Diagnostic Value of IgM and IgG Detection in COVID-19 Diagnosis by the Mobile Laboratory B-LiFE: A Massive Testing Strategy in the Piedmont Region
Abstract
1. Introduction
2. Material and Methods
Serological Test
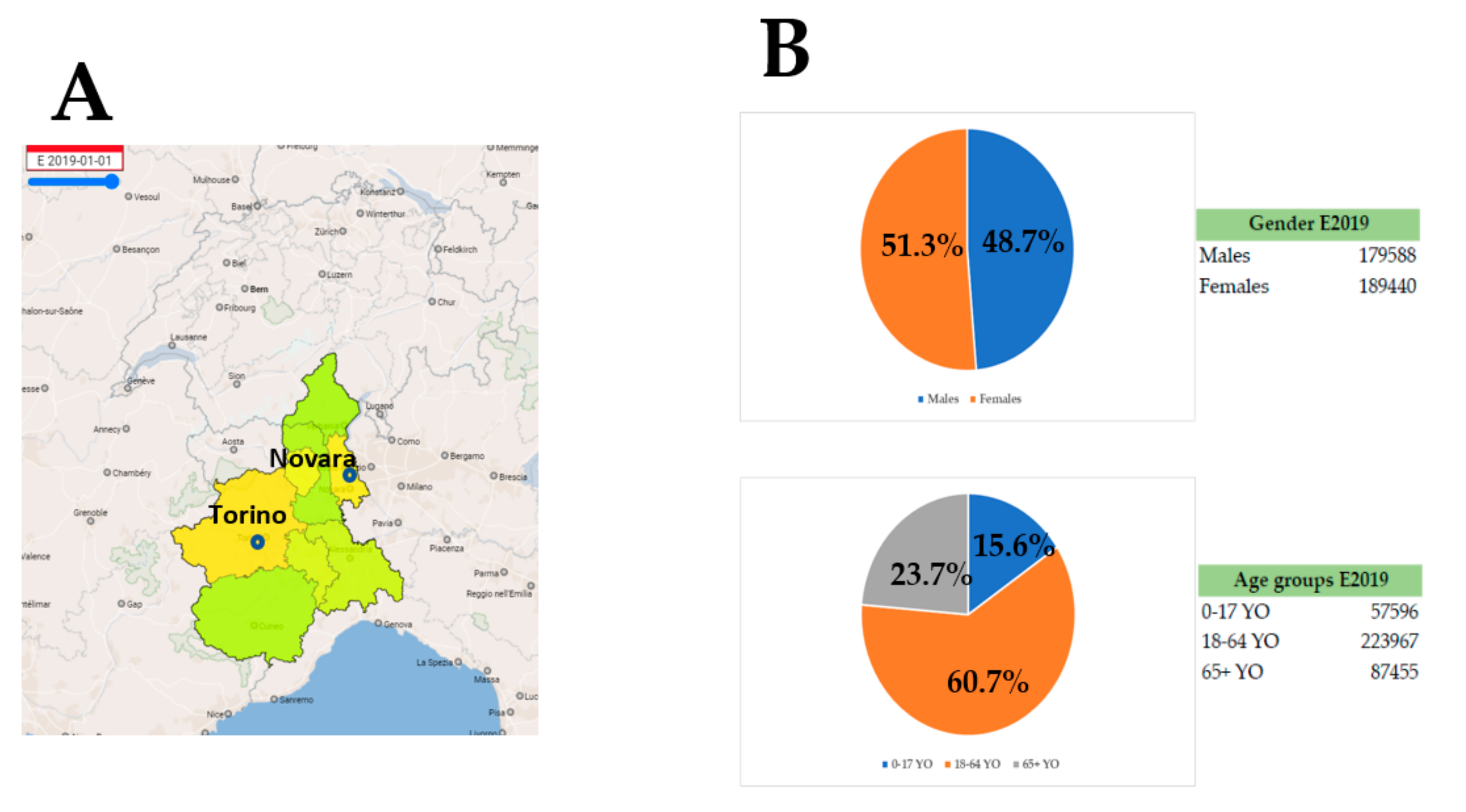
3. Results and Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gautam, A.; Kaphle, K.; Shrestha, B.; Phuyal, S. Susceptibility to SARS, MERS, and COVID-19 from animal health perspective. Open Vet. J. 2020, 10, 164–177. [Google Scholar] [CrossRef] [PubMed]
- Quirch, M.; Lee, J.; Rehman, S. Hazards of the cytokine storm and cytokine-targeted therapy in patients with COVID-19: Review. J. Med. Internet Res. 2020, 22, e20193. [Google Scholar] [CrossRef]
- Goncalves, B.C.; Lopes Barbosa, M.G.; Silva Olak, A.P.; Belebecha Terezo, N.; Nishi, L.; Watanabe, M.A.; Marinello, P.; Zendrini Rechenchoski, D.; Dejato Rocha, S.P.; Faccin-Galhardi, L.C. Antiviral therapies: Advances and perspectives. Fundam. Clin. Pharmacol. 2020, 35, 305–320. [Google Scholar] [CrossRef] [PubMed]
- de Wit, E.; van Doremalen, N.; Falzarano, D.; Munster, V.J. SARS and MERS: Recent insights into emerging coronaviruses. Nat. Rev. Microbiol. 2016, 14, 523–534. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Joyner, M.J.; Carter, R.E.; Senefeld, J.W.; Klassen, S.A.; Mills, J.R.; Johnson, P.W.; Theel, E.S.; Wiggins, C.C.; Bruno, K.A.; Klompas, A.M.; et al. Convalescent Plasma Antibody Levels and the Risk of Death from Covid-19. N. Engl. J. Med. 2021, 384, 1015–1027. [Google Scholar] [CrossRef]
- Deb, P.; Molla, M.M.A.; Rahman, K.M.S. An update to monoclonal antibody as therapeutic option against COVID-19. Biosaf. Health 2021. [Google Scholar] [CrossRef]
- Petersen, E.; Koopmans, M.; Go, U.; Hamer, D.H.; Petrosillo, N.; Castelli, F.; Storgaard, M.; Al Khalili, S.; Simonsen, L. Comparing SARS-CoV-2 with SARS-CoV and influenza pandemics. Lancet Infect. Dis. 2020, 20, e238–e244. [Google Scholar] [CrossRef]
- He, N.; Lu, Y.H.; Li, L.M.; Shen, H.B.; Yang, W.Z.; Feng, Z.J. Collaboration group for the emergency research project of novel coronavirus pneumonia for national natural science foundation of, C. [Epidemiological study design of asymptomatic infection of the 2019 novel coronavirus]. Zhonghua Liu Xing Bing Xue Za Zhi 2020, 41, E078. [Google Scholar] [CrossRef]
- Kumar, M.S.; Bhatnagar, T.; Manickam, P.; Kumar, V.S.; Rade, K.; Shah, N.; Kant, S.; Babu, G.R.; Zodpey, S.; Girish Kumar, C.P.; et al. National sero-surveillance to monitor the trend of SARS-CoV-2 infection transmission in India: Protocol for community-based surveillance. Indian J. Med. Res. 2020, 151, 419–423. [Google Scholar] [CrossRef]
- Amanat, F.; Stadlbauer, D.; Strohmeier, S.; Nguyen, T.H.O.; Chromikova, V.; McMahon, M.; Jiang, K.; Asthagiri Arunkumar, G.; Jurczyszak, D.; Polanco, J.; et al. A serological assay to detect SARS-CoV-2 seroconversion in humans. medRxiv 2020, 26, 1033–1036. [Google Scholar] [CrossRef] [PubMed]
- Okba, N.M.A.; Muller, M.A.; Li, W.; Wang, C.; GeurtsvanKessel, C.H.; Corman, V.M.; Lamers, M.M.; Sikkema, R.S.; de Bruin, E.; Chandler, F.D.; et al. Severe acute respiratory syndrome coronavirus 2-specific antibody responses in coronavirus disease patients. Emerg. Infect. Dis. 2020, 26, 1478–1488. [Google Scholar] [CrossRef]
- Ul-Rahman, A.; Shabbir, M.A.B.; Aziz, M.W.; Yaqub, S.; Mehmood, A.; Raza, M.A.; Shabbir, M.Z. A comparative phylogenomic analysis of SARS-CoV-2 strains reported from non-human mammalian species and environmental samples. Mol. Biol. Rep. 2020, 47, 9207–9217. [Google Scholar] [CrossRef] [PubMed]
- Huo, J.; Le Bas, A.; Ruza, R.R.; Duyvesteyn, H.M.E.; Mikolajek, H.; Malinauskas, T.; Tan, T.K.; Rijal, P.; Dumoux, M.; Ward, P.N.; et al. Neutralizing nanobodies bind SARS-CoV-2 spike RBD and block interaction with ACE2. Nat. Struct. Mol. Biol. 2020, 27, 846–854. [Google Scholar] [CrossRef]
- Tilocca, B.; Soggiu, A.; Sanguinetti, M.; Musella, V.; Britti, D.; Bonizzi, L.; Urbani, A.; Roncada, P. Comparative computational analysis of SARS-CoV-2 nucleocapsid protein epitopes in taxonomically related coronaviruses. Microbes. Infect. 2020, 22, 188–194. [Google Scholar] [CrossRef]
- Deshpande, G.R.; Sapkal, G.N.; Tilekar, B.N.; Yadav, P.D.; Gurav, Y.; Gaikwad, S.; Kaushal, H.; Deshpande, K.S.; Kaduskar, O.; Sarkale, P.; et al. Neutralizing antibody responses to SARS-CoV-2 in COVID-19 patients. Indian J. Med. Res. 2020, 152, 82–87. [Google Scholar] [CrossRef]
- Anna, F.; Goyard, S.; Lalanne, A.I.; Nevo, F.; Gransagne, M.; Souque, P.; Louis, D.; Gillon, V.; Turbiez, I.; Bidard, F.C.; et al. High seroprevalence but short-lived immune response to SARS-CoV-2 infection in Paris. Eur. J Immunol. 2021, 51, 180–190. [Google Scholar] [CrossRef]
- Bwire, G.M.; Majigo, M.V.; Njiro, B.J.; Mawazo, A. Detection profile of SARS-CoV-2 using RT-PCR in different types of clinical specimens: A systematic review and meta-analysis. J. Med. Virol. 2021, 93, 719–725. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Xu, Y.; Gao, R.; Lu, R.; Han, K.; Wu, G.; Tan, W. Detection of SARS-CoV-2 in Different Types of Clinical Specimens. JAMA 2020. [Google Scholar] [CrossRef]
- Montesinos, I.; Gruson, D.; Kabamba, B.; Dahma, H.; Van den Wijngaert, S.; Reza, S.; Carbone, V.; Vandenberg, O.; Gulbis, B.; Wolff, F.; et al. Evaluation of two automated and three rapid lateral flow immunoassays for the detection of anti-SARS-CoV-2 antibodies. J. Clin. Virol. 2020, 128, 104413. [Google Scholar] [CrossRef]
- Corman, V.M.; Drosten, C. Authors’ response: SARS-CoV-2 detection by real-time RT-PCR. Euro. Surveill. 2020, 25, 2001035. [Google Scholar] [CrossRef] [PubMed]
- Cassaniti, I.; Percivalle, E.; Sarasini, A.; Cambie, G.; Batisti Biffignandi, G.; Cereda, D.; Baldanti, F. Authors’ response: COVID-19: How accurate are seroprevalence studies? Eurosurveillance 2020, 25, 2001437. [Google Scholar] [CrossRef] [PubMed]
- Cassaniti, I.; Novazzi, F.; Giardina, F.; Salinaro, F.; Sachs, M.; Perlini, S.; Bruno, R.; Mojoli, F.; Baldanti, F.; Members of the San Matteo Pavia, C.-T.F.; et al. Performance of VivaDiag COVID-19 IgM/IgG Rapid Test is inadequate for diagnosis of COVID-19 in acute patients referring to emergency room department. J. Med. Virol. 2020, 92, 1724–1727. [Google Scholar] [CrossRef]
- Prazuck, T.; Colin, M.; Giache, S.; Gubavu, C.; Seve, A.; Rzepecki, V.; Chevereau-Choquet, M.; Kiani, C.; Rodot, V.; Lionnet, E.; et al. Evaluation of performance of two SARS-CoV-2 Rapid IgM-IgG combined antibody tests on capillary whole blood samples from the fingertip. PLoS ONE 2020, 15, e0237694. [Google Scholar] [CrossRef] [PubMed]
- Tomasik, P.; Krotki, F.; Jonca, M.; Anyszek, T. Rapid point-of-care antibody cassette tests for severe acute respiratory syndrome coronavirus 2: Practical considerations. Pol. Arch. Intern. Med. 2020, 130, 459–462. [Google Scholar] [CrossRef]
- Glasgow, A.; Glasgow, J.; Limonta, D.; Solomon, P.; Lui, I.; Zhang, Y.; Nix, M.A.; Rettko, N.J.; Zha, S.; Yamin, R.; et al. Engineered ACE2 receptor traps potently neutralize SARS-CoV-2. Proc. Natl. Acad. Sci. USA 2020, 45, 28046–28055. [Google Scholar] [CrossRef] [PubMed]
- Datta, P.K.; Liu, F.; Fischer, T.; Rappaport, J.; Qin, X. SARS-CoV-2 pandemic and research gaps: Understanding SARS-CoV-2 interaction with the ACE2 receptor and implications for therapy. Theranostics 2020, 10, 7448–7464. [Google Scholar] [CrossRef]
- Ding, S.; Laumaea, A.; Gasser, R.; Medjahed, H.; Pancera, M.; Stamatatos, L.; McGuire, A.; Bazin, R.; Finzi, A. Antibody binding to SARS-CoV-2 S glycoprotein correlates with, but does not predict neutralization. bioRxiv 2020, 12, 1214. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Lan, J.; Huang, B.; A, R.; Lu, M.; Wang, W.; Wang, W.; Li, W.; Deng, Y.; Wong, G.; et al. Lack of antibody-mediated cross-protection between SARS-CoV-2 and SARS-CoV infections. EBioMedicine 2020, 58, 102890. [Google Scholar] [CrossRef]
- Tan, C.W.; Chia, W.N.; Qin, X.; Liu, P.; Chen, M.I.; Tiu, C.; Hu, Z.; Chen, V.C.; Young, B.E.; Sia, W.R.; et al. A SARS-CoV-2 surrogate virus neutralization test based on antibody-mediated blockage of ACE2-spike protein-protein interaction. Nat. Biotechnol. 2020, 38, 1073–1078. [Google Scholar] [CrossRef]
- Liu, L.; Wang, P.; Nair, M.S.; Yu, J.; Rapp, M.; Wang, Q.; Luo, Y.; Chan, J.F.; Sahi, V.; Figueroa, A.; et al. Potent neutralizing antibodies against multiple epitopes on SARS-CoV-2 spike. Nature 2020, 584, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Zost, S.J.; Gilchuk, P.; Case, J.B.; Binshtein, E.; Chen, R.E.; Nkolola, J.P.; Schafer, A.; Reidy, J.X.; Trivette, A.; Nargi, R.S.; et al. Potently neutralizing and protective human antibodies against SARS-CoV-2. Nature 2020, 584, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.; Xing, S.; Ding, L.; Wang, Y.; Gu, C.; Wu, Y.; Rong, B.; Li, C.; Wang, S.; Chen, K.; et al. Human-IgG-Neutralizing Monoclonal Antibodies Block the SARS-CoV-2 Infection. Cell Rep. 2020, 32, 107918. [Google Scholar] [CrossRef] [PubMed]
- McAndrews, K.M.; Dowlatshahi, D.P.; Dai, J.; Becker, L.M.; Hensel, J.; Snowden, L.M.; Leveille, J.M.; Brunner, M.R.; Holden, K.W.; Hopkins, N.S.; et al. Heterogeneous antibodies against SARS-CoV-2 spike receptor binding domain and nucleocapsid with implications for COVID-19 immunity. JCI Insight 2020, 5, e142386. [Google Scholar] [CrossRef]
- Du, S.; Cao, Y.; Zhu, Q.; Yu, P.; Qi, F.; Wang, G.; Du, X.; Bao, L.; Deng, W.; Zhu, H.; et al. Structurally Resolved SARS-CoV-2 Antibody Shows High Efficacy in Severely Infected Hamsters and Provides a Potent Cocktail Pairing Strategy. Cell 2020, 183, 1013–1023. [Google Scholar] [CrossRef]
- Callow, K.A.; Parry, H.F.; Sergeant, M.; Tyrrell, D.A. The time course of the immune response to experimental coronavirus infection of man. Epidemiol. Infect. 1990, 105, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Reed, S.E. The behaviour of recent isolates of human respiratory coronavirus in vitro and in volunteers: Evidence of heterogeneity among 229E-related strains. J. Med. Virol. 1984, 13, 179–192. [Google Scholar] [CrossRef]
- Long, Q.X.; Tang, X.J.; Shi, Q.L.; Li, Q.; Deng, H.J.; Yuan, J.; Hu, J.L.; Xu, W.; Zhang, Y.; Lv, F.J.; et al. Clinical and immunological assessment of asymptomatic SARS-CoV-2 infections. Nat. Med. 2020, 26, 1200–1204. [Google Scholar] [CrossRef]
- Sharp, T.M.; Fischer, M.; Munoz-Jordan, J.L.; Paz-Bailey, G.; Staples, J.E.; Gregory, C.J.; Waterman, S.H. Dengue and Zika Virus Diagnostic Testing for Patients with a Clinically Compatible Illness and Risk for Infection with Both Viruses. MMWR Recomm. Rep. 2019, 68, 1–10. [Google Scholar] [CrossRef] [PubMed]





| Identity | National Registration Number |
|---|---|
| Date of first symptoms | Day/Month/Year |
| Date of last symptoms | Day/Month/Year |
| Cough | Yes/No |
| Anosmia | Yes/No |
| Dysgeusia | Yes/No |
| Headache | Yes/No |
| Muscle pain | Yes/No |
| Temperature | Yes/No |
| Respiratory distress | Yes/No |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nyabi, O.; Bentahir, M.; Ambroise, J.; Bearzatto, B.; Chibani, N.; Smits, B.; Durant, J.F.; Vybornov, A.; Thellin, O.; El Moualij, B.; et al. Diagnostic Value of IgM and IgG Detection in COVID-19 Diagnosis by the Mobile Laboratory B-LiFE: A Massive Testing Strategy in the Piedmont Region. Int. J. Environ. Res. Public Health 2021, 18, 3372. https://doi.org/10.3390/ijerph18073372
Nyabi O, Bentahir M, Ambroise J, Bearzatto B, Chibani N, Smits B, Durant JF, Vybornov A, Thellin O, El Moualij B, et al. Diagnostic Value of IgM and IgG Detection in COVID-19 Diagnosis by the Mobile Laboratory B-LiFE: A Massive Testing Strategy in the Piedmont Region. International Journal of Environmental Research and Public Health. 2021; 18(7):3372. https://doi.org/10.3390/ijerph18073372
Chicago/Turabian StyleNyabi, Omar, Mostafa Bentahir, Jérôme Ambroise, Bertrand Bearzatto, Nawfal Chibani, Benjamin Smits, Jean François Durant, Aleksandr Vybornov, Olivier Thellin, Benaissa El Moualij, and et al. 2021. "Diagnostic Value of IgM and IgG Detection in COVID-19 Diagnosis by the Mobile Laboratory B-LiFE: A Massive Testing Strategy in the Piedmont Region" International Journal of Environmental Research and Public Health 18, no. 7: 3372. https://doi.org/10.3390/ijerph18073372
APA StyleNyabi, O., Bentahir, M., Ambroise, J., Bearzatto, B., Chibani, N., Smits, B., Durant, J. F., Vybornov, A., Thellin, O., El Moualij, B., & Gala, J.-L. (2021). Diagnostic Value of IgM and IgG Detection in COVID-19 Diagnosis by the Mobile Laboratory B-LiFE: A Massive Testing Strategy in the Piedmont Region. International Journal of Environmental Research and Public Health, 18(7), 3372. https://doi.org/10.3390/ijerph18073372








