeria by microwave digestion and ICP–OES. Food Chem. 2002, 77, 439–447. [Google Scholar] [CrossRef]
- Combeau, C.; Carlier, M.F. Characterization of the Aluminum and Beryllium Fluoride Species Bound to F-actin and Microtubules at the Site of the y-Phosphate of the Nucleotide. J. Biol. Chem. 1989, 264, 1907–19021. [Google Scholar]
- Antonny, B.; Chabre, M. Characterization of the aluminum and beryllium fluoride species which activate transducin. Analysis of the binding and dissociation kinetics. J. Biol. Chem. 1992, 267, 6710–6718. [Google Scholar] [PubMed]
- Bigay, J.; Deterre, P.; Pfister, C.; Chabre, M. Fluoride complexes of aluminium or beryllium act on G-proteins as reversibly bound analogues of the gamma phosphate of GTP. EMBO J. 1987, 6, 2907–2913. [Google Scholar] [CrossRef] [PubMed]
- Chabre, M. Aluminofluoride and beryllofluoride complexes: A new phosphate analogs in enzymology. Trends Biochem. Sci. 1990, 15, 6–10. [Google Scholar] [CrossRef]
- Pietkiewicz, J.; Gamian, A.; Staniszewska, M.; Danielewicz, R. Inhibition of human muscle-specific enolase by methylglyoxal and irreversible formation of advanced glycation end products. J. Enzyme Inhib. Med. Chem. 2009, 24, 356–364. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gallicchio, M.A.; Bach, L.A. Advanced glycation end products inhibit Na+ K+ ATPase in proximal tubule epithelial cells: Role of cytosolic phospholipase A2alpha and phosphatidylinositol 4-phosphate 5-kinase gamma. Biochim. Biophys. Acta 2010, 1803, 919–930. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.L.; Lou, D.D.; Guan, Z.Z. Activation of the AGE/RAGE system in the brains of rats and in SH-SY5Y cells exposed to high level of fluoride might connect to oxidative stress. Neurotoxicol. Teratol. 2015, 48, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, M.; Kordi-Tamandani, D.M.; Sharifi, N.; Moazeni-Roodi, A.; Kaykhaei, M.A.; Narouie, B.; Torkmanzehi, A. Serum paraoxonase and arylesterase activities in metabolic syndrome in Zahedan, southeast Iran. Eur. J. Endocrinol. 2011, 164, 219–222. [Google Scholar] [CrossRef] [Green Version]
- Pavlov, V.A.; Ochani, M.; Gallowitsch-Puerta, M.; Ochani, K.; Huston, J.M.; Czura, C.J.; Al-Abed, Y.; Tracey, K.J. Central muscarinic cholinergic regulation of the systemic inflammatory response during endotoxemia. Proc. Natl. Acad. Sci. USA 2006, 103, 5219–5223. [Google Scholar] [CrossRef] [Green Version]
- Mabley, J.G.; Pacher, P.; Szabo, C. Activation of the cholinergic anti-inflammatory pathway reduces ricin-induced mortality and organ failure in mice. Mol. Med. 2009, 15, 166–172. [Google Scholar] [CrossRef]
- Søyseth, V.; Kongerud, J.; Broen, P.; Lilleng, P.; Boe, J. Bronchial responsiveness, eosinophilia, and short term exposure to air pollution. Arch. Dis. Child. 1995, 73, 418–422. [Google Scholar] [CrossRef]
- Søyseth, V.; Kongerud, J.; Ekstrand, J.; Boe, J. Relation between exposure to fluoride and bronchial responsiveness in aluminium potroom workers with work-related asthma-like symptoms. Thorax 1994, 49, 984–989. [Google Scholar] [CrossRef]
- Van Schoor, J.; Joos, G.F.; Pauwels, R.A. Indirect bronchial hyperresponsiveness in asthma:mechanisms, pharmacology and implications for clinical research. Eur. Respir. J. 2000, 16, 514–533. [Google Scholar] [CrossRef] [PubMed]
- Burrows, B.; Sears, M.R.; Flannery, E.M.; Herbison, G.P.; Holdaway, D. Relationships of bronchial responsiveness assessed by methacholine to serum IgE, lung function, symptoms, and diagnoses in 11-year-old New Zealand children. J. Allergy Clin. Immunol. 1992, 90, 376–385. [Google Scholar] [CrossRef]
- Joos, G.F.; O’Connor, B.; Anderson, S.D.; Chung, F.; Cockcoft, D.W.; Dahlen, B.; Di Maria, G.; Foresi, A.; Hargreave, F.E.; Holgate, S.T.; et al. Indirect airway challenges. Eur. Respir. J. 2003, 21, 1050–1068. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anderson, S.D.; Brannan, J.D. Methods for “indirect” challenge tests including exercise, eucapnic voluntary hyperpnes, and hypertonic aerolos. Clin. Rev. Allergy Immunol. 2003, 24, 27–54. [Google Scholar] [CrossRef]
- Nielson, K.G. Lung Function and bronchial responsiveness in young children. Dan Med. Bull. 2006, 53, 46–75. [Google Scholar]
- Rijchen, B.; Schouten, J.P.; Weiss, S.T.; Speizer, F.E.; Van der Lende, R. The relationship between airways responsiveness to histamine and pulmonary function level in a random population sample. Bull. Eur. Physiopathol. Respir. 1987, 23, 391–394. [Google Scholar] [CrossRef]
- Kongerud, J.; Soyseth, V. Methacholine responsiveness, respiratory symptoms and pulmonary function in aluminium potroom workers. Eur. Respir. J. 1991, 4, 159–166. [Google Scholar] [PubMed]
- Joseph-Bowen, J.; de Klerk, N.H.; Firth, M.J.; Kendall, G.E.; Holt, P.G.; Sly, P.D. Lung Function, Bronchial Responsiveness, and Asthma in a Community Cohort of 6-Year-Old Children. Am. J. Respir. Crit. Care Med. 2004, 169, 850–854. [Google Scholar] [CrossRef]
- Adinolfi, M. The development of the human blood-CSF-brain barrier. Dev. Med. Child Neurol. 1985, 27, 532–537. [Google Scholar] [CrossRef] [PubMed]
- Johanson, C.E. Permeability and vascularity of the developing brain: Cerebellum vs. cerebral cortex. Brain Res. 1980, 190, 3–16. [Google Scholar] [CrossRef]
- Sullivan, J.L.; Newton, R.B. Serum antioxidant activity in neonates. Arch. Dis. Child. 1988, 63, 748–750. [Google Scholar] [CrossRef]
- Gutteridge, J.M.C.; Stocks, J. Cacruloplasmin: Physiological and pathological perspectives. Crit. Rev. Clin. Lab. Sci. 1981, 14, 257–329. [Google Scholar] [CrossRef] [PubMed]
- National Research Council. Pesticides in the Diets of Infants and Children; National Academy Press: Washington, DC, USA, 1993. [Google Scholar]
- Dourson, M.; Charnley, G.; Scheuplein, R. Differential sensitivity of children and adults to chemical toxicity. II. Risk and regulation. Regul. Toxicol. Pharmacol. 2002, 35, 448–467. [Google Scholar] [CrossRef]
- Naik, U.S.; Gangadharan, C.; Abbagani, K.; Nagalla, B.; Dasari, N.; Manna, S.K. A study of nuclear transcription factor-kappa B in childhood autism. PLoS ONE 2011, 6, e19488. [Google Scholar] [CrossRef] [PubMed]
- Young, A.M.; Campbell, E.; Lynch, S.; Suckling, J.; Powis, S.J. Aberrant NF-kappaB expression in autism spectrum condition: A mechanism for neuroinflammation. Front. Psychiatry 2011, 2, 27. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Salam, O.M.E.; Youness, E.R.; Mohammed, N.A.; Elhamed, W.A.A. Nuclear Factor-Kappa B and Other Oxidative Stress Biomarkers in Serum of Autistic Children. Open J. Mol. Integr. Physiol. 2015, 5, 18–27. [Google Scholar] [CrossRef]
- Ghanizadeh, A. Nuclear factor kappa B may increase insight into the management of neuroinflammation and excitotoxicity in autism. Expert Opin. Ther. Targets 2011, 15, 781–783. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Li, X.; Zhou, W.; Lou, D.; Huang, D.; Li, Y.; Kang, Y.; Xiang, Y.; Li, T. Regulation of set gene expression by NF-κB. Mol. Neurobiol. 2016, 54, 4477–4485. [Google Scholar] [CrossRef]
- Lukiw, W.J.; Bazan, N.G. Strong nuclear factor-κB-DNA binding parallels cyclooxygenase-2 gene transcription in aging and in sporadic Alzheimer’s disease superior temporal lobe neocortex. J. Neurosci. Res. 1998, 53, 583–592. [Google Scholar] [CrossRef]
- Boissiere, F.; Hunot, S.; Faucheux, B.; Duyckaerts, C.; Hauw, J.J.; Agid, Y.; Hirsch, E.C. Nuclear translocation of NF-κB in cholinergic neurons of patients with Alzheimer’s disease. Neuroreport 1997, 8, 2849–2852. [Google Scholar] [CrossRef]
- Akiyama, H.; Nishimura, T.; Kondo, H.; Ikeda, K.; Hayashi, Y.; McGeer, P.L. Expression of the receptor for macrophage colony stimulating factor by brain microglia and its upregulation in brains of patients with Alzheimer’s disease and amyotrophic lateral sclerosis. Brain Res. 1994, 639, 171–174. [Google Scholar] [CrossRef]
- Hunot, S.; Brugg, B.; Ricard, D.; Michel, P.P.; Muriel, M.P.; Ruberg, M.; Faucheux, B.A.; Agid, Y.; Hirsch, E.C. Nuclear translocation of NF-κB is increased in dopaminergic neurons of patients with Parkinson disease. Proc. Natl. Acad. Sci. USA 1997, 94, 7531–7536. [Google Scholar] [CrossRef] [PubMed]
- Block, M.L.; Hong, J.S. Microglia and inflammation-mediated neurodegeneration: Multiple triggers with a common mechanism. Prog. Neurobiol. 2005, 76, 77–98. [Google Scholar] [CrossRef] [Green Version]
- Edwards, M.R.; Bartlett, N.W.; Clarke, D.; Birrell, M.; Belvisi, M.; Johnston, S.L. Targeting the NF-kappaB pathway in asthma and chronic obstructive pulmonary disease. Pharmacol. Ther. 2009, 121, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Barnes, P.J.; Karin, M. Nuclear factor-kappaB: A pivotal transcription factor in chronic inflammatory diseases. N. Engl. J. Med. 1997, 336, 1066–1071. [Google Scholar] [CrossRef] [PubMed]
- ISSAC Steering Committee. Worldwide variations in the prevalence of asthma symptoms: The International Study of Asthma and Allergies in Childhood (ISAAC). Eur. Respir. J. 1998, 12, 315–335. [Google Scholar] [CrossRef]
- Lai, C.K.W.; Beasley, R.; Crane, J.; Foliaki, S.; Shah, J.; Weiland, S.; The ISAAC Phase Three Study Group. Global variation in the prevalence and severity of asthma symptoms: Phase Three of the International Study of Asthma and Allergies in Childhood (ISAAC). Thorax 2009, 64, 476–483. [Google Scholar] [CrossRef]
- Burney, P.G.J.; Luczynska, C.; Chinn, S.; Jarvis, D. The European Community Respiratory Health Survey. Eur. Respir. J. 1994, 7, 954–960. [Google Scholar] [CrossRef] [Green Version]
- European Community Respiratory Health Survey (ECRHS). Variations in the prevalence of respiratory symptoms, self-reported asthma attacks, and use of asthma medication in the European Community Respiratory Health Survey (ECRHS). Eur. Respir. J. 1996, 9, 687–695. [Google Scholar] [CrossRef] [Green Version]
- Australian Institute of Health and Welfare. Australian National Infant Feeding Survey: Indicator Results; Cat. no. PHE 156; AIHW: Canberra, Australia, 2010. Available online: http://www.aihw.gov.au/publication-detail/?id=10737420927 (accessed on 19 March 2019).
- The longitudinal Study of Australian Children 2006–2007 Annual Report. Available online: https://growingupinaustralia.gov.au/sites/default/files/annualreport2006-07.pdf (accessed on 19 March 2019).
- OECD Family Database, CO1.5: Breastfeeding Rates. 2009. Available online: https://www.oecd.org/els/family/43136964.pdf (accessed on 15 March 2019).
- O’Donovan, S.M.; Murray, D.M.; Hourihane, J.B.; Kenny, L.C.; Irvine, A.D.; Kiely, M. Adherence with early infant feeding and complementary feeding guidelines in the Cork BASELINE Birth Cohort Study. Public Health Nutr. 2015, 18, 2864–2873. [Google Scholar] [CrossRef] [Green Version]
- New Zealand’s Breastfeeding Rates—Statistics from Breastfeeding: A Guide to Action; New Zealand Ministry of Health: Wellington, New Zealand, 2002. Available online: https://www.health.govt.nz/system/files/documents/publications/breastfeeding.pdf (accessed on 15 January 2019).
- Centers for Disease Control and Prevention. Racial and Ethnic Differences in Breastfeeding Initiation and Duration, by State National Immunization Survey, United States. 2004—2008. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/mm5911a2.htm (accessed on 15 January 2019).
- Williams, H.; McNicol, K.N. Prevalence, natural history and relationship of wheezy bronchitis and asthma in children. An epidemiological study. Br. Med. J. 1969, 4, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Akinbami, L.J.; Moorman, J.E.; Garbe, P.L.; Sondik, E.J. Status of Childhood Asthma in the United States, 1980–2007. Pediatrics 2009, 123, S131–S145. [Google Scholar] [CrossRef] [PubMed]
- Greally, P. Childhood Asthma: Prevalence and Presentation. Continuing Education Module 19: Child Health. WIN. 2012. Available online: https://www.inmo.ie/tempDocs/ChildHlth-Asthma%20PAGE41-42.pdf (accessed on 19 March 2019).
- Masoli, M.; Fabian, D.; Holt, S.; Beasley, R.; Global Initiative for Asthma (GINA) Program. The global burden of asthma: Executive summary of the GINA Dissemination Committee report. Allergy 2004, 59, 469–478. [Google Scholar] [CrossRef]
- Whelton, H.; Crowley, E.; O’Mullane, D.; Donaldson, M.; Kelleher, V.; Cronin, M. Dental caries and enamel fluorosis among the fluoridated and non-fluoridated populations in the Republic of Ireland in 2002. Community Dent. Health 2004, 21, 37–44. [Google Scholar]
- NCHS Data Brief Number 53, November 2010, Prevalence and Severity of Dental Fluorosis in the United States. 1999–2004. Available online: http://www.cdc.gov/nchs/data/databriefs/db53.htm (accessed on 19 January 2019).
- Do, L.G.; Spencer, A.J. Decline in the prevalence of dental fluorosis among South Australian children. Community Dent. Oral Epidemiol. 2007, 35, 282–291. [Google Scholar] [CrossRef]
- Obry-Musset, A.M. Epidemiology of dental caries in children. Arch. Pediatr. 1998, 5, 1145–1148. [Google Scholar] [CrossRef]
- Momeni, A.; Neuhäuser, A.; Renner, N.; Heinzel-Gutenbrunner, M.; Abou-Fidah, J.; Rasch, K.; Kröplin, M.; Fejerskov, O.; Pieper, K. Prevalence of dental fluorosis in German school children in areas with different preventive programmes. Caries Res. 2007, 41, 437–444. [Google Scholar] [CrossRef]
- Trabelsi, M.; Guermazi, F.; Zeghal, N. Effect of fluoride on thyroid function and cerebellar development in mice. Fluoride 2001, 34, 165–173. [Google Scholar]
- Bouaziz, H.; Ammar, E.; Ghorbel, H.; Ketata, S.; Jamoussi, K.; Ayadi, F.; Guermazi, F.; Zeghal, N. Effect of fluoride ingested by lactating mice on the thyroid function and bone maturation of their suckling pups. Fluoride 2004, 37, 133–142. [Google Scholar]
- Bouaziz, H.; Soussia, L.; Guermazi, F.; Zeghal, N. Fluoride-induced thyroid proliferative changes and their reversal in female mice and their pups. Fluoride 2005, 38, 185–192. [Google Scholar]
- Sun, Z.; Zhang, Y.; Xue, X.; Niu, R.; Wang, J. Maternal fluoride exposure during gestation and lactation decreased learning and memory ability, and glutamate receptor mRNA expressions of mouse pups. Hum. Exp. Toxicol. 2018, 37, 87–93. [Google Scholar] [CrossRef]
- Blaylock, R.L. Excitotoxicity: A possible central mechanism in fluoride neurotoxicity. Fluoride 2004, 37, 301–314. [Google Scholar]
- Dong, Y.T.; Wang, Y.; Wei, N.; Zhang, Q.F.; Guan, Z.Z. Deficit in learning and memory of rats with chronic fluorosis correlates with the decreased expressions of M1 and M3 muscarinic acetylcholine receptors. Arch. Toxicol. 2015, 89, 1981–1991. [Google Scholar] [CrossRef]
- Dong, Y.T.; Wei, N.; Qi, A.L.; Liu, X.H.; Chen, D.; Zeng, A.A.; Guan, Z.Z. Attenuating effect of Vitamin E on the deficit of learning and memory of rats with chronic fluorosis: The mechanism may involve Muscarinic Acetylcholine Receptors. Fluoride 2017, 50, 354–364. [Google Scholar]
- Raedler, T.J.; Bymaster, F.P.; Tandon, R.; Copolov, D.; Dean, B. Towards a muscarinic hypothesis of schizophrenia. Mol. Psychiatry 2007, 12, 232–246. [Google Scholar] [CrossRef]
- Gibbons, A.S.; Scarr, E.; McLean, C.; Sundram, S.; Dean, B. Decreased muscarinic receptor binding in the frontal cortex of bipolar disorder and major depressive disorder subjects. J. Affect. Disord. 2008, 116, 184–191. [Google Scholar] [CrossRef]
- Medeiros, R.; Kitazawa, M.; Caccamo, A.; Baglietto-Vargas, D.; Estrada-Hernandez, T.; Cribbs, D.H.; Fisher, A.; LaFerla, F.M. Loss of Muscarinic M1 Receptor Exacerbates Alzheimer’s Disease–Like Pathology and Cognitive Decline. Am. J. Pathol. 2011, 179, 980–991. [Google Scholar] [CrossRef]
- Miyakawa, T.; Yamada, M.; Duttaroy, A.; Wess, J. Hyperactivity and intact hippocampus-dependent learning in mice lacking the M1 muscarinic acetylcholine receptor. J. Neurosci. 2001, 21, 5239–5250. [Google Scholar] [CrossRef] [PubMed]
- Gautam, D.; Han, S.J.; Hamdan, F.F.; Jeon, J.; Li, B.; Li, J.H.; Cui, Y.; Mears, D.; Lu, H.; Deng, C.; et al. A critical role for β cell M3muscarinic acetylcholine receptors in regulating insulin release and blood glucose homeostasis in vivo. Cell Metab. 2006, 3, 449–461. [Google Scholar] [CrossRef] [PubMed]
- Smit, B.J.; Kok, J.H.; Vulsma, T.; Briet, J.M.; Boer, K.; Wiersinga, W.M. Neurologic development of the newborn and young child in relation to maternal thyroid function. Acta Paediatr. 2000, 89, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Glinoer, D.; Delange, F. The potential repercussions of maternal, fetal, and neonatal hypothyroxinemia on the progeny. Thyroid 2000, 10, 871–887. [Google Scholar] [CrossRef] [PubMed]
- Willoughby, K.A.; McAndrews, M.P.; Rovet, J.F. Effects of Maternal Hypothyroidism on Offspring Hippocampus and Memory. Thyroid 2014, 24, 576–584. [Google Scholar] [CrossRef] [PubMed]
- Gillberg, I.C.; Gillberg, C.; Kopp, S. Hypothyroidism and autism spectrum disorders. J. Child Psychol. Psychiatry 1992, 33, 531–542. [Google Scholar] [CrossRef]
- Vermiglio, F.; Lo Presti, V.P.; Moleti, M.; Sidoti, M.; Tortorella, G.; Scaffidi, G.; Castagna, M.G.; Mattina, F.; Violi, M.A.; Crisà, A.; et al. Attention deficit and hyperactivity disorders in the offspring of mothers exposed to mild-moderate iodine deficiency: A possible novel iodine deficiency disorder in developed countries. J. Clin. Endocrinol. Metab. 2004, 89, 6054–6060. [Google Scholar] [CrossRef] [PubMed]
- Instanes, J.T.; Halmøy, A.; Engeland, A.; Haavik, J.; Furu, K.; Klungsøyr, K. Attention-Deficit/Hyperactivity Disorder in Offspring of Mothers with Inflammatory and Immune System Diseases. Biol. Psychiatry 2017, 81, 452–459. [Google Scholar] [CrossRef] [PubMed]
- Abel, M.H.; Ystrom, E.; Caspersen, I.H.; Meltzer, H.M.; Aase, H.; Torheim, L.E.; Askeland, R.B.; Reichborn-Kjennerud, T.; Brantsæter, A.L. Maternal Iodine Intake and Offspring Attention-Deficit/Hyperactivity Disorder: Results from a Large Prospective Cohort Study. Nutrients 2017, 9, 1239. [Google Scholar] [CrossRef] [PubMed]
- Getahun, D.; Jacobsen, S.J.; Fassett, M.J.; Wing, D.A.; Xiang, A.H.; Chiu, V.Y.; Peltier, M.R. Association between maternal hypothyroidism and autism spectrum disorders in children. Pediatr. Res. 2018, 83, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Andersen, S.L.; Laurberg, P.; Wu, C.S.; Olsen, J. Attention deficit hyperactivity disorder and autism spectrum disorder in children born to mothers with thyroid dysfunction: A Danish nationwide cohort study. Int. J. Obstet. Gynaecol. 2014, 121, 1365–1374. [Google Scholar] [CrossRef]
- Gyllenberg, D.; Sourander, A.; Surcel, H.M.; Hinkka-Yli-Salomäki, S.; McKeague, I.W.; Brown, A.S. Hypothyroxinemia During Gestation and Offspring Schizophrenia in a National Birth Cohort. Biol. Psychiatry 2016, 79, 962–970. [Google Scholar] [CrossRef] [PubMed]
- Andersen, S.L.; Laurberg, P.; Wu, C.S.; Olsen, J. Maternal Thyroid Dysfunction and Risk of Seizure in the Child: A Danish Nationwide Cohort Study. J. Pregnancy 2013, 2013, 636705. [Google Scholar] [CrossRef]
- Liu, X.; Andersen, S.L.; Olsen, J.; Agerbo, E.; Schlünssen, V.; Dharmage, S.C.; Munk-Olsen, T. Maternal hypothyroidism in the perinatal period and childhood asthma in the offspring. Allergy 2018, 73, 932–939. [Google Scholar] [CrossRef]
- Aydin, G.; Ciçek, E.; Akdoğan, M.; Gökalp, O. Histopathological and biochemical changes in lung tissues of rats following administration of fluoride over several generations. J. Appl. Toxicol. 2003, 23, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Karaoz, E.; Oncu, M.; Gulle, K.; Kanter, M.; Gultekin, F.; Karaoz, S.; Mumcu, E. Effect of chronic fluorosis on lipid peroxidation and histology of kidney tissues in first- and second-generation rats. Biol. Trace Elem. Res. 2004, 102, 199–208. [Google Scholar] [CrossRef]
- Oncu, M.; Gulle, K.; Karaoz, E.; Gultekin, F.; Karaoz, S.; Karakoyun, I.; Mumcu, E. Effect of chronic fluorosis on lipid peroxidation and histology of lung tissues in first and second generation rats. Toxicol. Ind. Health 2006, 22, 375–380. [Google Scholar] [CrossRef] [PubMed]
- Oncü, M.; Kocak, A.; Karaoz, E.; Darici, H.; Savik, E.; Gultekin, F. Effect of long-term fluoride exposure on lipid peroxidation and histology of testes in first- and second-generation rats. Biol. Trace Elem. Res. 2007, 118, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Aluru, N.; Karchner, S.I.; Glazer, L. Early Life Exposure to Low Levels of AHR Agonist PCB126 (3,3′,4,4′,5-Pentachlorobiphenyl) Reprograms Gene Expression in Adult Brain. Toxicol. Sci. 2017, 160, 386–397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stankiewicz, A.M.; Swiergiel, A.H.; Lisowski, P. Epigenetics of stress adaptations in the brain. Brain Res. Bull. 2013, 98, 76–92. [Google Scholar] [CrossRef] [Green Version]
- Bravo, J.A.; Dinan, T.G.; Cryan, J.F. Early-life stress induces persistent alterations in 5-HT1A receptor and serotonin transporter mRNA expression in the adult rat brain. Front. Mol. Neurosci. 2014, 7, 24. [Google Scholar] [CrossRef]
- Mpofana, T.; Daniels, W.M.; Mabandla, M.V. Exposure to Early Life Stress Results in Epigenetic Changes in Neurotrophic Factor Gene Expression in a Parkinsonian Rat Model. Parkinsons Dis. 2016, 2016, 6438783. [Google Scholar] [CrossRef]
- Cohen, S.; Janicki-Deverts, D.; Doyle, W.; Miller, G.E.; Frank, E.; Rabin, B.S.; Turner, R.B. Chronic stress, glucocorticoid receptor resistance, inflammation, and disease risk. Proc. Natl. Acad. Sci. USA 2012, 109, 5995–5999. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.; Xia, T.; Zhang, M.; He, W.; He, P.; Chen, X.; Yang, K.; Wang, A. Screening of Environmental Response Genes Related to Dental Fluorosis. Fluoride 2006, 39, 195–201. [Google Scholar]
- Swift, R.G.; Polymeropoulos, M.H.; Torres, R.; Swift, M. Predisposition of Wolfram syndrome heterozygotes to psychiatric illness. Mol. Psychiatry 1998, 3, 86–91. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Swift, M.; Swift, R.G. Psychiatric disorders and mutations at the Wolfram syndrome locus. Biol. Psychiatry 2000, 47, 787–793. [Google Scholar] [CrossRef]
- Swift, M.; Swift, R.G. Wolframin mutations and hospitalization for psychiatric illness. Mol. Psychiatry 2005, 10, 799–803. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harding, H.P.; Ron, D. Endoplasmic reticulum stress and the development of diabetes: A review. Diabetes 2002, 51, S455–S461. [Google Scholar] [CrossRef] [PubMed]
- Rohayem, J.; Ehlers, C.; Wiedemann, B.; Holl, R.; Oexle, K.; Kordonouri, O.; Salzano, G.; Meissner, T.; Burger, W.; Schober, E.; et al. Diabetes and neurodegeneration in Wolfram syndrome: A multicenter study of phenotype and genotype. Diabetes Care 2011, 34, 1503–1510. [Google Scholar] [CrossRef] [PubMed]
- Mayer-Davis, E.J.; Dabelea, D.; Lawrence, J.M. Incidence Trends of Type 1 and Type 2 Diabetes among Youths, 2002–2012. N. Engl. J. Med. 2017, 377, 301. [Google Scholar] [CrossRef] [PubMed]
- Neurath, C.; Limeback, H.; Osmunson, B.; Connett, M.; Kanter, V.; Wells, C.R. Dental Fluorosis Trends in US Oral Health Surveys: 1986 to 2012. JDR Clin. Transl. Res. 2019. [Google Scholar] [CrossRef] [PubMed]
- Wiener, R.C.; Shen, C.; Findley, P.; Tan, X.; Sambamoorthi, U. Dental Fluorosis over Time: A comparison of National Health and Nutrition Examination Survey data from 2001–2002 and 2011–2012. J. Dent. Hyg. 2018, 92, 23–29. [Google Scholar]
- Chafe, R.; Aslanov, R.; Sarkar, A.; Gregory, P.; Comeau, A.; Newhook, L.A. Association of type 1 diabetes and concentrations of drinking water components in Newfoundland and Labrador, Canada. BMJ Open Diabetes Res. Care 2018, 6, e000466. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fluegge, K. Community water fluoridation predicts increase in age-adjusted incidence and prevalence of diabetes in 22 states from 2005 and 2010. J. Water Health 2016, 14, 864–877. [Google Scholar] [CrossRef] [Green Version]
- Harding, H.P.; Zeng, H.; Zhang, Y.; Jungries, R.; Chung, P.; Plesken, H.; Sabatini, D.D.; Ron, D. Diabetes mellitus and exocrine pancreatic dysfunction in perk-/-mice reveals a role for translational control in secretory cell survival. Mol. Cell. 2001, 7, 1153–1163. [Google Scholar] [CrossRef]
- Mathers, C.; Loncar, D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006, 28, e442. [Google Scholar] [CrossRef] [PubMed]
- Jemal, A.; Ward, E.; Hao, Y.; Thun, M. Trends in the leading causes of death in the United States, 1970–2002. JAMA 2005, 294, 1255–1259. [Google Scholar] [CrossRef]
- OECD. Health at a Glance 2011; OECD Indicators, Ed.; OECD Publishing: Paris, France, 2011; Available online: https://www.oecd.org/els/health-systems/49105858.pdf (accessed on 22 January 2019).
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Global Cancer Data by Country; World Cancer Research Fund, American Institute for Cancer Research: Washington, DC, USA, 2018; Available online: https://www.wcrf.org/dietandcancer/cancer-trends/data-cancer-frequency-country (accessed on 22 January 2019).
- Malfatti, C.R.; Burgos, L.T.; Rieger, A.; Rüdger, C.L.; Túrmina, J.A.; Pereira, R.A.; Pavlak, J.L.; Silva, L.A.; Osiecki, R. Decreased erythrocyte NA+, K+-ATPase activity and increased plasma TBARS in prehypertensive patients. Sci. World J. 2012, 2012, 348246. [Google Scholar] [CrossRef]
- Sun, L.; Gao, Y.; Liu, H.; Zhang, W.; Ding, Y.; Li, B.; Li, M.; Sun, D. An assessment of the relationship between excess fluoride intake from drinking water and essential hypertension in adults residing in fluoride endemic areas. Sci. Total Environ. 2013, 443, 864–869. [Google Scholar] [CrossRef]
- Haghighat, G.A.; Yunesian, M.; Amini, H. Hypertension and drinking water fluoride? Is there a relationship? Fluoride 2012, 45 Pt 1, 167. [Google Scholar]
- Tomar, S.; Mishra, S.; Choudhary, M.; Chauhan, D.S.; Yadav, P.; Singh, V.P.; Joshi, D.K.; Tripathi, S.; Tomar, S.; Tomar, A. Increased oxidative burden in hypertensive retinopathy patients and its association with fluoride exposure in population of Rajasthan India. Fluoride 2012, 45, 207–208. [Google Scholar]
- Amini, H.; Taghavi Shahri, S.M.; Amini, M.; Ramezani Mehrian, M.; Mokhayeri, Y.; Yunesian, M. Drinking water fluoride and blood pressure? An environmental study. Biol. Trace Elem. Res. 2011, 144, 157–163. [Google Scholar] [CrossRef]
- Singh, K.P.; Dash, R.J.; Varma, J.S.; Singh, M.; Gauba, K.; Dhillon, M.S. Incidence of cardiovascular abnormalities in endemic skeletal fluorosis. Fluoride 1998, 31, S22. [Google Scholar]
- Bera, I.; Sabatini, R.; Auteri, P.; Flace, P.; Sisto, G.; Montagnani, M.; Potenza, M.A.; Marasciulo, F.L.; Carratu, M.R.; Coluccia, A.; et al. Neurofunctional effects of developmental sodium fluoride exposure in rats. Eur. Rev. Med. Pharmacol. Sci. 2007, 11, 211–224. [Google Scholar] [PubMed]
- Omóbòwálé, T.O.; Oyagbemi, A.A.; Alaba, B.A.; Ola-Davies, O.E.; Adejumobi, O.A.; Asenuga, E.R.; Ajibade, T.O.; Adedapo, A.A.; Yakubu, M.A. Ameliorative effect of Azadirachta indica on sodium fluoride-induced hypertension through improvement of antioxidant defence system and upregulation of extracellular signal regulated kinase 1/2 signaling. J. Basic Clin. Physiol. Pharmacol. 2018, 29, 155–164. [Google Scholar] [CrossRef]
- Oyagbemi, A.A.; Omobowale, T.O.; Asenuga, E.R.; Adejumobi, A.O.; Ajibade, T.O.; Ige, T.M.; Ogunpolu, B.S.; Adedapo, A.A.; Yakubu, M.A. Sodium fluoride induces hypertension and cardiac complications through generation of reactive oxygen species and activation of nuclear factor kappa beta. Environ. Toxicol. 2017, 32, 1089–1101. [Google Scholar] [CrossRef] [PubMed]
- Oyagbemi, A.A.; Omobowale, T.O.; Ola-Davies, O.E.; Asenuga, E.R.; Ajibade, T.O.; Adejumobi, O.A.; Arojojoye, O.A.; Afolabi, J.M.; Ogunpolu, B.S.; Falayi, O.O.; et al. Quercetin attenuates hypertension induced by sodium fluoride via reduction in oxidative stress and modulation of HSP 70/ERK/PPARγ signaling pathways. Biofactors 2018, 44, 465–479. [Google Scholar] [CrossRef]
- Oyagbemi, A.A.; Omobowale, T.O.; Ola-Davies, O.E.; Asenuga, E.R.; Ajibade, T.O.; Adejumobi, O.A.; Afolabi, J.M.; Ogunpolu, B.S.; Falayi, O.O.; Saba, A.B.; et al. Luteolin-mediated Kim-1/NF-kB/Nrf2 signaling pathways protects sodium fluoride-induced hypertension and cardiovascular complications. Biofactors 2018, 44, 518–531. [Google Scholar] [CrossRef]
- Oyagbemi, A.A.; Omobowale, T.O.; Ola-Davies, O.E.; Asenuga, E.R.; Ajibade, T.O.; Adejumobi, O.A.; Afolabi, J.M.; Ogunpolu, B.S.; Falayi, O.O.; Ayodeji, F.; et al. Ameliorative effect of Rutin on sodium fluoride-induced hypertension through modulation of Kim-1/NF-κB/Nrf2 signaling pathway in rats. Environ. Toxicol. 2018, 33, 1284–1297. [Google Scholar] [CrossRef]
- Garcia, S.I.; Dabsys, S.M.; Martinez, V.N.; Delorenzi, A.; Santajuliana, D.; Nahmod, V.E.; Finkielman, S.; Pirola, C.J. Thyrotropin-releasing hormone hyperactivity in the preoptic area of spontaneously hypertensive rats. Hypertension 1995, 26, 1105–1110. [Google Scholar] [CrossRef]
- Sugi, K.; Much, M.W.; Field, M.; Chang, E.B. Inhibition of Na+, K+-ATPase by interferon gamma down-regulates intestinal epithelial transport and barrier function. Gastroenterology 2001, 120, 1393–1403. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Han, Y.; Dy, A.B.C.; Hagerman, R.J. The Gut Microbiota and Autism Spectrum Disorders. Front. Cell. Neurosci. 2017, 11, 120. [Google Scholar] [CrossRef]
- Malin, A.J.; Till, C. Exposure to fluoridated water and attention deficit hyperactivity disorder prevalence among children and adolescents in the United States: An ecological association. Environ. Health 2015, 14, 17. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.E.; Giust, J.M.; Kronenberger, W.G.; Dunn, D.W. Epilepsy and attention-deficit hyperactivity disorder: Links, risks, and challenges. Neuropsychiatr. Dis. Treat. 2016, 12, 287–296. [Google Scholar]
- Bashash, M.; Thomas, D.; Hu, H.; Martinez-Mier, E.A.; Sanchez, B.N.; Basu, N.; Peterson, K.E.; Ettinger, A.S.; Wright, R.; Zhang, Z.; et al. Prenatal Fluoride Exposure and Cognitive Outcomes in Children at 4 and 6-12 Years of Age in Mexico. Environ. Health Perspect. 2017, 125, 097017. [Google Scholar] [CrossRef] [PubMed]
- Bashash, M.; Marchand, M.; Hu, H.; Till, C.; Martinez-Mier, E.A.; Sanchez, B.N.; Basu, N.; Peterson, K.E.; Green, R.; Schnaas, L.; et al. Prenatal fluoride exposure and attention deficit hyperactivity disorder (ADHD) symptoms in children at 6–12 years of age in Mexico City. Environ. Int. 2018, 121, 658–666. [Google Scholar] [CrossRef]
- Kanık Yüksek, S.; Aycan, Z.; Öner, Ö. Evaluation of Iodine Deficiency in Children with Attention Deficit/Hyperactivity Disorder. J. Clin. Res. Pediatr. Endocrinol. 2016, 8, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Konikowska, K.; Regulska-Ilow, B.; Rózańska, D. The influence of components of diet on the symptoms of ADHD in children. Rocz. Panstw. Zakl. Hig. 2012, 63, 127–134. [Google Scholar] [PubMed]
- Błażewicz, A.; Makarewicz, A.; Korona-Glowniak, I.; Dolliver, W.; Kocjan, R. Iodine in autism spectrum disorders. J. Trace Elem. Med. Biol. 2016, 34, 32–37. [Google Scholar] [CrossRef]
- Hamza, R.T.; Hewedi, D.H.; Sallam, M.T. Iodine deficiency in Egyptian autistic children and their mothers: Relation to disease severity. Arch. Med. Res. 2013, 44, 555–561. [Google Scholar] [CrossRef]
- Sullivan, K.M. Iodine deficiency as a cause of autism. J. Neurol. Sci. 2009, 276, 202. [Google Scholar] [CrossRef] [PubMed]
- Rytter, D.; Andersen, S.L.; Bech, B.H.; Halldorsson, T.I.; Henriksen, T.B.; Laurberg, P.; Olsen, S.F. Maternal thyroid function in pregnancy may program offspring blood pressure, but not adiposity at 20 y of age. Pediatr. Res. 2016, 80, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Census 2011 Ireland and Northern Ireland. Central Statistics Office and Northern Ireland Statistics Research Agency. June 2014. Available online: https://www.cso.ie/en/media/csoie/census/documents/north-south-spreadsheets/Census2011IrelandandNorthernIrelandwebversion1.pdf (accessed on 15 March 2019).
- Balanda, K.P.; Wilde, J. Inequalities in Mortality 1989–1998. A Report on All-Ireland Mortality Data; Institute of Public Health in Ireland: Dublin, Ireland, 2001. [Google Scholar]
- Donnelly, D.W.; Gavin, A.T.; Comber, H. Cancer in Ireland 1994–2004: A Summary Report; National Cancer Registry/Northern Ireland Cancer Registry: Cork/Belfast, UK, 2009; Available online: https://www.ncri.ie/sites/ncri/files/pubs/CancerinIreland1994-2004(SummaryReport).pdf (accessed on 15 March 2019).
- All Ireland Cancer Atlas 1995–2007; National Cancer Registry/Northern Ireland Cancer Registry: Cork/Belfast, UK, 2011; Available online: https://www.ncri.ie/publications/cancer-atlases-and-geographic-studies/all-ireland-cancer-atlas-1995-2007 (accessed on 15 March 2019).
- Takahashi, K.; Akiniwa, K.; Narita, K. Regression Analysis of Cancer Incidence Rates and Water Fluoride in the U.S.A. based on IACR/IARC (WHO) Data (1978–1992). J. Epidemiol. 2001, 11, 170–179. [Google Scholar] [CrossRef] [PubMed]
- OECD/EU. Health at a Glance: Europe 2018: State of Health in the EU Cycle; OECD Publishing: Paris, France, 2018; Available online: https://doi.org/10.1787/health_glance_eur-2018-en (accessed on 19 March 2019).
- Torrey, E.F.; McGuire, M.; O’Hare, A.; Walsh, D.; Spellman, M.P. Endemic psychosis in Western Ireland. Am. J. Psychiatry 1984, 141, 966–970. [Google Scholar] [PubMed]
- Torrey, E.F. Prevalence studies in schizophrenia. Br. J. Psychiatry 1987, 150, 598–608. [Google Scholar] [CrossRef] [PubMed]
- Behan, C.; Kennelly, B.; O’Callaghan, E. The economic cost of schizophrenia in Ireland: A cost of illness study. Ir. J. Psychol. Med. 2008, 25, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Women and Men in Ireland 2013; Central Statistics Office: Blackrock, Ireland, 2013; Available online: https://www.cso.ie/en/releasesandpublications/ep/pwamii/womenandmeninireland2013/healthlist/health/ (accessed on 19 March 2019).
- Goldner, E.; Hsu, L.; Waraich, P.; Somers, J. Prevalence and incidence studies of schizophrenic disorders: A systematic review of the literature. Can. J. Psychiatry 2002, 47, 833–843. [Google Scholar] [CrossRef] [PubMed]
- Winder, D.G.; Sweatt, J.D. Roles of serine/threonine phosphatases in hippocampal synaptic plasticity. Nat. Rev. Neurosci. 2001, 2, 461–474. [Google Scholar] [CrossRef]
- Macedoni-Lukšič, M.; Petrič, I.; Cestnik, B.; Urbančič, T. Developing a Deeper Understanding of Autism: Connecting Knowledge through Literature Mining. Autism Res. Treat. 2011, 2011, 307152. [Google Scholar] [CrossRef]
- Runyan, J.D.; Moore, A.N.; Dash, P.K. A role for prefrontal calcium-sensitive protein phosphatase and kinase activities in working memory. Learn. Mem. 2005, 12, 103–110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lian, Q.; Ladner, C.J.; Magnuson, D.; Lee, J.M. Selective changes of calcineurin (protein phosphatase 2B) activity in Alzheimer’s disease cerebral cortex. Exp. Neurol. 2001, 167, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.; Chattarji, S.; Barbarosie, M.; Rondi-Reig, L.; Philpot, B.D.; Miyakawa, T.; Bear, M.F.; Tonegawa, S. Forebrain-specific calcineurin knockout selectively impairs bidirectional synaptic plasticity and working/episodic-like memory. Cell 2001, 107, 617–629. [Google Scholar] [CrossRef]
- Miyakawa, T.; Leiter, L.M.; Gerber, D.J.; Gainetdinov, R.R.; Sotnikova, T.D.; Zeng, H.; Caron, M.G.; Tonegawa, S. Conditional calcineurin knockout mice exhibit multiple abnormal behaviors related to schizophrenia. Proc. Natl. Acad. Sci. USA 2003, 100, 8987–8992. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mayes, S.D.; Calhoun, S.L.; Mayes, R.D.; Molitoris, S. Autism and ADHD: Overlapping and discriminating symptoms. Res. Autism Spectr. Disord. 2012, 6, 277–285. [Google Scholar] [CrossRef]
- Leitner, Y. The co-occurrence of autism and attention deficit hyperactivity disorder in children—What do we know? Front. Hum. Neurosci. 2014, 8, 268. [Google Scholar] [CrossRef] [PubMed]
- Miranda, A.; Berenguer, C.; Roselló, B.; Baixauli, I.; Colomer, C. Social Cognition in Children with High-Functioning Autism Spectrum Disorder and Attention-Deficit/Hyperactivity Disorder. Associations with Executive Functions. Front. Psychol. 2017, 8, 1035. [Google Scholar] [CrossRef] [PubMed]
- Colombi, C.; Ghaziuddin, M. Neuropsychological Characteristics of Children with Mixed Autism and ADHD. Autism Res. Treat. 2017, 2017, 5781781. [Google Scholar] [CrossRef] [PubMed]
- Costello, R.B.; Elin, R.J.; Rosanoff, A.; Wallace, T.C.; Guerrero-Romero, F.; Hruby, A.; Lutsey, P.L.; Nielsen, F.H.; Rodriguez-Moran, M.; Song, Y.; et al. Perspective: The case for an evidence-based reference interval for serum magnesium: The time has come. Adv. Nutr. 2016, 7, 977–993. [Google Scholar] [CrossRef]
- Whang, R.; Ryder, K.W. Frequency of hypomagnesemia and hypermagnesemia. Requested vs routine. JAMA 1990, 263, 3063–3064. [Google Scholar] [CrossRef] [PubMed]

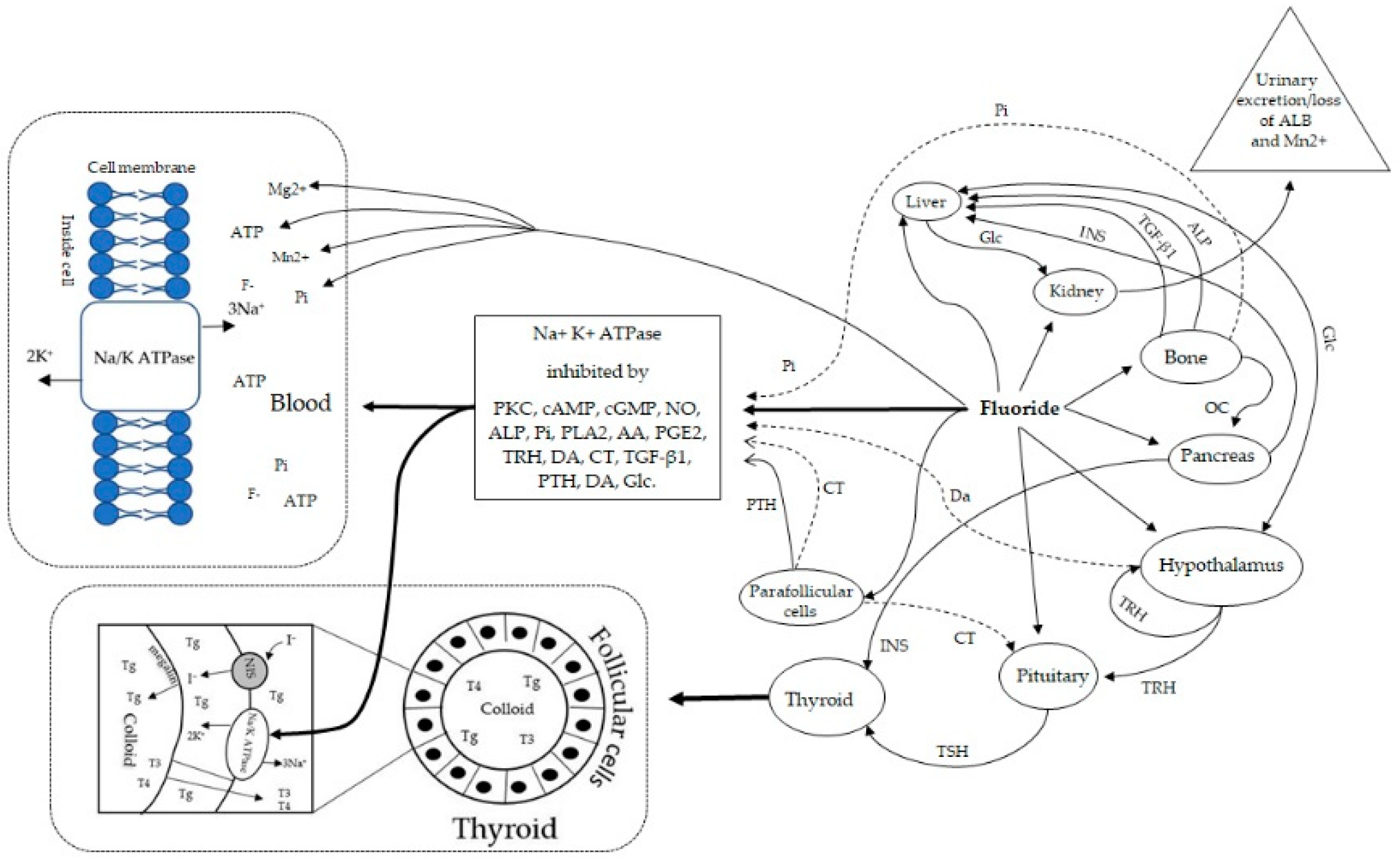
| Factor | Effect of F− | Effect on Na+, K+-ATPase Activity |
|---|---|---|
| ATP | ↓ | ATP is required for NKA homeostasis. Lower bioavailability of ATP leads to inhibition of enzyme activity |
| ENO1 | ↓ | Enolase is necessary for glycolysis and ATP production. Inhibition of enolase leads to loss of NKA activity |
| PKC | ↑ | PKC phosphorylates the α-1 subunit of NKA leading to inhibition of activity. |
| cAMP | ↑ | Inhibits NKA activity by decreasing bioavailability of ATP and enhancing phosphorylation of the α-1 subunit of NKA |
| Cn | ↓ | Regulates the dephosphorylation of NKA. Phosphorylation of NKA inhibits enzyme activity. Hence, inhibition or activation of Cn regulates enzymatic activity. Requires Calmodulin and Manganese for structural stability and full activity. |
| CaM | ↑ | Inhibits Na+, K+-ATPase activity by enhancing phosphorylation |
| Mn2+ | ↓ | Mn2+ is also an activator of Cn and its binding to Cn is required for functional stability and enzyme activity. Loss of Mn2+ inhibits Cn expression and impairs Cn activity leading to enhanced phosphorylation of NKA. Phosphorylation inhibits NKA activity. |
| Mg2+ | ↓ | Mg2+ facilitates the binding of ATP to NKA thereby providing the chemical energy required for enzyme activity. |
| cGMP | ↑ | Inhibits NKA activity |
| NO | ↑ | Inhibits NKA activity |
| Pi | ↑ | Inhibits NKA activity directly as well as inhibiting the phosphatase activity of Cn. |
| RANKL | ↑ | Inhibits NKA indirectly by increasing osteoclast number, bone resorption and Pi release |
| ALP | ↑ | ALP regulates Pi release, thereby indirectly inhibiting NKA activity. ALP activity in turn stimulated by Calcitonin. |
| TGF-β1 | ↑ | Inhibits NKA activity. Calcitonin has been found to be a potent stimulator of TGF-β1 protein synthesis as well as TGF-β1 mRNA expression. |
| CT | ↑ | Inhibits NKA activity indirectly by upregulating TGF-β1 and ALP activity. |
| DA | ↑ | Inhibits NKA activity. PKC and cAMP signalling further contribute to dopaminergic inhibition of NKA. |
| TRH | ↑ | Inhibits NKA activity indirectly by inducing DA release. |
| PTH | ↑ | Inhibits NKA activity directly. PTH also inhibits NKA activity indirectly through activation of PKC, cAMP, PLA2 and PKA dependent pathways. |
| PLA2 | ↑ | Inhibits NKA activity. |
| PGE2 | ↑ | Inhibits NKA activity. |
| BgL | ↑ | Inhibits NKA activity, via activation of PKC, PLA2 and PGE2. |
| AGEs | ↑ | Inhibits NKA activity. |
| TSH | ↑ | TSH induces cAMP production and cAMP inhibits NKA activity by reducing ATP bioavailability and enhancing phosphorylation of the alpha-1 subunit of NKA |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Waugh, D.T. Fluoride Exposure Induces Inhibition of Sodium-and Potassium-Activated Adenosine Triphosphatase (Na+, K+-ATPase) Enzyme Activity: Molecular Mechanisms and Implications for Public Health. Int. J. Environ. Res. Public Health 2019, 16, 1427. https://doi.org/10.3390/ijerph16081427
Waugh DT. Fluoride Exposure Induces Inhibition of Sodium-and Potassium-Activated Adenosine Triphosphatase (Na+, K+-ATPase) Enzyme Activity: Molecular Mechanisms and Implications for Public Health. International Journal of Environmental Research and Public Health. 2019; 16(8):1427. https://doi.org/10.3390/ijerph16081427
Chicago/Turabian StyleWaugh, Declan Timothy. 2019. "Fluoride Exposure Induces Inhibition of Sodium-and Potassium-Activated Adenosine Triphosphatase (Na+, K+-ATPase) Enzyme Activity: Molecular Mechanisms and Implications for Public Health" International Journal of Environmental Research and Public Health 16, no. 8: 1427. https://doi.org/10.3390/ijerph16081427




