Modeling Effects of Spatial Heterogeneities and Layered Exposure Interventions on the Spread of COVID-19 across New Jersey
Abstract
:1. Introduction
1.1. Background: Characterizing Pathways of COVID-19 Transmission
1.2. Modeling COVID-19: Computational Approaches
1.3. New Jersey as a “Microcosm” of COVID-19 Spread Heterogeneities
2. Materials and Methods
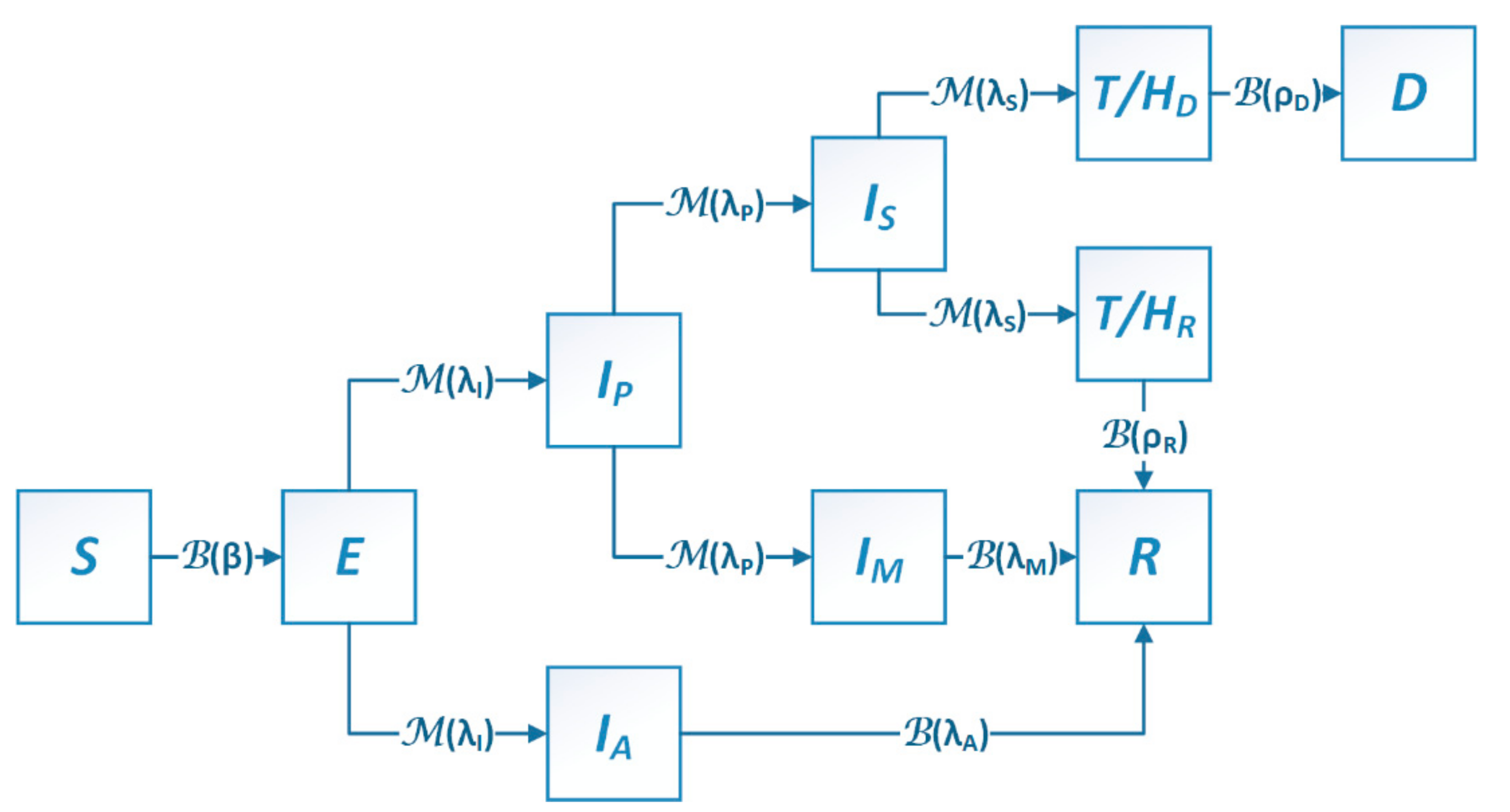
2.1. Stochastic SEIR Model
2.2. Model Calibration
2.3. Simulation
3. Results
3.1. Spatiotemporal Analysis of COVID-19 Deaths across New Jersey
3.2. Comparison of Predicted and Reported Confirmed Deaths
3.3. Realistic and Counterfactual Intervention Scenarios
4. Discussion
4.1. Spatially Heterogeneous Transmission Rate
4.2. Reproduction Number at County Level
4.3. Reducing COVID-19 Fatality by Layered Exposure-Relevant Interventions
4.4. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahmad, F.B.; Anderson, R.N. The leading causes of death in the US for 2020. JAMA 2021, 325, 1829–1830. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, F.B.; Cisewski, J.A.; Miniño, A.; Anderson, R.N. Provisional mortality data—United States, 2020. Morb. Mortal. Wkly. Rep. 2021, 70, 519–522. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Yao, L.; Wei, T.; Tian, F.; Jin, D.-Y.; Chen, L.; Wang, M. Presumed asymptomatic carrier transmission of COVID-19. JAMA 2020, 323, 1406–1407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, P.; Zhu, J.; Zhang, Z.; Han, Y. A familial cluster of infection associated with the 2019 novel coronavirus indicating possible person-to-person transmission during the incubation period. J. Infect. Dis. 2020, 221, 1757–1761. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deziel, N.C.; Allen, J.G.; Scheepers, P.T.; Levy, J.I. The COVID-19 pandemic: A moment for exposure science. J. Expo. Sci. Environ. Epidemiol. 2020, 30, 591–593. [Google Scholar] [CrossRef] [PubMed]
- CDC. How COVID-19 Spreads. 2020. Available online: https://www.cdc.gov/coronavirus/2019-ncov/prevent-getting-sick/how-covid-spreads.html (accessed on 6 October 2020).
- Morawska, L.; Cao, J. Airborne transmission of SARS-CoV-2: The world should face the reality. Environ. Int. 2020, 139, 105730. [Google Scholar] [CrossRef] [PubMed]
- Morawska, L.; Milton, D.K. It is time to address airborne transmission of coronavirus disease 2019 (COVID-19). Clin. Infect. Dis. 2020, 71, 2311–2313. [Google Scholar] [CrossRef] [PubMed]
- Fears, A.C.; Klimstra, W.B.; Duprex, P.; Hartman, A.; Weaver, S.C.; Plante, K.S.; Mirchandani, D.; Plante, J.A.; Aguilar, P.V.; Fernández, D. Persistence of severe acute respiratory syndrome coronavirus 2 in aerosol suspensions. Emerg. Infect. Dis. 2020, 26, 2168. [Google Scholar] [CrossRef]
- Fennelly, K.P. Particle sizes of infectious aerosols: Implications for infection control. Lancet Resp. Med. 2020, 8, 914–924. [Google Scholar] [CrossRef]
- Van Doremalen, N.; Bushmaker, T.; Morris, D.H.; Holbrook, M.G.; Gamble, A.; Williamson, B.N.; Tamin, A.; Harcourt, J.L.; Thornburg, N.J.; Gerber, S.I. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N. Engl. J. Med. 2020, 382, 1564–1567. [Google Scholar] [CrossRef]
- CDC. Scientific Brief: SARS-CoV-2 and Potential Airborne Transmission. 2020. Available online: https://www.cdc.gov/coronavirus/2019-ncov/more/scientific-brief-sars-cov-2.html (accessed on 15 June 2020).
- Kim, Y.-I.; Kim, S.-G.; Kim, S.-M.; Kim, E.-H.; Park, S.-J.; Yu, K.-M.; Chang, J.-H.; Kim, E.J.; Lee, S.; Casel, M.A.B. Infection and rapid transmission of SARS-CoV-2 in ferrets. Cell Host Microbe 2020, 27, 704–709. [Google Scholar] [CrossRef] [PubMed]
- Richard, M.; Kok, A.; de Meulder, D.; Bestebroer, T.M.; Lamers, M.M.; Okba, N.M.; van Vlissingen, M.F.; Rockx, B.; Haagmans, B.L.; Koopmans, M.P. SARS-CoV-2 is transmitted via contact and via the air between ferrets. Nat. Commun. 2020, 11, 1–6. [Google Scholar] [CrossRef]
- Shi, J.; Wen, Z.; Zhong, G.; Yang, H.; Wang, C.; Huang, B.; Liu, R.; He, X.; Shuai, L.; Sun, Z. Susceptibility of ferrets, cats, dogs, and other domesticated animals to SARS–coronavirus 2. Science 2020, 368, 1016–1020. [Google Scholar] [CrossRef] [Green Version]
- Chia, P.Y.; Coleman, K.K.; Tan, Y.K.; Ong, S.W.X.; Gum, M.; Lau, S.K.; Lim, X.F.; Lim, A.S.; Sutjipto, S.; Lee, P.H. Detection of air and surface contamination by SARS-CoV-2 in hospital rooms of infected patients. Nat. Commun. 2020, 11, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Lednicky, J.A.; Lauzardo, M.; Fan, Z.H.; Jutla, A.S.; Tilly, T.B.; Gangwar, M.; Usmani, M.; Shankar, S.N.; Mohamed, K.; Eiguren-Fernandez, A. Viable SARS-CoV-2 in the air of a hospital room with COVID-19 patients. MedrXiv 2020, 4, 1–20. [Google Scholar] [CrossRef]
- Liu, Y.; Ning, Z.; Chen, Y.; Guo, M.; Liu, Y.; Gali, N.K.; Sun, L.; Duan, Y.; Cai, J.; Westerdahl, D. Aerodynamic analysis of SARS-CoV-2 in two Wuhan hospitals. Nat. Med. 2020, 582, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Pei, G.; Taylor, M.; Rim, D. Human exposure to respiratory aerosols in a ventilated room: Effects of ventilation condition, emission mode, and social distancing. Sustain. Cities Soc. 2021, 73, 103090. [Google Scholar] [CrossRef]
- Adenaiye, O.O.; Lai, J.; de Mesquita, P.J.B.; Hong, F.H.; Youssefi, S.; German, J.R.; Tai, S.-H.S.; Albert, B.J.; Schanz, M.; Weston, S. Infectious SARS-CoV-2 in Exhaled Aerosols and Efficacy of Masks During Early Mild Infection. Clin. Infect. Dis. 2021. [Google Scholar] [CrossRef] [PubMed]
- Port, J.; Yinda, C.K.; Avanzato, V.; Schulz, J.; Holbrook, M.; van Doremalen, N.; Shaia, C.; Fischer, R.; Munster, V. Increased aerosol transmission for B. 1.1. 7 (alpha variant) over lineage A variant of SARS-CoV-2. bioRxiv 2021. [Google Scholar] [CrossRef]
- Morawska, L.; Tang, J.W.; Bahnfleth, W.; Bluyssen, P.M.; Boerstra, A.; Buonanno, G.; Cao, J.; Dancer, S.; Floto, A.; Franchimon, F. How can airborne transmission of COVID-19 indoors be minimised? Environ. Int. 2020, 142, 105832. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, A.; Eqan, M.; Pervez, S.; Alghamdi, H.A.; Tabinda, A.B.; Yasar, A.; Brindhadevi, K.; Pugazhendhi, A. COVID-19 and frequent use of hand sanitizers; human health and environmental hazards by exposure pathways. Sci. Total Environ. 2020, 742, 140561. [Google Scholar] [CrossRef] [PubMed]
- Gostin, L.O.; Wiley, L.F. Governmental Public Health Powers During the COVID-19 Pandemic: Stay-at-home Orders, Business Closures, and Travel Restrictions. JAMA 2020, 323, 2137–2138. [Google Scholar] [CrossRef]
- Lazzerini, M.; Putoto, G. COVID-19 in Italy: Momentous decisions and many uncertainties. Lancet Glob. Health 2020, 8, 641–642. [Google Scholar] [CrossRef] [Green Version]
- Pan, A.; Liu, L.; Wang, C.; Guo, H.; Hao, X.; Wang, Q.; Huang, J.; He, N.; Yu, H.; Lin, X.; et al. Association of Public Health Interventions With the Epidemiology of the COVID-19 Outbreak in Wuhan, China. JAMA 2020, 323, 1915–1923. [Google Scholar] [CrossRef] [Green Version]
- Horton, R. Offline: COVID-19 is not a pandemic. Lancet 2020, 396, 874. [Google Scholar] [CrossRef]
- Bialek, S.; Boundy, E.; Bowen, V.; Chow, N.; Cohn, A.; Dowling, N.; Ellington, S.; Gierke, R.; Hall, A.; Macneil, J.; et al. Severe Outcomes Among Patients with Coronavlrus Disease 2019 (COVID-19)—United States, February 12–March 16, 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 343–346. [Google Scholar]
- Onder, G.; Rezza, G.; Brusaferro, S. Case-Fatality Rate and Characteristics of Patients Dying in Relation to COVID-19 in Italy. JAMA 2020, 323, 1775–1776. [Google Scholar] [CrossRef]
- Richardson, S.; Hirsch, J.S.; Narasimhan, M.; Crawford, J.M.; McGinn, T.; Davidson, K.W.; Barnaby, D.P.; Becker, L.B.; Chelico, J.D.; Cohen, S.L. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA 2020, 323, 2052–2059. [Google Scholar] [CrossRef]
- Gandhi, M.; Yokoe, D.S.; Havlir, D.V. Asymptomatic Transmission, the Achilles’ Heel of Current Strategies to Control Covid-19. N. Engl. J. Med. 2020, 382, 2158–2160. [Google Scholar] [CrossRef]
- Chen, T.; Wu, D.; Chen, H.; Yan, W.; Yang, D.; Chen, G.; Ma, K.; Xu, D.; Yu, H.; Wang, H.; et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: Retrospective study. BMJ 2020, 368, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Koh, D. Occupational risks for COVID-19 infection. Occup. Med. 2020, 70, 3–5. [Google Scholar] [CrossRef] [Green Version]
- Chowkwanyun, M.; Reed, A.L., Jr. Racial health disparities and Covid-19—Caution and context. N. Engl. J. Med. 2020, 383, 201–203. [Google Scholar] [CrossRef]
- Owen, W.F.; Carmona, R.; Pomeroy, C. Failing another national stress test on health disparities. JAMA 2020, 323, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Metcalf, C.J.E.; Morris, D.H.; Park, S.W. Mathematical models to guide pandemic response. Science 2020, 369, 368–369. [Google Scholar] [CrossRef] [PubMed]
- Ross, R. The Prevention of Malaria; John Murray: London, UK, 1910. [Google Scholar]
- Kermack, W.O.; McKendrick, A.G. A contribution to the mathematical theory of epidemics. Proc. R. Soc. Lond. Ser. A 1927, 115, 700–721. [Google Scholar]
- Schneckenreither, G.; Popper, N.; Zauner, G.; Breitenecker, F. Modelling SIR-type epidemics by ODEs, PDEs, difference equations and cellular automata—A comparative study. Simul. Model. Pract. Theory 2008, 16, 1014–1023. [Google Scholar] [CrossRef]
- Bacaër, N. A Short History of Mathematical Population Dynamics; Springer Science & Business Media: New York, NY, USA, 2011. [Google Scholar]
- Chalub, F.A.; Souza, M.O. The SIR epidemic model from a PDE point of view. Math. Comput. Model. 2011, 53, 1568–1574. [Google Scholar] [CrossRef] [Green Version]
- Zhuang, Q.; Wang, J. A spatial epidemic model with a moving boundary. Infect. Dis. Model. 2021, 6, 1046–1060. [Google Scholar] [CrossRef]
- CDC. COVID-19 Mathematical Modeling. Available online: https://www.cdc.gov/coronavirus/2019-ncov/covid-data/mathematical-modeling.html (accessed on 6 October 2021).
- Giordano, G.; Blanchini, F.; Bruno, R.; Colaneri, P.; Di Filippo, A.; Di Matteo, A.; Colaneri, M. Modelling the COVID-19 epidemic and implementation of population-wide interventions in Italy. Nat. Med. 2020, 26, 855–860. [Google Scholar] [CrossRef]
- Childs, M.L.; Kain, M.P.; Harris, M.J.; Kirk, D.; Couper, L.; Nova, N.; Delwel, I.; Ritchie, J.; Becker, A.D.; Mordecai, E.A. The impact of long-term non-pharmaceutical interventions on COVID-19 epidemic dynamics and control: The value and limitations of early models. Proc. R. Soc. B 2021, 288, 20210811. [Google Scholar] [CrossRef]
- Bonabeau, E. Agent-based modeling: Methods and techniques for simulating human systems. Proc. Natl. Acad. Sci. USA 2002, 99, 7280–7287. [Google Scholar] [CrossRef] [Green Version]
- Grefenstette, J.J.; Brown, S.T.; Rosenfeld, R.; DePasse, J.; Stone, N.T.; Cooley, P.C.; Wheaton, W.D.; Fyshe, A.; Galloway, D.D.; Sriram, A. FRED (A Framework for Reconstructing Epidemic Dynamics): An open-source software system for modeling infectious diseases and control strategies using census-based populations. BMC Public Health 2013, 13, 1–14. [Google Scholar] [CrossRef] [Green Version]
- COVID, I.; Murray, C.J. Forecasting COVID-19 impact on hospital bed-days, ICU-days, ventilator-days and deaths by US state in the next 4 months. MedRxiv 2020, 1–26. Available online: https://www.medrxiv.org/content/10.1101/2020.1103.1127.20043752v20043751 (accessed on 6 October 2021).
- Petropoulos, F.; Makridakis, S. Forecasting the novel coronavirus COVID-19. PLoS ONE 2020, 15, e0231236. [Google Scholar] [CrossRef] [PubMed]
- Altieri, N.; Barter, R.L.; Duncan, J.; Dwivedi, R.; Kumbier, K.; Li, X.; Netzorg, R.; Park, B.; Singh, C.; Tan, Y.S. Curating a COVID-19 data repository and forecasting county-level death counts in the United States. 2020. Available online: https://arxiv.org/abs/2005.07882 (accessed on 6 October 2021).
- Gu, Y. COVID-Projections. Available online: https://covid19-projections.com/about/#historical-performance (accessed on 6 October 2021).
- Wong, F.; Collins, J.J. Evidence that coronavirus superspreading is fat-tailed. Proc. Natl. Acad. Sci. USA 2020, 117, 29416–29418. [Google Scholar] [CrossRef] [PubMed]
- Allen, L.J.; Burgin, A.M. Comparison of deterministic and stochastic SIS and SIR models in discrete time. Math. Biosci. 2000, 163, 1–33. [Google Scholar] [CrossRef]
- Britton, T. Stochastic epidemic models: A survey. Math. Biosci. 2010, 225, 24–35. [Google Scholar] [CrossRef] [PubMed]
- West, R.W.; Thompson, J.R. Models for the simple epidemic. Math. Biosci. 1997, 141, 29–39. [Google Scholar] [CrossRef]
- Li, M.Y.; Muldowney, J.S. Global stability for the SEIR model in epidemiology. Math. Biosci. 1995, 125, 155–164. [Google Scholar] [CrossRef]
- Chang, S.; Harding, N.; Cliff, O.; Prokopenko, M. Modelling transmission and control of the COVID-19 pandemic in Australia. arXiv 2020, arXiv:2003.10218. [Google Scholar] [CrossRef]
- Chatterjee, K.; Chatterjee, K.; Kumar, A.; Shankar, S. Healthcare impact of COVID-19 epidemic in India: A stochastic mathematical model. Med. J. Armed Forces India 2020, 76, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, N.; Laydon, D.; Nedjati Gilani, G.; Imai, N.; Ainslie, K.; Baguelin, M.; Bhatia, S.; Boonyasiri, A.; Cucunuba Perez, Z.; Cuomo-Dannenburg, G. Report 9: Impact of non-pharmaceutical interventions (NPIs) to reduce COVID19 mortality and healthcare demand. Imp. Coll. Lond. 2020, 1–20. [Google Scholar]
- Kucharski, A.J.; Russell, T.W.; Diamond, C.; Liu, Y.; Edmunds, J.; Funk, S.; Eggo, R.M.; Sun, F.; Jit, M.; Munday, J.D.; et al. Early dynamics of transmission and control of COVID-19: A mathematical modelling study. Lancet Infect. Dis. 2020, 20, 553–558. [Google Scholar] [CrossRef] [Green Version]
- Matrajt, L.; Leung, T. Evaluating the Effectiveness of Social Distancing Interventions to Delay or Flatten the Epidemic Curve of Coronavirus Disease. Emerg. Infect. Dis. 2020, 26, 1740–1748. [Google Scholar] [CrossRef] [PubMed]
- Kain, M.P.; Childs, M.L.; Becker, A.D.; Mordecai, E.A. Chopping the tail: How preventing superspreading can help to maintain COVID-19 control. Epidemics 2021, 34, 100430. [Google Scholar] [CrossRef] [PubMed]
- Thomas, L.J.; Huang, P.; Yin, F.; Luo, X.I.; Almquist, Z.W.; Hipp, J.R.; Butts, C.T. Spatial Heterogeneity Can Lead to Substantial Local Variations in COVID-19 Timing and Severity. arXiv 2020, arXiv:2005.09850, 24180–24187. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Yamamoto, N. Using a partial differential equation with Google Mobility data to predict COVID-19 in Arizona. arXiv 2020, arXiv:2006.16928. [Google Scholar] [CrossRef]
- Bertaglia, G.; Boscheri, W.; Dimarco, G.; Pareschi, L. Spatial spread of COVID-19 outbreak in Italy using multiscale kinetic transport equations with uncertainty. arXiv 2021, arXiv:2106.07262. [Google Scholar] [CrossRef]
- Bertaglia, G.; Pareschi, L. Hyperbolic compartmental models for epidemic spread on networks with uncertain data: Application to the emergence of Covid-19 in Italy. arXiv 2021, arXiv:2105.14258. [Google Scholar] [CrossRef]
- Viguerie, A.; Lorenzo, G.; Auricchio, F.; Baroli, D.; Hughes, T.J.; Patton, A.; Reali, A.; Yankeelov, T.E.; Veneziani, A. Simulating the spread of COVID-19 via a spatially-resolved susceptible–exposed–infected–recovered–deceased (SEIRD) model with heterogeneous diffusion. Appl. Math. Lett. 2021, 111, 106617. [Google Scholar] [CrossRef]
- Viguerie, A.; Veneziani, A.; Lorenzo, G.; Baroli, D.; Aretz-Nellesen, N.; Patton, A.; Yankeelov, T.E.; Reali, A.; Hughes, T.J.; Auricchio, F. Diffusion–reaction compartmental models formulated in a continuum mechanics framework: Application to COVID-19, mathematical analysis, and numerical study. Comput. Mech. 2020, 66, 1131–1152. [Google Scholar] [CrossRef] [PubMed]
- JHU (Johns Hopkins University). Death rates from coronavirus (COVID-19) in the United States as of November 6, 2020, by state. Available online: https://www.statista.com/statistics/1109011/coronavirus-covid19-death-rates-us-by-state/ (accessed on 6 October 2021).
- Caflisch, R.E. Monte carlo and quasi-monte carlo methods. Acta Numer. 1998, 1998, 1–49. [Google Scholar] [CrossRef] [Green Version]
- Chopin, N.; Papaspiliopoulos, O. An Introduction to Sequential Monte Carlo; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar]
- Gerber, M.; Chopin, N. Sequential quasi-Monte Carlo. J. R. Statist. Soc. B 2015, 77, 509–579. [Google Scholar] [CrossRef] [Green Version]
- Lemieux, C. Monte Carlo and Quasi-Monte Carlo Sampling; Springer Science & Business Media: New York, NY, USA, 2009. [Google Scholar]
- Endo, A.; van Leeuwen, E.; Baguelin, M.J.E. Introduction to particle Markov-chain Monte Carlo for disease dynamics modellers. Epidemics 2019, 29, 100363. [Google Scholar] [CrossRef]
- King, A.A.; Nguyen, D.; Ionides, E.L. Statistical inference for partially observed Markov processes via the R package pomp. 2015. Available online: https://arxiv.org/abs/1509.00503 (accessed on 6 October 2021).
- CEBM (Center for Evidence-Based Medicine). Declining death rate from COVID-19 in hospitals in England. Available online: https://www.cebm.net/covid-19/declining-death-rate-from-covid-19-in-hospitals-in-england/ (accessed on 6 October 2021).
- NJDOH. Has the State Used Data to Make Decisions and Slow the Spread of COVID-19? Available online: https://covid19.nj.gov/faqs/nj-information/reopening-guidance-and-restrictions/has-the-state-used-data-to-make-decisions-and-slow-the-spread-of-covid-19 (accessed on 6 October 2021).
- Safarishahrbijari, A.; Teyhouee, A.; Waldner, C.; Liu, J.; Osgood, N.D. Predictive accuracy of particle filtering in dynamic models supporting outbreak projections. BMC Infect. Dis. 2017, 17, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Ionides, E.L.; Nguyen, D.; Atchadé, Y.; Stoev, S.; King, A.A. Inference for dynamic and latent variable models via iterated, perturbed Bayes maps. Proc. Natl. Acad. Sci. USA 2015, 112, 719–724. [Google Scholar] [CrossRef] [Green Version]
- Sy, K.T.L.; White, L.F.; Nichols, B.E. Population density and basic reproductive number of COVID-19 across United States counties. medRxiv 2020, 1–17. Available online: https://www.medrxiv.org/content/10.1101/2020.1106.1112.20130021v20130021 (accessed on 6 October 2021).
- Anderson, R.M.; Anderson, B.; May, R.M. Infectious Diseases of Humans: Dynamics and Control; Oxford University Press, 1992; Available online: https://aasldpubs.onlinelibrary.wiley.com/doi/abs/10.1002/hep.1840150131 (accessed on 6 October 2021).
- Hu, H.; Nigmatulina, K.; Eckhoff, P. The scaling of contact rates with population density for the infectious disease models. Math. Biosci. 2013, 244, 125–134. [Google Scholar] [CrossRef]
- Grave, M.; Viguerie, A.; Barros, G.F.; Reali, A.; Coutinho, A.L. Assessing the spatio-temporal spread of COVID-19 via compartmental models with diffusion in Italy, USA, and Brazil. arXiv 2021, arXiv:2102.07208. [Google Scholar] [CrossRef]
- Delamater, P.L.; Street, E.J.; Leslie, T.F.; Yang, Y.T.; Jacobsen, K.H. Complexity of the basic reproduction number (R0). Emerg. Infect. Dis. 2019, 25, 1–4. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Gayle, A.A.; Wilder-Smith, A.; Rocklöv, J. The reproductive number of COVID-19 is higher compared to SARS coronavirus. J. Travel Med. 2020, 27, 1–4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanche, S.; Lin, Y.T.; Xu, C.; Romero-Severson, E.; Hengartner, N.; Ke, R. High Contagiousness and Rapid Spread of Severe Acute Respiratory Syndrome Coronavirus 2. Emerg. Infect. Dis. 2020, 26, 1470–1477. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Li, M.; Lv, G.; Lu, Z.K. Monitoring Transmissibility and Mortality of COVID-19 in Europe. Int. J. Infect. Dis. 2020, 95, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Ridenhour, B.; Kowalik, J.M.; Shay, D.K. Unraveling R0: Considerations for public health applications. Am. J. Public Health 2018, 108, S445–S454. [Google Scholar] [CrossRef]
- Nishiura, H.; Chowell, G. The effective reproduction number as a prelude to statistical estimation of time-dependent epidemic trends. In Mathematical and Statistical Estimation Approaches in Epidemiology; Springer: Berlin/Heidelberg, Germany, 2009; pp. 103–121. [Google Scholar]
- NJDOH. New Jersey COVID-19 Dashboard. Available online: https://www.nj.gov/health/cd/topics/covid2019_dashboard.shtml (accessed on 6 October 2021).
- Obadia, T.; Haneef, R.; Boëlle, P.-Y. The R0 package: A toolbox to estimate reproduction numbers for epidemic outbreaks. BMC Med. Inform. Decis. Mak. 2012, 12, 1–9. [Google Scholar] [CrossRef]
- rt.live. New Jersey Effective Reproduction Rate (Rt). Available online: https://rt.live/us/NJ (accessed on 6 October 2021).
- Zhang, R.; Li, Y.; Zhang, A.L.; Wang, Y.; Molina, M. Identifying airborne transmission as the dominant route for the spread of COVID-19. Proc. Natl. Acad. Sci. USA 2020, 117, 14857–14863. [Google Scholar] [CrossRef]
- Fisher, K.A.; Barile, J.P.; Guerin, R.J.; Esschert, K.L.V.; Jeffers, A.; Tian, L.H.; Garcia-Williams, A.; Gurbaxani, B.; Thompson, W.W.; Prue, C.E. Factors associated with cloth face covering use among adults during the COVID-19 pandemic—United States, April and May 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 933–937. [Google Scholar] [CrossRef] [PubMed]
- Davies, N.G.; Klepac, P.; Liu, Y.; Prem, K.; Jit, M.; Eggo, R.M.; CMMID COVID-19 working group. Age-dependent effects in the transmission and control of COVID-19 epidemics. Nat. Med. 2020, 26, 1205–1211. [Google Scholar] [CrossRef]
- Kraemer, M.U.; Yang, C.-H.; Gutierrez, B.; Wu, C.-H.; Klein, B.; Pigott, D.M.; Open COVID-19 Data Working Group; du Plessis, L.; Faria, N.R.; Li, R. The effect of human mobility and control measures on the COVID-19 epidemic in China. Science 2020, 368, 493–497. [Google Scholar] [CrossRef] [Green Version]
- Pei, S.; Kandula, S.; Shaman, J. Differential effects of intervention timing on COVID-19 spread in the United States. Sci. Adv. 2020, 6, eabd6370. [Google Scholar] [CrossRef] [PubMed]
- Naughton, S.X.; Raval, U.; Harary, J.M.; Pasinetti, G.M. The role of the exposome in promoting resilience or susceptibility after SARS-CoV-2 infection. J. Expo. Sci. Environ. Epidemiol. 2020, 30, 776–777. [Google Scholar] [CrossRef] [PubMed]
- Georgopoulos, P.G.; Ren, X.; Mi, Z. Socioexposomics of COVID-19: The Case of New Jersey. In Proceedings of the International Society of Exposure Science (ISES) Conference, Virtual Conference, 30 August–2 September 2021. [Google Scholar]
- Dashtbali, M.; Mirzaie, M. A compartmental model that predicts the effect of social distancing and vaccination on controlling COVID-19. Sci. Rep. 2021, 11, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Moghadas, S.M.; Vilches, T.N.; Zhang, K.; Wells, C.R.; Shoukat, A.; Singer, B.H.; Meyers, L.A.; Neuzil, K.M.; Langley, J.M.; Fitzpatrick, M.C. The impact of vaccination on COVID-19 outbreaks in the United States. medRxiv 2021. [Google Scholar] [CrossRef]
- Rachaniotis, N.P.; Dasaklis, T.K.; Fotopoulos, F.; Tinios, P. A two-phase stochastic dynamic model for COVID-19 mid-term policy recommendations in Greece: A pathway towards mass vaccination. Int. J. Environ. Res. Public Health 2021, 18, 2497. [Google Scholar] [CrossRef] [PubMed]









| County/ State | Total Deaths | Deaths per 100,000 |  | Geographic Region | Total Deaths | Deaths per 100,000 |  |
|---|---|---|---|---|---|---|---|
| New Jersey | 14,068 | 158 |  | ||||
| Bergen | 1768 | 189 |  | Gateway Region | 8458 | 197 |  |
| Essex | 1868 | 234 |  | ||||
| Hudson | 1336 | 198 |  | ||||
| Middlesex | 1218 | 147 |  | ||||
| Passaic | 1098 | 218 |  | ||||
| Union | 1170 | 210 |  | ||||
| Monmouth | 763 | 123 |  | Shore Region | 1715 | 140 |  |
| Ocean | 952 | 158 |  | ||||
| Hunterdon | 69 | 55 |  | Skyland Region | 1559 | 130 |  |
| Morris | 684 | 138 |  | ||||
| Somerset | 487 | 147 |  | ||||
| Sussex | 159 | 113 |  | ||||
| Warren | 160 | 151 |  | ||||
| Burlington | 440 | 99 |  | Delaware River Region | 1866 | 111 |  |
| Camden | 541 | 107 |  | ||||
| Gloucester | 211 | 72 |  | ||||
| Mercer | 589 | 159 |  | ||||
| Salem | 85 | 136 |  | ||||
| Atlantic | 237 | 89 |  | Southern Shore Region | 470 | 92 |  |
| Cape May | 90 | 97 |  | ||||
| Cumberland | 143 | 95 |  |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ren, X.; Weisel, C.P.; Georgopoulos, P.G. Modeling Effects of Spatial Heterogeneities and Layered Exposure Interventions on the Spread of COVID-19 across New Jersey. Int. J. Environ. Res. Public Health 2021, 18, 11950. https://doi.org/10.3390/ijerph182211950
Ren X, Weisel CP, Georgopoulos PG. Modeling Effects of Spatial Heterogeneities and Layered Exposure Interventions on the Spread of COVID-19 across New Jersey. International Journal of Environmental Research and Public Health. 2021; 18(22):11950. https://doi.org/10.3390/ijerph182211950
Chicago/Turabian StyleRen, Xiang, Clifford P. Weisel, and Panos G. Georgopoulos. 2021. "Modeling Effects of Spatial Heterogeneities and Layered Exposure Interventions on the Spread of COVID-19 across New Jersey" International Journal of Environmental Research and Public Health 18, no. 22: 11950. https://doi.org/10.3390/ijerph182211950
APA StyleRen, X., Weisel, C. P., & Georgopoulos, P. G. (2021). Modeling Effects of Spatial Heterogeneities and Layered Exposure Interventions on the Spread of COVID-19 across New Jersey. International Journal of Environmental Research and Public Health, 18(22), 11950. https://doi.org/10.3390/ijerph182211950






