Inactivation of Indigenous Microorganisms and Salmonella in Korean Rice Cakes by In-Package Cold Plasma Treatment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of KRC
2.2. Preparation of Salmonella Strains and Inoculation
2.3. In-Package CP Treatment
2.4. Microbial Inactivation Test
2.5. Investigation of Bacterial Inactivation Mechanism
2.6. Chromaticity Measurement
2.7. Texture Analysis
2.8. Statistical Analysis
3. Results and Discussion
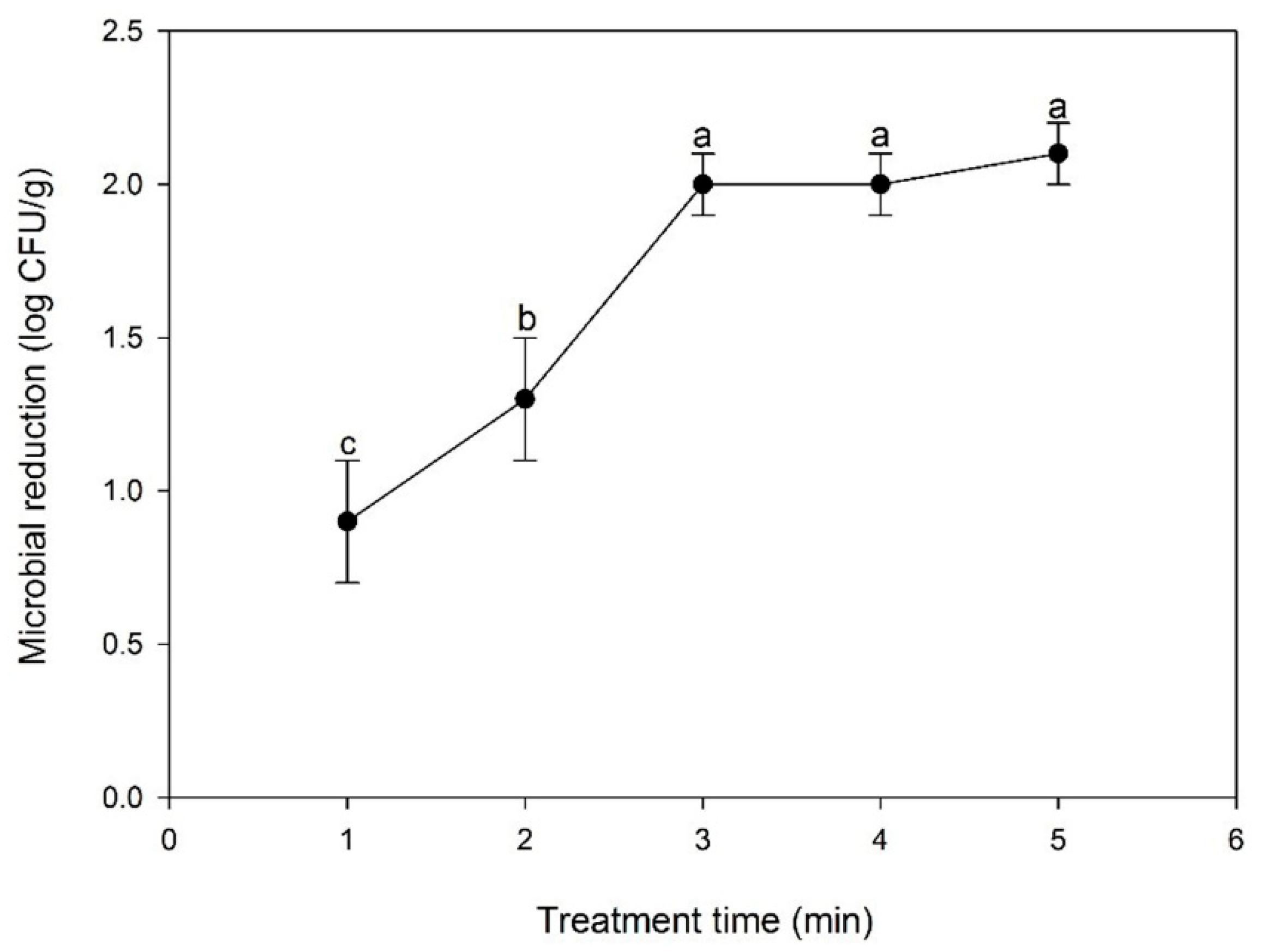
3.1. Effect of CP Treatment Time on Bacterial Inactivation
3.2. Effect of Shaking on CP Treatment Efficiency
3.3. Effect of KRC Number on CP Treatment Efficiency
3.4. Evaluation of KRC Color and Texture
3.5. Control of Indigenous Yeast/Mold and Salmonella Contamination
3.6. Salmonella Inactivation Mechanism
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Lee, S.Y.; Chung, H.J.; Shin, J.H.; Dougherty, R.H.; Kangi, D.H. Survival and growth of foodborne pathogens during cooking and storage of oriental-style rice cakes. J. Food Prot. 2006, 69, 3037–3042. [Google Scholar] [CrossRef]
- Cheon, H.S.; Cho, W.-I.; Lee, S.J.; Chung, M.-S.; Choi, J.-B. Acidic and steaming treatments of tteokbokki rice cake to improve its microbial and textural properties. Korean J. Food Sci. Technol. 2017, 49, 502–506. [Google Scholar]
- Jung, H.; Yuk, H.-G.; Yoon, W.B. Effect of LED light on the inactivation of Bacillus cereus for extending shelf-life of extruded rice cake and simulation of the patterns of LED irradiation by various arrays of LEDs. J. Appl. Biol. Chem. 2019, 62, 181–186. [Google Scholar] [CrossRef]
- Cho, S.; Chang, M.; Lee, Y.; Ha, S.; Kim, G. Changes in residual chlorine content on fresh-cut iceberg lettuce after chlorine treatment. Korean J. Hortic. Sci. Technol. 2010, 28, 77–81. [Google Scholar]
- Fernandez, A.; Noriega, E.; Thompson, A. Inactivation of Salmonella enterica serovar Typhimurium on fresh produce by cold atmospheric gas plasma technology. Food Microbiol. 2013, 33, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.J.; Park, J.D.; Lee, H.Y.; Kum, J.S. Effect of grapefruit seed extracts and acid regulation agents on the qualities of Topokkidduk. J. Korean Soc. Food Sci. Nutr. 2013, 42, 948–956. [Google Scholar] [CrossRef]
- Lee, J.-W.; Lee, H.-H.; Rhim, J.-W. Shelf life extension of white rice cake and wet noodle by the treatment with chitosan. Korean J. Food Sci. Technol. 2000, 32, 828–833. [Google Scholar]
- Park, C.-S.; Choi, M.; Park, G.-S. Effect of Paecilomyces japonica on the microbiological quality and shelf-life of Jeungpyun. Korean J. Food Cook. Sci. 2004, 20, 561–567. [Google Scholar]
- Pankaj, S.K.; Wan, Z.; Keener, K.M. Effects of Cold Plasma on Food Quality: A Review. Foods 2018, 7, 4. [Google Scholar] [CrossRef]
- Chauhan, O.; Unni, L. Pulsed electric field (PEF) processing of foods and its combination with electron beam processing. In Electron Beam Pasteurization and Complementary Food Processing Technologies; Woodhead Publishing: Cambridge, UK, 2015; pp. 157–184. [Google Scholar]
- Liao, X.; Liu, D.; Xiang, Q.; Ahn, J.; Chen, S.; Ye, X.; Ding, T. Inactivation mechanisms of non-thermal plasma on microbes: A review. Food Control 2017, 75, 83–91. [Google Scholar] [CrossRef]
- Misra, N.; Pankaj, S.; Segat, A.; Ishikawa, K. Cold plasma interactions with enzymes in foods and model systems. Trends Food Sci. Technol. 2016, 55, 39–47. [Google Scholar] [CrossRef]
- Misra, N.; Jo, C. Applications of cold plasma technology for microbiological safety in meat industry. Trends Food Sci. Technol. 2017, 64, 74–86. [Google Scholar] [CrossRef]
- Hertwig, C.; Meneses, N.; Mathys, A. Cold atmospheric pressure plasma and low energy electron beam as alternative nonthermal decontamination technologies for dry food surfaces: A review. Trends Food Sci. Technol. 2018, 77, 131–142. [Google Scholar] [CrossRef]
- Olatunde, O.O.; Benjakul, S. Nonthermal processes for shelf-life extension of seafoods: A revisit. Compr. Rev. Food Sci. Food Saf. 2018, 17, 892–904. [Google Scholar] [CrossRef] [PubMed]
- Roh, S.H.; Lee, S.Y.; Park, H.H.; Lee, E.S.; Min, S.C. Effects of the treatment parameters on the efficacy of the inactivation of Salmonella contaminating boiled chicken breast by in-package atmospheric cold plasma treatment. Int. J. Food Microbiol. 2019, 293, 24–33. [Google Scholar] [CrossRef]
- Min, S.C.; Roh, S.H.; Niemira, B.A.; Boyd, G.; Sites, J.E.; Fan, X.; Sokorai, K.; Jin, T.Z. In-package atmospheric cold plasma treatment of bulk grape tomatoes for microbiological safety and preservation. Food Res. Int. 2018, 108, 378–386. [Google Scholar] [CrossRef]
- Pal, P.; Kaur, P.; Singh, N.; Kaur, A.; Misra, N.; Tiwari, B.K.; Cullen, P.J.; Virdi, A.S. Effect of nonthermal plasma on physico-chemical, amino acid composition, pasting and protein characteristics of short and long grain rice flour. Food Res. Int. 2016, 81, 50–57. [Google Scholar] [CrossRef]
- Thirumdas, R.; Saragapani, C.; Ajinkya, M.; Deshmukh, R.; Annapure, U. Influence of low pressure cold plasma on cooking and textural properties of brown rice. Innov. Food Sci. Emerg. Technol. 2016, 37, 53–60. [Google Scholar] [CrossRef]
- Thirumdas, R.; Trimukhe, A.; Deshmukh, R.; Annapure, U. Functional and rheological properties of cold plasma treated rice starch. Carbohydr. Polym. 2017, 157, 1723–1731. [Google Scholar] [CrossRef]
- Kim, S.Y.; Bang, I.H.; Min, S.C. Effects of packaging parameters on the inactivation of Salmonella contaminating mixed vegetables in plastic packages using atmospheric dielectric barrier discharge cold plasma treatment. J. Food Eng. 2019, 242, 55–67. [Google Scholar] [CrossRef]
- Kim, D.-K.; Kang, D.-H. Elevated inactivation efficacy of a pulsed UVC light-emitting diode system for foodborne pathogens on selective media and food surfaces. Appl. Environ. Microbiol. 2018, 84. [Google Scholar] [CrossRef]
- Kang, J.-W.; Kang, D.-H. Increased resistance of Salmonella enterica serovar Typhimurium and Escherichia coli O157: H7 to 222-nanometer krypton-chlorine excilamp treatment by acid adaptation. Appl. Environ. Microbiol. 2019, 85. [Google Scholar] [CrossRef] [PubMed]
- Moutiq, R.; Misra, N.; Mendonca, A.; Keener, K. In-package decontamination of chicken breast using cold plasma technology: Microbial, quality and storage studies. Meat Sci. 2020, 159, 107942. [Google Scholar] [CrossRef] [PubMed]
- Baier, M.; Janßen, T.; Wieler, L.H.; Ehlbeck, J.; Knorr, D.; Schlüter, O. Inactivation of Shiga toxin-producing Escherichia coli O104: H4 using cold atmospheric pressure plasma. J. Biosci. Bioeng. 2015, 120, 275–279. [Google Scholar] [CrossRef] [PubMed]
- Min, S.C.; Roh, S.H.; Boyd, G.; Sites, J.E.; Uknalis, J.; Fan, X.; Niemira, B.A. Inactivation of Escherichia coli O157: H7 and aerobic microorganisms in romaine lettuce packaged in a commercial polyethylene terephthalate container using atmospheric cold plasma. J. Food Prot. 2017, 80, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Wiertzema, J.; Peng, P.; Cheng, Y.; Liu, J.; Mao, Q.; Ma, Y.; Anderson, E.; Chen, P.; Baumler, D.J. Effects of intense pulsed light on Cronobacter sakazakii inoculated in non-fat dry milk. J. Food Eng. 2018, 238, 178–187. [Google Scholar] [CrossRef]
- Dey, A.; Rasane, P.; Choudhury, A.; Singh, J.; Maisnam, D.; Rasane, P. Cold plasma processing: A review. J. Chem. Pharm. Sci. 2016, 9, 2980–2984. [Google Scholar]
- Pignata, C.; D’angelo, D.; Fea, E.; Gilli, G. A review on microbiological decontamination of fresh produce with nonthermal plasma. J. Appl. Microbiol. 2017, 122, 1438–1455. [Google Scholar] [CrossRef]
- Jung, H.; Yu, C.R.; Park, H.W.; Yoon, W.B. Effect of acid soaking and thermal sterilization on the shape and quality characteristics of Tteokbokki rice cake. Korean J. Food Nutr. 2018, 31, 737–750. [Google Scholar]
- Sarangapani, C.; Devi, Y.; Thirundas, R.; Annapure, U.S.; Deshmukh, R.R. Effect of low-pressure plasma on physico-chemical properties of parboiled rice. LWT Food Sci. Technol. 2015, 63, 452–460. [Google Scholar] [CrossRef]
- López, M.; Calvo, T.; Prieto, M.; Múgica-Vidal, R.; Muro-Fraguas, I.; Alba-Elías, F.; Alvarez-Ordóñez, A. A review on non-thermal atmospheric plasma for food preservation: Mode of action, determinants of effectiveness, and applications. Front. Microbiol. 2019, 10, 622. [Google Scholar] [CrossRef]
- Surowsky, B.; Fischer, A.; Schlueter, O.; Knorr, D. Cold plasma effects on enzyme activity in a model food system. Innov. Food Sci. Emerg. Technol. 2013, 19, 146–152. [Google Scholar] [CrossRef]
- Han, L.; Patil, S.; Boehm, D.; Milosavljevic, V.; Cullen, P.J.; Bourke, P. Mechanisms of Inactivation by High-Voltage Atmospheric Cold Plasma Differ for Escherichia coli and Staphylococcus aureus. Appl. Environ. Microbiol. 2016, 82, 450–458. [Google Scholar] [CrossRef] [PubMed]
- Varilla, C.; Marcone, M.; Annor, G.A. Potential of Cold Plasma Technology in Ensuring the Safety of Foods and Agricultural Produce: A Review. Foods 2020, 9, 1435. [Google Scholar] [CrossRef] [PubMed]
- Ziuzina, D.; Han, L.; Cullen, P.J.; Bourke, P. Cold plasma inactivation of internalised bacteria and biofilms for Salmonella enterica serovar Typhimurium, Listeria monocytogenes and Escherichia coli. Int. J. Food Microbiol. 2015, 210, 53–61. [Google Scholar] [CrossRef] [PubMed]

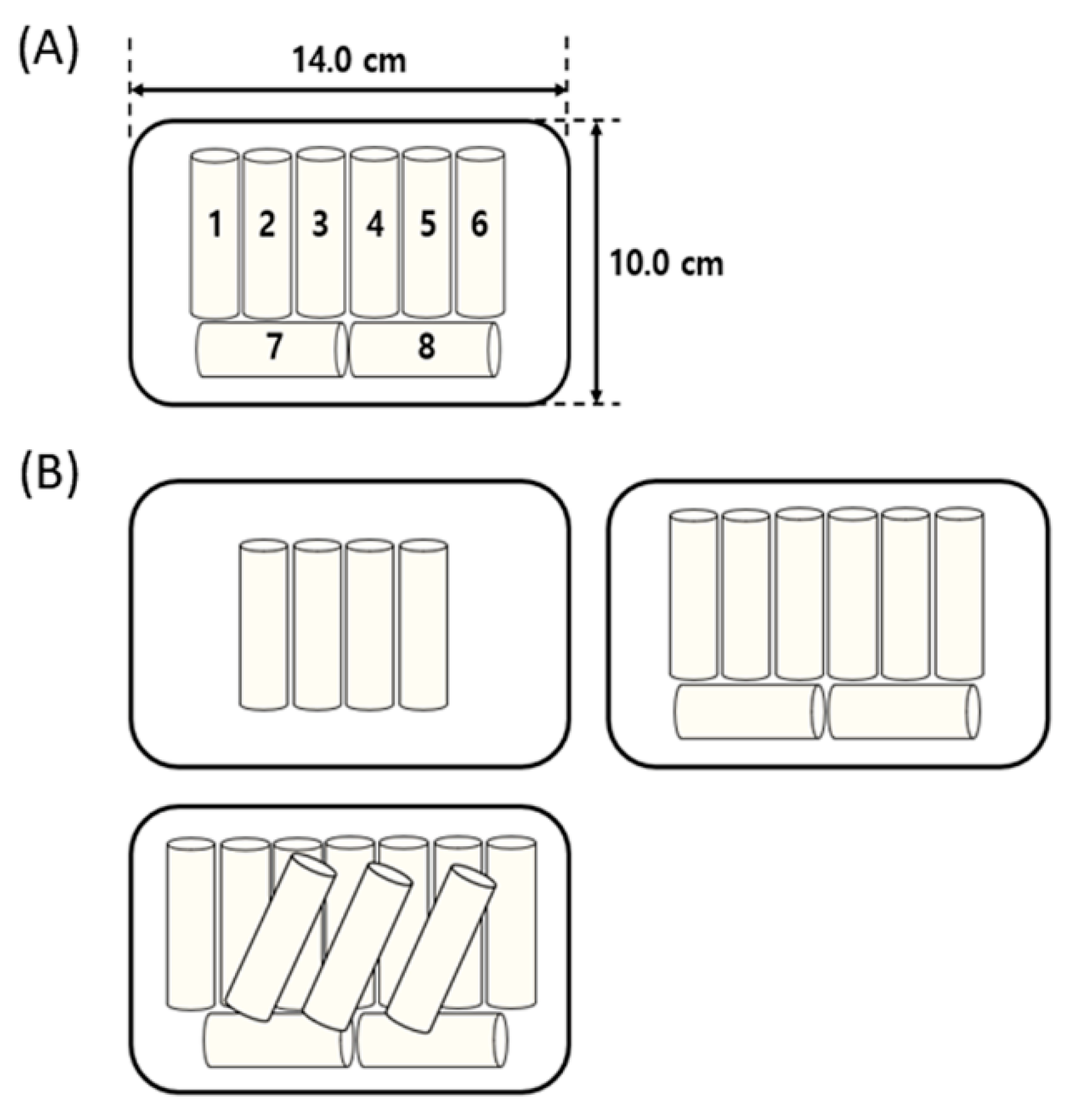
| Sample Position (Numbers Designated in Figure 2) | Microbial Reduction (log CFU/g) |
|---|---|
| 1 | 1.0 ± 0.3 a |
| 2 | 0.9 ± 0.3 a |
| 3 | 0.9 ± 0.1 a |
| 4 | 1.0 ± 0.3 a |
| 5 | 0.8 ± 0.1 a |
| 6 | 0.9 ± 0.0 a |
| 7 | 0.9 ± 0.1 a |
| 8 | 1.0 ± 0.4 a |
| Number of Samples (Pieces) | Microbial Reduction (log CFU/g) |
|---|---|
| 4 | 1.9 ± 0.1 a |
| 8 | 2.0 ± 0.1 a |
| 12 | 1.4 ± 0.1 b |
| Treatment | Fluorescence | ||
|---|---|---|---|
| Cell Membrane Integrity | Cell Lipid Peroxidation | Intracellular ROS | |
| Untreated | 303.83 ± 64.48 b | 4282.68 ± 3.71 b | 1036.54 ± 29.25 a |
| CP-treated | 420.88 ± 6.61 a | 6393.00 ± 188.72 a | 1097.17 ± 1.74 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, J.H.; Bai, J.; Min, S.C. Inactivation of Indigenous Microorganisms and Salmonella in Korean Rice Cakes by In-Package Cold Plasma Treatment. Int. J. Environ. Res. Public Health 2021, 18, 3360. https://doi.org/10.3390/ijerph18073360
Kang JH, Bai J, Min SC. Inactivation of Indigenous Microorganisms and Salmonella in Korean Rice Cakes by In-Package Cold Plasma Treatment. International Journal of Environmental Research and Public Health. 2021; 18(7):3360. https://doi.org/10.3390/ijerph18073360
Chicago/Turabian StyleKang, Joo Hyun, Jaewoo Bai, and Sea C. Min. 2021. "Inactivation of Indigenous Microorganisms and Salmonella in Korean Rice Cakes by In-Package Cold Plasma Treatment" International Journal of Environmental Research and Public Health 18, no. 7: 3360. https://doi.org/10.3390/ijerph18073360






