Effect of Combined Physical and Cognitive Interventions on Executive Functions in Older Adults: A Meta-Analysis of Outcomes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Eligibility Criteria
2.3. Data Extraction and Analysis
2.4. Evaluation of Methodological Quality
3. Results
3.1. Included Studies
3.2. Combined Interventions vs. the Control Group
3.3. Combined Interventions vs. Cognitive Intervention Alone
3.4. Combined Interventions vs. Physical Exercise Intervention Alone
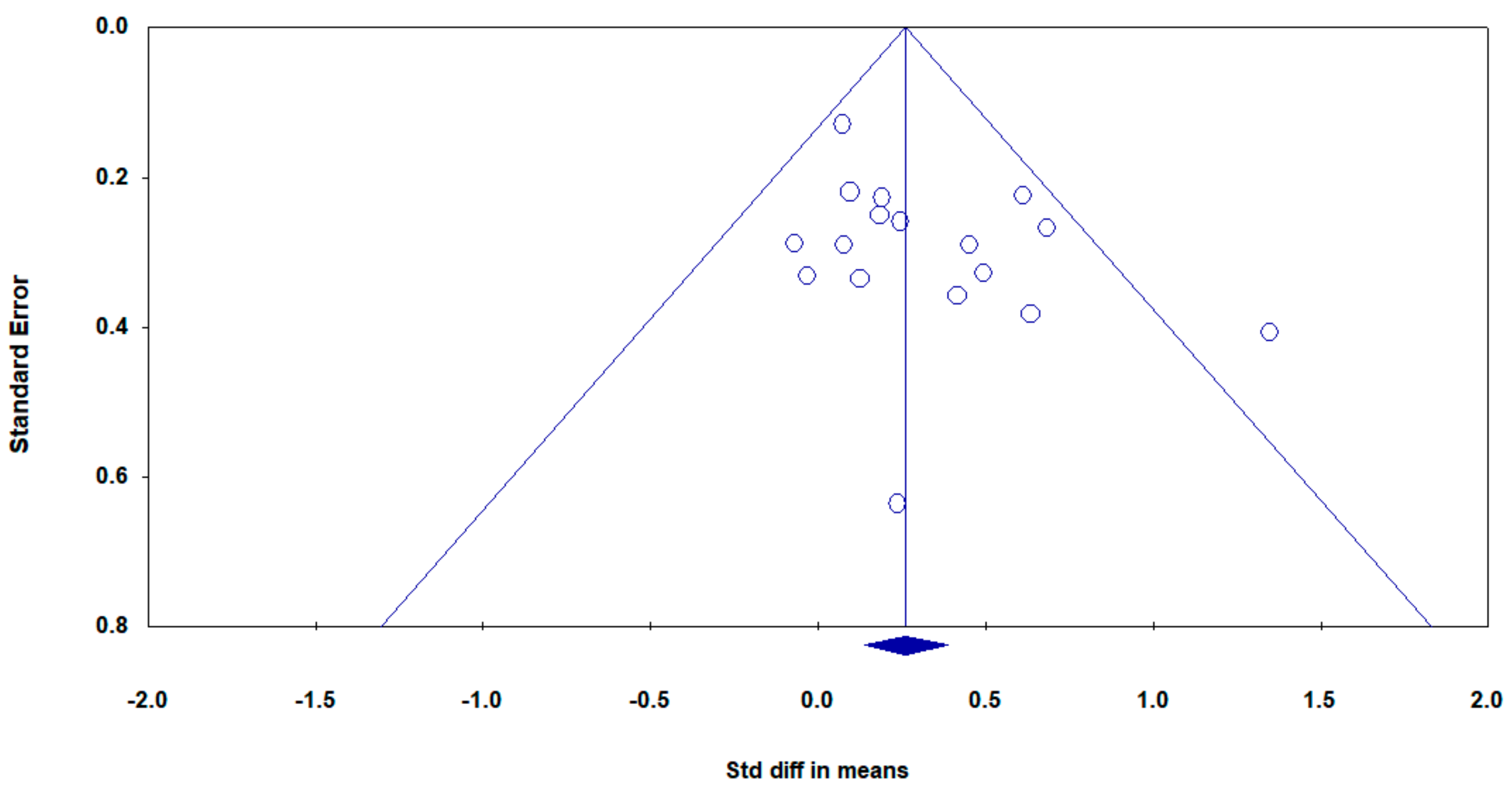
3.5. Sensitivity Analyses
3.6. Effect of Combined Interventions on Sub-Functions of EFs
3.7. Moderator Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Diamond, A. Executive functions. Ann. Rev. Psychol. 2013, 64, 135–168. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baddeley, A. Working Memory: Theories, Models, and Controversies. Ann. Rev. Psychol. 2012, 63, 1–29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Simpson, A.; Riggs, K.J.; Beck, S.R.; Gorniak, S.L.; Wu, Y.; Abbott, D.; Diamond, A. Refining the understanding of inhibitory processes: How response prepotency is created and overcome. Dev. Sci. 2011, 15, 62–73. [Google Scholar] [CrossRef] [Green Version]
- Miyake, A.; Friedman, N.P.; Emerson, M.J.; Witzki, A.H.; Howerter, A.; Wager, T.D. The Unity and Diversity of Executive Functions and Their Contributions to Complex “Frontal Lobe” Tasks: A Latent Variable Analysis. Cogn. Psychol. 2000, 41, 49–100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miyake, A.; Friedman, N.P. The Nature and Organization of Individual Differences in Executive Functions: Four General Conclusions. Curr. Dir. Psychol. Sci. 2012, 21, 8–14. [Google Scholar] [CrossRef]
- Macpherson, S.E.; Phillips, L.H.; Della, S.S. Age, executive function, and social decision making: A dorsolateral prefrontal theory of cognitive aging. Psychol. Aging 2002, 17, 598–609. [Google Scholar] [CrossRef]
- Elderkin-Thompson, V.; Ballmaier, M.; Hellemann, G.; Pham, D.; Kumar, A. Executive function and MRI prefrontal volumes among healthy older adults. Neuropsychology 2008, 22, 626–637. [Google Scholar] [CrossRef]
- Abe, T.; Fujii, K.; Hyodo, K.; Kitano, N.; Okura, T. Effects of acute exercise in the sitting position on executive function evaluated by the Stroop task in healthy older adults. J. Phys. Ther. Sci. 2018, 30, 609–613. [Google Scholar] [CrossRef]
- Carlson, M.C.; Saczynski, J.S.; Rebok, G.W.; Seeman, T.; Glass, T.A.; McGill, S.; Tielsch, J.M.; Frick, K.D.; Hill, J.; Fried, L.P. Exploring the effects of an “everyday” activity program on executive function and memory in older adults: Experience Corps. Gerontology 2008, 48, 793–801. [Google Scholar] [CrossRef] [Green Version]
- Bixby, W.R.; Spalding, T.W.; Haufler, A.J.; Deeny, S.P.; Mahlow, P.T.; Zimmerman, J.B.; Hatfield, B.D. The Unique Relation of Physical Activity to Executive Function in Older Men and Women. Med. Sci. Sports Exerc. 2007, 39, 1408–1416. [Google Scholar] [CrossRef]
- Best, J.R.; Nagamatsu, L.S.; Liu—Ambrose, T. Improvements to executive function during exercise training predict maintenance of physical activity over the following year. Front. Hum. Neurosci. 2014, 8, 353–362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Öhman, H.; Savikko, N.; Strandberg, T.E.; Pitkälä, K.H. Effect of Physical Exercise on Cognitive Performance in Older Adults with Mild Cognitive Impairment or Dementia: A Systematic Review. Dement. Geriatr. Cogn. Disord. 2014, 38, 347–365. [Google Scholar] [CrossRef] [PubMed]
- Toril, P.; Reales, J.M.; Mayas, J.; Ballesteros, S. Video Game Training Enhances Visuospatial Working Memory and Episodic Memory in Older Adults. Front. Hum. Neurosci. 2016, 10, 206–219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Toril, P.; Reales, J.M.; Ballesteros, S. Video game training enhances cognition of older adults: A meta-analytic study. Psychol. Aging 2014, 29, 706–716. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shatil, E.; Mikulecka, J.; Bellotti, F.; Bureš, V. Novel Television-Based Cognitive Training Improves Working Memory and Executive Function. PLoS ONE 2014, 9, e101472. [Google Scholar] [CrossRef] [Green Version]
- Haimov, I.; Shatil, E. Cognitive training improves sleep quality and cognitive function among older adults with insomnia. PLoS ONE 2013, 8, e61390. [Google Scholar] [CrossRef] [Green Version]
- Kim, M.-Y.; Kang, J.-W.; Park, S.-M.; Park, H.Y.; Park, J.-H. The Effect of Computer-Assisted Cognitive Training for Improving Cognitive Function in Older Adults: A Systematic Review. J. Korean Soc. Occup. Ther. 2016, 24, 81–94. [Google Scholar] [CrossRef]
- Chu-Man, L.; Chang, M.-Y.; Chu, M.-C. Effects of mahjong on the cognitive function of middle-aged and older people. Int. J. Geriatr. Psychiatry 2015, 30, 995–997. [Google Scholar] [CrossRef]
- Panphunpho, S.; Thavichachart, N.; Kritpet, T. Positive effects of Ska game practice on cognitive function among older adults. J. Med Assoc. Thail. Chotmaihet Thangphaet 2013, 96, 358–364. [Google Scholar]
- Zhang, F.; Kaufman, D. Cognitive benefits of older adults’ digital gameplay: A critical review. Gerontechnology 2016, 15, 3–16. [Google Scholar] [CrossRef]
- Youn, J.H.; Park, S.; Lee, J.Y.; Cho, S.-J.; Kim, J.; Ryu, S.-H. Cognitive Improvement in Older Adults with Mild Cognitive Impairment: Evidence from a Multi-Strategic Metamemory Training. J. Clin. Med. 2020, 9, 362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.Y.; Li, L.; Xiao, J.Q.; He, C.; Lyu, X.L.; Gao, L.; Yang, X.W.; Cui, X.G.; Fan, L.H. Cognitive Training in Older Adults with Mild Cognitive Impairment. Biomed. Environ. Sci. 2016, 29, 356–364. [Google Scholar] [PubMed]
- Hill, N.T.M.; Mowszowski, L.; Naismith, S.L.; Chadwick, V.L.; Valenzuela, M.; Lampit, A. Computerized Cognitive Training in Older Adults with Mild Cognitive Impairment or Dementia: A Systematic Review and Meta-Analysis. Am. J. Psychiatry 2017, 174, 329–340. [Google Scholar] [CrossRef] [PubMed]
- Eggenberger, P.; Schumacher, V.; Angst, M.; Theill, N.; de Bruin, E.D. Does multicomponent physical exercise with simultaneous cognitive training boost cognitive performance in older adults? A 6-month randomized controlled trial with a 1-year follow-up. Clin. Interv. Aging 2015, 10, 1335–1349. [Google Scholar] [CrossRef] [Green Version]
- Kalbe, E.; Roheger, M.; Paluszak, K.; Meyer, J.; Becker, J.; Fink, G.R.; Kukolja, J.; Rahn, A.; Szabados, F.; Wirth, B.; et al. Effects of a Cognitive Training With and Without Additional Physical Activity in Healthy Older Adults: A Follow-Up 1 Year After a Randomized Controlled Trial. Front. Aging Neurosci. 2018, 10, 407–420. [Google Scholar] [CrossRef]
- Barcelos, N.; Shah, N.; Cohen, K.; Hogan, M.J.; Mulkerrin, E.; Arciero, P.; Cohen, B.D.; Kramer, A.F.; Anderson-Hanley, C. Aerobic and Cognitive Exercise (ACE) Pilot Study for Older Adults: Executive Function Improves with Cognitive Challenge While Exergaming. J. Int. Neuropsychol. Soc. 2015, 21, 768–779. [Google Scholar] [CrossRef]
- Okamura, H.; Otani, M.; Shimoyama, N.; Fujii, T. Combined Exercise and Cognitive Training System for Dementia Patients: A Randomized Controlled Trial. Dement. Geriatr. Cogn. Disord. 2018, 45, 318–325. [Google Scholar] [CrossRef]
- Curlik, D.; Shors, T.J. Training your brain: Do mental and physical (MAP) training enhance cognition through the process of neurogenesis in the hippocampus? Neuropharmacology 2013, 64, 506–514. [Google Scholar] [CrossRef]
- Fissler, P.; Küster, O.; Schlee, W.; Kolassa, I.-T. Novelty Interventions to Enhance Broad Cognitive Abilities and Prevent Dementia: Synergistic approaches for the facilitation of positive plastic change. Prog. Brain Res. 2013, 207, 403–434. [Google Scholar] [CrossRef] [Green Version]
- Barban, F.; Annicchiarico, R.; Melideo, M.; Federici, A.; Lombardi, M.G.; Giuli, S.; Ricci, C.; Adriano, F.; Griffini, I.; Silvestri, M.; et al. Reducing Fall Risk with Combined Motor and Cognitive Training in Elderly Fallers. Brain Sci. 2017, 7, 19. [Google Scholar] [CrossRef] [Green Version]
- Law, L.L.; Barnett, F.; Yau, M.K.; Gray, M. Effects of combined cognitive and exercise interventions on cognition in older adults with and without cognitive impairment: A systematic review. Ageing Res. Rev. 2014, 15, 61–75. [Google Scholar] [CrossRef]
- Donnezan, L.C.; Perrot, A.; Belleville, S.; Bloch, F.; Kemoun, G. Effects of simultaneous aerobic and cognitive training on executive functions, cardiovascular fitness and functional abilities in older adults with mild cognitive impairment. Ment. Heal. Phys. Act. 2018, 15, 78–87. [Google Scholar] [CrossRef] [Green Version]
- Bo, W.; Lei, M.; Tao, S.; Jie, L.T.; Qian, L.; Lin, F.Q.; Ping, W.X. Effects of combined intervention of physical exercise and cognitive training on cognitive function in stroke survivors with vascular cognitive impairment: A randomized controlled trial. Clin. Rehabil. 2018, 33, 54–63. [Google Scholar] [CrossRef] [PubMed]
- Legault, C.; Jennings, J.M.; Katula, J.A.; Dagenbach, D.; Gaussoin, S.A.; Sink, K.M.; Rapp, S.R.; Rejeski, W.J.; Shumaker, S.A.; A Espeland, M. Designing clinical trials for assessing the effects of cognitive training and physical activity interventions on cognitive outcomes: The Seniors Health and Activity Research Program Pilot (SHARP-P) Study, a randomized controlled trial. BMC Geriatr. 2011, 11, 27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fossati, M.; Amati, F.; Painot, D.; Reiner, M.; Haenni, C.; Golay, A. Cognitive-behavioral therapy with simultaneous nutritional and physical activity education in obese patients with binge eating disorder. Eat. Weight. Disord. Stud. Anorexia Bulim. Obes. 2004, 9, 134–138. [Google Scholar] [CrossRef] [PubMed]
- Laatar, R.; Kachouri, H.; Borji, R.; Rebai, H.; Sahli, S. Combined physical-cognitive training enhances postural performances during daily life tasks in older adults. Exp. Gerontol. 2018, 107, 91–97. [Google Scholar] [CrossRef]
- Knaepen, K.; Goekint, M.; Heyman, E.; Meeusen, R. Neuroplasticity—Exercise-Induced Response of Peripheral Brain-Derived Neurotrophic Factor: A systematic review of experimental studies in human subjects. Sports Med. 2010, 40, 765–801. [Google Scholar] [CrossRef]
- Berryman, C.; Stanton, T.; Bowering, K.J.; Tabor, A.; McFarlane, A.; Moseley, G.L.; Moseley, G. Do people with chronic pain have impaired executive function? A meta-analytical review. Clin. Psychol. Rev. 2014, 34, 563–579. [Google Scholar] [CrossRef]
- Rahe, J.; Becker, J.; Fink, G.R.; Kessler, J.; Kukolja, J.; Rahn, A.; Rosen, J.B.; Szabados, F.; Wirth, B.; Kalbe, E. Cognitive training with and without additional physical activity in healthy older adults: Cognitive effects, neurobiological mechanisms, and prediction of training success. Front. Aging Neurosci. 2015, 7, 187–201. [Google Scholar] [CrossRef] [PubMed]
- Verhagen, A.P.; de Vet, H.C.; de Bie, R.A.; Kessels, A.G.; Boers, M.; Bouter, L.M.; Knipschild, P.G. The Delphi list: A criteria list for quality assessment of randomized clinical trials for conducting systematic reviews developed by Delphi consensus. J. Clin. Epidemiol. 1998, 51, 1235–1241. [Google Scholar] [CrossRef] [Green Version]
- Kayama, H.; Okamoto, K.; Nishiguchi, S.; Yamada, M.; Kuroda, T.; Aoyama, T.; Penichet, V.; Wolbring, G. Effect of a Kinect-Based Exercise Game on Improving Executive Cognitive Performance in Community-Dwelling Elderly: Case Control Study. J. Med. Internet Res. 2014, 16, e61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schoene, D.; Lord, S.R.; Delbaere, K.; Severino, C.; Davies, T.A.; Smith, S.T. A Randomized Controlled Pilot Study of Home-Based Step Training in Older People Using Videogame Technology. PLoS ONE 2013, 8, e57734. [Google Scholar] [CrossRef] [PubMed]
- Hiyamizu, M.; Morioka, S.; Shomoto, K.; Shimada, T. Effects of dual task balance training on dual task performance in elderly people: A randomized controlled trial. Clin. Rehabil. 2011, 26, 58–67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shatil, E. Does combined cognitive training and physical activity training enhance cognitive abilities more than either alone? A four-condition randomized controlled trial among healthy older adults. Front. Aging Neurosci. 2013, 5, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Lam, L.; Chau, R.C.; Wong, B.M.; Fung, A.W.T.; Tam, C.W.; Leung, G.T.Y.; Kwok, T.C.; Leung, T.Y.; Ng, S.; Chan, W.M. A 1-Year Randomized Controlled Trial Comparing Mind Body Exercise (Tai Chi) With Stretching and Toning Exercise on Cognitive Function in Older Chinese Adults at Risk of Cognitive Decline. J. Am. Med Dir. Assoc. 2012, 13, 568.e15–568.e20. [Google Scholar] [CrossRef]
- Sungkarat, S.; Boripuntakul, S.; Chattipakorn, N.; Watcharasaksilp, K.; Lord, S.R. Effects of Tai Chi on Cognition and Fall Risk in Older Adults with Mild Cognitive Impairment: A Randomized Controlled Trial. J. Am. Geriatr. Soc. 2016, 65, 721–727. [Google Scholar] [CrossRef]
- Singh, M.A.F.; Gates, N.J.; Saigal, N.; Wilson, G.C.; Meiklejohn, J.; Brodaty, H.; Wen, W.; Singh, M.A.F.; Baune, B.T.; Suo, C.; et al. The Study of Mental and Resistance Training (SMART) Study—Resistance Training and/or Cognitive Training in Mild Cognitive Impairment: A Randomized, Double-Blind, Double-Sham Controlled Trial. J. Am. Med. Dir. Assoc. 2014, 15, 873–880. [Google Scholar] [CrossRef]
- Park-Na, H.R.; Hiroyuki, S.; Kim-Jung, M.K. Combined Intervention of Physical Activity, Aerobic Exercise, and Cognitive Exercise Intervention to Prevent Cognitive Decline for Patients with Mild Cognitive Impairment: A Randomized Controlled Clinical Study. J. Clin. Med. 2019, 8, 940. [Google Scholar] [CrossRef] [Green Version]
- Maillot, P.; Perrot, A.; Hartley, A. Effects of interactive physical-activity video-game training on physical and cognitive function in older adults. Psychol. Aging 2012, 27, 589–600. [Google Scholar] [CrossRef] [Green Version]
- Barnes, D.E.; Santos-Modesitt, W.; Poelke, G.; Kramer, A.F.; Castro, C.; Middleton, L.E.; Yaffe, K. The mental activity and exercise (MAX) trial a randomized controlled trial to enhance cognitive function in older adults. JAMA Intern. Med. 2013, 173, 797–804. [Google Scholar] [CrossRef] [Green Version]
- Mrakic-Sposta, S.; di Santo, S.G.; Franchini, F.; Arlati, S.; Zangiacomi, A.; Greci, L.; Moretti, S.; Jesuthasan, N.; Marzorati, M.; Rizzo, G.; et al. Effects of Combined Physical and Cognitive Virtual Reality-Based Training on Cognitive Impairment and Oxidative Stress in MCI Patients: A Pilot Study. Front. Aging Neurosci. 2018, 10, 282–292. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brinke, L.F.T.; Best, J.R.; Chan, J.L.C.; Ghag, C.; Erickson, K.I.; Handy, T.C.; Liu, A.T. The Effects of Computerized Cognitive Training with and without Physical Exercise on Cognitive Function in Older Adults: An 8-week Randomized Controlled Trial. J. Gerontol. Ser. A Boil. Sci. Med. Sci. 2019, 75, 755–763. [Google Scholar] [CrossRef]
- Shah, T.M.; Verdile, G.; Sohrabi, H.R.; Campbell, A.; Putland, E.; Cheetham, C.; Dhaliwal, S.; Weinborn, M.; Maruff, P.; Darby, D.; et al. A combination of physical activity and computerized brain training improves verbal memory and increases cerebral glucose metabolism in the elderly. Transl. Psychiatry 2014, 4, e487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bae, S.; Lee, S.; Jung, S.; Makino, K.; Harada, K.; Harada, K.; Shinkai, Y.; Chiba, I.; Shimada, H. The effect of a multicomponent intervention to promote community activity on cognitive function in older adults with mild cognitive impairment: A randomized controlled trial. Complement. Ther. Med. 2019, 42, 164–169. [Google Scholar] [CrossRef]
- Nishiguchi, S.; Yamada, M.; Tanigawa, T.; Sekiyama, K.; Kawagoe, T.; Suzuki, M.; Yoshikawa, S.; Abe, N.; Otsuka, Y.; Nakai, R.; et al. A 12-Week Physical and Cognitive Exercise Program Can Improve Cognitive Function and Neural Efficiency in Community-Dwelling Older Adults: A Randomized Controlled Trial. J. Am. Geriatr. Soc. 2015, 63, 1355–1363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Linde, K.; Alfermann, D. Single versus combined cognitive and physical activity effects on fluid cognitive abilities of healthy older adults: A 4-month randomized controlled trial with follow-up. J. Aging Phys. Act. 2014, 22, 302–313. [Google Scholar] [CrossRef] [PubMed]
- Rahe, J.; Petrelli, A.; Kaesberg, S.; Fink, G.R.; Kessler, J.; Kalbe, E. Effects of cognitive training with additional physical activity compared to pure cognitive training in healthy older adults. Clin. Interv. Aging 10, 297–310. [CrossRef] [Green Version]
- Hagovska, M.; Nagyova, I. The transfer of skills from cognitive and physical training to activities of daily living: A randomised controlled study. Eur. J. Ageing 2016, 14, 133–142. [Google Scholar] [CrossRef]
- Zhu, X.; Yin, S.; Lang, M.; He, R.; Li, J. The more the better? A meta-analysis on effects of combined cognitive and physical intervention on cognition in healthy older adults. Ageing Res. Rev. 2016, 31, 67–79. [Google Scholar] [CrossRef]
- Gheysen, F.; Poppe, L.; Desmet, A.; Swinnen, S.; Cardon, G.; de Bourdeaudhuij, I.; Chastin, S.; Fias, W. Physical activity to improve cognition in older adults: Can physical activity programs enriched with cognitive challenges enhance the effects? A systematic review and meta-analysis. Int. J. Behav. Nutr. Phys. Act. 2018, 15, 63. [Google Scholar] [CrossRef] [Green Version]
- Walsh, J.N.; Manor, B.; Hausdorff, J.; Novak, V.; Lipsitz, L.; Gow, B.; Macklin, E.A.; Peng, C.-K.; Wayne, P.M. Impact of Short- and Long-term Tai Chi Mind-Body Exercise Training on Cognitive Function in Healthy Adults: Results from a Hybrid Observational Study and Randomized Trial. Glob. Adv. Heal. Med. 2015, 4, 38–48. [Google Scholar] [CrossRef] [Green Version]
- Holtzer, R.; Shuman, M.; Mahoney, J.R.; Lipton, R.; Verghese, J. Cognitive Fatigue Defined in the Context of Attention Networks. Aging Neuropsychol. Cogn. 2010, 18, 108–128. [Google Scholar] [CrossRef]
- Gutchess, A. Plasticity of the aging brain: New directions in cognitive neuroscience. Science 2014, 346, 579–582. [Google Scholar] [CrossRef]
- Hedge, C.; Powell, G.; Sumner, P. The mapping between transformed reaction time costs and models of processing in aging and cognition. Psychol. Aging 2018, 33, 1093–1104. [Google Scholar] [CrossRef]
- Rabbitt, P. Do “Frontal Tests” Measure Executive Function? Issues of Assessment and Evidence from Fluency Tests. In Methodology of Frontal and Executive Function; Routledge: London, UK, 2004; pp. 193–215. [Google Scholar]
- Northey, J.M.; Cherbuin, N.; Pumpa, K.L.; Smee, D.J.; Rattray, B. Exercise interventions for cognitive function in adults older than 50: A systematic review with meta-analysis. Br. J. Sports Med. 2017, 52, 154–160. [Google Scholar] [CrossRef]
- Kramer, A.F.; Colcombe, S.J. Fitness Effects on the Cognitive Function of Older Adults: A Meta-Analytic Study—Revisited. Perspect. Psychol. Sci. 2018, 13, 213–217. [Google Scholar] [CrossRef] [Green Version]
- Friedman, N.P.; Miyake, A.; Altamirano, L.J.; Corley, R.P.; Young, S.E.; Rhea, S.A.; Hewitt, J.K. Stability and change in executive function abilities from late adolescence to early adulthood: A longitudinal twin study. Dev. Psychol. 2016, 52, 326–340. [Google Scholar] [CrossRef]
- McEwen, S.C.; Siddarth, P.; Abedelsater, B.; Kim, Y.; Mui, W.; Wu, P.; Emerson, N.D.; Lee, J.; Greenberg, S.; Shelton, T.; et al. Simultaneous Aerobic Exercise and Memory Training Program in Older Adults with Subjective Memory Impairments. J. Alzheimer’s Dis. 2018, 62, 795–806. [Google Scholar] [CrossRef] [Green Version]




| Characteristics | Intervention Methods | EFs Measure Tasks | Control Group Activities | Study Quality | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Study | Sample Size | Age (Mean) | Cognitive Status | Comparison | Cognitive Intervention | Physical Intervention | Combination Mode | Intervention Plan | |||
| Hiyamizu (2012) | 36 | >65 (71.6) | Health | CPI vs. CG | memory, visual search, verbal fluency training | Balance exercise | Simultaneous | 60 min/session, 2 sessions/week, 12 weeks | Stroop Task, TMT A, TMT B | NO | 8 |
| Legault (2011) | 73 | 70–85 (76.4) | Health | CPI vs. PI vs. CI vs. CG | Memory training | Aerobic and flexibility exercises | Separate | 50–150 min/session, 3 sessions/week, 24 weeks | 2-Back Test, Flanker Task, Task Switching, TMT A, TMT B, Self-Ordered Pointing Task | NO | 9 |
| Maillot (2012) | 30 | 65–78 (73.5) | Health | CPI vs. CG | Nintendo Wii game | Wii Sports, Wii Fit, Mario & Sonic on Olympic Games. | Simultaneous | 60 min/session, 2 sessions/week, 12 weeks | TMT A, TMT B; Stroop Test, Letter Sets Test, Matrix Reasoning Test, Digit-Symbol Substitution | NO | 9 |
| Barnes (2013) | 126 | ≥65 (73.4) | Health | CPI vs. CI vs. PI vs. CG | Visual and auditory processing speed training | Aerobic exercise | Separate | 60 min/session, 6 sessions/week, 12 weeks | TMT B, Erikson Flanker Test | Health video education activities | 11 |
| Shatil (2013) | 122 | 65–93 (76.8) | Health | CPI vs. CI vs. PI vs. CG | Cognitive game (CogniFit) | Aerobic, strength, and flexibility exercises | Separate | 40–45 min/session, 6 sessions/week, 16 weeks | CogniFit neuropsycholog-ical evaluation | Join a book club | 7 |
| Nishiguchi (2015) | 48 | ≥60 (73.3) | Health | CPI vs. CG | verbal fluency, cognitive-motor training | Step exercises, stretching, and strength | Simultaneous | 90 min/session, 1 session/week, 12 weeks | TMT A, TMT B | NO | 9 |
| Rahe (2015)a | 68 | 50–85 (68.4) | Health | CPI vs. CI | Working memory, fluency, inhibition, planning training | Strength, flexibility, coordination, and endurance exercises | Separate | 90 min/session, 2 sessions/week, 7 weeks | Regensburger Wort Flüssigkeits-Test, Stroop Test, WAIS-II (DSB) | N/C | 8 |
| Rahe (2015)b | 30 | 50–85 (66.7) | Health | CPI vs. CI | memory, attention, EFs training | Strength, flexibility, and balance exercises | Separate | 90 min/session, 2 sessions/week, 7 weeks | Complex Figure Test, TMT A, TMT B | N/C | 9 |
| Kalbe (2018) | 55 | 50–85 (68.1) | Health | CPI vs. CI | Memory, attention, EFs training | Strength, flexibility, and balance exercises | Separate | 90 min/session, 2 sessions/week, 7 weeks | Stroop Test, WAIS-II (DSB),Regensburger Wort Flüssigkeits-Test | N/C | 10 |
| Lam (2012) | 261 | ≥65 (77.8) | MCI | CPI vs. CG | Memory, attention training | Tai Chi | Simultaneous | 30 min/session, 3 sessions/week, 12 months | TMT B | Stretching exercise | 9 |
| Hagovska (2017) | 80 | ≥65 (67) | MCI | CPI vs. PI | Attention, memory, EFs training | Different forms of walking | Simultaneous | 30 min/session, 2 sessions/week, 10 weeks | Stroop Test, TMT A | N/C | 10 |
| Sungkarat (2017) | 66 | ≥60 (67.9) | MCI | CPI vs. CG | Memory, attention training | Tai Chi | Simultaneous | 50 min/session, 3 sessions/week, 12 weeks | TMT A, TMT B | Education activities | 9 |
| Mrakic (2018) | 10 | ≥65 (73.3) | MCI | CPI vs. CG | Memory, visuospatial ability training | Aerobic exercise | Simultaneous | 40–45 min/session, 3 sessions/week, 6 weeks | TMT A | NO | 8 |
| Singh (2014) | 86 | 55–89 (70.1) | MCI | CPI vs. CI vs. PI vs. CG | Memory, attention, EFs, cognitive processing speed training | Resistance training | Separate | 60–100 min/session, 2 sessions/week, 24 weeks | WAIS-III, Controlled Oral Words Association Test | Stretching and education activities | 12 |
| Park (2019) | 49 | >60 (71.6) | MCI | CPI vs. CG | Phrase play, memory play, arithmetic training | Aerobic, balance, stretching | Simultaneous | 110 min/session, 2 sessions/week, 24 weeks | Symbol–Digit Substitution Test | NO | 10 |
| Kayama (2014) | 41 | ≥65 | Health | CPI vs. CG | Placement problem solving (Sudoku) | Tai chi | Simultaneous | 75–80 min/session, 1 session/week, 12 weeks | TMT A, TMT B | Strength and balance training | 7 |
| Brinke (2020) | 124 | 65–85 (72.4) | Health | CPI vs. CI vs. CG | Cognitive game (Fit Brains) | Brisk walking | Separate | 60 min/session, 3 sessions/week, 8 weeks | DCCS, Flanker Test, TMT A, TMT B, Stroop Test | Regular activities | 9 |
| Schoene (2013) | 32 | ≥65 (77.9) | Health | CPI vs. CG | Attention training | Step training | Simultaneous | 15–20 min/session, 2–3 sessions/week, 8 weeks | TMT A, TMT B | Regular activities | 10 |
| Linde (2014) | 55 | 60–75 (67.1) | Health | CPI vs. CI vs. PI vs. CG | Short-term memory, information processing speed, logical reasoning training | Aerobic, endurance, and strength training | Separate | 60–90 min/session, 2 sessions/week, 16 weeks | Leistungs-Prüf-System | NO | 11 |
| Shah (2014) | 172 | 60–85 (67.4) | Health | CPI vs. CI vs. PI vs. CG | Auditory-based BFP, Visual-based IP | Walking, strength training | Separate | 60 min/session, 10 sessions/week, 16 weeks | Groton Maze Learning, Controlled Oral Word Association Test | NO | 9 |
| Bae (2019) | 83 | ≥65 (75.9) | MCI | CPI vs. CG | “KENKOJISEICHI” system | “KENKOJISEICHI” system | Separate | 90 min/session, 2 sessions/week, 24 weeks | TMT B | NO | 9 |
| Comparison | Outcomes | No. of Studies | SMD | 95% Confidence Interval | I2 (%) | Homogeneity Test | ||
|---|---|---|---|---|---|---|---|---|
| Q | df | p | ||||||
| Combined intervention versus Control group | Response inhibition | 8 | 0.29 ** | 0.10 to 0.48 | 65.6 | 20.35 | 7 | 0.005 |
| Set-shifting | 13 | 0.24 ** | 0.10 to 0.37 | 34.03 | 18.19 | 12 | 0.11 | |
| Complex EFs | 8 | 0.34 ** | 0.13 to 0.56 | 63.67 | 19.27 | 7 | 0.007 | |
| Overall | 29 | 0.27 ** | 0.17 to 0.37 | 52.18 | 58.55 | 28 | 0.001 | |
| Combined intervention versus Cognitive intervention | Response inhibition | 9 | 0.09 | −0.09 to 0.26 | 0 | 5.16 | 8 | 0.74 |
| Set-shifting | 5 | 0.04 * | 0.04 to 0.50 | 0 | 1.15 | 4 | 0.87 | |
| Complex EFs | 7 | 0.07 | −0.14 to 0.28 | 0 | 2.2 | 6 | 0.9 | |
| Overall | 21 | 0.13 * | 0.01 to 0.25 | 0 | 10.6 | 20 | 0.96 | |
| Combined intervention versus Physical intervention | Response inhibition | 6 | 0.06 | −0.15 to 0.26 | 33.03 | 7.47 | 5 | 0.19 |
| Set-shifting | 4 | 0.26* | 0.01 to 0.52 | 0 | 2.04 | 3 | 0.56 | |
| Complex EFs | 5 | 0.21 | −0.04 to 0.46 | 0 | 3.56 | 4 | 0.47 | |
| Overall | 15 | 0.16 | 0.02 to 0.29 | 5.43 | 14.8 | 14 | 0.39 | |
| Moderator | Level | No. of Studies | SMD | 95% CI | I2 | Homogeneity Test | ||
|---|---|---|---|---|---|---|---|---|
| Q | df | p | ||||||
| Mode of combination | Sequential | 9 | 0.27 ** | 0.10 to 0.44 | 0 | 0.01 | 1 | 0.94 |
| Simultaneous | 8 | 0.26 ** | 0.08 to 0.44 | 49.53 | ||||
| Cognitive status | Healthy | 11 | 0.33 ** | 0.16 to 0.50 | 23.28 | 1.22 | 1 | 0.27 |
| MCI | 6 | 0.19 * | 0.01 to 0.37 | 6.74 | ||||
| Control group | Active | 8 | 0.27 ** | 0.11 to 0.43 | 14.52 | 0.03 | 1 | 0.86 |
| Passive | 9 | 0.25 * | 0.05 to 0.45 | 29.78 | ||||
| Intervention length | Long (≥24 weeks) | 5 | 0.11 | −0.07 to 0.29 | 0 | 4.92 | 2 | 0.09 |
| Medium (12–23 weeks) | 8 | 0.37 ** | 0.17 to 0.56 | 39 | ||||
| Short (<12 weeks) | 4 | 0.44 * | 0.12 to 0.75 | 0 | ||||
| Frequency | High (>3 sessions/week) | 3 | 0.21 | −0.07 to 0.48 | 0 | 0.2 | 1 | 0.66 |
| Low (≤3 sessions/week) | 14 | 0.28 * | 0.14 to 0.41 | 32.92 | ||||
| Session duration | Long (>60 min) | 7 | 0.20 | −0.02 to 0.41 | 0 | 4.13 | 2 | 0.13 |
| Medium (>30 to ≤60 min) | 8 | 0.41 ** | 0.22 to 0.61 | 30.47 | ||||
| Short (≤30 min) | 2 | 0.11 | −0.13 to 0.35 | 0 | ||||
| Study quality | High (≥9 scores) | 13 | 0.26 ** | 0.13 to 0.39 | 36.65 | 0.01 | 1 | 0.91 |
| Low (<9 scores) | 4 | 0.28 | −0.05 to 0.61 | 0 | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, W.; Zang, M.; Klich, S.; Kawczyński, A.; Smoter, M.; Wang, B. Effect of Combined Physical and Cognitive Interventions on Executive Functions in Older Adults: A Meta-Analysis of Outcomes. Int. J. Environ. Res. Public Health 2020, 17, 6166. https://doi.org/10.3390/ijerph17176166
Guo W, Zang M, Klich S, Kawczyński A, Smoter M, Wang B. Effect of Combined Physical and Cognitive Interventions on Executive Functions in Older Adults: A Meta-Analysis of Outcomes. International Journal of Environmental Research and Public Health. 2020; 17(17):6166. https://doi.org/10.3390/ijerph17176166
Chicago/Turabian StyleGuo, Wei, Ming Zang, Sebastian Klich, Adam Kawczyński, Małgorzata Smoter, and Biye Wang. 2020. "Effect of Combined Physical and Cognitive Interventions on Executive Functions in Older Adults: A Meta-Analysis of Outcomes" International Journal of Environmental Research and Public Health 17, no. 17: 6166. https://doi.org/10.3390/ijerph17176166
APA StyleGuo, W., Zang, M., Klich, S., Kawczyński, A., Smoter, M., & Wang, B. (2020). Effect of Combined Physical and Cognitive Interventions on Executive Functions in Older Adults: A Meta-Analysis of Outcomes. International Journal of Environmental Research and Public Health, 17(17), 6166. https://doi.org/10.3390/ijerph17176166







