Identification of Nontuberculous Mycobacteria in Drinking Water in Cali, Colombia
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jarlier, V.; Nikaido, H. Mycobacterial cell wall: Structure and role in natural resistance to antibiotics. FEMS Microbiol. Lett. 1994, 123, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Faria, S.; Joao, I.; Jordao, L. General Overview on Nontuberculous Mycobacteria, Biofilms, and Human Infection. J. Pathog. 2015, 2015, 809014. [Google Scholar] [CrossRef]
- Gebert, M.J.; Delgado-Baquerizo, M.; Oliverio, A.M.; Webster, T.M.; Nichols, L.M.; Honda, J.R.; Chan, E.D.; Adjemian, J.; Dunn, R.R.; Fierer, N. Ecological Analyses of Mycobacteria in Showerhead Biofilms and Their Relevance to Human Health. MBio 2018, 9, e01614-18. [Google Scholar] [CrossRef]
- Parte, A.C. LPSN—List of Prokaryotic names with Standing in Nomenclature (bacterio.net), 20 years on. Int. J. Syst. Evol. Microbiol. 2018, 68, 1825–1829. [Google Scholar] [CrossRef]
- Tortoli, E. Impact of genotypic studies on mycobacterial taxonomy: The new mycobacteria of the 1990s. Clin. Microbiol. Rev. 2003, 16, 319–354. [Google Scholar] [CrossRef] [PubMed]
- Griffith, D.E.; Aksamit, T.; Brown-Elliott, B.A.; Catanzaro, A.; Daley, C.; Gordin, F.; Holland, S.M.; Horsburgh, R.; Huitt, G.; Iademarco, M.F.; et al. An official ATS/IDSA statement: Diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am. J. Respir. Crit. Care Med. 2007, 175, 367–416. [Google Scholar] [CrossRef]
- To, K.; Cao, R.; Yegiazaryan, A.; Owens, J.; Venketaraman, V. General Overview of Nontuberculous Mycobacteria Opportunistic Pathogens: Mycobacterium avium and Mycobacterium abscessus. J. Clin. Med. 2020, 9, 2541. [Google Scholar] [CrossRef]
- Prince, D.S.; Peterson, D.D.; Steiner, R.M.; Gottlieb, J.E.; Scott, R.; Israel, H.L.; Figueroa, W.G.; Fish, J.E. Infection with Mycobacterium avium complex in patients without predisposing conditions. N. Engl. J. Med. 1989, 321, 863–868. [Google Scholar] [CrossRef]
- Henry, M.T.; Inamdar, L.; O’Riordain, D.; Schweiger, M.; Watson, J.P. Nontuberculous mycobacteria in non-HIV patients: Epidemiology, treatment and response. Eur. Respir. J. 2004, 23, 741–746. [Google Scholar] [CrossRef]
- Donohue, M.J. Increasing nontuberculous mycobacteria reporting rates and species diversity identified in clinical laboratory reports. BMC Infect. Dis. 2018, 18, 163. [Google Scholar] [CrossRef] [PubMed]
- Brode, S.K.; Daley, C.L.; Marras, T.K. The epidemiologic relationship between tuberculosis and non-tuberculous mycobacterial disease: A systematic review. Int. J. Tuberc. Lung Dis. 2014, 18, 1370–1377. [Google Scholar] [CrossRef] [PubMed]
- van Ingen, J.; Bendien, S.A.; de Lange, W.C.; Hoefsloot, W.; Dekhuijzen, P.N.; Boeree, M.J.; van Soolingen, D. Clinical relevance of non-tuberculous mycobacteria isolated in the Nijmegen-Arnhem region, The Netherlands. Thorax 2009, 64, 502–506. [Google Scholar] [CrossRef] [PubMed]
- Adjemian, J.; Olivier, K.N.; Seitz, A.E.; Holland, S.M.; Prevots, D.R. Prevalence of nontuberculous mycobacterial lung disease in U.S. Medicare beneficiaries. Am. J. Respir. Crit. Care Med. 2012, 185, 881–886. [Google Scholar] [CrossRef]
- Zweijpfenning, S.M.H.; Ingen, J.V.; Hoefsloot, W. Geographic Distribution of Nontuberculous Mycobacteria Isolated from Clinical Specimens: A Systematic Review. Semin. Respir. Crit. Care Med. 2018, 39, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Schiff, H.F.; Jones, S.; Achaiah, A.; Pereira, A.; Stait, G.; Green, B. Clinical relevance of non-tuberculous mycobacteria isolated from respiratory specimens: Seven year experience in a UK hospital. Sci. Rep. 2019, 9, 1730. [Google Scholar] [CrossRef] [PubMed]
- Prevots, D.R.; Loddenkemper, R.; Sotgiu, G.; Migliori, G.B. Nontuberculous mycobacterial pulmonary disease: An increasing burden with substantial costs. Eur. Respir. J. 2017, 49, 1700374. [Google Scholar] [CrossRef]
- Rivero-Lezcano, O.M.; González-Cortés, C.; Mirsaeidi, M. The unexplained increase of nontuberculous mycobacteriosis. Int. J. Mycobacteriol. 2019, 8, 1–6. [Google Scholar] [CrossRef]
- Honda, J.R.; Virdi, R.; Chan, E.D. Global Environmental Nontuberculous Mycobacteria and Their Contemporaneous Man-Made and Natural Niches. Front. Microbiol. 2018, 9, 2029. [Google Scholar] [CrossRef]
- Chou, M.P.; Clements, A.C.; Thomson, R.M. A spatial epidemiological analysis of nontuberculous mycobacterial infections in Queensland, Australia. BMC Infect. Dis. 2014, 14, 279. [Google Scholar] [CrossRef] [PubMed]
- Correa, N.E.; Cataño, J.C.; Mejía, G.I.; Realpe, T.; Orozco, B.; Estrada, S.; Vélez, A.; Vélez, L.; Barón, P.; Guzmán, A.; et al. Outbreak of mesotherapy-associated cutaneous infections caused by Mycobacterium chelonae in Colombia. Jpn. J. Infect. Dis. 2010, 63, 143–145. [Google Scholar]
- Llerena, C.; Valbuena, A.; Zabaleta, A.P. Mycobacterioses identified in the National Reference Laboratory of Colombia from 2012 to 2016. Biomedica 2018, 38, 87–94. [Google Scholar] [CrossRef]
- Delgado, L.E.; Escobar, D.R.; Hoyos, D.M.; Luna, L.; Pacheco-Lopez, R.; Ferro, B. Nontuberculous mycobacteria in patients registered in a tuberculosis control program in Southwestern Colombia, 2014–2017. Interdiscip. J. Epidemiol. Public Health 2019, 2, e–017. [Google Scholar] [CrossRef]
- Tichenor, W.S.; Thurlow, J.; McNulty, S.; Brown-Elliott, B.A.; Wallace, R.J., Jr.; Falkinham, J.O., 3rd. Nontuberculous Mycobacteria in household plumbing as possible cause of chronic rhinosinusitis. Emerg. Infect. Dis. 2012, 18, 1612–1617. [Google Scholar] [CrossRef] [PubMed]
- Falkinham, J.O., 3rd. Nontuberculous mycobacteria from household plumbing of patients with nontuberculous mycobacteria disease. Emerg. Infect. Dis. 2011, 17, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Adékambi, T.; Drancourt, M.; Raoult, D. The rpoB gene as a tool for clinical microbiologists. Trends Microbiol. 2009, 17, 37–45. [Google Scholar] [CrossRef] [PubMed]
- CLSI. Performance Standards for Susceptibility Testing of Mycobacteria, Nocardia spp., and Other Aerobic Actinomycetes, 1st ed.; CLSI supplement M62; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2018. [Google Scholar]
- Ministerio de la protección social, Ministerio de Ambiente, Vivienda y Desarrollo Territorial. Resolución Número 2115. República de Colombia. 2007. Available online: https://www.minambiente.gov.co/images/GestionIntegraldelRecursoHidrico/pdf/normativa/Res_2115_de_2007.pdf (accessed on 16 February 2021).
- Perez-Martinez, I.; Aguilar-Ayala, D.A.; Fernandez-Rendon, E.; Carrillo-Sanchez, A.K.; Helguera-Repetto, A.C.; Rivera-Gutierrez, S.; Estrada-Garcia, T.; Cerna-Cortes, J.F.; Gonzalez, Y.M.J.A. Occurrence of potentially pathogenic nontuberculous mycobacteria in Mexican household potable water: A pilot study. BMC Res. Notes 2013, 6, 531. [Google Scholar] [CrossRef]
- Tortone, C.A.; Oriani, D.S.; Staskevich, A.S.; Oriani, A.S.; Gino, L.M.; Marfil, M.J.; Nava Vargas, A.; Gioffré, A.K.; Zumárraga, M.J. Species diversity of non-tuberculous mycobacteria isolated from aquatic environments of General Pico city, Province of La Pampa (Argentina). Rev. Argent. Microbiol. 2019, 51, 259–267. [Google Scholar] [CrossRef]
- Oriani, A.S.; Marfil, M.J.; Zumárraga, M.J.; Baldini, M.D. Prevalence and species diversity of nontuberculous mycobacteria in drinking water supply system of Bahía Blanca City, Argentina. Int J. Mycobacteriol. 2019, 8, 138–145. [Google Scholar] [CrossRef]
- Donohue, M.J.; Mistry, J.H.; Donohue, J.M.; O’Connell, K.; King, D.; Byran, J.; Covert, T.; Pfaller, S. Increased Frequency of Nontuberculous Mycobacteria Detection at Potable Water Taps within the United States. Environ. Sci. Technol. 2015, 49, 6127–6133. [Google Scholar] [CrossRef]
- Loret, J.F.; Dumoutier, N. Non-tuberculous mycobacteria in drinking water systems: A review of prevalence data and control means. Int. J. Hyg. Environ. Health 2019, 222, 628–634. [Google Scholar] [CrossRef] [PubMed]
- Thomson, R.M.; Carter, R.; Tolson, C.; Coulter, C.; Huygens, F.; Hargreaves, M. Factors associated with the isolation of Nontuberculous mycobacteria (NTM) from a large municipal water system in Brisbane, Australia. BMC Microbiol. 2013, 13, 89. [Google Scholar] [CrossRef]
- Hussein, Z.; Landt, O.; Wirths, B.; Wellinghausen, N. Detection of non-tuberculous mycobacteria in hospital water by culture and molecular methods. Int. J. Med. Microbiol. 2009, 299, 281–290. [Google Scholar] [CrossRef]
- Le Dantec, C.; Duguet, J.P.; Montiel, A.; Dumoutier, N.; Dubrou, S.; Vincent, V. Occurrence of mycobacteria in water treatment lines and in water distribution systems. Appl. Environ. Microbiol. 2002, 68, 5318–5325. [Google Scholar] [CrossRef]
- Mi, Z.; Dai, Y.; Xie, S.; Chen, C.; Zhang, X. Impact of disinfection on drinking water biofilm bacterial community. J. Environ. Sci. 2015, 37, 200–205. [Google Scholar] [CrossRef]
- Norton, C.D.; LeChevallier, M.W. A pilot study of bacteriological population changes through potable water treatment and distribution. Appl. Environ. Microbiol. 2000, 66, 268–276. [Google Scholar] [CrossRef]
- Norton, C.D.; LeChevallier, M.W.; Falkinham, J.O., 3rd. Survival of Mycobacterium avium in a model distribution system. Water Res. 2004, 38, 1457–1466. [Google Scholar] [CrossRef]
- LeChevallier, M.W.; Lowry, C.D.; Lee, R.G.; Gibbon, D.L. Examining the Relationship Between Iron Corrosion and the Disinfection of Biofilm Bacteria. J. Am. Water Work. Assoc. 1993, 85, 111–123. [Google Scholar] [CrossRef]
- Hoefsloot, W.; van Ingen, J.; Andrejak, C.; Angeby, K.; Bauriaud, R.; Bemer, P.; Beylis, N.; Boeree, M.J.; Cacho, J.; Chihota, V.; et al. The geographic diversity of nontuberculous mycobacteria isolated from pulmonary samples: An NTM-NET collaborative study. Eur. Respir. J. 2013, 42, 1604–1613. [Google Scholar] [CrossRef]
- Gupta, N.; Mittal, A.; Niyas, V.K.M.; Banerjee, S.; Ray, Y.; Kodan, P.; Malla, S.; Khot, W.; Fazal, F.; Singh, B.K.; et al. Nontuberculous mycobacteria: A report of eighteen cases from a tertiary care center in India. Lung India 2020, 37, 495–500. [Google Scholar] [CrossRef]
- Gonzalez-Santiago, T.M.; Drage, L.A. Nontuberculous Mycobacteria: Skin and Soft Tissue Infections. Dermatol. Clin. 2015, 33, 563–577. [Google Scholar] [CrossRef]
- Sharma, K.; Gautam, N.; Sharma, M.; Dogra, M.; Bajgai, P.; Tigari, B.; Sharma, A.; Gupta, V.; Sharma, S.P.; Singh, R. Ocular mycobacteriosis-dual infection of M. tuberculosis complex with M. fortuitum and M. bovis. J. Ophthalmic Inflamm. Infect. 2017, 7, 2. [Google Scholar] [CrossRef]
- Kurokawa, K.; Harada, N.; Sasano, H.; Takagi, H.; Takei, S.; Nakamura, A.; Kamada, K.; Yoshida, A.; Kikuchi, K.; Takahashi, K. Pulmonary infection due to fluoroquinolone-resistant Mycolicibacterium fortuitum: A case report. BMC Infect. Dis. 2020, 20, 866. [Google Scholar] [CrossRef] [PubMed]
- Okamori, S.; Asakura, T.; Nishimura, T.; Tamizu, E.; Ishii, M.; Yoshida, M.; Fukano, H.; Hayashi, Y.; Fujita, M.; Hoshino, Y.; et al. Natural history of Mycobacterium fortuitum pulmonary infection presenting with migratory infiltrates: A case report with microbiological analysis. BMC Infect. Dis. 2018, 18, 1. [Google Scholar] [CrossRef]
- Pradier, M.; Boucher, A.; Robineau, O.; Chachaty, E.; Meybeck, A.; Senneville, E. Mycobacterium mucogenicum bacteremia: Major role of clinical microbiologists. BMC Infect. Dis. 2018, 18, 646. [Google Scholar] [CrossRef]
- Otchere, I.D.; Asante-Poku, A.; Osei-Wusu, S.; Aboagye, S.Y.; Yeboah-Manu, D. Isolation and characterization of nontuberculous mycobacteria from patients with pulmonary tuberculosis in Ghana. Int. J. Mycobacteriol. 2017, 6, 70–75. [Google Scholar] [CrossRef]
- Adékambi, T.; Foucault, C.; La Scola, B.; Drancourt, M. Report of two fatal cases of Mycobacterium mucogenicum central nervous system infection in immunocompetent patients. J. Clin. Microbiol. 2006, 44, 837–840. [Google Scholar] [CrossRef]
- Pereira, S.G.; Alarico, S.; Tiago, I.; Reis, D.; Nunes-Costa, D.; Cardoso, O.; Maranha, A.; Empadinhas, N. Studies of antimicrobial resistance in rare mycobacteria from a nosocomial environment. BMC Microbiol. 2019, 19, 62. [Google Scholar] [CrossRef]
- Ben Salah, I.; Adékambi, T.; Drancourt, M. Mycobacterium phocaicum in therapy pool water. Int. J. Hyg. Environ. Health 2009, 212, 439–444. [Google Scholar] [CrossRef]
- Wethasinghe, J.; Hotu, S.; Taylor, S.; Anderson, G.; Wong, C. Mycobacterium phocaicum and Mycobacterium avium-intracellulare in a patient with hot tub lung. Respirol. Case Rep. 2015, 3, 19–21. [Google Scholar] [CrossRef][Green Version]
- Cooksey, R.C.; Jhung, M.A.; Yakrus, M.A.; Butler, W.R.; Adékambi, T.; Morlock, G.P.; Williams, M.; Shams, A.M.; Jensen, B.J.; Morey, R.E.; et al. Multiphasic approach reveals genetic diversity of environmental and patient isolates of Mycobacterium mucogenicum and Mycobacterium phocaicum associated with an outbreak of bacteremias at a Texas hospital. Appl. Environ. Microbiol. 2008, 74, 2480–2487. [Google Scholar] [CrossRef]
- Zheng, H.W.; Pang, Y.; He, G.X.; Song, Y.Y.; Zhao, Y.L. Antimicrobial Susceptibility Testing and Molecular Characterization of Mycobacterium fortuitum Isolates in China. Biomed. Environ. Sci. 2017, 30, 376–379. [Google Scholar] [CrossRef]
- Kim, S.Y.; Moon, S.M.; Jhun, B.W.; Kwon, O.J.; Huh, H.J.; Lee, N.Y.; Lee, S.H.; Shin, S.J.; Kasperbauer, S.H.; Huitt, G.A.; et al. Species Distribution and Macrolide Susceptibility of Mycobacterium fortuitum Complex Clinical Isolates. Antimicrob. Agents Chemother. 2019, 63. [Google Scholar] [CrossRef]
- Pérez-Vidal, A. Estrategias de Implementación de los Planes de Seguridad del Agua para la Gestión del Riesgo en Sistemas de Abastecimiento de Agua Potable. Universidad del Valle 2013. Available online: https://opac.univalle.edu.co/cgi-olib/?infile=details.glu&loid=834909&rs=7139723&hitno=2 (accessed on 4 March 2021).
- Parques Nacionales Naturales de Colombia. Operación Farallones Acciones Interinstitucionales en Implementación para Controlar la Minería Ilegal de Oro 2015. Available online: https://www.parquesnacionales.gov.co/portal/es/operacion-farallones-acciones-interinstitucionales-en-implementacion-para-controlar-la-mineria-ilegal-de-oro-en-el-parque-nacional-natural-farallones-de-cali/ (accessed on 4 March 2021).
- Pérez-Vidal, A.; Escobar-Rivera, J.C.; Torres-Lozada, P. Development and implementation of a water-safety plan for drinking-water supply system of Cali, Colombia. Int. J. Hyg. Environ. Health 2020, 224, 113422. [Google Scholar] [CrossRef]
- Montoya-Pachongo, C.; Douterelo, I.; Noakes, C.; Camargo-Valero, M.A.; Sleigh, A.; Escobar-Rivera, J.C.; Torres-Lozada, P. Field assessment of bacterial communities and total trihalomethanes: Implications for drinking water networks. Sci. Total Environ. 2018, 616–617, 345–354. [Google Scholar] [CrossRef] [PubMed]
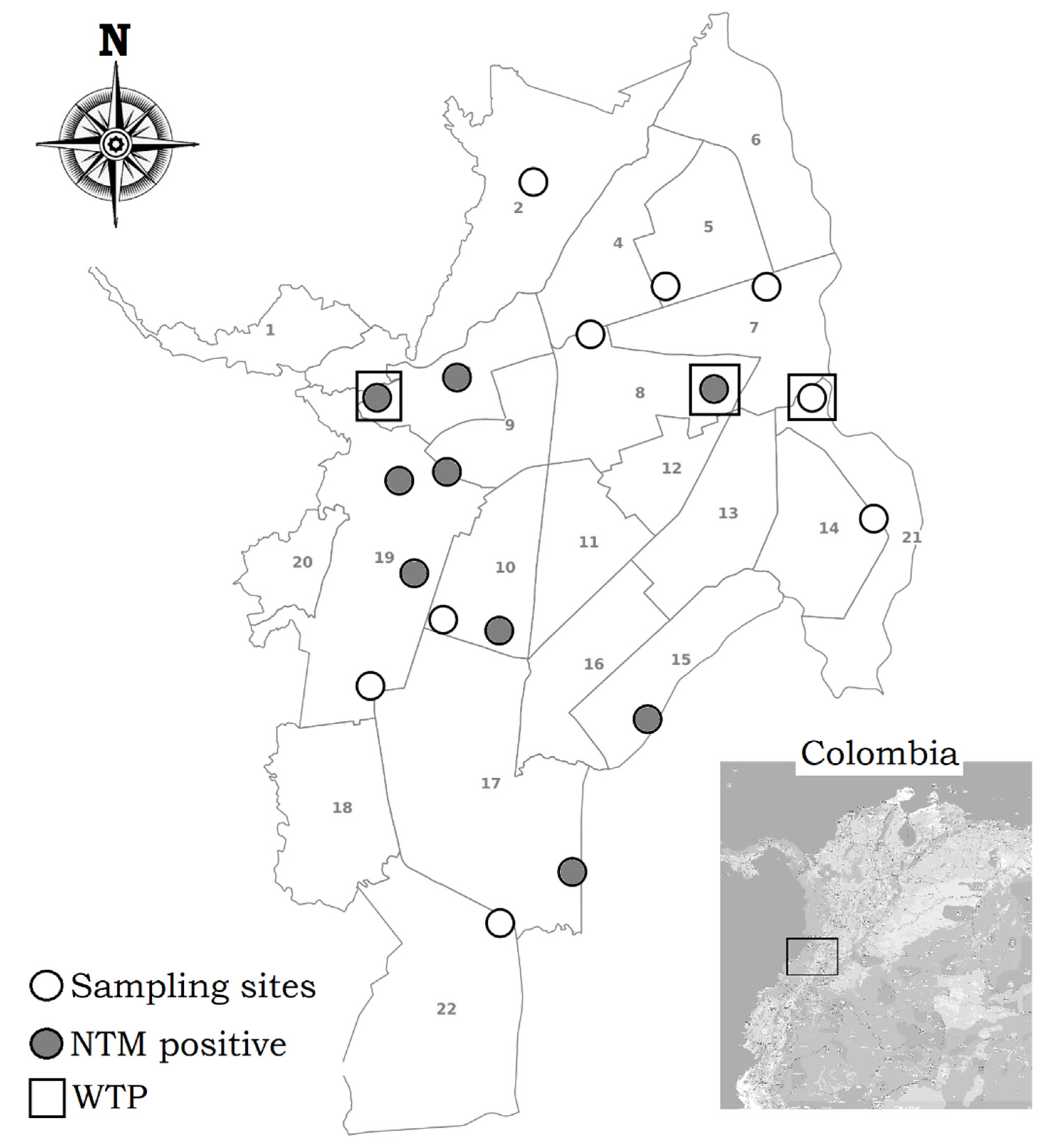
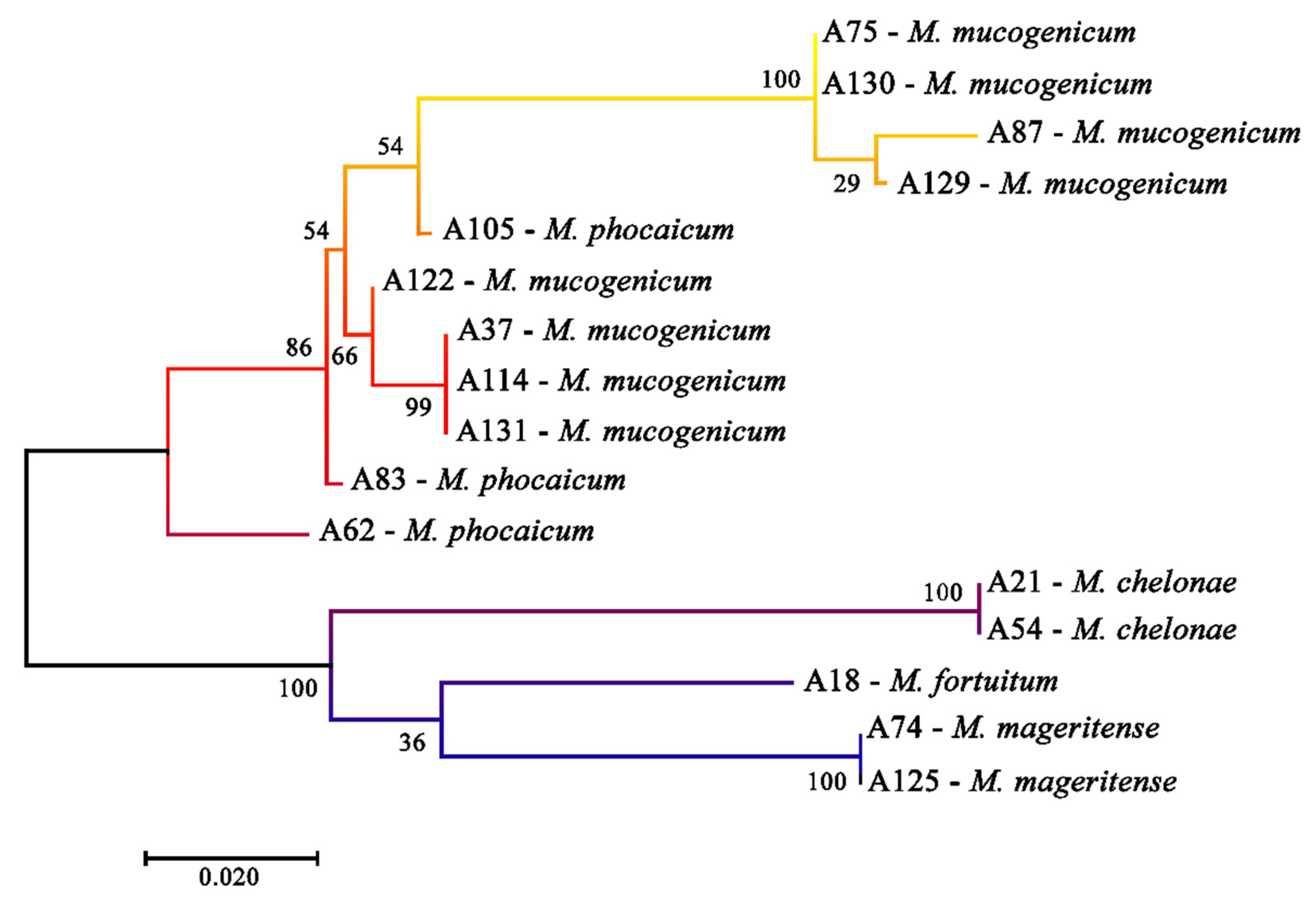
| Sampling Point ID | WTP/Neighborhood | Supplied by WTP | Storage Tank | Floor in the Building | NTM Species Isolated |
|---|---|---|---|---|---|
| 1 | WTP Río Cali | — | — | — | A87-M. mucogenicum |
| 2 | WTP Río Cauca | — | — | — | A18-M. fortuitum A75-M. mucogenicum A74-M. mageritense |
| 3 | WTP Puerto Mallarino | — | — | — | — |
| 4 | Urbanización Río Lili | Puerto Mallarino * Río Cauca | Yes | 4 | — |
| 5 | Los Cámbulos | Río Cauca * Puerto Mallarino | Yes | 2 | A21-M. chelonae A54-M. chelonae |
| 6 | Industrial | Río Cauca * Puerto Mallarino | No | 1 | |
| 7 | Las Granjas | Puerto Mallarino | No | 1 | A62-M. phocaicum |
| 8 | Panamericano | Puerto Mallarino * Río Cauca | No | 1 | — |
| 9 | Refugio | Río Cauca * Puerto Mallarino | Yes | 6 | — |
| 10 | Salomia | Puerto Mallarino | No | 2 | — |
| 11 | San Fernando | Río Cali | No | 1 | A37-M. mucogenicum A83-M. phocaicum |
| 12 | Vipasa ** | Puerto Mallarino Río Cauca | No | 1 | — |
| 13 | Alameda | Río Cauca * Puerto Mallarino | No | 1 | A105-M. phocaicum |
| 14 | Valle del Lili | Puerto Mallarino | Yes | 3 | A122-M. mucogenicum |
| 15 | Alfonso López ** | Puerto Mallarino Río Cauca | No | 2 | — |
| 16 | San Pedro | Río Cali | Yes | 1 | A114-M. mucogenicum A130-M. mucogenicum A131-M. mucogenicum |
| 17 | Puertas del Sol | Puerto Mallarino | No | 1 | — |
| 18 | Ciudad Córdoba | Puerto Mallarino | No | 1 | A125-M. mageritense A129-M. mucogenicum |
| Parameter | Median (Interquartile Range) | Mean (Standard Deviation) | Accepted Parameters in Colombian Regulations [27] |
|---|---|---|---|
| Temperature (°C) (n = 33) | 25.2 (24.6–25.4) | 24.9 (1.1) | Not regulated |
| pH (n = 33) | 7.3 (7.0–7.9) | 7.4 (0.5) | 6.5–9.0 |
| Conductivity (uS/cm) (n = 32) | 136.4 (128.3–150.4) | 139.8 (18.8) | Up to 1000 |
| Antimicrobial Agent | MIC (mg/L) Breakpoints | MIC (mg/L) for RGM Isolates | ||||||
|---|---|---|---|---|---|---|---|---|
| S | I | R | M. fortuitum A18 | M. chelonae A21, A54 | M. mucogenicum A37,A75, A87, A114 | M. phocaicum A62, A83, A105 | M. mageritense A74 | |
| Amikacin | ≤16 | 32 | ≥64 | S | I, S | S, S, S, S | S, S, S | S |
| Tobramycin | ≤4 | 8 | ≥16 | R | S, S | I, R, R, R | R, S, I | R |
| Doxycycline | ≤1 | 2–8 | ≥16 | S | R, R | R, R, S, S | I, R, S | R |
| Ciprofloxacin | ≤1 | 2 | ≥4 | S | R, I | S, R, S, S | R, R, R | R |
| Moxifloxacin | ≤1 | 2 | ≥4 | S | R, R | S, I, S, S | S, S, S | S |
| Clarithromycin | ≤2 | 4 | ≥8 | R | S, S | S, S, S, R | S, S, S | R |
| Linezolid | ≤8 | 16 | ≥32 | S | R, S | S, S, S, R | S, S, S | S |
| Imipenem | ≤4 | 8 | ≥16 | S | R, R | S, S, I, S | I, S, S | R |
| Cefoxitin | ≤16 | 32–64 | ≥128 | I | R, R | S, S, S, S | I, S, S | I |
| Sulfamethoxazole | ≤32 | - | ≥64 | S | R, R | S, S, S, R | S, S, S | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dávalos, A.F.; Garcia, P.K.; Montoya-Pachongo, C.; Rengifo, A.; Guerrero, D.; Díaz-Ordoñez, L.; Díaz, G.; Ferro, B.E. Identification of Nontuberculous Mycobacteria in Drinking Water in Cali, Colombia. Int. J. Environ. Res. Public Health 2021, 18, 8451. https://doi.org/10.3390/ijerph18168451
Dávalos AF, Garcia PK, Montoya-Pachongo C, Rengifo A, Guerrero D, Díaz-Ordoñez L, Díaz G, Ferro BE. Identification of Nontuberculous Mycobacteria in Drinking Water in Cali, Colombia. International Journal of Environmental Research and Public Health. 2021; 18(16):8451. https://doi.org/10.3390/ijerph18168451
Chicago/Turabian StyleDávalos, Andrés F., Pamela K. Garcia, Carolina Montoya-Pachongo, Andrea Rengifo, Daniela Guerrero, Lorena Díaz-Ordoñez, Gustavo Díaz, and Beatriz E. Ferro. 2021. "Identification of Nontuberculous Mycobacteria in Drinking Water in Cali, Colombia" International Journal of Environmental Research and Public Health 18, no. 16: 8451. https://doi.org/10.3390/ijerph18168451
APA StyleDávalos, A. F., Garcia, P. K., Montoya-Pachongo, C., Rengifo, A., Guerrero, D., Díaz-Ordoñez, L., Díaz, G., & Ferro, B. E. (2021). Identification of Nontuberculous Mycobacteria in Drinking Water in Cali, Colombia. International Journal of Environmental Research and Public Health, 18(16), 8451. https://doi.org/10.3390/ijerph18168451






