Effect of Polyols and Selected Dental Materials on the Ability to Create a Cariogenic Biofilm–On Children Caries-Associated Streptococcus Mutans Isolates
Abstract
:1. Introduction
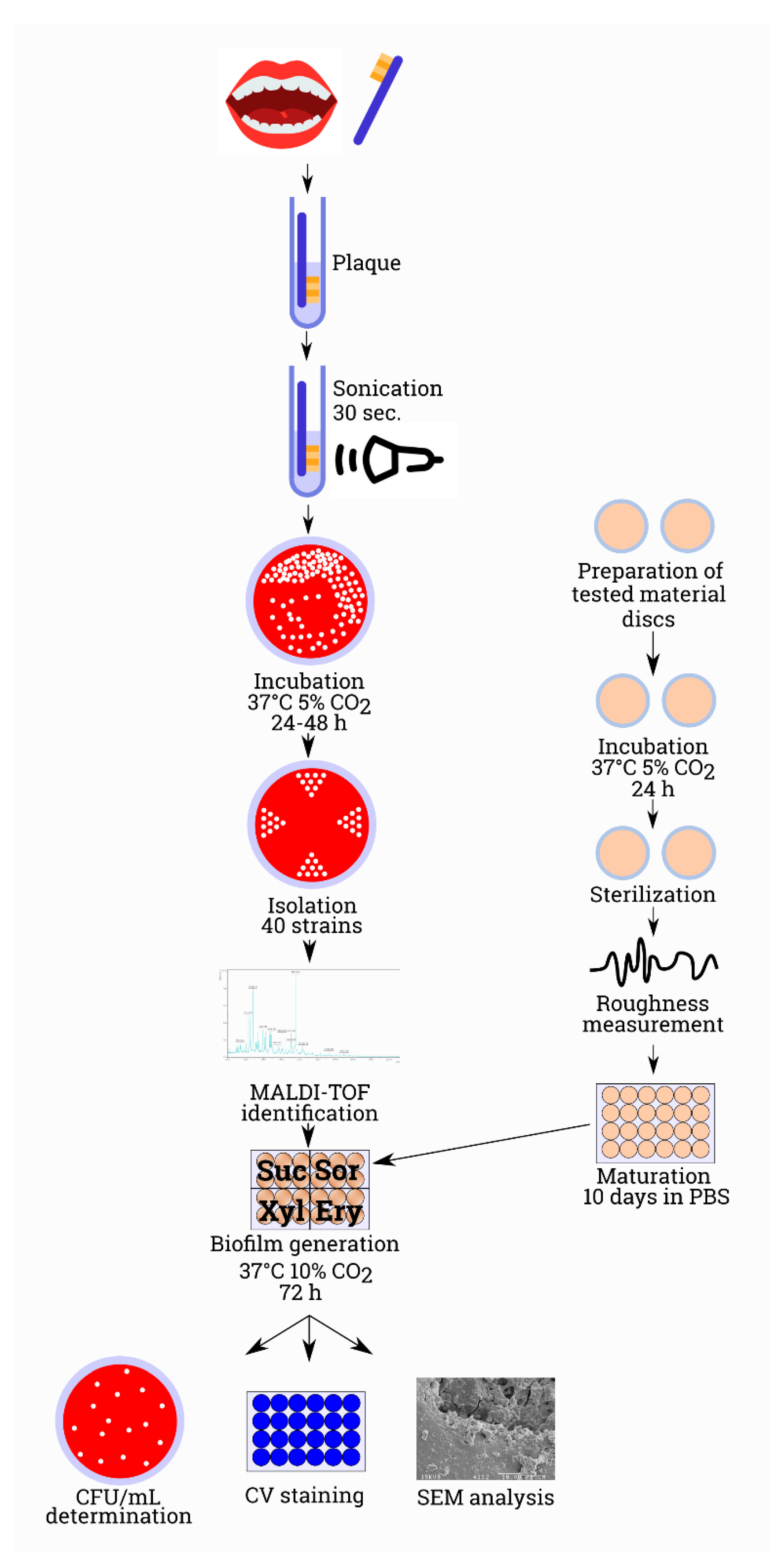
2. Materials and Methods
2.1. Ethical Aspects
2.2. Participants of the Study
2.3. Dental Plaque Collection
2.4. Laboratory Analyses and Proceeding with Dental Materials
2.5. Microbiological Analysis
2.6. In Vitro Biofilm Formation
2.7. In Vitro Biofilm Model
2.8. Statistical Analysis
3. Results
3.1. Microbiological Identification
3.2. Surface of Tested Materials
3.3. Biofilm Biomass Assessment
3.4. Measurement of Total Biomass Expressed as OD (Optical Density)
3.5. Determination of the Amount of Live Microorganisms in a Biofilm
3.6. Morphological Characteristics of Biofilms
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A

References
- Chen, K.J.; Gao, S.S.; Duangthip, D.; Lo, E.C.M.; Chu, C.H. Prevalence of early childhood caries among 5-year-old children: A systematic review. J. Investig. Clin. Dent. 2019, 10, e12376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eskandarizadeh, A.; Mohammadzadeh, I.; Shahravan, A.; Bavafa, M.; Kakooei, S.; Torabi, M. Prevention of secondary caries by a new antibacterial compound. Dent. Res. J. (Isfahan) 2020, 17, 40–47. [Google Scholar]
- de Ávila, N.M.; Bottezini, P.A.; Nicoloso, G.F.; de Araujo, F.B.; Ardenghi, T.M.; Lenzi, T.L.; Casagrande, L. Prevalence of defective restorations and factors associated with re-intervention in primary teeth: A retrospective university-based study. Int. J. Paediatr. Dent. 2019, 29, 566–572. [Google Scholar] [CrossRef] [PubMed]
- Nedeljkovic, I.; De Munck, J.; Vanloy, A.; Declerck, D.; Lambrechts, P.; Peumans, M.; Teughels, W.; Van Meerbeek, B.; Van Landuyt, K.L. Secondary caries: Prevalence, characteristics, and approach. Clin. Oral Investig. 2019, 24, 683–691. [Google Scholar] [CrossRef]
- Petersen, R.C.; Reddy, M.S.; Liu, P.-R. Fiber-Reinforced Composites: A Breakthrough in Practical Clinical Applications with Advanced Wear Resistance for Dental Materials. EC Dent. Sci. 2018, 17, 430–459. [Google Scholar]
- Hutchison, C.; Cave, V. 10 year comparison of glass ionomer and composite resin restoration materials in class 1 and 2 cavities. Evid. Based. Dent. 2019, 20, 113–114. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, Y.; Hu, Y.; Huang, F.; Xiao, Y. Quaternary ammonium compounds in dental restorative materials. Dent. Mater. J. 2018, 37, 183–191. [Google Scholar] [CrossRef] [Green Version]
- Cazzaniga, G.; Ottobelli, M.; Ionescu, A.C.; Paolone, G.; Gherlone, E.; Ferracane, J.L.; Brambilla, E. In vitro biofilm formation on resin-based composites after different finishing and polishing procedures. J. Dent. 2017, 67, 43–52. [Google Scholar] [CrossRef]
- Paglia, L. The sweet danger of added sugars. Eur. J. Paediatr. Dent. 2019, 20, 89. [Google Scholar]
- Von Philipsborn, P.; Stratil, J.M.; Burns, J.; Busert, L.K.; Pfadenhauer, L.M.; Polus, S.; Holzapfel, C.; Hauner, H.; Rehfuess, E. Environmental interventions to reduce the consumption of sugar-sweetened beverages and their effects on health. Cochrane Database Syst. Rev. 2019, 6, CD012292. [Google Scholar] [CrossRef]
- Singh, J.; Rasane, P.; Kaur, S.; Kumar, V.; Dhawan, K.; Mahato, D.K.; Malhotra, S.; Sarma, C.; Kaur, D.; Bhattacharya, J. Nutritional Interventions and Considerations for the development of low calorie or sugar free foods. Curr. Diabetes Rev. 2019. [Google Scholar] [CrossRef]
- De Cock, P.; Mäkinen, K.; Honkala, E.; Saag, M.; Kennepohl, E.; Eapen, A. Erythritol Is More Effective Than Xylitol and Sorbitol in Managing Oral Health Endpoints. Int. J. Dent. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Fitch, C.; Keim, K.S. Position of the Academy of Nutrition and Dietetics: Use of Nutritive and Nonnutritive Sweeteners. J. Acad. Nutr. Diet. 2012, 112, 739–758. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Carder, H.M.; Wendlandt, A.E. Synthesis of rare sugar isomers through site-selective epimerization. Nature 2020, 578, 403–408. [Google Scholar] [CrossRef]
- Jin, X.; Yin, B.; Xia, Q.; Fang, T.; Shen, J.; Kuang, L.; Yang, C. Catalytic Transfer Hydrogenation of Biomass-Derived Substrates to Value-Added Chemicals on Dual-Function Catalysts: Opportunities and Challenges. ChemSusChem 2019, 12, 71–92. [Google Scholar] [CrossRef]
- Zhan, L. Rebalancing the Caries Microbiome Dysbiosis: Targeted Treatment and Sugar Alcohols. Adv. Dent. Res. 2018, 29, 110–116. [Google Scholar] [CrossRef] [Green Version]
- Colombo, S.; Gallus, S.; Beretta, M.; Lugo, A.; Scaglioni, S.; Colombo, P.; Paglia, M.; Gatto, R.; Marzo, G.; Caruso, S.; et al. Prevalence and determinants of early childhood caries in Italy. Eur. J. Paediatr. Dent. 2019, 20, 267–273. [Google Scholar]
- Shmueli, A.; Moskovitz, M.; Halperson, E.; Ram, D.; Fux-Noy, A. Epidemiology and Clinical Management of Early Childhood Caries in Israel. Front. Public Heal. 2019, 7, 280. [Google Scholar] [CrossRef]
- Amalia, R.; Chairunisa, F.; Alfian, M.F.; Supartinah, A. Indonesia: Epidemiological Profiles of Early Childhood Caries. Front. Public Heal. 2019, 7, 210. [Google Scholar] [CrossRef] [Green Version]
- Martignon, S.; Usuga-Vacca, M.; Cortés, F.; Cortes, A.; Gamboa, L.F.; Jacome-Lievano, S.; Ruiz-Carrizosa, J.A.; González-Carrera, M.C.; Restrepo-Perez, L.F.; Ramos, N. Risk factors for early childhood caries experience expressed by ICDAS criteria in Anapoima, Colombia: A cross-sectional study. Acta Odontol. Latinoam. 2018, 31, 58–66. [Google Scholar]
- O’Connell, L.M.; Santos, R.; Springer, G.; Burne, R.A.; Nascimento, M.M.; Richards, V.P. Site-Specific Profiling of the Dental Mycobiome Reveals Strong Taxonomic Shifts during Progression of Early-Childhood Caries. Appl. Environ. Microbiol. 2020, 86. [Google Scholar] [CrossRef] [PubMed]
- Tseveenjav, B.; Furuholm, J.; Mulic, A.; Valen, H.; Maisala, T.; Turunen, S.; Varsio, S.; Auero, M.; Tjäderhane, L. Survival of extensive restorations in primary molars: 15-year practice-based study. Int. J. Paediatr. Dent. 2018, 28, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Mendes da Silva, C.; Figueiredo, M.C.; Casagrande, L.; Larissa Lenzi, T. Survival and Associated Risk Factors of Atraumatic Restorative Treatment Restorations in Children with Early Childhood Caries. J. Dent. Child. (Chic) 2020, 87, 12–17. [Google Scholar]
- Yengopal, V.; Harnekar, S.Y.; Patel, N.; Siegfried, N. WITHDRAWN: Dental fillings for the treatment of caries in the primary dentition. Cochrane Database Syst. Rev. 2016, 10, CD004483. [Google Scholar]
- Askar, H.; Brouwer, F.; Lehmensiek, M.; Paris, S.; Schwendicke, F. The association between loading of restorations and secondary caries lesions is moderated by the restoration material elasticity. J. Dent. 2017, 58, 74–79. [Google Scholar] [CrossRef]
- Hetrodt, F.; Lausch, J.; Meyer-Lueckel, H.; Conrads, G.; Apel, C. Evaluation of restorative materials containing preventive additives in a secondary caries model in vitro. Caries Res. 2019, 53, 447–456. [Google Scholar] [CrossRef]
- Kuper, N.K.; Montagner, A.F.; van de Sande, F.; Bronkhorst, E.M.; Opdam, N.J.; Huysmans, M.C. Secondary caries development in in situ gaps next to composite and amalgam. Caries Res. 2015, 49, 557–563. [Google Scholar] [CrossRef]
- Askar, H.; Tu, Y.K.; Paris, S.; Yeh, Y.C.; Schwendicke, F. Risk of caries adjacent to different restoration materials: Systematic review of in situ studies. J. Dent. 2017, 56, 1–10. [Google Scholar] [CrossRef]
- Askar, H.; Krois, J.; Göstemeyer, G.; Bottenberg, P.; Zero, D.; Banerjee, A.; Schwendicke, F. Secondary caries: What is it, and how it can be controlled, detected, and managed? Clin. Oral Investig. 2020, 24, 1869–1876. [Google Scholar] [CrossRef]
- Mäkinen, K.K.; Isotupa, K.P.; Kivilompolo, T.; Mäkinen, P.-L.; Murtomaa, S.; Petäjä, J.; Toivanen, J.; Söderling, E. The effect of polyol-combinant saliva stimulants on S. mutans levels in plaque and saliva of patients with mental retardation. Spec. Care Dent. 2002, 22, 187–193. [Google Scholar] [CrossRef] [Green Version]
- Kuper, N.K.; Opdam, N.J.; Bronkhorst, E.M.; Huysmans, M.C. The influence of approximal restoration extension on the development of secondary caries. J. Dent. 2012, 40, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Balkaya, H.; Arslan, S.; Pala, K. A randomized, prospective clinical study evaluating effectiveness of a bulk-fill composite resin, a conventional composite resin and a reinforced glass ionomer in Class II cavities: One-year results. J. Appl. Oral Sci. 2019, 27, e20180678. [Google Scholar] [CrossRef] [PubMed]
- Cooper, A.M.; O’Malley, L.A.; Elison, S.N.; Armstrong, R.; Burnside, G.; Adair, P.; Dugdill, L.; Pine, C. Primary school-based behavioural interventions for preventing caries. Cochrane Database Syst. Rev. 2013, 5, CD009378. [Google Scholar] [CrossRef] [PubMed]
- Dikmen, B. Icdas II Criteria (International Caries Detection and Assessment System). J. Istanbul Univ. Fac. Dent. 2015, 49, 63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krzysciak, W.; Papiez, M.; Jurczak, A.; Koscielniak, D.; Vyhouskaya, P.; Zagórska-Swiezy, K.; Skalniak, A. Relationship between pyruvate kinase activity and cariogenic biofilm formation in streptococcus mutans biotypes in caries patients. Front. Microbiol. 2017, 8, 856. [Google Scholar] [CrossRef] [Green Version]
- Kleverlaan, C.J.; Van Duinen, R.N.B.; Feilzer, A.J. Mechanical properties of glass ionomer cements affected by curing methods. Dent. Mater. 2004, 20, 45–50. [Google Scholar] [CrossRef]
- R Development Core Team 3.0.1. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013; Available online: https://www.R-project.org (accessed on 22 May 2020).
- Hughes, C.V.; Dahlan, M.; Papadopolou, E.; Loo, C.Y.; Pradhan, N.S.; Lu, S.C.; Mathney, J.M.J.; Bravoco, A.; Kent, R.L.; Tanner, A.C.R. Aciduric microbiota and mutans streptococci in severe and recurrent severe early childhood caries. Pediatr. Dent. 2012, 34, e16–e23. [Google Scholar]
- Tanner, A.C.R.; Kent, R.L.; Holgerson, P.L.; Hughes, C.V.; Loo, C.Y.; Kanasi, E.; Chalmers, N.I.; Johansson, I. Microbiota of Severe Early Childhood Caries before and after Therapy. J. Dent. Res. 2011, 90, 1298–1305. [Google Scholar] [CrossRef] [Green Version]
- Tanner, A.C.R.; Sonis, A.L.; Lif Holgerson, P.; Starr, J.R.; Nunez, Y.; Kressirer, C.A.; Paster, B.J.; Johansson, I. White-spot Lesions and Gingivitis Microbiotas in Orthodontic Patients. J. Dent. Res. 2012, 91, 853–858. [Google Scholar] [CrossRef] [Green Version]
- Tanner, A.C.R. Anaerobic culture to detect periodontal and caries pathogens. J. Oral Biosci. 2015, 57, 18–26. [Google Scholar] [CrossRef] [Green Version]
- Tanner, A.C.R.; Kressirer, C.A.; Faller, L.L. Understanding Caries from the Oral Microbiome Perspective. J. Calif. Dent. Assoc. 2016, 44, 437–446. [Google Scholar] [PubMed]
- Loesche, W.J. Role of Streptococcus mutans in human dental decay. Microbiol. Rev. 1986, 50, 353–380. [Google Scholar] [CrossRef] [PubMed]
- Kressirer, C.A.; Smith, D.J.; King, W.F.; Dobeck, J.M.; Starr, J.R.; Tanner, A.C.R. Scardovia wiggsiae and its potential role as a caries pathogen. J. Oral Biosci. 2017, 59, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, Y.; Bowen, W.H.; Burne, R.A.; Kuramitsu, H.K. Role of the Streptococcus-mutans gtf genes in caries induction in the specific-pathogen-free rat model. Infect. Immun. 1993, 61, 3811–3817. [Google Scholar] [CrossRef] [Green Version]
- Krzyściak, W.; Pluskwa, K.K.; Pia¸tkowski, J.; Krzyściak, P.; Jurczak, A.; Kościelniak, D.; Skalniak, A. The usefulness of biotyping in the determination of selected pathogenicity determinants in Streptococcus mutans. BMC Microbiol. 2014, 14, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Riley, P.; Moore, D.; Ahmed, F.; Sharif, M.O.; Worthington, H. V Xylitol-containing products for preventing dental caries in children and adults. Cochrane Database Syst. Rev. 2015, 26, CD010743. [Google Scholar] [CrossRef]
- Janakiram, C.; Deepan Kumar, C.V.; Joseph, J. Xylitol in preventing dental caries: A systematic review and meta-analyses. J. Nat. Sci. Biol. Med. 2017, 8, 16–21. [Google Scholar] [CrossRef] [Green Version]
- Burt, B.A. The use of sorbitol- and xylitol-sweetened chewing gum in caries control. J. Am. Dent. Assoc. 2006, 137, 190–196. [Google Scholar] [CrossRef] [Green Version]
- Söderling, E.M.; Hietala-Lenkkeri, A.M. Xylitol and erythritol decrease adherence of polysaccharide-producing oral streptococci. Curr. Microbiol. 2010, 60, 25–29. [Google Scholar] [CrossRef]
- Kościelniak, D.; Gregorczyk-Maga, I.; Jurczak, A.; Staszczyk, M.; Kołodziej, I.; Magacz, M.; Kędziora, K.; Papież, M.; Vyhouskaya, P.; Jamka-Kasprzyk, M.; et al. Low concentration of xylitol improves children tooth protection against Streptococcus mutans biofilm formation. Oral Health Care 2019, 4. [Google Scholar] [CrossRef]
- Radmerikhi, S.; Azul, E.; Fajardo, K.; Formantes, B. Antimicrobial effect of different xylitol concentrations on Streptococcus mutans and Lactobacillus acidophilus count. J. Restor. Dent. 2013, 1, 95. [Google Scholar] [CrossRef]
- Salli, K.M.; Forssten, S.D.; Lahtinen, S.J.; Ouwehand, A.C. Influence of sucrose and xylitol on an early Streptococcus mutans biofilm in a dental simulator. Arch. Oral Biol. 2016, 70, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Soderling, E.; Isokangas, P.; Pienihäkkinen, K.; Tenovuo, J. Influence of Maternal Xylitol Consumption on Acquisition of Mutans Streptococci by Infants. J. Dent. Res. 2000, 79, 882–887. [Google Scholar] [CrossRef] [PubMed]
- Holgerson, P.L.; Sjöström, I.; Stecksén-Blicks, C.; Twetman, S. Dental plaque formation and salivary mutans streptococci in schoolchildren after use of xylitol-containing chewing gum. Int. J. Paediatr. Dent. 2007, 17, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Marghalani, A.A.; Guinto, E.; Phan, M.; Dhar, V.; Tinanoff, N. Effectiveness of Xylitol in Reducing Dental Caries in Children. Pediatr. Dent. 2017, 39, 103–110. [Google Scholar]
- Trahan, L. Xylitol: A review of its action on mutans streptococci and dental plaque--its clinical significance. Int. Dent. J. 1995, 45, 77–92. [Google Scholar]
- Lee, S.-H.; Choi, B.-K.; Kim, Y.-J. The cariogenic characters of xylitol-resistant and xylitol-sensitive Streptococcus mutans in biofilm formation with salivary bacteria. Arch. Oral Biol. 2012, 57, 697–703. [Google Scholar] [CrossRef]
- Takahashi-Abbe, S.; Abbe, K.; Takahashi, N.; Tamazawa, Y.; Yamada, T. Inhibitory effect of sorbitol on sugar metabolism of Streptococcus mutans in vitro and on acid production in dental plaque in vivo. Oral Microbiol. Immunol. 2001, 16, 94–99. [Google Scholar] [CrossRef]
- Mäkinen, K.K.; Saag, M.; Isotupa, K.P.; Olak, J.; Nõmmela, R.; Söderling, E.; Mäkinen, P.L. Similarity of the effects of erythritol and xylitol on some risk factors of dental caries. Caries Res. 2005, 39, 207–215. [Google Scholar] [CrossRef]
- Chen, L.; Suh, B.I.; Yang, J. Antibacterial dental restorative materials: A review. Am. J. Dent. 2018, 31, 6B–12B. [Google Scholar]
- Du, X.; Huang, X.; Huang, C.; Frencken, J.E.; Yang, T. Inhibition of early biofilm formation by glass-ionomer incorporated with chlorhexidine in vivo: A pilot study. Aust. Dent. J. 2012, 57, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.P.; Ge, Y.; Zhou, X.D.; Xu, H.H.K.; Weir, M.D.; Zhang, K.K.; Wang, H.H.; Hannig, M.; Rupf, S.; Li, Q.; et al. Effect of anti-biofilm glass-ionomer cement on Streptococcus mutans biofilms. Int. J. Oral Sci. 2016, 8, 76–83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Makvandi, P.; Gu, J.T.; Zare, E.N.; Ashtari, B.; Moeini, A.; Tay, F.R.; Niu, L. na Polymeric and inorganic nanoscopical antimicrobial fillers in dentistry. Acta Biomater. 2020, 101, 69–101. [Google Scholar] [CrossRef] [PubMed]
- Eick, S.; Glockmann, E.; Brandl, B.; Pfister, W. Adherence of Streptococcus mutans to various restorative materials in a continuous flow system. J. Oral Rehabil. 2004, 31, 278–285. [Google Scholar] [CrossRef]
- Buergers, R.; Rosentritt, M.; Handel, G. Bacterial adhesion of Streptococcus mutans to provisional fixed prosthodontic material. J. Prosthet. Dent. 2007, 98, 461–469. [Google Scholar] [CrossRef]
- Hahnel, S.; Ionescu, A.C.; Cazzaniga, G.; Ottobelli, M.; Brambilla, E. Biofilm formation and release of fluoride from dental restorative materials in relation to their surface properties. J. Dent. 2017, 60, 14–24. [Google Scholar] [CrossRef]
- Amend, S.; Frankenberger, R.; Lücker, S.; Domann, E.; Krämer, N. Secondary caries formation with a two-species biofilm artificial mouth. Dent. Mater. 2018, 34, 786–796. [Google Scholar] [CrossRef]
- Bourbia, M.; Ma, D.; Cvitkovitch, D.G.; Santerre, J.P.; Finer, Y. Cariogenic bacteria degrade dental resin composites and adhesives. J. Dent. Res. 2013, 92, 989–994. [Google Scholar] [CrossRef]
- Li, Y.; Carrera, C.; Chen, R.; Li, J.; Patricia, L.; Joel, D.; Jones, R.S.; Aparicio, C.; Fok, A. Degradation in the dentin-composite interface subjected to multi-species biofilm challenges. Acta Biomater. 2015, 10, 1–20. [Google Scholar] [CrossRef] [Green Version]
- Borges, M.A.P.; Matos, I.C.; Mendes, L.C.; Gomes, A.S.; Miranda, M.S. Degradation of polymeric restorative materials subjected to a high caries challenge. Dent. Mater. 2011, 27, 244–252. [Google Scholar] [CrossRef]
- Ilie, O.; van Loosdrecht, M.C.M.; Picioreanu, C. Mathematical modelling of tooth demineralisation and pH profiles in dental plaque. J. Theor. Biol. 2012, 309, 159–175. [Google Scholar] [CrossRef] [PubMed]
- Banting, D.W. The diagnosis of root caries. J. Dent. Educ. 2001, 65, 991–996. [Google Scholar] [PubMed]
- De Paula, A.B.; De Fúcio, S.B.P.; Alonso, R.C.B.; Ambrosano, G.M.B.; Puppin-Rontani, R.M. Influence of chemical degradation on the surface properties of nano restorative materials. Oper. Dent. 2014, 39, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Wang, S.; Peng, X.; Hu, Y.; Ren, B.; Li, M.; Hao, L.; Feng, M.; Cheng, L.; Zhou, X. Effects of water and microbial-based aging on the performance of three dental restorative materials. J. Mech. Behav. Biomed. Mater. 2018, 80, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, R.P.D.S.; Pereira-Cenci, T.; Silva, W.M.D.; Coelho-De-Souza, F.H.; Demarco, F.F.; Cenci, M.S. Effect of cariogenic biofilm challenge on the surface hardness of direct restorative materials in situ. J. Dent. 2012, 40, 359–363. [Google Scholar] [CrossRef]
- De Paula, A.B.; Fucio, S.B.P.; Ambrosano, G.M.B.; Alonso, R.C.B.; Sardi, J.C.O.; Puppin-Rontan, R.M. Biodegradation and abrasive wear of nano restorative material. Oper. Dent. 2011, 36, 670–677. [Google Scholar] [CrossRef]
- Krämer, N.; Schmidt, M.; Lücker, S.; Domann, E.; Frankenberger, R. Glass ionomer cement inhibits secondary caries in an in vitro biofilm model. Clin. Oral Investig. 2018, 22, 1019–1031. [Google Scholar] [CrossRef]
- Rethman, M.P.; Beltrán-Aguilar, E.D.; Billings, R.J.; Hujoel, P.P.; Katz, B.P.; Milgrom, P.; Sohn, W.; Stamm, J.W.; Watson, G.; Wolff, M.; et al. Nonfluoride caries-preventive agents: Executive summary of evidence-based clinical recommendations. J. Am. Dent. Assoc. 2011, 142, 1065–1071. [Google Scholar] [CrossRef] [Green Version]
- EAPD Guidelines on Prevention of Early Childhood Caries: An EAPD Policy Document. Available online: https://www.eapd.eu/uploads/1722F50D_file.pdf (accessed on 22 May 2020).




| Material | 5% Sucrose | 5% Xylitol | 5% Sorbitol | 5% Erythritol |
|---|---|---|---|---|
| Control | 0.172 (0.030) a | 0.154 (0.012) a | 0.155 (0.010) a | 0.149 (0.011) a |
| Composite | 0.321 (0.031) a | 0.271 (0.033) a | 0.256 (0.023) a | 0.247 (0.014) b |
| Glass ionomer | 1.321 (0.024) a | 1.121 (0.025) a | 0.967 (0.018) a | 0.887 (0.053) b |
| Character | Sucrose | Xylitol | Sorbitol | Erythritol | (ANOVA) p |
|---|---|---|---|---|---|
| Time | OD540 mean ± SD (median ± QD) | ||||
| 12 h | 0.11 ± 0.01 (0.11 ± 0.01) | 0.10 ± 0.01 ** (0.11 ± 0.01) | 0.10 ± 0.01 **** (0.10 ± 0.01) | 0.09 ± 0.01 **** (0.09 ± 0.01) | <0.0001 |
| 24 h | 0.12 ± 0.01 (0.12 ± 0.00) | 0.11 ± 0.02 (0.12 ± 0.01) | 0.10 ± 0.01 *** (0.11 ± 0.01) | 0.08 ± 0.01 **** (0.08 ± 0.01) | <0.0001 |
| 48 h | 0.12 ± 0.01 (0.12 ± 0.01) | 0.11 ± 0.02 (0.12 ± 0.01) | 0.11 ± 0.01 ** (0.11 ± 0.01) | 0.08 ± 0.01 **** (0.08 ± 0.00) | <0.0001 |
| 72 h | 0.14 ± 0.01 (0.14 ± 0.01) | 0.12 ± 0.01 **** (0.13 ± 0.01) | 0.11 ± 0.01 **** (0.12 ± 0.01) | 0.08 ± 0.01 **** (0.08 ± 0.01) | <0.0001 |
| Log(CFU/mL) mean ± SD (median ± QD) | |||||
| 12 h | 1.32 ± 0.05 (1.32 ± 0.05) | 1.12 ± 0.07 *** (1.13 ± 0.06) | 0.92 ± 0.10 *** (0.93 ± 0.05) | 0.85 ± 0.12 *** (0.85 ± 0.08) | <0.0001 |
| 24 h | 1.34 ± 0.04 (1.34 ± 0.02) | 1.22 ± 0.05 *** (1.22 ± 0.03) | 1.14 ± 0.09 *** (1.15 ± 0.08) | 0.86 ± 0.22 *** (0.90 ± 0.12) | <0.0001 |
| 48 h | 1.51 ± 0.16 (1.48 ± 0.09) | 1.26 ± 0.06 *** (1.25 ± 0.03) | 1.24 ± 0.07 *** (1.24 ± 0.04) | 0.84 ± 0.21 *** (0.85 ± 0.08) | <0.0001 |
| 72 h | 1.70 ± 0.07 (1.71 ± 0.03) | 1.36 ± 0.06 *** (1.35 ± 0.04) | 1.31 ± 0.07 *** (1.30 ± 0.04) | 0.84 ± 0.10 *** (0.85 ± 0.06) | <0.0001 |
| Character | Sucrose | Xylitol | Sorbitol | Erythritol | (ANOVA) p |
|---|---|---|---|---|---|
| Time | OD540 mean ± SD (median ± QD) | ||||
| 12 h | 0.08 ± 0.01 (0.08 ± 0.01) | 0.07 ± 0.01 (0.07 ± 0.01) | 0.06 ± 0.01 **** (0.06 ± 0.01) | 0.06 ± 0.01 **** (0.06 ± 0.00) | <0.0001 |
| 24 h | 0.08 ± 0.00 (0.08 ± 0.00) | 0.07 ± 0.01 ** (0.07 ± 0.01) | 0.06 ± 0.01 **** (0.06 ± 0.01) | 0.06 ± 0.01 **** (0.06 ± 0.00) | <0.0001 |
| 48 h | 0.08 ± 0.01 (0.08 ± 0.00) | 0.07 ± 0.01 (0.07 ± 0.01) | 0.06 ± 0.01 **** (0.06 ± 0.01) | 0.06 ± 0.01 **** (0.05 ± 0.01) | <0.0001 |
| 72 h | 0.08 ± 0.00 (0.08 ± 0.00) | 0.08 ± 0.01 (0.08 ± 0.01) | 0.06 ± 0.01 **** (0.06 ± 0.01) | 0.05 ± 0.01 **** (0.05 ± 0.01) | <0.0001 |
| Log(CFU/mL) mean ± SD (median ± QD) | |||||
| 12 h | 1.26 ± 0.09 (1.26 ± 0.05) | 1.31 ± 0.09 (1.31 ± 0.05) | 0.97 ± 0.16 **** (0.95 ± 0.13) | 0.83 ± 0.13 **** (0.84 ± 0.10) | <0.0001 |
| 24 h | 1.27 ± 0.06 (1.26 ± 0.05) | 1.33 ± 0.08 (1.34 ± 0.05) | 1.10 ± 0.15 **** (1.15 ± 0.15) | 0.80 ± 0.11 **** (0.81 ± 0.09) | <0.0001 |
| 48 h | 1.28 ± 0.05 (1.29 ± 0.03) | 1.32 ± 0.07 (1.32 ± 0.05) | 1.17 ± 0.12 **** (1.20 ± 0.10) | 0.77 ± 0.12 **** (0.75 ± 0.09) | <0.0001 |
| 72 h | 1.28 ± 0.05 (1.29 ± 0.03) | 1.35 ± 0.06 ** (1.36 ± 0.05) | 1.15 ± 0.11 **** (1.18 ± 0.08) | 0.74 ± 0.12 **** (0.73 ± 0.09) | <0.0001 |
| Characteristic | Sucrose | Xylitol | Sorbitol | Erythritol | (ANOVA) p |
|---|---|---|---|---|---|
| Time | OD540 mean ± SD (median ± QD) | ||||
| 12 h | 0.12 ± 0.00 (0.12 ± 0.00) | 0.13 ± 0.01 (0.13 ± 0.00) | 0.12 ± 0.00 (0.12 ± 0.00) | 0.10 ± 0.01 **** (0.11 ± 0.01) | <0.0001 |
| 24 h | 0.13 ± 0.01 (0.13 ± 0.00) | 0.14 ± 0.01 **** (0.14 ± 0.01) | 0.13 ± 0.01 (0.13 ± 0.00) | 0.11 ± 0.01 **** (0.11 ± 0.00) | <0.0001 |
| 48 h | 0.14 ± 0.00 (0.14 ± 0.00) | 0.15 ± 0.01 **** (0.15 ± 0.01) | 0.13 ± 0.01 ** (0.13 ± 0.00) | 0.11 ± 0.01 **** (0.11 ± 0.01) | <0.0001 |
| 72 h | 0.15 ± 0.01 (0.15 ± 0.00) | 0.18 ± 0.02 **** (0.17 ± 0.01) | 0.14 ± 0.01 ** (0.14 ± 0.00) | 0.11 ± 0.01 **** (0.11 ± 0.01) | <0.0001 |
| Log(CFU/mL) mean ± SD (median ± QD) | |||||
| 12 h | 1.39 ± 0.14 (1.39 ± 0.07) | 1.49 ± 0.15 ** (1.47 ± 0.06) | 1.31 ± 0.12 (1.31 ± 0.08) | 1.33 ± 0.12 (1.33 ± 0.07) | <0.0001 |
| 24 h | 1.43 ± 0.09 (1.43 ± 0.05) | 1.64 ± 0.19 **** (1.57 ± 0.07) | 1.36 ± 0.11 (1.35 ± 0.06) | 1.34 ± 0.12 * (1.35 ± 0.07) | <0.0001 |
| 48 h | 1.61 ± 0.13 (1.64 ± 0.11) | 1.85 ± 0.19 **** (1.85 ± 0.11) | 1.46 ± 0.15 **** (1.44 ± 0.08) | 1.35 ± 0.12 **** (1.36 ± 0.07) | <0.0001 |
| 72 h | 1.71 ± 0.12 (1.73 ± 0.07) | 2.24 ± 0.29 **** (2.23 ± 0.16) | 1.53 ± 0.15 *** (1.51 ± 0.05) | 1.36 ± 0.13 **** (1.38 ± 0.08) | <0.0001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Staszczyk, M.; Jurczak, A.; Magacz, M.; Kościelniak, D.; Gregorczyk-Maga, I.; Jamka-Kasprzyk, M.; Kępisty, M.; Kołodziej, I.; Kukurba-Setkowicz, M.; Krzyściak, W. Effect of Polyols and Selected Dental Materials on the Ability to Create a Cariogenic Biofilm–On Children Caries-Associated Streptococcus Mutans Isolates. Int. J. Environ. Res. Public Health 2020, 17, 3720. https://doi.org/10.3390/ijerph17103720
Staszczyk M, Jurczak A, Magacz M, Kościelniak D, Gregorczyk-Maga I, Jamka-Kasprzyk M, Kępisty M, Kołodziej I, Kukurba-Setkowicz M, Krzyściak W. Effect of Polyols and Selected Dental Materials on the Ability to Create a Cariogenic Biofilm–On Children Caries-Associated Streptococcus Mutans Isolates. International Journal of Environmental Research and Public Health. 2020; 17(10):3720. https://doi.org/10.3390/ijerph17103720
Chicago/Turabian StyleStaszczyk, Małgorzata, Anna Jurczak, Marcin Magacz, Dorota Kościelniak, Iwona Gregorczyk-Maga, Małgorzata Jamka-Kasprzyk, Magdalena Kępisty, Iwona Kołodziej, Magdalena Kukurba-Setkowicz, and Wirginia Krzyściak. 2020. "Effect of Polyols and Selected Dental Materials on the Ability to Create a Cariogenic Biofilm–On Children Caries-Associated Streptococcus Mutans Isolates" International Journal of Environmental Research and Public Health 17, no. 10: 3720. https://doi.org/10.3390/ijerph17103720






