1. Introduction
Frailty represents a state of age-related physiological vulnerability, which results from the body’s reduced ability to cope with adverse health situations, such as hospitalizations, falls, and functional reduction [
1,
2], with a high prevalence in women [
3,
4]. According to the frailty phenotype, a pre-frail older person presents one or two of the following criteria: unintentional weight loss, muscle weakness, exhaustion, low physical activity, and slowness [
5]. Factors such as chronic inflammation, obesity, muscle quality, physical inactivity, and inadequate nutrition may culminate in adverse outcomes in the musculoskeletal function of the physically frail older adults [
6,
7,
8].
Multi-domain interventions (a combination of more than one intervention, e.g., exercise and nutrition interventions) [
9,
10,
11,
12], as well as multi-component physical training (e.g., a combination of resistance exercise, aerobics, balance, and flexibility) [
13], are recommended to manage physical frailty in older adults.
Exergames emerge as a safe and viable possibility for physical training in the frail older population due to the benefits in balance and mobility and muscle mass gain, mainly when associated with resistance training [
14,
15,
16]. In community-dwelling older women, exergames show high adherence and compliance to physical training. There were gains in both knee and ankle muscle strength, quadriceps cross-sectional area, and gastrocnemius architecture, which are essential aspects to improve functionality, and to prevent sarcopenia and physical frailty [
17,
18,
19].
Higher protein intake also appears to reduce the risk of frailty in women [
20] and increase muscle mass, strength, and function [
21,
22]. Although the rationale for supplementing protein in older adults is considered strong for multiple factors, including their reduced anabolic response to protein intake and their elevated prevalence of inflammatory and catabolic conditions associated with aging [
23,
24], the substantial information on the association between frailty and dietary protein comes from observational studies. Randomized clinical trials in support of high protein are scanty [
24], suggesting that large clinical trials in heterogeneous populations are necessary to quantify the clinical benefit of nutrition on different clinical outcomes, with and without exercise [
23].
When combined with resistance and/or multi-component exercise training, higher protein intake has been more effective than single domain interventions [
10], improving frailty status, lean mass, muscle strength, and mobility in frail older adults [
25]. However, most studies have significant limitations, such as including frail and non-frail older people with different degrees of nutritional impairment [
21,
22,
26] in the same cohort, variation in the amount and quality of protein provided, and not presenting the participant’s nutrient intake [
11,
27]. Furthermore, the effects of multi-component interventions combining progressive resistance training using virtual games with protein supplementation on reducing frailty criteria or reversing pre-frailty status and improving musculoskeletal function in community-dwelling older people have not yet been investigated.
This study hypothesized that multi-component interventions based on virtual games combined with protein supplementation improve body composition and musculoskeletal function more than single domain interventions, such as virtual game training or protein supplementation. Multi-component interventions can also reduce and/or reverse the pre-frail state reinforcing the benefits of combining interventions. Therefore, this randomized controlled trial aimed to verify the effects of progressive resistance training with exergames combined or not with protein supplementation on pre-frailty status and criteria, body composition (appendicular muscle mass (ASM); appendicular muscle mass index (ASMI); plasma levels of (IL)-6 and musculoskeletal function (strength, mass, and architecture) in pre-frail older women.
3. Results
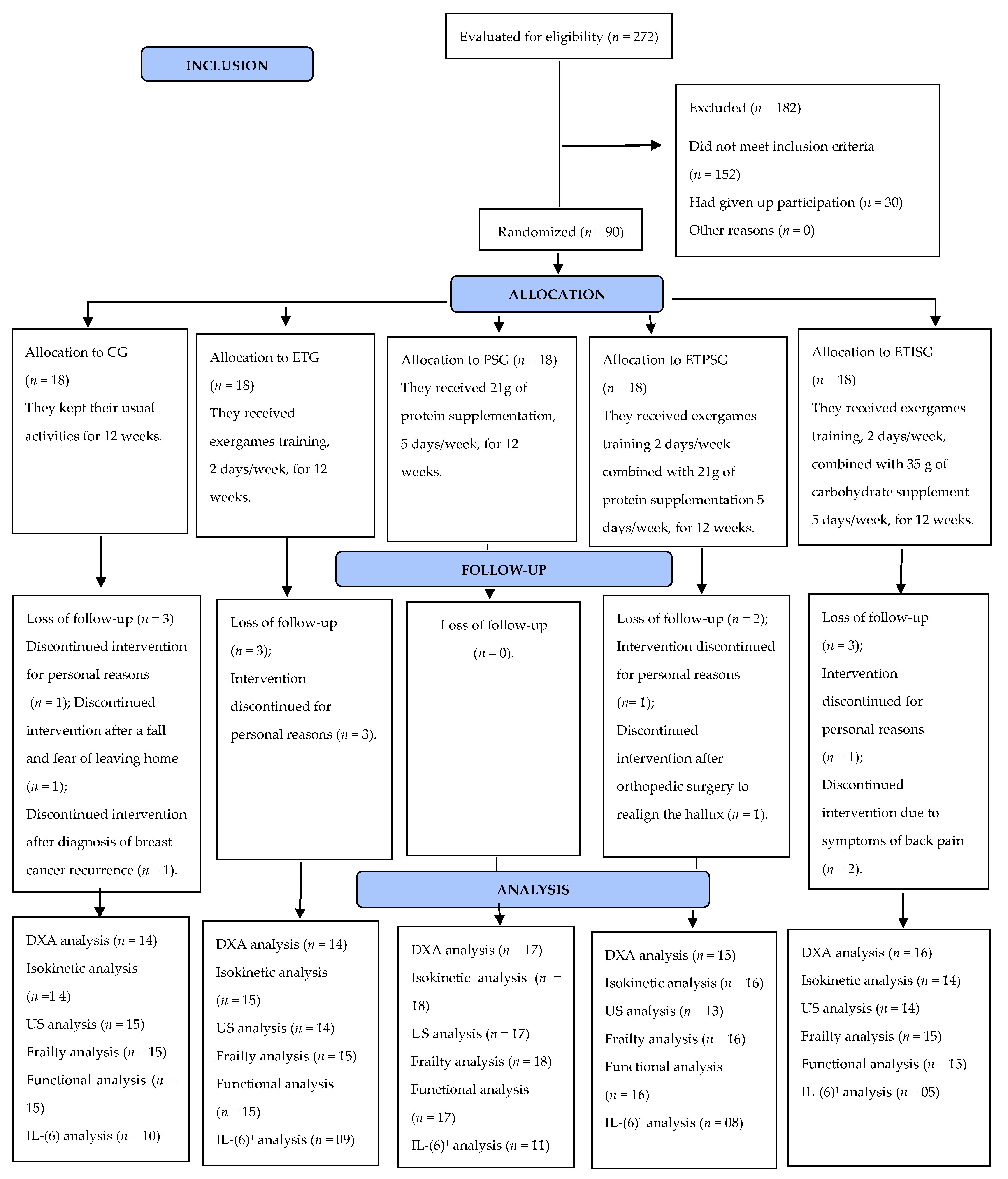
Of the 272 older women assessed, 182 older women were excluded from the study because they did not meet the inclusion criteria. Ninety older women with a mean age of 71.2 ± 4.5 years, classified as pre-frail according to the frailty phenotype, were randomized. Regarding the frailty criteria at the pre-intervention time point, 50% (
n = 45) scored reduced strength (HS), followed by fatigue/exhaustion (38.8%,
n = 35), weight loss (15.5%,
n = 14), low energy expenditure (3.3%,
n = 3), and slowness (3.3%,
n = 3). As for the number of frailty criteria, 72.2% (
n = 65) scored on one criterion for frailty and 27.7% (
n = 25) scored on two criteria. All other investigated characteristics showed no significant difference between the five intervention groups at baseline (
Table 1).
The analysis losses after intervention are depicted in
Figure 1.
A significant interaction (
p = 0.028) was observed on protein intake per kilogram weight in the CG with reduced by about 22% after 12 weeks compared to the pre-time point (1.1 g/kg vs. 0.9 g/kg;
p = 0.015;
d = 0.81). In addition, there was a significant increase in the percentage of protein intake between groups (
p = 0.000) of 17.1% vs. 20.1% in PSG. A significant interaction (
p = 0.049) was also observed on lipid intake in the CG, with a 33.5% reduction in total fat intake. In relation to energy intake, we observed a difference between groups following the experiment. There was a 23.8% reduction in energy intake from 1766.7 kcal to 1426.5 kcal (
p = 0.004,
d = 1.07) in the CG and 12.7% increase in total energy intake in the PSG from 1504.6 kcal to 1723.1 kcal (
p = 0.001,
d = −0.60). As for total protein intake (g/day), a significant 22% increase was observed in the PSG of 63.9 g vs. 82.9 g (
p = 0.006,
d = −1.45) and 28% increase in the ETPSG of 58.1 g vs. 81.5 g (
p = 0.06,
d = −0.84) between groups post experiment was also perceived. Furthermore, a 9.6% increase in carbohydrate intake was observed within groups in the ETISG (
p = 0.013,
d = −0.78) (
Table 2).
The adherence rate to the use of protein and/or isoenergetic supplementation was classified as Intake B, with average supplement intake ranging from 50.1% to 74.9% of that prescribed.
In comparisons of body composition analyzed by DXA, we observed a significant 3.5% reduction in android fat in within-group comparisons (
p = 0.024) in the PSG after the intervention (44.3 ± 7.1 vs. 42.8 ± 6.4%, Δ = −0.4 ± 0.7%;
p = 0.045;
d = 0.21). We also observed a significant difference between groups (
p = 0.001) and interaction (
p = 0.001) in the Up lim MM (kg) with a 2.8% reduction in ETG (3.7 ± 0.9 vs. 3.6 ± 0.9 kg; Δ = −0.1 ± 0.2 kg;
p = 0.002,
d = 0.2), after training. A 3.7% reduction was observed in the ASM of the ETG (16.7 ± 3.4 vs. 16.1 ± 3.3 kg, Δ = −0.6 ± 0.1 kg,
p = 0.02,
d = 0.26) and ASMI (6.8 ± 0.9 vs. 6.5 ± 0.9 kg/m
2, Δ = −0.3 ± 0.0,
p = 0.03,
d = 0.35) after physical training. No significant difference was observed in the other variables for within group, between group, and interaction variables (
Table 3).
There was no significant difference in any variables when comparing within- and between-group, interactions, and medial gastrocnemius muscle architecture (
Table 4).
Concerning the plasma levels of (IL)-6, there was no significant difference in any of the comparisons. A within-group difference was observed for PT of the ankle dorsiflexors at 60°/s ankle (
p = 0.031), with an 11.4% increase in ETPSG (16.3 ± 2.5 vs. 18.4 ± 4.2 Nm, Δ = 2.13 ± 3.4,
p intragroup = 0.021,
d = −0.58). There was a significant change in the HS from pre- to post-intervention (
p = 0.008), with a 13.7% increase in ETG (20.1 ± 7.2 vs. 23.3 ± 6.2 kg, Δ = 3.2 ± 4.9,
p intragroup = 0.004,
d = −0.48). However, no significant difference was observed at the two time points (pre- and post-intervention) when evaluated between intervention groups (
Table 5).
There was a significant reduction in the number of participants with exhaustion criterion, being 100% in the ETG (pre:
n = 7, 46.7% versus. post:
n = 0, 0.0%,
p = 0.016), 75% in the PSG (pre:
n = 8, 44.4% vs. post:
n = 2, 11.1%,
p = 0.031), and 100% in the ETPSG (pre:
n = 7, 43.8% versus. post:
n = 0, 0.0%,
p = 0.016) groups. No significant difference was observed in other frailty criteria both pre- and post-experiment. Regarding the frailty classification, in CG, seven subjects (46.7%) changed from pre-frail to non-frail and one (6.7%) from pre-frail to frail (
p = 0.016); in ETG, 11 (73.3%) participants changed status from pre-frail to non-frail (
p = 0.001); in ETPSG, 10 (55.6%) changed from pre-frail to non-frail and 1 (5.6%) from pre-frail to frail (
p = 0.000); in ETISG, seven (43.8%) changed from pre-frail to non-frail (
p = 0.016) (
Table 6).
4. Discussion
Physical training exergames associated with protein supplementation (WiiProtein) reversed the pre-frailty state, reduced fatigue/exhaustion, and increased ankle dorsiflexors torque without difference between groups. The single domain intervention with exergames increased the handgrip strength, decreased ASM and ASMI (within group), and reduced the Up Lim MM (kg) in relation to the other groups, although this difference was subtle when compared to the other intervention groups. Meanwhile, the single domain with protein supplementation decreased android fat, without significant difference in the muscle mass in prefrail community-dwelling older women.
Other studies that evaluated multi-domain interventions, combining physical training and calorie-protein [
11] supplementation or nutrition education [
12], also found a reduction in the criteria and prevalence of physical frailty. However, none of these studies mentioned which frailty criteria were improved after interventions. Muscle weakness and self-reported fatigue/exhaustion are two Frailty Phenotype criteria frequent in frail and pre-frail older, and the most prevalent in the present study [
11,
40,
41]. Even without difference when compared to the control group, we must highlight the reduction of fatigue/exhaustion because the majority of participants were included reporting this criterion. Thus, when we found, at post intervention (exergames or protein supplementation or the association of both), that fatigue/exhaustion had ameliorated and this outcome was not observed in the control group, we suggest an important clinical relevance of WiiProtein, isolated or in combination. We might suppose that multifactorial intervention (WiiProtein) should be used for pre-frail older women while a reduction in fatigue/exhaustion and increase in muscle strength are associated with delayed progression of frailty, as detected in the present study with pre-frailty reversion [
42].
The WiiProtein interventions were not different from control group, instead of improvements more pronounced in the dorsiflexion torque. The study demonstrated the importance of protein supplementation, alone or associated with exercise and social integration during the study in the management of pre-frailty in women. Thus, even if older women were unable to exercise, protein supplementation alone was sufficient to improve fatigability, preventing negative prognosis.
Multi-component physical training, especially with the addition of progressive resistance, is a strategy to improve muscle strength, physical performance, balance, and flexibility, as well as to reverse the state of frailty in pre-frail and frail older people [
13,
43,
44]. Interventions associated with protein supplementation can favor better outcomes [
9]. Consistent with these findings, in our study, only the pre-frail older women in the ETG and ETPSG had muscle strength gains when compared to other intervention groups. However, the combination of exergames and protein supplementation resulted in the maintenance of lean body mass without additional gains. Contrarily, exercise training without supplementation demonstrated a decline in Up lim MM (kg) and ASM mass after intervention. One study observed an increase in strength and muscle mass in frail older people after resistance training intervention associated with 30 g of protein supplementation for 24 weeks [
22]. This study suggests that frail older people under resistance training should take protein supplementation to increase skeletal muscle mass. Therefore, we did not observe an increase in lean body mass nor changes in muscle architecture in our non-intervention group. This may be associated with a low supply of protein supplementation (21 g/day), or a training protocol with exergames, which may have been insufficient in frequency (2×/week) and duration (12 weeks), since some studies have identified benefits when the physically frail perform physical training three times per week, for 30–45 min/session, for at least 16 weeks [
45].
The physical training with virtual games proposed in this study emphasize on exercises for the lower limbs, with focus on the gastrocnemius muscle (e.g., palm tree, chair), with a progressive increase in load (resistance) of the exercises every two weeks, in addition to the stimulus through protein supplementation. Nevertheless, no changes in muscle architecture were verified after 12 weeks of intervention. Even though the muscle architecture presented no change, by assessing muscle architecture using ultrasonography (US), it was found that pre-frail older women had reduced MT and FL of the medial gastrocnemius at the pre-intervention time point. This factor indicates sarcopenia, considering the cutoff points of 1.5 cm for MT found in sarcopenic older individuals [
33,
46]. A study conducted with community-dwelling older women who underwent physical training with virtual games found a 1.3% increase in the quadriceps cross-sectional area [
17]. The mentioned study used magnetic resonance imaging, a non-invasive method that is considered the gold standard for assessing muscle quality and quantity [
47]. Other studies verified an 8.7% increase in the thickness of the medial gastrocnemius muscle after 12 weeks of virtual dance training performed three times a week in community-dwelling older women [
18]. Further, there were gains in MT, PA, and FL of the medial gastrocnemius after training for eight weeks with ballroom dancing (three times per week for 60 min) [
48].
Regardless of the investigated muscle architecture changes, the ETPSG participants increased the dorsiflexors’ torque, reinforcing the non-linearity of the relationship between muscle mass and strength, and suggesting neural gains [
49]. The improvement in ankle dorsiflexion strength may have resulted from the use of an overload vest beginning in the third week of physical training, with load progression every two weeks. In addition, there was a probable effect of lower limb resistance exercises (e.g., palm tree, chair) and neuromotor exercises (e.g., balance bubble, snowboard slalom). We also assume that the physical training associated with protein supplementation contributed to the positive results, although no muscle mass gains were observed in the Lo lim MM (kg). Once, despite a significant effect, an increase in the strength of the plantiflexors was observed in the training groups associated with protein as isocaloric supplementation. Other studies investigating the effects of physical training through virtual games also found gains in Lo lim MM (kg) strength in both pre-frail community-dwelling older women [
16] and moderately active community-dwelling older women [
18]. Santos et al. (2019) attributed the results to the progression of the vest’s load used by participants. Gallo et al. (2019) verified a 16.3% increase in ankle plantar flexor peak torque, as well as an increase in muscle mass detected by ultrasonography (US) of the gastrocnemius muscle and calf circumference measurement after 12 weeks of physical training (three times per week) with virtual dance (Xbox 360
®) in community-dwelling older women. However, these studies lacked an assessment of protein intake and performance of the nutritional intervention.
Therefore, we assume that the protein supply in this study probably was insufficient to stimulate protein synthesis and generate additional gains in gastrocnemius muscle thickness, especially when considering older women with likely an excess in intramuscular fat. Although protein supplementation raised protein intake by 23% in the PSG group and 29% in the ETPSG group, mean protein intake after intervention (1.1 ± 0.2 g/kg/d) was below expected (≥1.2 g/kg/day) [
50] in both groups. In addition, as the pre-frail older women were overweight, the average amount of protein offered was 0.28 g/kg/day in only one additional meal. However, it was recommended that the subjects have at least three meals containing 0.4 g of protein/kg of body mass to maximize myofibrillar protein synthesis [
51].
Nonetheless, increasing a dose of at least 20 g of protein/day appeared to represent a promising strategy to prevent and treat sarcopenia in the older population [
52], considering that older individuals have difficulty adhering to large doses of protein supplementation. This strategy also contributes to lower body adiposity [
53,
54]. Our data showed that protein supplementation reduced android fat mass in PSG participants by 3.5% and preserved muscle mass.
Our data showed that protein supplementation reduced android fat mass in PSG participants by 3.5%. In addition to the excess body adiposity found in the study participants, they had prediabetes and higher cytokine level, which was observed by increased glycated hemoglobin levels (A1c) and IL-6, respectively. Thus, insulin resistance can be stimulated by the inflammatory response, which could trigger increased intramuscular fat, negatively affecting musculoskeletal function over time [
55]. Therefore, protein supplementation could be advantageous for older women in order to preserve muscle mass, improve insulin action, and reduce the development and worsening of chronic diseases such as Type II diabetes [
22]. In our study, the aim of IL-6 assessment was to investigate the effects of multidomain intervention on factors that could intervene in the body composition and muscle function. As previously reported, lower muscle mass and strength have been associated to higher plasma concentrations of IL-6, to frailty, and to poor health outcomes in older adults [
56,
57]. Instead, although participants of the present study have presented higher IL-6 levels, no effect was found for multidomain intervention.
The adherence to supplements offered in our study was lower than observed by other studies [
21,
22]. Although Tieland et al. (2012) controlled the use of supplements, in a similar way to ours, the product offered in this study was presented in liquid form, which may have facilitated adherence to consumption. While Park et al. (2018) offered the supplement in powder form and their adherence to the use of the supplements was controlled by the rate of non-consumed products. The modification of eating habits in older people is a great challenge. In addition, the adaptation to the organoleptic and physical characteristics of the product and the participant’s need for portioning (preparing the product and the quantity to use) may have contributed to the lower adherence to consumption in our study. It is noteworthy that, in this study, the participants were periodically encouraged (at least once a week) to use the product properly, through verbal guidance on the days of physical training and by telephone to the participants who were not undergoing training. Moreover, all participants had recipe suggestions (book with recipes tested by the researchers, with isocaloric or protein supplements). The difficulty of adherence found in this study may approach the limitations found in clinical practice. Thus, for future studies, it is suggested to evaluate the barriers and difficulties of adherence to supplementation.
Finally, physical training with exergames, encompassing balance and progressive resistance exercises, proved to be safe and motivating for community-based pre-frail older women, as observed by the high adherence rate to the study (87.7%). The practice of exercise using exergames has been classified as fun and motivating by aged individuals. Thus, the type of physical training chosen, exergames, per se, stimulates adherence. In addition, interventions with more than one domain (e.g., physical training and nutritional supplementation) appear promising in treating physical frailty
4.1. Strengths and Limitations
The present study has relevant strengths. It is a randomized controlled clinical trial, conducted only with community women who were classified as pre-frail. The assessment of participants’ food intake followed a standardized and recommended methodology. Participants showed good adherence to the study, and adherence to physical training with exergames and the supplement, and there were no reports of adverse effects with the use of the protein supplement. Finally, the study had a low dropout rate from the interventions. Limitations of the study include the 12-week intervention period, as well as frequency and progression of resistance, which may have been insufficient to increase the lean body mass of pre-frail older women, and the fatigue/exhaustion criterion was assessed with two questions derived from Fried’s frailty phenotype. Thus, for future research, we suggest that this criterion be evaluated by other methods including physical and cognitive function to detect the interactions between performance fatigability and perceived fatigability and by a multi-professional team, including psychology professionals. In addition, a myoelectrical analysis should be included to elucidate the mechanisms of the strength increase induced by training with virtual games on neuromuscular aspects. Finally, the study was conducted only with pre-frail women, so the results cannot be generalized to pre-frail men.
4.2. Implications for Clinical Practice
Our results indicated that physical training with exergames associated or not associated with protein supplementation, for 12 weeks, was able to reverse the state of frailty and reduce the report of fatigue/exhaustion after the intervention. Self-reported exhaustion appears to emerge in the development of early frailty. Furthermore, the feeling of fatigue/exhaustion can trigger behavioral changes, making individuals less active, predisposing to a vicious cycle that would direct toward frailty [
58]. Therefore, early identification of this criterion through a quick and easy-to-perform instrument, such as by applying two questions taken from the Center for Epidemiologic Studies Depression Scale (CES-D), is considered a valid instrument for clinical studies [
5,
42,
59] and should be implemented in clinical practice. Regarding the practical implications, the type of physical training can contribute to enhancing psychosocial effects if played with other players. Additionally, the benefits can be applied to cognitive functioning due to the dual task required by the virtual game. Finally, exergames were well accepted by pre-frail older women. Physical exercise using technological resources such as exergames can be a strategy to increase adherence to physical training and probably contribute to the exchange of experiences with younger family members [
60].