Antimicrobial Resistance in Humans, Animals, Water and Household Environs in Rural Andean Peru: Exploring Dissemination Pathways through the One Health Lens
Abstract
1. Introduction
2. Methods
2.1. Study Site
2.2. Study Design
2.2.1. Sample Collection
Human and Animal Fecal Samples
Environmental Samples
2.2.2. Laboratory Analysis of Samples
Antibiotic Susceptibility Testing
Extended Spectrum Beta Lactamases (ESBL) Detection and Confirmation
2.2.3. Questionnaires
2.3. Data Analysis
2.4. Ethics
3. Results
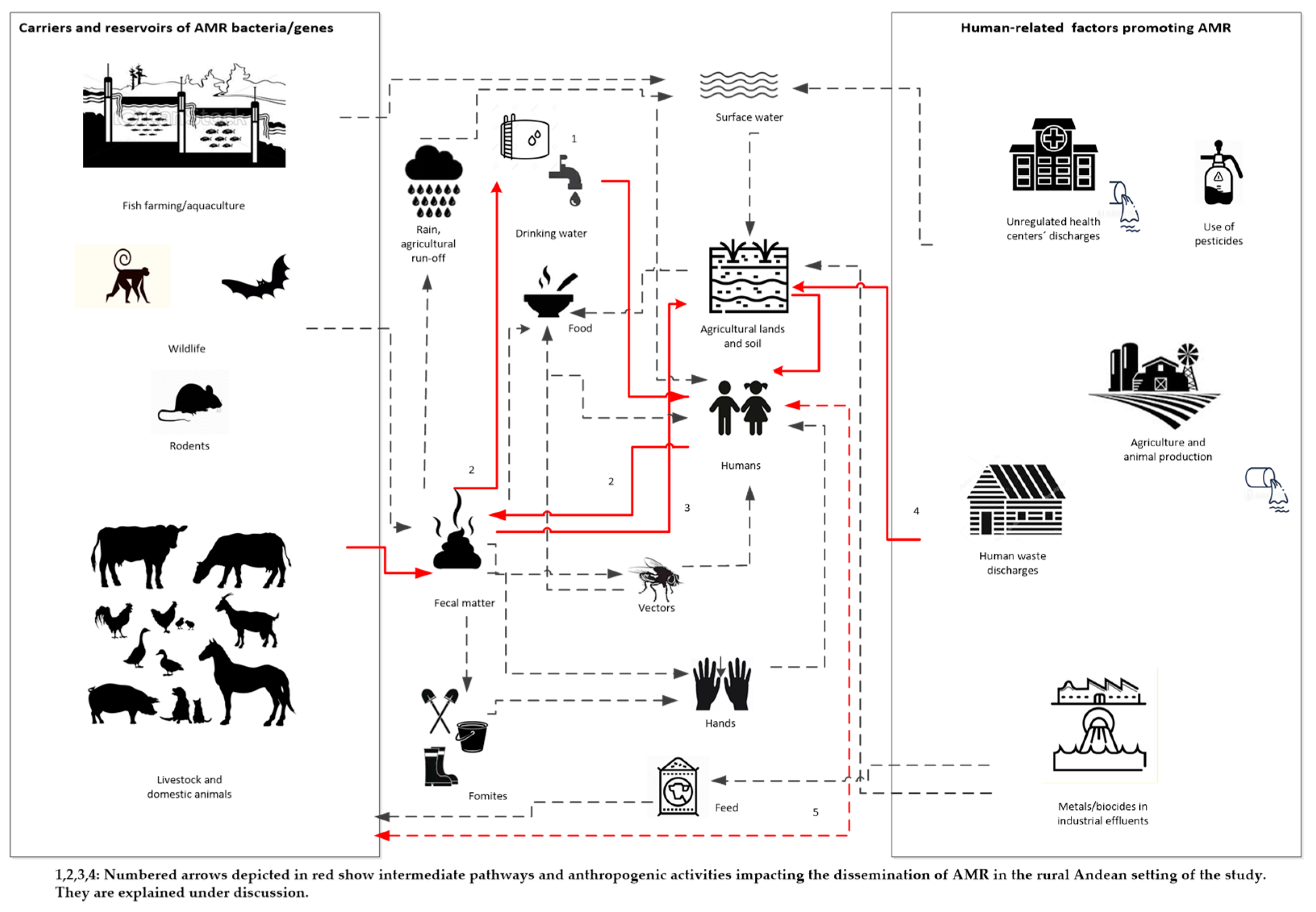
3.1. AMR Dissemination Pathways in Rural Settings
3.2. Setting Description
3.3. Water Samples
3.4. Soil Samples
3.5. Child Fecal Samples
3.6. Animal Fecal Samples
3.7. Multidrug Resistance Profiles
3.8. Detection of ESBL Resistance Genes
4. Discussion
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AMR | Antimicrobial resistance |
| LMIC | Low- and middle-income countries |
| CFU | colonies forming units |
| CLSI | Clinical and Laboratory Standards Institute |
| JASS | community water supply and irrigation committee |
| HWT | Household water treatment |
| MDR | Multidrug-resistant |
References
- Pokharel, S.; Raut, S.; Adhikari, B. Tackling antimicrobial resistance in low-income and middle-income countries. BMJ Glob. Health 2019, 4, 4–6. [Google Scholar] [CrossRef]
- Dadgostar, P. Antimicrobial resistance: Implications and costs. Infect. Drug Resist. 2019, 12, 3903–3910. [Google Scholar] [CrossRef]
- World Health Organization. Global Action Plan on Antimicrobial Resistance; World Health Organization: Geneva, Switzerland, 2015; ISBN 9789241509763. [Google Scholar]
- Ochoa, T.J.; Egoavil, M.; Castillo, M.E.; Reyes, I.; Chaparro, E.; Silva, W.; Campos, F.; Sáenz, A. Invasive pneumococcal diseases among hospitalized children in lima, Peru. Rev. Panam. Salud Publica/Pan Am. J. Public Health 2010, 28, 121–127. [Google Scholar] [CrossRef][Green Version]
- Pan American Health Organization. Tuberculosis in the Americas 2018 Regional Report; Pan American Health Organization: Washington, DC, USA, 2018. [Google Scholar]
- Tien, V.; Punjabi, C.; Holubar, M.K. Antimicrobial resistance in sexually transmitted infections. J. Travel Med. 2020, 27, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Szmolka, A.; Nagy, B. Multidrug resistant commensal Escherichia coli in animals and its impact for public health. Front. Microbiol. 2013, 4, 1–13. [Google Scholar] [CrossRef]
- Van Schaik, W. The human gut resistome. Philos. Trans. R. Soc. B 2015, 370, 20140087. [Google Scholar] [CrossRef]
- Purohit, M.R.; Lindahl, L.F.; Diwan, V.; Marrone, G.; Lundborg, C.S. High levels of drug resistance in commensal E. coli in a cohort of children from rural central India. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Pons, M.J.; Mosquito, S.; Gomesa, C.; Del Valle, L.J.; Ochoa, T.J.; Ruiz, J. Analysis of quinolone-resistance in commensal and diarrheagenic Escherichia coli isolates from infants in Lima, Peru. Trans. R. Soc. Trop. Med. Hyg. 2014, 108, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Miranda, J.; Pinto, J.; Faustino Arias, D.M.; Sánchez-Jacinto, B.; Ramirez, F. Antimicrobial resistance of uropathogens in older adults in a private clinic in Lima, Peru. Rev. Peru. Med. Exp. Salud Publica 2019, 36, 87–92. [Google Scholar] [CrossRef]
- Marcos-Carbajal, P.; Galarza-Pérez, M.; Huancahuire-Vega, S.; Otiniano-Trujillo, M.; Soto-Pastrana, J. Comparación de los perfiles de resistencia antimicrobiana de Escherichia coli uropatógena e incidencia de la producción de betalactamasas de espectro extendido en tres establecimientos privados de salud de Perú. Biomédica 2020, 40, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Von Wintersdorff, C.J.H.; Penders, J.; Van Niekerk, J.M.; Mills, N.D.; Majumder, S.; Van Alphen, L.B.; Savelkoul, P.H.M.; Wolffs, P.F.G. Dissemination of antimicrobial resistance in microbial ecosystems through horizontal gene transfer. Front. Microbiol. 2016, 7, 1–10. [Google Scholar] [CrossRef]
- Pons, M.J.; Mosquito, S.; Ochoa, T.J.; Vargas, M.; Molina, M.; Lluque, A.; Gil, A.I.; Ecker, L.; Barletta, F.; Lanata, C.F.; et al. Niveles de resistencia a quinolonas y otros antimicrobianos en cepas de Escherichia coli comensales en niños de la zona periurbana de Lima, Perú. Rev. Peru. Med. Exp. Salud Publica 2012, 29, 82–86. [Google Scholar] [CrossRef]
- Alzamora, M.C.; Echevarría, A.C.; Ferraro, V.M.; Riveros, M.D.; Zambruni, M.; Ochoa, T.J. Resistencia Antimicrobiana de cepas comensales de Escherichia coli en niños de dos comunidades rurales peruanas. Rev. Peru. Med. Exp. Salud Publica 2019, 36, 459–464. [Google Scholar] [CrossRef]
- Kalter, H.D.; Gilman, R.H.; Moulton, L.H.; Cullotta, A.R.; Cabrera, L.; Velapatiño, B. Risk factors for antibiotic-resistant Escherichia coli carriage in young children in Peru: Community-based cross-sectional prevalence study. Am. J. Trop. Med. Hyg. 2010, 82, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Larson, A.; Hartinger, S.M.; Riveros, M.; Salmon-Mulanovich, G.; Hattendorf, J.; Verastegui, H.; Huaylinos, M.L.; Mäusezahl, D. Antibiotic-resistant Escherichia coli in drinking water samples from rural andean households in Cajamarca, Peru. Am. J. Trop. Med. Hyg. 2019, 100, 1–6. [Google Scholar] [CrossRef]
- World Health Organization. Antimicrobial Resistance: An Emerging Water, Sanitation and Hygiene Issue; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Gonzáles Mendoza, J.; Maguiña Vargas, C.; de Gonzáles Ponce, F.M. Resistance to antibacterial agents: A serious problem. Acta Med. Peru. 2019, 36, 145–151. [Google Scholar] [CrossRef]
- Organización Panamericana de la Salud. Plan de Accion Sobre la Resistencia a los Antimicrobianos; Organización Panamericana de la Salud: Washington, DC, USA, 2015. [Google Scholar]
- World Health Organization. Antimicrobial Resistance: Global Report on Surveillance; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- Palhares, J.C.P.; Kich, J.D.; Bessa, M.C.; Biesus, L.L.; Berno, L.G.; Triques, N.J. Salmonella and antimicrobial resistance in an animal-based agriculture river system. Sci. Total Environ. 2014, 472, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Miranda, C.D.; Zemelman, R. Antimicrobial multiresistance in bacteria isolated from freshwater Chilean salmon farms. Sci. Total Environ. 2002, 293, 207–218. [Google Scholar] [CrossRef]
- Bengtsson-Palme, J.; Kristiansson, E.; Larsson, D.G.J. Environmental factors influencing the development and spread of antibiotic resistance. FEMS Microbiol. Rev. 2018, 42, 68–80. [Google Scholar] [CrossRef]
- Singer, A.C.; Shaw, H.; Rhodes, V.; Hart, A. Review of antimicrobial resistance in the environment and its relevance to environmental regulators. Front. Microbiol. 2016, 7, 1–22. [Google Scholar] [CrossRef]
- Redding, L.E.; Cubas-Delgado, F.; Sammel, M.D.; Smith, G.; Galligan, D.T.; Levy, M.Z.; Hennessy, S. Antibiotic residues in milk from small dairy farms in rural Peru. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2014, 31, 1001–1008. [Google Scholar] [CrossRef]
- World Organisation for Animal Health One Health at a Glance. Available online: https://www.oie.int/en/for-the-media/onehealth/ (accessed on 9 March 2021).
- World Organisation for Animal Health International Partnership to Address Human-Animal-Environment Health Risks Gets a Boost. Available online: https://www.oie.int/en/for-the-media/press-releases/detail/article/international-partnership-to-address-human-animal-environment-health-risks-gets-a-boost/ (accessed on 9 March 2021).
- World Health Organization Global Framework for Development & Stewardship to Combat Antimicrobial Resistance: Draft. Available online: https://www.who.int/publications/m/item/global-framework-for-development-stewardship-to-combat-antimicrobial-resistance-draft (accessed on 9 March 2021).
- World Health Organization. WHO Report on Surveillance of Antibiotic Consumption 2016–2018 Early Implementation; World Health Organization: Geneva, Switzerland, 2018; ISBN 9789241514880. [Google Scholar]
- Dirección General de Medicamentos Insumos y Drogas (DIGEMID). Plan Nacional Contra la Resistencia a los Antimicrobianos; DIGEMID: Lima, Peru, 2017. [Google Scholar]
- Elías, R.; Berenguel, R.; Beraún, Y.; Enrique, C.; Vásquez, P. Gestión y vigilancia sanitaria de la fauna silvestre en el Perú. Salud Tecnol. Vet. 2020, 8, 19–26. [Google Scholar] [CrossRef]
- Schneider, M.C.; Aguilera, X.P.; Smith, R.M.; Moynihan, M.J.; Da Silva, J.B.; Aldighieri, S.; Almiron, M. Importance of animal/human health interface in potential Public Health Emergencies of International Concern in the Americas. Rev. Panam. Salud Publica/Pan Am. J. Public Health 2011, 29, 371–379. [Google Scholar] [CrossRef]
- Queenan, K.; Garnier, J.; Nielsen, L.R.; Buttigieg, S.; De Meneghi, D.; Holmberg, M.; Zinsstag, J.; Rüegg, S.; Häsler, B.; Kock, R. Roadmap to a one health agenda 2030. CAB Rev. 2017, 12, 17. [Google Scholar] [CrossRef]
- Rosa, G.; Huaylinos, M.L.; Gil, A.; Lanata, C.; Clasen, T. Assessing the consistency and microbiological effectiveness of household water treatment practices by urban and rural populations claiming to treat their water at home: A case study in Peru. PLoS ONE 2014, 9, e114997. [Google Scholar] [CrossRef]
- Hartinger, S.M.; Nuño, N.; Hattendorf, J.; Verastegui, H.; Karlen, W.; Ortiz, M.; Mäusezahl, D. A factorial cluster-randomised controlled trial combining home-environmental and early child development interventions to improve child health and development: Rationale, trial design and baseline findings. BMC Med Res. Methodol. 2020, 20, 1–12. [Google Scholar] [CrossRef] [PubMed]
- OXFAM-DELAGUA. Oxfam–Delagua Portable Water Testing Kit-Users Manual-Version 4.3; OXFAM-DELAGUA: Wiltshire, UK, 2012; pp. 1–64. [Google Scholar]
- MacFaddin, J.F. Biochemical Tests for Identification of Medical Bacteria, 3rd ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2000. [Google Scholar]
- Clinical and Laboratory Standards Institute (CLSI) Performance Standards for Antimicrobial Susceptibility Testing. 30th ed. Available online: https://clsi.org/standards/products/microbiology/documents/m100/ (accessed on 8 March 2021).
- Autoridad Nacional del Agua Comparison of Four Phenotypic Methods to Detect Extended-Spectrum Betalactamases. Available online: https://www.ana.gob.pe/normatividad/resolucion-administrativa-no-656-2017-ana-aaa-xipaala-bap (accessed on 8 February 2021).
- Belaaouaj, A.; Lapoumeroulie, C.; Caniça, M.M.; Vedel, G.; Névot, P.; Krishnamoorthy, R.; Paul, G. Nucleotide sequences of the genes coding for the TEM-like β-lactamases IRT-1 and IRT-2 (formerly called TRI-1 and TRI-2). FEMS Microbiol. Lett. 1994, 120, 75–80. [Google Scholar] [CrossRef][Green Version]
- Pitout, J.D.D.; Thomson, K.S.; Hanson, N.D.; Ehrhardt, A.F.; Moland, E.S.; Sanders, C.C. β-lactamases responsible for resistance to expanded-spectrum cephalosporins in Klebsiella pneumoniae, Escherichia coli, and Proteus mirabilis isolates recovered in South Africa. Antimicrob. Agents Chemother. 1998, 42, 1350–1354. [Google Scholar] [CrossRef]
- Jouini, A.; Vinué, L.; Ben Slama, K.; Sáenz, Y.; Klibi, N.; Hammami, S.; Boudabous, A.; Torres, C. Characterization of CTX-M and SHV extended-spectrum β-lactamases and associated resistance genes in Escherichia coli strains of food samples in Tunisia. J. Antimicrob. Chemother. 2007, 60, 1137–1141. [Google Scholar] [CrossRef]
- Batchelor, M.; Hopkins, K.; Threlfall, E.J.; Clifton-Hadley, F.A.; Stallwood, A.D.; Davies, R.H.; Liebana, E. blaCTX-M genes in clinical Salmonella isolates recovered from humans in England and Wales from 1992 to 2003. Antimicrob. Agents Chemother. 2005, 49, 1319–1322. [Google Scholar] [CrossRef]
- World Health Organization. Antimicrobial Resistance. Available online: https://www.who.int/health-topics/antimicrobial-resistance (accessed on 30 March 2021).
- Magiorakos, A.P.; Srinivasan, A.; Carey, R.B.; Carmeli, Y.; Falagas, M.E.; Giske, C.G.; Harbarth, S.; Hindler, J.F.; Kahlmeter, G.; Olsson-Liljequist, B.; et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: An international expert proposal for interim standard definitions for acquired resistance. Clin. Microbiol. Infect. 2012, 18, 268–281. [Google Scholar] [CrossRef] [PubMed]
- Lebov, J.; Grieger, K.; Womack, D.; Zaccaro, D.; Whitehead, N.; Kowalcyk, B.; MacDonald, P.D.M. A framework for One Health research. One Health 2017, 3, 44–50. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for Drinking Water, 4th ed.; WHO: Geneva, Switzerland, 2017; ISBN 9789241549950. [Google Scholar]
- Ministerio de Salud. Reglamento de la Calidad del Agua para Consumo Humano; Ministerio de Salud: Lima, Peru, 2011; pp. 1–46. [Google Scholar]
- MVCS. DATASS: Modelo Para la Toma de Decisiones en Saneamiento Sistema de Diagnóstico Sobre Abastecimiento de Agua y Saneamiento en el Ámbito Rural; Ministerio de Vivienda, Construcción y Saneamiento: Lima, Peru, 2019.
- Bartram, J.; Bos, R.; Dufour, A. (Eds.) Animal Waste, Water Quality and Human Health; World Health Organizations: London, UK, 2013; Volume 12, ISBN 9781780401232.
- CARE-Perú. Diagnóstico de Saneamiento Integral de la Región Cajamarca; CARE-Peru: Lima, Peru, 2008. [Google Scholar]
- Gil, A.I.; Lanata, C.F.; Hartinger, S.M.; Mäusezahl, D.; Padilla, B.; Ochoa, T.J.; Lozada, M.; Pineda, I.; Verastegui, H. Fecal contamination of food, water, hands, and kitchen utensils at the household level in rural areas of Peru. J. Environ. Health 2014, 76, 102–106. [Google Scholar]
- WHO. Managing Water in the Home: Accelerated Health Gains from Improved Water Supply; World Health Organization: Geneva, Switzerland, 2002; Volume 8, pp. 1–83. [Google Scholar]
- Granados-Chinchilla, F.; Rodríguez, C. Tetracyclines in Food and Feedingstuffs: From Regulation to Analytical Methods, Bacterial Resistance, and Environmental and Health Implications. J. Anal. Methods Chem. 2017, 2017. [Google Scholar] [CrossRef]
- Gu, Y.; Shen, S.; Han, B.; Tian, X.; Yang, F.; Zhang, K. Family livestock waste: An ignored pollutant resource of antibiotic resistance genes. Ecotoxicol. Environ. Saf. 2020, 197, 110567. [Google Scholar] [CrossRef]
- Wang, L.; Wang, J.; Wang, J.; Zhu, L.; Yang, L.; Yang, R. Distribution characteristics of antibiotic resistant bacteria and genes in fresh and composted manures of livestock farms. Sci. Total Environ. 2019, 695, 133781. [Google Scholar] [CrossRef]
- Mishra, M.; Arukha, A.P.; Patel, A.K.; Behera, N.; Mohanta, T.K.; Yadav, D. Multi-drug resistant coliform: Water sanitary standards and health hazards. Front. Pharmacol. 2018, 9, 1–8. [Google Scholar] [CrossRef]
- Agensi, A.; Tibyangye, J.; Tamale, A.; Agwu, E.; Amongi, C. Contamination Potentials of Household Water Handling and Storage Practices in Kirundo Subcounty, Kisoro District, Uganda. J. Environ. Public Health 2019, 2019, 1–8. [Google Scholar] [CrossRef]
- Levy, K.; Nelson, K.L.; Hubbard, A.; Eisenberg, J.N.S. Following the water: A controlled study of drinking water storage in Northern Coastal Ecuador. Environ. Health Perspect. 2008, 116, 1533–1540. [Google Scholar] [CrossRef]
- Ercumen, A.; Pickering, A.J.; Kwong, L.H.; Arnold, B.F.; Parvez, S.M.; Alam, M.; Sen, D.; Islam, S.; Kullmann, C.; Chase, C.; et al. Animal Feces Contribute to Domestic Fecal Contamination: Evidence from E. coli Measured in Water, Hands, Food, Flies, and Soil in Bangladesh. Environ. Sci. Technol. 2017, 51. [Google Scholar] [CrossRef]
- Zambrano, L.D.; Levy, K.; Menezes, N.P.; Freeman, M.C. Human diarrhea infections associated with domestic animal husbandry: A systematic review and meta-analysis. Trans. R. Soc. Trop. Med. Hyg. 2014, 108, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention Shigellosis-Chapter 4-2020 Yellow Book|Travelers’ Health|CDC. Available online: https://wwwnc.cdc.gov/travel/yellowbook/2020/travel-related-infectious-diseases/shigellosis (accessed on 31 March 2021).
- Ortega-Paredes, D.; Haro, M.; Leoro-Garzón, P.; Barba, P.; Loaiza, K.; Mora, F.; Fors, M.; Vinueza-Burgos, C.; Fernández-Moreira, E. Multidrug-resistant Escherichia coli isolated from canine faeces in a public park in Quito, Ecuador. J. Glob. Antimicrob. Resist. 2019, 18, 263–268. [Google Scholar] [CrossRef]
- Ferrer, N.; Folch, A.; Masó, G.; Sanchez, S.; Sanchez-Vila, X. What are the main factors influencing the presence of faecal bacteria pollution in groundwater systems in developing countries? J. Contam. Hydrol. 2020, 228, 103556. [Google Scholar] [CrossRef] [PubMed]
| N | Mean [SD] or % (N) | |
|---|---|---|
| Demographic characteristics | 40 | |
| Number of inhabitants per household | 5.0 [1.44] | |
| Number of children under 6 per household | 1.4 [0.53] | |
| Household characteristics | ||
| Adobe wall type | ||
| - Coated adobe or rammed earth | 60 (24) | |
| - Uncoated adobe or rammed earth | 22.5 (9) | |
| Latrines w/o ventilation | ||
| - Septic tank | 22.5 (9) | |
| - Latrine | 75 (30) | |
| Piped water supply | ||
| - Public water supply system/piped water in the house | 72.5 (29) | |
| - Public water supply system/piped water outside the house | 20 (8) | |
| - Public water supply system/piped water outside the house but inside the building | 2.5 (1) | |
| - Surface water, spring | 5 (2) | |
| Energy source | ||
| - Electricity | 87.5 (35) | |
| - Candle | 5 (2) | |
| - Solar Panel | 7.5 (3) | |
| Household Water Treatment | ||
| Boiling | 60 (24) | |
| Chlorine or bleach | 12.5 (5) | |
| None | 27.5 (11) | |
| Animal Management and treatment | ||
| The last time the animal was treated; did the animal receive any antibiotic? | 72.5 (29) | |
| Antibiotic used for the treatment | ||
| - Amoxicillin | 3.1 (1) | |
| - “Biomizona“ 1 | 21.8 (7) | |
| - “Ciclosona” 2 | 50 (16) | |
| - “Emicina” 3 | 3.1 (1) | |
| - “Hipradoxi S” 4 | 3.1 (1) | |
| - “Hipralona” 5 | 6.2 (2) | |
| - “Quinolaba” 5 | 6.2 (2) | |
| - “Tylogen” 6 | 6.2 (2) | |
| Where did they get the antibiotic? | ||
| - Directly form a veterinarian | 18.7 (6) | |
| - Directly from a veterinarian technician | 50 (16) | |
| - From a neighbour or relative | 3.1 (1) | |
| - At a local veterinary store | 18.7 (6) | |
| - At a veterinary store in the area | 0 | |
| - At a pharmacy | 0 | |
| - Other place | 9.3 (3) |
| Coliforms (Count) | Water from Reservoir (N = 26) % (N) | Main Household’s Water (N = 40) % (N) | Child’s Drinking Water (N = 40) % (N) |
|---|---|---|---|
| Thermotolerant coliform count (IQR 1st–3rd Quantile) | 0–3.75 | 0–10.5 | 0–9.5 |
| Thermotolerant coliform (CFU/mL)—mean (SD) | 14.3 (59.2) | 36.2 (108.4) | 104.1 (373.5) |
| Total positive thermotolerant sample | 34.6 (9) | 45 (18) | 32.5 (13) |
| Total thermotolerant bacterial isolates * | N = 14 % (n) | N = 28 % (n) | N = 27 % (n) |
| Total positive Enterobacteriaceae isolates | 92.8 (13) | 82.1 (23) | 74.0 (20) |
| E. coli | 92.8 (13) | 57.1 (16) | 44.4 (12) |
| Klebsiella spp. | 0 | 10.7 (3) | 14.8 (4) |
| Enterobacter spp. | 0 | 14.8 (4) | 14.2 (4) |
| Water from Reservoir | Main Household’s Water | Child’s Drinking Water | |||
|---|---|---|---|---|---|
| E. coli N = 13 | E. coli N = 16 | Klebsiella spp. N = 3 | E. coli N = 12 | Klebsiella spp. N = 4 | |
| Antibiotic | Resistance % (N) | Resistance % (N) | Resistance % (N) | ||
| Amoxicillin-clavulanic acid | 0 | 0 | 0 | 0 | 0 |
| Ampicillin | 0 | 18.8 (3) | 100 (3) | 25 (3) | 100 (4) |
| Aztreonam | 0 | 0 | 0 | 0 | 0 |
| Cefotaxime | 15.4 (2) | 0 | 0 | 8.3 (1) | 0 |
| Cefoxitin | 0 | 0 | 0 | 0 | 0 |
| Chloramphenicol | 7.7 (1) | 12.5 (2) | 0 | 25 (3) | 0 |
| Ciprofloxacin | 0 | 6.3 (1) | 33.3 (1) | 16.7 (2) | 25 (1) |
| Gentamicin | 0 | 6.3 (1) | 0 | 8.3 (1) | 0 |
| Nalidixic acid | 0 | 18.8 (3) | 0 | 16.7 (2) | 0 |
| Trimethoprim-sulfamethoxazole | 0 | 0 | 33.3 (1) | 8.3 (1) | 25 (1) |
| Tetracycline | 15.4 (2) | 31.3 (5) | 33.3 (1) | 41.7 (5) | 25 (1) |
| Ceftriazone | 0 | 0 | 0 | 11.1 (1) | 0 |
| Cefepime | 0 | 0 | 0 | 8.3 (1) | 0 |
| Imipenem | 0 | 0 | 0 | 0 | 0 |
| AMR to at least one antibiotic 1 | 30.8(4) | 43.8 (3) | 100 (3) | 41.7 (5) | 100 (4) |
| Multidrug resistance 2 | 0 (0) | 18.6 (3) | 33.3 (1) | 33.3 (4) | 25 (1) |
| Coliforms | Soil (N = 40) % (n) | Child Faeces (N = 40) % (n) | Animal Faeces (N = 80) % (n) |
|---|---|---|---|
| Thermotolerant coliforms | 100 (40) | 97.5 (39) | 67.5 (54) |
| Total thermotolerant bacterial isolates | N = 83 % (n) | N = 98 % (n) | N = 116 % (n) |
| E. coli | 43.3 (36) | 45.3 (44) | 38.7 (45) |
| Klebsiella spp. | 4.8 (4) | 11.3 (11) | 5.1 (6) |
| Enterobacter spp. | 24.1 (20) | 6.2 (6) | 4.3 (5) |
| Citrobacter spp. | 9.6 (8) | 9.2 (9) | 16.3 (19) |
| Soil | Child Faeces | Animal Faeces | ||||
|---|---|---|---|---|---|---|
| E. coli N = 36 | Klebsiella spp. N = 4 | E. coli N = 44 | Klebsiella spp. N = 11 | E. coli N = 45 | Klebsiella spp. N = 6 | |
| Antibiotic | Resistance % (N) | Resistance % (N) | Resistance % (N) | |||
| Amoxicillin-clavulanic acid | 0 | 25 (1) | 0 | 9.1 (1) | 4.4 (2) | 0 |
| Ampicillin | 11.1 (4) | 75 (3) | 34.1 (15) | 54.5 (6) | 11.1 (5) | 50 (3) |
| Aztreonam | 0 | 0 | 2.3 (1) | 0 | 2.2 (1) | 0 |
| Cefotaxime | 0 | 0 | 0 | 0 | 0 | 0 |
| Cefoxitin | 2.8 (1) | 25 (1) | 0 | 9.1 (1) | 4.4 (2) | 0 |
| Chloramphenicol | 2.8 (1) | 0 | 4.5 (2) | 0 | 11.1 (5) | 0 |
| Ciprofloxacin | 5.5 (2) | 0 | 11.4 (5) | 0 | 8.8 (4) | 0 |
| Gentamicin | 0 | 0 | 2.3 (1) | 0 | 0 | 0 |
| Nalidixic acid | 5.5 (2) | 0 | 13.6 (6) | 0 | 20 (9) | 0 |
| Trimethoprim-sulfamethoxazole | 5.5 (2) | 0 | 20.5 (9) | 9.1 (1) | 11.1 (5) | 0 |
| Tetracycline | 25.0 (9) | 0 | 25.0 (11) | 9.1 (1) | 26.6 (12) | 0 |
| Ceftriazone | 0 | 0 | 2.3 (1) | 0 | 0 | 0 |
| Cefepime | 0 | 0 | 0 | 0 | 0 | 0 |
| Imipenem | 0 | 0 | 0 | 0 | 0 | 0 |
| AMR1 | 33.3 (12) | 75 (3) | 52.3 (23) | 54.6 (6) | 37.7 (17) | 50 (3) |
| MDR2 | 2.8 (1) | 0 (0) | 15.9 (7) | 0 (0) | 0 (0) | 0 (0) |
| Source Type *** | MDR | Antimicrobial Class ** |
|---|---|---|
| Child Water | AMP, TE y C | Penicillin |
| Child Water | SXT, TE, C | Sulfonamides, tetracycline, quinolone |
| Child Water | NA, CIP y TE | Quinolone, tetracycline |
| Child Water * | AMP, CTX, CRO, FEP, NA, CIP, TE, C y CN | Penicillin, 3rd & 4th generation cephalosporin, quinolone, tetracycline |
| HH Water Source | NA, TE, C y CN | Quinolone, tetracycline |
| HH Water Source | NA, CIP, SXT, TE, C | Quinolone, sulfonamides, tetracycline |
| HH Water Source | AMP, SXT, TE | Penicillin, quinolone, tetracycline |
| Soil | AMP, NA, CIP, SXT, TE, C | Penicillin, quinolone, sulfonamides, tetracycline |
| Child faeces | AMP, NA, CIP, SXT, TE y CN | Penicillin, quinolone, sulfonamides, tetracycline |
| Child faeces | AMP, SXT, TE | Penicillin, sulfonamides, tetracycline |
| Child faeces | AMP, TE, C | Penicillin, tetracycline, quinolone |
| Child faeces | AMP, SXT, TE | Penicillin, sulfonamides, tetracycline |
| Child faeces | CRO, CIP, SXT | 3rd generation cephalosporin, quinolone, sulfonamides |
| Child faeces | AMP, SXT, TE | Penicillin, sulfonamides |
| Child faeces | AMP, NA, CIP, SXT, TE | Penicillin, quinolone, sulfonamides, tetracycline |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hartinger, S.M.; Medina-Pizzali, M.L.; Salmon-Mulanovich, G.; Larson, A.J.; Pinedo-Bardales, M.; Verastegui, H.; Riberos, M.; Mäusezahl, D. Antimicrobial Resistance in Humans, Animals, Water and Household Environs in Rural Andean Peru: Exploring Dissemination Pathways through the One Health Lens. Int. J. Environ. Res. Public Health 2021, 18, 4604. https://doi.org/10.3390/ijerph18094604
Hartinger SM, Medina-Pizzali ML, Salmon-Mulanovich G, Larson AJ, Pinedo-Bardales M, Verastegui H, Riberos M, Mäusezahl D. Antimicrobial Resistance in Humans, Animals, Water and Household Environs in Rural Andean Peru: Exploring Dissemination Pathways through the One Health Lens. International Journal of Environmental Research and Public Health. 2021; 18(9):4604. https://doi.org/10.3390/ijerph18094604
Chicago/Turabian StyleHartinger, Stella M., Maria Luisa Medina-Pizzali, Gabriela Salmon-Mulanovich, Anika J. Larson, María Pinedo-Bardales, Hector Verastegui, Maribel Riberos, and Daniel Mäusezahl. 2021. "Antimicrobial Resistance in Humans, Animals, Water and Household Environs in Rural Andean Peru: Exploring Dissemination Pathways through the One Health Lens" International Journal of Environmental Research and Public Health 18, no. 9: 4604. https://doi.org/10.3390/ijerph18094604
APA StyleHartinger, S. M., Medina-Pizzali, M. L., Salmon-Mulanovich, G., Larson, A. J., Pinedo-Bardales, M., Verastegui, H., Riberos, M., & Mäusezahl, D. (2021). Antimicrobial Resistance in Humans, Animals, Water and Household Environs in Rural Andean Peru: Exploring Dissemination Pathways through the One Health Lens. International Journal of Environmental Research and Public Health, 18(9), 4604. https://doi.org/10.3390/ijerph18094604






