Environmental Impact of Phosphogypsum-Derived Building Materials
Abstract
:1. Introduction
2. Materials and Methods
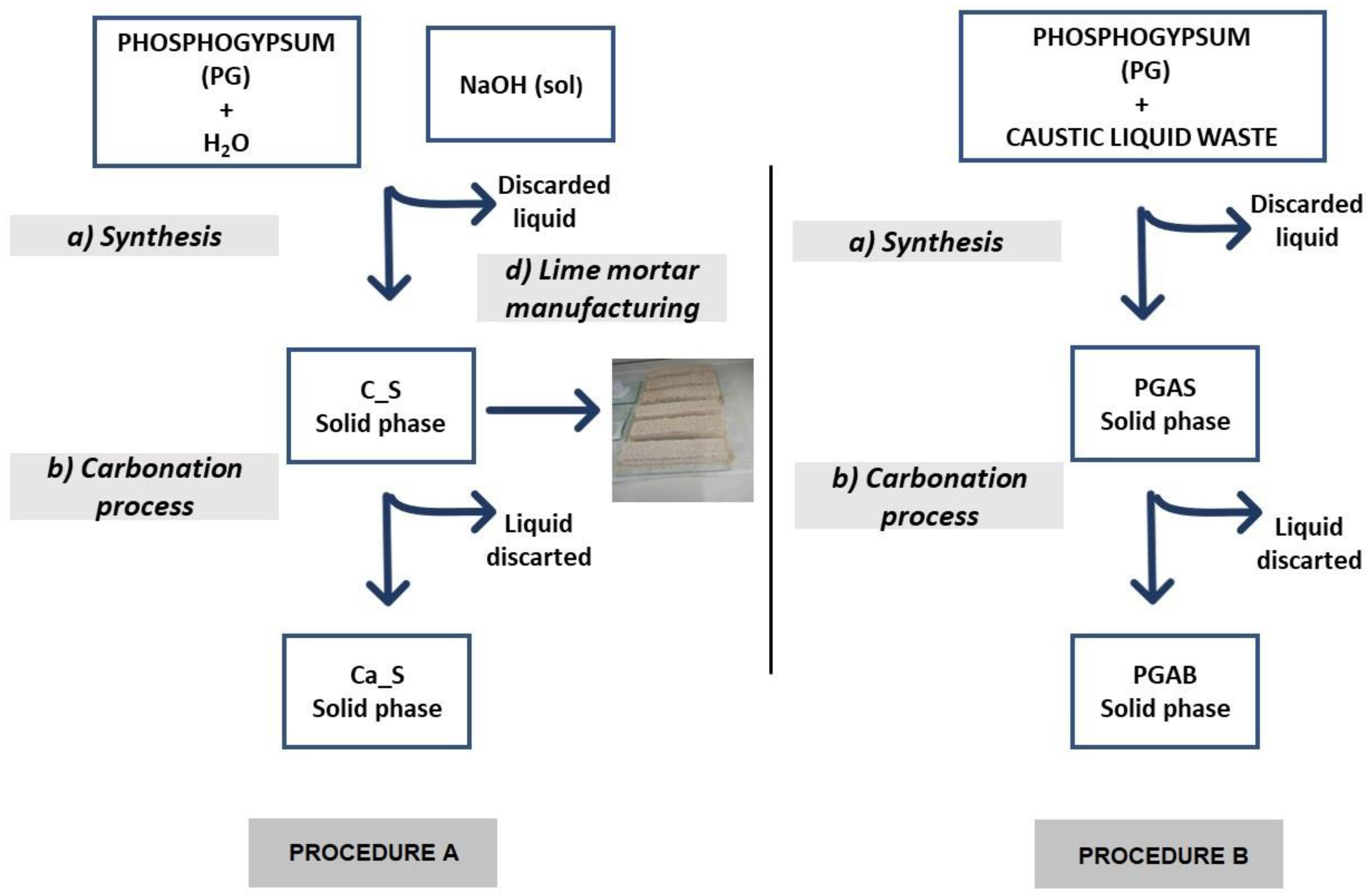
2.1. Procedure A
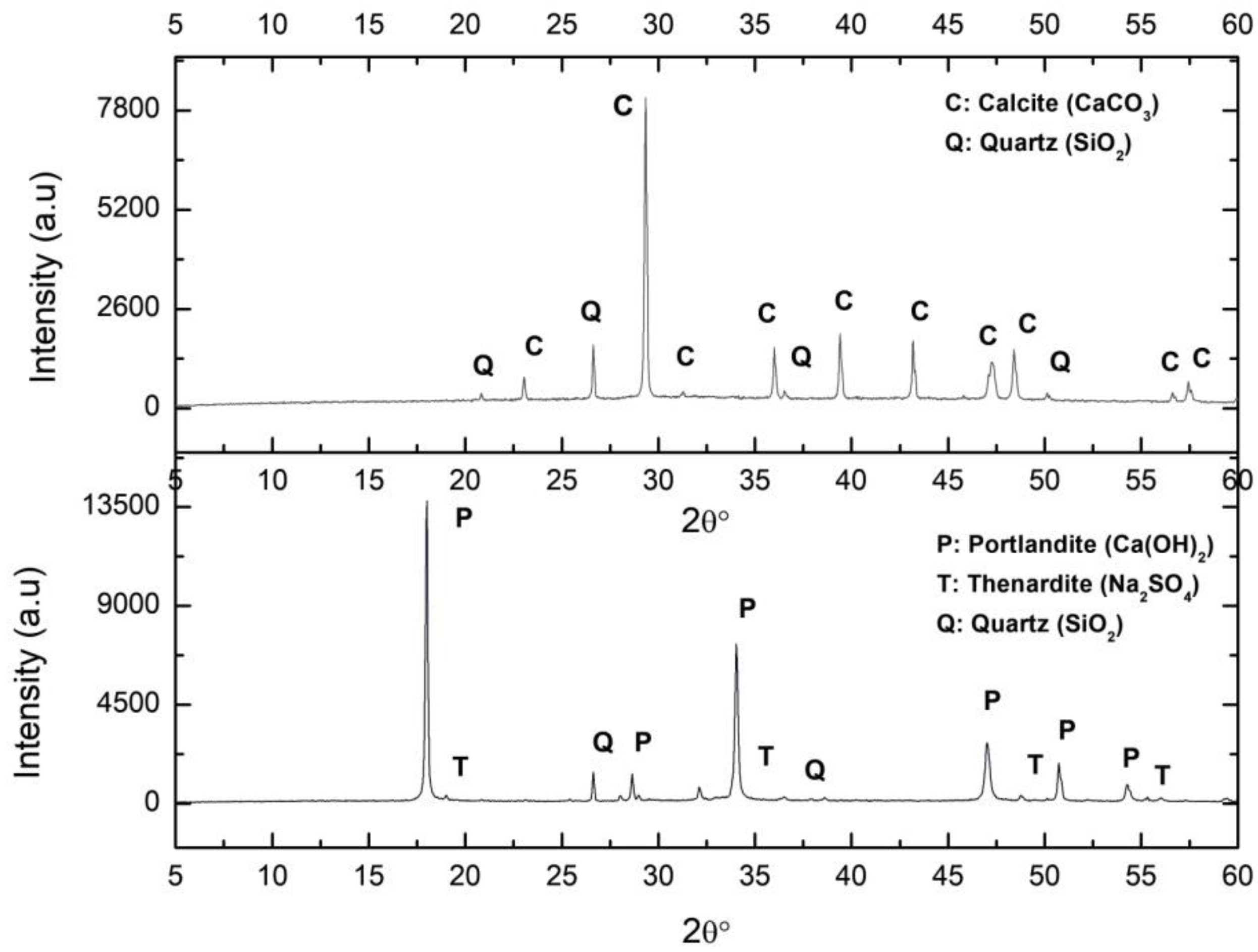
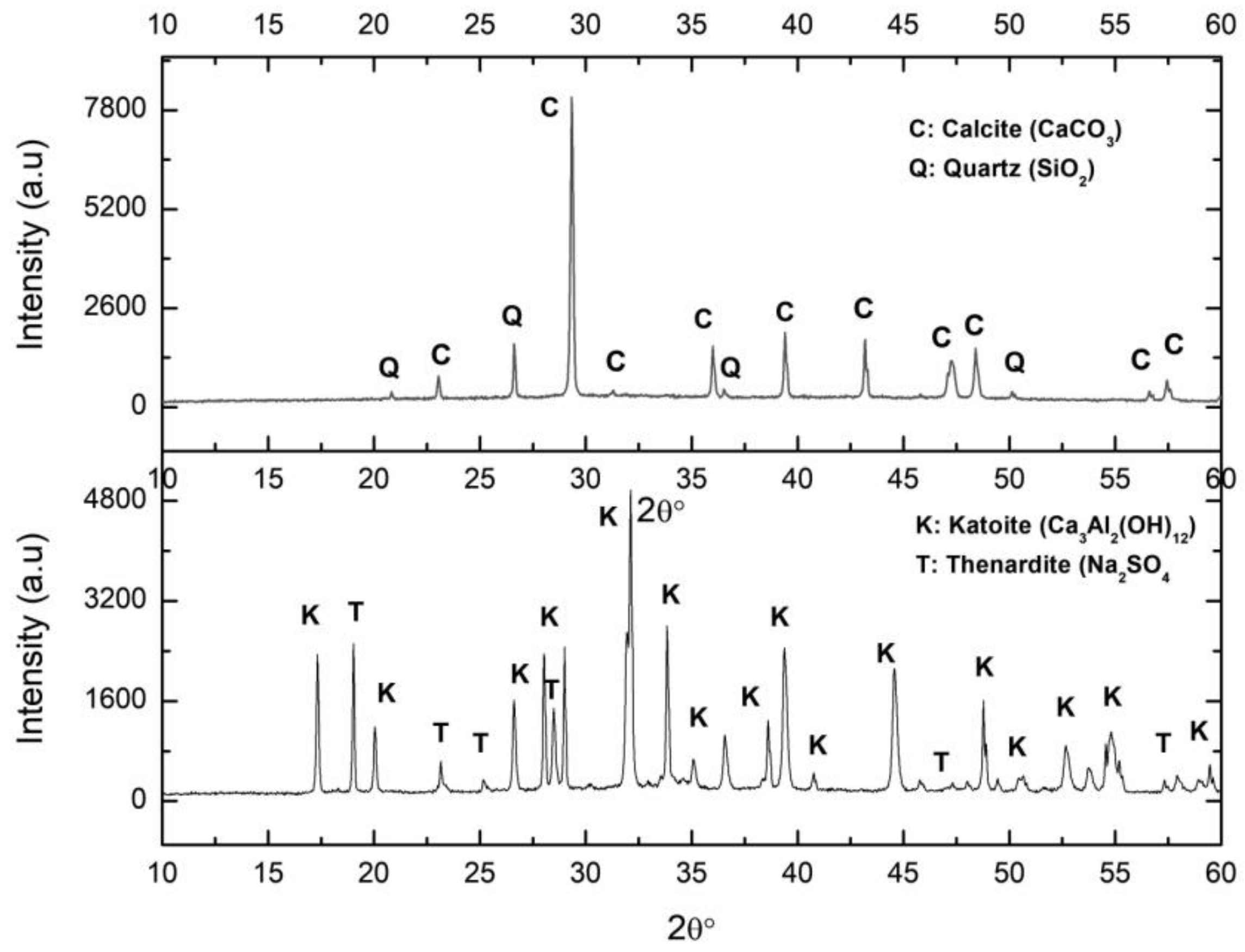
2.1.1. Synthesis
2.1.2. Carbonation Process
2.1.3. Lime Mortar Manufacturing
2.2. Procedure B
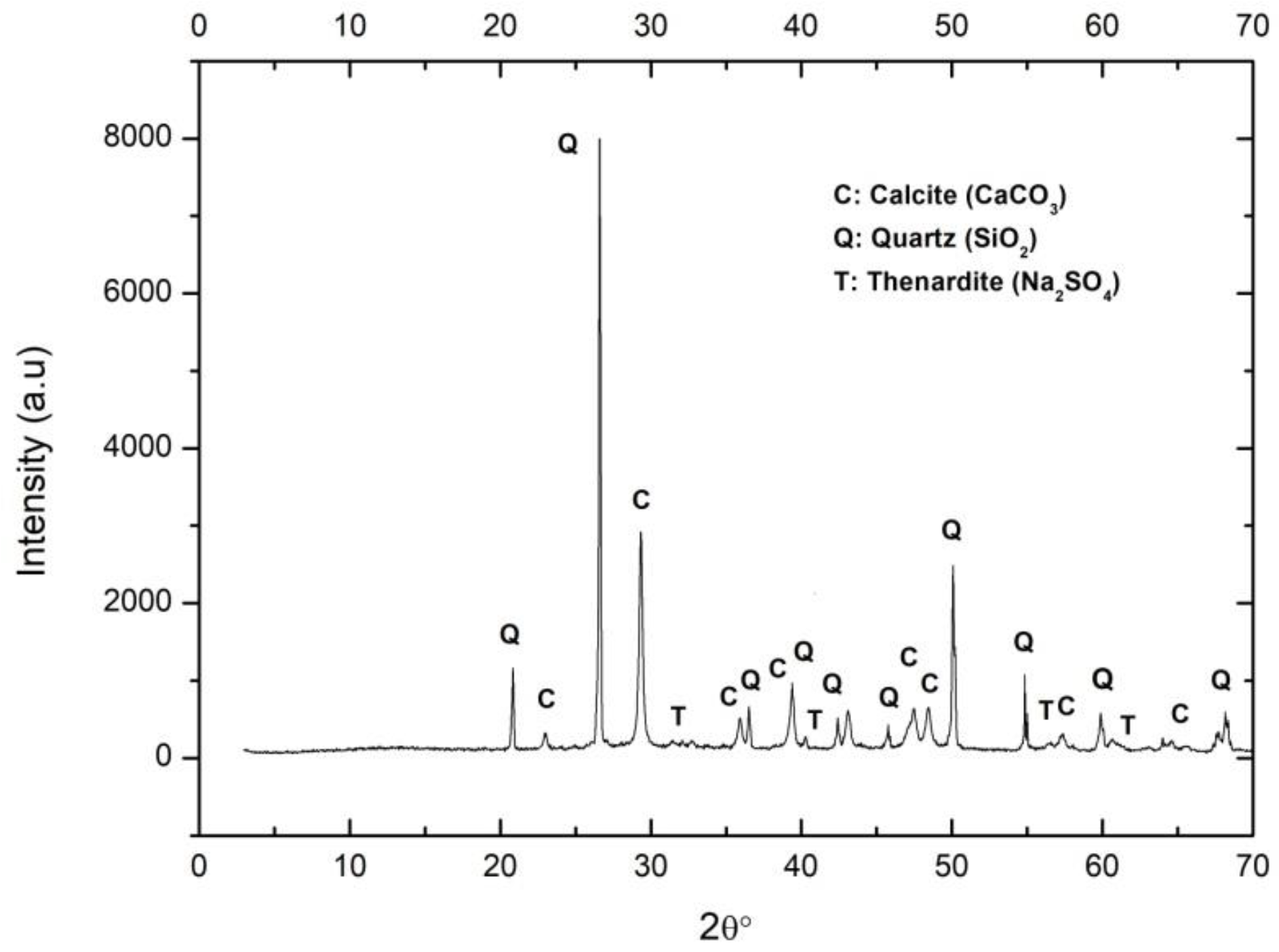
2.2.1. Synthesis
2.2.2. Carbonation Process
2.3. Characterization Techniques
3. Results and Discussion
3.1. Evolution of Environmental Risk
3.1.1. Evaluation of Major Elements and Trace Elements from the Treatment of PG with a Soda in Solution
3.1.2. Contents of Major and Trace Elements in the Treatment of PG with Aluminium Residue
3.2. Radiological Evolution
3.2.1. Procedure A
3.2.2. Procedure B
3.3. Radiological Risk Indexes
4. Conclusions
4.1. PG
4.2. Procedure A
4.3. Procedure B
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Contreras, M.; Pérez-López, R.; Gázquez, M.J.; Morales-Flórez, V.; Santos, A.; Esquivias, L.; Bolívar, J.P. Fractionation and fluxes of metals and radionuclides during the recycling process of phosphogypsum wastes applied to mineral CO2 sequestration. Waste Manag. 2015, 45, 412–419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Villa-Alfageme, M.; Mosqueda, F.; Hurtado, S.; Mantero, J.; Manjón, G.; Periañez, R.; Vaca, F.; García-Tenorio, R. Contamination and restoration of an estuary affected by phosphogypsum releases. Sci. Total Environ. 2009, 408, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Corisco, J.A.G.; Mihalik, J.; Madruga, M.J.; Prudencio, M.I.; Marques, R.; Santos, M.; Reis, M. Natural radionuclides, rare earths and heavy metals transferred to the wild vegetation covering a phosphogypsum stockpile at Barreiro, Portugal. Water Soil Pollut. 2017, 228, 235. [Google Scholar] [CrossRef]
- Macías, F.; Cánovas, C.R.; Cruz-Hernández, P.; Carrero, S.; Asta, M.P.; Nieto, J.M.; Pérez-López, R. An anomalous metal-rich phosphogypsum: Characterization and classification according to international regulations. J. Hazard. Mater. 2017, 331, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Bolívar, J.P.; Martín, J.E.; García-Tenorio, R.; Pérez-Moreno, J.P.; Mas, J.L. Behaviour and fluxes of natural radionuclides in the production process of a phosphoric acid plant. Appl. Radiat. Isot. 2009, 67, 345–356. [Google Scholar] [CrossRef]
- Lieberman, R.N.; Izquierdo, M.; Córdoba, P.; Moreno-Palmerola, N.; Querol, X.; Sánchez de la Campa, A.M.; Font, O.; Cohen, H.; Knop, Y.; Torres-Sánchez, R.; et al. The geochemical evolution of brines from phosphogypsum deposits in Huelva (SW Spain) and its environmental implications. Sci. Total Environ. 2020, 700, 134444. [Google Scholar] [CrossRef]
- Torres-Sánchez, R.; Sánchez-Rodas, D.; Sánchez de la Campa, A.M.; de la Rosa, J.D. Long term geochemical variation of brines derived from a major phosphogypsum pond of SW Europe. J. Environ. Manag. 2020, 254, 109832. [Google Scholar] [CrossRef]
- Esquivias, L.; Morales-Flórez, V.; Santos, A. Carbon dioxide sequestration by phosphogypsum based procedur. In Carbon Dioxide Sequestration in Cementitious Construction Materials; Woodhead Publishing: Amsterdam, The Netherlands, 2018; pp. 199–223. [Google Scholar] [CrossRef]
- Saadaoui, E.; Ghazel, N.; Romdhane, C.B.; Massoudi, N. Phosphogypsum: Potential uses and problems-a review. Int. J. Environ. Stu. 2017, 74, 558–567. [Google Scholar] [CrossRef]
- Tayibi, H.; Gascó, C.; Navarro, N.; López-Delgado, A.; Álvarez, A.; Yagüe, L.; Alguacil, F.J.; López, F.A. Valorisation of phosphogypsum as building material: Radiological aspects. Mat. Const. 2011, 61, 503–515. [Google Scholar] [CrossRef] [Green Version]
- Flores-Alés, V.; Jiménez-Bayarri, V.; Pérez-Fargallo, A. The influence of the incorporation of crushed glass on the properties and high temperature behaviour of cement mortars. Bol. Soc. Esp. Ceram. Vidr. 2018, 57, 257–265. [Google Scholar] [CrossRef]
- Flores-Alés, V.; Alducin-Ochoa, J.M.; Martín-del-Río, J.J.; Torres-González, M.; Jiménez-Bayarri, V. Physical-mechanical behaviour and transformations at high temperature in a cement mortar with waste glass as aggregate. J. Build. Engin. 2020, 101158. [Google Scholar] [CrossRef]
- Liew, K.M.; Sojobi, A.O.; Zhang, L.W. Green concrete: Prospects and challenges. Constr. Build. Mater. 2017, 156, 1063–1095. [Google Scholar] [CrossRef]
- Gijbels, K.; Nguyen, H.; Kinnunen, P.; Schroeyers, W.; Pontikes, Y.; Schreurs, S.; Illikainen, M. Feasibility of incorporating phosphogypsum in ettringite-based binder from ladle slag. J. Clean. Prod. 2019, 237, 117793. [Google Scholar] [CrossRef]
- Ajam, L.; Ben El Haj Hassen, A.; Reguigui, N. Phosphogypsum utilization in fired bricks: Radioactivity assessment and durability. J. Build. Engin. 2019, 26, 100928. [Google Scholar] [CrossRef]
- Cárdenas-Escudero, C.; Morales-Flórez, V.; Pérez-López, R.; Santos, A.; Esquivias, L. Procedure to use phosphogypsum industrial waste for mineral CO2 sequestration. J. Hazard Mater. 2011, 196, 431–435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Msila, X.; Billing, D.; David, G.; Barnard, W. Capture and storage of CO2 into waste phosphogypsum: The modified Merseburg process. Clean. Tech. Environ. Policy 2016, 18, 2709–2715. [Google Scholar] [CrossRef]
- Mattila, H.P.; Zevenhoven, R. Mineral carbonation of phosphogypsum waste for production of useful carbonate and sulfate salts. Fron. Energy Res. 2015, 3, 48. [Google Scholar] [CrossRef] [Green Version]
- Romero-Hermida, I.; Morales-Flórez, V.; Santos, A.; Villena, A.; Esquivias, L. Technological proposals for recycling industrial wastes for environmental applications. Minerals 2014, 4, 746–757. [Google Scholar] [CrossRef] [Green Version]
- Xie, H.P.; Wang, J.L.; Hou, Z.M.; Wang, Y.F.; Liu, T.; Tang, L.; Jiang, W. CO2 sequestration through mineral carbonation of waste phosphogypsum using the technique of membrane electrolysis. Environ. Earth Sci. 2016, 75, 1216. [Google Scholar] [CrossRef]
- Zhao, S.; Ma, L.; Yang, J.; Zheng, D.; Liu, H.; Yang, J. Mechanism of CO2 capture technology based on the phosphogypsum reduction thermal decomposition process. Energy Fuels 2017, 31, 9824–9832. [Google Scholar] [CrossRef]
- Romero-Hermida, I.; Santos, A.; Pérez-López, R.; García-Tenorio, R.; Esquivias, L.; Morales-Flórez, V. New method for carbon dioxide mineralization based on phosphogypsum and aluminium-rich industrial wastes resulting in valuable carbonated by-products. J.CO2 Util. 2017, 18, 15–22. [Google Scholar] [CrossRef] [Green Version]
- Bueno-Marí, R.; Almeida, A.P.G.; Navarro, J.C. Emerging zoonoses: Eco-epidemiology, involved mechanisms, and public health implications. Front. Public Health 2015, 3, 39–49. [Google Scholar]
- US-EPA. 2018. Available online: https://www.epa.gov/radiation/technologically-enhanced-naturally-occurring-radioactive-materials-tenorm (accessed on 19 November 2019).
- Bolívar, J.P.; Pérez-Moreno, J.P.; Mas, J.L.; Martín, J.E.; San Miguel, E.G.; García-Tenorio, R. External radiation assessment in a wet phosphoric acid production plant. Appl. Radiat. Isotopes 2009, 67, 1930–1938. [Google Scholar] [CrossRef] [PubMed]
- Mazzilli, B.; Palmiro, V.; Saueia, C.; Nisti, M.B. Radiochemical characterization of Brazilian phosphogypsum. J. Environ. Radioact. 2000, 49, 113–122. [Google Scholar] [CrossRef]
- Gijbels, K.; Landsberger, S.; Samyn, P.; Iacobescu, R.I.; Pontikes, Y.; Schreurs, S.; Schroeyers, W. Radiological and non-radiological leaching assessment of alkali-activated materials containing ground granulated blast furnace slag and phosphogypsum. Sc. Total Environ. 2019, 660, 1098–1107. [Google Scholar] [CrossRef] [PubMed]
- Pérez-López, R.; Nieto, J.M.; López-Coto, I.; Aguado, J.L.; Bolívar, J.P.; Santisteban, M. Dinamics of contaminants in phosphogypsum of the fertilizer industry of Huelva (SW Spain): From phosphate rock ore to the environment. Appl. Geochem. 2010, 25, 705–715. [Google Scholar] [CrossRef]
- Tayibi, H.; Choura, M.; López, F.A.; Alguacil, F.J.; López-Delgado, A. Environmental impact and management of phosphogypsum. J. Environ. Manag. 2009, 90, 2377–2386. [Google Scholar] [CrossRef] [Green Version]
- Romero-Hermida, M.I.; Borrero-López, A.M.; Alejandre, F.J.; Flores-Alés, V.; Santos, A.; Franco, J.M.; Esquivias, L. Phosphogypsum waste lime as a promising substitute of commercial limes: A rheological approach. Cem. Conc. Comp. 2019, 95, 205–216. [Google Scholar] [CrossRef]
- Intrakamhaeng, V.; Clavier, K.A.; Townsend, T.G. Hazardous waste characterization implications of updating the toxicity characteristic list. J. Hazard. Mater. 2020, 383, 121171. [Google Scholar] [CrossRef]
- UNE-EN 1015-3:2000/A1:2005/A2:2007. Methods of Test for Mortar for Masonry. Part 3: Determination of Consistence of Fresh Mortar; UNE-EN: Madrid, Spain, 2007. [Google Scholar]
- UNE-EN 1015-2:1999/A1:2007. Methods of Test for Mortar for Masonry-Part 2: Bulk Sampling of Mortars and Preparation of Test Mortars; UNE-EN: Madrid, Spain, 2007. [Google Scholar]
- US-EPA. Handbook for Stabilization/Solidification of Hazardous Wastes, EPA 540-2-86-001; Environmental Protection Agency: Washington, DC, USA, 1986. [Google Scholar]
- Rudnick, R.L.; Gao, S. Composition of the continental crust. Crust 2003, 3, 1–64. [Google Scholar]
- Drinking Water Directive. Council Directive 98/83/EC of 3 November 1998 on the Quality of Water Intended for Human Consumption. 1998. Available online: https://ec.europa.eu/environment/water/waterdrink/legislation_en.html (accessed on 19 November 2019).
- Alhers, M.; Kuhlmann, B.; Wiggershaus-Eschert, S.; Zullei-Seibert, N. Daten und Informationen zu Wasser-inhaltsstoffen; DVGW-Schriftenreihe Wasser: Eschborn, Germany, 1993; p. 48. [Google Scholar]
- Martínez-López, C.; Mejía-Arcila, J.M.; Torres-Agredo, J. Mejía-de-Gutiérrez, R. Evaluation of the toxicity characteristics of two industrial wastes valorized by geopolymerization process. DYNA 2015, 190, 74. [Google Scholar] [CrossRef]
- Macías, F.; Pérez-López, R.; Ruiz-Cánovas, C.; Carrero, S.; Cruz-Hernández, P. Environmental assessment and management of phosphogypsum according to european and United States of America regulation. Procedia Earth Planet. Sci. 2017, 17, 666–669. [Google Scholar] [CrossRef] [Green Version]
- Ruiz-Cánovas, C.; Pérez-López, R.; Millán, R.; Nieto, J.M. Preliminary study of Huelva phosphogypsum stacks as a potential source of elements of economic interest. Geogaceta 2017, 62, 103–106. [Google Scholar]
- Suárez, F.M.; Perez-Lopez, R.; Ruiz-Canovas, C. Evaluación Ambiental de los Fosfoyesos de Huelva Según Normativas de Europa y Norteamérica. Mineral. Soc. J. 2015, 20, 85–86. [Google Scholar] [CrossRef]
- UNE-EN 459-2: 1994. Cement and Lime Test Methods. Part 1. Determination of Mechanical Resistance; UNE-EN: Madrid, Spain, 1994. [Google Scholar]
- UNE-EN 196-2: 2014. Cement and Lime Test Methods. Part 2: Chemical Analysis of Cements; UNE-EN: Madrid, Spain, 2014. [Google Scholar]
- Carvalho, F.P. Disposal of phosphogypsum waste containing enhanced levels of radioactivity. In Proceedings of the International Last Conference on Management of Radioactive Waste from non-power applications–Sharing the IAEA-CN-87/6, Saint Paul’s Bay, Malta, 5–9 November 2001; pp. 67–68. [Google Scholar]
- Bolivar, J.P.; García-Tenorio, R.; Garcia-Leon, M. On the fractionation of natural radioactivity in the production of phosphoric acid by the wet acid method. J. Radioanal. Nucl. Chem. 1996, 214, 77–78. [Google Scholar] [CrossRef]
- International Atomic Energy Agency-Annual Report for 2004. Available online: https://www.iaea.org/es/publications/reports/annual-report-2004 (accessed on 10 December 2019).
- US-EPA. Recommended Water Quality Criteria. 2002. Available online: https://www.elaw.org/es/content/us-%E2%80%93-us-epa-national-recommended-water-quality-criteria-2002 (accessed on 10 December 2019).
- US-EPA. Guidelines for Exposure Assessment. 1992. Available online: https://cfpub.epa.gov/ncea/risk/recordisplay.cfm?deid=15263 (accessed on 10 December 2019).
- US-EPA. National Primary Drinking Water Regulations; Radionuclides; Final Rule. Part II. 40 CFR Parts 9, 141, and 142. 2000. Available online: https://nepis.epa.gov/Exe/ZyPURL.cgi?Dockey=2000238H.TXT (accessed on 10 December 2019).
- Royal Decree, 314/2016, de 29 de Julio, por el que se Establecen los Criterios Sanitarios de la Calidad del Agua de Consumo Humano. Official State Gazette (State Agency BOE), nº 183 (2016), Madrid (in Spanish). Available online: https://www.boe.es/eli/es/rd/2016/07/29/314 (accessed on 10 December 2019).
- Al Attar, L.; Al-Oudat, M.; Kanakri, S.; Budeir, Y.; Khalily, H.; Al Hamwi, A. Radiological impacts of phosphogypsum. J. Eenviron. Manag. 2011, 92, 2151–2158. [Google Scholar] [CrossRef]
- Bolívar, J.P.; García-Tenorio, R.; Vaca, F. Radio ecological study of and estuarine system located in the south of Spain. Water Res. 2000, 34, 2941–2950. [Google Scholar] [CrossRef]
- UNSCEAR. United Nations Scientific Committee on the Effects of Atomic Radiation; Sources and Effects of Ionizing Radiation: New York, NY, USA, 1993. [Google Scholar]
- Miró, C.; Ávila, J.M.; García, M.; Pastor-Villegas, S.J. Riesgos debido a la radiactividad natural de pizarras de construcción. Inf. Tecnol. 2010, 21, 9–16. [Google Scholar] [CrossRef]
- EC-European Commission, Radiation Protection. 112-radiological Protection Principles Concerning the Natural Radioactivity of Building Materials, Directorate-General Environment, Nuclear Safety and Civil Protection; EC-European Commission, Radiation Protection: Brussels, Belgium, 1999. [Google Scholar]
- Belivermis, M.; Kilic, N.; Cotuk, Y.; Topcuoglu, S. The effects of physicochemical properties on gamma emitting natural radionuclide levels in the soil profile of Istanbul. Environ. Monit. Assess. 2010, 163, 15–26. [Google Scholar] [CrossRef]
- Beretka, J.; Mathew, P.J. Natural radioactivity of Australian building materials, waste and byproducts. Health Phys. 1985, 48, 87–95. [Google Scholar] [CrossRef] [PubMed]
- El-Taher, A. Gamma spectroscopic analysis and associated radiation hazards of building materials used in Egypt. Radiat. Prot. Dosim. 2009, 138, 166–173. [Google Scholar] [CrossRef] [PubMed]
- Gázquez-González, M.J. Caracterización y Valorización de Residuos Generados en la Industria de Producción de Dióxido de Titanio. Ph.D. Thesis, Universidad de Huelva, Huelva, Spain, 2011. [Google Scholar]
- Krieger, R. Radioactivity of construction materials. Betonw. Fert. Tech. 1981, 47, 468. [Google Scholar]
- International Commission on Radiological Protection. Internacional Commision on Radiological Protection. Recommendations of ICRP; Publication 26; Pergamon Press: Oxford, UK, 1977. [Google Scholar]
| Majority Elements (wt. %) | PG | C_S | PGAS | PGAB |
|---|---|---|---|---|
| Fe2O3 | nd | nd | nd | 0.01 ± 0.01 |
| MnO | nd | nd | nd | nd |
| MgO | nd | nd | nd | nd |
| CaO | 32 ± 1 | 49 ± 2 | 21 ± 1 | 32 ± 2 |
| Na2O | 0.01 ± 0.01 | 12.5 ± 0.6 | 20.5 ± 0.7 | 2.29 ± 0.08 |
| K2O | 0.02 ± 0.01 | nd | 0.02 ± 0.01 | 0.03 ± 0.01 |
| TiO2 | nd | nd | nd | nd |
| P2O5 | 0.65 ± 0.02 | 1.02 ± 0.01 | 0.38 ± 0.01 | 0.47 ± 0.01 |
| SO3 | 46 ± 3 | 13.6 ± 0.2 | 27 ± 2 | 14.4 ± 0.9 |
| Cl | nd | nd | nd | nd |
| F | nd | nd | nd | nd |
| SrO | nd | nd | nd | nd |
| BaO | nd | nd | nd | nd |
| LOI | 18.4 ± 0.4 | 19.4 ± 0.2 | 14.4 ± 0.2 | 33 ± 0.1 |
| Metal | PG (mg/kg) | L_f (mg/L) | Limits Allowed (mg/kg) | TCLP (U S—EPA) | U S—EPA | WHO | EU |
|---|---|---|---|---|---|---|---|
| V | 2.9 ± 0.7 | 0.0161 ± 0.0001 | 97 | 0.05 | |||
| Cr | 6.3 ±0.2 | < 0.023 | 92 | 5.0 | 0.10 | 0.05 | 0.05 |
| Co | <0.6 | <0.0024 | 17.3 | ||||
| Ni | <3 | 0.026 ± 0.001 | 47 | 0.02 | 0.02 | ||
| Zn | <42 | 0.600 ± 0.007 | 67 | 5.0 | 3.0 | ||
| As | <0.6 | 0.0052 ± 0.0004 | 4.8 | 5.0 | 0.05 | 0.01 | 0.01 |
| Se | <30 | <0.006 | 0.09 | 1.0 | |||
| Sr | 360 ± 10 | 1.89 ± 0.03 | 320 | 4.0 | |||
| Cd | 1.8 ± 0.4 | 0.0072 ± 0.0007 | 0.09 | 1.0 | 0.005 | 0.003 | 0.005 |
| Ba | 37 ± 1 | 0.0433 ± 0.0008 | 628 | 100.0 | 2.0 | 0.30 | |
| Pb | 1.8 ± 0.1 | 0.0053 ± 0.0001 | 17 | 5.0 | 0.015 | 0.01 | 0.01 |
| Th | 1.1 ± 0.2 | <0.0013 | 10.5 | ||||
| U | 5 ± 1 | 0.0081 ± 0.0001 | 2.7 |
| METAL | C_S (mg/kg) | L_p (mg/L) | Ca_S (mg/kg) | L_c (mg/L) | Mortar (mg/kg) | L_m (mg/L) |
|---|---|---|---|---|---|---|
| V | <0.1 | <0.007 | <0.1 | <0.007 | 0.3 ± 0.1 | 0.109 ± 0.001 |
| Cr | 13.8 ± 0.2 | 0.083 ± 0.003 | 11.5 ± 0.4 | <0.023 | 9 ± 2 | <0.023 |
| Co | 0.275 ± 0.004 | <0.0024 | 0.23 ± 0.01 | 0.0048 ± 0.0002 | 0.165 ± 0.03 | 0.0049 ± 0.0002 |
| Ni | 1.80 ± 0.02 | 0.023 ± 0.001 | 2.3 ± 0.1 | 0.046 ± 0.001 | 0.347 ± 0.05 | 0.039 ± 0.001 |
| Zn | 10.1 ± 0.2 | 0.67 ± 0.02 | 9.7 ± 0.5 | <0.214 | 9.6 ± 1.2 | <0.221 |
| As | 7 ± 2 | <0.002 | 5.2 ± 0.4 | <0.002 | 3.9 ± 1.1 | <0.002 |
| Se | 2.36 ± 0.08 | <0.0059 | 2.33±0.08 | <0.006 | 1.2 ± 0.7 | <0.0061 |
| Sr | 413 ± 6 | 2.17 ± 0.05 | 353 ± 1 | 3.03 ± 0.04 | 170 ± 20 | 2.31 ± 0.01 |
| Cd | 3.37 ± 0.04 | <0.0008 | 3.6 ± 0.1 | <0.0008 | 1.4 ± 0.4 | <0.0008 |
| Ba | 83 ± 2 | 0.062 ± 0.002 | 72 ± 1 | 0.1203 ± 0.0009 | 36 ± 2 | 0.0658 ± 0.0004 |
| Pb | 3.25 ± 0.05 | 0.0066 ± 0.0003 | 3.5 ± 0.2 | <0.0009 | 1.43 ± 0.04 | <0.0009 |
| Th | 1.5 ± 0.2 | <0.0013 | 2.69 ± 0.08 | <0.0013 | 0.32 ± 0.05 | <0.0013 |
| U | 10.7 ± 0.3 | <0.0012 | 9.7 ± 0.3 | 0.0508 ± 0.0003 | 3.9 ± 0.3 | 0.0302 ± 0.0003 |
| Metal | PGAS (mg/kg) | L_k (mg/L) | PGAB (mg/kg) | L_ck (mg/L) |
|---|---|---|---|---|
| V | 8.0 ± 0.3 | <0.007 | 5.8 ± 0.3 | 0.0225 ± 0.04 |
| Cr | 5.3 ± 0.4 | <0.023 | 6.7 ± 0.6 | 0.030 ± 0.004 |
| Co | <0.6 | <0.0024 | <0.6 | <0.0024 |
| Ni | <3 | 0.0082 ± 0.0003 | <3 | 0.0050 ± 0.0004 |
| Zn | <41 | 0.0740 ± 0.0005 | <41 | <0.212 |
| As | 1.8 ± 0.8 | <0.002 | 1.4 ± 0.2 | <0.002 |
| Se | <30 | <0.006 | <30 | <0.0059 |
| Sr | 248 ± 10 | 1.69 ± 0.02 | 345 ± 5 | 1.46 ± 0.1 |
| Cd | 1.1 ± 0.2 | <0.0008 | 1.6 ± 0.2 | <0.0008 |
| Ba | 27 ± 3 | 0.0344 ± 0.0001 | 36 ± 1 | 0.0024 ± 0.02 |
| Pb | 1.5 ± 0.2 | <0.0009 | 1.6 ± 0.1 | <0.0009 |
| Th | 0.9 ± 0.1 | <0.0013 | 1.1 ± 0.1 | <0.0013 |
| U | 2.5 ± 0.3 | <0.0012 | 2.9 ± 0.2 | <0.0012 |
| Sample | 226Ra | 210Pb | 40K | 232Th | 235U |
|---|---|---|---|---|---|
| Raw Material | |||||
| PG (Bq/kg) | (5.68 ± 0.25) 102 | (7.2 ± 0.3)102 | <111 | 4 ± 1 | 19 ± 3 |
| TCLP (Bq/L) | 0.65 ± 0.15 (ŋ = 2.3%) | 4.0 ± 1.3(ŋ = 11.2%) | <3 | <1 | <2 |
| PG + (Na(OH) | |||||
| Dissolution reaction | |||||
| C_S (Bq/kg) | (7.0 ± 0.3) 102 | (1.03 ± 0.05) 103 | 16 ± 5 | 8 ± 1 | 28 ± 3 |
| TCLP (Bq/L) | <1 | <3 | <3 | <1 | <2 |
| Carbonation reaction | |||||
| Ca_S (Bq/kg) | (7.9 ± 0.3) 102 | (1.01 ± 0.07) 103 | <65 | 8 ± 1 | 20 ± 4 |
| TCLP (Bq/L) | 1.3 ± 0.4 (ŋ = 3.3%) | 2.2 ± 1.1 (ŋ = 4.3%) | <2 | <0.25 | <0.2 |
| Mortar production | |||||
| Mortar (Bq/kg) | (2.32 ± 0.10) 102 | (3.65 ± 0.17) 102 | 80 ± 17 | 5.6 ± 0.5 | 8 ± 2 |
| TCLP (Bq/L) | 0.85 ± 0.25 (ŋ = 7.3%) | 2 ± 1 (ŋ = 11.0%) | <2.5 | <1 | <1 |
| PG + Caustic wastes. | |||||
| Dissolution reaction | |||||
| PGAS (Bq/kg) | (4.45 ± 0.19) 102 | (3.90 ± 0.18) 102 | <61 | 4 ± 1 | 10 ± 2 |
| TCLP (Bq/L) | 0.9 ± 0.2 (ŋ = 4.3%) | <3.5 | <4 | <1 | <2 |
| Carbonation reaction | |||||
| PGAB (Bq/kg) | (4.17 ± 0.17) 102 | (4.1 ± 0.3) 102 | <27 | 5 ± 1 | 10 ± 2 |
| TCLP (Bq/L) | 3 ± 1 (ŋ = 14.4%) | <3 | <3 | <1 | <2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romero-Hermida, M.I.; Flores-Alés, V.; Hurtado-Bermúdez, S.J.; Santos, A.; Esquivias, L. Environmental Impact of Phosphogypsum-Derived Building Materials. Int. J. Environ. Res. Public Health 2020, 17, 4248. https://doi.org/10.3390/ijerph17124248
Romero-Hermida MI, Flores-Alés V, Hurtado-Bermúdez SJ, Santos A, Esquivias L. Environmental Impact of Phosphogypsum-Derived Building Materials. International Journal of Environmental Research and Public Health. 2020; 17(12):4248. https://doi.org/10.3390/ijerph17124248
Chicago/Turabian StyleRomero-Hermida, M. I., V. Flores-Alés, S. J. Hurtado-Bermúdez, A. Santos, and L. Esquivias. 2020. "Environmental Impact of Phosphogypsum-Derived Building Materials" International Journal of Environmental Research and Public Health 17, no. 12: 4248. https://doi.org/10.3390/ijerph17124248






