Haemophilia in France: Modelisation of the Clinical Pathway for Patients
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Setting
- −
- Hospices Civils de Lyon (HCL), Groupement Hospitalier Est, is a coordinator site from the HRC and the French network on inherited bleeding disorders (MHEMO) with 3000 consultations per year.
- −
- Assistance Publique des Hôpitaux de Paris (APHP)—Hôpital Bicêtre is a reference centre for haemophilia and other constitutional coagulation protein deficiencies. Nearly 600 patients with haemophilia are followed in this reference centre for rare haemophilia diseases.
- −
- The Lille University Hospital—Lille Regional Haemophilia Treatment Centre (CTL) is one of the five French sites labelled national reference centre for haemophilia and other rare coagulation deficiencies. This centre is composed of several functional units: (i) biological haemostasis; (ii) cardiological haemorrhagic pathology; (iii) haemorrhagic pathology Jeanne de Flandre; (iv) thrombotic pathology; (v) Willebrand UF, vi/Transfusion Safety & Hemovigilance; (vi) immuno-haematology. It should be noted that there is no doctor assigned to each patient, which favours the versatility of medical professionals. It is the doctor on duty who manages, with shared advice.
- −
- The Metropole Savoy Hospital Centre in Chambery (CHC, France) is a CRC-MHC. It should be noted that it is relatively rare to have a haemophilia service in a general hospital, as is the case here. The geography (mountains) partly explains this situation. The number of populations increases by a factor of 100 during the winter vacations (skiing).
2.3. Data Collection
- −
- Interview with the head of the department or his/her representative, who was able to explain the context and history of the organization.
- −
- Visits to the departments concerned: 30 min–1 h.
- −
- Interviews with the different actors of the pathway (collective or individual): doctor, nurse, paediatrician, haematologist, biologist, physiotherapist, pharmacist, etc.: 30 and 45 min.
- −
- Work observations (free time involved) (secretarial/consultation/team meeting, patients, others): observations 1 h or more.
- −
- Debriefing with the Head of Department: 15 min or more depending on availability.
2.4. Network Validation
2.5. Outcomes
2.6. Sample Size
2.7. Analysis
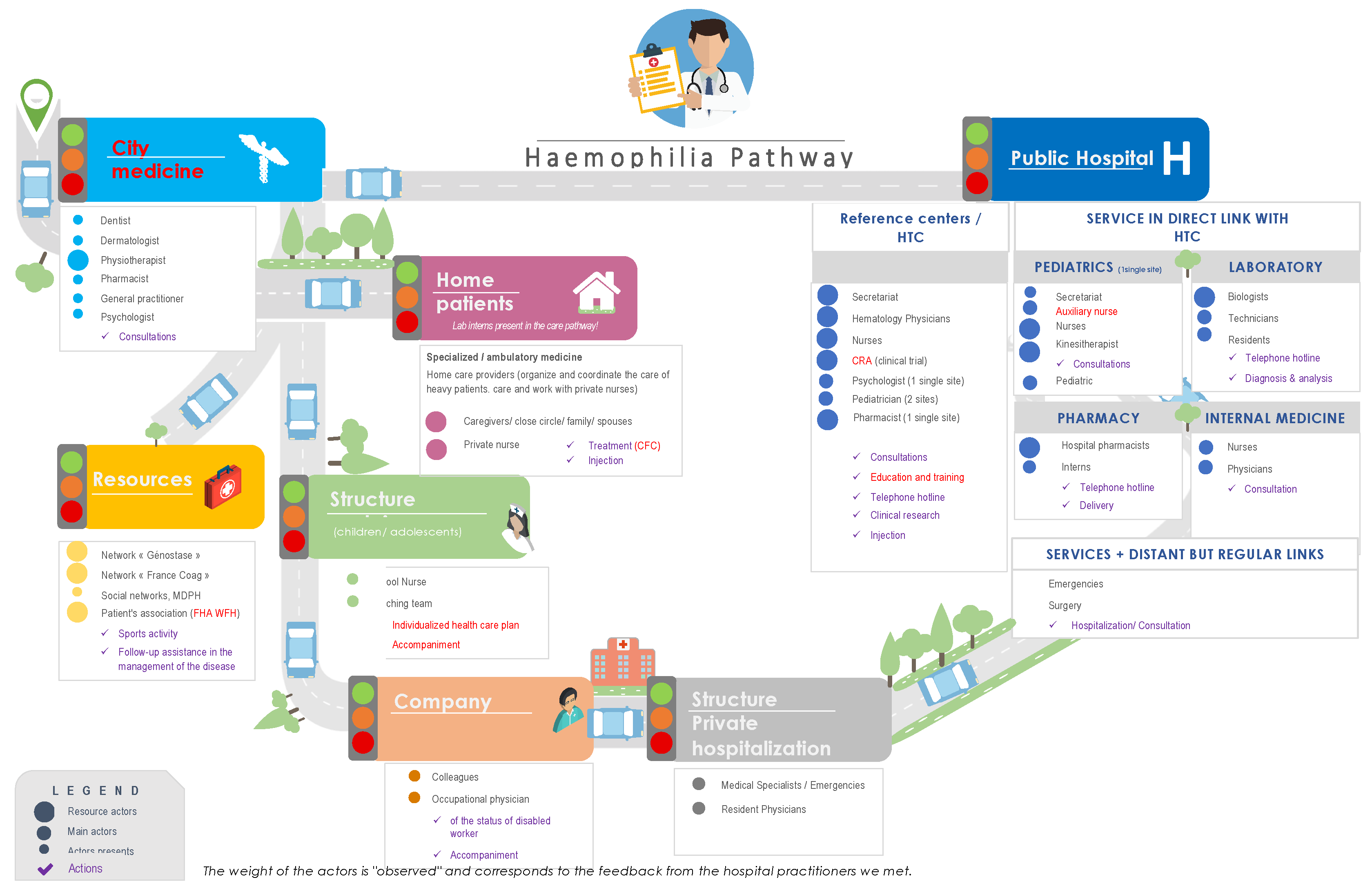
3. Results
- −
- Nurses: mainly for treatment injections for patients who do not practice self-injection, or who do not have a caregiver for this practice.
- −
- Physiotherapists: although their role is recognized by caregivers in haemophilia services for managing the consequences of hemarthrosis, they are not very present in the institutions.
- −
- General practitioner is not very present; his or her interactions with the specialized service are very rare.
- −
- Patient’s close family and friends are particularly concerned by the disease, and even involved in its management.
3.1. Several Specific Elements Have Emerged
- −
- The estrangement in the relationship between patients and health care teams due to the development of prophylaxis with long-acting treatments.
- −
- Questions about the risk of patients escaping if new treatments are dispensed in pharmacies.
- −
- The haemophilia service sometimes has difficulty getting along with certain other services, particularly emergency rooms, where patients are occasionally poorly recognized and misdirected.
- −
- Poor recognition of women with haemophilia: not only “carriers” but also “affected”: 1/3 of them have factor 8 deficiency. Additionally, 90% of these women are not believed in the emergency room.
- −
- A loss of awareness of the disease on the part of patients, due to the lengthening of the time between treatments or easier administration methods, leading to risky behaviours (such as non-adapted physical activity, non-management of joint pain, etc.).
- −
- The importance of therapeutic education of the patient, and of his entourage, to maintain an awareness of the disease and a good management of it.
- −
- The importance of broad-based support for haemophilia patients, including both physiotherapists and psychologists.
- −
- The link between hospital nurses and private nurses is almost non-existent.
- −
- The link between city doctors and hospital doctors is almost non-existent.
- −
- Adherence to medication was defined as satisfactory in children, average in adolescents, and poorer in adults.
3.2. Obstacles and Barriers
3.2.1. Within the Specialized Service
3.2.2. With Other Hospital Services
3.2.3. With External Hospital or Private Services
3.2.4. With Community Health Care Providers
3.2.5. Between the Stakeholders in General Practice
3.2.6. Links between Patients with Haemophilia and Existing Support Structures
3.2.7. School Care
3.2.8. Professional Structures
3.2.9. The Immediate Environment—Family
3.2.10. The Impact of Therapeutic Innovations
4. Discussion
- −
- The importance of the hospital in the current course with the predominance of its actors, who may differ from one site to another, and the close links occasionally difficult to establish with the patient’s life outside the hospital. This is a constant observed in industrialized countries. Procedures, rules for collaboration between EHTCs and EHCCCs must be written, shared and made available to the public [31]. While the implementation of the haemophilia treatment centre model is dependent on the availability of a relevant range of medical and paramedical services, states and territories depending on resources, patient population demographics, centralization and organization of services, most people with haemophilia receive care through specialized centres [32]. For example, in Australia, these centres were established in 1998 to play a leading role within their hospital, city and outlying areas to ensure optimal care and equitable distribution of professional and therapeutic resources [33].
- −
- Links outside the hospital of various categories. The links are occasionally not strong enough to be maintained in the long term, reinforcing the importance and the necessary autonomy of the patient in the management of their illness. The links are occasionally non-existent or created on the basis of opportunities or historical configurations and often dependent persons. Certainly, the World Federation of Haemophilia recommends that patients with haemophilia be managed in a specialized comprehensive care centre [34]. However, sharing best practices with other healthcare professionals external to hospitals allows for better patient-centred approaches, and improved adherence and health outcomes [35]. Today, individuals with haemophilia can look forward to a virtually normal life expectancy and quality of life [25]. In France, 131 national reference centres for rare diseases must organize partnership in the delivery of care and the coordination of the health and social care network. Patient education, information and training of professionals, patients and their families are also among their missions. This raises the question of the extent to which the care of specific diseases or highly specialized interventions are restricted to a limited number of specific centres or health care providers. The treatment of haemophilia is evolving and changed the role of external stakeholders [36]. As a result of progress in disease management, healthcare professionals who are not haemophilia specialists are more likely to encounter people with haemophilia [37]. Nurses, for example, are the first point of contact for people with haemophilia and provide more care to patients and their families than any other specialist on the team [38,39]. They are to place greater emphasis on psychosocial support, therapeutic education, shared decision making, and patient support and advocacy [40].
- −
- Intra-hospital interactions linked to opportunities, specific historical organizations and collaborative choices that are not always possible (limited flexibility) in terms of time, human or financial resources, availability, etc. The mission of French clinical networks of reference centres is to structure and coordinate the activities between the national and regional centres, the technical platforms, or any other structure involved in care for patients. EHTC must have access to multidisciplinary support, locally or in conjunction with EHCCCs as orthopaedics and physiotherapy, dental care, surgery, obstetrics and gynaecology, hepatology, infectious diseases, paediatric, clinical psychology and social worker [17,19,32].
- −
- There is considerable variability despite a willingness to describe a “meta-course”, a “standardized” description that makes it possible to visualize all of the participants who revolve around the patient and the places where patients with haemophilia go. Through daily self-management, patients with haemophilia become experts and partners in their own care. As such, they should be considered as separate essential members of the comprehensive care team [11]. However, main risks of disruption in this process target parameters such as patient typology, geographical accessibility, characteristics of the patient’s disease, non-medical environment and therefore the patient’s environment. The necessary search for balance between standardization of the care action and adaptation to the specificity of each situation, and the precious pursuit of “standardized singularity”: the personalization of standardized steps will contribute to the reduction in health inequalities [31].
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ferraris, V.A.; Boral, L.I.; Cohen, A.J.; Smyth, S.S.; White, G.C. Consensus Review of the Treatment of Cardiovascular Disease in People with Hemophilia A and B. Cardiol. Rev. 2015, 23, 53–68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iorio, A.; Stonebraker, J.S.; Chambost, H.; Makris, M.; Coffin, D.; Herr, C.; Germini, F. Data and Demographics Committee of the World Federation of Hemophilia Establishing the Prevalence and Prevalence at Birth of Hemophilia in Males: A Meta-Analytic Approach Using National Registries. Ann. Intern. Med. 2019, 171, 540–546. [Google Scholar] [CrossRef] [PubMed]
- Stonebraker, J.S.; Bolton-Maggs, P.H.B.; Brooker, M.; Evatt, B.; Iorio, A.; Makris, M.; O’Mahony, B.; Skinner, M.W.; Coffin, D.; Pierce, G.F.; et al. The World Federation of Hemophilia Annual Global Survey 1999–2018. Haemophilia 2020, 26, 591–600. [Google Scholar] [CrossRef] [PubMed]
- Mühlbacher, A.C.; Sadler, A.; Lamprecht, B.; Juhnke, C. Patient Preferences in the Treatment of Hemophilia A: A Latent Class Analysis. PLoS ONE 2021, 16, e0256521. [Google Scholar] [CrossRef]
- Cavazza, M.; Kodra, Y.; Armeni, P.; De Santis, M.; López-Bastida, J.; Linertová, R.; Oliva-Moreno, J.; Serrano-Aguilar, P.; Posada-de-la-Paz, M.; Taruscio, D.; et al. Social/Economic Costs and Quality of Life in Patients with Haemophilia in Europe. Eur. J. Health Econ. 2016, 17 (Suppl. S1), 53–65. [Google Scholar] [CrossRef]
- Randuineau, P.; Stieltjes, N.; Perut, V.; Paubel, P.; Lopez, I. Cost of clotting factors in hospitalization. Ann. Pharm. Fr. 2018, 76, 361–367. [Google Scholar] [CrossRef]
- Laurendeau, C.; Goudemand, J.; Trossaert, M.; Polack, B.; Varin, R.; Godard, C.; Hadim, F.; Detournay, B. Costs and Management of Patients with Hemophilia A in France: The Hemraude Study. Eur. J. Health Econ. 2021. [Google Scholar] [CrossRef]
- Yeung, C.H.T.; Santesso, N.; Pai, M.; Kessler, C.; Key, N.S.; Makris, M.; Navarro-Ruan, T.; Soucie, J.M.; Schünemann, H.J.; Iorio, A. Care Models in the Management of Haemophilia: A Systematic Review. Haemophilia 2016, 22 (Suppl. S3), 31–40. [Google Scholar] [CrossRef] [Green Version]
- Srivastava, A.; Santagostino, E.; Dougall, A.; Kitchen, S.; Sutherland, M.; Pipe, S.W.; Carcao, M.; Mahlangu, J.; Ragni, M.V.; Windyga, J.; et al. WFH Guidelines for the Management of Hemophilia, 3rd Edition. Haemophilia 2020, 26 (Suppl. S6), 1–158. [Google Scholar] [CrossRef]
- Van der Ham, A.; van Merode, F.; Ruwaard, D.; van Raak, A. Identifying Integration and Differentiation in a Hospital’s Logistical System: A Social Network Analysis of a Case Study. BMC Health Serv. Res. 2020, 20, 857. [Google Scholar] [CrossRef]
- Ansteatt, K.T.; Unzicker, C.J.; Hurn, M.L.; Olaiya, O.O.; Nugent, D.J.; Tarantino, M.D. The Need for Comprehensive Care for Persons with Chronic Immune Thrombocytopenic Purpura. J. Blood Med. 2020, 11, 457–463. [Google Scholar] [CrossRef]
- Burke, T.; Asghar, S.; O’Hara, J.; Sawyer, E.K.; Li, N. Clinical, Humanistic, and Economic Burden of Severe Hemophilia B in the United States: Results from the CHESS US and CHESS US+ Population Surveys. Orphanet J. Rare Dis. 2021, 16, 143. [Google Scholar] [CrossRef]
- Rocks, S.; Berntson, D.; Gil-Salmerón, A.; Kadu, M.; Ehrenberg, N.; Stein, V.; Tsiachristas, A. Cost and Effects of Integrated Care: A Systematic Literature Review and Meta-Analysis. Eur. J. Health Econ. 2020, 21, 1211–1221. [Google Scholar] [CrossRef]
- De Moerloose, P.; Fischer, K.; Lambert, T.; Windyga, J.; Batorova, A.; Lavigne-Lissalde, G.; Rocino, A.; Astermark, J.; Hermans, C. Recommendations for Assessment, Monitoring and Follow-up of Patients with Haemophilia. Haemophilia 2012, 18, 319–325. [Google Scholar] [CrossRef]
- Page, D. Comprehensive Care for Hemophilia and Other Inherited Bleeding Disorders. Transfus. Apher. Sci. 2019, 58, 565–568. [Google Scholar] [CrossRef]
- Héon-Klin, V. European Reference Networks for Rare Diseases: What Is the Conceptual Framework? Orphanet J. Rare Dis. 2017, 12, 137. [Google Scholar] [CrossRef] [Green Version]
- Colvin, B.T.; Astermark, J.; Fischer, K.; Gringeri, A.; Lassila, R.; Schramm, W.; Thomas, A.; Ingerslev, J. Inter Disciplinary Working Group European Principles of Haemophilia Care. Haemophilia 2008, 14, 361–374. [Google Scholar] [CrossRef]
- Fischer, K.; Hermans, C. European Haemophilia Therapy Standardisation Board The European Principles of Haemophilia Care: A Pilot Investigation of Adherence to the Principles in Europe. Haemophilia 2013, 19, 35–43. [Google Scholar] [CrossRef]
- Hoos, A.; Anderson, J.; Boutin, M.; Dewulf, L.; Geissler, J.; Johnston, G.; Joos, A.; Metcalf, M.; Regnante, J.; Sargeant, I.; et al. Partnering With Patients in the Development and Lifecycle of Medicines: A Call for Action. Ther. Innov. Regul. Sci. 2015, 49, 929–939. [Google Scholar] [CrossRef]
- Noone, D.; O’Mahony, B.; Peyvandi, F.; Makris, M.; Bok, A. Evolution of Haemophilia Care in Europe: 10 Years of the Principles of Care. Orphanet J. Rare Dis. 2020, 15, 184. [Google Scholar] [CrossRef]
- O’Mahony, B.; Noone, D.; Giangrande, P.L.F.; Prihodova, L. Haemophilia Care in Europe: A Survey of 19 Countries. Haemophilia 2011, 17, 35–40. [Google Scholar] [CrossRef] [Green Version]
- Schrijvers, G.; van Hoorn, A.; Huiskes, N. The Care Pathway: Concepts and Theories: An Introduction. Int. J. Integr. Care 2012, 12, e192. [Google Scholar] [CrossRef]
- Chaudoir, S.R.; Dugan, A.G.; Barr, C.H.I. Measuring Factors Affecting Implementation of Health Innovations: A Systematic Review of Structural, Organizational, Provider, Patient, and Innovation Level Measures. Implement Sci. 2013, 8, 22. [Google Scholar] [CrossRef] [Green Version]
- Flottorp, S.A.; Oxman, A.D.; Krause, J.; Musila, N.R.; Wensing, M.; Godycki-Cwirko, M.; Baker, R.; Eccles, M.P. A Checklist for Identifying Determinants of Practice: A Systematic Review and Synthesis of Frameworks and Taxonomies of Factors That Prevent or Enable Improvements in Healthcare Professional Practice. Implement. Sci. 2013, 8, 35. [Google Scholar] [CrossRef] [Green Version]
- Berntorp, E.; Fischer, K.; Hart, D.P.; Mancuso, M.E.; Stephensen, D.; Shapiro, A.D.; Blanchette, V. Haemophilia. Nat. Rev. Dis. Primers 2021, 7, 45. [Google Scholar] [CrossRef]
- Calvez, T.; Biou, M.; Costagliola, D.; Jullien, A.M.; Laurian, Y.; Rossi, F.; Rothschild, C.; Sié, P. Suivi Thérapeutique National des Hémophiles Group The French Haemophilia Cohort: Rationale and Organization of a Long-Term National Pharmacosurveillance System. Haemophilia 2001, 7, 82–88. [Google Scholar] [CrossRef]
- Calvez, T.; Chambost, H.; d’Oiron, R.; Dalibard, V.; Demiguel, V.; Doncarli, A.; Gruel, Y.; Huguenin, Y.; Lutz, P.; Rothschild, C.; et al. Analyses of the FranceCoag Cohort Support Differences in Immunogenicity among One Plasma-Derived and Two Recombinant Factor VIII Brands in Boys with Severe Hemophilia A. Haematologica 2018, 103, 179–189. [Google Scholar] [CrossRef] [Green Version]
- Chambost, H.; Gaboulaud, V.; Coatmélec, B.; Rafowicz, A.; Schneider, P.; Calvez, T. Suivi thérapeutique National des Hémophiles (SNH) Group What Factors Influence the Age at Diagnosis of Hemophilia? Results of the French Hemophilia Cohort. J. Pediatr. 2002, 141, 548–552. [Google Scholar] [CrossRef]
- Doncarli, A.; Doncarli, A.; Demiguel, V.; Ghez, M.; Doussin, A.; Gautier, P.; Faradji, A.; Lambert, T.; Peynet, J.; Donadieu, J.; et al. Premier État Des Lieux Du Suivi de La Population Hémophile En France (Cohorte FranceCoag), 1994–2005. Bull. Epidemiol. Hebd. 2006, 39, 291–294. [Google Scholar]
- Doncarli, A.; Demiguel, V.; Guseva Canu, I.; Goulet, V.; Bayart, S.; Calvez, T.; Castet, S.; Dalibard, V.; Demay, Y.; Frotscher, B.; et al. FranceCoag: A 22-Year Prospective Follow-up of the National French Cohort of Patients with Inherited Bleeding Disorders. Eur. J. Epidemiol. 2019, 34, 521–532. [Google Scholar] [CrossRef]
- Giangrande, P.; Calizzani, G.; Menichini, I.; Candura, F.; Mannucci, P.M.; Makris, M. The European Standards of Haemophilia Centres. Blood Transfus 2014, 12 (Suppl. S3), s525–s530. [Google Scholar] [CrossRef] [PubMed]
- Parker, C.S.; Tsai, J.; Siddiqi, A.-A.; Atrash, H.K.; Richardson, L.C. Meeting the Emerging Public Health Needs of Persons with Blood Disorders. Am. J. Prev. Med. 2014, 47, 658–663. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- National Blood Authority. Guidelines for the Management of Haemophilia in Australia. Available online: https://www.blood.gov.au/haemophilia-guidelines (accessed on 2 November 2021).
- Srivastava, A.; Brewer, A.K.; Mauser-Bunschoten, E.P.; Key, N.S.; Kitchen, S.; Llinas, A.; Ludlam, C.A.; Mahlangu, J.N.; Mulder, K.; Poon, M.C.; et al. Guidelines for the Management of Hemophilia. Haemophilia 2013, 19, e1–e47. [Google Scholar] [CrossRef] [PubMed]
- Van Balen, E.C.; Krawczyk, M.; Gue, D.; Jackson, S.; Gouw, S.C.; van der Bom, J.G.; Sawatzky, R. Patient-Centred Care in Haemophilia: Patient Perspectives on Visualization and Participation in Decision-Making. Haemophilia 2019, 25, 938–945. [Google Scholar] [CrossRef]
- Konkle, B.A.; Skinner, M.; Iorio, A. Hemophilia Trials in the Twenty-First Century: Defining Patient Important Outcomes. Res. Pract. Thromb. Haemost. 2019, 3, 184–192. [Google Scholar] [CrossRef] [Green Version]
- Potì, S.; Palareti, L.; Cassis, F.R.; Brondi, S. Health Care Professionals Dealing with Hemophilia: Insights from the International Qualitative Study of the HERO Initiative. J. Multidiscip. Healthc. 2019, 12, 361–375. [Google Scholar] [CrossRef] [Green Version]
- Pollard, D.; Harrison, C.; Dodgson, S.; Holland, M.; Khair, K. The UK Haemophilia Specialist Nurse: Competencies Fit for Practice in the 21st Century. Haemophilia 2020, 26, 622–630. [Google Scholar] [CrossRef] [Green Version]
- Contributor, N.T. The Changing Role of the Specialist Haemophilia Nurse. Nurs. Times 2021, 117, 50–52. [Google Scholar]
- Khair, K.; Pollard, D.; Harrison, C.; Hook, S.; O’Driscoll, M.; Holland, M. HOw Patients View Extended Half-Life Products: Impressions from Real-World Experience (The HOPE Study). Haemophilia 2019, 25, 814–820. [Google Scholar] [CrossRef]
- Hemophar—Hemophar. Available online: https://hemophar.fr (accessed on 2 November 2021).
- Weyand, A.C.; James, P.D. Sexism in the Management of Bleeding Disorders. Res. Pract. Thromb. Haemost. 2021, 5, 51–54. [Google Scholar] [CrossRef]
- Van Galen, K.P.M.; d’Oiron, R.; James, P.; Abdul-Kadir, R.; Kouides, P.A.; Kulkarni, R.; Mahlangu, J.N.; Othman, M.; Peyvandi, F.; Rotellini, D.; et al. A New Hemophilia Carrier Nomenclature to Define Hemophilia in Women and Girls: Communication from the SSC of the ISTH. J. Thromb. Haemost. 2021, 19, 1883–1887. [Google Scholar] [CrossRef]
- Benson, G.; Auerswald, G.; Dolan, G.; Duffy, A.; Hermans, C.; Ljung, R.; Morfini, M.; Šalek, S.Z. Diagnosis and Care of Patients with Mild Haemophilia: Practical Recommendations for Clinical Management. Blood Transfus. 2018, 16, 535–544. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beny, K.; du Sartz de Vigneulles, B.; Carrouel, F.; Bourgeois, D.; Gay, V.; Negrier, C.; Dussart, C. Haemophilia in France: Modelisation of the Clinical Pathway for Patients. Int. J. Environ. Res. Public Health 2022, 19, 646. https://doi.org/10.3390/ijerph19020646
Beny K, du Sartz de Vigneulles B, Carrouel F, Bourgeois D, Gay V, Negrier C, Dussart C. Haemophilia in France: Modelisation of the Clinical Pathway for Patients. International Journal of Environmental Research and Public Health. 2022; 19(2):646. https://doi.org/10.3390/ijerph19020646
Chicago/Turabian StyleBeny, Karen, Benjamin du Sartz de Vigneulles, Florence Carrouel, Denis Bourgeois, Valérie Gay, Claude Negrier, and Claude Dussart. 2022. "Haemophilia in France: Modelisation of the Clinical Pathway for Patients" International Journal of Environmental Research and Public Health 19, no. 2: 646. https://doi.org/10.3390/ijerph19020646





