Novel Insights on the Role of Nitric Oxide in the Ovary: A Review of the Literature
Abstract
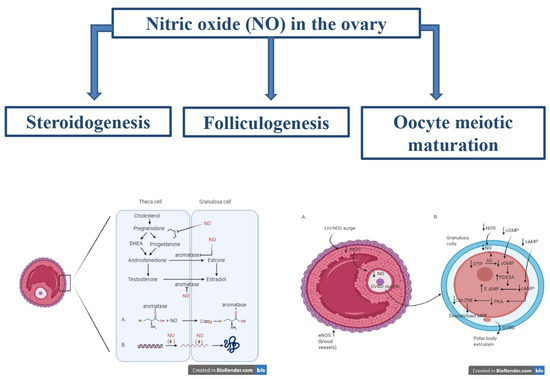
:1. Introduction
2. Nitric Oxide Roles
2.1. NOS Expression in the Ovary
2.2. Nitric Oxide and Steroidogenesis
2.3. Nitric Oxide and Folliculogenesis
2.4. Nitric Oxide and Oocyte Meiotic Maturation
2.5. Nitric Oxide and IVF
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Wu, G.; Morris, S.M. Arginine metabolism: Nitric oxide and beyond. Biochem. J. 1998, 15, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Basini, G.; Grasselli, F. Nitric oxide in follicle development and oocyte competence. Reproduction 2015, 150, R1–R9. [Google Scholar] [CrossRef]
- Morris, S.M.; Billiar, T.R. New insights into the regulation of inducible nitric oxide synthesis. Am. J. Physiol. 1994, 266, E829–E839. [Google Scholar] [CrossRef]
- Khorram, O. Nitric oxide and its role in blastocyst implantation. Rev. Endocr. Metab. Disord. 2002, 3, 145–149. [Google Scholar] [CrossRef] [PubMed]
- Rosselli, M.; Keller, P.J.; Dubey, R.K. Role of nitric oxide in the biology, physiology and pathophysiology of reproduction. Hum. Reprod. Update 1998, 4, 3–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guzik, T.J.; Korbut, R.; Adamek-Guzik, T. Nitric oxide and superoxide in inflammation and immune regulation. J. Physiol. Pharmacol. 2003, 54, 469–487. [Google Scholar]
- Vega, M.; Johnson, M.C.; Díaz, H.A.; Urrutia, L.R.; Troncoso, J.L.; Devoto, L. Regulation of human luteal steroidogenesis in vitro by nitric oxide. Endocrine 1998, 8, 185–191. [Google Scholar] [CrossRef]
- Roy, P.; Ramana, V.G.; Naidu, M.U.R.; Rani, P.U. Recent trends in the nitrergic nervous system. Ind. J. Pharmacol. 2005, 37, 69. [Google Scholar]
- Ducsay, C.A.; Myers, D.A. eNOS activation and NO function: Differential control of steroidogenesis by nitric oxide and its adaptation with hypoxia. J. Endocrinol. 2011, 210, 259–269. [Google Scholar] [CrossRef] [Green Version]
- Dave, S.; Farrance, D.P.; Whitehead, S.A. Evidence that nitric oxide inhibits steroidogenesis in cultured rat granulosa cells. Clin. Sci. 1997, 92, 277–284. [Google Scholar] [CrossRef] [Green Version]
- Nath, P.; Maitra, S. Physiological relevance of nitric oxide in ovarian functions: An overview. Gen. Comp. Endocrinol. 2019, 279, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Grasselli, F.; Ponderato, N.; Basini, G.; Tamanini, C. Nitric oxide synthase expression and nitric oxide/cyclic GMP pathway in swine granulosa cells. Domest. Anim. Endocrinol. 2001, 20, 241–252. [Google Scholar] [CrossRef]
- Van Voorhis, B.J.; Dunn, M.S.; Snyder, G.D.; Weiner, C.P. Nitric oxide: An autocrine regulator of human granulosa-luteal cell steroidogenesis. Endocrinology 1994, 135, 1799–1806. [Google Scholar] [CrossRef] [PubMed]
- Ahsan, S.; Lacey, M.; Whitehead, S.A. Interactions between interleukin-1 beta, nitric oxide and prostaglandin E2 in the rat ovary: Effects on steroidogenesis. Eur. J. Endocrinol. 1997, 137, 293–300. [Google Scholar] [CrossRef] [Green Version]
- Mitsube, K.; Mikuni, M.; Matousek, M.; Brännström, M. Effects of a nitric oxide donor and nitric oxide synthase inhibitors on luteinizing hormone-induced ovulation in the ex-vivo perfused rat ovary. Hum. Reprod. 1999, 14, 2537–2543. [Google Scholar] [CrossRef] [Green Version]
- Dong, Y.L.; Gangula, P.R.; Fang, L.; Yallampalli, C. Nitric oxide reverses prostaglandin-induced inhibition in ovarian progesterone secretion in rats. Hum. Reprod. 1999, 14, 27–32. [Google Scholar] [CrossRef] [Green Version]
- Masuda, M.; Kubota, T.; Aso, T. Effects of nitric oxide on steroidogenesis in porcine granulosa cells during different stages of follicular development. Eur. J. Endocrinol. 2001, 144, 303–308. [Google Scholar] [CrossRef] [Green Version]
- Kagabu, S.; Kodama, H.; Fukuda, J.; Karube, A.; Murata, M.; Tanaka, T. Inhibitory effects of nitric oxide on the expression and activity of aromatase in human granulosa cells. Mol. Hum. Reprod. 1999, 5, 396–401. [Google Scholar] [CrossRef] [Green Version]
- Basini, G.; Tamanini, C. Interrelationship between nitric oxide and prostaglandins in bovine granulosa cells. Prostaglandins. Other. Lipid. Mediat. 2001, 66, 179–202. [Google Scholar] [CrossRef]
- Lin, L.L.; Wartmann, M.; Lin, A.Y.; Knopf, J.L.; Seth, A.; Davis, R.J. CPLA2 is phosphorylated and activated by MAP Kinase. Cell 1993, 72, 269–278. [Google Scholar] [CrossRef]
- Van Voorhis, B.J.; Moore, K.; Strijbos, P.J.; Nelson, S.; Baylis, S.A.; Grzybicki, D.; Weiner, C.P. Expression and localization of inducible and endothelial nitric oxide synthase in the rat ovary. Effects of gonadotropin stimulation in vivo. J. Clin. Investig. 1995, 96, 2719–2726. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jablonka-Shariff, A.; Olson, L.M. Hormonal regulation of nitric oxide synthases and their cell-specific expression during follicular development in the rat ovary. Endocrinology 1997, 138, 460–468. [Google Scholar] [CrossRef] [PubMed]
- Basini, G.; Baratta, M.; Ponderato, N.; Bussolati, S.; Tamanini, C. Is nitric oxide an autocrine modulator of bovine granulosa cell function? Reprod. Fertil. Dev. 1998, 10, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Matsumi, H.; Koji, T.; Yano, T.; Yano, N.; Tsutsumi, O.; Momoeda, M.; Osuga, Y.; Taketani, Y. Evidence for an inverse relationship between apoptosis and inducible nitric oxide synthase expression in rat granulosa cells: A possible role of nitric oxide in ovarian follicle atresia. Endocr. J. 1998, 45, 745–751. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Q.; Yano, T.; Matsumi, H.; Osuga, Y.; Yano, N.; Xu, J.; Wada, O.; Koga, K.; Fujiwara, T.; Kugu, K.; et al. Cross-Talk between Fas/Fas ligand system and nitric oxide in the pathway subserving granulosa cell apoptosis: A possible regulatory mechanism for ovarian follicle atresia. Endocrinology 2005, 146, 808–815. [Google Scholar] [CrossRef] [Green Version]
- Dineva, J.D.; Vangelov, I.M.; Nikolov, G.G.; Konakchieva, R.Ts.; Ivanova, M.D. Nitric oxide stimulates the production of atrial natriuretic peptide and progesterone by human granulosa luteinized cells with an antiapoptotic effect. Endocr. Regul. 2008, 42, 45–51. [Google Scholar]
- Zamberlam, G.; Portela, V.; De Oliveira, J.F.; Gonçalves, P.B.; Price, C.A. Regulation of inducible nitric oxide synthase expression in bovine ovarian granulosa cells. Mol. Cell. Endocrinol. 2011, 335, 189–194. [Google Scholar] [CrossRef]
- Matsumi, H.; Yano, T.; Osuga, Y.; Kugu, K.; Tang, X.; Xu, J.P.; Yano, N.; Kurashima, Y.; Ogura, T.; Taketani, Y.; et al. Regulation of nitric oxide synthase to promote cytostasis in ovarian follicular development. Biol. Reprod. 2000, 63, 141–146. [Google Scholar] [CrossRef] [Green Version]
- Rong, L.; Albertini, D.F. The road to maturation: Somatic cell interaction and self-organization of the mammalian oocyte. Nat. Rev. Mol. Cell. Biol. 2013, 14, 141–152. [Google Scholar]
- Albertini, D.F.; Sanfins, A.; Combelles, C.M. Origins and manifestations of oocyte maturation competencies. Reprod. Biomed. Online 2003, 6, 410–415. [Google Scholar] [CrossRef]
- Larose, H.; Shami, A.N.; Abbott, H.; Manske, G.; Lei, L.; Hammoud, S.S. Gametogenesis: A journey from inception to conception. Curr. Top. Dev. Biol. 2019, 132, 257–310. [Google Scholar] [PubMed]
- Nakamura, Y.; Yamagata, Y.; Sugino, N.; Takayama, H.; Kato, H. Nitric oxide inhibits oocyte meiotic maturation. Biol. Reprod. 2002, 67, 1588–1592. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hattori, M.; Nishida, N.; Takesue, K.; Kato, Y.; Fujihara, N. FSH suppression of nitric oxide synthesis in porcine oocytes. J. Mol. Endocrinol. 2000, 24, 65–73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamagata, Y.; Nakamura, Y.; Sugino, N.; Harada, A.; Takayama, H.; Kashida, S.; Kato, H. Alterations in nitrate/nitrite and nitric oxide synthase in preovulatory follicles in gonadotropin-primed immature rat. Endocrinology 2002, 49, 219–226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jablonka-Shariff, A.; Olson, L.M. The role of nitric oxide in oocyte meiotic maturation and ovulation: Meiotic abnormalities of endothelial nitric oxide synthase knock-out mouse oocytes. Endocrinology 1998, 139, 2944–2954. [Google Scholar] [CrossRef]
- Jablonka-Shariff, A.; Basuray, R.; Olson, L.M. Inhibitors of nitric oxide synthase influence oocyte maturation in rats. J. Soc. Gynecol. Investig. 1999, 6, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Jablonka-Shariff, A.; Olson, L.M. Nitric oxide is essential for optimal meiotic maturation of murine cumulus-oocyte complexes in vitro. Mol. Reprod. Dev. 2000, 55, 412–421. [Google Scholar] [CrossRef]
- Sengoku, K.; Takuma, N.; Horikawa, M.; Tsuchiya, K.; Komori, H.; Sharifa, D.; Tamate, K.; Ishikawa, M. Requirement of nitric oxide for murine oocyte maturation, embryo development and trophoblast outgrowth in vitro. Mol. Reprod. Dev. 2001, 58, 262–268. [Google Scholar] [CrossRef]
- Tao, Y.; Fu, Z.; Zhang, M.; Xia, G.; Yang, J.; Xie, H. Immunohistochemical localization of inducible and endothelial nitric oxide synthase in porcine ovaries and effects of NO on antrum formation and oocyte meiotic maturation. Mol. Cell. Endocrinol. 2004, 222, 93–103. [Google Scholar] [CrossRef]
- Tao, Y.; Xie, H.; Hong, H.; Chen, X.; Jang, J.; Xia, G. Effects of nitric oxide synthase inhibitors on porcine oocyte meiotic maturation. Zygote 2005, 13, 1–9. [Google Scholar] [CrossRef]
- Huo, L.J.; Liang, C.; Yu, L.Z.; Zhong, Z.S.; Yang, Z.; Fan, H.Y.; Chen, D.Y.; Sun, Q.Y. Inducible nitric oxide synthase-derived nitric oxide regulates germinal vesicle breakdown and first polar body emission in the mouse oocyte. Reproduction 2005, 129, 403–409. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pandey, A.N.; Chaube, S.K. Reduction of nitric oxide level leads to spontaneous resumption of meiosis in diplotene-arrested rat oocytes cultured in vitro. Exp. Biol. Med. 2015, 240, 15–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bu, S.; Xia, G.; Tao, Y.; Lei, L.; Zhou, B. Dual effects of nitric oxide on meiotic maturation of mouse cumulus cell-enclosed oocytes in vitro. Mol. Cell. Endocrinol. 2003, 207, 21–30. [Google Scholar] [CrossRef]
- Tripathi, A.; Khatun, S.; Pandey, A.N.; Mishra, S.K.; Chaube, R.; Shrivastav, T.G.; Chaube, S.K. Intracellular levels of hydrogen peroxide and nitric oxide in oocytes at various stages of meiotic cell cycle and apoptosis. Free Radic. Res. 2009, 43, 287–294. [Google Scholar] [CrossRef]
- Bu, S.; Xie, H.; Tao, Y.; Wang, J.; Xia, G. Nitric oxide influences the maturation of cumulus cell-enclosed mouse oocytes cultured in spontaneous maturation medium and hypoxanthine-supplemented medium through different signaling pathways. Mol. Cell. Endocrinol. 2004, 223, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Goeseels, S.B. Effects of manipulating the nitric oxide/cyclic GMP pathway on bovine oocyte meiotic resumption in vitro. Theriogenology 2007, 15, 693–701. [Google Scholar] [CrossRef]
- Botigelli, R.C.; Lancellotti Schwarz, K.; Zaffalon, F.G.; Del Collado, M.; Cavallari Castro, F.; Fernandes, H.; Lima Verde Leal, C. Influence of nitric oxide and phosphodiesterases during in vitro maturation of bovine oocytes on meiotic resumption and embryo production. Zygote 2017, 25, 321–330. [Google Scholar] [CrossRef]
- Sela-Abramovich, S.; Galiani, D.; Nevo, N.; Dekel, N. Inhibition of rat oocyte maturation and ovulation by nitric oxide: Mechanism of action. Biol. Reprod. 2008, 78, 1111–1118. [Google Scholar] [CrossRef] [Green Version]
- Kim, B.H.; Kim, C.H.; Jung, K.Y.; Jeon, B.H.; Ju, E.J.; Choo, Y.K. Involvement of nitric oxide during in vitro fertilization and early embryonic development in mice. Arch. Pharm. Res. 2004, 27, 86–93. [Google Scholar] [CrossRef]
- Santana, P.D.; Silva, T.V.; Da Costa, N.N.; Da Silva, B.B.; Carter, T.F.; Cordeiro, S.; Da Silva, B.J.; Santos, S.; Herculano, A.M.; Miranda, S.; et al. Supplementation of bovine embryo culture medium with L-arginine improves embryo quality via nitric oxide production. Mol. Reprod. Dev. 2014, 81, 918–927. [Google Scholar] [CrossRef]
- Barroso, R.P.; Osuamkpe, C.; Nagamani, M.; Yallampalli, C. Nitric oxide inhibits development of embryos and implantation in mice. Mol. Hum. Reprod. 1998, 4, 503–507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tranguch, S.; Steuerwald, N.; Huet-Hudson, Y.M. Nitric oxide synthase production and nitric oxide regulation of preimplantation embryo development. Biol. Reprod. 2003, 68, 1538–1544. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gouge, R.C.; Marshburn, P.; Gordon, B.E.; Nunley, W.; Huet-Hudson, Y.M. Nitric oxide as a regulator of embryonic development. Biol. Reprod. 1998, 58, 875–879. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, H.W.; Jiang, W.S.; Tzeng, C.R. Nitric oxide as a regulator in preimplantation embryo development and apoptosis. Fertil. Steril. 2001, 75, 1163–1171. [Google Scholar] [CrossRef]
- Battaglia, C.; Ciotti, P.; Notarangelo, L.; Fratto, R.; Facchinetti, F.; De Aloysio, D. Embryonic production of nitric oxide and its role in Implantation: A pilot study. J. Assist. Reprod. Genet. 2003, 20, 449–454. [Google Scholar] [CrossRef] [Green Version]
- Lipari, C.W.; Garcia, J.E.; Zhao, Y.; Thrift, K.; Vaidya, D.; Rodriguez, A. Nitric Oxide Metabolite Production in the Human Preimplantation Embryo and Successful Blastocyst Formation. Fertil. Steril. 2009, 91, 1316–1318. [Google Scholar] [CrossRef] [Green Version]
- Gallinelli, A.; NIcoli, A.; Capodanno, F.; Valli, B.; Facchinetti, F.; La Sala, G.B. Nitric oxide as an early marker of human embryo metabolic cleavage in ART using fresh and thawed oocytes. Eur. J. Obstet. Gynecol. Reprod. Biol. 2009, 142, 48–52. [Google Scholar] [CrossRef]
- Ohl, J.; Lefebvre-Maounoury, C.; Wittemer, C.; Nisand, G.; Laurent, M.C.; Hoffmann, P. Nitric oxide donors for patients undergoing IVF. A prospective, double-blind, randomized, placebo-controlled trial. Hum. Reprod. 2002, 17, 2615–2620. [Google Scholar] [CrossRef] [Green Version]
- Lee, K.S.; Joo, B.S.; Na, Y.J.; Yoon, M.S.; Choi, O.H.; Kim, W.W. Relationships between concentrations of tumor necrosis factor-alpha and nitric oxide in follicular fluid and oocyte quality. J. Assist. Reprod. Genet. 2000, 17, 222–228. [Google Scholar] [CrossRef]
- Lee, T.H.; Wu, M.Y.; Chen, M.J.; Chao, K.H.; Ho, H.N.; Yang, Y.S. Nitric oxide is associated with poor embryo quality and pregnancy outcome in in vitro fertilization cycles. Fertil. Steril. 2004, 82, 126–131. [Google Scholar] [CrossRef]
- Vignini, A.; Turi, A.; Giannubilo, S.R.; Pescosolido, D.; Scognamiglio, P.; Zanconi, S.; Silvi, C.; Mazzanti, L.; Tranquilli, A.L. Follicular fluid nitric oxide (NO) concentrations in stimulated cycles: The relationship to embryo grading. Arch. Gynecol. Obstet. 2008, 277, 229–232. [Google Scholar] [CrossRef] [PubMed]
- Bergandi, L.; Basso, G.; Evangelista, F.; Canosa, S.; Dalmasso, P.; Aldieri, E.; Revelli, A.; Benedetto, C.; Ghigo, D. Inducible nitric oxide synthase and heme oxygenase 1 are expressed in human cumulus cells and may be used as biomarkers of oocyte competence. Reprod. Sci. 2014, 21, 1370–1377. [Google Scholar] [CrossRef] [PubMed]
- Dunnam, R.C.; Hill, M.J.; Lawson, D.M.; Dunba, J.C. Ovarian hormone secretory response to gonadotropins and nitric oxide following chronic nitric oxide deficiency in the rat. Biol. Reprod. 1999, 60, 959–963. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fridén, B.E.; Runesson, E.; Hahlin, M.; Brännström, M. Evidence for nitric oxide acting as a luteolytic factor in the human corpus luteum. Mol. Hum. Reprod. 2000, 6, 397–403. [Google Scholar] [CrossRef]
- Fang, L.; Chang, H.M.; Cheng, J.C.; Leung, P.C.; Sun, Y.P. Nitric oxide and cGMP induce COX-2 expression and PGE2 production in human granulosa cells through CREB signaling pathway. J. Clin. Endocrinol. Metab. 2015, 100, E262–E269. [Google Scholar] [CrossRef] [Green Version]
- Banerjee, J.; Shaeib, F.; Maitra, D.; Saed, G.M.; Dai, J.; Diamond, M.P.; Abu-Soud, H.M. Peroxynitrite affects the cumulus cell defense of metaphase II mouse oocytes leading to disruption of the spindle structure in vitro. Fertil. Steril. 2013, 100, 578–584. [Google Scholar] [CrossRef]
- Khan, S.N.; Shaeib, F.; Thakur, M.; Jeelani, R.; Awonuga, A.O.; Goud, P.T.; Abu-Soud, H.M. Peroxynitrite deteriorates oocyte quality through disassembly of microtubule organizing centers. Free Radic. Biol. Med. 2016, 91, 275–280. [Google Scholar] [CrossRef]
- El-Sehemy, A.; Postovit, L.M.; Fu, Y.X. Nitric oxide signaling in human ovarian cancer: A potential therapeutic target. Nitric Oxide 2016, 54, 30–37. [Google Scholar] [CrossRef]
- Goteri, G.; Lucarini, G.; Zizzi, A.; Rubini, C.; Di Primio, R.; Tranquilli, A.L.; Ciavattini, A. Proangiogenetic molecules, hypoxia-inducible factor-1α and nitric oxide synthase isoforms in ovarian endometriotic cysts. Virchows Arch. 2010, 456, 703–710. [Google Scholar] [CrossRef]
- Meng, C. Nitric oxide (NO) levels in patients with polycystic ovary syndrome (PCOS): A meta-analysis. J. Int. Med. Res. 2019, 47, 4083–4094. [Google Scholar] [CrossRef]
- Watanabe, T.; Akishita, M.; Toba, K.; Kozaki, K.; Eto, M.; Sugimoto, N.; Kiuchi, T.; Hashimoto, M.; Shirakawa, W.; Ouchi, Y. Influence of sex and age on serum nitrite/nitrate concentration in healthy subjects. Clin. Chim. Acta. 2000, 301, 169–179. [Google Scholar] [CrossRef]
- Ghasemi, A.; Zahedi Asl, S.; Mehrabi, Y.; Saadat, N.; Azizi, F. Serum nitric oxide metabolite levels in a general healthy population: Relation to sex and age. Life Sci. 2008, 83, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Tehrani, F.R.; Behboudi-Gandevani, S.; Gasemi, A.; Azizi, F. Association between serum concentrations of nitric oxide and transition to menopause. Acta. Obstet. Gynecol. Scand. 2015, 94, 708–714. [Google Scholar] [CrossRef] [PubMed]
- Chedraui, P.; Escobar, G.S.; Ramírez, C.; Pérez-López, F.R.; Hidalgo, L.; Mannella, P.; Genazzani, A.; Simoncini, T. Nitric oxide and pro-inflammatory cytokine serum levels in postmenopausal women with the metabolic syndrome. Gynecol. Endocrinol. 2012, 28, 787–791. [Google Scholar] [CrossRef] [PubMed]
- Habermeyer, M.; Roth, A.; Guth, S.; Diel, P.; Engel, K.H.; Epe, B.; Furst, P.; Heinz, V.; Humpf, H.U.; Eisenbrand, G.; et al. Nitrate and nitrite in the diet: How to assess their benefit and risk for human health. Mol. Nutr. Food Res. 2015, 59, 106–128. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, J.O.; Carlstrom, M.; Larsen, F.J.; Weitzberg, E. Roles of dietary inorganic nitrate in cardiovascular health and disease. Cardiovasc. Res. 2011, 89, 525–532. [Google Scholar] [CrossRef] [PubMed]
- Carter, P.; Gray, L.J.; Troughton, J.; Khunti, K.; Davies, M.J. Fruit and vegetable intake and incidence of type 2 diabetes mellitus: Systematic review and meta-analysis. Brit. Med. J. 2010, 341, c4229. [Google Scholar] [CrossRef] [Green Version]
- Kapil, V.; Milsom, A.B.; Okorie, M.; Maleki-Toyserkani, S.; Akram, F.; Rehman, F.; Arghandawi, S.; Pearl, V.; Benjamin, N.; Loukogeorgakis, S.; et al. Inorganic nitrate supplementation lowers blood pressure in humans: Role for nitrite-derived NO. Hypertension 2010, 56, 274–281. [Google Scholar] [CrossRef] [Green Version]


| Authors | Animal | Model | NO Donors Used | NOS Inhibitors Used | Effects |
|---|---|---|---|---|---|
| Dave et al., 1997 [10] | Rat | In-vitro granulosa/lutein cells culture | -SNP: 10−3 mol/L, 10−4 mol/L -SNAP: 10−3 mol/L, 10−4 mol/L | -Progesterone synthesis inhibition in the presence of the nitric oxide donors in a concentration-dependent manner. | |
| Ahsan et al., 1997 [14] | Rat | In-vitro culture of dispersed ovarian cells | -SNP: 10−4 M and 10−3 M | -Progesterone synthesis in cultures of ovarian dispersates dose dependently inhibited by SNP and inversely related to the concentrations of nitrites measured in the culture medium. -Progesterone production stimulated by PGE2 (5 mM). | |
| Mitsube et al., 1999 [15] | Rat | Ex-vivo perfused pre-ovulatory ovary | -Spermine NONOate: 10 µmol/L and 100 µmol/L | -L-NMMA: 300 µmol/L -AG: 300 µmol/L and 1 mmol/L | -Steroid concentration in the perfusion media not affected by the NO inhibitors. -NONOate (100 µmol/L) increased progesterone production. |
| Dong et al., 1999 [16] | Rat | In-vitro cultured ovaries | -DETA/NO: 10–6, 10–5, 10–4 M -DETA: 10–4M | -L-NAME: 10–4 M | -Dose-dependent increase in progesterone synthesis, with a concomitant decrease in ovarian oestradiol secretion caused by DETA/NO. -No effects on progesterone and oestradiol secretion by L-NAME (10−4 M). |
| Masuda et al., 2001 [17] | Porcine | In-vitro granulosa cells derived from small-sized (<3 mm) and medium-sized (3–5 mm) ovarian follicles | -NOC18: 0.01–1.0 mmol/L | -LNMMA: 0.01–1.0 mmol/L | -NOC18 suppressed the basal and gonadotropin-stimulated E2 and inhibited progesterone release from small-sized follicles. -No significant effect on progesterone release from medium-sized follicles by NOC18. -Significant release of E2 and progesterone from both small-sized and medium-sized follicles in the presence of gonadotropin by LNMMA. |
| Van-Voorhis et al., 1994 [13] | Human | In-vitro granulosa-luteal cell | -SNAP -S-nitroso glutathione | -LNMMA -N-nitro-arginase methyl ester. | -Inhibitors of NO synthase significantly increased estradiol secretion by granulosa-luteal cells. -NO donors caused a dose-dependent decrease in both estradiol and progesterone secretion. |
| Kagabu et al., 1999 [18] | Human | In-vitro ovarian granulosa-luteal cells | -SNAP: 10−5 M, 10−4 M, 10−3 M -NOC12: 10−4 M, 10−3M | -Aromatase activity significantly inhibited by treatment with SNAP (10−4 M, 10−3 M) and NOC12 (10−4 M, 10−3 M) in a dose-dependent manner. -Treatment with SNAP at 10−3 M decreased relative aromatase mRNA values and intracellular cyclic AMP concentrations. |
| Authors | Animal | Model | NO Donors Used | NOS Inhibitors Used | Effects |
|---|---|---|---|---|---|
| Van Voorhish et al., 1995 [21] | Rat | In vivo | -Highest levels of iNOS mRNA in unstimulated ovaries. -After gonadotropin injection, iNOS mRNA declined to undetectable levels in ovaries containing ovulatory follicles before increasing slightly in ovaries containing copora lutea. -iNOS expressed in granulosa cells of secondary follicles and small antral follicles. -eNOS mRNA levels increased after gonadotropin stimulation and peaked in ovaries containing ovulatory follicles before declining in the luteal phase. | ||
| Jablonka-Shariff et al., 1997 [22] | Rat | In vivo | -In immature ovaries and during follicular development, iNOS was expressed in the theca cell layer and stroma. -After ovulation, iNOS was present only in the external layers of the developing corpus luteum. | ||
| Basini et al., 1998 [23] | Bovine | In vitro culture of granulosa cells collected from follicles divided according to their size, small (<5 mm) and large (>8 mm). | -SNAP: 10−3 M, 10−4 M, 10−5M | -Significant inhibition of DNA fragmentation in all the cells with the highest concentration of SNAP. -Stimulation of cellular apoptosis in granulosa cells from large follicles with the lowest concentration of SNAP. | |
| Matsumi et al., 1998 [24] | Rat | In vitro culture of granulosa cells | -SNAP: 0.05 mM, 0.5 mM | -Inverse correlation between iNOS expression and apoptotic changes in rat granulosa cells. -The NO donor directly inhibited spontaneously occurring apoptosis. | |
| Chen et al., 2005 [25] | Rat | In vitro culture of granulosa cells | -SNAP: 0.05 mM, 0.2 mM, 0.5 mM | -The induction of apoptosis in granulosa cells by 100 ng/ml rFasL in the presence of interferon-gamma was blocked by the concomitant addition of SNAP in a dose-dependent manner. -rFasL significantly up-regulated caspase-3, -8, and -9 activities in granulosa cells, which were attenuated by concurrent treatment with SNAP (0.5 mM). | |
| Dineva et al., 2008 [26] | Human | In vitro human granulosa/luteinized cells | -SNP: 0.5 mM | -AG: 0.5 mM | -SNP treatment significantly lowered the caspase-3 activity that significantly increased after AG treatment. |
| Matsumi et al., 2000 [28] | Rat | In vitro culture of granulosa cells | -SNAP: 0.05 mM, 0.2 mM, 0.5 mM | -The in vitro induction of granulosa cell apoptosis by 10−6 M buserelin was inhibited by the addition of SNAP (0.5 mM) -The percentage of apoptotic cells increased by the addition of 10−6 M buserelin but this increase was reverted by supplementation of SNAP (0.5 mM) -Dose-dependent inhibition of DNA synthesis mediated by SNAP. At 0.5 mM, SNAP caused a 90% inhibition in DNA synthesis. |
| Authors | Animal | Model | NO Donors Used | NOS Inhibitors Used | Effects |
|---|---|---|---|---|---|
| Nakamura et al., 2002 [32] | Rat | In-vitro cultured ovaries and denuded oocytes | -SNAP: 500 μM | -AG; 0, 1 mM, 10 mM, 100 mM | -The percentage of oocytes at the germinal vesicle stage decreased in the group receiving 10 and 100 mM AG compared with the control group after 5 h of incubation. This GVBD-promoting effect of 100 mM AG was significantly reversed by the addition of 500 μM SNAP. -SNAP dose-dependently inhibited GVBD in denuded oocytes, and this effect of SNAP was reversed by the addition of hemoglobin |
| Yamagata et al., 2002 [34] | Rat | In vitro culture of granulosa cell and theca cell layers | -SNAP: 10 −5 M, 10 −4 M, 5 × 10−4 M | -Significant decrease in nitrate/nitrite concentration in the follicular fluid 5 and 10 h after hCG injection. -Significant decrease in iNOS mRNA expression 5 and 10 h after hCG injection. eNOS mRNA expression increased 5 and 10 h after hCG injection. -Decreased progesterone production and increased DNA fragmentation of granulosa cells with SNAP (10−4 M or 5 × 10−4 M). | |
| Jablonka-Shariff et al., 1998 [35] | Mouse | In vivo (mice knock-out for the eNOS gene) | -eNOS knock-out females showed a significant reduction in ovulatory efficiency compared with wild type females. -eNOS deficiency impaired not only ovulation, but also oocyte meiotic maturation. -Fewer oocytes from eNOS knock-out mice entered metaphase II of meiosis, with a greater percentage in metaphase I or morphologically atypical relative to those in wild type mice. | ||
| Jablonka-Shariff et al., 1999 [36] | Rat | In-vivo | -L-NAME: 100 mg/kg administered orally -L-NIL, 30 mg/kg administered orally | -Fewer ovulated oocytes at metaphase II obtained from rats treated with NOS inhibitors, and a significantly greater percentage of oocytes displayed atypical morphology as compared with control oocytes. | |
| Jablonka-Shariff et al., 2000 [37] | Mouse | In-vitro cultured COCs | -L-NAME: 0.1 mM, 0.5 mM, or 1 mM | -eNOS-KO mice contained fewer COCs relative to wild type mice. Maturation of COCs from eNOS-KO mice or wild type oocytes treated with L-NAME resulted in a lower percentage of oocytes at M II stage and a higher percentage of oocytes at M I or atypical stages compared with those from WT. - Abnormalities in the distribution of maternal chromosomes in MII stage-oocytes. | |
| Sengoku et al., 2001 [38] | Mouse | In-vitro culture of oocytes/embryos | -SNP: ranging from 10−3 M to 10−7 M ÿ | -L-NAME: ranging from 10−3 M to 10−7 M | -Low concentrations of SNP (10−7 M) significantly stimulated meiotic maturation to metaphase II stages in CEOs. -L-NAME (10−3 M and 10−5 M) demonstrated a significant suppression in the resumption of meiosis. This inhibition was reversed by the addition of SNP. -No development beyond the four-cell stage was observed by the addition of a high concentration of SNP (10−3 M). -Inhibition of embryo development, especially the conversion of morulae to blastocysts, was also observed in the treatment with lower doses of SNP (10−5 and 10−7 M). -Inhibition of NO by an NOS inhibitor resulted in the dose-dependent inhibition of embryo development and hatching rates, but the concomitant addition of SNP with L-NAME reversed the inhibitory effect by each SNP or L-NAME treatment. -A low concentration of SNP (10−7 M) but not a high concentration of SNP (10−3 M) significantly stimulated trophoblast outgrowth, whereas the addition of L-NAME suppressed the spreading of blastocysts in a dose-dependent manner |
| Tao et al., 2004 [39] | Porcine | In-vitro cultured pre-antral follicles, COCs aspirated from medium follicles (3–6 mm in diameter), ovarian tissues, CL, CA and COCs from small (1–2 mm in diameter), medium (3–6 mm), and large follicles (7–10 mm) | -SNP: 0.1 M, 0.3 M, 0.5 M or 1 mM | -AG: 10 mM -L-NAME: 1 mM | -0.3, 0.5 or 1 mM SNP significantly inhibited antrum formation. -AG markedly inhibited porcine oocyte meiotic resumption while L-NAME inhibited first polar body (PB1) extrusion. -Porcine ovaries had distinct cell-specific expression of both eNOS and iNOS. |
| Tao et al., 2005 [40] | Porcine | In- vitro cultured CEOs and DOs. | -AG -L-NNA -L-NAME | -AG suppressed cumulus expansion and inhibited CEOs to resume meiosis but did not inhibit cumulus cell DNA fragmentation. -LNNA and L-NAME delayed cumulus expansion, inhibited cumulus cell DNA fragmentation, and inhibited CEOs to resume meiosis. -No effects on DO. | |
| Huo et al., 2005 [41] | Mouse | In vitro culture of denuded and cumulus-enclosed GV-intact oocytes | -AG, 0, 1 mM, 10 mM, 50 mM | -AG significantly blocked the GVBD of the DOs in a dose-dependent manner. -AG blocked the PB1 emission of oocytes in a dose-dependent manner. | |
| Pandey et al., 2015 [42] | Rat | In-vitro cultured oocytes | -SNAP: 0.1 mmol/L, 0.2 mmol/L, 0.3 mmol/L, and 0.4 mmol/L | -AG 0.0, 2.5 µmol/L, 5.0 µmol/L, 10.0 µmol/L, 20.0 µmol/L, and 40.0 µmol/L | -SNAP inhibited the spontaneous resumption of meiosis from diplotene arrest in a concentration-dependent manner. -AG induced the spontaneous resumption of meiosis in a concentration-dependent manner. -AG treatment resulted in a significant reduction in the level of iNOS expression in oocytes undergoing resumption of meiosis after 3 h of in vitro culture. |
| Bu et al., 2003 [43] | Mouse | In-vitro cultured CEOs and DOs | -SNP: ranging from 0.1-4 mM | -L-NAME: ranging from 10−5 M to 10−2 M -L-NNA: ranging from 10−5 M to 10−2 M. | -Low concentrations of SNP (10−7, 10−6, 10−5 M) significantly stimulated the oocyte meiotic maturation of CEOs but had no effect on DOs. -High concentrations of SNP (0.1 mM–4 mM) in the CEOs cultured in maturation medium resulted in a lower percentage of oocytes at the PB1 stage and a higher percentage of atypical oocytes in a dose-dependent manner compared with the control. -Treatment with SNP (1 mM) resulted in a significant inhibitory effect on the formation of PB1. -The concentration of SNP (1 mM) significantly delayed GVBD during the first 5 h of the culture period. The concomitant addition of L-NAME with SNP did not reverse the inhibitory effect of SNP on CEOs. |
| Tripathi et al., 2009 [44] | Rat | In-vitro culture of oocytes | -Tonic level of H2O2 induced the meiotic resumption of diplotene-arrested oocytes and a further increase may have led to apoptosis. -Reduction in iNOS expression and total nitrite level associated with meiotic resumption in diplotene-arrested oocytes, but induced apoptosis in aged oocytes. | ||
| Bu et al., 2004 [45] | Mouse | In-vitro cultured CEOs | -SNP: 1 mM, 10 µM. | -SNP (1 mM) significantly delayed GVBD during the first 5 h of the incubation period -SNP (1 mM) inhibited the formation of PB1 at the end of 24 h of incubation. -SNP (10µM) stimulated the meiotic maturation of oocytes by overcoming the inhibition of hypoxantine. | |
| Groessels et al., 2007 [46] | Bovine | In vitro cultured CEOs | -SNP: 100 µM, 500 µM | -AG: 1 mM,10 mM and 50 mM | -AG (10 and 50 mM) and SNP (100 and 500 mM) significantly inhibited GVBD after 7 h of culture. -SNP (0.01 mM) stimulated GVBD. |
| Authors | Model | NO Donors Used | NOS Inhibitors Used | Effects |
|---|---|---|---|---|
| Kim et al., 2004 [49] | Mice (In vivo) | -L-NAME (0.5, 1, 5 and 10 mM) | -L-NAME inhibited the fertilization rate and the early embryonic development by treating sperm or oocytes. -Fertilization rate and early embryonic development were reduced when L-NAME or L-arginine was added to the culture media of embryos. -Microinjection of L-NAME into the fertilized embryos inhibited in a dose-dependent manner early embryonic development, but only by high concentrations of L-arginine. | |
| Santana et al., 2014 [50] | Bovine (in vitro embryo culture) | -L-NAME (10 mM) -L-arginine (1 mM, 10 mM, or 50 mM) | -Supplementation with L-NAME from Day 1 to 8 of the culture decreased blastocyst and hatching rates. -L-arginine (50 mM) added from Day 1 to Day 8 decreased the blastocyst rates; in contrast, when added from Day 5 to 8, L-arginine (1 mM) improved the embryo hatching rates and quality. -Positive correlation between NO levels in the medium during this culture period and increased embryo hatching rates and quality. | |
| Barroso et al., 1998 [51] | Mice (In vitro embryo culture and in vivo) | -DETA/NO: 0.001 mM, 0.01 mM, 0.1 mM, 1mM -DETA: 0.1 mM, 1 mM In-vivo: S.c. implantation of miniosmotic pumps containing either saline or different concentrations of DETA/NO or DETA (0.2, 0.4, and 0.8 mM) to deliver a daily dose of 5, 10 or 20 µmol per animal. | -None of the embryos progressed beyond the 4-cell stage when exposed to DETA/NO (0.1 or 1.0 mM). -Embryo development unaffected by lower (0.001 and 0.01 mM) concentrations of DETA/NO, after 48 h preincubation with DETA/NO or DETA only. -Embryo implantation inhibition with the infusion of DETA/NO in a dose-dependent manner. -No implantation sites observed with the infusion of a daily dose of 20 µmol DETA/NO, compared with the control or DETA-treated mice. | |
| Tranguch et al., 2003 [52] | Mice (In vitro embryo culture) | -SNP: from 0.1 to 500 mM | -l-NA: from 125 to 500 mM | -All three NOS isoforms were expressed in two-cell, four-cell, morula, and blastocyst embryos. -Blastocyst-stage embryos isolated on the midmorning of Day 4 of pregnancy expressed only nNOS and eNOS, whereas those isolated in the midafternoon expressed all three NOS isoforms. |
| Gouge et al., 1998 [53] | Mice (in vitro embryo culture) | -l-NA: 500 µM | -Preimplantation murine embryos produced NO, reversibly inhibited by the culture of embryos in medium containing L-NA. -L-NA inhibits normal embryo development. | |
| Chen et al., 2001 [54] | Mice (in vitro embryo culture) | -SNP: 0.1 µM, 1 µM, 10 µM | -L-NAME: 0.1 µM, 1 µM, 10 µM | -L-NAME inhibited blastocyst development in a concentration-dependent manner and SNP (0.1 µM) reversed this effect. -Excessive NO (> or = 10 µM) induced apoptosis in the mouse embryos. -The inhibitory effect of L-NAME was reversed by 8-Br-cGMP that rescued the embryo growth. -ODQ inhibited the embryo development in a dose-dependent manner (0.1 µM–100 µM) but had no effect on NO-induced embryo apoptosis. |
| Authors | Type of Study | Population Considered | Effects |
|---|---|---|---|
| Battaglia et al., 2003 [55] | Observational (pilot) study | -23 women undergoing an IVF-ET program | -Embryonic secretion of NO demonstrated by higher mean nitrite/nitrate concentrations present in the culture medium of each embryo -The mean nitrite/nitrate concentrations in the embryo culture medium were significantly higher in patients that became pregnant. |
| Lipari et al., 2009 [56] | Observational study | -11 women undergoing an IVF-ET program | -Nitric oxide metabolite levels in the insemination media were higher in embryos that progressed to blastocysts by culture day 5 compared with those that did not. |
| Gallinelli et al., 2008 [57] | Study population | -179 women undergoing an IVF-ET program: -123 women with fresh oocytes. -56 oocyte thawing cycles | -Higher NO production in embryos derived from ICSI than from IVF after 52 h of culture. -Embryos derived from fresh oocytes produced more NO than embryos from thawed oocytes after 48 and 52 h of culture. |
| Ohl et al., 2002 [58] | Prospective, double-blind, randomized, placebo-controlled trial | -138 patients undergoing an IVF-ET program: -70 patients treated with nitroglycerin-68 with placebo | -No improved implantation or pregnancy rates after NTG treatment on the day before embryo transfer. |
| Lee et al., 2000 [59] | Observational study | -43 patients undergoing an IVF-ET program | -No significant correlation between the concentrations of NO and TNF-alpha in follicular fluid. -NO levels in follicular fluid were altered in infertility-associated diseases. -TNF-alpha levels but not NO levels influenced oocyte quality. |
| Lee et al., 2004 [60] | Prospective, case-control study | -36 patients undergoing an IVF-ET program: -18 patients undergoing an IVF-ET program with tubal or peritoneal factor -18 female partners from couples with male factor infertility | -Higher follicular NO levels associated with advanced fragmentation of embryos. -Higher serum NO levels found among non-pregnant patients with tubal or peritoneal factor infertility. |
| Vignini et al., 2008 [61] | Observational study | -15 patients undergoing an IVF-ET program | -Mean concentration of the NO follicular fluid was significantly higher in patients with embryos showing significant or severe fragmentation or blastomeres of distinctively unequal size than those with good-quality embryos. -Direct correlation between follicular NO and embryo grading and an inverse correlation between follicular NO and serum 17beta-estradiol. |
| Bergandi et al., 2014 [62] | Observational study | -40 women undergoing an IVF-ET program | -iNOS and HO-1 mRNAs and proteins significantly higher in cumulus cells (CCs) corresponding to oocytes that were not fertilized in comparison to CCs whose corresponding oocytes showed normal fertilization. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Budani, M.C.; Tiboni, G.M. Novel Insights on the Role of Nitric Oxide in the Ovary: A Review of the Literature. Int. J. Environ. Res. Public Health 2021, 18, 980. https://doi.org/10.3390/ijerph18030980
Budani MC, Tiboni GM. Novel Insights on the Role of Nitric Oxide in the Ovary: A Review of the Literature. International Journal of Environmental Research and Public Health. 2021; 18(3):980. https://doi.org/10.3390/ijerph18030980
Chicago/Turabian StyleBudani, Maria Cristina, and Gian Mario Tiboni. 2021. "Novel Insights on the Role of Nitric Oxide in the Ovary: A Review of the Literature" International Journal of Environmental Research and Public Health 18, no. 3: 980. https://doi.org/10.3390/ijerph18030980