1. Introduction
The world’s largest energy consumption is produced by coal, making it the second-largest energy source [
1]. South Africa (SA) has been ranked the fifth producer and consumer of coal and 86% of the energy produced in SA is derived from it [
1]. In SA, coal has been one of the reasons behind the country’s improved financial and economic growth due to its availability and cost-effectiveness. The rise in energy demand has exposed the use of coal as a sustainable energy source which causes a potential health hazard to humans and the environment due to the release of a by-product known as coal tar [
2]. Thousands of coking processes are responsible for the release of coal tar and gas fuel production, which are being derived from coal transformation [
3].
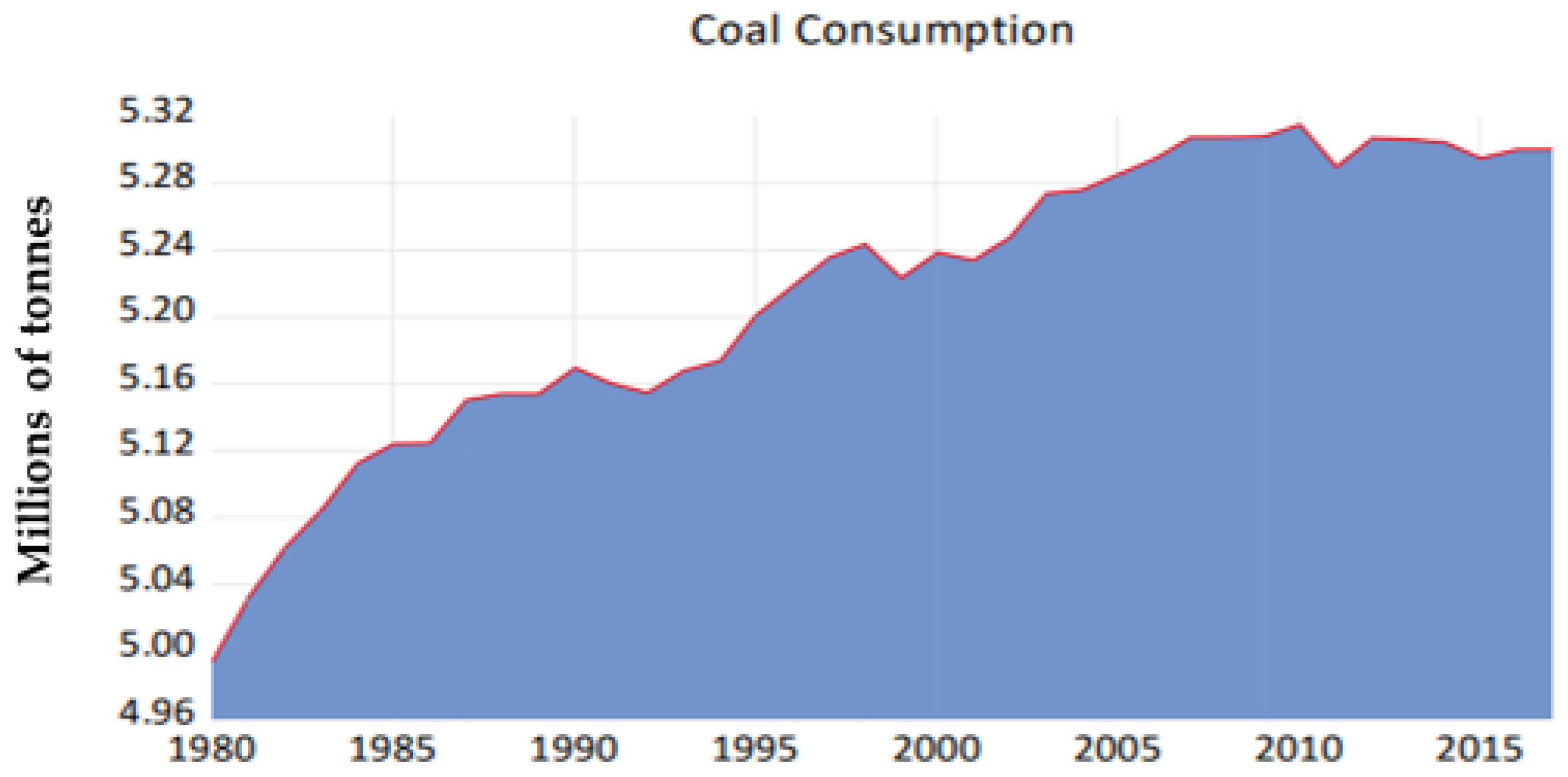
Figure 1 represents the usage of coal in SA from the year 1980 to 2015 and it has the potential to release a by-product that consists of polycyclic aromatic hydrocarbons (PAHs).
Coal tar is a by-product of coal that is released from various processes of the coal gas and coking process. It is semi-liquid in nature with a density of 1.15 to 1.4 g/cm
3 and has a foul smell [
4].
Polycyclic aromatic hydrocarbons (PAHs) are a group of organic compounds that are life-threatening because of their chemical structures, that consist of benzene rings bonded in a linear and angular position [
5]. Though there are hundreds of PAH compounds, only 16 PAHs are commonly identified in various wastewater and water sources [
6]. See also
Table 1.
PAHs are released from various industries, from petroleum, coal tar and oil spills that end up settling in the environment and water streams, making them hazardous due to their toxicity level [
7]. According to Wlodarczyk-Makula [
8], the leaching of the pollutants in the various water sources contain PAHs with different chemical properties and concentration ranges from 46 to 70 g/d. Świetlik et al. [
9] investigated the photodegradation of PAHs in distilled water and river water. Before the experiment, solids were removed from the river water using a nylon membrane with an opening size of 0.45 µm to prevent any reaction that may occur between the PAHs and the water. It was found that the PAHs were more concentrated in river water compared to distilled water. This was attributed to the physiochemistry of the river water, the temperature and the pH level [
9].
Based on the major effluents that occur in various industries, PAHs have attracted a lot of attention due to their presence in wastewater and other water sources. Their deposition into the environment has led to major health concerns such as carcinogenic, mutagenic and teratogenic symptoms. The solubility of PAHs is dependent on various factors such as the temperature, pH, ionic strength and water matrix of the dissolved organic carbon [
10]. It has been reported by Mojiri, Zhou, Ohashi, Ozaki and Kindaichi [
5] that the concentration of pyrene in water in South Africa is about 1,118,000 × 10
−6 mg/L and 8,310,000 × 10
−6 mg/L for benzo(b)fluoranthene in sewage wastewater.
This study aims to assess the influence of the physiochemical properties of receiving water sources, such as sewage, and acid and alkaline mine drainage, on the release of PAHs from coal tar.
Table 2 shows the molecular weight of the PAHs that are mostly found in water sources and recognized by the World Health Organization (WHO). A few PAHs have been identified to be life-threatening due to their extreme effects on humans, as established by their chemical properties depicted in
Table 2.
It has been reported by Mojiri, Zhou, Ohashi, Ozaki and Kindaichi [
5] that, in an aquatic environment, the PAHs concentrations were 0.03 × 10
−6 mg/L to 83,100,000 × 10
−6 mg/L. Among these PAHs, the lower molecular weight species, such as naphthalene, which consist of two to three rings, are not as cancerous as other PAHs of four to ten ring structures, such as chrysene, pyrene and others [
11,
12].
Table 3 shows the PAHs content in various water sources. However, drinking water was pointed out to have a higher PAHs presence compared to groundwater.
During the leaching process, PAHs normally migrate into the soil surface and groundwater where most of the PAHs are adsorbed or biodegraded in the existing minerals or enzymes in the soil. The remaining PAHs then largely channel into the drinking water resulting in it seeming to be more polluted than the groundwater [
13,
14].
Phase Partition Equilibrium (Raoult’s Law) and Dissolution of Kinetics
Raoult’s law can be applied for the water samples and the non-miscible organic phase (PAHs), both having an ideal behaviour [
15] that best fits the study aim. The ideal behaviour happens to be linear, whereby the more the coal is immersed in the water deposit, will result in more PAHs released in that water. Raoult’s law [
16] can best describe the influence on the leaching process of various parameters; however, only pH and contact time were investigated in the study to determine their influence on the leaching process.
The distribution of the pollutant “coal tar”, that contains PAHs, into water sources (sewage wastewater, acidic and alkaline mine drainage) is controlled by the molecular diffusion of the coal tar interphase [
17]. Since the viscosity of the coal tar is also considered, the global mass transfer is controlled by the slowest rate [
18]. This can be attributed to the release of the PAHs in the water source by ageing. The slower the diffusion of the (coal tar) water interface, the higher the concentration of PAHs in the water source [
3,
14].
3. Results and Discussion
In
Table 4, the results are shown for the water sources analyzed in the study.
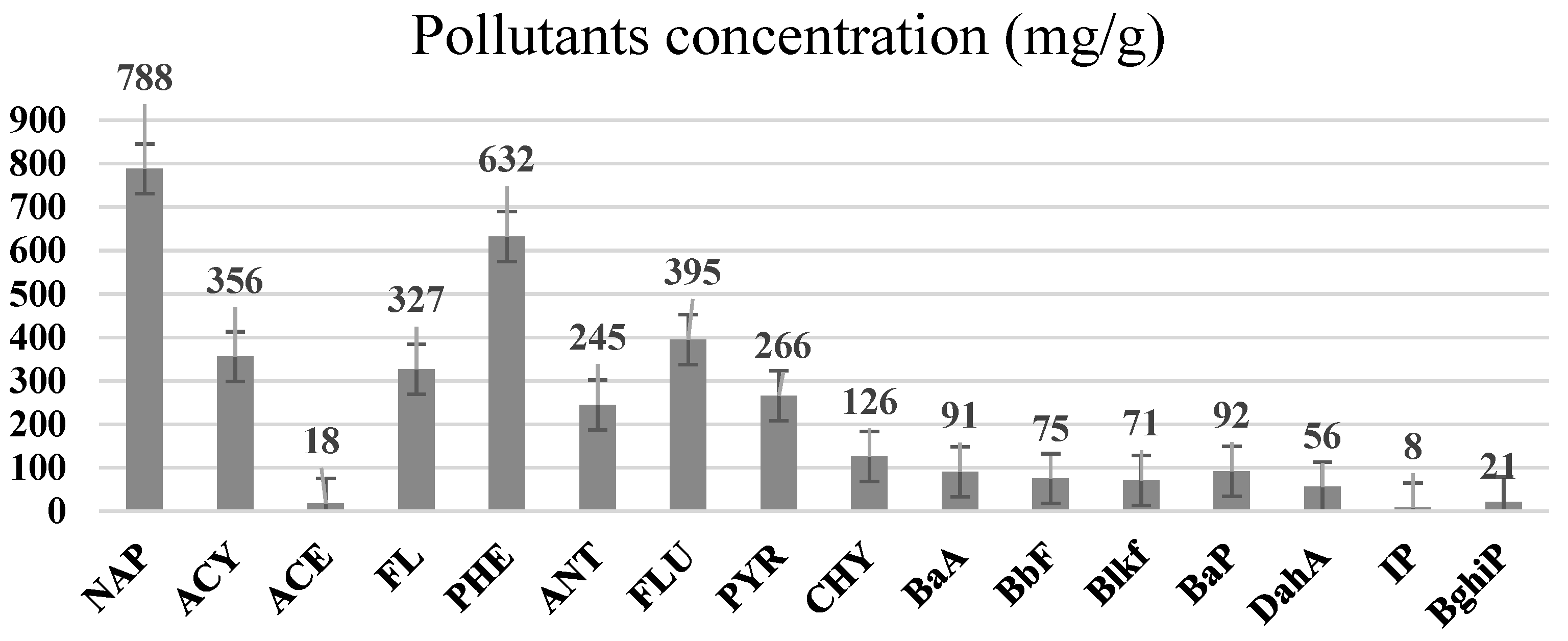
Figure 2 represents the PAHs content in the coal tar before the leaching process was conducted. It was found that, from the coal mass of 116.3 mg with 2 mL of hexane added as a solvent, when the PAHs were observed using the GC–MS technique, a total of 16 PAHs were detected. Naphthalene, followed by phenanthrene, fluoranthene and acenaphthylene, were found to be in the highest concentrations, compared to the remaining PAHs. The overall PAHs concentration, identified from the coal tar used, was found to be 2916.47 mg/L.
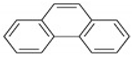
Naphthalene and phenanthrene were the simplest and most water-soluble molecules of the PAH group observed, and this serves as a model for the dissolution of the light hydroxylated aromatic compounds. The highest dissolution occurred in the acid mine drainage sample. The transfer kinetics of pollutants from the coal tar in the water was the highest at the lower molecular weights of PAHs. The experimental results of this study also showed ageing between the coal tar and the various water samples, resulting in the progressive release of PAHs. The deposit of coal tar in the acidic mine drainage should be given more attention, as PAHs were proven to be released most effectively with the acid mine drainage, which prompted the release of PAHs hazardous to the environment and to humans. According to Sharma and Lee [
20], naphthalene was found to be the most abundant PAH in the coal tar, which was also proven in the present study, as can be seen in
Figure 3.
According to Makelane et al. [
21], pyrene was found to be most concentrated in sewage wastewater, which was also proven in the present study.
All the results on the leaching process of the PAHs from the various water sources are tabulated in
Table 5,
Table 6 and
Table 7. A total of 16 PAHs were detected in the three water sources; however, acenaphthene was not detected in any of the three water sources used in the present study, albeit a concentration of 18.46 mg/L was found when the coal tar was analyzed. It was noted that the concentration of the PAHs increased with the ageing process. Ageing (or time) was noted to be a factor to be considered in the physiochemical conditions between the water source and the PAHs. The presence of the PAHs in the water source increases as ageing increases. The lower molecular weight PAHs, with two to three rings, were identified to be highly soluble in the acidic media and slightly soluble in the sewage wastewater, and less soluble in the alkaline media (
Table 6). Since PAHs are non-polar and have no charge, the electron pairs on the aromatic rings play a role in the physiochemical reaction of the PAHs and the water forming radicals’ reaction that triggered the PAHs to be released.
Table 5,
Table 6 and
Table 7 represent the experimental results obtained during the leaching process. It should also be noted that all experiments were conducted in triplicate.
Figure 4 represents the PAHs dissolution in various water sources and how effective a role Raoult’s law plays in the leaching process. The immersion of coal tar in the various water sources used, with their different physiochemistry, resulted in obtaining different PAHs values, as shown in
Figure 4. According to Boulangé, Lorgeoux, Biache, Michel, Michels and Faure [
3], there are factors such as temperature, ionic strength, pH and dissolved organic carbon. However, in this study, the pH and the dissolved organic carbon were considered in the leaching. There is a need to understand the driving forces of these factors:
The dissolved organic carbon concentration can be influenced by pH [
22]; the effect of pH does not influence the neutral organic compound. Neutral organic compounds contain functional groups such as hydroxyl, ether, ketone, lactone, aldehyde and ester because of the oxygen present in them, which can easily react with water to form hydrogen bonds. The presence of these functional groups increases the solubility in water [
3].
This study does agree with Boulangé, Lorgeoux, Biache, Michel, Michels and Faure [
3], as it labelled the nature of the dissolved PAHs as being acidic by having them dissolved or favored in an acidic mine drainage at a lower pH.
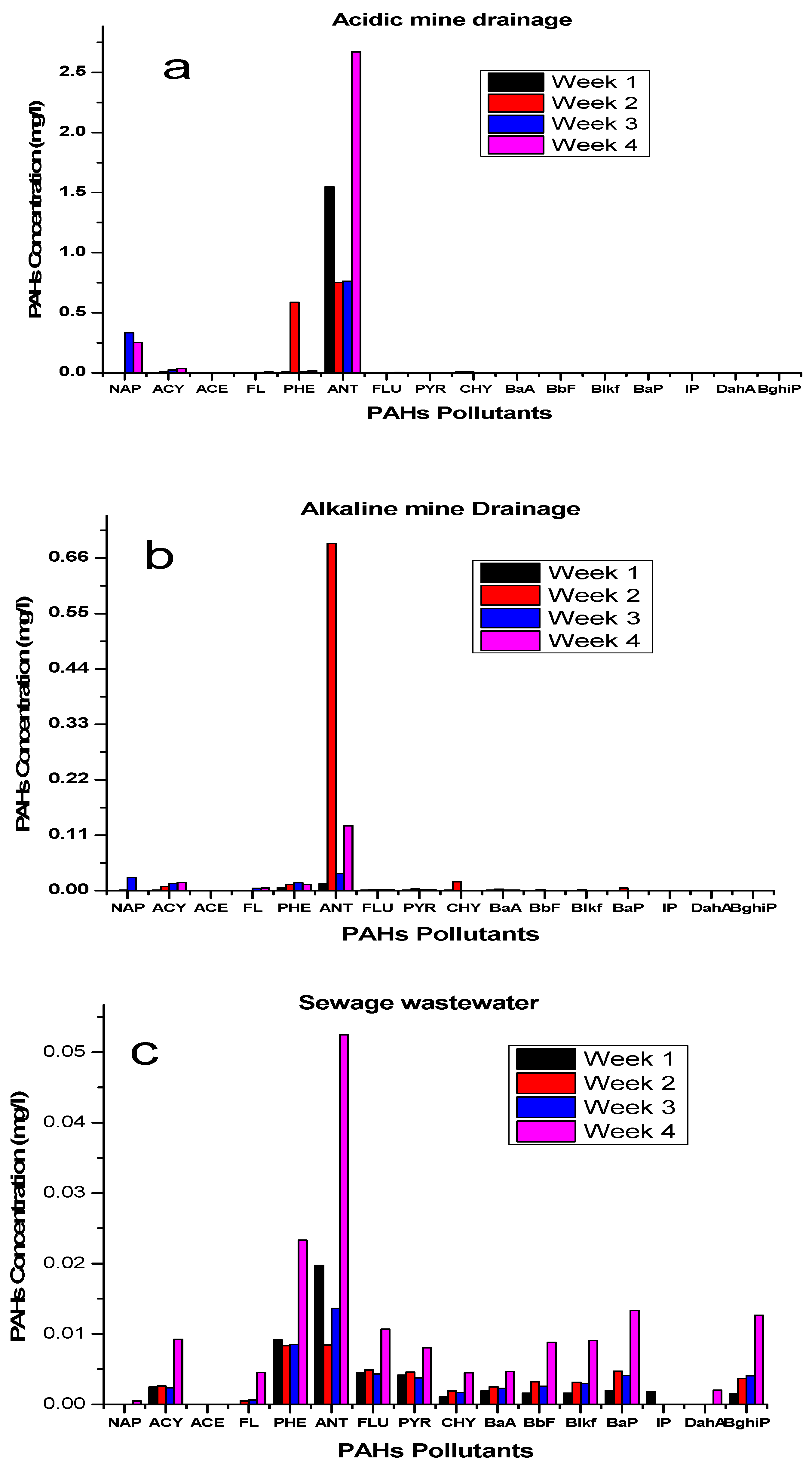
Influence of Ageing of Coal Tar/Various Water Sources Interphase
Figure 5 represents how ageing influenced the release of the different PAHs and the different water sources. The ageing phenomenon of the coal tar/water sources interphase was found to be an important aspect that has a significant consequence for the long-term fate of coal tar in various water sources, with an increase in the release of higher PAHs concentrations. During the leaching process, anthracene was highly dissolved in the acidic mine drainage and sewage wastewater at week 4. The ageing of PAHs in sewage wastewater agreed with Cai, Ding, Zhang, Wang, Wang, Ren and Dong [
14], who mentioned that the longer PAHs are in water, the more they reach an unacceptable level over time. Since time is directly proportional to ageing, as time increases, the PAHs concentration in the coal tar availability reduces, resulting in a greater dissolution.
Since coal is still currently being used as one of the most substantial sources of energy in SA, there is still a prediction of a large release of coal tar that also exhibits PAHs. An intensive consideration should be implemented to avoid coal tar being channeled into various waters. The processes involving the transformation of coal should be conducted far away from water sources, allowing ageing to occur between the by-product (coal tar) and the soil. In doing so, the exposure of PAHs on the surface will be no risk to humans, as it was reported by Patel et al. [
23] that the concentrations of 1 ng/L and 11 μg/L of PAHs in drinking water, as per the WHO regulations, are acceptable.
Since billions of rands have and still are being used on coal for energy consumption and production, millions of rands also play a part in remediating the PAHs from the environment and water.
4. Conclusions
In coal tar-contamination in various water sources, the main mechanism involved in the PAHs release is known to be dissolution; this is described by Raoult’s law, which states that the equilibrium concentration of a compound in a water source is a function of the compound’s water solubility and its molar fraction in the initial phase in contact with water. The acidic media was proven to have the highest release of PAHs, with a total concentration of 7.1 mg/L, followed by 1.2 mg/L for the sewage, and lastly, 0.32 mg/L for the alkaline mine drainage. Furthermore, it was found that the lower molecular weight PAHs were more soluble in the acidic water as it resulted in releasing a higher PAH concentration.
During the leaching on the acidic mine drainage, it was observed that a pH of 2.77 and dissolved organic carbon of 2.25 mg/L, resulted in the release of 7.1 mg/L of PAHs from the coal tar. The reaction of the π–π bonding also plays a vital role in the reaction of the aromatic compound of the PAHs with the acidic water molecules. PAHs happened to be more soluble in the acidic mine drainage compared to the alkaline, and slightly soluble in sewage depending on the pH and the dissolved organic carbon. Since PAHs are non-polar and only expected to be soluble in an aromatic’s solvent however, the pH and dissolved organic carbon are factors to be considered when anticipating the solubility of PAHs in wastewater and water sources.