Occurrence and Antimicrobial Resistance Traits of Escherichia coli from Wild Birds and Rodents in Singapore
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection, Isolation and Identification
2.2. Antimicrobial Susceptibility Testing of E. coli Isolates
2.3. Extended Spectrum Beta-Lactamases (ESBL) Testing for E. coli Isolates
2.4. Genotypic Characterisation by Whole Genome Sequencing
2.5. Statistical Analysis
3. Results
3.1. Occurrence of E. coli in Wild Birds and Rodents
3.2. Antimicrobial Resistance in E. coli Isolated from Wild Birds and Rodents
3.3. Distribution of Resistance Genes in Eight MDR E. coli Isolates from Wild Birds and Rodents
3.4. Comparison between Phenotypic and Genotypic Characteristics of MDR E. coli Isolates
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Van den Bogaard, A.E.; Stobberingh, E.E. Epidemiology of resistance to antibiotics: Links between animals and humans. Int. J. Antimicrob. Agents 2000, 14, 327–335. [Google Scholar] [CrossRef]
- Marinho, C.M.; Santos, T.; Gonçalves, A.; Poeta, P.; Igrejas, G. A Decade-Long Commitment to Antimicrobial Resistance Surveillance in Portugal. Front. Microbiol. 2016, 7, 1650. [Google Scholar] [CrossRef] [PubMed]
- Jindal, A.K.; Pandya, K.; Khan, I.D. Antimicrobial resistance: A public health challenge. Med. J. Armed Forces Ind. 2015, 71, 178–181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Poirel, L.; Madec, J.Y.; Lupo, A.; Schink, A.K.; Kieffer, N.; Nordmann, P.; Schwarz, S. Antimicrobial Resistance in Escherichia coli. Microbiol. Spectr. 2018, 6, 4:1–4:27. [Google Scholar]
- Rasheed, M.U.; Thajuddin, N.; Ahamed, P.; Teklemariam, Z.; Jamil, K. Antimicrobial Drug Resistance in Strains of Escherichia coli isolated from Food Sources. Rev. Inst. Med. Trop. São Paulo 2014, 56, 341–346. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation. Global Priority List of Antibiotic-Resistant Bacteria to Guide Research, Discovery, and Development of New Antibiotics 2017. Available online: http://apps.who.int/medicinedocs/documents/s23171en/s23171en.pdf (accessed on 14 May 2020).
- Guo, S.; Tay, M.Y.F.; Aung, K.T.; Seow, K.L.G.; Ng, L.C.; Purbojati, R.W.; Drautz-Moses, D.I.; Schuster, S.C.; Schlundt, J. Phenotypic and genotypic characterization of antimicrobial resistant Escherichia coli isolated from ready-to-eat food in Singapore using disk diffusion, broth microdilution and whole genome sequencing methods. Food Control 2019, 99, 89–97. [Google Scholar] [CrossRef]
- Hsu, L.-Y.; Tan, T.-Y.; Jureen, R.; Koh, T.-H.; Krishnan, P.; Lin, R.T.-P.; Tee, N.W.-S.; Tambyah, P.A. Antimicrobial Drug Resistance in Singapore Hospitals. Emerg. Infect. Dis. J. 2007, 13, 1944. [Google Scholar] [CrossRef]
- Wang, J.; Ma, Z.-B.; Zeng, Z.-L.; Yang, X.-W.; Huang, Y.; Liu, J.-H. The role of wildlife (wild birds) in the global transmission of antimicrobial resistance genes. Zool. Res. 2017, 38, 55–80. [Google Scholar] [CrossRef] [Green Version]
- Ho, P.-L.; Lo, W.-U.; Lai, E.L.; Law, P.Y.; Leung, S.M.; Wang, Y.; Chow, K.-H. Clonal diversity of CTX-M-producing, multidrug-resistant Escherichia coli from rodents. J. Med. Microbiol. 2015, 64, 185–190. [Google Scholar] [CrossRef]
- Lane, D.J. 16S/23S rRNA sequencing in nucleic acid techniques. In Nucleic Acid Techniques in Bacterial Systematics; Stackebrandt, E., Goodfellow, M., Eds.; John Wiley and Sons: New York, NY, USA, 1991; pp. 115–148. [Google Scholar]
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing, 30th ed.; CLSI Supplement M100; CLSI: Wayne, PA, USA, 2020. [Google Scholar]
- Exner, M.; Bhattacharya, S.; Christiansen, B.; Gebel, J.; Goroncy-Bermes, P.; Hartemann, P.; Heeg, P.; Ilschner, C.; Kramer, A.; Larson, E.; et al. Antibiotic resistance: What is so special about multidrug-resistant Gram-negative bacteria? GMS Hyg. Infect. Control 2017, 12, Doc05. [Google Scholar]
- Bankevich, A.; Nurk, S.; Antipov, D.; Gurevich, A.A.; Dvorkin, M.; Kulikov, A.S.; Lesin, V.M.; Nikolenko, S.I.; Pham, S.; Prjibelski, A.D.; et al. SPAdes: A new genome assembly algorithm and its applications to single-cell sequencing. J. Comput. Biol. 2012, 19, 455–477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zankari, E.; Hasman, H.; Cosentino, S.; Vestergaard, M.; Rasmussen, S.; Lund, O.; Aarestrup, F.M.; Larsen, M.V. Identification of acquired antimicrobial resistance genes. J. Antimicrob. Chemother. 2012, 67, 2640–2644. [Google Scholar] [CrossRef] [PubMed]
- Zurfluh, K.; Albini, S.; Mattmann, P.; Kindle, P.; Nüesch-Inderbinen, M.; Stephan, R.; Vogler, B.R. Antimicrobial resistant and extended-spectrum β-lactamase producing Escherichia coli in common wild bird species in Switzerland. Microbiol. Open 2019, 8, e845. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shobrak, M.Y.; Abo-Amer, A.E. Role of wild birds as carriers of multi-drug resistant Escherichia coli and Escherichia vulneris. Braz. J. Microbiol. 2014, 45, 1199–1209. [Google Scholar] [CrossRef] [Green Version]
- Nkogwe, C.; Raletobana, J.; Stewart-Johnson, A.; Suepaul, S.; Adesiyun, A. Frequency of Detection of Escherichia coli, Salmonella spp., and Campylobacter spp. in the Faeces of Wild Rats Rattus spp.) in Trinidad and Tobago. Vet. Med. Int. 2011, 2011, 686923. [Google Scholar] [CrossRef] [Green Version]
- Himsworth, C.G.; Zabek, E.; Desruisseau, A.; Parmley, E.J.; Reid-Smith, R.; Jardine, C.M.; Tang, P.; Patrick, D.M. Prevalence and Characteristics of Escherichia Coli and Salmonella spp. in the Feces of Wild Urban Norway and Black Rats (Rattus Norvegicus and Rattus Rattus) from an Inner-City Neighborhood of Vancouver, Canada. J. Wildl. Dis. 2015, 51, 589–600. [Google Scholar] [CrossRef]
- National Environment Agency. What is the Difference Between a Rat and a Shrew? Should the Public Be Worried about the Presence of Shrews? 2016. Available online: https://www.nea.gov.sg/media/nea-vox/index/what-is-the-difference-between-a-rat-and-a-shrew-should-the-public-be-worried-about-the-presence-of-shrews (accessed on 14 July 2020).
- Arnold, K.E.; Williams, N.J.; Bennett, M. ‘Disperse abroad in the land’: The role of wildlife in the dissemination of antimicrobial resistance. Biol. Lett. 2016, 12, 20160137. [Google Scholar] [CrossRef] [Green Version]
- Aung, K.T.; Chen, H.J.; Chau, M.L.; Yap, G.; Lim, X.F.; Humaidi, M.; Chua, C.; Yeo, G.; Yap, H.M.; Oh, J.Q.; et al. Salmonella in Retail. Food and Wild Birds in Singapore-Prevalence, Antimicrobial Resistance, and Sequence Types. Int. J. Environ. Res. Public Health 2019, 16, 4235. [Google Scholar] [CrossRef] [Green Version]
- Varga, C.; Rajić, A.; McFall, M.E.; Reid-Smith, R.J.; Deckert, A.E.; Pearl, D.L.; Avery, B.P.; Checkley, S.L.; McEwen, S.A. Comparison of antimicrobial resistance in generic Escherichia coli and Salmonella spp. cultured from identical fecal samples in finishing swine. Can. J. Vet. Res. 2008, 72, 181–187. [Google Scholar]
- Mathew, A.; Jackson, F.; Saxton, A. Effects of antibiotic regimens on resistance of Escherichia coli and Salmonella serovar Typhimurium in swine. J. Swine Health Prod. 2002, 10, 7–13. [Google Scholar]
- Chopra, I.; Roberts, M. Tetracycline antibiotics: Mode of action, applications, molecular biology, and epidemiology of bacterial resistance. Microbiol. Mol. Biol. Rev. 2001, 65, 232–260. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diren Sigirci, B.; Celik, B.; Başaran Kahraman, B.; Bagcigil, A.F.; Ak, S. Tetracycline Resistance of Enterobacteriaceae Isolated from Feces of Synanthropic Birds. J. Exot. Pet Med. 2019, 28, 13–18. [Google Scholar] [CrossRef]
- Nessim, N. Antibiotics in 2019: Everything You Need to Know. Available online: https://www.solvhealth.com/blog/antibiotics-everything-you-need-to-know (accessed on 14 March 2020).
- Pozzi, S.P.; Ben-David, Z. Use of amoxicillin + clavulanic acid combination in Veterinary Medicine and possible antibiotic-resistance in human pathogens; A world-wide overview. Isr. J. Vet. Med. 2002, 57, 95. [Google Scholar]
- Iredell, J.; Brown, J.; Tagg, K. Antibiotic resistance in Enterobacteriaceae: Mechanisms and clinical implications. Bmj 2016, 352, h6420. [Google Scholar] [CrossRef]
- Zwe, Y.H.; Tang, V.C.Y.; Aung, K.T.; Gutiérrez, R.A.; Ng, L.C.; Yuk, H.-G. Prevalence, sequence types, antibiotic resistance and, gyrA mutations of Salmonella isolated from retail fresh chicken meat in Singapore. Food Control 2018, 90, 233–240. [Google Scholar] [CrossRef]
- Hornish, R.E.; Kotarski, S.F. Cephalosporins in veterinary medicine—Ceftiofur use in food animals. Curr. Top. Med. Chem. 2002, 2, 717–731. [Google Scholar] [CrossRef]
- Fritzenwanker, M.; Imirzalioglu, C.; Herold, S.; Wagenlehner, F.M.; Zimmer, K.P.; Chakraborty, T. Treatment Options for Carbapenem- Resistant Gram-Negative Infections. Dtsch Arztebl Int. 2018, 115, 345–352. [Google Scholar]
- Chua, K.Y.L.; Stewardson, A.J. Individual and community predictors of urinary ceftriaxone-resistant Escherichia coli isolates, Victoria, Australia. Antimicrob. Resist. Infect. Control 2019, 8, 36. [Google Scholar] [CrossRef] [Green Version]
- Cheng, G.; Ning, J.; Ahmed, S.; Huang, J.; Ullah, R.; An, B.; Hao, H.; Dai, M.; Huang, L.; Wang, X.; et al. Selection and dissemination of antimicrobial resistance in Agri-food production. Antimicrob. Resist. Infect. Control 2019, 8, 158. [Google Scholar] [CrossRef] [Green Version]
- Edgar, R.; Bibi, E. A single membrane-embedded negative charge is critical for recognizing positively charged drugs by the Escherichia coli multidrug resistance protein MdfA. EMBO J. 1999, 18, 822–832. [Google Scholar] [CrossRef] [Green Version]
- Sutcliffe, J.A.; Leclercq, R. Mechanisms of Resistance to Macrolides, Lincosamides, and Ketolides. In Macrolide Antibiotics; Schönfeld, W., Kirst, H.A., Eds.; Birkhäuser Basel: Basel, Switzerland, 2002; pp. 281–317. [Google Scholar]
- Edgar, R.; Bibi, E. MdfA, an Escherichia coli multidrug resistance protein with an extraordinarily broad spectrum of drug recognition. J. Bacteriol. 1997, 179, 2274–2280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noguchi, N.; Takada, K.; Katayama, J.; Emura, A.; Sasatsu, M. Regulation of transcription of the mph(A) gene for macrolide 2′-phosphotransferase I in Escherichia coli: Characterization of the regulatory gene mphR(A). J. Bacteriol. 2000, 182, 5052–5058. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Handrova, L.; Kmet, V. ; Kmet, V. Antibiotic resistance and virulence factors of Escherichia coli from eagles and goshawks. J. Environ. Sci. Health Part B 2019, 54, 605–614. [Google Scholar] [CrossRef] [PubMed]
- Huy, H.; Koizumi, N.; Ung, T.; Le, T.; Nguyen, H.; Hoang, P.; Nguyen, C.; Khong, T.; Hasebe, F.; Haga, T.; et al. Antibiotic-resistant Escherichia coli isolated from urban rodents in Hanoi, Vietnam. J. Vet. Med. Sci. 2020, 82, 653–660. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leski, T.A.; Bangura, U.; Jimmy, D.H.; Ansumana, R.; Lizewski, S.E.; Stenger, D.A.; Taitt, C.R.; Vora, G.J. Multidrug-resistant tet(X)-containing hospital isolates in Sierra Leone. Int. J. Antimicrob. Agents 2013, 42, 83–86. [Google Scholar] [CrossRef] [PubMed]
- Clark, N.C.; Olsvik, O.; Swenson, J.M.; Spiegel, C.A.; Tenover, F.C. Detection of a streptomycin/spectinomycin adenylyltransferase gene (aadA) in Enterococcus faecalis. Antimicrob. Agents Chemother. 1999, 43, 157–160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marcelino, V.; Wille, M.; Hurt, A.; González-Acuña, D.; Klaassen, M.; Eden, J.-S.; Shi, M.; Iredell, J.; Sorrell, T.; Holmes, E. Meta-transcriptomics reveals a diverse antibiotic resistance gene pool in avian microbiomes. BMC Biol. 2019, 17, 31:1–31:11. [Google Scholar] [CrossRef] [Green Version]
- Hassan, R.; Tantawy, M.; Gouda, N.A.; Elzayat, M.G.; Gabra, S.; Nabih, A.; Diab, A.A.; El-Hadidi, M.; Bakry, U.; Shoeb, M.R.; et al. Genotypic characterization of multiple drug resistant Escherichia coli isolates from a pediatric cancer hospital in Egypt. Sci. Rep. 2020, 10, 4165. [Google Scholar] [CrossRef]
- Van Hoek, A.H.A.M.; Mevius, D.; Guerra, B.; Mullany, P.; Roberts, A.P.; Aarts, H.J.M. Acquired antibiotic resistance genes: An overview. Front. Microbiol. 2011, 2, 203. [Google Scholar] [CrossRef] [Green Version]
- Lu, J.; Zhang, J.; Xu, L.; Liu, Y.; Li, P.; Zhu, T.; Cheng, C.; Lu, S.; Xu, T.; Yi, H.; et al. Spread of the florfenicol resistance floR gene among clinical Klebsiella pneumoniae isolates in China. Antimicrob. Resist. Infect. Control 2018, 7, 127. [Google Scholar] [CrossRef] [Green Version]
- Cloeckaert, A.; Baucheron, S.; Flaujac, G.; Schwarz, S.; Kehrenberg, C.; Martel, J.-L.; Chaslus-Dancla, E. Plasmid-Mediated Florfenicol Resistance Encoded by the floR Gene in Escherichia coli Isolated from Cattle. Antimicrob. Agents Chemother. 2000, 44, 2858–2860. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olawale, O.A.; Obasola, E.F.; Yvonne, A. Antibiotic resistance and resistance genes in Escherichia coli from poultry farms, southwest Nigeria. J. Infect. Dev. Ctries. 2014, 8, 1103–1112. [Google Scholar]
- Aeksiri, N.; Toanan, W.; Sawikan, S.; Suwannarit, R.; Pungpomin, P.; Khieokhajonkhet, A.; Niumsup, P.R. First Detection and Genomic Insight into mcr-1 Encoding Plasmid-Mediated Colistin-Resistance Gene in Escherichia coli ST101 Isolated from the Migratory Bird Species Hirundo rustica in Thailand. Microb. Drug Resist. 2019, 25, 1437–1442. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Shoichet, B.; Bonnet, R. Structure, function, and inhibition along the reaction coordinate of CTX-M beta-lactamases. J. Am. Chem. Soc. 2005, 127, 5423–5434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, H.; Sun, F.; Chen, J.; Ou, Q.; Feng, W.; Yong, X.; Xia, P. Epidemiology of CTX-M-type extended-spectrum beta-lactamase (ESBL)-producing nosocomial-Escherichia coli infection in China. Ann. Clin. Microbiol. Antimicrob. 2015, 14, 1–5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, H.; Kim, J.; Ryu, S.; Jeon, B. Predominance of blaCTX-M-65 and blaCTX-M-55 in extended-spectrum β-lactamase-producing Escherichia coli from raw retail chicken in South Korea. J. Glob. Antimicrob. Resist. 2019, 17, 216–220. [Google Scholar] [CrossRef]
- Jacoby, G.A. Mechanisms of resistance to quinolones. Clin. Infect. Dis. 2005, 41 (Suppl. 2), S120–S126. [Google Scholar] [CrossRef] [Green Version]
- Oh, J.Y.; Kwon, Y.K.; Tamang, M.D.; Jang, H.K.; Jeong, O.M.; Lee, H.S.; Kang, M.S. Plasmid-Mediated Quinolone Resistance in Escherichia coli Isolates from Wild Birds and Chickens in South Korea. Microb. Drug Resist. 2016, 22, 69–79. [Google Scholar] [CrossRef]
- Literak, I.; Micudova, M.; Tausova, D.; Cizek, A.; Dolejska, M.; Papousek, I.; Prochazka, J.; Vojtech, J.; Borleis, F.; Guardone, L.; et al. Plasmid-mediated quinolone resistance genes in fecal bacteria from rooks commonly wintering throughout Europe. Microb. Drug Resist. 2012, 18, 567–573. [Google Scholar] [CrossRef]
- Kim, H.B.; Wang, M.; Park, C.H.; Kim, E.-C.; Jacoby, G.A.; Hooper, D.C. oqxAB Encoding a Multidrug Efflux Pump in Human Clinical Isolates of Enterobacteriaceae. Antimicrob. Agents Chemother. 2009, 53, 3582–3584. [Google Scholar] [CrossRef] [Green Version]
- Ruiz, J. Mechanisms of resistance to quinolones: Target alterations, decreased accumulation and DNA gyrase protection. J. Antimicrob. Chemother. 2003, 51, 1109–1117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Z.; Meng, X.; Wang, Y.; Xia, X.; Wang, X.; Xi, M.; Meng, J.; Shi, X.; Wang, D.; Yang, B. Presence of qnr, aac(6′)-Ib, qepA, oqxAB, and mutations in gyrase and topoisomerase in nalidixic acid-resistant Salmonella isolates recovered from retail chicken carcasses. Foodborne Pathog. Dis. 2014, 11, 698–705. [Google Scholar] [CrossRef] [PubMed]
- Gow, S.P.; Waldner, C.L.; Harel, J.; Boerlin, P. Associations between Antimicrobial Resistance Genes in Fecal Generic Escherichia coli Isolates from Cow-Calf Herds in Western Canada. Appl. Environ. Microbiol. 2008, 74, 3658. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lou, Y.; Liu, H.; Zhang, Z.; Pan, Y.; Zhao, Y. Mismatch between antimicrobial resistance phenotype and genotype of pathogenic Vibrio parahaemolyticus isolated from seafood. Food Control 2016, 59, 207–211. [Google Scholar] [CrossRef]
- Fernández, L.; Hancock, R.E.W. Adaptive and mutational resistance: Role of porins and efflux pumps in drug resistance. Clin. Microbiol. Rev. 2012, 25, 661–681. [Google Scholar]
- Kindle, P.; Zurfluh, K.; Nuesch-Inderbinen, M.; von Ah, S.; Sidler, X.; Stephan, R.; Kummerlen, D. Phenotypic and genotypic characteristics of Escherichia coli with non-susceptibility to quinolones isolated from environmental samples on pig farms. Porcine Health Manag. 2019, 5, 9. [Google Scholar] [CrossRef]
- Nguyen, D.T.; Kanki, M.; Nguyen, P.D.; Le, H.T.; Ngo, P.T.; Tran, D.N.; Le, N.H.; Dang, C.V.; Kawai, T.; Kawahara, R.; et al. Prevalence, antibiotic resistance, and extended-spectrum and AmpC beta-lactamase productivity of Salmonella isolates from raw meat and seafood samples in Ho Chi Minh City, Vietnam. Int. J. Food Microbiol. 2016, 236, 115–122. [Google Scholar] [CrossRef]
- Argudín, M.A.; Deplano, A.; Meghraoui, A.; Dodémont, M.; Heinrichs, A.; Denis, O.; Nonhoff, C.; Roisin, S. Bacteria from Animals as a Pool of Antimicrobial Resistance Genes. Antibiotics 2017, 6, 12. [Google Scholar] [CrossRef]
- Dolejska, M.; Literak, I. Wildlife is overlooked in the epidemiology of medically important antimicrobial resistant bacteria. Antimicrob. Agents Chemother. 2019, 63, 1167-19. [Google Scholar] [CrossRef] [Green Version]
| Sample ID | Sample Type | Source | AK30 | AMC30 | AMP10 | CRO30 | C30 | CIP5 | CN10 | MEM10 | NA30 | NOR10 | SXT25 | TE30 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C1776# | Wild Bird | Crow | S | S | R | S | R | R | S | S | R | R | R | R |
| C1775 | Wild Bird | Myna | S | S | R | S | S | S | S | S | S | S | S | S |
| 8657-0407-0411-1 | Rodent | Rodent | S | S | S | S | S | S | S | S | S | S | S | S |
| 8657-0948-0411 | Rodent | Rodent | S | S | I | S | S | S | S | S | S | S | S | S |
| 8645-0135# | Rodent | Rodent | S | S | R | S | R | S | S | S | S | S | R | R |
| C1797# | Wild Bird | Crested Goshawk | S | S | I | S | R | R | S | S | R | R | S | R |
| C1809-1 | Wild Bird | Hooded Pitta | S | S | I | S | S | S | S | S | S | S | S | S |
| C1789 | Wild Bird | Crow | S | S | I | S | S | S | S | S | S | S | S | S |
| C1798 | Wild Bird | Black-naped oriole | S | S | I | S | S | S | S | S | S | S | S | S |
| HHK-M04-060417-1 | Rodent | Rodent | S | S | S | S | S | S | S | S | S | S | S | S |
| HHK-M04-060417-3 | Rodent | Rodent | S | S | I | S | S | S | S | S | S | S | S | S |
| 8642-0159-0410-1 | Rodent | Rodent | S | S | S | S | S | S | S | S | S | S | S | S |
| C1742# | Wild Bird | Yellow Bittern | S | I | R | S | R | S | S | S | S | S | S | R |
| C1779 | Wild Bird | Crow | S | S | S | S | S | S | S | S | S | S | S | S |
| C1805-1# | Wild Bird | Crow | S | I | R | S | R | S | S | S | S | S | S | R |
| 8657-0146-0224 | Rodent | Rodent | S | S | S | S | S | S | S | S | S | S | S | S |
| NHA-M02-230117 | Rodent | Rodent | S | S | S | S | S | S | S | S | S | S | S | S |
| 8657-0258-0411-1 | Rodent | Rodent | S | S | S | S | S | S | S | S | S | S | S | S |
| 7574-0224-0224 | Rodent | Rodent | S | S | I | S | S | S | S | S | S | S | S | S |
| C1802-1 | Wild Bird | Crow | S | S | S | S | S | S | R | S | S | S | S | R |
| 8646-0251-0301 | Rodent | Rodent | S | S | I | S | S | S | S | S | S | S | S | S |
| HHK-M05-160317 | Rodent | Rodent | S | S | S | S | S | S | S | S | S | S | S | S |
| C1743E | Wild Bird | Black Bittern | S | S | S | S | S | S | S | S | S | S | S | S |
| C1781 | Wild Bird | Black Bittern | S | S | I | S | S | S | S | S | S | S | S | S |
| HHK-M07-060417-1 | Rodent | Rodent | S | I | R | S | S | S | S | S | S | S | S | R |
| C1806-1 | Wild Bird | Pied Fantail | S | S | S | S | S | S | S | S | S | S | S | S |
| C1736 | Wild Bird | Brahmin Kite | S | S | S | S | S | S | S | S | S | S | S | S |
| 8645-0205-0411-1 | Rodent | Rodent | S | S | S | S | S | S | S | S | S | S | S | S |
| C1795 | Wild Bird | Sparrow Hawk | S | S | I | S | S | S | S | S | R | S | S | S |
| C1794 | Wild Bird | Grey Heron | S | I | R | S | S | S | S | S | I | S | S | R |
| NRS-M02-150317 | Rodent | Rodent | S | S | S | S | S | S | S | S | S | S | S | S |
| C1783 | Wild Bird | Scops Owl | S | S | S | S | S | S | S | S | S | S | S | S |
| C1770 | Wild Bird | Myna | S | S | S | S | S | S | S | S | S | S | S | R |
| C1758# | Wild Bird | Black Bittern | S | I | R | S | S | S | S | S | S | S | R | R |
| C1757 | Wild Bird | Yellow Bittern | S | S | I | S | S | S | S | S | S | S | S | S |
| C1750 | Wild Bird | Sparrow Hawk | S | I | R | S | S | S | S | S | S | S | S | S |
| C1737 | Wild Bird | Crow | S | S | I | S | S | S | S | S | S | S | S | S |
| C1740 | Wild Bird | Crow | S | R | R | S | S | S | S | S | S | S | S | S |
| C1803-2 | Wild Bird | Crow | S | S | I | S | S | S | S | S | S | S | S | S |
| 8656-0339-0224 | Rodent | Rodent | S | S | S | S | S | S | S | S | S | S | S | S |
| TAH-M01-210317 | Rodent | Rodent | S | S | S | S | S | S | S | S | R | S | S | S |
| HHK-M04-060417-1A | Rodent | Rodent | S | S | S | S | S | S | S | S | S | S | S | S |
| 8655-0114# | Rodent | Rodent | S | R | R | S | S | R | S | S | R | R | S | R |
| C1738 | Wild Bird | Crow | S | S | I | S | S | S | S | S | S | S | S | S |
| C1722# | Wild Bird | Crow | S | R | R | R | R | I | R | S | R | S | R | R |
| HHK-M06-060417-1 | Rodent | Rodent | S | S | S | S | S | S | S | S | S | S | S | S |
| Types of Samples | Percentage of Samples Positive for E. coli | Sample Name (Scientific Name) | No. of E. coli Isolates |
|---|---|---|---|
| Wild Birds | 27.1% (26/96) [95% CI: 19.2–36.7%] | Crow (Corvus spp.) | Ten |
| Black Bittern (Ixobrychus flavicollis) | Three | ||
| Myna (Acridotheres spp.) | Two | ||
| Sparrow Hawk (Accipiter spp.) | Two | ||
| Yellow Bittern (Ixobrychus sinensis) | Two | ||
| Black-naped oriole (Oriolus chinensis) | One | ||
| Brahminy Kite (Haliastur indus) | One | ||
| Crested Goshawk (Accipiter trivirgatus) | One | ||
| Grey Heron (Ardea cinerea) | One | ||
| Hooded Pitta (Pitta sordida) | One | ||
| Pied Fantail (Rhipidura javanica) | One | ||
| Scops Owl (Otus spp.) | One | ||
| Rodents | 14.8% (20/135) [95% CI: 9.8–21.8%] | Black Rat/Brown Rat/House Mouse (Rattus rattus/Rattus norvegicus/Mus musculus) | 20 |
| Wild Birds | Rodents | Z-Score |
|---|---|---|
| 80.8% (21/26) [95% CI: 62.1–91.5%] | 40.0% (8/20) [95% CI: 21.9–61.3%] | 2.8 (p < 0.05) |
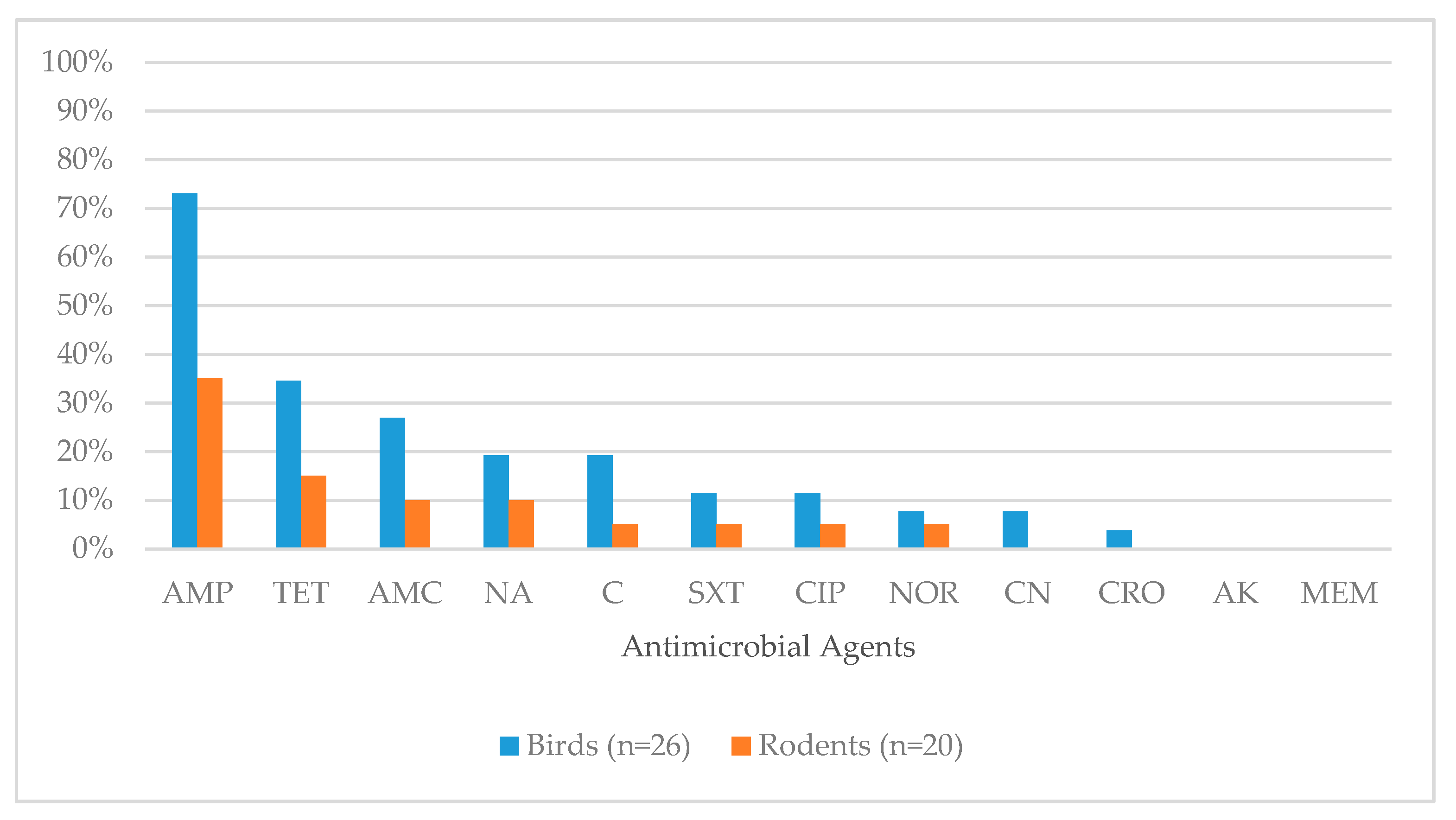
| Antimicrobial Class | Antimicrobial Agent Tested in the Study | Percentage of Isolates Showing Resistant Phenotype (n) | |
|---|---|---|---|
| Wild Birds (26) | Rodents (20) | ||
| Penicillins | Ampicillin | 73.1% (19/26) | 35.0% (7/20) |
| Tetracyclines | Tetracycline | 34.6% (9/26) | 15.0% (3/20) |
| Beta-lactam/beta-lactamase Inhibitor Combinations | Amoxicillin/Clavulanic acid | 26.9% (7/26) | 10.0% (2/20) |
| Quinolones | Nalidixic acid (Quinolone) | 19.2% (5/26) | 10.0% (2/20) |
| Phenicols | Chloramphenicol | 19.2% (5/26) | 5.0% (1/20) |
| Folate Pathway Synthesis | Sulphamethoxazole/Trimethoprim | 11.5% (3/26) | 5.0% (1/20) |
| Quinolones | Ciprofloxacin (Fluoroquinolone) | 11.5% (3/26) | 5.0% (1/20) |
| Quinolones | Norfloxacin (Fluoroquinolone) | 7.7% (2/26) | 5.0% (1/20) |
| Aminoglycosides | Gentamicin | 7.7% (2/26) | 0.0% |
| Third Generation Cephalosporins | Ceftriaxone | 3.8% (1/26) | 0.0% |
| Aminoglycosides | Amikacin | 0.0% | 0.0% |
| Carbapenems | Meropenem | 0.0% | 0.0% |
| Isolate ID | Sample Source | Sample Description | Resistance Genes (n) | Aminoglycoside | ESBL | Quinolone | Fosfomycin | MLS a | Phenicol | Sulphonamide | Tetracycline | Trimethoprim | Chromosomal Point Mutations |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C1722 | Wild bird | Crow | 17 | aac(3)-IV, aadA2, aph(3′)-Ia, aph(3″)-Ib, aph(4)-Ia, aph(6)-Id, | blaCTX-M-65, blaTEM-1B | - | fosA3 | mdf(A) | cmlA1, floR | sul2, sul3 | tet(A), tet(X) | dfrA12 | gyrA (Ser83Leu) |
| C1742 | Wild bird | Yellow Bittern | 7 | aph(3′)-Ia | blaTEM-176 | qnrS1 | - | mdf(A) | floR | - | tet(A) | dfrA14 | - |
| C1758 | Wild bird | Black Bittern | 4 | aph(6)-Id | qnrS1 | - | mdf(A) | - | - | tet(A) | - | - | |
| C1776 | Wild bird | Crow | 11 | aadA1, aadA2, aph(3″)-Ib, aph(6)-Id | blaTEM-1B | oqxB | - | mdf(A), mph(A) | cmlA1 | sul3 | tet(A) | - | gyrA, (Ser83Leu), gyrA(Asp87Asn) & parC (Ser80Ile). |
| C1797 | Wild bird | Crested Goshawk | 3 | - | - | - | - | mdf(A) | catA1 | - | tet(B) | - | gyrA (Ser83Leu), gyrA(Asp87Asn) & parC (Ser80Ile). |
| C1805-1 | Wild bird | Crow | 7 | aadA1 | blaTEM-1B | qnrS1 | - | mdf(A) | floR | sul3 | tet(A) | - | - |
| 8645-0135 | Rodent | Rodent | 11 | aadA1, aadA2 | blaTEM-1B | qnrS1 | - | mdf(A) | cmlA1, floR | sul2, sul3 | tet(A) | dfrA12 | - |
| 8655-0114 | Rodent | Rodent | 1 | - | - | - | - | mdf(A) | - | - | - | - | - |
| Isolate ID | Sample Source | Sample Description | Beta-lactams | Quinolones | Phenicols | Tetracyclines | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Phenotype | Genotype | Phenotype | Genotype | Phenotype | Genotype | Phenotype | Genotype | |||
| C1722 | Wild bird | Crow | AMC *, AMP, CRO | blaCTX-M-65, blaTEM-1B | CIP, NA | Chromosomal mutations gyrA (Ser83Leu) | C | cmlA1, floR | TE | tet(A), tet(X) |
| C1742 | Wild bird | Yellow Bittern | AMC *, AMP | blaTEM-176 | - | qnrS1 | C | floR | TE | tet(A) |
| C1758 | Wild bird | Black Bittern | AMC *, AMP | - | - | qnrS1 | - | - | TE | tet(A) |
| C1776 | Wild bird | Crow | AMP | blaTEM-1B | CIP, NA, NOR | oqxB, Chromosomal mutations gyrA (Ser83Leu) gyrA (Asp87Asn) & parC (Ser80Ile) | C | cmlA1 | TE | tet(A) |
| C1797 | Wild bird | Crested Goshawk | AMP | - | CIP, NA, NOR | Chromosomal mutations gyrA (Ser83Leu) gyrA (Asp87Asn) & parC (Ser80Ile) | C | catA1 | TE | tet(B) |
| C1805_1 | Wild bird | Crow | AMP, AMC * | blaTEM-1B | - | qnrS1 | C | floR | TE | tet(A) |
| 8645_0135 | Rodent | Rodent | AMP | blaTEM-1B | - | qnrS1 | C | cmlA1, floR | TE | tet(A) |
| 8655_0114 | Rodent | Rodent | AMC *, AMP | - | CIP, NA, NOR | - | - | - | TE | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ong, K.H.; Khor, W.C.; Quek, J.Y.; Low, Z.X.; Arivalan, S.; Humaidi, M.; Chua, C.; Seow, K.L.G.; Guo, S.; Tay, M.Y.F.; et al. Occurrence and Antimicrobial Resistance Traits of Escherichia coli from Wild Birds and Rodents in Singapore. Int. J. Environ. Res. Public Health 2020, 17, 5606. https://doi.org/10.3390/ijerph17155606
Ong KH, Khor WC, Quek JY, Low ZX, Arivalan S, Humaidi M, Chua C, Seow KLG, Guo S, Tay MYF, et al. Occurrence and Antimicrobial Resistance Traits of Escherichia coli from Wild Birds and Rodents in Singapore. International Journal of Environmental Research and Public Health. 2020; 17(15):5606. https://doi.org/10.3390/ijerph17155606
Chicago/Turabian StyleOng, Kar Hui, Wei Ching Khor, Jing Yi Quek, Zi Xi Low, Sathish Arivalan, Mahathir Humaidi, Cliff Chua, Kelyn L. G. Seow, Siyao Guo, Moon Y. F. Tay, and et al. 2020. "Occurrence and Antimicrobial Resistance Traits of Escherichia coli from Wild Birds and Rodents in Singapore" International Journal of Environmental Research and Public Health 17, no. 15: 5606. https://doi.org/10.3390/ijerph17155606






