Hardness-Dependent Water Quality Criteria for Protection of Freshwater Aquatic Organisms for Silver in China
Abstract
:1. Introduction
2. Materials and Methods
2.1. Toxicity Data Collection and Selection
2.2. Hardness Adjustment of ATVs
2.3. Statistical Analysis and the CMC Derivation by SSD Method
2.4. The CCC Derivation by the Acute-to-Chronic Ratio (ACR) Method
3. Results and Discussion
3.1. The Database of ATVs and CTVs of Silver to Freshwater Aquatic Organisms
3.2. Derivation of the WQC for Silver
3.2.1. Correlations between Water Hardness and Toxicity of Silver
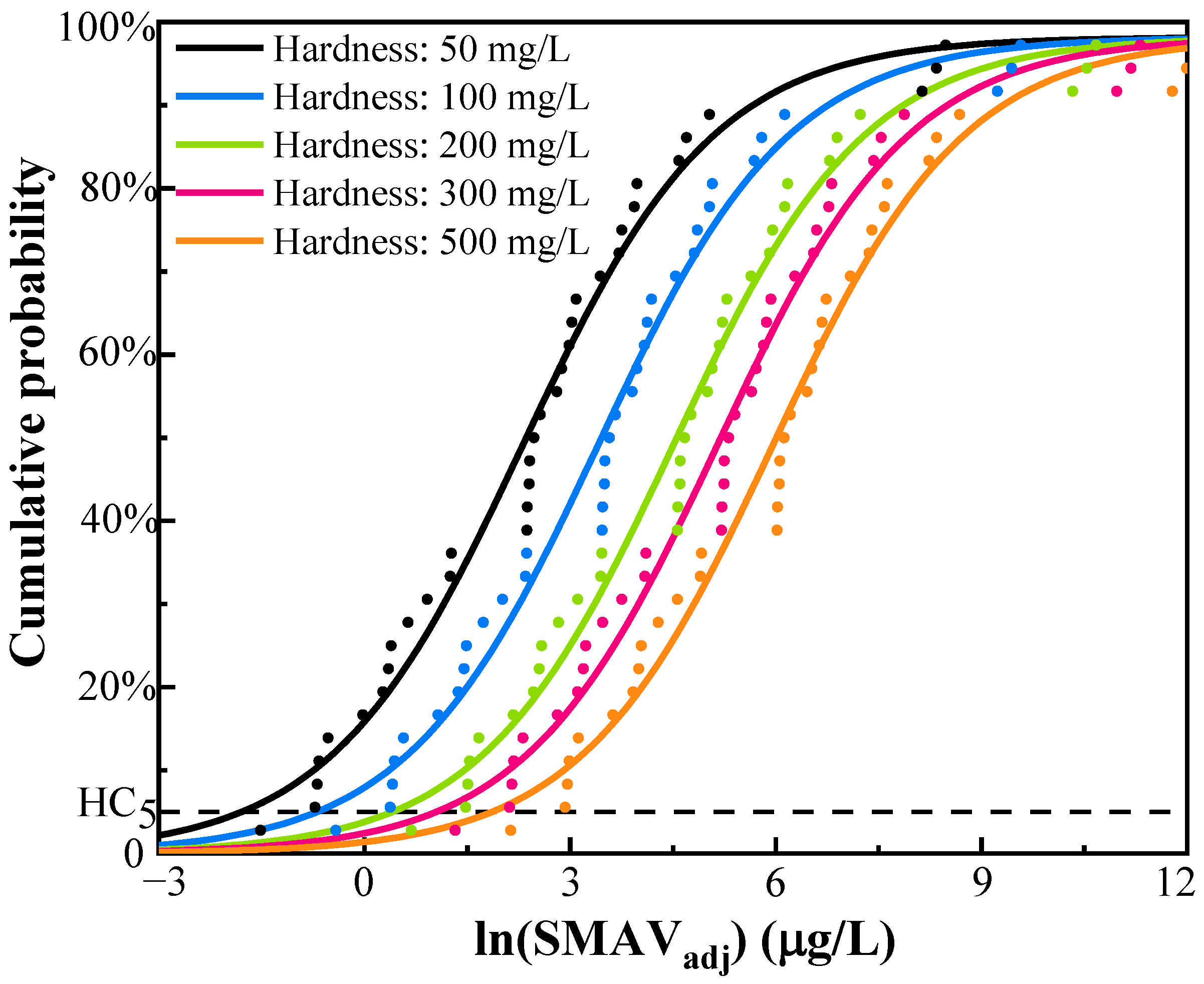
3.2.2. Hardness-Dependent WQC for Silver
| Rank | Species | Phyla | Families | N | Hardness (mg/L) | ATV (μg/L) | SMAVadj (μg/L) |
|---|---|---|---|---|---|---|---|
| 1 | Oryzias latipes | Chordata | Adrianichthyidae | 2 | 40 | 0.14–0.17 | 0.66 |
| 2 | Lymnaea luteola | Mollusca | Lymnaea | 1 | 195 | 4.2 | 1.46 |
| 3 | Lebistes reticulatus | Chordata | Poeciliidae | 1 | 250 | 6.44 | 1.51 |
| 4 | Duttaphrynus melanostictus | Chordata | Bufonidae | 1 | 185 | 4.1 | 1.55 |
| 5 | Puntius sophore | Chordata | Cyprinidae | 1 | 250 | 7.55 | 1.77 |
| 6 | Cyprinus carpio | Chordata | Cyprinidae | 1 | 118 | 3.8 | 2.92 |
| 7 | Moina dubia | Arthropoda | Moinidae | 1 | 109 | 4.5 | 3.93 |
| 8 | Daphnia magna | Arthropoda | Daphniidae | 24 | 35–255 | 0.25–49 | 4.27 |
| 9 | Channa punctatus | Chordata | Channidae | 1 | 250 | 18.89 | 4.42 |
| 10 | Hyalella azteca | Arthropoda | Hyalellidae | 2 | 35.2–47.8 | 1–1.9 | 5.66 |
| 11 | Tubifex tubifex | Annelida | Tubificid | 1 | 245 | 31 | 7.49 |
| 12 | Cyclops varicans | Arthropoda | Cyclopidae | 1 | 109 | 12 | 10.47 |
| 13 | Cottus bairdi | Chordata | Cottidae | 2 | 30–250 | 5.3–13.6 | 10.66 |
| 14 | Jordanella floridae | Chordata | Istiophoridae | 2 | 44.3–48 | 9.2–9.6 | 32.05 |
| 15 | Alona affinis | Arthropoda | Chydoridae | 1 | 109 | 37 | 32.28 |
| 16 | Monopterus albus | Chordata | Synbranchidae | 1 | 21 | 2.8 | 33.21 |
| 17 | Pimephales promelas | Chordata | Cyprinidae | 30 | 25–255 | 2.15–270 | 33.41 |
| 18 | Isonychia bicolor | Arthropoda | Isonychiidae | 1 | 35.2 | 6.8 | 35.58 |
| 19 | Ceriodaphnia reticulata | Arthropoda | Daphniidae | 1 | 45 | 11 | 38.99 |
| 20 | Daphnia pulex | Arthropoda | Daphniidae | 1 | 45 | 14 | 49.63 |
| 21 | Simocephalus vetulus | Arthropoda | Daphniidae | 1 | 45 | 15 | 53.17 |
| 22 | Oncorhynchus mykiss | Chordata | Salmonidae | 26 | 26–255 | 6.9–280 | 59.28 |
| 23 | Ictalurus punctatus | Chordata | Ictaluridae | 1 | 44.8 | 17.3 | 61.76 |
| 24 | Cambarus diogenes | Arthropoda | Cambaridae | 1 | 100 | 65.85 | 65.85 |
| 25 | Chironomus tentans | Arthropoda | Chironomidae | 1 | 25 | 10.4 | 93.58 |
| 26 | Gambusia affinis | Chordata | Poeciliidae | 1 | 35.2 | 23.5 | 122.94 |
| 27 | Lepomis macrochirus | Chordata | Centrarchidae | 3 | 35.2–44.7 | 13–64 | 128.30 |
| 28 | Ceriodaphnia dubia | Arthropoda | Daphniidae | 3 | 80–172 | 77.6–839.95 | 153.37 |
| 29 | Macrobrachium nipponense | Arthropoda | Palaemonidae | 1 | 104 | 170 | 159.76 |
| 30 | Scenedesmus dimorphus | Chlorophyta | Scenedesmaceae | 1 | 11.3 | 9.3 | 294.55 |
| 31 | Euphlyctis hexadactylus | Chordata | Ranidae | 1 | 20 | 25.7 | 329.35 |
| 32 | Aplexa hypnorum | Mollusca | Physidae | 2 | 44.7–50.4 | 83–241 | 460.72 |
| 33 | Tanytarsus dissimilis | Arthropoda | Chironomidae | 1 | 48 | 3200 | 10,240.45 |
| 34 | Philodina acuticornis | Aschelminthes | Philodinidae | 1 | 25 | 1400 | 12,597.16 |
| 35 | Gammarus pseudolimnaeus | Arthropoda | Gammaridae | 1 | 48 | 4500 | 14,400.63 |
| Species | N | Hardness (mg/L) | CTV (μg/L) | SACRs |
|---|---|---|---|---|
| D. magna | 8 | 35–180 | 2.6–29 | 0.75 |
| O. mykiss | 3 | 27.5–37 | 0.12–12 | 39.36 |
| Oreochromis niloticus | 1 | 340 | 50 | - |
| P. promelas | 3 | 30.5–206 | 0.53–98 | 4.06 |
| Hardness (mg/L) | a | b | x0 | Adj R2 | HC5 (μg/L) | CMC (μg/L) | CCC (μg/L) |
|---|---|---|---|---|---|---|---|
| 50 | 0.9819 | 1.4004 | 2.3066 | 0.9797 | 0.17 | 0.08 | 0.02 |
| 100 | 0.9819 | 1.4004 | 3.4051 | 0.9797 | 0.50 | 0.25 | 0.05 |
| 200 | 0.9819 | 1.4004 | 4.5036 | 0.9797 | 1.50 | 0.75 | 0.15 |
| 300 | 0.9819 | 1.4004 | 5.1461 | 0.9797 | 2.86 | 1.43 | 0.29 |
| 500 | 0.9819 | 1.4004 | 5.9557 | 0.9797 | 6.42 | 3.21 | 0.65 |
3.3. The Comparison with Other WQC and Water Quality Standards for Silver
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nitayaphat, W.; Jintakosol, T. Removal of silver(I) from aqueous solutions by chitosan/bamboo charcoal composite beads. J. Clean. Prod. 2015, 87, 850–855. [Google Scholar] [CrossRef]
- Akgül, M.; Karabakan, A.; Acar, O.; Yürüm, Y. Removal of silver (I) from aqueous solutions with clinoptilolite. Microporous Mesoporous Mater. 2006, 94, 99–104. [Google Scholar] [CrossRef] [Green Version]
- Cantuaria, M.L.; de Almeida Neto, A.F.; Nascimento, E.S.; Vieira, M.G.A. Adsorption of silver from aqueous solution onto pre-treated bentonite clay: Complete batch system evaluation. J. Clean. Prod. 2016, 112, 1112–1121. [Google Scholar] [CrossRef]
- Ahamed, M.E.H.; Mbianda, X.Y.; Marjanovic, L.; Mulaba-Bafubiandi, A.F. Ion imprinted polymers for the selective extraction of silver(I) ions in aqueous media: Kinetic modeling and isotherm studies. React. Funct. Polym. 2013, 73, 474–483. [Google Scholar] [CrossRef]
- El-Shorbagy, H.G.; El-Kousy, S.M.; Elwakeel, K.Z.; El-Ghaffar, M.A.A. Eco-friendly Chitosan Condensation Adduct Resins for Removal of Toxic Silver Ions from Aqueous Medium. J. Ind. Eng. Chem. 2021, 100, 410–421. [Google Scholar] [CrossRef]
- USEPA. Ambient Water Quality Criteria for Silver; US Environmental Protection Agency: Washington, DC, USA, 1980.
- Hogstrand, C.; Galvez, F.; Wood, C.M. Toxicity, silver accumulation and metallothionein induction in freshwater rainbow trout during exposure to different silver salts. Environ. Toxicol. Chem. 2010, 15, 1102–1108. [Google Scholar] [CrossRef]
- Evans, D.H.; Piermarini, P.M.; Choe, K.P. The multifunctional fish gill: Dominant site of gas exchange, osmoregulation, acid-base regulation, and excretion of nitrogenous waste. Physiol. Rev. 2005, 85, 97–177. [Google Scholar] [CrossRef]
- Xiang, Q.; Gao, Y.; Li, Q.; Ling, J.; Chen, L. Proteomic profiling reveals the differential toxic responses of gills of common carp exposed to nanosilver and silver nitrate. J. Hazard. Mater. 2020, 394, 122562. [Google Scholar] [CrossRef] [PubMed]
- Wood, C.M.; Hogstrand, C.; Galvez, F.; Munger, R.S. The physiology of waterborne silver toxicity in freshwater rainbow trout (Oncorhynchus mykiss) 2. The effects of silver thiosulfate. Aquat. Toxicol. 1996, 35, 111–125. [Google Scholar] [CrossRef]
- Bianchini, A.; Wood, C.M. Mechanism of acute silver toxicity in Daphnia magna. Environ. Toxicol. Chem. 2010, 22, 1361–1367. [Google Scholar] [CrossRef]
- Hogstrand, C.; Wood, C.M. Toward a better understanding of the bioavailability, physiology, and toxicity of silver in fish: Implications for water quality criteria. Environ. Toxicol. Chem. 1998, 17, 547–561. [Google Scholar] [CrossRef]
- Wood, E. Homeostasis and Toxicology of Essential Metals; Academic Press: Cambridge, MA, USA, 2012. [Google Scholar]
- CCME. Canadian Water Quality Guidelines for the Protection of Aquatic Life: Silver; Canadian Council of Ministers of the Environment: Winnipeg, MB, Canada, 2015. [Google Scholar]
- Anzecc, A. Australian and New Zealand Guidelines for Fresh and Marine Water Quality; Australian and New Zealand Environment and Conservation Council and Agriculture and Resource Management Council of Australia and New Zealand: Canberra, Australia, 2000. [Google Scholar]
- Lu, C.; Yang, S.; Yan, Z.; Ling, J.; Jiao, L.; He, H.; Zheng, X.; Jin, W.; Fan, J. Deriving aquatic life criteria for PBDEs in China and comparison of species sensitivity distribution with TBBPA and HBCD. Sci. Total Environ. 2018, 640–641, 1279–1285. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Mu, Y.; Wu, F.; Zhang, R.; Su, H.; Giesy, J.P. Derivation of marine water quality criteria for metals based on a novel QICAR-SSD model. Environ. Sci. Pollut. Res. 2015, 22, 4297–4304. [Google Scholar] [CrossRef]
- Yang, S.; Xu, F.; Wu, F.; Wang, S.; Zheng, B. Development of PFOS and PFOA criteria for the protection of freshwater aquatic life in China. Sci. Total Environ. 2014, 470–471, 677–683. [Google Scholar] [CrossRef]
- Yin, D.; Jin, H.; Yu, L.; Hu, S. Deriving freshwater quality criteria for 2,4-dichlorophenol for protection of aquatic life in China. Environ. Pollut. 2003, 122, 217–222. [Google Scholar] [CrossRef]
- Yan, Z.; Yang, N.; Wang, X.; Wang, W.; Meng, S.; Liu, Z. Preliminary analysis of species sensitivity distribution based on gene expression effect. Sci. China Earth Sci. 2012, 55, 907–913. [Google Scholar] [CrossRef]
- Yang, S.; Yan, Z.; Xu, F.; Wang, S.; Wu, F. Development of freshwater aquatic life criteria for Tetrabromobisphenol A in China. Environ. Pollut. 2012, 169, 59–63. [Google Scholar] [CrossRef]
- Zheng, L.; Liu, Z.; Yan, Z.; Yi, X.; Zhang, J.; Zhang, Y.; Zheng, X.; Zhu, Y. Deriving water quality criteria for trivalent and pentavalent arsenic. Sci. Total Environ. 2017, 587–588, 68–74. [Google Scholar] [CrossRef]
- Lasko, C.L.; Hurst, M.P. An Investigation into the Use of Chitosan for the Removal of Soluble Silver from Industrial Wastewater. Environ. Sci. Technol. 1999, 33, 3622–3626. [Google Scholar] [CrossRef]
- Stephen, C.; Mount, D.; Hansen, D.; Gentile, J.; Chapman, G.; Brungs, W. Guidelines for Deriving Numerical National Water Quality Criteria for the Protection of Aquatic Organisms and Their Uses; US Environmental Protection Agency: Washington, DC, USA, 1985; Volume 45.
- MEEC. Technical Guideline for Deriving Water Quality Criteria for the Protection of Freshwater Aquatic Organisms; HJ 831-2017; Ministry of Ecology and Environment of China: Beijing, China, 2017.
- Yan, Z.; Zheng, X.; Fan, J.; Zhang, Y.; Wang, S.; Zhang, T.; Sun, Q.; Huang, Y. China national water quality criteria for the protection of freshwater life: Ammonia. Chemosphere 2020, 251, 126379. [Google Scholar] [CrossRef]
- MEEC. Freshwater Aquatic Biological Water Quality Criteria-Ammonia Nitrogen; Ministry of Ecology and Environment of China: Beijing, China, 2019.
- MEEC. Freshwater Aquatic Biological Water Quality Criteria-Cadmium; Ministry of Ecology and Environment of China: Beijing, China, 2019.
- MEEC. Freshwater Aquatic Biological Water Quality Criteria-Phenol; Ministry of Ecology and Environment of China: Beijing, China, 2020.
- Chen, Y.; Dai, Q.; Chen, C.; Tai, J.; Pi, Y. Toxicity of silver ions to fish and other aquatic animals. J. Fish. China 1991, 15, 55–61. [Google Scholar]
- Purcell, T.W.; Peters, J.J. Sources of Silver in the Environment. Environ. Toxicol. Chem. 1998, 17, 539–546. [Google Scholar] [CrossRef]
- Van Vlaardingen, P.; Traas, T.; Wintersen, A.; Aldenberg, T. ETX 2.0. A Program to Calculate Hazardous Concentrations and Fraction Affected, Based on Normally Distributed Toxicity Data; RIVM Rapport 601501028; National Institute for Public Health and the Environment (RIVM): Bilthoven, The Netherlands, 2005. [Google Scholar]
- ECB. Technical Guidance Document on Risk Assessment-Partii; Institute for Health and Consumer Protection: Ispra, Italy, 2003. [Google Scholar]
- Ciffroy, P.; Brebbia, C.A. Methods For Calculating PNECs Using Species Sensitivity Distribution (SSD) With Various Hypothesis On The Way To Handle Ecotoxicity Data. Environ. Health Risk 2007, 11, 9. [Google Scholar]
- Venkat, A.; Bielmyer-Fraser, G.K.; Amerson, A. Influence of Water Hardness on Accumulation and Effects of Silver in the Green Alga, Raphidocelis subcapitata. Ga. J. Sci. Off. Publ. Ga. Acad. Sci. 2016, 74, 5. [Google Scholar]
- Chen, Y.; Yu, S.; Tang, S.; Li, Y.; Liu, H.; Zhang, X.; Su, G.; Li, B.; Yu, H.; Giesy, J.P. Site-specific water quality criteria for aquatic ecosystems: A case study of pentachlorophenol for Tai Lake, China. Sci. Total Environ. 2016, 541, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Li, Z.; Gibson, M.; Gao, H. Ecological risk assessment of nonylphenol in coastal waters of China based on species sensitivity distribution model. Chemosphere 2014, 104, 113–119. [Google Scholar] [CrossRef]
- Hernando, M.D.; Mezcua, M.; Fernández-Alba, A.R.; Barceló, D. Environmental risk assessment of pharmaceutical residues in wastewater effluents, surface waters and sediments. Talanta 2006, 69, 334–342. [Google Scholar] [CrossRef]
- Park, T.; Lee, J.; Lee, M.; Park, C.; Lee, C.; Moon, S.; Chung, J.; Cui, R.; An, Y.-J.; Yeom, D.-H.; et al. Development of water quality criteria of ammonia for protecting aquatic life in freshwater using species sensitivity distribution method. Sci. Total Environ. 2018, 634, 934–940. [Google Scholar] [CrossRef]
- Guo, L.; Li, Z.; Gao, P.; Hu, H.; Gibson, M. Ecological risk assessment of bisphenol A in surface waters of China based on both traditional and reproductive endpoints. Chemosphere 2015, 139, 133–137. [Google Scholar] [CrossRef]
- Wen, J.; Cui, X.; Gibson, M.; Li, Z. Water quality criteria derivation and ecological risk assessment for triphenyltin in China. Ecotoxicol. Environ. Saf. 2018, 161, 397–401. [Google Scholar] [CrossRef]
- Elphick, J.R.F.; Bergh, K.D.; Bailey, H.C. Chronic toxicity of chloride to freshwater species: Effects of hardness and implications for water quality guidelines. Environ. Toxicol. Chem. 2011, 30, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Diamond, J.M.; Mackler, D.G.; Collins, M.; Gruber, D. Derivation of a freshwater silver criteria for the New River, Virginia, using representative species. Environ. Toxicol. Chem. 1990, 9, 1425–1434. [Google Scholar] [CrossRef]
- De, P.; De, H.; Posthumus, R.; Crommentuijn, T. Risk limits for boron, silver, titanium, tellurium, uranium and organosilicon compounds in the framework of EU Directive 76/464/EEC. Rijksinst. Voor Volksgezond. En Milieu Rivm 1999, 601, 005. [Google Scholar]
- MOHC. Standards for drinking Water Quality (GB5749-2006); Standards Press of China: Beijing, China, 2006. [Google Scholar]
- MEEC. Discharge Standard of Pollutants for Municipal Wastewater Treatment Plant (GB 18918-2002); Ministry of Ecology and Environment of China: Beijing, China, 2002.
- Ding, T.; Du, S.; Zhang, Y.; Wang, H.; Zhang, Y.; Cao, Y.; Zhang, J.; He, L. Hardness-dependent water quality criteria for cadmium and an ecological risk assessment of the Shaying River Basin, China. Ecotoxicol. Environ. Saf. 2020, 198, 110666. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Sun, F.; Liu, Q.; Zhao, X.; Song, K. Development of regional water quality criteria of lead for protecting aquatic organism in Taihu Lake, China. Ecotoxicol. Environ. Saf. 2021, 222, 112479. [Google Scholar] [CrossRef] [PubMed]
- Clark, N.J.; Boyle, D.; Eynon, B.P.; Handy, R.D. Dietary exposure to silver nitrate compared to two forms of silver nanoparticles in rainbow trout: Bioaccumulation potential with minimal physiological effects. Environ. Sci. Nano 2019, 6, 1393–1405. [Google Scholar] [CrossRef] [Green Version]
- Joo, H.S.; Kalbassi, M.R.; Yu, I.J.; Lee, J.H.; Johari, S.A. Bioaccumulation of silver nanoparticles in rainbow trout (Oncorhynchus mykiss): Influence of concentration and salinity. Aquat. Toxicol. 2013, 140, 398–406. [Google Scholar]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, Q.; Feng, C.; Xia, P.; Bai, Y. Hardness-Dependent Water Quality Criteria for Protection of Freshwater Aquatic Organisms for Silver in China. Int. J. Environ. Res. Public Health 2022, 19, 6067. https://doi.org/10.3390/ijerph19106067
Jin Q, Feng C, Xia P, Bai Y. Hardness-Dependent Water Quality Criteria for Protection of Freshwater Aquatic Organisms for Silver in China. International Journal of Environmental Research and Public Health. 2022; 19(10):6067. https://doi.org/10.3390/ijerph19106067
Chicago/Turabian StyleJin, Qi, Chenglian Feng, Peng Xia, and Yingchen Bai. 2022. "Hardness-Dependent Water Quality Criteria for Protection of Freshwater Aquatic Organisms for Silver in China" International Journal of Environmental Research and Public Health 19, no. 10: 6067. https://doi.org/10.3390/ijerph19106067
APA StyleJin, Q., Feng, C., Xia, P., & Bai, Y. (2022). Hardness-Dependent Water Quality Criteria for Protection of Freshwater Aquatic Organisms for Silver in China. International Journal of Environmental Research and Public Health, 19(10), 6067. https://doi.org/10.3390/ijerph19106067








