Impacts of Pesticides on Oral Cavity Health and Ecosystems: A Review
Abstract
1. Introduction
2. General Characteristics of Pesticides
3. Oral Cavity
3.1. Oral Microbiota Diversities
Oral Microbiota and Immune Response
3.2. Dysbiosis and Associated Oral Pathologies
4. General Effects of Pesticides on the Oral Cavity
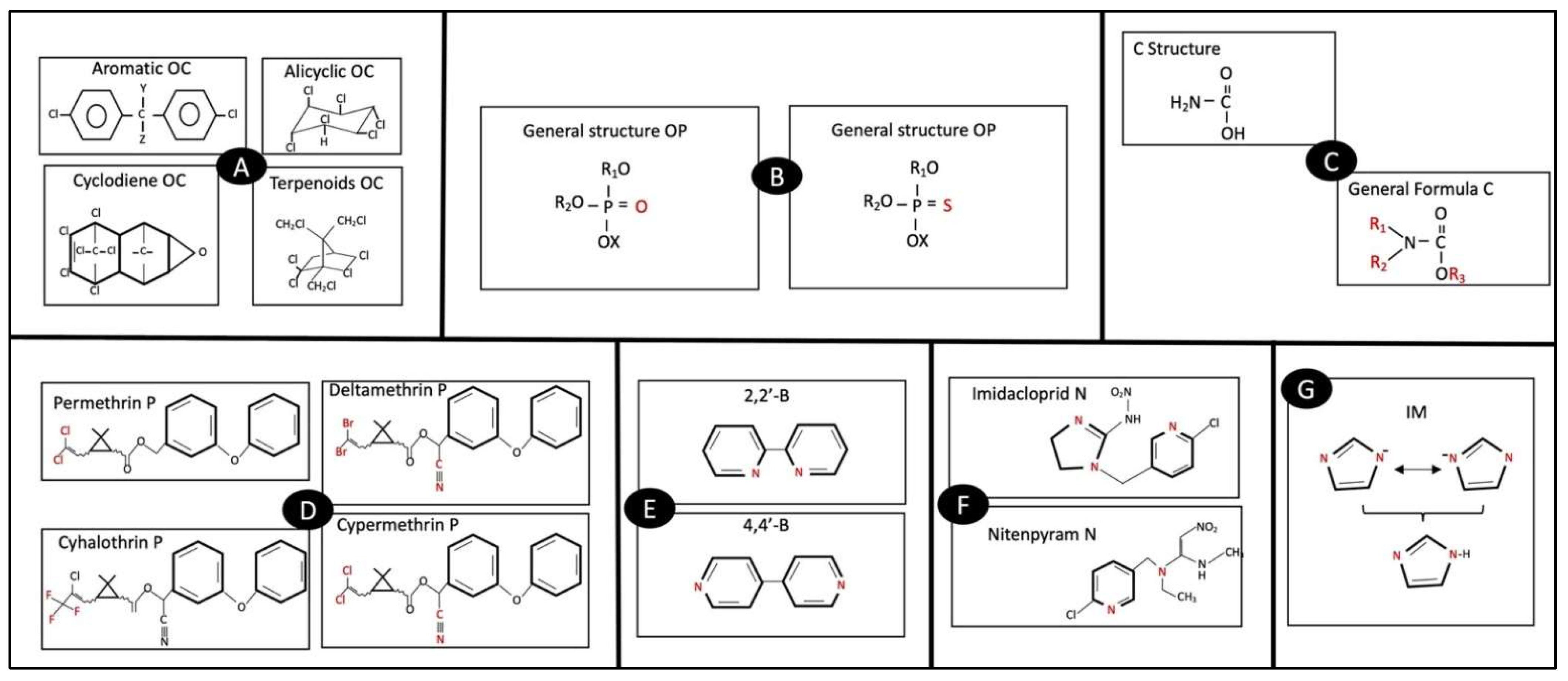
4.1. Effects Based on Type of Pesticide in the Oral Cavity
4.2. Effects of Pesticides on the Oral Microbiome
4.2.1. Effect of OP and B on Oral Microbiome
4.2.2. Analysis of the Effect of Pesticides on Bacteria
4.2.3. Analysis of the Effect of Pesticides on Phyla
5. Scope for Future Research
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Peres, M.A.; Macpherson, L.M.; Weyant, R.J.; Daly, B.; Venturelli, R.; Mathur, M.R.; Listl, S.; Celeste, R.K.; Guarnizo-Herreño, C.C.; Kearns, C.; et al. Oral diseases: A global public health challenge. Lancet 2019, 394, 249–260. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Oral Health. 2022. Available online: https://www.who.int/news-room/fact-sheets/detail/oral-health (accessed on 12 May 2022).
- Petti, S.; Glendor, U.; Andersson, L. World traumatic dental injury prevalence and incidence, a meta-analysis-One billion living people have had traumatic dental injuries. Dent. Traumatol. 2018, 34, 71–86. [Google Scholar] [CrossRef] [PubMed]
- Shah, R. Pesticides and Human Health. In Emerging Contaminants; IntechOpen: London, UK, 2020; pp. 1–22. [Google Scholar]
- Kalyabina, V.P.; Esimbekova, E.N.; Kopylova, K.V.; Kratasyuk, V.A. Pesticides: Formulants, distribution pathways and effects on human health—A review. Toxicol Rep. 2021, 8, 1179–1192. [Google Scholar] [PubMed]
- European Commission. EU Pesticides Database. 2022. Available online: https://ec.europa.eu/food/plant/pesticides/eu-pesticides-database/active-substances/?event=search.as (accessed on 31 May 2022).
- Damalas, C.A.; Eleftherohorinos, I.G. Pesticide Exposure, Safety Issues, and Risk Assessment Indicators. Int. J. Environ. Res. Public Health 2011, 8, 1402–1419. [Google Scholar]
- Fatunsin, O.T.; Oyeyiola, A.O.; Moshood, M.O.; Akanbi, L.M.; Fadahunsi, D.E. Dietary risk assessment of organophosphate and carbamate pesticide residues in commonly eaten food crops. Sci. Afr. 2020, 8, e00442. [Google Scholar] [CrossRef]
- Silva-Madera, R.J.; Salazar-Flores, J.; Peregrina-Lucano, A.A.; Mendoza-Michel, J.; Ceja-Gálvez, H.R.; Rojas-Bravo, D.; Reyna-Villela, M.Z.; Torres-Sánchez, E.D. Pesticide contamination in drinking and surface water in the Cienega, Jalisco, México. Water Air Soil Pollut. 2021, 232, 1–13. [Google Scholar] [CrossRef]
- Tang, W.; Wang, D.; Wang, J.; Wu, Z.; Li, L.; Huang, M.; Xu, S.; Yan, D. Pyrethroid pesticide residues in the global environment: An overview. Chemosphere 2018, 191, 990–1007. [Google Scholar] [CrossRef]
- Klaimala, P.; Khunlert, P.; Chuntib, P.; Pundee, R.; Kallayanatham, N.; Nankongnab, N.; Kongtip, P.; Woskie, S. Pesticide residues on children’s hands, home indoor surfaces, and drinking water among conventional and organic farmers in Thailand. Environ. Monit. Assess. 2022, 194, 1–15. [Google Scholar] [CrossRef]
- Kaushik, P.; Kaushik, G. An assessment of structure and toxicity correlation in organochlorine pesticides. J. Hazard. Mater. 2007, 143, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.; Zettel, V.H.; Ward, N.I. The The organophosphate pesticides. J. Nutr. Environ. Med. 1995, 5, 325–339. [Google Scholar] [CrossRef]
- Gosh, A.K.; Brindisi, M. Organic carbamates in drug design and medicinal chemistry. J. Med. Chem. 2015, 58, 2895–2940. [Google Scholar] [CrossRef]
- Gajendiran, A.; Abraham, J. An overview of pyrethroid insecticides. Front. Biol. 2018, 13, 79–90. [Google Scholar] [CrossRef]
- Brewer, K.J.; Swavey, S. Comprehensive Coordination Chemistry II. Bipyridine. Fundamentals: Ligands, Complexes, Synthesis, Purification, and Structure; Thany, S.H., Lever, A.B.P., Eds.; Springer: New York, NY, USA, 2010; pp. 75–83. [Google Scholar]
- Thany, S.H. Neonicotinoid insecticides. In Insect Nicotinic Acetylcholine Receptors; Springer: New York, NY, USA, 2010; pp. 101–110. [Google Scholar]
- Ali, I.; Lone, M.N.; Aboul-Enein, H.Y. Imidazole’s as potential anticancer agents. MedChemCom 2017, 8, 1742–1773. [Google Scholar] [CrossRef]
- Díaz, O.; Aguilar, D.C.C.R.B. Los pesticidas; clasificación, necesidad de un manejo integrado y alternativas para reducir su consumo indebido: Una revisión. Rev. Cient. Agroecosistemas 2018, 6, 15–30. [Google Scholar]
- Yang, W.; Ni, W.; Jin, L.; Liu, J.; Li, Z.; Wang, L.; Ren, A. Determination of organochlorine pesticides in human umbilical cord and association with orofacial clefts in offspring. Chemosphere 2021, 266, 129188. [Google Scholar] [CrossRef]
- Thammachai, A.; Sapbamrer, R.; Rohitrattana, J.; Tongprasert, S.; Hongsibsong, S.; Wangsan, K. Differences in Knowledge, Awareness, Practice, and Health Symptoms in Farmers Who Applied Organophosphates and Pyrethroids on Farms. Front. Public Health 2022, 10, 80. [Google Scholar] [CrossRef]
- Djekkoun, N.; Lalau, J.D.; Bach, V.; Depeint, F.; Khorsi-Cauet, H. Chronic oral exposure to pesticides and their consequences on metabolic regulation: Role of the microbiota. Eur. J. Nutr. 2021, 60, 4131–4149. [Google Scholar] [CrossRef]
- Jackson, E.; Shoemaker, R.; Larian, N.; Cassis, L. Adipose tissue as a site of toxin accumulation. Compr. Physiol. 2017, 7, 1085. [Google Scholar]
- Barton, H.A.; Pastoor, T.P.; Baetcke, K.; Chambers, J.E.; Diliberto, J.; Doerrer, N.G.; Driver, J.H.; Hastings, C.E.; Iyengar, S.; Krieger, R.; et al. The acquisition and application of absorption, distribution, metabolism, and excretion (ADME) data in agricultural chemical safety assessments. Crit. Rev. Toxicol. 2006, 36, 9–35. [Google Scholar] [CrossRef] [PubMed]
- Belsey, N.A.; Cordery, S.F.; Bunge, A.L.; Guy, R.H. Assessment of dermal exposure to pesticide residues during re-entry. Environ. Sci. Technol. 2011, 45, 4609–4615. [Google Scholar] [CrossRef]
- Parizi, J.L.S.; Tolardo, A.J.; Lisboa, A.C.G.; Barravieira, B.; Mello, F.D.A.; Rossi, R.C.; Nai, G.A. Evaluation of buccal damage associated with acute inhalation exposure to 2,4-dichlorophenoxyacetic acid (2,4-D) in mice. BMC Vet. Res. 2020, 16, 244. [Google Scholar] [CrossRef]
- Ptasiewicz, M.; Grywalska, E.; Mertowska, P.; Korona-Głowniak, I.; Poniewierska-Baran, A.; Niedźwiedzka-Rystwej, P.; Chałas, R. Armed to the Teeth—The Oral Mucosa Immunity System and Microbiota. Int. J. Mol. Sci. 2022, 23, 882. [Google Scholar] [CrossRef]
- Waasdorp, M.; Krom, B.P.; Bikker, F.J.; van Zuijlen, P.P.M.; Niessen, F.B.; Gibbs, S. The bigger picture: Why oral mucosa heals better than skin. Biomolecules 2021, 11, 1165. [Google Scholar] [CrossRef]
- Sepulveda, S.J.; Medina, H.R.M. Cavidad oral. In Histología, Biología Celular y Tisular, 6th ed.; McGraw Hill Interamericana: Mexico City, Mexico, 2014; pp. 452–458. [Google Scholar]
- Şenel, S. An Overview of Physical, Microbiological and Immune Barriers of Oral Mucosa. Int. J. Mol. Sci. 2021, 22, 7821. [Google Scholar] [CrossRef] [PubMed]
- Moutsopoulos, N.M.; Konkel, J.E. Tissue-Specific Immunity at the Oral Mucosal Barrier. Trends Immunol. 2018, 39, 276–287. [Google Scholar] [CrossRef]
- Roblegg, E.; Coughran, A.; Sirjani, D. Saliva: An all-rounder of our body. Eur. J. Pharm. Biopharm. 2019, 142, 133–141. [Google Scholar] [CrossRef]
- Barrios, C.E.; Vila, V.G.; Martinez, S.E.; Tutuy, A.J.E. Ph Salival como factor asociado a la caries dental. Rev. Fac. Odontol. Univ. Nac. 2017, 10, 13–19. [Google Scholar] [CrossRef]
- Chimenos-Küstner, E.; Giovannoni, M.L.; Schemel-Suárez, M. Dysbiosis as a determinant factor of systemic and oral pathology: Importance of micorbiome. Med. Clín. 2017, 149, 305–309. [Google Scholar] [CrossRef]
- Scotti, E.; Boué, S.; Sasso, G.L.; Zanetti, F.; Belcastro, V.; Poussin, C.; Sierro, N.; Battey, J.; Gimalac, A.; Ivanov, N.V.; et al. Exploring the microbiome in health and disease: Implications for toxicology. Toxicol. Res. Appl. 2017, 1, 1–37. [Google Scholar] [CrossRef]
- Jakubovics, N.S.; Goodman, S.D.; Mashburn-Warren, L.; Stafford, G.P.; Cieplik, F. The dental plaque biofilm matrix. Periodontol. 2000 2021, 86, 32–56. [Google Scholar] [CrossRef] [PubMed]
- Dhir, S. Biofilm and dental implant: The microbial link. J. Indian Soc. Periodontol. 2013, 17, 5. [Google Scholar] [CrossRef]
- Barboza-Solís, C.; Acuña-Amador, L.A. The Oral Microbiota: A Literature Review for Updating Professionals in Dentistry. Part I. Odovtos Int. J. Dent. Sci. 2020, 22, 59–68. [Google Scholar] [CrossRef]
- Giambò, F.; Teodoro, M.; Costa, C.; Fenga, C. Toxicology and Microbiota: How Do Pesticides Influence Gut Microbiota? A Review. Int. J. Environ. Res. Public Health 2021, 18, 5510. [Google Scholar] [CrossRef] [PubMed]
- Valm, A.M. The Structure of Dental Plaque Microbial Communities in the Transition from Health to Dental Caries and Periodontal Disease. J. Mol. Biol. 2019, 431, 2957–2969. [Google Scholar] [CrossRef]
- Barboza-Solís, C.; Acuña-Amador, L.A. The oral microbiota: A literature review for updating profesional s in dentistry-Part II. Odovtos Int. J. Dent. Sci. 2021, 23, 45–56. [Google Scholar] [CrossRef]
- Stanaway, I.B.; Wallace, J.C.; Shojaie, A.; Griffith, W.C.; Hong, S.; Wilder, C.S.; Green, F.H.; Tsai, J.; Knight, M.; Workman, T.; et al. Human oral buccal microbiomes are associated with farmworker status and azinphos-methyl agricultural pesticide exposure. Appl. Environ. Microbiol. 2017, 83, 2149–2165. [Google Scholar] [CrossRef]
- Sulijaya, B.; Takahashi, N.; Yamazaki, K.; Yamazaki, K. Nutrition as Adjunct Therapy in Periodontal Disease Management. Curr. Oral. Health Rep. 2019, 6, 61–69. [Google Scholar] [CrossRef]
- Cruz Quintana, S.M.; Díaz Sjostrom, P.; Arias Socarrás, D.; Mazón Baldeón, G.M. Microbiota of oral cavity ecosystems. Rev. Cuba. Estomatol. 2017, 54, 84–99. [Google Scholar]
- Gomaa, E.Z. Human gut microbiota/microbiome in health and diseases: A review. Antonie Van Leeuwenhoek 2020, 113, 2019–2040. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Zhan, J.; Liu, D.; Luo, M.; Han, J.; Liu, X.; Liu, C.; Cheng, Z.; Zhou, Z.; Wang, P. Organophosphorus pesticide chlorpyrifos intake promotes obesity and insulin resistance through impacting gut and gut microbiota. Microbiome 2019, 7, 1–15. [Google Scholar] [CrossRef]
- Jin, Y.; Zeng, Z.; Wu, Y.; Zhang, S.; Fu, Z. Oral Exposure of Mice to Carbendazim Induces Hepatic Lipid Metabolism Disorder and Gut Microbiota Dysbiosis. Toxicol. Sci. 2015, 147, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.; Zeng, Z.; Fu, Z.; Jin, Y. Oral imazalil exposure induces gut microbiota dysbiosis and colonic inflammation in mice. Chemosphere 2016, 160, 349–358. [Google Scholar] [CrossRef]
- Tonetti, M.S.; Greenwell, H.; Kornman, K.S. Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. J. Periodontol. 2018, 89, S159–S172. [Google Scholar] [CrossRef] [PubMed]
- Socransky, S.S.; Haffajee, A.D.; Cugini, M.A.; Smith, C.; Kent, R.L. Microbial complexes in subgingival plaque. J. Clin. Periodontol. 1998, 25, 134–144. [Google Scholar] [CrossRef]
- Lee, D.H.; Jacobs, D.R.; Kocher, T. Associations of serum concentrations of persistent organic pollutants with the prevalence of periodontal disease and subpopulations of white blood cells. Environ. Health Perspect. 2008, 116, 1558–1562. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fernandes, L.C.; Santos, A.G.; Sampaio, T.B.; Sborgi, S.; Prediger, R.; Ferro, M.; Franco, G.; Lipinski, L.; Miyoshi, E. Exposure to paraquat associated with periodontal disease causes motor damage and neurochemical changes in rats. Hum. Exp. Toxicol. 2021, 40, 81–89. [Google Scholar] [CrossRef]
- de Souza, A.; Medeiros, A.D.R.; de Souza, A.C.; Wink, M.; Siqueira, I.R.; Ferreira, M.B.C.; Fernandes, L.; Hidalgo, M.P.L.; da Silva Torres, I.L. Avaliação do impacto da exposição a agrotóxicos sobre a saúde de população rural: Vale do Taquari (RS, Brasil). Ciênc. Saúde Coletiva 2011, 16, 3519–3528. [Google Scholar] [CrossRef] [PubMed]
- Shiue, I. Urinary heavy metals, phthalates, phenols, thiocyanate, parabens, pesticides, polyaromatic hydrocarbons but not arsenic or polyfluorinated compounds are associated with adult oral health: USA NHANES, 2011–2012. Environ. Sci. Pollut. Res. 2015, 22, 15636–15645. [Google Scholar] [CrossRef]
- Condette, C.J.; Bach, V.; Mayeur, C.; Gay-Quéheillard, J.; Khorsi-Cauet, H. Chlorpyrifos Exposure during Perinatal Period Affects Intestinal Microbiota Associated with Delay of Maturation of Digestive Tract in Rats. J. Pediatr. Gastroenterol. Nutr. 2015, 61, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Fang, B.; Li, J.W.; Zhang, M.; Ren, F.Z.; Pang, G.F. Chronic chlorpyrifos exposure elicits diet-specific effects on metabolism and the gut microbiome in rats. Food Chem. Toxicol. 2018, 111, 144–152. [Google Scholar] [CrossRef]
- Birnbaum, S.S.L.; Schulz, N.K.E.; Tate, A.T. Interactions between evolved pesticide resistance and pesticide exposure influence immunity against pathogens. bioRxiv 2022. [Google Scholar] [CrossRef]
- Yi, S.W.; Ohrr, H. Agent Orange exposure and cancer incidence in Korean Vietnam veterans: A prospective cohort study. Cancer 2014, 120, 3699–3706. [Google Scholar] [CrossRef]
- Chi, C.C.; Chou, C.T.; Liang, W.Z.; Jan, C.R. Effect of the pesticide, deltamethrin, on Ca2+ signaling and apoptosis in OC2 human oral cancer cells. Drug Chem. Toxicol. 2013, 37, 25–31. [Google Scholar] [CrossRef]
- Zhang, N.; Wang, B.; Zhang, Z.; Chen, X.; Huang, Y.; Liu, Q.; Zhang, H. Occurrence of neonicotinoid insecticides and their metabolites in tooth samples collected from south China: Associations with periodontitis. Chemosphere 2021, 264, 128498. [Google Scholar] [CrossRef] [PubMed]
- Anusha, J.; Moudgil, K. Accidental paraquat induced hypersalivation: A case report. DARU J. Pharm. Sci. 2019, 27, 885–888. [Google Scholar] [CrossRef]
- Arora, N.; Dhibar, D.P. Quaternion of paraquat poisoning: Icterus, oral ulceration, acute respiratory and renal failure. Postgrad. Med. J. 2022, 98, 150. [Google Scholar] [CrossRef]
- Kumar, S. Paraquat tongue. Indian J. Gastroenterol. 2016, 35, 321. [Google Scholar] [CrossRef] [PubMed]
- Lo, J.; Poon, L.Y.C. Systemic Paraquat Intoxication Presenting with Peripheral Ulcerative Keratitis: A Case Report and Literature Review. Ocul. Immunol. Inflamm. 2020, 28, 871–875. [Google Scholar] [CrossRef]
- Madhan, B.; Arunprasad, G.; Krishnan, B. Paraquat tongue. BMJ Case Rep. 2014, 2014, 206581. [Google Scholar] [CrossRef] [PubMed]
- Tu, P.; Gao, B.; Chi, L.; Lai, Y.; Bian, X.; Ru, H.; Lu, K. Subchronic low-dose 2,4-D exposure changed plasma acylcarnitine levels and induced gut microbiome perturbations in mice. Sci. Rep. 2019, 9, 4363. [Google Scholar] [CrossRef]
- Inserm. Glyphosate and Glyphosate-Based Herbicides. Extract from “Pesticides and Health Effects: New Data.” Collect Expert Report 2021. Available online: https://www.inserm.fr/wp-content/uploads/inserm-expertiscollective-pesticides2021-glyphosate-en.pdf (accessed on 16 May 2022).
- Ackermann, W.; Coenen, M.; Schrödl, W.; Shehata, A.A.; Krüger, M. The influence of glyphosate on the microbiota and production of botulinum neurotoxin during ruminal fermentation. Curr. Microbiol. 2014, 70, 374–382. [Google Scholar] [PubMed]
- Lozano, V.L.; Defarge, N.; Rocque, L.M.; Mesnage, R.; Hennequin, D.; Cassier, R.; de Vendômois, J.S.; Panoff, J.-M.; Séralini, G.-E.; Amiel, C. Sex-dependent impact of Roundup on the rat gut microbiome. Toxicol. Rep. 2018, 5, 96–107. [Google Scholar]
- Cobanoglu, H.; Coskun, M.; Coskun, M.; Çayir, A. Results of buccal micronucleus cytome assay in pesticide-exposed and non-exposed group. Environ. Sci. Pollut. Res. 2019, 26, 19676–19683. [Google Scholar]
- Zhan, J.; Liang, Y.; Liu, D.; Ma, X.; Li, P.; Liu, C.; Liu, X.; Wang, P.; Zhou, Z. Antibiotics may increase triazine herbicide exposure risk via disturbing gut microbiota. Microbiome 2018, 6, 1–13. [Google Scholar]
- Gao, B.; Chi, L.; Tu, P.; Gao, N.; Lu, K. The Carbamate Aldicarb Altered the Gut Microbiome, Metabolome, and Lipidome of C57BL/6J Mice. Chem. Res. Toxicol. 2019, 32, 67–79. [Google Scholar] [PubMed]
- de Bortoli, G.M.; de Azevedo, M.B.; da Silva, L.B. Cytogenetic biomonitoring of Brazilian workers exposed to pesticides: Micronucleus analysis in buccal epithelial cells of soybean growers. Mutat. Res. Toxicol. Environ. Mutagen. 2009, 675, 1–4. [Google Scholar]
- Tarvainen, L.; Kyyrönen, P.; Kauppinen, T.; Pukkala, E. Cancer of the mouth and pharynx, occupation and exposure to chemical agents in Finland [in 1971–95]. Int. J. Cancer 2008, 123, 653–659. [Google Scholar] [PubMed]
- Tirelli, V.; Catone, T.; Turco, L.; Di Consiglio, E.; Testai, E.; De Angelis, I. Effects of the pesticide clorpyrifos on an in vitro model of intestinal barrier. Toxicol. Vitr. 2007, 21, 308–313. [Google Scholar]
- Mao, Q.; Manservisi, F.; Panzacchi, S.; Mandrioli, D.; Menghetti, I.; Vornoli, A.; Bua, L.; Falcioni, L.; Lesseur, C.; Chen, J.; et al. The Ramazzini Institute 13-week pilot study on glyphosate and Roundup administered at human-equivalent dose to Sprague Dawley rats: Effects on the microbiome. Environ. Health 2018, 17, 1–12. [Google Scholar]
- Zhou, M.; Zhao, J. A Review on the Health Effects of Pesticides Based on Host Gut Microbiome and Metabolomics. Front. Mol. Biosci. 2021, 8, 632955. [Google Scholar]

| Pesticide Characteristics | Chemical Characteristics | Organism Characteristics | |||
|---|---|---|---|---|---|
| Chemical composition |
| OC |
| H |
|
| Target organism |
| OP |
| I |
|
| Formulation |
| C |
| FN |
|
| Persistence in the environment |
| P |
| A |
|
| Level of toxicity |
| N |
| R |
|
| Anatomical Site | Microbiome | Pathology | Predominant Bacteria |
|---|---|---|---|
| Tongue | Streptococcus salivarius and S. parasanguinis, Streptococcus mitis, Streptococcus mucilaginosus, Actinomyces spp., Lactobacillus, Neisseria, Fusobacterium, Haemophilus | Caries | Etiological bacteria: Streptococcus mutans Other bacteria:Bifidobacterium, Prevotella, Propionibacterium, Scardovia, Actinomyces, Scardovia wiggsiae (childhood caries), Veillonella, Rothia, Leptotrichia (enamel caries), S. sanguinis, Atopobium, Schlegelella, Pseudoramibacter, Lactobacillus (dentin caries). |
| Gingival sulcus | Acinetobacter, Haemophilus, Moraxella, Streptococcus, Granulicatella, Gemella, Treponema | ||
| Buccal mucosa, keratinized gum and hard palate | Streptococcus mutans, S. viridans, Staphylococcus epidermidis, Kokuria, Micrococcus, Streptococcus mitis, Streptococcus sanguis, Simonsiella, Streptococcus salivarius | ||
| Soft palate | Haemophilus, Corynebacterium, Neisseria, Streptococcus pyogenes, S. viridans | Periodontitis | Red complex:Porphyromonas gingivalis, Tannerella forsythia, Treponema denticola Orange complex:Prevotella intermedia, Campylobacter Yellow complex: S. salivarius Other bacteria present:Staphylococcus aureus, Aggregatibacter actinomycetemcomitans, Filifactor alocis, Peptoanaerobacter stomatis; Firmicutes phylum (Dialister spp., Megasphaera spp., Selenomonas spp.); Desulfobulbus, Synergiste. |
| Tooth surface | Streptococcus, Actinomyces, Corynebacterium, Capnocytophaga, Lautropia, Rothia, Campylobacter, Granulicatella, Kingella, Leptotrichia | ||
| Dental plaque | S. sanguinis, S. gordonii, Fusobacterium nucleatum, Rothia | ||
| Biofilm | Stabilizes with Firmicutes, dominant Actinobacterias | Oral cancer | Capnocytophaga gingivalis, Fusobacterium spp., Streptococcus spp., Peptostreptococcus spp., P. gingivalis, Prevotella spp. |
| Saliva | It has high variability, but predominate Veinonella spp., Actinomyces as well as S. mitis, S. salivarius, Granulicatella adiacens, N. flavescens, R. mucilaginosa, P. melaninogenica |
| Pesticide | Type of Study | Alteration in the Oral Cavity | Mechanism | Reference | |
|---|---|---|---|---|---|
| OC | DDE HCH Oxychlordane Trans-Nonachlor | Cross-sectional study (human) |
|
| [51] |
| Cohort study |
|
| [58] | ||
| 2, 4 D | Murine model |
|
| [26] | |
| P | Deltamethrin | Cell culture |
|
| [59] |
| N | Clothianidin Dinotefuran Acetamiprid 1 MethyL 3 Tetrahydro Furimethyl Urea (UF) | Descriptive study |
|
| [60] |
| B | Paraquat | Case report |
|
| [61] |
| Case report |
|
| [62] | ||
| Case report |
|
| [63] | ||
| Case report and literature review |
|
| [64] | ||
| Case report |
| [65] | |||
| Murine model |
| [52] | |||
| BIOLOGICAL AGENT ANALYSIS | |||||
|---|---|---|---|---|---|
| Bacterium | Basic Description | Type of Study | Exposure Pesticide | Alteration Due to Exposure | Reference |
| Streptococcusspp.(F) | It is associated with eubiosis, although it is found in a high proportion of cancer patients (Gram-positive, anaerobic) | Bovine rumen | Glyphosate (OP) | (−) colonization | [68] |
| Farm workers | Azinphos methyl (OP) | (−) colonization | [35,42] | ||
| Lactobacillusspp.(F) | Lactobacillus spp. is said to be more susceptible to chlorpyrifos than other bacterial species. Its reduction contributes to acidosis. Lactobacillus spp. stimulates globulin production and counteracts infections (Gram-positive) | Review | Glyphosate (OP) | (−) colonization | [67] |
| Murine model/Review | Glyphosate (OP) | (−) colonization | [22,39,55,69] | ||
| Farm workers | Azinphos methyl (OP) | (−) colonization | [42] | ||
| Murine model | Chlorpyrifos (OP) | (−) colonization | [22,55,69] | ||
| Review | Chlorpyrifos (OP) | (−) colonization | [39] | ||
| Review | Imidacloprid (N) | (+) colonization | [39] | ||
| Murine model | Nitenpyram (N) | (−) colonization | [77] | ||
| Murine model | Imazalil (IM) | (−) colonization | [48] | ||
| Granulicatella(F) | In a state of eubiosis, it predominates in saliva, on tooth surfaces, and in the gingival sulcus (Gram-positive) | Murine model | Diazinon (OP) | (−) colonization | [42] |
| Corynebacterium (A) | In eubiosis it is found on the tooth surface and soft palate (Gram-positive) | Review | Glyphosate (OP) | (−) colonization | [39] |
| Prevotella(B) | In eubiosis it is present in the oral cavity, mainly in saliva. In dysbiosis, it is related to caries, periodontitis, and oral cancer (Gram-negative, anaerobic) | Review | Permethrin (P) | (−) colonization in triplicate | [39] |
| Bifidobacterium(A) | In eubiosis is found in the oral cavity, in dysbisosis is related to the development of caries. It is reported that it stimulates the immune response and favors the protection of the mucosal barrier (Gram-positive, anaerobic) | Review | Glyphosate (OP) | Susceptible | [67] |
| Murine model | Chlorpyrifos (OP) | (−) colonization | [22,55] | ||
| Review | Chlorpyrifos (OP) | (−) colonization | [39] | ||
| Murine model | Imazalil (IM) | (−) colonization | [48] | ||
| Porphyromonas(B) | In eubiosis it is found in almost all oral niches. In dysbiosis it occurs in caries, gingivitis, periodontitis, and oral cancer (Gram-negative, anaerobic) | Review | Permethrin (P) | (−) colonization in triplicate | [39] |
| Desulfovibrio(P) | Sulfate-reducing bacteria, converts sulfate to sulfur, toxic to the cell by stimulating the destruction of the oral mucosa. (Gram-negative) | Murine model | Carbendazim (C) | (+) colonization | [47] |
| Murine model/Review | Nitenpyram (N) | (−) colonization | [39,77] | ||
| Murine model | Imazalil (IM) | (+) colonization | [48] | ||
| Fusobacterium(Fu) | Bacteria that are related to damage at the membrane, In dysbiosis, periodontitis is prevalent, increases an inflammatory response in the host. (Gram-negative, anaerobic) | Review | Trichlorfon (OP) | (−) colonization | [39] |
| Diazinon (OP) | (+) colonization | ||||
| Murine model | Imazalil (IM) | (+) colonization | [77] | ||
| PHYLUM ANALYSIS | |||||
| Bacteroidetes | Murine model | Tetrachlorodibenzofuran(TCDF) (OC) | (+) colonization | [22] | |
| Murine model | DDE (OC) | (−) colonization | [22] | ||
| Review | DDE (OC) | (−) colonization | [39,77] | ||
| Review | 2, 4 D (OC) | (+) colonization | [39] | ||
| Murine model | PCP(OP) | (+) colonization | [22] | ||
| Review | Glyphosate (OP) | (+) colonization | [67] | ||
| Murine model | Glyphosate (OP) | (+) colonization | [69] | ||
| Review | Glyphosate (OP) | (−) colonization | [39] | ||
| Murine model | Chlorpyrifos (OP) | (+) colonization | [22,55,69] | ||
| Review | Chlorpyrifos (OP) | (−) colonization | [39] | ||
| Review | Diazinon (OP) | (+) colonization | [39] | ||
| Murine model | Carbendazim (C) | (−) colonization | [22,47] | ||
| Review | Permethrins (P) | (−) colonization in triplicate | [39] | ||
| Review | Imidacloprid (N) | (+) colonization in triplicate | [39] | ||
| Murine model | Imazalil (IM) | (−) colonization | [48] | ||
| Review | Imazalil (IM) | (+) colonization | [77] | ||
| Firmicutes | Murine model | TCDF (OC) | (−) colonization | [22] | |
| Review | DDE (OC) | (+) colonization | [39,77] | ||
| Murine model | PCP (OC) | (−) colonization | [22] | ||
| Murine model | DDT (OC) | (+) colonization | [22] | ||
| Review | Dieldrin (OC) | (−) colonization | [39] | ||
| Murine model | Glyphosate (OP) | (−) colonization | [22,69] | ||
| Review | Glyphosate (OP) | (+) colonization | [39] | ||
| Murine model | Chlorpyrifos (OP) | (−) colonization | [22] | ||
| Murine model | Carbendazim (C) | (+) colonization | [22,47] | ||
| Murine model | Imazalil (IM) | (−) colonization | [48] | ||
| Proteobacteria | Review | DDE (OC) | (−) colonization | [39] | |
| Review | DDE (OC) | (+) colonization | [77] | ||
| Murine model | DDT (OC) | (−) colonization | [22] | ||
| Review | Glyphosate (OP) | (−) colonization | [39] | ||
| Review | Chlorpyrifos (OP) | (+) colonization | [39] | ||
| Murine model | Carbendazim (C) | (+) colonization | [22,47] | ||
| Review | Imidacloprid (N) | (−) colonization | [39] | ||
| Review | Nitenpyram (N) | (−) colonization | [39] | ||
| Actinobacteria | Review | DDE (OC) | (−) colonization | [77] | |
| Review | Glyphosate (OP) | (−) colonization | [39] | ||
| Review | Chlorpyrifos (OP) | (+) colonization | [39] | ||
| Murine model | Carbendazim (C) | (+) colonization | [22,47] | ||
| Murine model | Imazalil (IM) | (−) colonization | [48] | ||
| Verrucomicrobia | Review | DDE (OC) | (−) colonization | [77] | |
| Murine model | Carbendazim (C) | (−) colonization | [22] | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salazar-Flores, J.; Lomelí-Martínez, S.M.; Ceja-Gálvez, H.R.; Torres-Jasso, J.H.; Torres-Reyes, L.A.; Torres-Sánchez, E.D. Impacts of Pesticides on Oral Cavity Health and Ecosystems: A Review. Int. J. Environ. Res. Public Health 2022, 19, 11257. https://doi.org/10.3390/ijerph191811257
Salazar-Flores J, Lomelí-Martínez SM, Ceja-Gálvez HR, Torres-Jasso JH, Torres-Reyes LA, Torres-Sánchez ED. Impacts of Pesticides on Oral Cavity Health and Ecosystems: A Review. International Journal of Environmental Research and Public Health. 2022; 19(18):11257. https://doi.org/10.3390/ijerph191811257
Chicago/Turabian StyleSalazar-Flores, Joel, Sarah M. Lomelí-Martínez, Hazael R. Ceja-Gálvez, Juan H. Torres-Jasso, Luis A. Torres-Reyes, and Erandis D. Torres-Sánchez. 2022. "Impacts of Pesticides on Oral Cavity Health and Ecosystems: A Review" International Journal of Environmental Research and Public Health 19, no. 18: 11257. https://doi.org/10.3390/ijerph191811257
APA StyleSalazar-Flores, J., Lomelí-Martínez, S. M., Ceja-Gálvez, H. R., Torres-Jasso, J. H., Torres-Reyes, L. A., & Torres-Sánchez, E. D. (2022). Impacts of Pesticides on Oral Cavity Health and Ecosystems: A Review. International Journal of Environmental Research and Public Health, 19(18), 11257. https://doi.org/10.3390/ijerph191811257







