Nutshells as Efficient Biosorbents to Remove Cadmium, Lead, and Mercury from Contaminated Solutions
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Biosorbents Preparation
2.3. Nutshell Characterization by Fourier-Transform Infrared Spectroscopy
2.4. Design of the Sorption Experiments
2.5. Quantification of Cadmium, Lead, and Mercury in Solution
2.6. Kinetic Modelling Applied to the Results
3. Results
3.1. Best Nutshells to Remove Cd, Pb, and Hg from Spiked Ultrapure Water
3.2. Effect of Solution Chemistry on Removal of Cd, Hg, and Pb by Hazelnuts
3.3. Effect of Washing Procedure
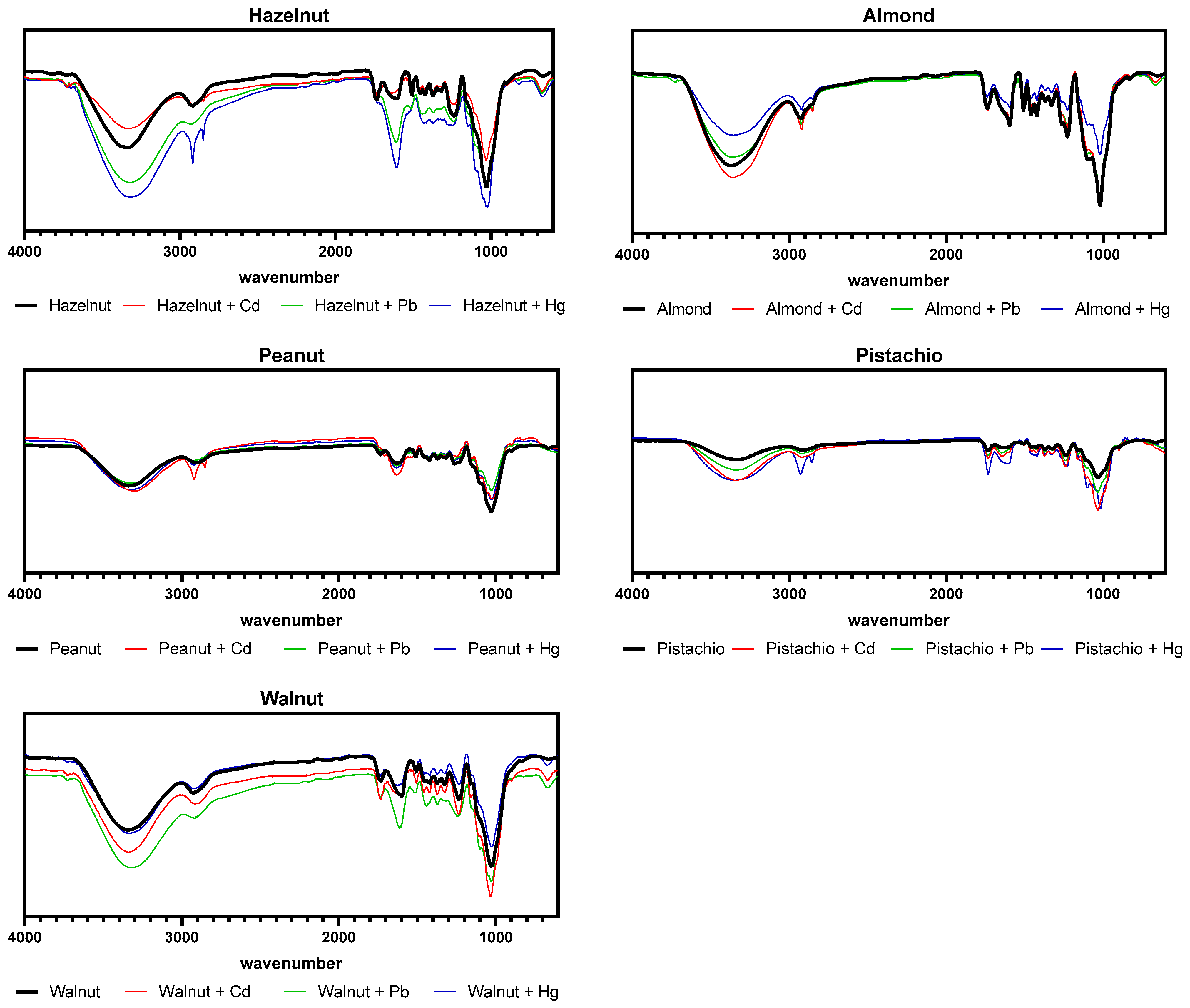
3.4. Major Functional Groups in Nutshells before and after Exposure to Cd, Pb, and Hg
4. Discussion
4.1. Removal Differences among Elements and Biosorbents
4.2. Effect of Ionic Strength and Metal Competition on the Removal of Cd, Pb, and Hg by Hazelnut Shells
4.3. Effect of Pre-Treatment with Hot Water on Removal Efficiency of Biosorbents
4.4. Food and Agricultural Wastes as Biosorbents in the Remediation of Contaminated Waters
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Febrianto, J.; Kosasih, A.N.; Sunarso, J.; Ju, Y.H.; Indraswati, N.; Ismadji, S. Equilibrium and kinetic studies in adsorption of heavy metals using biosorbent: A summary of recent studies. J. Hazard. Mater. 2009, 162, 616–645. [Google Scholar] [CrossRef]
- Rozumová, L.; Životský, O.; Seidlerová, J.; Motyka, O.; Šafařík, I.; Šafaříková, M. Magnetically modified peanut husks as an effective sorbent of heavy metals. J. Environ. Chem. Eng. 2016, 4, 549–555. [Google Scholar] [CrossRef]
- Adamu, C.I.; Nganje, T.N.; Edet, A. Heavy metal contamination and health risk assessment associated with abandoned barite mines in Cross River State, southeastern Nigeria. Environ. Nanotechnol. Monit. Manag. 2015, 3, 10–21. [Google Scholar] [CrossRef]
- Chowdhury, S.; Mazumder, M.A.J.; Al-Attas, O.; Husain, T. Heavy metals in drinking water: Occurrences, implications, and future needs in developing countries. Sci. Total Environ. 2016, 569, 476–488. [Google Scholar] [CrossRef]
- Jiang, L.; Zhou, W.; Liu, D.; Liu, T.; Wang, Z. Biosorption isotherm study of Cd2+, Pb2+ and Zn2+ biosorption onto marine bacterium Pseudoalteromonas sp. SCSE709-6 in multiple systems. J. Mol. Liq. 2017, 247, 230–237. [Google Scholar] [CrossRef]
- Farooq, U.; Kozinski, J.A.; Khan, M.A.; Athar, M. Biosorption of heavy metal ions using wheat based biosorbents—A review of the recent literature. Bioresour. Technol. 2010, 101, 5043–5053. [Google Scholar] [CrossRef] [PubMed]
- Abdullah, E.J. Quality Assessment for Shatt Al-Arab River Using Heavy Metal Pollution Index and Metal Index. J. Environ. Earth Sci. 2013, 3, 114–120. [Google Scholar]
- Singh, U.K.; Kumar, B. Pathways of heavy metals contamination and associated human health risk in Ajay River basin, India. Chemosphere 2017, 174, 183–199. [Google Scholar] [CrossRef]
- WHO. Guidelines for Drinking-Water Quality, 4th ed.; Incorporating the 1st Addendum; WHO: Geneva, Switzerland, 2017; p. 564. ISBN 978-92-4-154995-0. [Google Scholar]
- Decree-Law No. 236/98 of the Portuguese Ministry of the Environment of 1 August Establishing Water Quality Standards. Diário da República I Série 1998, 176, 3676–3722.
- AMBIENTE Decree-Law No. 152/2017. Diário da República 2017, 235, 6555–6576.
- Agency for Toxic Substances and Disease Registry ATSDR’s Substance Priority List. Available online: https://www.atsdr.cdc.gov/spl/#2019spl (accessed on 20 December 2020).
- Witek-Krowiak, A.; Szafran, R.G.; Modelski, S. Biosorption of heavy metals from aqueous solutions onto peanut shell as a low-cost biosorbent. Desalination 2011, 265, 126–134. [Google Scholar] [CrossRef]
- Peng, S.H.; Wang, R.; Yang, L.Z.; He, L.; He, X.; Liu, X. Biosorption of copper, zinc, cadmium and chromium ions from aqueous solution by natural foxtail millet shell. Ecotoxicol. Environ. Saf. 2018, 165, 61–69. [Google Scholar] [CrossRef]
- Yahaya, Y.A.; Mat Don, M.; Bhatia, S. Biosorption of copper (II) onto immobilized cells of Pycnoporus sanguineus from aqueous solution: Equilibrium and kinetic studies. J. Hazard. Mater. 2009, 161, 189–195. [Google Scholar] [CrossRef]
- Pehlivan, E.; Altun, T.; Cetin, S.; Iqbal Bhanger, M. Lead sorption by waste biomass of hazelnut and almond shell. J. Hazard. Mater. 2009, 167, 1203–1208. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.L.; Chen, C. Biosorbents for heavy metals removal and their future. Biotechnol. Adv. 2009, 27, 195–226. [Google Scholar] [CrossRef] [PubMed]
- Fomina, M.; Gadd, G.M. Biosorption: Current perspectives on concept, definition and application. Bioresour. Technol. 2014, 160, 3–14. [Google Scholar] [CrossRef]
- Robalds, A.; Naja, G.M.; Klavins, M. Highlighting inconsistencies regarding metal biosorption. J. Hazard. Mater. 2016, 304, 553–556. [Google Scholar] [CrossRef]
- Ismail, I.; Moustafa, T. Biosorption of heavy metals. In Heavy Metals: Sources, Toxicity and Remediation Techniques; Nova Science Publishers: Hauppauge, NY, USA, 2016; Volume 7, pp. 131–174. ISBN 9781634847667. [Google Scholar]
- Gadd, G.M. Biosorption: Critical review of scientific rationale, environmental importance and significance for pollution treatment. J. Chem. Technol. Biotechnol. 2009, 84, 13–28. [Google Scholar] [CrossRef]
- Dai, Y.; Sun, Q.; Wang, W.; Lu, L.; Liu, M.; Li, J.; Yang, S.; Sun, Y.; Zhang, K.; Xu, J.; et al. Utilizations of agricultural waste as adsorbent for the removal of contaminants: A review. Chemosphere 2018, 211, 235–253. [Google Scholar] [CrossRef]
- De Freitas, G.R.; da Silva, M.G.C.; Vieira, M.G.A. Biosorption technology for removal of toxic metals: A review of commercial biosorbents and patents. Environ. Sci. Pollut. Res. 2019, 26, 19097–19118. [Google Scholar] [CrossRef]
- Pavan, F.A.; Lima, I.S.; Lima, É.C.; Airoldi, C.; Gushikem, Y. Use of Ponkan mandarin peels as biosorbent for toxic metals uptake from aqueous solutions. J. Hazard. Mater. 2006, 137, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Sud, D.; Mahajan, G.; Kaur, M.P. Agricultural waste material as potential adsorbent for sequestering heavy metal ions from aqueous solutions—A review. Bioresour. Technol. 2008, 99, 6017–6027. [Google Scholar] [CrossRef]
- Maaloul, N.; Oulego, P.; Rendueles, M.; Ghorbal, A.; Díaz, M. Novel biosorbents from almond shells: Characterization and adsorption properties modeling for Cu (II) ions from aqueous solutions. J. Environ. Chem. Eng. 2017, 5, 2944–2954. [Google Scholar] [CrossRef]
- International Nut & Dried Fruit Council. Nuts & Dried Fruits—Statistical Yearbook 2019–2020. Stat. Yearb. 2020, 80, 5–7. [Google Scholar]
- Khir, R.; Pan, Z. Walnuts. In Integrated Processing Technologies for Food and Agricultural By-Products; Elsevier: Amsterdam, The Netherlands, 2019; pp. 391–411. ISBN 9780128141397. [Google Scholar]
- Vaghetti, J.C.P.; Lima, E.C.; Royer, B.; da Cunha, B.M.; Cardoso, N.F.; Brasil, J.L.; Dias, S.L.P. Pecan nutshell as biosorbent to remove Cu (II), Mn (II) and Pb (II) from aqueous solutions. J. Hazard. Mater. 2009, 162, 270–280. [Google Scholar] [CrossRef]
- Cimino, G.; Passerini, A.; Toscano, G. Removal of toxic cations and Cr (VI) from aqueous solution by hazelnut shell. Water Res. 2000, 34, 2955–2962. [Google Scholar] [CrossRef]
- Feizi, M.; Jalali, M. Removal of heavy metals from aqueous solutions using sunflower, potato, canola and walnut shell residues. J. Taiwan Inst. Chem. Eng. 2015, 54, 125–136. [Google Scholar] [CrossRef]
- Abdelfattah, I.; Ismail, A.A.; Sayed, F.A.; Almedolab, A.; Aboelghait, K.M. Biosorption of heavy metals ions in real industrial wastewater using peanut husk as efficient and cost effective adsorbent. Environ. Nanotechnol. Monit. Manag. 2016, 6, 176–183. [Google Scholar] [CrossRef]
- Bessa, A.; Henriques, B.; Gonçalves, G.; Irurueta, G.; Pereira, E.; Marques, P.A.A.P. Graphene oxide/polyethyleneimine aerogel for high-performance mercury sorption from natural waters. Chem. Eng. J. 2020, 398, 125587. [Google Scholar] [CrossRef]
- Henriques, B.; Lopes, C.B.; Figueira, P.; Rocha, L.S.; Duarte, A.C.; Vale, C.; Pardal, M.A.; Pereira, E. Bioaccumulation of Hg, Cd and Pb by Fucus vesiculosus in single and multi-metal contamination scenarios and its effect on growth rate. Chemosphere 2017, 171, 208–222. [Google Scholar] [CrossRef] [PubMed]
- Henriques, B.; Rodrigues, S.M.; Coelho, C.; Cruz, N.; Duarte, A.C.; Römkens, P.F.A.M.; Pereira, E. Risks associated with the transfer of toxic organo-metallic mercury from soils into the terrestrial feed chain. Environ. Int. 2013, 59, 408–417. [Google Scholar] [CrossRef]
- Costa, M.; Henriques, B.; Pinto, J.; Fabre, E.; Viana, T.; Ferreira, N.; Amaral, J.; Vale, C.; Pinheiro-Torres, J.; Pereira, E. Influence of salinity and rare earth elements on simultaneous removal of Cd, Cr, Cu, Hg, Ni and Pb from contaminated waters by living macroalgae. Environ. Pollut. 2020, 266, 115374. [Google Scholar] [CrossRef]
- Fabre, E.; Lopes, C.B.; Vale, C.; Pereira, E.; Silva, C.M. Valuation of banana peels as an effective biosorbent for mercury removal under low environmental concentrations. Sci. Total Environ. 2020, 709, 135883. [Google Scholar] [CrossRef]
- Ho, Y.-S. Review of second-order models for adsorption systems. J. Hazard. Mater. 2006, 136, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.S.; Ng, J.C.Y.; Mckay, G. Kinetics of pollutant sorption by biosorbents: Review. Sep. Purif. Methods 2000, 29, 189–232. [Google Scholar] [CrossRef]
- Gozaydin, G.; Yuksel, A. Valorization of hazelnut shell waste in hot compressed water. Fuel Process. Technol. 2017, 166, 96–106. [Google Scholar] [CrossRef]
- Li, X.; Liu, Y.; Hao, J.; Wang, W. Study of almond shell characteristics. Materials 2018, 11, 1782. [Google Scholar] [CrossRef]
- Şencan, A.; Karaboyacı, M.; Kılıç, M. Determination of lead (II) sorption capacity of hazelnut shell and activated carbon obtained from hazelnut shell activated with ZnCl2. Environ. Sci. Pollut. Res. 2015, 22, 3238–3248. [Google Scholar] [CrossRef]
- Li, R.; Zhang, Y.; Chu, W.; Chen, Z.; Wang, J. Adsorptive removal of antibiotics from water using peanut shells from agricultural waste. RSC Adv. 2018, 8, 13546–13555. [Google Scholar] [CrossRef]
- Cui, H.; Liu, X.; Li, K.; Cao, T.T.; Cui, C.; Wang, J.Y. Mechanism of Hg (II), Cd (II) and Pb (II) ions sorption from aqueous solutions by Aspergillus niger spores. Sep. Sci. Technol. 2020, 55, 848–859. [Google Scholar] [CrossRef]
- Costa, M.; Henriques, B.; Pinto, J.; Fabre, E.; Dias, M.; Soares, J.; Carvalho, L.; Vale, C.; Pinheiro-Torres, J.; Pereira, E. Influence of toxic elements on the simultaneous uptake of rare earth elements from contaminated waters by estuarine macroalgae. Chemosphere 2020, 126562. [Google Scholar] [CrossRef]
- Powell, K.J.; Brown, P.L.; Byrne, R.H.; Gajda, T.; Hefter, G.; Leuz, A.K.; Sjöberg, S.; Wanner, H. Chemical speciation of environmentally significant metals with inorganic ligands. Part 4: The Cd2+ + OH-, Cl-, co32-, So42-, and Po43- systems (IUPAC technical report). Pure Appl. Chem. 2011, 83, 1163–1214. [Google Scholar] [CrossRef]
- Brady, J.M.; Tobin, J.M. Binding of hard and soft metal ions to Rhizopus arrhizus biomass. Enzyme Microb. Technol. 1995, 17, 791–796. [Google Scholar] [CrossRef]
- Nieboer, E.; Richardson, D.H.S. The replacement of the nondescript term “heavy metals” by a biologically and chemically significant classification of metal ions. Environ. Pollution. Ser. B Chem. Phys. 1980, 1, 3–26. [Google Scholar] [CrossRef]
- Zheng, Q.; Zhou, T.; Wang, Y.; Cao, X.; Wu, S.; Zhao, M.; Wang, H.; Xu, M.; Zheng, B.; Zheng, J.; et al. Pretreatment of wheat straw leads to structural changes and improved enzymatic hydrolysis. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Altun, T.; Pehlivan, E. Removal of Cr (VI) from aqueous solutions by modified walnut shells. Food Chem. 2012, 132, 693–700. [Google Scholar] [CrossRef]
- Segovia-Sandoval, S.J.; Ocampo-Pérez, R.; Berber-Mendoza, M.S.; Leyva-Ramos, R.; Jacobo-Azuara, A.; Medellín-Castillo, N.A. Walnut shell treated with citric acid and its application as biosorbent in the removal of Zn (II). J. Water Process Eng. 2018, 25, 45–53. [Google Scholar] [CrossRef]
- Anwar, J.; Shafique, U.; Waheed-uz-Zaman; Salman, M.; Dar, A.; Anwar, S. Removal of Pb (II) and Cd (II) from water by adsorption on peels of banana. Bioresour. Technol. 2010, 101, 1752–1755. [Google Scholar] [CrossRef]
- Schiewer, S.; Balaria, A. Biosorption of Pb2+ by original and protonated citrus peels: Equilibrium, kinetics, and mechanism. Chem. Eng. J. 2009, 146, 211–219. [Google Scholar] [CrossRef]
- Asuquo, E.D.; Martin, A.D. Sorption of cadmium (II) ion from aqueous solution onto sweet potato (Ipomoea batatas L.) peel adsorbent: Characterisation, kinetic and isotherm studies. J. Environ. Chem. Eng. 2016, 4, 4207–4228. [Google Scholar] [CrossRef]
- Zein, R.; Suhaili, R.; Earnestly, F.; Indrawati; Munaf, E. Removal of Pb (II), Cd (II) and Co (II) from aqueous solution using Garcinia mangostana L. fruit shell. J. Hazard. Mater. 2010, 181, 52–56. [Google Scholar] [CrossRef]
- Garba, Z.N.; Ugbaga, N.I.; Abdullahi, A.K. Evaluation of optimum adsorption conditions for Ni (II) and Cd (II) removal from aqueous solution by modified plantain peels (MPP). Beni-Suef Univ. J. Basic Appl. Sci. 2016, 5, 170–179. [Google Scholar] [CrossRef]
- Moussavi, G.; Barikbin, B. Biosorption of chromium (VI) from industrial wastewater onto pistachio hull waste biomass. Chem. Eng. J. 2010, 162, 893–900. [Google Scholar] [CrossRef]
- Wu, Y.; Pang, H.; Liu, Y.; Wang, X.; Yu, S.; Fu, D.; Chen, J.; Wang, X. Environmental remediation of heavy metal ions by novel-nanomaterials: A review. Environ. Pollut. 2019, 246, 608–620. [Google Scholar] [CrossRef] [PubMed]
- Freitas, O.M.M.; Martins, R.J.E.; Delerue-Matos, C.M.; Boaventura, R.A.R. Removal of Cd (II), Zn (II) and Pb (II) from aqueous solutions by brown marine macro algae: Kinetic modelling. J. Hazard. Mater. 2008, 153, 493–501. [Google Scholar] [CrossRef]
- Shaaban, A.F.; Fadel, D.A.; Mahmoud, A.A.; Elkomy, M.A.; Elbahy, S.M. Synthesis and characterization of dithiocarbamate chelating resin and its adsorption performance toward Hg (II), Cd (II) and Pb (II) by batch and fixed-bed column methods. J. Environ. Chem. Eng. 2013, 1, 208–217. [Google Scholar] [CrossRef]
- Rambabu, K.; Thanigaivelan, A.; Bharath, G.; Sivarajasekar, N.; Banat, F.; Show, P.L. Biosorption potential of Phoenix dactylifera coir wastes for toxic hexavalent chromium sequestration. Chemosphere 2020, in press. [Google Scholar] [CrossRef]
- Hossain, M.A.; Ngo, H.H.; Guo, W.S.; Setiadi, T. Adsorption and desorption of copper (II) ions onto garden grass. Bioresour. Technol. 2012, 121, 386–395. [Google Scholar] [CrossRef] [PubMed]
- Abdolali, A.; Guo, W.S.; Ngo, H.H.; Chen, S.S.; Nguyen, N.C.; Tung, K.L. Typical lignocellulosic wastes and by-products for biosorption process in water and wastewater treatment: A critical review. Bioresour. Technol. 2014, 160, 57–66. [Google Scholar] [CrossRef]



| Hazelnut | Almond | Peanut | Pistachio | Walnut | ||
|---|---|---|---|---|---|---|
| Ultrapure water | Mono-element | x | x | x | x | x |
| Equimolar | ||||||
| Mono-element | x | x | x | x | x | |
| Discharge | ||||||
| Mineral water | Mono-element | x | ||||
| Equimolar | ||||||
| Mono-element | x | |||||
| Discharge |
| Cd | Pb | Hg | |||||
|---|---|---|---|---|---|---|---|
| 2 µmol L−1 | 200 µg L−1 | 2 µmol L−1 | 1000 µg L−1 | 2 µmol L−1 | 50 µg L−1 | ||
| (225 µg L−1) | (415 µg L−1) | (400 µg L−1) | |||||
| Hazelnut | q (µg g−1) | 425 | 324 | 714 | 1725 | 680 | 63 |
| Removal (%) | 98 | 97 | 97 | 97 | 90 | 78 | |
| Almond | q (µg g−1) | 327 | 354 | 679 | 1570 | 607 | 67 |
| Removal (%) | 81 | 87 | 89 | 79 | 79 | 68 | |
| Peanut | q (µg g−1) | 433 | 386 | 729 | 1414 | 506 | 89 |
| Removal (%) | 97 | 94 | 92 | 81 | 65 | 88 | |
| Pistachio | q (µg g−1) | 389 | 331 | 669 | 1450 | 633 | 62 |
| Removal (%) | 88 | 94 | 91 | 96 | 77 | 72 | |
| Walnut | q (µg g−1) | 378 | 373 | 480 | 1706 | 609 | 71 |
| Removal (%) | 93 | 89 | 72 | 86 | 77 | 74 | |
| PFO | PSO | ||||||
|---|---|---|---|---|---|---|---|
| R2 | k1 | qe (µg g−1) | R2 | k2 | qe (µg g−1) | ||
| Cd | Ultrapure | 0.990 | 0.423 | 408 | 0.977 | 1.29 × 10−3 | 449 |
| Mineral | 0.992 | 0.144 | 297 | 0.999 | 4.60 × 10−4 | 349 | |
| Mineral Mix | 0.986 | 0.115 | 265 | 0.997 | 3.82 × 10−4 | 319 | |
| Pb | Ultrapure | 0.985 | 0.269 | 693 | 0.987 | 4.89 × 10−4 | 767 |
| Mineral | 0.995 | 0.150 | 703 | 0.992 | 2.01 × 10−4 | 826 | |
| Mineral Mix | 0.998 | 0.135 | 670 | 0.996 | 1.84 × 10−4 | 795 | |
| Hg | Ultrapure | 0.986 | 0.265 | 656 | 0.987 | 4.86 × 10−4 | 731 |
| Mineral | 0.993 | 0.161 | 529 | 0.996 | 3.00 × 10−4 | 615 | |
| Mineral Mix | 0.996 | 0.170 | 571 | 0.993 | 3.08 × 10−4 | 656 | |
| Hazelnut | Almond | Peanut | Pistachio | Walnut | ||
|---|---|---|---|---|---|---|
| Equimolar | Cd | n.r. | n.r. | n.r. | n.r. | n.r. |
| Pb | 0.10 | n.r. | n.r. | n.r. | 0.11 | |
| Hg | 0.16 | 0.19 | 0.16 | n.r. | 0.24 | |
| Discharge | Cd | n.r. | n.r. | n.r. | n.r. | n.r. |
| Pb | 0.13 | n.r. | n.r. | n.r. | 0.22 | |
| Hg | 0.21 | n.r. | 0.23 | n.r. | n.r. |
| Hazelnut a,b,c | Almond d,e | Peanut a,f | Pistachio d,g | Walnut d,h | |
|---|---|---|---|---|---|
| Lignin | 29.6–40.1 | 25.5–39.3 | 27.6–30.6 | 16.3–23.6 | 27.2–43.7 |
| Cellulose | 22.9–37.5 | 34.4–38.5 | 37.5–38.9 | 37.5–38.1 | 36.4–42.4 |
| Hemicellulose | 12.7–24.9 | 14.0–28.8 | 15.4–16.3 | 25.3–31.4 | 10.3–27.9 |
| Reference | Biosorbent | Matrix | Contaminant | Maximum Removal |
|---|---|---|---|---|
| [30] | Hazelnut (4 g L−1) | Distilled water | Cd 14.5 mg L−1 | 92.4% (5 h) |
| [16] | Hazelnut (6.3 g L−1) Almond (6.3 g L−1) | Distilled water | Pb 207 mg L−1 | Hazelnut: 90% (4 h) Almond: 68% (4 h) |
| [31] | Walnut (2 g L−1) | Distilled water | Cd 100 mg L−1 | 45% (3.3 h) |
| [29] | Pecan nut (1–15 g L−1) | Distilled water | Pb 100 mg L−1 | 95% (6 h) |
| [32] | Peanut (5 g L−1) | Wastewater | Cd 46 mg L−1 Pb 0.26 mg L−1 | Cd: 38% (1 h) Pb: <99% (1 h) |
| [2] | Peanut (4 g L−1) | Deionized water | Cd 500 mg L−1 Pb 500 mg L−1 | Cd: 6.14% (1 h) Pb: 22.6% (1 h) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dias, M.; Pinto, J.; Henriques, B.; Figueira, P.; Fabre, E.; Tavares, D.; Vale, C.; Pereira, E. Nutshells as Efficient Biosorbents to Remove Cadmium, Lead, and Mercury from Contaminated Solutions. Int. J. Environ. Res. Public Health 2021, 18, 1580. https://doi.org/10.3390/ijerph18041580
Dias M, Pinto J, Henriques B, Figueira P, Fabre E, Tavares D, Vale C, Pereira E. Nutshells as Efficient Biosorbents to Remove Cadmium, Lead, and Mercury from Contaminated Solutions. International Journal of Environmental Research and Public Health. 2021; 18(4):1580. https://doi.org/10.3390/ijerph18041580
Chicago/Turabian StyleDias, Mariana, João Pinto, Bruno Henriques, Paula Figueira, Elaine Fabre, Daniela Tavares, Carlos Vale, and Eduarda Pereira. 2021. "Nutshells as Efficient Biosorbents to Remove Cadmium, Lead, and Mercury from Contaminated Solutions" International Journal of Environmental Research and Public Health 18, no. 4: 1580. https://doi.org/10.3390/ijerph18041580
APA StyleDias, M., Pinto, J., Henriques, B., Figueira, P., Fabre, E., Tavares, D., Vale, C., & Pereira, E. (2021). Nutshells as Efficient Biosorbents to Remove Cadmium, Lead, and Mercury from Contaminated Solutions. International Journal of Environmental Research and Public Health, 18(4), 1580. https://doi.org/10.3390/ijerph18041580










