Use of Prescribed Drugs to Treat Chronic Diseases during Pregnancy in Outpatient Care in Switzerland between 2014 and 2018: Descriptive Analysis of Swiss Health Care Claims Data
Abstract
:1. Introduction
2. Methods
2.1. Data Source
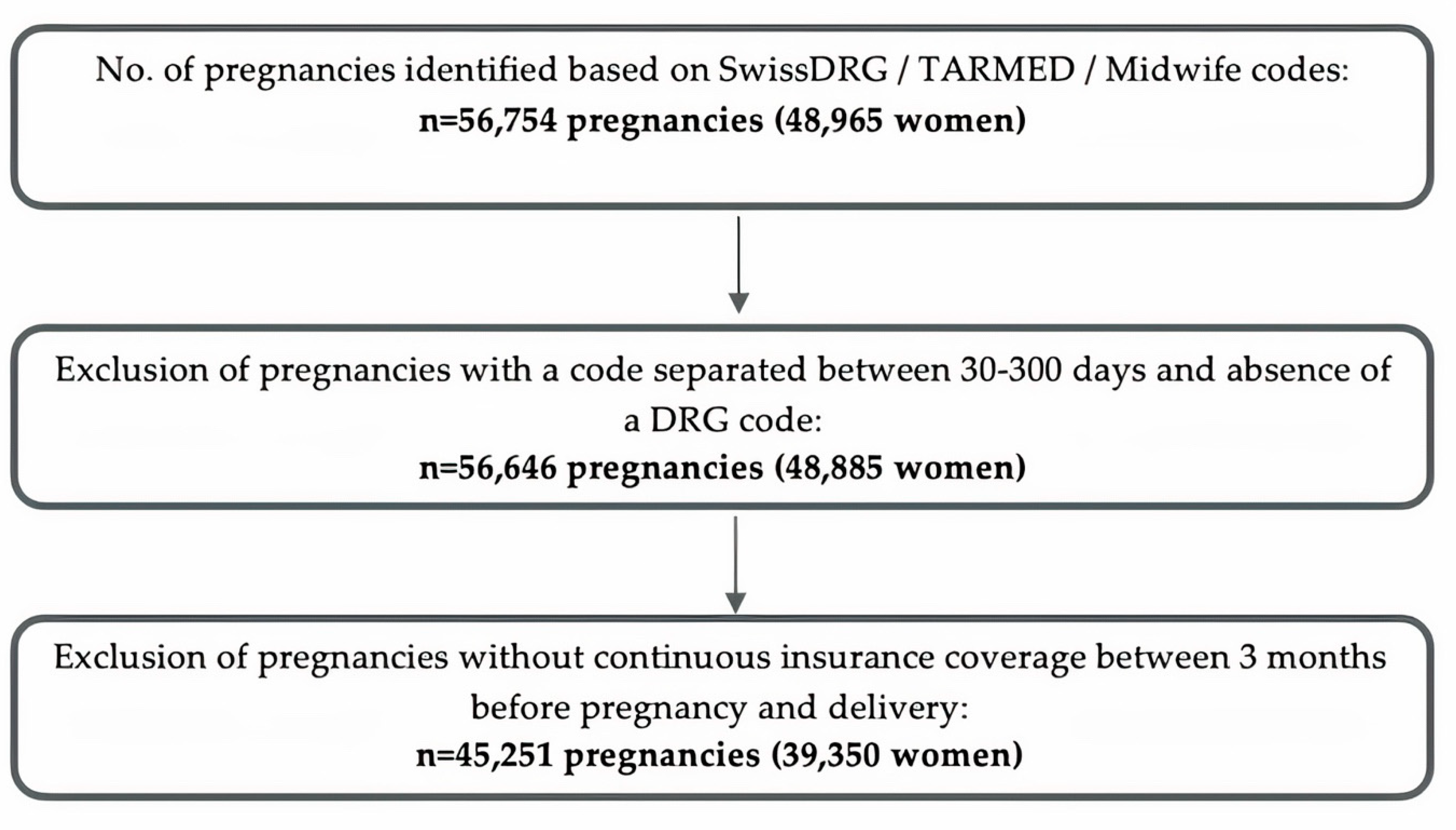
2.2. Pregnancy Cohort
2.2.1. Identification of Pregnancies and Delivery Date
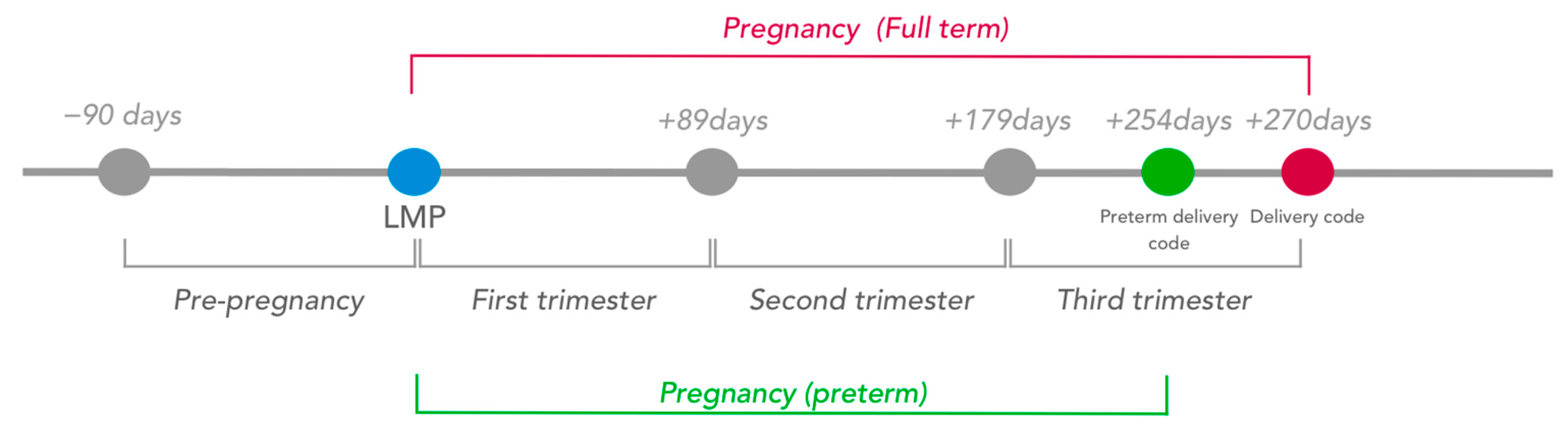
2.2.2. Estimation of the Date of the Last Menstrual Period and Pregnancy Trimesters
2.3. Demographics and Characteristics
Exposure to Drugs
2.4. Statistical Analyses
2.5. Protocol Approvals
3. Results
| Year | N of Weighted Deliveries in Our Study Population | Weighted Age at Delivery in the Cohort (Weighted sd) | Age at Delivery in Switzerland (BfS) | Weighted Percentage of Caesarean Section in the Cohort (%, 95% CI) | Percentage of Caesarean Section in Switzerland (BfS) |
|---|---|---|---|---|---|
| 2014 | 71,933 | 31.96 (5.04) | 31.7 | 34.4 (33.4, 35.5) | 33.7 |
| 2015 | 71,844 | 31.97 (5.15) | 31.8 | 34.3 (33.3, 35.4) | 33.3 |
| 2016 | 74,149 | 31.93 (5.11) | 31.8 | 33.4 (32.4, 34.5) | 33.2 |
| 2017 | 79,610 | 32.06 (5.14) | 31.9 | 33.4 (32.4, 34.6) | 32.3 |
| 2018 | 71,836 | 32.15 (5.00) | 32.0 | 33.1 (32.0, 34.2) | 32.1 |
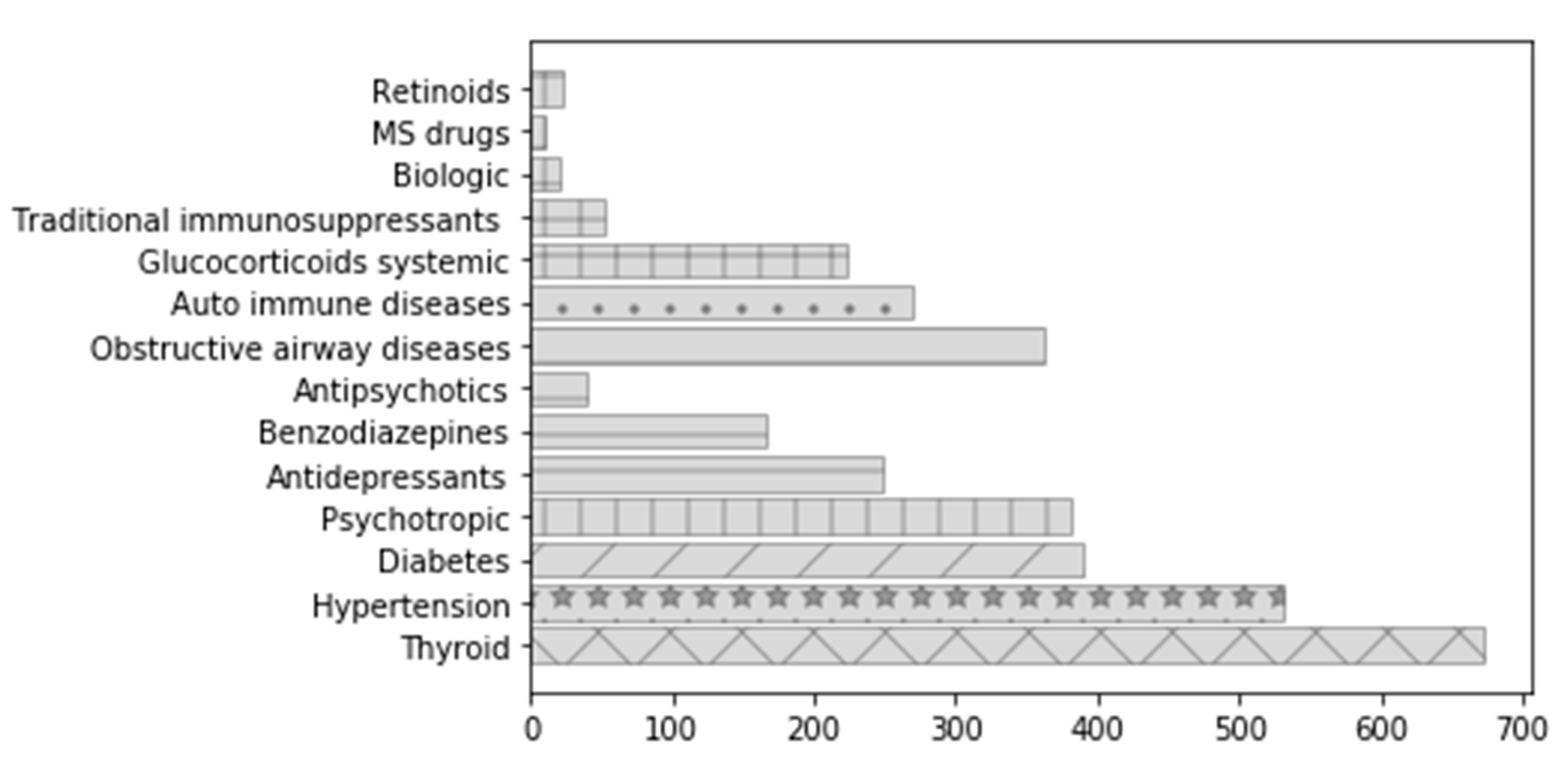
3.1. Prevalence of Drug Exposure
| ATC Code | Drug Substance | Pre-Pregnancy N (/10,000) | 95% CI | T1 N (/10,000) | 95% CI | T2 N (/10,000) | 95% CI | T3 N (/10,000) | 95% CI | T1–T3 N (/10,000) | 95% CI |
|---|---|---|---|---|---|---|---|---|---|---|---|
| At least one drug from any of the following drug groups | 43,179 (1169.0) | 1136.0–1202.0 | 39,978 (1008.6) | 1050.4–1114.3 | 41,601 (1126.3) | 1093.8–1158.7 | 57,980 (1569.7) | 1531.5–1607.9 | 81,715 (2212.3) | 2168.4–2256.1 | |
| H03 | Thyroid therapy | 10,581 (286.5) | 270.1–302.9 | 17,570 (475.7) | 454.3–497.0 | 19,111 (517.4) | 495.3–539.5 | 17,677 (478.6) | 457.3–499.8 | 24,874 (673.4) | 648.3–698.5 |
| H03A | Thyroid preparations | 10,223 (276.8) | 260.6–292.9 | 17,206 (465.8) | 444.7–486.9 | 18,834 (509.9) | 488–531.8 | 17,470 (473) | 451.8–494.1 | 24,418 (661.1) | 636.2–686 |
| H03AA01 | Levothyroxine sodium | 10,087 (273.1) | 257.1–289.1 | 17,081 (462.4)) | 441.4–483.4 | 18,690(506) | 484.2–527.8 | 17,365 (470.1) | 449–491.2 | 24,228 (655.9) | 631.2–680.7 |
| H03AA02 | Liothyronine sodium | 0.0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 |
| H03B | Antithyroid preparations | 388 (10.5) | 7.4–13.6 | 378 (10.2) | 7.2–13.3 | 284 (7.7) | 5–10.4 | 207 (5.6) | 3.4–7.8 | 491 (13.3) | 9.7–16.9 |
| H03BA02 | Propylthiouracil | 174 (4.7) | 2.6–6.8 | 285 (7.7) | 5–10.5 | 137 (3.7) | 1.8–5.6 | 88 (2.4) | 0.9–3.8 | 328 (8.9) | 5.9–11.8 |
| H03BB01 | Carbimazole | 255 (6.9) | 4.3–9.5 | 174 (4.7) | 2.7–6.7 | 164 (4.4) | 2.4–6.5 | 126 (3.4) | 1.7–5.1 | 311 (8.4) | 5.6–11.2 |
| C02, C03, C07, C08, C09 | Anti-hypertensive drugs | 2612 (70.7) | 62.2–79.2 | 2290 (62) | 53.9–70.1 | 4716 (127.7) | 116.3–139 | 17,177 (465) | 443.5–486.6 | 19,594 (530.5) | 507.6–553.3 |
| C08 | Calcium channel blockers | 279 (7.6) | 4.7–10.4 | 405 (11) | 7.9–14.1 | 2917 (79) | 70.2–87.7 | 14,585 (394.9) | 375.2–414.5 | 15,778 (427.2) | 406.8–447.5 |
| C08CA05 | Nifedipine | 157 (4.3) | 2.1–6.4 | 289 (7.8) | 5.2–10.5 | 2827 (76.5) | 67.9–85.1 | 14,423 (390.5) | 371–410 | 15,531 (420.5) | 400.3–440.7 |
| C08CA01 | Amlodipine | 94 (2.5) | 0.8–4.3 | 106 (2.9) | 1.2–4.5 | 85 (2.3) | 0.7–3.9 | 154 (4.2) | 2–6.3 | 233 (6.3) | 3.7–8.9 |
| C07 | Beta-blocking agents | 1651 (44.7) | 37.9–51.5 | 1847 (50) | 42.6–57.4 | 1860 (50.4) | 43–57.8 | 2878 (77.9) | 68.6–87.3 | 4193 (113.5) | 102.4–124.6 |
| C07AG01 | Labetalol | 225 (6.1) | 3.7–8.5 | 670 (18.1) | 13.3–23 | 987 (26.7) | 20.8–32.6 | 2019 (54.7) | 46.5–62.8 | 2484 (67.2) | 58.2–76.3 |
| C07AB02 | Metoprolol | 555 (15) | 11.2–18.9 | 640 (17.3) | 13.3–21.3 | 520 (14.1) | 10.7–17.5 | 521 (14.1) | 10.6–17.6 | 986 (26.7) | 21.8–31.6 |
| C09 | RAA acting agents | 610 (16.5) | 12.3–20.7 | 227 (6.1) | 3.7–8.6 | 105 (2.8) | 1.1–4.5 | 72 (1.9) | 0.6–3.2 | 341 (9.2) | 6.3–12.2 |
| C09CA06 | Candesartan | 59 (1.6) | 0.4–2.8 | 54 (1.5) | 0.3–2.6 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 54 (1.5) | 0.3–2.6 |
| C09AA03 | Lisinopril | 87 (2.4) | 0.9–3.8 | 46 (1.2) | 0.0–2.5 | 30 (0.8) | 0.0–1.8 | 13 (0.4) | 0.0–0.9 | 58 (1.6) | 0.2–2.9 |
| C03 | Diuretics | 277 (7.5) | 4.7–10.3 | 105 (2.8) | 1.1–4.6 | 59 (1.6) | 0.4–2.8 | 126 (3.4) | 1.7–5.1 | 271 (7.3) | 4.6–10 |
| C03CA01 | Furosemide | 77 (2.1) | 0.7–3.5 | 18 (0.5) | 0.0–1.0 | 27 (0.7) | 0.0–1.6 | 95 (2.6) | 1.0–4.1 | 139 (3.8) | 1.9–5.6 |
| C03CA04 | Torasemide | 94 (2.5) | 0.6–4.4 | 58 (1.6) | 0.1–3.1 | 32 (0.9) | 0.0–1.8 | 31 (0.8) | 0.1–1.6 | 115 (3.1) | 1.2–5.0 |
| C02 | Antihypertensives | 58 (1.6)) | 0.3–2.9 | 17 (0.5) | 0.0–1.4 | 17 (0.5) | 0.0–1.4 | 24 (0.6) | 0.0–1.6 | 24 (0.6) | 0.0–1.6 |
| C02AB01 | Methyldopa | 0 (0.0) | 0.0–0.0 | 17 (0.5) | 0.0–1.4 | 17 (0.5) | 0.0–1.4 | 25 (0.6) | 0.0–1.6 | 24 (0.6) | 0.0–1.6 |
| C02CA04 | Doxazosin | 0 (0.0) | 0.0–0.0 | 17 (0.5) | 0.0–1.4 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 17 (0.5) | 0.0–1.4 |
| N06A, N05A, N05AN, N05BA, N05CD, N05CF | Psychotropic drugs | 15,794 (427.6) | 407.2–448 | 9222 (249.7) | 234.1–265.2 | 6468 (175.1) | 162.2–188 | 6236 (168.8) | 156.3–181.4 | 14,084 (381.3) | 362.2–400.4 |
| N06A | Antidepressants | 9442 (255.6) | 239.9–271.3 | 6488 (175.6) | 162.7–188.6 | 4572 (123.8) | 112.9–134.6 | 3940 (106.7) | 96.9–116.4 | 9150 (247.7) | 232.4–263 |
| N06AB | Selective serotonin reuptake inhibitors | 5632 (152.5) | 140.4–164.6 | 4143 (112.2) | 101.7–122.6 | 3263 (88.3) | 79.1–97.6 | 2831 (76.6) | 68.3–85 | 5952 (161.1) | 148.6–173.7 |
| N06AB10 | Escitalopram | 2313 (62.6) | 54.9–70.3 | 1422 (38.5) | 32.4–44.6 | 972 (26.3) | 21.5–31.1 | 912 (24.7) | 20–29.4 | 2054 (55.6) | 48.4–62.8 |
| N06AB06 | Sertraline | 1324 (35.8) | 30.0–41.7 | 1093 (29.6) | 24–35.1 | 1148 (31.1) | 25.5–36.7 | 1075 (29.1) | 23.9–34.3 | 1817 (49.2) | 42.2–56.2 |
| N06AX | Other antidepressants | 3871 (104.8) | 94.8–114.8 | 2353 (63.7) | 56.1–71.3 | 1344 (36.4) | 30.7–42 | 1092 (29.6) | 24.5–34.6 | 3360 (91) | 81.9–100.1 |
| N06AX25 | Hyperici herba | 935 (104.8) | 20.2–30.4 | 609 (16.5) | 12.6–20.4 | 519 (14.1) | 10.5–17.6 | 487 (13.2) | 9.7–16.6 | 1291 (35) | 29.3–40.6 |
| N06AX16, N06AX21 | Serotonin and noradrenaline reuptake inhibitors (duloxetine. venlafaxine) | 1155 (31.3) | 26–36.6 | 835 (22.6) | 18.2–27 | 433 (11.7) | 8.6–14.9 | 329 (8.9) | 6.1–11.7 | 946 (25.6) | 20.8–30.4 |
| N06AX11, N06AX03 | Alpha antagonists (mirtazapine. mianserine) | 584 (15.8) | 12.0–14.3 | 370 (10.0) | 7.0–13.0 | 203 (5.5) | 3.2–7.8 | 122 (3.3) | 1.7–4.9 | 508 (13.8) | 10.2–17.3 |
| N06AA | Non-selective monoamine reuptake inhibitors | 574 (15.5) | 11.6–19.4 | 396 (10.7) | 7.4–14.1 | 138 (3.7) | 1.8–5.6 | 137 (3.7) | 1.9–5.5 | 506 (13.7) | 10–17.4 |
| N06AA06 | Trimipramine | 321 (8.7) | 5.7–11.7 | 203 (5.5) | 3.1–7.9 | 53 (1.4) | 0.1–2.8 | 45 (1.2) | 0.0–2.4 | 238 (6.4) | 3.8–9.0 |
| N06AA09 | Amitriptyline | 163 (4.4) | 2.3–6.5 | 113 (3.1) | 1.4–4.7 | 78 (2.1) | 0.9–3.4 | 92 (2.5) | 1.1–3.9 | 181 (4.9) | 2.8–7.0 |
| N05A | Antipsychotics | 1881 (50.9) | 43.9–58.0 | 1099 (29.8) | 24.3–35.2 | 699 (18.9) | 14.8–23.1 | 761 (20.6) | 16.2–24.9 | 1424 (38.6) | 32.4–44.8 |
| N05AH04 | Quetiapine | 749 (20.3) | 15.9–24.6 | 506 (13.7) | 10.2–17.2 | 433 (11.7) | 8.5–8.5 | 456 (12.3) | 9.0–15.6 | 650 (17.6) | 13.6–21.6 |
| N05AX12 | Aripiprazole | 267 (7.2)) | 4.5–10 | 173 (4.7) | 2.5–6.9 | 108 (2.9) | 1.3–4.6 | 125 (3.4) | 1.6–5.2 | 251 (6.8) | 4.1–9.5 |
| N05AH03 | Olanzapine | 177 (4.8) | 2.5–7.1 | 139 (3.8) | 1.5–6.0 | 49 (1.3) | 0.1–2.5 | 87 (2.4) | 0.9–3.8 | 190 (5.1) | 2.6–7.7 |
| N05AX08 | Risperidone | 103 (2.8) | 1.2–4.4 | 47 (1.3) | 0.3–2.2 | 21 (0.6) | 0.0–1.2 | 18 (0.5) | 0.0–1.2 | 79 (2.1) | 0.8–3.4 |
| N05AD01 | Haloperidol | 70 (1.9) | 0.6–3.2 | 62 (1.7) | 0.1–3.3 | 28 (0.8) | 0.0–1.6 | 23 (0.6) | 0.01.5 | 64 (1.7) | 0.9–4.7 |
| N05AD08 | Droperidol | 390 (10.6) | 7.3–13.8 | 35 (0.9) | 0.1–1.8 | 12 (0.3) | 0.0–0.8 | 6 (0.2) | 0.0–0.5 | 53 (1.4) | 0.4–2.5 |
| N05AF03 | Chlorprothixene | 81 (2.2) | 0.8–3.6 | 64 (1.7) | 0.4–3.1 | 29 (0.8) | 0.0–1.7 | 23 (0.6) | 0.0–1.5 | 104 (2.8) | 0.4–3.1 |
| N05AN01 | Lithium | 17 (0.5) | 0.0–1.1 | 20 (0.5) | 0.0–1.1 | 10 (0.3) | 0.0–0.8 | 10 (0.3) | 0.0–0.8 | 29 (0.8) | 0.0–1.6 |
| N05BA, N05CD, N05CF | Benzodiazepine derivatives/related drugs | 7509 (203.3) | 188.9–217.6 | 3492 (94.5) | 84.7–104.4 | 1926 (52.1) | 45–59.2 | 2180 (59) | 51.3–66.8 | 6141 (166.3) | 153.3–179.2 |
| N05BA, N05CD | Benzodiazepine derivatives | 6260 (169.5) | 156.3–182.7 | 2656 (71.9) | 63.4–80.4 | 1642 (44.5) | 37.9–51 | 1749 (47.4) | 40.4–54.3 | 4958 (134.2) | 122.6–145.8 |
| N05BA06 | Lorazepam | 2788 (75.5) | 66.7–84.3 | 1390 (37.6) | 31.4–43.9 | 853 (23.1) | 18.3–27.9 | 1034 (28) | 22.6–33.4 | 2813 (76.2) | 67.3–85.0 |
| N05CD08 | Midazolam | 1963 (53.1) | 45.6–60.7 | 360 (9.7) | 6.8–12.7 | 68 (1.8) | 0.8–3.0 | 113 (3.1) | 1.2–4.9 | 514 (13.9) | 10.3–17.6 |
| N05CF | Benzodiazepine related drugs | 1606 (43.5) | 37–50 | 989 (26.8) | 21.4–32.1 | 337 (9.1) | 6.1–12.0 | 517 (14) | 10.3–17.7 | 1486 (40.2) | 33.7–46.7 |
| N05CF02 | Zolpidem | 1425 (38.6) | 32.5–44.7 | 908 (24.6)) | 19.5–29.6 | 333 (9) | 6.1–12.0 | 511 (13.8) | 10.1–17.5 | 1486 (40.2) | 31.6–44 |
| N05CF01 | Zopiclone | 200 (5.4) | 2.9–7.9 | 87 (2.4) | 0.5–4.2 | 4 (0.1) | 0.0–0.3 | 6 (0.2) | 0.0–0.5 | 97 (2.6) | 0.7–4.5 |
| A10 | Anti-diabetics | 3592 (97.2) | 87.6–106.9 | 3227 (87.4) | 78–96.8 | 4540 (122.9) | 112–133.9 | 12,659 (342.7) | 324.8–360.7 | 14,398 (389.8) | 370.7–408.9 |
| A10A | Insulins and analogues | 1050 (28.4) | 23.2–33.7 | 1598 (43.3) | 36.7–49.8 | 4060 (109.9) | 99.7–120.2 | 12,258 (331.9) | 314.3–349.5 | 12,752 (345.2) | 327.3–363.2 |
| A10AE | Long acting | 498 (13.5) | 9.8–17.2 | 866 (23.4) | 18.5–28.4 | 2434 (65.9) | 58.2–73.6 | 7328 (198.4) | 185.1–211.7 | 7903 (214) | 200.1227.8 |
| A10AB | Fast acting | 922 (25) | 20.1–29.8 | 1281 (34.7) | 28.9–28.9 | 2104 (57) | 49.5–40.4 | 5812 (157.3) | 145.1–169.6 | 6085 (164.7) | 152.2–177.3 |
| A10AC | Intermediate acting | 75 (2) | 0.6–3.5 | 203 (5.5) | 3.2–7.8 | 1027 (27.8) | 22.3–33.3 | 3688 (99.8) | 89.9–109.8 | 3978 (107.7) | 97.3–118.1 |
| A10B | Blood glucose-lowering drugs | 2650 (71.7) | 63.5–80 | 1802 (48.8) | 41.7–55.9 | 595 (16.1) | 12–20.3 | 608 (16.5) | 12.1–20.8 | 2243 (60.7) | 52.8–68.6 |
| A10BA02 | Metformin | 2483 (67.2) | 59.2–75.2 | 1726 (46.7) | 39.8–53.7 | 569 (15.4) | 11.3–19.5 | 541 (14.6) | 10.5–18.8 | 2086 (56.5) | 48.9–64.1 |
| A10BJ02 | Liraglutide | 63 (1.7) | 0.5–2.9 | 32 (0.9) | 0.1–1.6 | 6 (0.2) | 0.0–0.5 | 18 (0.5) | 0.0–1.2 | 56 (1.5) | 0.4–2.6 |
| A10BB01 | Glibenclamide | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 10 (0.3) | 0.0–0.8 | 36 () | 0.0–1.9 | 46 (1.2) | 0.2–2.4 |
| R03 | Obstructive airway diseases | 7332 (198.5) | 184.6–212.3 | 5775 (156.3) | 143.9–168.7 | 6345 (171.8) | 158.9–184.7 | 6077 (164.5) | 151.9–177.2 | 13,414 (363.2) | 344.4381.9 |
| R03AK | Adrenergics with glucocorticoids or other drugs (excl anticholinergics) | 5115 (138.5) | 126.9–150.1 | 3566 (96.5) | 86.7–106.4 | 3950 (106.9) | 96.7–117.1 | 3572 (96.7) | 86.8–106.6 | 7925 (214.6) | 200.1–229 |
| R03AK07 | Formoterol and budesonide | 3396 (91.9) | 82.7–101.2 | 2219 (60.1) | 52.5–67.6 | 2618 (70.9) | 62.8–78.9 | 2242 (60.7) | 53.1–68.3 | 5337 (144.5) | 132.8–156.1 |
| R03AK06 | Salmeterol and fluticasone | 1496 (40.5) | 33.9–47.1 | 1158 (31.4) | 25.5–37.2 | 1249 (33.8) | 27.7–39.9 | 1199 (32.5) | 26.4–38.5 | 2429 (65.8) | 57.3–74.2 |
| R03AC | Selective B2 adrenoreceptor agonists | 2714 (73.5) | 64.9–82.1 | 2499 (67.7) | 59.4–75.9 | 2636 (71.4) | 63.1–79.6 | 2271 (61.5) | 53.8–69.2 | 5947 (161) | 148.3–173.7 |
| R03AC02 | Salbutamol | 2362 (63.9) | 55.8–72.1 | 2132 (57.7) | 50.1–65.4 | 2297 (62.2) | 54.4–70 | 2046 (55.4) | 48–62.8 | 5289 (143.2) | 131.2–155.2 |
| R03AC13 | Formoterol | 182 (4.9) | 3–6.9 | 244 (6.6) | 4.1–9.2 | 139 (3.8) | 2.1–5.5 | 123 (3.3) | 1.7–5.0 | 348 (9.4) | 6.4–12.5 |
| R03BA | Inhaled Glucocorticoids | 596 (16.1) | 12.3–19.9 | 758 (20.5) | 16.1–24.9 | 899 (24.3) | 19.7–29.0 | 758 (20.5) | 16.2–24.8 | 2011 (54.4) | 47.3–61.6 |
| R03BA02 | Budesonide | 227 (6.1) | 3.8–8.4 | 410 (11.1) | 7.8–14.4 | 479 (13) | 9.7–16.3 | 410 (11.1) | 8.0–14.2 | 1156 (31.3) | 26.0–36.6 |
| R03BA05 | Fluticasone | 210 (5.7) | 3.3–8.1 | 191(5.2) | 2.9–7.4 | 269 (7.3) | 4.7–9.8 | 189 (5.1) | 3.1–7.1 | 514 (13.9) | 10.3–17.6 |
| R03CC | Selective beta-2 adrenoreceptor agonists | 20 (0.5) | 0.0–1.2 | 12 (0.3) | 0.0–0.8 | 6 (0.2) | 0.0–0.5 | 412 (11.2) | 8.2–14.1 | 424 (11.5) | 8.4–14.5 |
| R03CC05 | Hexoprenaline | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 395 (10.7) | 7.7–13.6 | 395 (10.7) | 7.7 |
| R03CC02 | Salbutamol | 20 (0.5) | 0.0–1.2 | 12 (0.3) | 0.0–0.8 | 6 (0.2) | 0.0–0.5 | 17 (0.5) | 0.0–1.0 | 29 (0.8) | 0.1–1.5 |
| R03BB | Anticholinergics | 198 (5.4) | 2.9–7.8 | 171 (4.6) | 2.5–6.8 | 165 (4.5) | 2.1–6.8 | 146 (4) | 2.0–5.9 | 394 (10.7) | 7.4–14.0 |
| R03BB01 | Ipratropium bromide | 144 (3.9) | 1.8–6.0 | 136 (3.7) | 1.8–5.5 | 129 (3.5) | 1.5–5.5 | 127 (3.4) | 1.6–5.3 | 341 (9.2) | 6.2–12.3 |
| R03BB04 | Tiotropium bromide | 27 (0.7) | 0.0–1.7 | 24 (0.6) | 0.0–1.4 | 13 (0.4) | 0.0–1.1 | 19 (0.5) | 0.0–1.3 | 43 (1.2) | 0.1–2.2 |
| R03AL | Adrenergics in combination with anticholinergics | 120 (3.2) | 1.7–4.8 | 94 (2.5) | 1.0–4.1 | 108 (2.9) | 1.2–4.6 | 141 (3.8) | 2.1–5.5 | 305 (8.3) | 5.5–10.9 |
| R03AL02 | Salbutamol and ipratropium bromide | 114 (3.1) | 1.5–4.7 | 89 (2.4) | 0.9–3.9 | 73 (2) | 0.7–3.2 | 124 (3.4) | 1.8–4.9 | 259 (7) | 4.6–9.4 |
| R03DC (R03DC03) | Leukotriene receptor antagonists (Montelukast) | 339 (9.2) | 6.2–12.2 | 214 (5.8) | 3.5–8.1 | 105 (2.8) | 1.1–4.5 | 51 (1.4) | 0.2–2.5 | 278 (7.5) | 4.8–10.2 |
| R03DX | Other systemic drugs for obstructive airway diseases | 99 (2.7) | 1.0–4.3 | 69 (1.9) | 0.4–3.3 | 13 (0.4) | 0.0–1.0 | 27 (0.7) | 0.0–1.7 | 69 (1.9) | 0.4–3.3 |
| R03DX05 | Omalizumab | 80 (2.2) | 0.7–3.6 | 49 (1.3) | 0.1–2.5 | 13 (0.4) | 0.0–1.0 | 27 (0.7) | 0.0–1.7 | 49 (1.3) | 0.1–2.5 |
| R03DX09 | Mepolizumab | 19 (0.5) | 0.0–1.3 | 19 (0.5) | 0.0–1.3 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 19 (0.5) | 0.0–1.3 |
| H02AB, A07EC, P01BA02, L03, L04AD, AX, AA, J06BA | Drugs to treat auto-immune diseases | 8560 (231.7) | 216.9–246.6 | 5104 (138.2) | 126.8–149.6 | 3414 (92.4) | 83.1–101.7 | 4593 (124.3) | 113.3–135.4 | 9947 (269.3) | 253.2–285–4 |
| H02AB | Systemic glucocorticoids | 6831 (184.9) | 171.7–198.2 | 3673 (99.4) | 89.7–109.2 | 2309 (62.5) | 54.9–70.1 | 3375 (91.4) | 81.9–100.9 | 8237 (223) | 208.3–237.7 |
| L04AD, AX, AA, A07EC, P01BA02, L01XC02 | Traditional immunosuppressants | 1647 (44.6) | 38.1–51.0 | 1466 (39.7) | 33.6–45.8 | 1295 (35.1) | 29.3–40.8 | 1338 (36.2) | 30.4–42.0 | 1951 (52.8) | 45.8–59.9 |
| A07EC02 | Mesalazine | 549 (44.6)) | 11.1–18.6 | 541 (14.6) | 10.9–18.4 | 552 (14.9) | 11.2–18.7 | 623 (16.9) | 12.9–20.9 | 845 (22.9) | 18.2–27.5 |
| A07EC01 | Sulfasalazine | 197 (5.3) | 3.0–7.6 | 187 (5.1) | 2.8–7.3 | 146 (4) | 2.1–5.8 | 128 (3.5) | 1.7–5.2 | 203 (5.5) | 3.2–7.8 |
| P01BA02 | Hydroxychloroquine | 324 (8.8) | 6.1–11.5 | 320 (8.7) | 5.9–11.4 | 300 (8.1) | 5.4–10.8 | 333 (9) | 6.1–11.9 | 404 (10.9) | 7.8–14.1 |
| L04AX0 | Azathioprine | 490 (13.3) | 9.6–17.0 | 384 (10.4) | 7.3–13.5 | 376 (10.2) | 6.9–13.5 | 345 (9.3) | 6.4–12.3 | 484 (13.1) | 9.4–16.8 |
| L04AD02 | Tacrolimus | 41 (1.1) | 0.1–2.1 | 38 (1) | 0.1–1.9 | 47 (1.3) | 0.2–2.3 | 47 (1.3) | 0.2–2.3 | 47 (1.3) | 0.2–2.3 |
| L04AD01 | Ciclosporin | 6 (0.2) | 0.0–0.5 | 9 (0.2) | 0.0–0.7 | 0 (0.0) | 0.0–0.0 | 13 (0.4) | 0.0–0.8 | 21 (0.6) | 0.0–1.2 |
| L04AX03, L01BA01 | Methotrexate | 70 (1.9) | 0.5–3.3 | 13 (0.4) | 0.0–1.0 | 0 (0.0) | 0.0–0.0 | 11 (0.3) | 0.0–0.7 | 24 (0.6) | 0.0–1.4 |
| L04AX03 | Methotrexate (low dose) | 20 (0.5) | 0.0–1.3 | 13 (0.4) | 0.0–1.0 | 0 (0.0) | 0.0–0.0 | 11 (0.3) | 0.0–0.7 | 24 (0.6) | 0.0–1.4 |
| L01BA01 | Methotrexate (high dose) | 51 (1.4) | 0.2–2.5 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 |
| L04AA06 | Myophenolic acid | 11 (0.3) | 0.0–0.8 | 11 (0.3) | 0.0–0.8 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 11 (0.3) | 0.0–0.9 |
| L04AA29 | Tofacitinib | 8 (0.2) | 0.0–0.6 | 20 (0.5) | 0.0–1.3 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 20 (0.5) | 0.0–1.3 |
| L04AB, AC | Biologic DMARDS | 821 (22.2) | 17.5–27 | 682 (18.5) | 14.3–22.7 | 438 (11.9) | 8.6–15.1 | 316 (8.6) | 5.7–11.4 | 725 (19.6) | 15.3–24.0 |
| L04AB | TNF-alpha inhibitors | 764 (20.7) | 16.1–25.3 | 661 (17.9) | 13.8–22.0 | 430 (11.6) | 8.4–14.9 | 316 (8.6) | 5.7–11.4 | 704 (19.1) | 14.8–23.3 |
| L04AB05 | Certolizumab | 143 (3.9) | 2.0–5.7 | 164 (4.4) | 2.4–6.4 | 156 (4.2) | 2.3–6.2 | 146 (4) | 2.1–5.8 | 228 (6.2) | 3.8–8.5 |
| L04AB02 | Infliximab | 207 (5.6) | 3.3–7.9 | 177 (4.8) | 2.8–6.8 | 148 (4) | 2.2–5.8 | 96 (2.6) | 1.1–4.2 | 177 (4.8) | 2.7–6.8 |
| L04AB01 | Etanercept | 174 (4.7) | 2.5–7.0 | 131 (3.5) | 1.8–5.3 | 55 (1.5) | 0.4–2.6 | 25 (0.7) | 0.0–1.5 | 149 (4) | 2.1–5.9 |
| L04AB04 | Adalimumab | 179 (4.8) | 2.4–7.3 | 158 (4.3) | 2.0–6.5 | 91 (2.5) | 0.8–4.1 | 38 (1) | 0.0–2.1 | 177 (4.8) | 2.4–7.2 |
| L04AB06 | Golimumab | 66 (1.8) | 0.4–3.1 | 30 (0.8) | 0.0–1.7 | 10 (0.3) | 0.0–0.8 | 10 (0.3) | 0.0–0.8 | 30 (0.8) | 0.0–1.7 |
| L04AC | Interleukin inhibitors | 64 (1.7) | 0.4–3.0 | 30 (0.8) | 0.0–1.8 | 8 (0.2) | 0.0–0.6 | 0 (0.0) | 0.0–0.0 | 30 (0.8) | 0.0–1.8 |
| L04AC07 | Tocilizumab | 45 (1.2) | 0.1–2.3 | 23 (0.6) | 0.0–1.5 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 23 (0.6) | 0.0–1.5 |
| L04AC05 | Ustekinumab | 8 (0.2) | 0.0–0.6 | 8 (0.2) | 0.0–0.6 | 8 (0.2) | 0.0–0.6 | 0 (0.0) | 0.0–0.0 | 8 (0.2) | 0.0–0.6 |
| L04AA33 | Vedolizumab | 17 (0.5) | 0.0–1.1 | 16 (0.4) | 0.0–1.0 | 6 (0.2) | 0.0–0.5 | 6 (0.2) | 0.0–0.5 | 16 (0.4)) | 0.0–1.0 |
| L01XC02, L04AA26 | B cell therapy | 6 (0.2) | 0.0–0.5 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 |
| L01XC02 | Rituximab | 6 (0.2) | 0.0–0.5 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 |
| L04AA26 | Belimumab | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 |
| J06BA02 | Immunoglobulins. normal for intravascular administration | 23 (0.6) | 0.0–1.4 | 12 (0.3) | 0.0–0.8 | 40 (1.1) | 0.3–1.9 | 88 (2.4) | 0.9–3.9 | 101 (2.7) | 1.1–4.3 |
| L04AA23, L03AX13, L03AB07, L04AX07, L04AA27 | Multiple sclerosis specific drugs | 538 (14.6) | 10.9–18.3 | 303 (8.2) | 5.7–10.8 | 40 (1.1) | 0.1–2.0 | 48 (1.3) | 0.4–2.3 | 316 (8.6) | 5.9–11.2 |
| L04AA23 | Natalizumab | 163 (4.4) | 2.5–6.3 | 111 (3) | 1.4–4.6 | 9 (0.2) | 0.0–0.7 | 0 (0.0) | 0.0–0.0 | 111 (3) | 1.4–4.6 |
| L03AX13 | Glatiramer acetate | 169 (4.4) | 2.6–6.5 | 94 (2.5) | 1.1–4.0 | 31 (0.8) | 0.0–1.7 | 30 (0.8) | 0.0–1.6 | 100 (2.7) | 1.2–4.2 |
| L03AB07 | Interferon beta-1 a | 182 (4.9) | 2.5–7.3 | 74 (2) | 0.8–3.2 | 0 (0.0) | 0.0–0.0 | 13 (0.4) | 0.0–0.8 | 80 (2.2) | 0.9–3.4 |
| L04AX07 | Dimethylfumarate | 41 (1.1) | 0.2–2.0 | 25 (0.7) | 0.0–1.3 | 0 (0.0) | 0.0–0.0 | 6 (0.2) | 0.0–0.5 | 25 (0.7) | 0.0–1.3 |
| L04AA27 | Fingolimod | 27 (0.7) | 0.0–1.6 | 14 (0.4) | 0.0–1.1 | 0 (0.0) | 0.0–0.0 | 19 (0.5) | 0.0–1.3 | 19 (0.5) | 0.0–1.3 |
| J05AR + J05AE01/03/08/10 + J05AF05/07/11 + J05AG01/04/05 + J05AX08/12 | HIV antiretrovirals | 477 (12.9) | 483 (13.1) | 610 (16.5) | 671 (18.2) | 760 (20.6) | |||||
| J05AR03 | Tenofovir disoproxil and emtracitabine | 223 (6.0) | 3.7–8.4 | 206 (5.6) | (3.4–7.8) | 235 (6.4) | 4.1–8.6 | 267 (7.)2 | 4.8–9.7 | 304 (8.2) | 5.6–10.9 |
| J05AE03 | Ritonavir | 112 (3.0) | 1.5–4.5 | 99 (2.7) | 1.4–4.0 | 179 (4.8) | 2.9–6.8 | 192 (5.2) | 3.2–7.2 | 205 (5.5) | 3.5–7.6 |
| D10BA, D10AD | Retinoids for acne treatment | 1641 (44.4) | 38.1–50.8 | 591 (16.0) | 12.3–19.7 | 136 (3.7) | 1.9–5.5 | 105 (2.8) | 1.1–4.6 | 789 (21.4) | 16.9–25.8 |
| D10BA01 | Isotretinoin (oral) | 153 (4.1) | 2.2–6.0 | 52 (1.4) | 0.2–2.6 | 0 (0.0) | 0.0–0.0 | 0 (0.0) | 0.0–0.0 | 52 (1.4) | 0.2–2.7 |
| D10AD | Topical retinoids | 1488 (40.3) | 34.2–46.3 | 539 (14.6) | 11.1–18.1 | 136 (3.7) | 1.9–5.5 | 105 (2.8) | 1.1–4.6 | 737 (20.0) | 15.7–24.2 |
3.2. Thyroid Therapy
3.2.1. Anti-Hypertensive Drugs
3.2.2. Psychotropic Drugs
3.3. Anti-Diabetics
3.3.1. Obstructive Airway Diseases
3.3.2. Drugs to Treat Auto-Immune Diseases
Systemic Glucocorticoids and Traditional Immunosuppressants
Biologic Immunosuppressants and Intravenous Immunoglobulins (IVIG)
3.3.3. Drugs to Treat Multiple Sclerosis
3.4. Human Immunodeficiency Virus (HIV) Antiretrovirals
3.5. Retinoids
| Drugs with Known or Potential Adverse Reaction (ATC Code) | Warnings Regarding Use during Critical Period | Risk Period | Pregnancies Exposed during Risk Period during Study Period (N) | Pregnancies Exposed during Risk Period during Study Period (%, 95%CI) |
|---|---|---|---|---|
| Methotrexate (L01BA01, L04AX03) | Risk of abortion. Malformations including face, skull, central nervous system, limbs, heart. | T1 (maximal risk between 8–10 GW) | 13 | 0.4 (0.0–1.0/10,000) |
| Mycophenolic acid (L04AA06) | Risk of abortion. Malformations including cleft palate, ear deformities in up to 25% of exposed foetuses. | T1 | 11 | 0.3 (0.0–0.8/10,000) |
| Cyclophosphamide (L01AA01) | Malformations including the face, skull, eyes and central nervous system. Hematologic anomalies (anemia, leucopenia) in the newborn. | T1 | 0 | 0.0 (0.0–0.0/10,000) |
| Carbimazole (H03BB01) | Malformations including aplasia cutis, choanal atreasia, tracheo-esophageal fistula in up to 4% of exposed foetuses. | T1 (maximal risk between 6–10 GW) | 174 | 4.7 (2.7–6.7/10,000) |
| Lithium (N05AN01) | Increased risk of heart malformation, especially the Ebstein malformation in 2.5% of exposed babies (including 1% of Ebstein disease vs. 0.005%). | T1 (until GW 10) | 19.6 | 0.5 (0.0–0.1/10,000) |
| Dolutegravir (J05AR13) 1 | Suspicion of an increased risk of neural tube defects. | T1 (Maximal risk between 0–4 GW) | 16 | 0.4 (0.4–1.0/10,000) |
| Isotretinoin | Malformations including the heart, central nervous system, face, ear, eyes and thymus in up to 20% of exposed foetuses. | T1 | 52 | 1.4 (0.2–2.6/10,000) |
| Drugs with Known or Potential Adverse Reaction (ATC Code) | Warnings Regarding Use during Critical Period | Risk Period | Pregnancies Exposed during Risk Period during Study Period (N) | Pregnancies Exposed during Risk Period during Study Period/10,000 95%CI) |
|---|---|---|---|---|
| Agents acting on the RAA system. (C09) | Renal toxicity which can induce oligo/anamnios, with potential pulmonary, skull hypoplasia and reduction of the extremities. Neonatal renal insufficiency. | T2 and T3 | 105, 72 | 2.8 (1.1–4.5/10,000) 1.9 (0.6–3.2/10,000) |
| Psychotropic drugs - Antidepressants; - Antipsychotics; - Benzodiazepines and related drugs (N06A, N05A, N05AN, N05BA, N05CD, N05CF) | -1/3 newborns present transient neonatal symptoms (serotoninergic toxicity or weaning symptoms); - Risk of transient extra-pyramidal symptoms in the newborn; - Prenatal exposure to benzodiazepines with long half-life in may cause impregnation symptoms (sleepiness, suction problems, hypotension, respiratory distress), whereas short half-life substances may cause weaning symptoms (agitation, hyperexcitability). | T3 | 779 | 172.2/10,000 |
| Rituximab (L01XC02) | Neonatal B cell depletion | T2 and T3 | 0, 0 | 0.0 (0.0–0.0/10,000) |
| Belimumab (L04AA26) | Neonatal B cell depletion | T2 and T3 | 0, 0 | 0.0 (0.0–0.0/10,000) |
| Drugs with Potential Adverse Reaction (ATC Code) | Warnings Regarding Use during Critical Period | Risk Period | Pregnancies Exposed during Risk Period during Study Period (N) | Pregnancies Exposed during Risk Period during Study Period (%, 95%CI) |
|---|---|---|---|---|
| Propylthiouracil (H03BA02) | T1 | 285 | 7.7 (5–10.5/10,000) | |
| Mitoxantrone (L01DB07) | Contraception is recommended 4 months after the last administration. | Pre-pregnancy and T1. | 0, 0 | 0.0 (0.0–0.0/10,000) |
| Fingolimod (L04AA27) | Contraception is recommended 2 months after the last administration. | 27, 14 | 0.7 (0.0–1.6/10,000) 0.4 (0.0–1.1/10,000) | |
| Teriflunomide (L04AA31) | 0, 0 | 0.0 (0.0–0.0/10,000) | ||
| Leflunomide (L04AA13) | 0, 0 | 0.0 (0.0–0.0/10,000) | ||
| Dimethylfumarate (L04AX07) | Contraception is recommended 3.5 months after the last administration. | 41, 25 | 1.1 (0.2–2.0/10,000), 0.7 (0.0–1.3/10,000) | |
| Cladribine (L01BB04, L04AA40) | 0, 0 | 0.0 (0.0–0.0/10,000) | ||
| Alemtuzumab (L04AA34) | Contraception is recommended 4 months after the last administration. | 0, 0 | 0.0 (0.0–0.0/10,000) | |
| Tofacitinib (L04AA29) [22] | 8, 20 | 0.2 (0.2–0.6/10,000), 0.5 (0.2–1.3/10,000) | ||
| Baricitinib (L04AA37) [23] | 0, 0 | 0.0 (0.0–0.0/10,000) |
4. Discussion
4.1. Thyroid Therapy
4.2. Hypertension
4.3. Psychotropic Drugs
4.4. Anti-Diabetics
4.5. Obstructive Airway Diseases
4.5.1. Drugs to Treat Auto-Immune Diseases
Glucocorticoids and Nzon-Biologic Immunosuppressants
Biologic Immunosuppressants and Intravenous Immunoglobulins (ivig)
4.5.2. Drugs to Treat Multiple Sclerosis
4.6. HIV Antiretrovirals
4.7. Retinoids
4.8. Clinical and Policy Implications
4.9. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Appendix A
| Codes | Definition | Type of Delivery |
|---|---|---|
| TarMed codes version (V01.08.00, 01.08.01, 01.09) | Grossesse et obstétrique | |
| 22.2110 | Surveillance de la naissance et conduite de l’accouchement, risque normal | Vaginal delivery |
| 22.2120 | +Césarienne secondaire | Cesarean delivery |
| 22.2130 | +Hystérectomie lors d’une césarienne | Cesarean delivery |
| 22.2200 | Surveillance de la naissance et conduite de l’accouchement, haut risque | Vaginal delivery |
| 22.2210 | Surveillance de la naissance et conduite de l’accouchement, très haut risque | Vaginal delivery |
| 22.2410 | Césarienne, planifiée ou primaire | Cesarean delivery |
| 22.2420 | Césarienne itérative | Cesarean delivery |
| SwissDRG Codes | MDC 14: Grossesse, naissance et suites de couches | |
| O01A (V3.0, V4.0, V5.0, V6.0) | Césarienne avec plusieurs diagnostics de complication, durée de la grossesse jusqu’à 25 semaines complètes ou avec thérapie intra-utérine | Cesarean delivery |
| O01A (V7.0) | Césarienne et dialyse, ou thérapie intra-utérine complexe du fœtus | Cesarean delivery |
| O01B (V3.0, V4.0) | Césarienne avec plusieurs diagnostics de complication, durée de la grossesse de 26 à 33 semaines complètes, sans thérapie intra-utérine ou avec diagnostic de complication, jusqu’à 25 semaines complètes, ou thromboembolie pendant la période de gestation avec procédure opératoire | Cesarean delivery |
| O01B (V5.0) | Césarienne avec plusieurs diagnostics de complication, durée de la grossesse de 26 à 33 semaines complètes, jusqu’à 25 semaines complètes, ou thromboembolie pendant la période de gestation avec procédure opératoire ou procédure complexe | Cesarean delivery |
| O01B (V6.0, V7.0) | Césarienne avec plusieurs diagnostics de complication, durée de la grossesse de 26 à 33 semaines ou CC extrêmement sévères ou diagnostic complexe ou procédure de complication, jusqu’à 33 semaines de grossesse ou diagnostic complexe et CC extrêmement sévères, ou jusqu’à 25 semaines de grossesse et diagnostic de complication | Cesarean delivery |
| O01C (V3.0, V4.0, V5.0) | Césarienne avec plusieurs diagnostics de complication, durée de la grossesse > 33 semaines complètes, sans thérapie intra-utérine ou avec diagnostic de complication, de 26 à 33 semaines ou avec diagnostic complexe ou jusqu’à 33 semaines ou avec diagnostic complexe, avec CC extrêmement sévères | Cesarean delivery |
| O01C (V6.0, V7.0) | Césarienne secondaire avec plusieurs diagnostics de complication ou procédure complexe, ou jusqu’à 33 semaines de grossesse ou diagnostic complexe ou diagnostic de complication et grossesse de 26 à 33 semaines ou diagnostic complexe | Cesarean delivery |
| O01D (V3.0, V4.0, V5.0) | Césarienne avec plusieurs diagnostics de complication, durée de la grossesse > 33 semaines complètes, sans thérapie intra-utérine ou avec diagnostic de complication, de 26 à 33 semaines ou avec diagnostic complexe ou jusqu’à 33 semaines ou avec diagnostic complexe, sans CC extrêmement sévères | Cesarean delivery |
| O01D (V6.0, V7.0) | Césarienne secondaire avec diagnostic de complication, durée de la grossesse plus de 33 semaines complètes | Cesarean delivery |
| O01E (V3.0, V4.0, V5.0) | Césarienne avec diagnostic de complication, durée de la grossesse plus de 33 semaines complètes, sans diagnostic complexe | Cesarean delivery |
| O01E (V6.0, V7.0) | Césarienne avec plusieurs diagnostics de complication ou procédure complexe, ou jusqu’à 33 semaines de grossesse ou diagnostic complexe, ou diagnostic de complication et grossesse de 26 à 33 semaines ou diagnostic complexe, ou césarienne secondaire | Cesarean delivery |
| O01F (V3.0, V4.0, V5.0) | Césarienne sans diagnostic de complication, durée de la grossesse plus de 33 semaines complètes, sans diagnostic complexe | Cesarean delivery |
| O01F (V6.0 V7.0) | Césarienne avec diagnostic de complication, durée de la grossesse plus de 33 semaines complètes | Cesarean delivery |
| O01G (V6.0, V7.0) | Césarienne, durée de la grossesse > 33 semaines complètes | Cesarean delivery |
| O01H (V7.0) | Césarienne, durée de la grossesse plus de 33 semaines complètes | Cesarean delivery |
| O02A (V3.0, V4.0) | Accouchement par voie basse avec procédure opératoire de complication, durée de la grossesse jusqu’à 33 semaines complètes ou avec thérapie intra-utérine | Vaginal delivery |
| O02A (V5.0, V6.0, V7.0) | Accouchement par voie basse avec procédure opératoire de complication, avec thérapie intra-utérine ou traitement complexe de soins intensifs > 119 points ou procédure de complication ou procédure complexe | Vaginal delivery |
| O02B (V3.0, V4.0) | Accouchement par voie basse avec procédure opératoire de complication, durée de la grossesse plus de 33 semaines complètes, sans thérapie intra-utérine | Vaginal delivery |
| O02B (V5.0, V6.0, V7.0) | Accouchement par voie basse avec procédure opératoire de complication | Vaginal delivery |
| O60A (V3.0) | Accouchement par voie basse avec plusieurs diagnostics de complication, au moins une complication sévère, durée de la grossesse jusqu’à 33 semaines complètes ou avec procédure de complication | Vaginal delivery |
| 060A (V 4.0, V5.0, V6.0, V7.0) | Accouchement par voie basse avec plusieurs diagnostics de complication, au moins une complication sévère, durée de la grossesse jusqu’à 33 semaines complètes ou avec procédure de complication ou thromboembolie pendant la période de gestation | Vaginal delivery |
| 060B (V3.0) | Accouchement par voie basse avec plusieurs diagnostics de complication, au moins une complication sévère, durée de la grossesse plus de 33 semaines complètes, sans procédure de complication ou thromboembolie pendant la période de gestation sans procédure opératoire | Vaginal delivery |
| 060B (V4.0, V5.0) | Accouchement par voie basse avec plusieurs diagnostics de complication, au moins une complication sévère, durée de la grossesse plus de 33 semaines complètes, sans procédure de complication ou thromboembolie pendant la période de gestation | Vaginal delivery |
| O60B (V6.0, V7.0) | Accouchement par voie basse avec plusieurs diagnostics de complication, au moins une complication sévère, durée de la grossesse plus de 33 semaines complètes | Vaginal delivery |
| O60C (V3.0, V4.0, V5.0, V6.0, V7.0) | Accouchement par voie basse avec diagnostic de complication sévère ou moyennement sévère | Vaginal delivery |
| O60D (V3.0, V4.0) | Accouchement par voie basse sans diagnostic de complication | Vaginal delivery |
| 060D (V5.0, V6.0, V7.0) | Accouchement par voie basse | Vaginal delivery |
| Midwife codes | ||
| B1 | Leitung einer ambulanten Geburt | Vaginal delivery |
| B2 | Zweithebamme für ambulante Geburt oder Verlegung | Vaginal delivery |
| B3 | Verbrauchsmaterial für unvollendete ambulante Geburt | Vaginal delivery |
| B4 | Verbrauchsmaterial für ambulante Geburt | Vaginal delivery |
References
- Lyerly, A.D.; Little, M.O.; Faden, R.R. Pregnancy and Clinical Research. Hastings Cent. Rep. 2008, 38, 53. [Google Scholar] [CrossRef]
- Widnes, S.K.F.; Schjøtt, J. Advice on drug safety in pregnancy: Are there differences between commonly used sources of information? Drug Saf. 2008, 31, 799–806. [Google Scholar] [CrossRef] [Green Version]
- Noh, Y.; Yoon, D.; Song, I.; Jeong, H.E.; Bae, J.H.; Shin, J.-Y. Discrepancies in the Evidence and Recommendation Levels of Pregnancy Information in Prescription Drug Labeling in the United States, United Kingdom, Japan, and Korea. J. Womens Health 2018, 27, 1086–1092. [Google Scholar] [CrossRef]
- Dashraath, P.; Nielsen-Saines, K.; Madhi, S.A.; Baud, D. COVID-19 vaccines and neglected pregnancy. Lancet 2020, 396, e22. [Google Scholar] [CrossRef]
- Dathe, K.; Schaefer, C. The Use of Medication in Pregnancy. Dtsch. Ärzteblatt Int. 2019, 116, 783–790. [Google Scholar] [CrossRef]
- Lupattelli, A.; Spigset, O.; Twigg, M.J.; Zagorodnikova, K.; Mårdby, A.C.; Moretti, M.E.; Drozd, M.; Panchaud, A.; Hämeen-Anttila, K.; Rieutord, A.; et al. Medication use in pregnancy: A cross-sectional, multinational web-based study. BMJ Open 2014, 4, e004365. [Google Scholar] [CrossRef] [Green Version]
- Jølving, L.R.; Nielsen, J.; Kesmodel, U.S.; Nielsen, R.G.; Beck-Nielsen, S.S.; Nørgård, B.M. Prevalence of maternal chronic diseases during pregnancy—A nationwide population based study from 1989 to 2013. Acta Obstet. Gynecol. Scand. 2016, 95, 1295–1304. [Google Scholar] [CrossRef]
- Chronic Diseases in Pregnant Women: Prevalence and Birth Outcomes Based on the SNiP-Study|BMC Pregnancy and Childbirth|Full Text. Available online: https://bmcpregnancychildbirth.biomedcentral.com/articles/10.1186/1471-2393-14-75 (accessed on 16 November 2021).
- Chatterjee, S.; Kotelchuck, M.; Sambamoorthi, U. Prevalence of Chronic Illness in Pregnancy, Access to Care, and Health Care Costs: Implications for Interconception Care. Women’s Health Issues 2008, 18 (Suppl. S6), S107–S116. [Google Scholar] [CrossRef]
- Statistique O Fédéral de la. Age Moyen des Mères et des Pères à la Naissance de L’enfant Selon la Nationalité, 1971–2019|Tableau. Office Fédéral de la Statistique. Published 29 September 2020. Available online: https://www.bfs.admin.ch/bfs/fr/home/statistiques/catalogues-banques-donnees/tableaux.assetdetail.14387072.html (accessed on 13 September 2021).
- Gerbier, E.; Graber, S.M.; Rauch, M.; Marxer, C.A.; Meier, C.R.; Baud, D.; Winterfeld, U.; Blozik, E.; Surbek, D.; Spoendlin, J.; et al. Use of drugs to treat symptoms and acute conditions during pregnancy in outpatient care in Switzerland between 2014 and 2018: Analysis of Swiss health care claims data. Swiss Med. Wkly. 2021, 151, w30048. [Google Scholar]
- Spoendlin, J.; Blozik, E.; Graber, S.; Rauch, M.; Marxer, C.; Rüegg, S.; Meier, C.; Winterfeld, U.; Panchaud, A. Use of valproate in pregnancy and in women of childbearing age between 2014 and 2018 in Switzerland: A retrospective analysis of Swiss healthcare claims data. Swiss Med. Wkly. 2021, 151, w20386. [Google Scholar] [CrossRef]
- WHOCC—ATC/DDD Index. Available online: https://www.whocc.no/atc_ddd_index/ (accessed on 21 January 2021).
- Accouchement Extrahospitalier: État des Lieux et Perspectives. Revue Medicale Suisse. Available online: https://www.revmed.ch/revue-medicale-suisse/2017/revue-medicale-suisse-580/accouchement-extrahospitalier-etat-des-lieux-et-perspectives (accessed on 13 September 2021).
- Margulis, A.V.; Setoguchi, S.; Mittleman, M.A.; Glynn, R.J.; Dormuth, C.R.; Hernández-Díaz, S. Algorithms to estimate the beginning of pregnancy in administrative databases. Pharmacoepidemiol. Drug Saf. 2013, 22, 16–24. [Google Scholar] [CrossRef] [PubMed]
- CRAT—Centre de Référence sur les Agents Tératogènes Chez la Femme Enceinte. Available online: http://www.lecrat.fr/ (accessed on 26 November 2020).
- Embryotox—Sécurité des Médicaments Pendant la Grossesse et L’allaitement: Accueil. Available online: https://www.embryotox.de/ (accessed on 26 November 2020).
- Swissmedic 2019 © Copyright. (Direct) Healthcare Professional Communications. Available online: https://www.swissmedic.ch/swissmedic/fr/home/humanarzneimittel/marktueberwachung/healthcare-professional-communications.html (accessed on 26 March 2021).
- Lumley, T. Analysis of Complex Survey Samples. J. Stat. Softw. 2004, 9, 1–19. [Google Scholar] [CrossRef] [Green Version]
- R: The R Project for Statistical Computing. Available online: https://www.r-project.org/ (accessed on 26 March 2021).
- Statistique O Fédéral de la. Nombre et Taux de Césariennes Selon le Canton et la Région de Domicile—2007–2019|Tableau. Office Fédéral de la Statistique. Published 20 November 2020. Available online: https://www.bfs.admin.ch/bfs/fr/home/statistiques/catalogues-banques-donnees/tableaux.assetdetail.14841442.html (accessed on 17 November 2021).
- Clowse, M.E.B.; Feldman, S.R.; Isaacs, J.; Kimball, A.; Strand, V.; Warren, R.B.; Xibillé, D.; Chen, Y.; Frazier, D.; Geier, J.; et al. Pregnancy Outcomes in the Tofacitinib Safety Databases for Rheumatoid Arthritis and Psoriasis. Drug Saf. 2016, 39, 755–762. [Google Scholar] [CrossRef]
- Costanzo, G.; Firinu, D.; Losa, F.; Deidda, M.; Barca, M.P.; Del Giacco, S. Baricitinib exposure during pregnancy in rheumatoid arthritis. Ther. Adv. Musculoskelet. Dis. 2020, 12, 1759720X19899296. [Google Scholar] [CrossRef]
- Engeland, A.; Bjørge, T.; Klungsøyr, K.; Hjellvik, V.; Skurtveit, S.; Furu, K. Trends in prescription drug use during pregnancy and postpartum in Norway, 2005 to 2015. Pharmacoepidemiol. Drug Saf. 2018, 27, 995–1004. [Google Scholar] [CrossRef]
- Bérard, A.; Abbas-Chorfa, F.; Kassai, B.; Vial, T.; Nguyen, K.A.; Sheehy, O.; Schott, A.-M. The French Pregnancy Cohort: Medication use during pregnancy in the French population. PLoS ONE 2019, 14, e0219095. [Google Scholar] [CrossRef] [Green Version]
- Thyroid Disease in Pregnancy: ACOG Practice Bulletin, Number 223. Obstet. Gynecol. 2020, 135, e261. [CrossRef]
- Carbimazole/Thiamazole—Grossesse et Allaitement. Available online: https://lecrat.fr/spip.php?page=article&id_article=296 (accessed on 15 July 2021).
- Embryotox—Arzneimittelsicherheit in Schwangerschaft und Stillzeit: Carbimazol. Available online: https://www.embryotox.de/arzneimittel/details/carbimazol/ (accessed on 15 July 2021).
- Andersen, S.L.; Olsen, J.; Wu, C.S.; Laurberg, P. Birth defects after early pregnancy use of antithyroid drugs: A Danish nationwide study. J. Clin. Endocrinol. Metab. 2013, 98, 4373–4381. [Google Scholar] [CrossRef]
- Song, R.; Lin, H.; Chen, Y.; Zhang, X.; Feng, W. Effects of methimazole and propylthiouracil exposure during pregnancy on the risk of neonatal congenital malformations: A meta-analysis. PLoS ONE 2017, 12, e0180108. [Google Scholar] [CrossRef] [Green Version]
- Andersen, S.L.; Lönn, S.; Vestergaard, P.; Törring, O. Birth defects after use of antithyroid drugs in early pregnancy: A Swedish nationwide study. Eur. J. Endocrinol. 2017, 177, 369–378. [Google Scholar] [CrossRef]
- Alexander, E.K.; Pearce, E.N.; Brent, G.A.; Brown, R.S.; Chen, H.; Dosiou, C.; Grobman, W.A.; Laurberg, P.; Lazarus, J.H.; Mandel, S.J.; et al. 2017 Guidelines of the American Thyroid Association for the Diagnosis and Management of Thyroid Disease During Pregnancy and the Postpartum. Thyroid 2017, 27, 315–389. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Regitz-Zagrosek, V.; Roos-Hesselink, J.W.; Bauersachs, J.; Blomström-Lundqvist, C.; Cífková, R.; De Bonis, M.; Iung, B.; Johnson, M.R.; Kintscher, U.; Kranke, P.; et al. 2018 ESC Guidelines for the management of cardiovascular diseases during pregnancy: The Task Force for the Management of Cardiovascular Diseases during Pregnancy of the European Society of Cardiology (ESC). Eur. Heart J. 2018, 39, 3165–3241. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.A.; Magee, L.A.; Kenny, L.C.; Karumanchi, S.A.; McCarthy, F.P.; Saito, S.; Hall, D.R.; Warren, C.E.; Adoyi, G.; Ishaku, S. The hypertensive disorders of pregnancy: ISSHP classification, diagnosis & management recommendations for international practice. Pregnancy Hypertens. 2018, 13, 291–310. [Google Scholar] [CrossRef] [PubMed]
- La Tocolyse dans les Menaces D’accouchement Prématuré. Sggg.ch. Published 2021. Available online: https://www.sggg.ch/fileadmin/user_upload/41_Tocolyse_2013.pdf (accessed on 18 October 2021).
- Hypertension chez la Femme Enceinte. Revue Medicale Suisse. Available online: https://www.revmed.ch/revue-medicale-suisse/2007/revue-medicale-suisse-124/hypertension-chez-la-femme-enceinte (accessed on 10 September 2021).
- Inhibiteurs de Lenzyme de Conversion (IEC) et Antagonistes des Récepteurs de L’angiotensine II (Sartans)—Grossesse. Available online: https://lecrat.fr/spip.php?page=article&id_article=486 (accessed on 15 July 2021).
- Embryotox—Sécurité des Médicaments Pendant la Grossesse et L’allaitement: Énalapril. Available online: https://www.embryotox.de/arzneimittel/details/enalapril/ (accessed on 15 July 2021).
- Weber-Schoendorfer, C.; Kayser, A.; Tissen-Diabaté, T.; Winterfeld, U.; Eleftheriou, G.; Winkel, B.T.; Diav-Citrin, O.; Greenall, A.; Hoeltzenbein, M.; Schaefer, C. Fetotoxic risk of AT1 blockers exceeds that of angiotensin-converting enzyme inhibitors: An observational study. J. Hypertens. 2020, 38, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Huybrechts, K.F.; Bateman, B.T.; Palmsten, K.; Desai, R.J.; Patorno, E.; Gopalakrishnan, C.; Levin, R.; Mogun, H.; Hernandez-Diaz, S. Antidepressant use late in pregnancy and risk of persistent pulmonary hypertension of the newborn. JAMA 2015, 313, 2142–2151. [Google Scholar] [CrossRef]
- Etat des Connaissances sur le Citalopram—Grossesse. Available online: https://lecrat.fr/spip.php?page=article&id_article=226 (accessed on 15 July 2021).
- Embryotox—Arzneimittelsicherheit in Schwangerschaft und Stillzeit: Sertralin. Available online: https://www.embryotox.de/arzneimittel/details/sertralin/ (accessed on 15 July 2021).
- Overview|Antenatal and Postnatal Mental Health: Clinical Management and Service Guidance|Guidance|NICE. Available online: https://www.nice.org.uk/guidance/cg192 (accessed on 1 June 2021).
- Neuroleptiques/Antipsychotiques et Grossesse. Available online: https://lecrat.fr/spip.php?page=article&id_article=115 (accessed on 15 July 2021).
- Embryotox—Sécurité des Médicaments Pendant la Grossesse et L’allaitement: Quétiapine. Available online: https://www.embryotox.de/arzneimittel/details/quetiapin/ (accessed on 15 July 2021).
- Lithium—Grossesse et Allaitement. Available online: https://lecrat.fr/spip.php?page=article&id_article=360 (accessed on 15 July 2021).
- Embryotox—Sécurité des Médicaments Pendant la Grossesse et L’allaitement: Sels de Lithium. Available online: https://www.embryotox.de/arzneimittel/details/lithiumsalze/ (accessed on 15 July 2021).
- Anxiolytiques et Grossesse. Available online: https://lecrat.fr/spip.php?page=article&id_article=21 (accessed on 15 July 2021).
- AWMF: Détail. Available online: https://www.awmf.org/leitlinien/detail/ll/057-008.html (accessed on 17 November 2021).
- Recommendations|Diabetes in Pregnancy: Management from Preconception to the Postnatal Period|Guidance|NICE. Available online: https://www.nice.org.uk/guidance/ng3/chapter/Recommendations (accessed on 16 July 2021).
- Dépistage du Diabète Gestationnel. Sggg.ch. Available online: https://www.sggg.ch/fileadmin/user_upload/Dokumente/3_Fachinformationen/1_Expertenbriefe/Fr/37_Depistage_du_diabete_gestationnel_2011.pdf. (accessed on 18 October 2021).
- Metformin—Grossesse et Allaitement. Available online: https://lecrat.fr/spip.php?page=article&id_article=867 (accessed on 15 July 2021).
- Glibenclamide = Glyburide—Grossesse et Allaitement. Available online: https://lecrat.fr/spip.php?page=article&id_article=870 (accessed on 15 July 2021).
- 2020 GINA Main Report. Global Initiative for Asthma—GINA. Available online: https://ginasthma.org/gina-reports/ (accessed on 25 January 2021).
- Murphy, V.E.; Namazy, J.A.; Powell, H.; Schatz, M.; Chambers, C.; Attia, J.; Gibson, P.G. A meta-analysis of adverse perinatal outcomes in women with asthma. BJOG 2011, 118, 1314–1323. [Google Scholar] [CrossRef]
- Cornish, J.; Tan, E.; Teare, J.; Teoh, T.G.; Rai, R.; Clark, S.K.; Tekkis, P.P. A meta-analysis on the influence of inflammatory bowel disease on pregnancy. Gut 2007, 56, 830–837. [Google Scholar] [CrossRef] [Green Version]
- Petri, M. Pregnancy and Systemic Lupus Erythematosus. Best Pract. Res. Clin. Obstetr. Gynaecol. 2020, 64, 24–30. [Google Scholar] [CrossRef]
- Voskuhl, R.; Momtazee, C. Pregnancy: Effect on Multiple Sclerosis, Treatment Considerations, and Breastfeeding. Neurotherapeutics 2017, 14, 974–984. [Google Scholar] [CrossRef]
- Skorpen, C.G.; Hoeltzenbein, M.; Tincani, A.; Fischer-Betz, R.; Elefant, E.; Chambers, C.; Da Silva, J.; Nelson-Piercy, C.; Cetin, I.; Costedoat-Chalumeau, N.; et al. The EULAR points to consider for use of antirheumatic drugs before pregnancy, and during pregnancy and lactation. Ann. Rheum. Dis. 2016, 75, 795–810. [Google Scholar] [CrossRef] [Green Version]
- Mycophénolate—Grossesse et Allaitement. Available online: https://lecrat.fr/spip.php?page=article&id_article=423 (accessed on 15 July 2021).
- Methotrexate—Grossesse et Allaitement. Available online: https://lecrat.fr/spip.php?page=article&id_article=469 (accessed on 15 July 2021).
- Pattinson, R.C. A Meta-Analysis of the Use of Corticosteroids in Pregnancies Complicated by Preterm Premature Rupture of Membranes; Centre for Reviews and Dissemination: York, UK, 1999. [Google Scholar]
- Corticoïdes—Grossesse et Allaitement. Available online: https://lecrat.fr/spip.php?page=article&id_article=672 (accessed on 15 October 2021).
- Østensen, M.; Khamashta, M.; Lockshin, M.; Parke, A.; Brucato, A.; Carp, H.; Doria, A.; Rai, R.; Meroni, P.L.; Cetin, I.; et al. Anti-inflammatory and immunosuppressive drugs and reproduction. Arthritis Res. Ther. 2006, 8, 209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mariette, X.; Förger, F.; Abraham, B.; Flynn, A.D.; Moltó, A.; Flipo, R.-M.; van Tubergen, A.; Shaughnessy, L.; Simpson, J.; Teil, M.; et al. Lack of placental transfer of certolizumab pegol during pregnancy: Results from CRIB, a prospective, postmarketing, pharmacokinetic study. Ann. Rheum. Dis. 2018, 77, 228–233. [Google Scholar] [CrossRef] [PubMed]
- EU Approves Certolizumab Pegol for Pregnant & Nursing Patients. The Rheumatologist. Available online: https://www.the-rheumatologist.org/article/eu-approves-certolizumab-pegol-pregnant-nursing-patients/ (accessed on 18 October 2021).
- Certolizumab—Grossesse et Allaitement. Available online: https://lecrat.fr/spip.php?page=article&id_article=946 (accessed on 15 July 2021).
- Infliximab—Grossesse et Allaitement. Available online: https://lecrat.fr/spip.php?page=article&id_article=429 (accessed on 15 July 2021).
- Etanercept—Grossesse et Allaitement. Available online: https://lecrat.fr/spip.php?page=article&id_article=431 (accessed on 15 July 2021).
- Rituximab—Grossesse et Allaitement. Available online: https://lecrat.fr/spip.php?page=article&id_article=1105 (accessed on 15 July 2021).
- D’Mello, R.J.; Hsu, C.-D.; Chaiworapongsa, P.; Chaiworapongsa, T. Update on the Use of Intravenous Immunoglobulin in Pregnancy. NeoReviews 2021, 22, e7–e24. [Google Scholar] [CrossRef] [PubMed]
- Confavreux, C.; Hutchinson, M.; Hours, M.M.; Cortinovis-Tourniaire, P.; Moreau, T. Rate of pregnancy-related relapse in multiple sclerosis. Pregnancy in Multiple Sclerosis Group. N. Engl. J. Med. 1998, 339, 285–291. [Google Scholar] [CrossRef]
- Natalizumab—Grossesse et Allaitement. Available online: https://lecrat.fr/spip.php?page=article&id_article=1083 (accessed on 15 July 2021).
- Résumé des Caractéristiques du Produit—MITOXANTRONE ACCORD 2 mg/mL, Solution à Diluer Pour Perfusion—Base de Données Publique des Médicaments. Available online: https://base-donnees-publique.medicaments.gouv.fr/affichageDoc.php?specid=61760943&typedoc=R (accessed on 18 October 2021).
- Médicaments Utilisés dans les Formes très Actives de Sclérose en Plaques Récurrente. Haute Autorité de Santé. Available online: https://www.has-sante.fr/jcms/p_3067012/fr/medicaments-utilises-dans-les-formes-tres-actives-de-sclerose-en-plaques-recurrente (accessed on 19 April 2021).
- Léflunomide/Tériflunomide—Grossesse et Allaitement. Available online: https://lecrat.fr/spip.php?page=article&id_article=696 (accessed on 15 July 2021).
- Résumé des Caractéristiques du Produit. Available online: http://agence-prd.ansm.sante.fr/php/ecodex/rcp/R0356973.htm (accessed on 18 October 2021).
- Tecfidera, INN-Dimethyl Fumarate. Available online: https://www.ema.europa.eu/en/documents/product-information/tecfidera-epar-product-information_en.pdf (accessed on 10 September 2021).
- Mavenclad, INN-Cladribine. Available online: https://www.ema.europa.eu/en/documents/product-information/mavenclad-epar-product-information_en.pdf (accessed on 23 January 2022).
- Oh, J.; Achiron, A.; Celius, E.G.; Chambers, C.; Derwenskus, J.; Devonshire, V.; Hellwig, K.; Hutton, G.J.; McCombe, P.; Moore, M.; et al. Pregnancy outcomes and postpartum relapse rates in women with RRMS treated with alemtuzumab in the phase 2 and 3 clinical development program over 16 years. Mult. Scler. Relat. Disord. 2020, 43, 102146. [Google Scholar] [CrossRef]
- OFSP O Fédéral de la Santé Publique. Sida. Available online: https://www.bag.admin.ch/bag/fr/home/krankheiten/krankheiten-im-ueberblick/aids.html (accessed on 6 December 2021).
- Le VIH/Sida en Chiffres. Aide Suisse Contre le Sida. Available online: https://aids.ch/fr/questions-frequentes/vih-sida/chiffres/83 (accessed on 6 December 2021).
- HIV 2019. Available online: https://en.ssi.dk/surveillance-and-preparedness/surveillance-in-denmark/annual-reports-on-disease-incidence/hiv-2019 (accessed on 6 December 2021).
- What’s New in the Guidelines|NIH. Available online: https://clinicalinfo.hiv.gov/en/guidelines/perinatal/whats-new-guidelines (accessed on 16 November 2021).
- Stavudine (Zerit, d4T)|NIH. Available online: https://clinicalinfo.hiv.gov/en/guidelines/perinatal/stavudine-zerit-d4t (accessed on 16 November 2021).
- Swissmedic 2019 © Copyright. HPC—Correct Use of Retinoids in Dermatology. Available online: https://www.swissmedic.ch/swissmedic/en/home/humanarzneimittel/marktueberwachung/healthcare-professional-communications/archiv/hpc-_-die-sichere-anwendung-von-retinoiden-in-der-dermatologie.html (accessed on 19 January 2022).
- De Wit, M.L.; Rajulton, F. Education and timing of parenthood among Canadian women: A cohort analysis. Soc. Biol. 1992, 39, 109–122. [Google Scholar] [CrossRef]
- Schenkel, L.; Simões-Wüst, A.P.; Hösli, I.; von Mandach, U. Drugs in Pregnancy and Lactation—Medications Used in Swiss Obstetrics. Z. Geburtshilfe Neonatol. 2018, 222, 152–165. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gerbier, E.; Graber, S.M.; Rauch, M.; Marxer, C.A.; Meier, C.R.; Baud, D.; Winterfeld, U.; Blozik, E.; Surbek, D.; Spoendlin, J.; et al. Use of Prescribed Drugs to Treat Chronic Diseases during Pregnancy in Outpatient Care in Switzerland between 2014 and 2018: Descriptive Analysis of Swiss Health Care Claims Data. Int. J. Environ. Res. Public Health 2022, 19, 1456. https://doi.org/10.3390/ijerph19031456
Gerbier E, Graber SM, Rauch M, Marxer CA, Meier CR, Baud D, Winterfeld U, Blozik E, Surbek D, Spoendlin J, et al. Use of Prescribed Drugs to Treat Chronic Diseases during Pregnancy in Outpatient Care in Switzerland between 2014 and 2018: Descriptive Analysis of Swiss Health Care Claims Data. International Journal of Environmental Research and Public Health. 2022; 19(3):1456. https://doi.org/10.3390/ijerph19031456
Chicago/Turabian StyleGerbier, Eva, Sereina M. Graber, Marlene Rauch, Carole A. Marxer, Christoph R. Meier, David Baud, Ursula Winterfeld, Eva Blozik, Daniel Surbek, Julia Spoendlin, and et al. 2022. "Use of Prescribed Drugs to Treat Chronic Diseases during Pregnancy in Outpatient Care in Switzerland between 2014 and 2018: Descriptive Analysis of Swiss Health Care Claims Data" International Journal of Environmental Research and Public Health 19, no. 3: 1456. https://doi.org/10.3390/ijerph19031456







