Cataloging of Cd Allocation in Late Rice Cultivars Grown in Polluted Gleysol: Implications for Selection of Cultivars with Minimal Risk to Human Health
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Site and Characterization
2.2. Plant Material and Sample Collection
2.3. Soil and Plant Samples Analysis
2.4. Translocation and Bioaccumulation Factor
2.5. Statistical Analysis
3. Results and Discussion
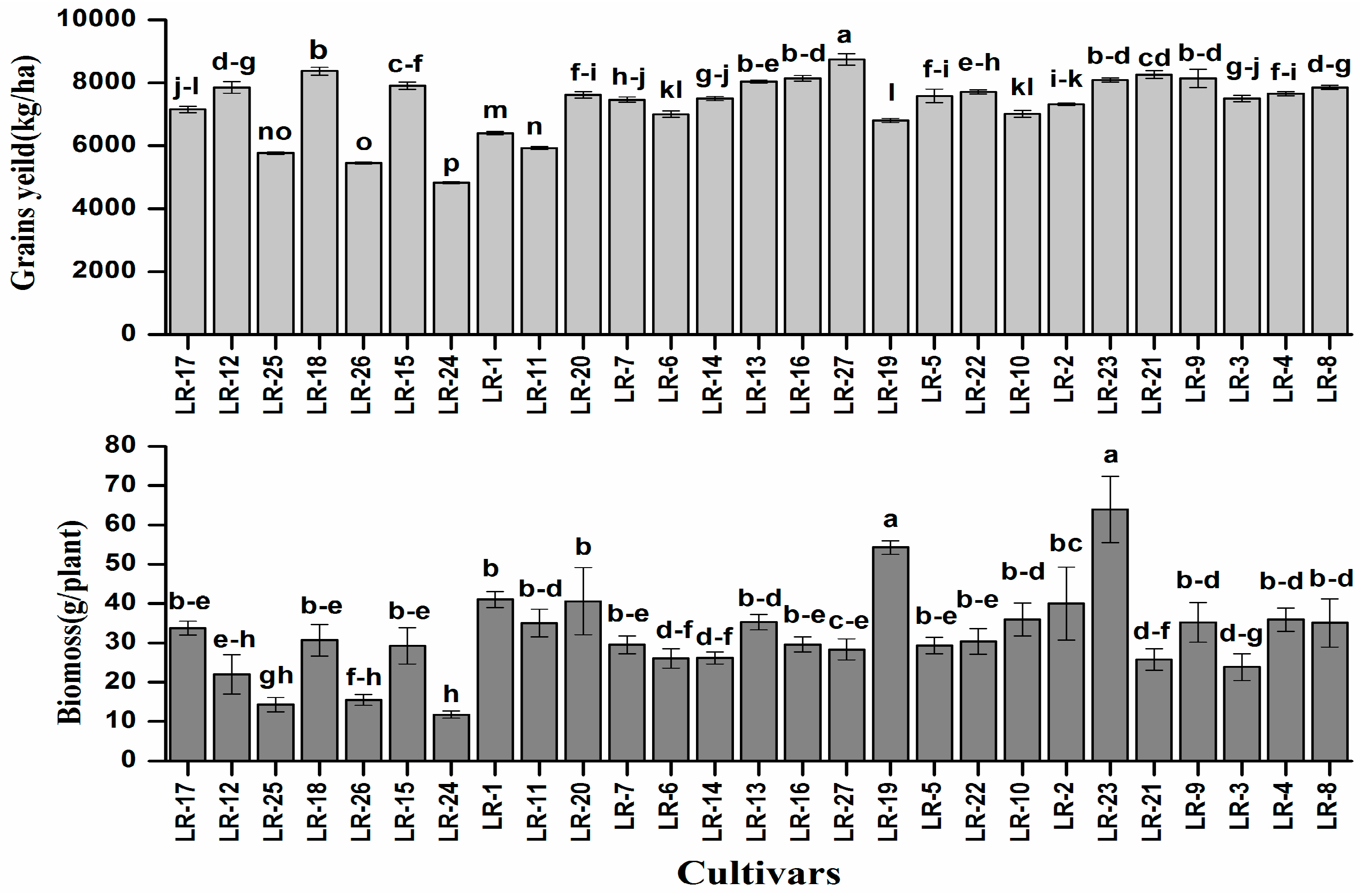
3.1. Plant Biomass and Grains Yield of Rice Cultivars
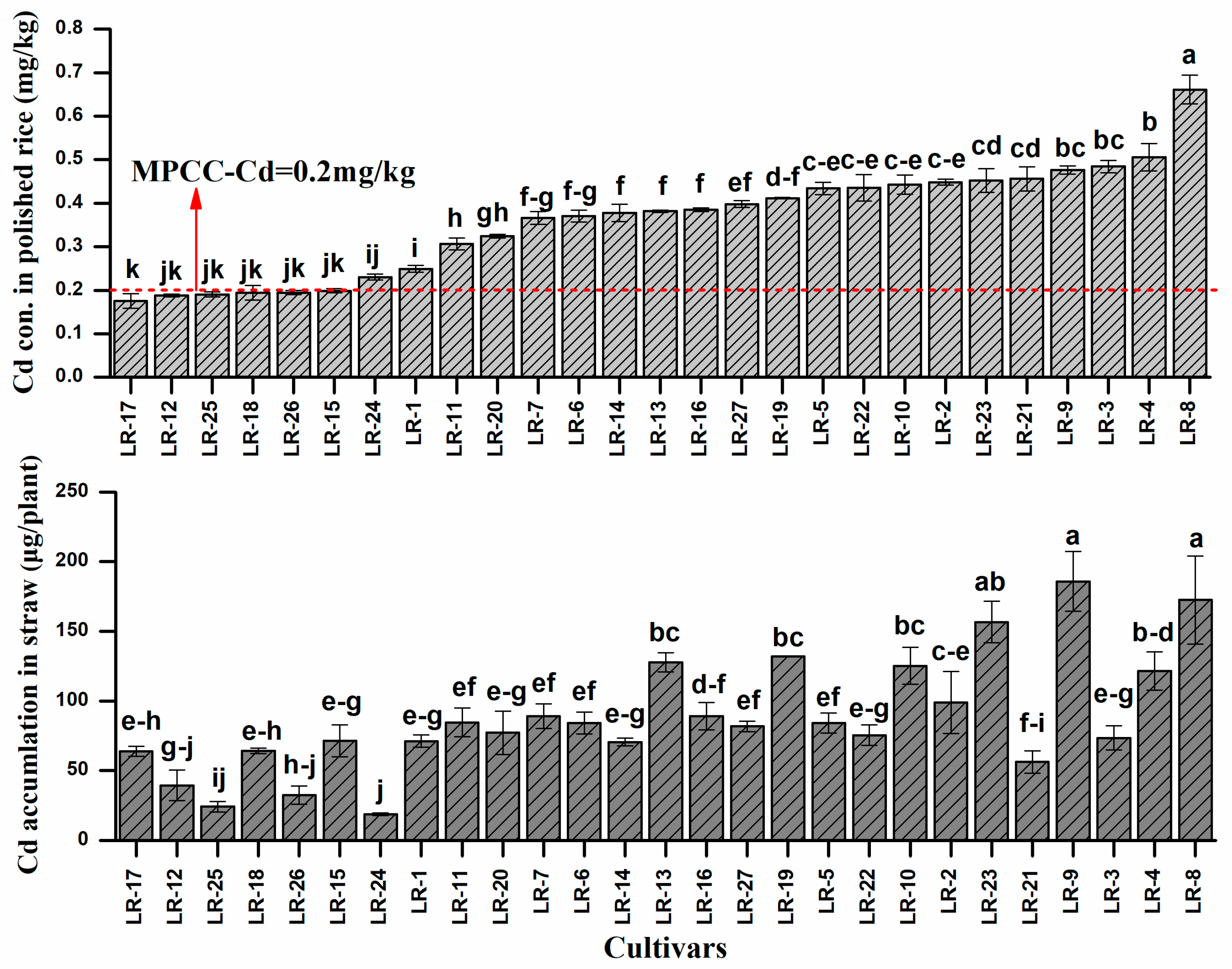
3.2. Cadmium Concentration in Plant Tissues
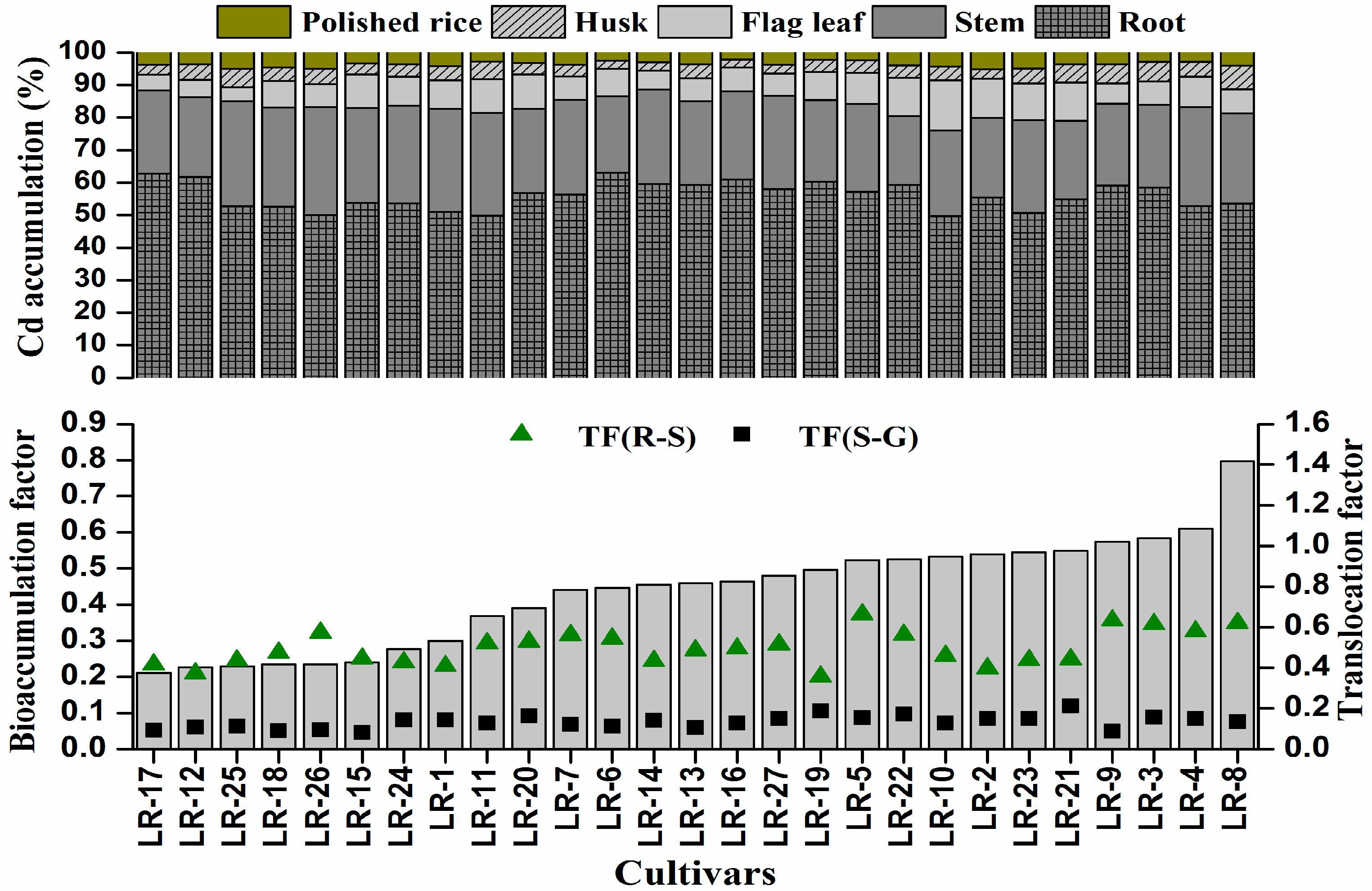
3.3. Variation in Accumulation, Translocation and Bioaccumulation of Cd among the Rice Cultivars
3.4. Correlation between Cd Concentration in Polished Rice and Cd Concentration in Other Parts of Plants
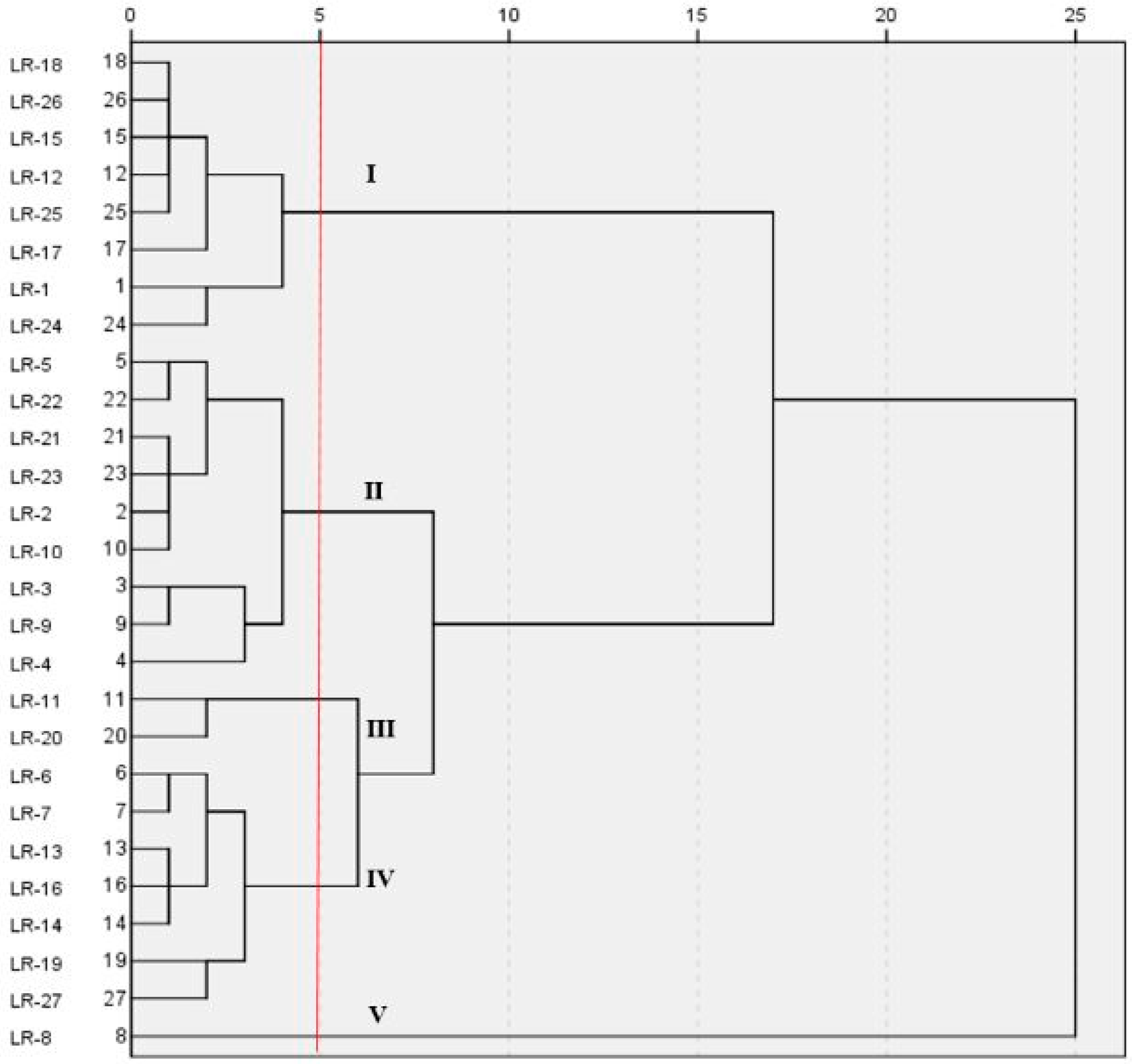
3.5. Genotypic Classification of Late Rice Cultivars Based on Cd Accumulation
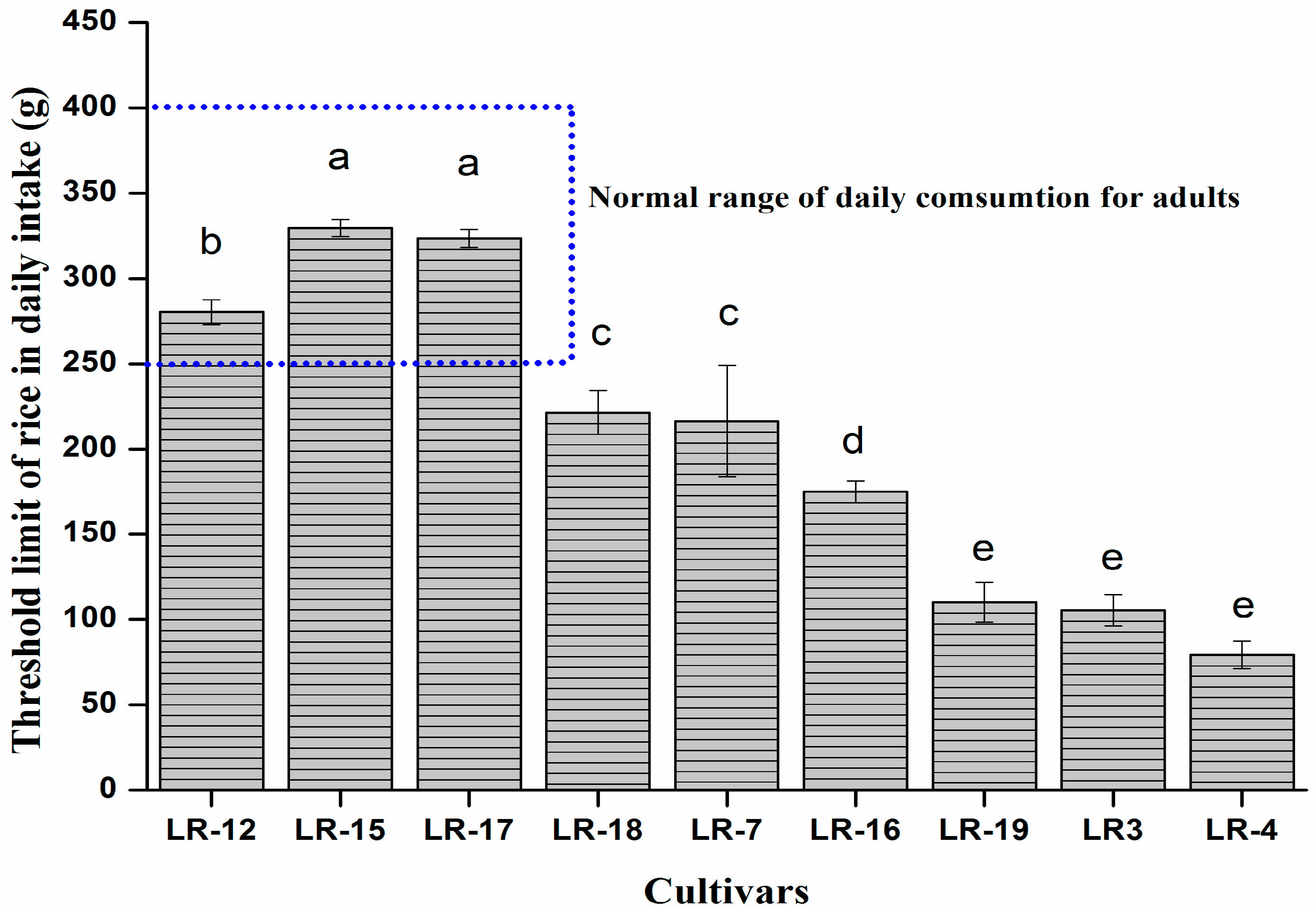
3.6. Selection Criteria for the Safe Cultivar
3.7. Health Risk Assessment Associated with Cd Ingestion
3.8. Phytic Acid, Protein Content and Mineral Nutrient Concentration in Polished Rice
4. Conclusion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Wang, F.Y.; Shi, Z.Y.; Xu, X.F.; Wang, X.G.; Li, Y.J. Contribution of AM inoculation and cattle manure to lead and cadmium phytoremediation by tobacco plants. Environ. Sci. Process. Impacts 2013, 15, 794–801. [Google Scholar] [CrossRef]
- Hamid, Y.; Tang, L.; Muhammad, I.S.; Cao, X.R.; Hussain, B.; Muhammad, Z.A.; Muhammad, U.; He, Z.L.; Yang, X.E. An explanation of soil amendments to reduce cadmium phytoavailability and transfer to food chain. Sci. Total Environ. 2019, 660, 80–96. [Google Scholar] [CrossRef]
- Hussain, B.; Lin, Q.; Hamid, Y.; Sanaullah, M.; Di, L.; Hashmi, M.; Yang, X.E. Foliage application of selenium and silicon nanoparticles alleviates Cd and Pb toxicity in rice (Oryza sativa L.). Sci. Total Environ. 2020, 712, 136497. [Google Scholar] [CrossRef] [PubMed]
- Krystofova, O.; Zitka, O.; Krizkova, S.; Hynek, D.; Shestivska, V.; Adam, V.; Hubalek, J.; Mackova, M.; Macek, T.; Zehnalek, J.; et al. Accumulation of cadmium by transgenic tobacco plants (Nicotiana tabacum L.) carrying yeast metallothione in gene revealed by electrochemistry. Int. J. Electrochem. Sci. 2012, 8, 886–907. [Google Scholar]
- Xie, L.H.; Tang, S.Q.; Wei, X.J.; Shao, G.N.; Jiao, G.A.; Sheng, Z.H. The cadmium and lead content of the grain produced by leading Chinese rice cultivars. Food Chem. 2017, 217, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.R.; Wang, X.Z.; Tong, W.B.; Gurajala, H.M.; Lu, M.; Hamid, Y.; Feng, Y.; He, Z.L.; Yang, X.E. Distribution, availability and translocation of heavy metals in soil oilseed rape (Brassica napus L.) system related to soil properties. Environ. Pollut. 2019, 252, 733–741. [Google Scholar] [CrossRef]
- Vatanpour, N.; Feizy, J.; Talouki, H.H.; Es’haghi, Z.; Scesi, L.; Malvandi, A.M. The high levels of heavy metal accumulation in cultivated rice from the Tajan river basin: Health and ecologic al risk assessment. Chemosphere 2020, 245, 125639. [Google Scholar] [CrossRef]
- Ying, H. The Exploring of Heavy Metal Pollution Source Apportionmentin Various Scale of Agriculture Soils. Ph.D. Thesis, Zhejiang University, Hangzhou, China, 2018; p. 147. [Google Scholar]
- Hamid, Y.; Tang, L.; Muhammad, Y.; Bilal, H.; Afsheen, Z.; Muhammad, Z.A.; He, Z.L.; Yang, X.E. Comparative efficacy of organic and inorganic amendments for cadmium and lead immobilization in contaminated soil under rice wheat cropping system. Chemosphere 2019, 214, 259–268. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Huang, D.; Liu, S.; Luo, Z.; Rao, Z.; Cao, X.; Ren, X. Accumulation and subcellular distribution of cadmium in ramie (Boehmerianivea L. Gaud.) planted one elevated soil cadmium contents. Plant Soil Environ. 2013, 59, 57–61. [Google Scholar]
- Hamid, Y.; Tang, L.; Lu, M.; Hussain, B.; Zehra, A.; Bilal, K.M.; He, Z.L.; Kumar, G.H.; Yang, X.E. Assessing the immobilization efficiency of organic and inorganic amendments for cadmium phytoavailability to wheat. J. Soils Sediments 2019, 19, 3708–3717. [Google Scholar] [CrossRef]
- Sebastian, A.; Prasad, M.N.V. Cadmium minimization in rice. A review. Agron. Sustain. Dev. 2014, 34, 155–173. [Google Scholar] [CrossRef]
- Hamid, Y.; Tang, L.; Wang, X.Z.; Bilal, H.; Muhammad, Y.; Muhammad, Z.A.; Yang, X.E. Immobilization of cadmium and lead in contaminated paddy field using inorganic and organic additives. Sci. Rep. 2018, 8, 17839. [Google Scholar] [CrossRef] [Green Version]
- Hamid, Y.; Tang, L.; Hussain, B.; Usman, M.; Lin, Q.; Rashid, M.S.; He, Z.L.; Yang, X.E. Organic soil additives for the remediation of cadmium contaminated soils and their impact on the soil-plant system: A review. Sci. Total Environ. 2020, 707, 136121. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.L.; Li, Y.E. Variation of cadmium tolerance and accumulation in different tobacco cultivars. J. Southwest Univ. 2007, 3, 110–114. (In Chinese) [Google Scholar]
- Wang, X.; Shi, Y.; Chen, X.; Huang, B. Screening of Cd-safe genotypes of Chinese cabbage in field condition and Cd accumulation in relation to organic acids in two typical genotypes under long-term Cd stress. Environ. Sci. Pollut. Res. 2015, 22, 16590–16599. [Google Scholar] [CrossRef]
- Xu, W.D.; Lu, G.N.; Dang, Z.; Liao, C.J.; Chen, Q.P.; Yi, X.Y. Uptake and distribution of Cd in sweet maize grown on contaminated soils: Afield-scale study. Bioinorg. Chem. Appl. 2013, 2013, 959764. [Google Scholar] [CrossRef] [Green Version]
- Page, V.; Feller, U. Selective transport of zinc, manganese, nickel, cobalt and cadmium in the root system and transfer to the leaves in young wheat plants. Ann. Bot.-Lond. 2005, 96, 425–434. [Google Scholar] [CrossRef] [Green Version]
- Alexander, P.D.; Alloway, B.J.; Dourado, A.M. Genotypic variations in the accumulation of Cd, Cu, Pb and Zn exhibited by six commonly grown vegetables. Environ. Pollut. 2006, 144, 736–745. [Google Scholar] [CrossRef]
- Liu, C.S.; Sun, B.Y.; Kan, S.H.; Zhang, Y.Z.; Deng, S.H.; Yang, G. Copper toxicity and accumulation in potted seedlings of three apple rootstock species: Implications for safe fruit production on copper-polluted soils. J. Plant Nutr. 2010, 34, 1268–1277. [Google Scholar] [CrossRef]
- Tang, L.; Hamid, Y.; Zehra, A.; Sahito, Z.A.; He, Z.L.; Khan, M.B.; Feng, Y.; Yang, X.E. Mechanisms of water regime effects on uptake of cadmium and nitrate by two ecotypes of water spinach (Ipomoea aquatica Forsk.) in contaminated soil. Chemosphere 2020, 246, 125798. [Google Scholar] [CrossRef]
- Arao, T.; Ae, N.; Sugiyama, M.; Takahashi, M. Genotypic differences in cadmium uptake and distribution in soybeans. Plant Soil. 2003, 251, 247–253. [Google Scholar] [CrossRef]
- Williams, P.N.; Lei, M.; Sun, G.; Huang, Q.; Lu, Y.; Deacon, C. Occurrence and partitioning of cadmium, arsenic and lead in mine impacted paddy rice: Hunan, China. Environ. Sci. Technol. 2009, 43, 637–642. [Google Scholar] [CrossRef] [PubMed]
- Analysis of Agricultural Meteorological Conditions of Quhou in 2019. Available online: http://nyncj.qz.gov.cn/art/2019/1/8/art_1594264_33925418.html (accessed on 19 May 2020).
- Li, H.S. Principle and Technology of Plant Physiological and Biochemical Experiment; Higher Education Press: Beijing, China, 2000. (In Chinese) [Google Scholar]
- Bao, S.D. Soil Agricultural Chemistry Analysis Method, 3rd ed.; China Agriculture Press: Beijing, China, 2008. (In Chinese) [Google Scholar]
- Dai, F.; Wang, J.; Zhang, S.; Xu, Z.; Zhang, G. Genotypic and environmental variation in phytic acid content and its relation to protein content and malt quality in barley. Food Chem. 2007, 105, 606–611. [Google Scholar] [CrossRef]
- Bai, Y.C.; Zuo, W.G.; Zhao, H.T.; Mei, L.J.; Gu, C.H.; Guan, Y.X.; Wang, X.K.; Gu, M.J.; Zang, C.Y.; Shan, Y.H. Distribution of heavy metals in maize and mudflat saline soil amended by sewage sludge. J. Soils Sediments 2017, 17, 1565–1578. [Google Scholar] [CrossRef]
- Wodle, T.B.; Weldemariam, S.G.; Tian, S.K. Maize cultivars relieve health risks of Cd-Polluted Soils: In vitro Cd bioaccessibility and bioavailability. Sci. Total Environ. 2020, 703, 134852. [Google Scholar]
- Reynolds, M.; Foulkes, J.; Furbank, R.; Griffiths, S.; King, J.; Murchie, E.; Parry, M.; Slafer, G. Achieving yield gains in wheat. Plant Cell Environ. 2012, 35, 1799–1823. [Google Scholar] [CrossRef]
- Abacar, J.D.; Zhao, M.L.; Xin, C.Z.; Cheng, Q.D.; She, T.; Zheng, H.L.; Shao, H.W.; Yan, F.D. Variation in yield and physicochemical quality traits among mutants of Japonica rice cultivar Wuyujing 3. Rice Sci. 2016, 23, 33–41. [Google Scholar] [CrossRef] [Green Version]
- Koutroubas, S.D.; Mazzini, F.; Pons, B.; Ntanos, D.A. Grain quality variation and relationships with morpho-physiological traits in rice (Oryza sativa L.) genetic resources in Europe. Field Crops Res. 2004, 86, 115–130. [Google Scholar] [CrossRef]
- Lu, M.; Cao, X.R.; Pan, J.Q.; Li, T.Q.; Khan, M.B.; Gurajala, H.K.; He, Z.L.; Yang, X.E. Identification of wheat (Triticum aestivum L.) genotypes for food safety on two different cadmium contaminated soils. Environ. Sci. Pollut. Res. 2019, 27, 7943–7956. [Google Scholar] [CrossRef]
- Ye, X.; Ma, Y.; Sun, B. Influence of soil type and genotype on Cd bioavailability and uptake by rice and implications for food safety. J. Environ. Sci. 2012, 24, 1647–1654. [Google Scholar] [CrossRef]
- Yan, Y.F.; Choi, D.H.; Kim, D.S.; Lee, B.W. Genotypic variation of cadmium accumulation and distribution in rice. J. Crop Sci. Biotechnol. 2010, 13, 69–73. [Google Scholar] [CrossRef]
- Davies., B.E.; Alloway, B.J. Heavy Metals in Soils; Blackie Academic: London, UK, 1995; pp. 206–223. [Google Scholar]
- Stoltz, E.; Greger, M. Accumulation properties of As, Cd, Cu, Pb and Zn by four wetland plant species growing on submerged mine tailings. Environ. Exp. Bot. 2002, 47, 271–280. [Google Scholar] [CrossRef]
- Zeng, F.R.; Mao, Y.; Cheng, W.D.; Wu, F.B.; Zhang, G.P. Genotypic and environmental variation in chromium, cadmium and lead concentrations in rice. Environ. Pollut. 2008, 153, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Liang, L.; Zhang, X.; Zhou, Q. Cultivar variations in cadmium and lead accumulation and distribution among 30 wheat (Triticum aestivum L.) cultivars. Environ. Sci. Pollut. Res. 2015, 22, 8432–8441. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.; Ye, Z.H.; Wong, M.H. Accumulation of lead, zinc, copper and cadmium by 12 wetland plant species thriving in metal-contaminated sites in China. Environ. Pollut. 2004, 132, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.X.; Ma, Z.L.; Ye, Z.H.; Guo, X.Y.; Qiu, R.L. Heavy metal (Pb, Zn) uptake and chemical changes in rhizosphere soils of four wetland plants with different radial oxygen loss. J. Environ. Sci. 2010, 22, 696–702. [Google Scholar] [CrossRef]
- Wu, C.Y.; Feng, Y.; Shohag, M.J.I.; Lu, L.L.; Wei, Y.Y.; Gao, C.; Yang, X.E. Characterization of 68 Zn uptake, translocation, and accumulation into developing grains and young leaves of high Zn-density rice genotype. J. Zhejiang Univ. Sci. B (Biomed. Biotechnol.) 2011, 12, 408–418. [Google Scholar] [CrossRef] [Green Version]
- Tanaka, K.; Fujimaki, S.; Fujiwara, T.; Yoneyama, T.; Hayashi, H. Quantitative estimation of the contribution of the phloem in cadmium transport to grains in rice plants (Oryza sativa L.). Soil Sci. Plant Nutr. 2007, 53, 72–77. [Google Scholar] [CrossRef] [Green Version]
- Yoon, J.K.; Cao, X.D.; Zhou, Q.X.; Ma, L.Q. Accumulation of Pb, Cu, and Zn in native plants growing on a contaminated Florida site. Sci. Total Environ. 2006, 368, 456–464. [Google Scholar] [CrossRef]
- Zhang, H.Z.; Guo, Q.J.; Yang, J.X.; Chen, T.B.; Chen, H.; Shen, J.X.; Zhu, G.X.; Wang, X.; Sun, Y.C. Cadmium and mineral nutrients accumulation in various genotypes of Castor under cadmium stress. Ecol. Environ. Sci. 2015, 24, 323–328. [Google Scholar]
- Liu, Y.; Zhang, C.; Zhao, Y.; Sun, S.; Liu, Z. Effects of growing seasons and genotypes on the accumulation of cadmium and mineral nutrients in rice grown in cadmium contaminated soil. Sci. Total Environ. 2017, 579, 1282–1288. [Google Scholar] [CrossRef] [PubMed]
- Nan, Z.R.; Zhao, C.Y.; Li, J.J.; Chen, F.H.; Sun, W. Relations between soil properties and selected heavy metal concentrations in spring wheat (Triticum a estivum L.) grown in contaminated soils. Water Air Soil Pollut. 2002, 133, 205–213. [Google Scholar] [CrossRef]
- Dala-Paula, B.M.; Custodio, F.B.; Knupp, E.A.N.; Palmieri, H.E.L.; Silva, J.B.B.; Gloria, M.B.A. Cadmium, copper and lead levels in different cultivars of lettuce and soil from urban agriculture. Environ. Pollut. 2018, 242, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Kang, Y.; Pan, W.; Zeng, L.; Zhang, Q.; Luo, J. Concentration and transportation of heavy metals in vegetables and risk assessment of human exposure to bioaccessible heavy metals in soil near a waste-incinerator site, South China. Sci. Total Environ. 2015, 521, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Zhai, F.Y.; He, Y.N.; Ma, G.S.; Li, Y.; Wang, Z.; Hu, Y.; Zhao, Y.; Cui, Y.; Li, Y.; Yang, Y. Study on the current status and trend of food consumption among Chinese population. Zhong Hua Liu Xing Bing Xue Zazhi 2015, 26, 485–488. [Google Scholar]
- Zheng, N.; Wang, X.Q.; Zhang, D.; Zheng, Z.; Zhang, S.Z. Population health risk due to dietary intake of heavy metals in the industrial area of Hulu dao city, China. Sci. Total Environ. 2007, 387, 96–104. [Google Scholar] [CrossRef]
- JECFA. Proceedings of the Joint FAO/WHO Expert Committee on Food Additives, Seventy-Third Meeting, Geneva, Switzerland, 8–17 June 2010; p. 2.
- Sarwar, N.; Saifullah, M.S.S.; Zia, M.H.; Naeem, A.; Bibi, S.; Farid, G. Role of mineral nutrition in minimizing cadmium accumulation by plants. J. Sci. Food Agric. 2010, 90, 925–937. [Google Scholar] [CrossRef]
- Shahid, M.; Shukla, A.K.; Bhattacharyya, P.; Tripathi, R.; Mohanty, S.; Kumar, A.; LaI, B.; Gautam, P.; Raja, R.; Panda, B.B.; et al. Micronutrients (Fe, Mn, Zn and Cu) balance under long-term application of fertilizer and manure in a tropical rice-rice system. J. Soils Sediments 2016, 16, 737–747. [Google Scholar] [CrossRef]
- Pinto, E.; Ferreira, I.M. Cation transporters/channels in plants: Tools for nutrient biofortification. J. Plant Physiol. 2015, 179, 64–82. [Google Scholar] [CrossRef] [Green Version]
- Egli, I.; Davidsson, L.; Zeder, C.; Walczyk, T.; Hurrell, R. Dephytinization of a complementary food based on wheat and soy increases zinc, but not copper, apparent absorption in adults. J. Nutr. 2014, 134, 1077–1081. [Google Scholar] [CrossRef]
- Hurrell, R.F.; Reddy, M.B.; Juillerat, M.A.; Cook, J.D. Degradation of phytic acid in cereal porridges improves iron absorption by human subjects. Am. J. Clin. Nutr. 2003, 77, 1213–1219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stmopen. Phytic acid content in different parts of grain and its correlation with rice quality traits. Adv. J. Food Sci. Technol. 2013, 2, 328–334. [Google Scholar]


| Properties | Values |
|---|---|
| Soil type | Red earth |
| pH | 5.83 ± 0.04 |
| Organic matter (%) | 7.25 ± 0.03 |
| Total N (g kg−1) | 2.10 ± 0.03 |
| Available P (mg kg−1) | 53.9 ± 4.69 |
| Available K (mg kg−1) | 120 ± 9.8 |
| Total Cd (mg kg−1) | 0.85 ± 0.06 |
| Available Cd (mg kg−1) | 0.34 ± 0.02 |
| Type | Cultivars | Cd Conc. (mg kg−1) | Grains Yield (kg Mu−1) | Cu (mg kg−1) | Fe (mg kg−1) | Mn (mg kg−1) | Zn (mg kg−1) | Protein % | Phytic Acid |
|---|---|---|---|---|---|---|---|---|---|
| LOW | LR-12 | 0.20 ± 0.00 d | 486.36 ± 0.94 b | 2.32 ± 0.08 e | 12.5 ± 1.71 d | 16.03 ± 0.22 ef | 18.73 ± 0.23 cd | 9.42 ± 0.07 c | 2.89 ± 0.06 b |
| LR-15 | 0.17 ± 0.00 d | 558.67 ± 1.11 a | 2.93 ± 0.06 bc | 14.29 ± 0.24 d | 26.24 ± 1.52 bc | 23.67 ± 0.42 b | 10.17 ± 0.08 b | 1.50 ± 0.33 cd | |
| LR-17 | 0.18 ± 0.00 d | 395.02 ± 1.04 g | 3.21 ± 0.11 a | 22.65 ± 0.99 a | 34.18 ± 1.71 a | 26.86 ± 1.03 a | 8.42 ± 0.25 d | 1.64 ± 0.17 cd | |
| MID | LR-18 | 0.26 ± 0.01 cd | 449.63 ± 6.29 c | 2.63 ± 0.04 d | 12.39 ± 0.35 d | 15.24 ± 0.08 ef | 16.76 ± 0.02 de | 8.70 ± 0.03 d | 1.31 ± 0.11 d |
| LR-7 | 0.28 ± 0.03 cd | 436.14 ± 0.86 d | 2.27 ± 0.03 e | 17.60 ± 0.42 c | 18.61 ± 0.21 def | 16.34 ± 0.15 e | 8.79 ± 0.08 d | 1.26 ± 0.25 d | |
| LR-16 | 0.33 ± 0.01 c | 413.69 ± 1.79 f | 2.28 ± 0.04 e | 12.63 ± 1.72 d | 13.33 ± 0.18 f | 17.41 ± 0.41 cde | 8.02 ± 0.14 e | 1.44 ± 0.06 cd | |
| HIGH | LR-19 | 0.54 ± 0.05 b | 409.92 ± 0.09 f | 2.73 ± 0.06 cd | 18.58 ± 0.94 bc | 29.80 ± 5.15 ab | 19.14 ± 1.4 c | 10.01 ± 0.07 b | 3.53 ± 0.13 a |
| LR3 | 0.56 ± 0.05 b | 426.84 ± 0.11 e | 3.06 ± 0.11 ab | 20.98 ± 1.29 ab | 24.44 ± 0.03 bcd | 17.91 ± 0.08 cde | 10.66 ± 0.03 a | 3.62 ± 0.11 a | |
| LR-4 | 0.74 ± 0.06 a | 492.56 ± 0.85 b | 2.82 ± 0.05 cd | 22.70 ± 0.87 a | 21.26 ± 1.43 cde | 17.39 ± 0.7 cde | 7.75 ± 0.15 e | 1.94 ± 0.06 c |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, Q.; Tong, W.; Hussain, B.; Hamid, Y.; Lu, M.; He, Z.; Yang, X. Cataloging of Cd Allocation in Late Rice Cultivars Grown in Polluted Gleysol: Implications for Selection of Cultivars with Minimal Risk to Human Health. Int. J. Environ. Res. Public Health 2020, 17, 3632. https://doi.org/10.3390/ijerph17103632
Lin Q, Tong W, Hussain B, Hamid Y, Lu M, He Z, Yang X. Cataloging of Cd Allocation in Late Rice Cultivars Grown in Polluted Gleysol: Implications for Selection of Cultivars with Minimal Risk to Human Health. International Journal of Environmental Research and Public Health. 2020; 17(10):3632. https://doi.org/10.3390/ijerph17103632
Chicago/Turabian StyleLin, Qiang, Wenbin Tong, Bilal Hussain, Yasir Hamid, Min Lu, Zhenli He, and Xiaoe Yang. 2020. "Cataloging of Cd Allocation in Late Rice Cultivars Grown in Polluted Gleysol: Implications for Selection of Cultivars with Minimal Risk to Human Health" International Journal of Environmental Research and Public Health 17, no. 10: 3632. https://doi.org/10.3390/ijerph17103632
APA StyleLin, Q., Tong, W., Hussain, B., Hamid, Y., Lu, M., He, Z., & Yang, X. (2020). Cataloging of Cd Allocation in Late Rice Cultivars Grown in Polluted Gleysol: Implications for Selection of Cultivars with Minimal Risk to Human Health. International Journal of Environmental Research and Public Health, 17(10), 3632. https://doi.org/10.3390/ijerph17103632







