Effect of Grandmaternal Smoking on Body Size and Proportions at Birth
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Exposure
2.3. Outcomes
2.4. Covariates
2.5. Statistical Analysis
2.6. Ethics Approval and Register Data Permit
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kiuru, S.; Gissler, M.; Heino, A. TILASTORAPORTTI 48/2020: Perinataalitilasto—Synnyttäjät, Synnytykset ja Vastasyntyneet 2019. Finnish Institute for Health and Welfare. 2020. Available online: https://www.julkari.fi/bitstream/handle/10024/140702/Tr48_20.pdf?sequence=1&isAllowed=y (accessed on 10 March 2021). (In Finnish).
- Rillamas-Sun, E.; Harlow, S.D.E.; Randolph, J.F., Jr. Grandmothers’ smoking in pregnancy and grandchildren’s birth weight: Comparisons by grandmother birth cohort. Matern. Child Health J. 2014, 18, 1691–1698. [Google Scholar] [CrossRef] [PubMed]
- Miller, L.L.; Pembrey, M.; Smith, G.D.; Northstone, K.; Golding, J. Is the growth of the fetus of a non-smoking mother influenced by the smoking of either grandmother while pregnant? PLoS ONE 2014, 9, e86781. [Google Scholar] [CrossRef] [PubMed]
- Ding, M.; Yuan, C.; Gaskins, A.J.; Field, A.E.; Missmer, S.A.; Michels, K.B.; Hu, F.; Zhang, C.; Gillman, M.W.; Chavarro, J. Smoking during pregnancy in relation to grandchild birth weight and BMI trajectories. PLoS ONE 2017, 12, e0179368. [Google Scholar] [CrossRef] [PubMed]
- Mejia-Lancheros, C.; Mehegan, J.; Murrin, C.M.; Kelleher, C.C.; Lifeways Cross-Generation Cohort Study Group. Smoking habit from the paternal line and grand-child’s overweight or obesity status in early childhood: Prospective findings from the lifeways cross-generation cohort study. Int. J. Obes. 2018, 42, 1853–1870. [Google Scholar] [CrossRef] [PubMed]
- Dougan, M.M.; Field, A.E.; Rich-Edwards, J.W.; Hankinson, S.E.; Glynn, R.J.; Willett, W.C.; Michels, K.B. Is grand-parental smoking associated with adolescent obesity? A three-generational study. Int. J. Obes. 2016, 40, 531–537. [Google Scholar] [CrossRef]
- Golding, J.; Northstone, K.; Gregory, S.; Miller, L.L.; Pembrey, M. The anthropometry of children and adolescents may be influenced by the prenatal smoking habits of their grandmothers: A longitudinal cohort study. Am. J. Hum. Biol. 2014, 26, 731–739. [Google Scholar] [CrossRef]
- Braback, L.; Lodge, C.J.; Lowe, A.J.; Dharmage, S.C.; Olsson, D.; Forsberg, B. Childhood asthma and smoking exposures before conception—A three-generational cohort study. Pediatr. Allergy Immunol. 2018, 29, 361–368. [Google Scholar] [CrossRef]
- Accordini, S.; Calciano, L.; Johannessen, A.; Portas, L.; Benediktsdottir, B.; Bertelsen, R.J.; Braback, L.; Carsin, A.E.; Dharmage, S.C.; Dratva, J.; et al. Ageing Lungs in European Cohorts (ALEC) Study A three-generation study on the association of tobacco smoking with asthma. Int. J. Epidemiol. 2018, 47, 1106–1117. [Google Scholar] [CrossRef]
- Lodge, C.J.; Bråbäck, L.; Lowe, A.J.; Dharmage, S.C.; Olsson, D.; Forsberg, B. Grandmaternal smoking increases asthma risk in grandchildren: A nationwide Swedish cohort. Clin. Exp. Allergy 2018, 48, 167–174. [Google Scholar] [CrossRef]
- Golding, J.; Ellis, G.; Gregory, S.; Birmingham, K.; Iles-Caven, Y.; Rai, D.; Pembrey, M. Grand-maternal smoking in pregnancy and grandchild’s autistic traits and diagnosed autism. Sci. Rep. 2017, 7, 46179. [Google Scholar] [CrossRef]
- Banderali, G.; Martelli, A.; Landi, M.; Moretti, F.; Betti, F.; Radaelli, G.; Lassandro, C.; Verduci, E. Short and long term health effects of parental tobacco smoking during pregnancy and lactation: A descriptive review. J. Transl. Med. 2015, 13, 1–7. [Google Scholar] [CrossRef]
- Ekblad, M.; Korkeila, J.; Lehtonen, L. Smoking during pregnancy affects foetal brain development. Acta Paediatr. 2015, 104, 12–18. [Google Scholar] [CrossRef]
- Buck, J.M.; Sanders, K.N.; Wageman, C.R.; Knopik, V.S.; Stitzel, J.A.; O’Neill, H.C. Developmental nicotine exposure pre-cipitates multigenerational maternal transmission of nicotine preference and ADHD-like behavioral, rhythmometric, neuro-pharmacological, and epigenetic anomalies in adolescent mice. Neuropharmacology 2019, 149, 66–82. [Google Scholar] [CrossRef]
- Leslie, F.M. Multigenerational epigenetic effects of nicotine on lung function. BMC Med. 2013, 11, 27. [Google Scholar] [CrossRef]
- Barua, S.; Junaid, M.A. Lifestyle, pregnancy and epigenetic effects. Epigenomics 2015, 7, 85–102. [Google Scholar] [CrossRef]
- Jung, Y.; Hsieh, L.S.; Lee, A.M.; Zhou, Z.; Coman, D.; Heath, C.J.; Hyder, F.; Mineur, Y.S.; Yuan, Q.; Goldman, D.; et al. An epigenetic mechanism mediates developmental nicotine effects on neuronal structure and behavior. Nat. Neurosci. 2016, 19, 905–914. [Google Scholar] [CrossRef]
- Rehan, V.K.; Liu, J.; Naeem, E.; Tian, J.; Sakurai, R.; Kwong, K.; Akbari, O.; Torday, J.S. Perinatal nicotine exposure induces asthma in second generation offspring. BMC Med. 2012, 10, 129. [Google Scholar] [CrossRef]
- Finnish Institute for Health and Welfare Medical Birth Register. Available online: https://www.thl.fi/fi/web/thlfi-en/statistics/information-on-statistics/register-descriptions/newborns (accessed on 10 March 2021).
- Rumrich, I.K.; Vähäkangas, K.; Viluksela, M.; Gissler, M.; Surcel, H.-M.; De Ruyter, H.; Jokinen, J.; Hänninen, O. The MATEX cohort—A Finnish population register birth cohort to study health effects of prenatal exposures. BMC Public Health 2017, 17, 871. [Google Scholar] [CrossRef]
- Sankilampi, U.; Hannila, M.-L.; Saari, A.; Gissler, M.; Dunkel, L. New population-based references for birth weight, length, and head circumference in singletons and twins from 23 to 43 gestation weeks. Ann. Med. 2013, 45, 446–454. [Google Scholar] [CrossRef]
- McLennan, J.E.; Gilles, F.H.; Neff, R. A model of growth of the human fetal brain. In The Developing Human Brain; Elsevier BV: Amsterdam, The Netherlands, 1983; pp. 43–58. [Google Scholar]
- Statistics Finland Finnish National Classification of Occupations. 2001. Available online: http://www.stat.fi/meta/luokitukset/ammatti/001-2001/kuvaus_en.html (accessed on 10 March 2021).
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. Available online: http://www.R-project.org/ (accessed on 11 March 2021).
- Stevenson, M. Package ‘epiR’. 2021, 97–100. 2021. Available online: https://cran.r-project.org/web/packages/epiR/epiR.pdf (accessed on 10 March 2021).
- Abraham, M.; Alramadhan, S.; Iniguez, C.; Duijts, L.; Jaddoe, V.W.V.; Dekker, H.; Herman, T.D.; Crozier, S.; Godfrey, K.M.; Hindmarsh, P. A systematic review of maternal smoking during pregnancy and fetal measurements with meta-analysis. PLoS ONE 2017, 12, e0170946. [Google Scholar] [CrossRef]
- Misra, D.P.; Astone, N.; Lynch, C.D. Maternal Smoking and Birth Weight: Interaction with parity and mother’s own in utero exposure to smoking. Epidemiology 2005, 16, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Barouki, R.; Melén, E.; Herceg, Z.; Beckers, J.; Chen, J.; Karagas, M.; Puga, A.; Xia, Y.; Chadwick, L.; Yan, W.; et al. Epigenetics as a mechanism linking developmental exposures to long-term toxicity. Environ. Int. 2018, 114, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Lamminpää, R.; Vehviläinen-Julkunen, K.; Gissler, M.; Heinonen, S. Smoking among older childbearing women—A marker of risky health behaviour a registry-based study in Finland. BMC Public Health 2013, 13, 1179. [Google Scholar] [CrossRef] [PubMed]
- Räisänen, S.; Gissler, M.; Sankilampi, U.; Saari, J.; Kramer, M.R.; Heinonen, S. Contribution of socioeconomic status to the risk of small for gestational age infants—A population-based study of 1,390,165 singleton live births in Finland. Int. J. Equity Health 2013, 12, 1–28. [Google Scholar] [CrossRef]
- Rumrich, I.; Vähäkangas, K.; Viluksela, M.; Gissler, M.; de Ruyter, H.; Hänninen, O. Effects of maternal smoking on body size and proportions at birth: A register-based cohort study of 1.4 million births. BMJ Open 2020, 10, e033465. [Google Scholar] [CrossRef]
- Jääskeläinen, M.; Virtanen, S. Tupakkatilasto 2018. Finnish Institute for Health and Welfare. 2019. Available online: https://www.julkari.fi/bitstream/handle/10024/138961/Tupakkatilasto%202018_korjattu%20taulukon%2011%20ja%209%20%2819.8.2020%29.pdf?sequence=9&isAllowed=y (accessed on 10 March 2021).
- Duodecim Tupakka- ja Nikotiiniriippuvuuden Ehkäisy ja Hoito. Available online: https://www.kaypahoito.fi/hoi40020?tab=suositus (accessed on 15 July 2019). (In Finnish).
- Kreyberg, I.; Nordhagen, L.S.; Bains, K.E.S.; Alexander, J.; Becher, R.; Carlsen, K.C.L.; Glavin, K. An update on prevalence and risk of snus and nicotine replacement therapy during pregnancy and breastfeeding. Acta Paediatr. 2019, 108, 1215–1221. [Google Scholar] [CrossRef]
- Laverty, A.A.; Filippidis, F.T.; Vardavas, C.I. Patterns, trends and determinants of e-cigarette use in 28 European Union Member States 2014–2017. Prev. Med. 2018, 116, 13–18. [Google Scholar] [CrossRef]
- Peterson, L.A.; Hecht, S.S. Tobacco, e-cigarettes, and child health. Curr. Opin. Pediatr. 2017, 29, 225–230. [Google Scholar] [CrossRef]
- Whittington, J.R.; Simmons, P.M.; Phillips, A.M.; Gammill, S.K.; Cen, R.; Magann, E.F.; Cardenas, V.M. The Use of Electronic Cigarettes in Pregnancy: A Review of the Literature. Obstet. Gynecol. Surv. 2018, 73, 544–549. [Google Scholar] [CrossRef]
- Kirkpatrick, M.G.; Cruz, T.B.; Unger, J.B.; Herrera, J.; Schiff, S.; Allem, J.-P. Cartoon-based e-cigarette marketing: Associations with susceptibility to use and perceived expectations of use. Drug Alcohol Depend. 2019, 201, 109–114. [Google Scholar] [CrossRef]
- Walley, S.C.; Wilson, K.M.; Winickoff, J.P.; Groner, J. A public health crisis: Electronic cigarettes, vape, and juul. Pediatrics 2019, 143, e20182741. [Google Scholar] [CrossRef]
- England, L.J.; Aagaard, K.; Bloch, M.; Conway, K.; Cosgrove, K.; Grana, R.; Gould, T.J.; Hatsukami, D.; Jensen, F.; Kandel, D.; et al. Developmental toxicity of nicotine: A transdisciplinary synthesis and implications for emerging tobacco products. Neurosci. Biobehav. Rev. 2017, 72, 176–189. [Google Scholar] [CrossRef]
- Gibbs, K.; Collaco, J.M.; McGrath-Morrow, S.A. Impact of tobacco smoke and nicotine exposure on lung development. Chest 2016, 149, 552–561. [Google Scholar] [CrossRef]
- Holbrook, B.D. The effects of nicotine on human fetal development. Birth Defects Res. Part C Embryo Today Rev. 2016, 108, 181–192. [Google Scholar] [CrossRef]
- Erickson, A.C.; Arbour, L.T. Heavy smoking during pregnancy as a marker for other risk factors of adverse birth outcomes: A population-based study in British Columbia, Canada. BMC Public Health 2012, 12, 102. [Google Scholar] [CrossRef]
- Lamminpaa, R.; Gissler, M.; Vehvilainen-Julkunen, K. Utilization of Large Data Sets in Maternal Health in Finland: A Case for Global Health Research. J. Perinat. Neonatal Nurs. 2017, 31, 236–243. [Google Scholar] [CrossRef]
- Leonardi-Bee, J.; Smyth, A.; Britton, J.; Coleman, T. Environmental tobacco smoke and fetal health: Systematic review and meta-analysis. Arch. Dis. Child. Fetal Neonatal Ed. 2008, 93, F351–F361. [Google Scholar] [CrossRef]
- Gissler, M.; Meriläinen, J.; Vuori, E.; Hemminki, E. Register based monitoring shows decreasing socioeconomic differences in Finnish perinatal health. J. Epidemiol. Community Health 2003, 57, 433–439. [Google Scholar] [CrossRef]
- Gissler, M.; Rahkonen, O.; Arntzen, A.; Cnattingius, S.; Andersen, A.-M.N.; Hemminki, E. Trends in socioeconomic differences in Finnish perinatal health 1991–2006. J. Epidemiol. Community Health 2009, 63, 420–425. [Google Scholar] [CrossRef]
- Cavazos-Rehg, P.A.; Krauss, M.J.; Spitznagel, E.L.; Schootman, M.; Cottler, L.B.; Bierut, L.J. Substance use and the risk for sexual intercourse with and without a history of teenage pregnancy among adolescent females. J. Stud. Alcohol Drugs 2011, 72, 194–198. [Google Scholar] [CrossRef]
- Darroch, J.E.; Singh, S.; Frost, J.J. Differences in teenage pregnancy rates among five developed countries: The roles of sexual activity and contraceptive use. Fam. Plan. Perspect. 2001, 33, 244. [Google Scholar] [CrossRef]
- Leppälahti, S.; Gissler, M.; Mentula, M.; Heikinheimo, O. Trends in teenage termination of pregnancy and its risk factors: A population-based study in Finland, 1987–2009. Hum. Reprod. 2012, 27, 2829–2836. [Google Scholar] [CrossRef]
| Category | Grandmother | Mother | Frequency | % |
|---|---|---|---|---|
| n-n | No smoking | No smoking | 11,848 | 49.4 |
| y-y | Any smoking | Any smoking | 3668 | 15.3 |
| y-n | Any smoking | No smoking | 2710 | 11.3 |
| n-y | No smoking | Any smoking | 5750 | 24.0 |
| Total | 23,976 | 100.0 |
| Model | Confounder |
|---|---|
| 1 | Maternal age, sex, maternal parity, and maternal socioeconomic status |
| 2 | Model 1 + grandmaternal age, grandmaternal parity, and grandmaternal socioeconomic status |
| 3 | Model 2 + maternal low birth weight and maternal preterm birth |
| 4 | Model 3 + maternal co-morbidities during pregnancy |
| Exposure Group | Population Size | Exposed (%) | Number Exposed/Number Unexposed | Minimum Detectable OR at Given Endpoint Incidence | ||
|---|---|---|---|---|---|---|
| 10% | 5% | 1% | ||||
| Any smoking grandmother and any smoking mother | 16,000 | 15 | 0.333 | 1.19 | 1.27 | 1.66 |
| Any smoking grandmother, no smoking mother | 11,000 | 11 | 0.225 | 1.26 | 1.38 | 1.97 |
| No smoking grandmother, any smoking mother | 17,000 | 24 | 0.475 | 1.15 | 1.22 | 1.56 |
| Mean (sd) or Count (%) | ||||
|---|---|---|---|---|
| No Smoking Grandmother or Mother | Any Smoking Grandmother, any Smoking Mother | Any Smoking Grandmother, no Smoking Mother | No Smoking Grandmother, any Smoking Mother | |
| N (%) of triads | 11,848 (49.4) | 3668 (15.3) | 2710 (11.3) | 5750 (24.0) |
| Grandmother | ||||
| Age (years) | 27.8 (5.6) | 25.6 (5.5) | 25.9 (5.6) | 27 (5.5) |
| Parity (multiparous) | 8215 (69.4%) | 2345 (64.1%) | 1622 (59.9%) | 3918 (68.2%) |
| Marital status (married, registered partnership) | 9031 (76.4%) | 1463 (40.1%) | 1268 (47%) | 3739 (65.3%) |
| Socioeconomic status | ||||
| Upper white collar | 766 (6.5%) | 77 (2.1%) | 64 (2.4%) | 249 (4.3%) |
| Lower white collar | 5298 (44.7%) | 1168 (31.8%) | 945 (34.9%) | 2324 (40.4%) |
| Blue collar | 2624 (22.1%) | 1276 (34.8%) | 956 (35.3%) | 1607 (27.9%) |
| Other socioeconomic status (e.g., student, farmer, self-employed) | 2721 (23%) | 903 (24.6%) | 576 (21.3%) | 1282 (22.3%) |
| No information on socioeconomic status | 439 (3.7%) | 244 (6.7%) | 169 (6.2%) | 288 (5%) |
| Mother | ||||
| Age (years) | 21.4 (2) | 20.3 (2.1) | 21.1 (2.2) | 20.5 (2) |
| Height (cm) | 165 (6) | 164 (5.9) | 164.2 (6) | 164.9 (5.9) |
| Weight (kg) | 64.4 (14) | 65.5 (15.6) | 66.2 (15.2) | 64.9 (14.9) |
| Parity (multiparous) | 3767 (31.8%) | 871 (23.8%) | 839 (31%) | 1156 (20.1%) |
| Marital status (married, registered partnership) | 4479 (37.8%) | 386 (10.5%) | 560 (20.7%) | 867 (15.1%) |
| Socioeconomic status | ||||
| Upper white collar | 83 (0.7%) | 8 (0.2%) | 12 (0.4%) | 25 (0.4%) |
| Lower white collar | 1700 (14.3%) | 257 (7%) | 309 (11.4%) | 554 (9.6%) |
| Blue collar | 1006 (8.5%) | 287 (7.8%) | 188 (6.9%) | 577 (10%) |
| Other socioeconomic status (e.g., student, farmer, self-employed) | 2302 (19.4%) | 869 (23.7%) | 521 (19.2%) | 1340 (23.3%) |
| No information on socioeconomic status | 6757 (57%) | 2247 (61.3%) | 1680 (62%) | 3254 (56.6%) |
| Birth weight (g) | 3588.5 (504.3) | 3365.6 (489.4) | 3368.8 (496.7) | 3585.7 (506) |
| Birth length (cm) | 50.2 (2.1) | 49.3 (2.1) | 49.3 (2.2) | 50.1 (2.1) |
| Gestational age (weeks) | 39.6 (1.5) | 39.4 (1.5) | 39.5 (1.6) | 39.6 (1.5) |
| Co-morbidities (yes) | 1435 (12.1%) | 515 (14%) | 406 (15%) | 702 (12.2%) |
| Child | ||||
| Gestational age (weeks) | 39.4 (1.7) | 39.4 (1.9) | 39.5 (1.6) | 39.4 (1.8) |
| Birth weight (g) | 3509.7 (510.3) | 3376.7 (543.9) | 3528.9 (496.8) | 3416 (530.8) |
| Birth length (cm) | 50 (2.3) | 49.4 (2.5) | 50.1 (2.3) | 49.6 (2.5) |
| Head circumference (cm) | 34.8 (1.5) | 34.5 (1.7) | 34.8 (1.5) | 34.6 (1.7) |
| Sex (male) | 6104 (51.5%) | 1833 (50%) | 1404 (51.8%) | 2968 (51.6%) |
| PTB (yes) | 511 (4.3%) | 194 (5.3%) | 115 (4.2%) | 290 (5%) |
| LBW (yes) | 325 (2.8%) | 169 (4.7%) | 73 (2.7%) | 213 (3.8%) |
| Small for gestational age (<10th percentile) # (yes) | 2805 (23.7%) | 1285 (35%) | 635 (23.4%) | 1877 (32.6%) |
| Small weight for gestational age (<10th percentile) (yes) | 1710 (14.4%) | 844 (23%) | 337 (12.4%) | 1223 (21.3%) |
| Small length for gestational age (<10th percentile) (yes) | 883 (7.5%) | 486 (13.3%) | 188 (6.9%) | 621 (10.8%) |
| Small head circumference for gestational age (<10th percentile) (yes) | 1660 (14.1%) | 729 (20.1%) | 384 (14.3%) | 1046 (18.4%) |
| Low ponderal index (<10th percentile) (yes) | 1098 (10.3%) | 359 (10.8%) | 243 (10%) | 583 (11.3%) |
| High head-to-length ratio (>90th percentile) (yes) | 1120 (10.6%) | 490 (14.9%) | 237 (10%) | 723 (14.1%) |
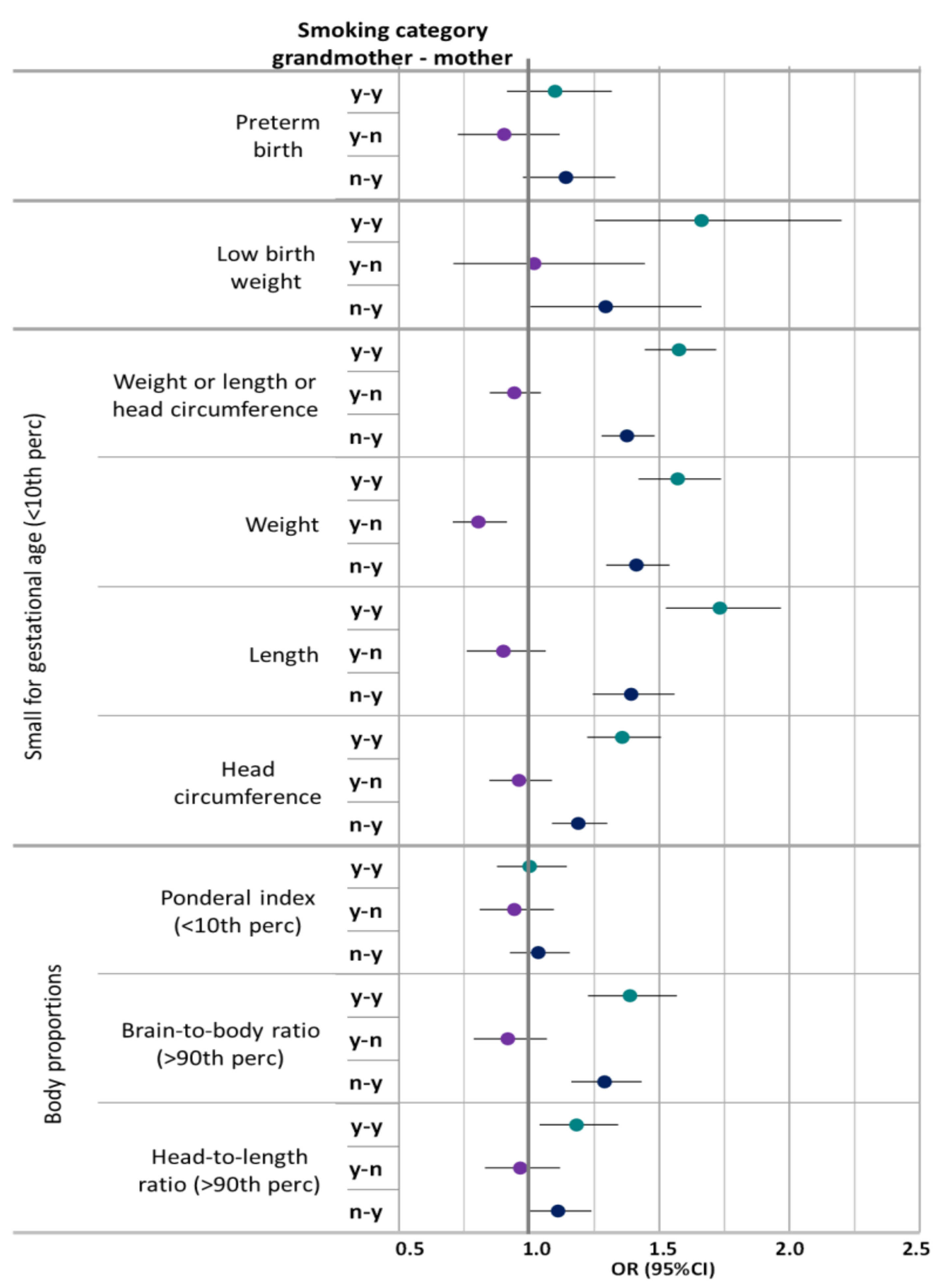
| OR (95%CI) | ||||||
|---|---|---|---|---|---|---|
| Model 3 * | Model 4 ^ | |||||
| Outcome | Any Smoking Mother and Grandmother | Any Smoking Grandmother and no Smoking Mother | No Smoking Grandmother and any Smoking Mother | Any Smoking Mother and Grandmother | Any Smoking Grandmother and no Smoking Mother | No Smoking Grandmother and any Smoking Mother |
| Preterm birth | 1.129 (0.94–1.351) | 0.944 (0.759–1.163) | 1.134 (0.971–1.323) | 1.1 (0.915–1.317) | 0.904 (0.726–1.117) | 1.141 (0.976–1.331) |
| Low birth weight | 1.658 (1.249–2.193) | 1.02 (0.709–1.446) | 1.292 (1.004–1.66) | 1.662 (1.253–2.2) | 1.019 (0.709–1.445) | 1.294 (1.005–1.662) |
| Small for gestational age | ||||||
| Birth weight(<10th percentile)) | 1.572 (1.421–1.738) | 0.807 (0.707–0.917) | 1.412 (1.296–1.539) | 1.571 (1.42–1.737) | 0.805 (0.706–0.915) | 1.412 (1.296–1.539) |
| Birth length (<10th percentile) | 1.727 (1.52–1.96) | 0.896 (0.756–1.058) | 1.393 (1.244–1.558) | 1.733 (1.525–1.967) | 0.901 (0.76–1.064) | 1.392 (1.244–1.558) |
| Head circumference (<10th percentile) | 1.351 (1.217–1.499) | 0.953 (0.841–1.079) | 1.19 (1.088–1.301) | 1.358 (1.223–1.507) | 0.961 (0.847–1.087) | 1.189 (1.087–1.3) |
| Any of the above# (<10th percentile) | 1.57 (1.438–1.713) | 0.938 (0.845–1.04) | 1.376 (1.278–1.482) | 1.576 (1.444–1.719) | 0.943 (0.849–1.045) | 1.376 (1.278–1.482) |
| Body proportions | ||||||
| Ponderal index (<10th percentile) | 1.003 (0.877–1.146) | 0.947 (0.813–1.099) | 1.035 (0.925–1.156) | 1.002 (0.876–1.145) | 0.943 (0.8100–1.095) | 1.035 (0.926–1.156) |
| Brain-to-body ratio (>90th percentile) | 1.395 (1.233–1.576) | 0.927 (0.794–1.077) | 1.288 (1.16–1.43) | 1.387 (1.226–1.568) | 0.918 (0.787–1.068) | 1.29 (1.162–1.432) |
| Head-to-length ratio (>90th percentile) | 1.176 (1.03–1.342) | 0.972 (0.833–1.131) | 1.126 (1.007–1.259) | 1.176 (1.029–1.341) | 0.969 (0.83–1.127) | 1.127 (1.007–1.259) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rumrich, I.K.; Hänninen, O.; Viluksela, M.; Vähäkangas, K. Effect of Grandmaternal Smoking on Body Size and Proportions at Birth. Int. J. Environ. Res. Public Health 2021, 18, 4985. https://doi.org/10.3390/ijerph18094985
Rumrich IK, Hänninen O, Viluksela M, Vähäkangas K. Effect of Grandmaternal Smoking on Body Size and Proportions at Birth. International Journal of Environmental Research and Public Health. 2021; 18(9):4985. https://doi.org/10.3390/ijerph18094985
Chicago/Turabian StyleRumrich, Isabell Katharina, Otto Hänninen, Matti Viluksela, and Kirsi Vähäkangas. 2021. "Effect of Grandmaternal Smoking on Body Size and Proportions at Birth" International Journal of Environmental Research and Public Health 18, no. 9: 4985. https://doi.org/10.3390/ijerph18094985
APA StyleRumrich, I. K., Hänninen, O., Viluksela, M., & Vähäkangas, K. (2021). Effect of Grandmaternal Smoking on Body Size and Proportions at Birth. International Journal of Environmental Research and Public Health, 18(9), 4985. https://doi.org/10.3390/ijerph18094985







