The Relationship between Physical Activity and Long COVID: A Cross-Sectional Study
Highlights
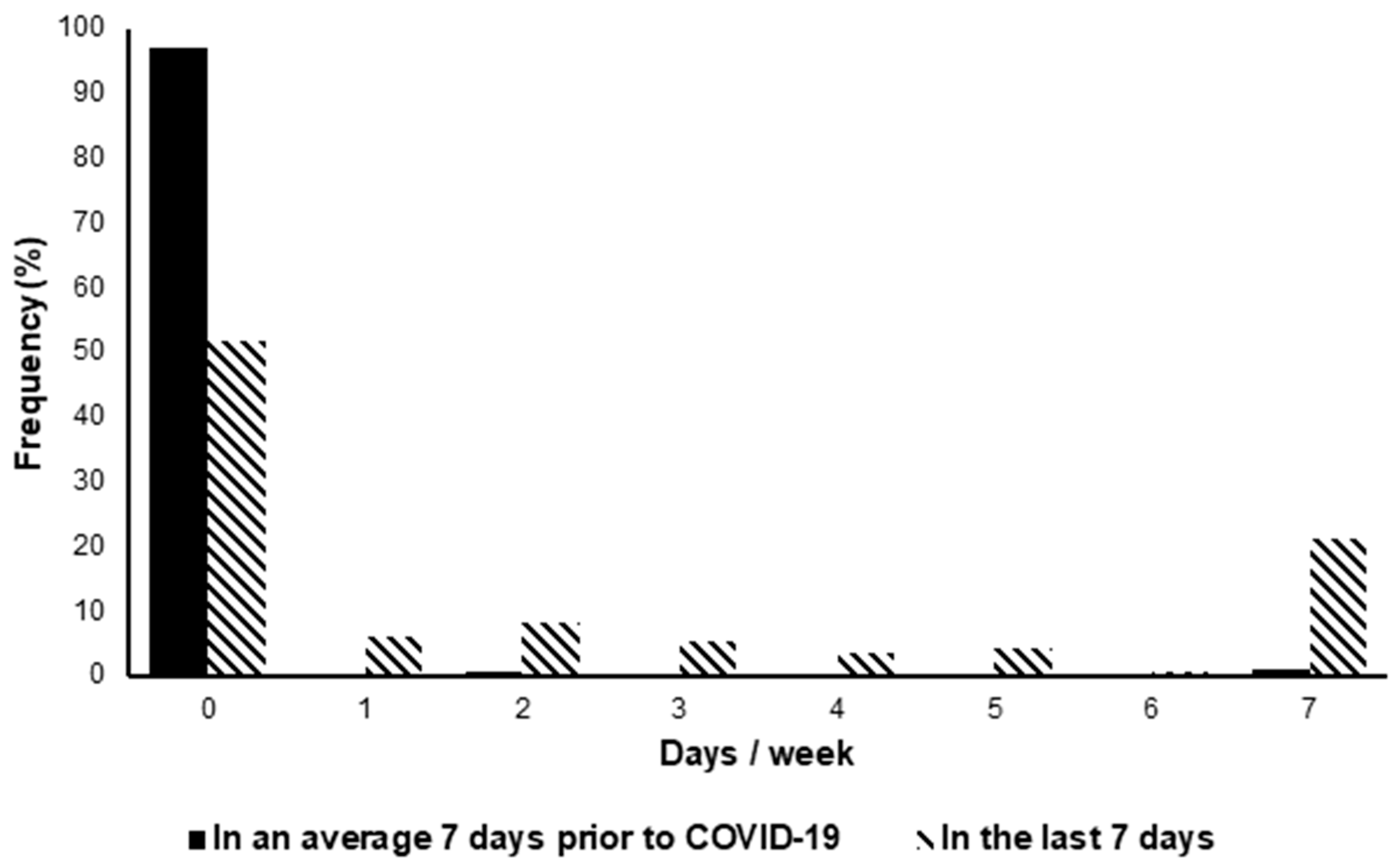
- Activity levels were significantly lower in individuals with LC post-COVID-19 compared to pre-COVID-19.
- Almost half of those with LC surveyed were no longer able to complete ADLs without assistance.
- Engaging in PA generally made LC symptoms worse; however, in a small minority, PA improves symptoms.
- There was inconsistent advice from healthcare professionals on how to be physically active.
- Clinicians should recognize that returning to pre-COVID PA levels may be challenging for many with LC.
- The priority should be placed on supporting patients and their families and restoring people's independence.
- Clinicians should take an individualized approach to recommending PA in those with LC, using principles of pacing.
- PA’s potential benefits should be balanced against adverse effects, including the worsening of LC symptoms.
- Policymakers need to consider the multitude of effects LC can have at both the individual and societal level.
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Procedures
2.3. Dependent Measures and Statistical Analyses
3. Results
4. Discussion
4.1. Physical Activity Patterns and Independence
4.2. PA’s Effect on LC
4.3. Recommendations by HCPs
4.4. Limitations
4.5. Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A. Summary Box
Appendix A.1. What Are the New Findings?
- Activity levels were significantly lower in individuals with LC post COVID-19 compared to pre COVID-19, with the average participant reporting 26 min of activity in the last week
- Only 8.13% of participants met the UK PA guidelines
- Almost half of those with LC surveyed in this study were no longer able to complete ADLs without assistance
- Engaging in PA generally make LC symptoms worse however, in a small minority PA improves symptoms
- There was inconsistent advice on physically active from healthcare professionals
Appendix A.2. How Might It Impact on Clinical Practice in the Future?
- Clinicians should recognise that return to pre-COVID PA levels may be challenging for many with LC and the priority should be that of supporting patients, their families and restoring patients independence
- Clinicians should take an individualised approach to recommending PA in those with LC, using principles of pacing, balancing its potential benefits against adverse effects, including the worsening of LC symptoms
- Policy makers need to consider the multitude of effects LC can have at both the individual and societal level as well as providing HCPs with clear, safe advice to care for their patients
References
- Davis, H.E.; Assaf, G.S.; McCorkell, L.; Wei, H.; Low, R.J.; Re’em, Y.; Redfield, S.; Austin, J.P.; Akrami, A. Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. eClinicalMedicine 2021, 38, 101019. [Google Scholar] [CrossRef] [PubMed]
- NICE. COVID-19 Rapid Guideline: Managing the Long-Term Effects of COVID-19 2020. Available online: https://www.nice.org.uk/guidance/NG188 (accessed on 26 July 2021).
- Ziauddeen, N.; Gurdasani, D.; O’Hara, M.E.; Hastie, C.; Roderick, P.; Yao, G.; Alwan, N.A. Characteristics of Long COVID: Findings from a social media survey. medRxiv 2021, 2021, 21253968. [Google Scholar]
- Whitaker, M.; Elliott, J.; Chadeau-Hyam, M.; Riley, S.; Darzi, A.; Cooke, G.; Ward, H.; Elliott, P. Persistent symptoms following SARS-CoV-2 in-fection in a random community sample of 508,707 people. medRxiv 2021, 2021, 21259452. [Google Scholar]
- NIHR. Living with COVID-19—Second Review 2021. Available online: https://evidence.nihr.ac.uk/themedreview/living-with-covid19-second-review/ (accessed on 15 August 2021).
- Katz, S. Assessing self-maintenance: Activities of daily living, mobility, and instrumental activities of daily living. J. Am. Geriatr. Soc. 1983, 31, 721–727. [Google Scholar] [CrossRef]
- Bowling, A.; Gabriel, Z.; Dykes, J.; Dowding, L.M.; Evans, O.; Fleissig, A.; Banister, D.; Sutton, S. Let’s ask them: A national survey of definitions of quality of life and its enhancement among people aged 65 and over. Int. J. Aging Hum. Dev. 2003, 56, 269–306. [Google Scholar] [CrossRef]
- Fiske, A.; Wetherell, J.L.; Gatz, M. Depression in Older Adults. Ann. Rev. Clin. Psychol. 2009, 5, 363–389. [Google Scholar] [CrossRef]
- Jimeno-Almazán, A.; Pallarés, J.; Buendía-Romero, Á.; Martínez-Cava, A.; Franco-López, F.; Martínez, B.S.-A.; Bernal-Morel, E.; Courel-Ibáñez, J. Post-COVID-19 Syndrome and the Potential Benefits of Exercise. Int. J. Environ. Res. Public Health 2021, 18, 5329. [Google Scholar] [CrossRef]
- Powell, K.E.; King, A.C.; Buchner, D.M.; Campbell, W.W.; DiPietro, L.; Erickson, K.I.; Hillman, C.H.; Jakicic, J.M.; Janz, K.F.; Katzmarzyk, P.T.; et al. The Scientific Foundation for the Physical Activity Guidelines for Americans, 2nd Edition. J. Phys. Act. Health 2018, 16, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Shuwa, H.A.; Shaw, T.N.; Knight, S.B.; Wemyss, K.; McClure, F.A.; Pearmain, L.; Prise, I.; Jagger, C.; Morgan, D.J.; Khan, S.; et al. Alterations in T and B cell function persist in convalescent COVID-19 patients. N. Y. Med. J. 2021, 2, 720–735.e4. [Google Scholar] [CrossRef]
- Campbell, J.P.; Turner, J.E. Debunking the Myth of Exercise-Induced Immune Suppression: Redefining the Impact of Exercise on Immunological Health Across the Lifespan. Front. Immunol. 2018, 9, 648. [Google Scholar] [CrossRef]
- Humphreys, H.; Kilby, L.; Kudiersky, N.; Copeland, R. Long COVID and the role of physical activity: A qualitative study. BMJ Open 2021, 11, e047632. [Google Scholar] [CrossRef] [PubMed]
- Salman, D.; Vishnubala, D.; Le Feuvre, P.; Beaney, T.; Korgaonkar, J.; Majeed, A.; McGregor, A.H. Returning to physical activity after COVID-19. BMJ 2021, 372, m4721. [Google Scholar] [CrossRef] [PubMed]
- Décary, S.; Gaboury, I.; Poirier, S.; Garcia, C.; Simpson, S.; Bull, M.; Brown, D.; Daigle, F. Humility and Acceptance: Working Within Our Limits With Long COVID and Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. J. Orthop. Sports Phys. Ther. 2021, 51, 197–200. [Google Scholar] [CrossRef] [PubMed]
- NICE. Chronic Fatigue Syndrome/Myalgic Encephalomyelitis (or Encephalopathy): Diagnosis and Management: NICE. 2007. Available online: https://www.nice.org.uk/guidance/cg53 (accessed on 26 July 2021).
- Geraghty, K.; Hann, M.; Kurtev, S. Myalgic encephalomyelitis/chronic fatigue syndrome patients’ reports of symptom changes following cognitive behavioural therapy, graded exercise therapy and pacing treatments: Analysis of a primary survey compared with secondary surveys. J. Health Psychol. 2019, 24, 1318–1333. [Google Scholar] [CrossRef]
- RCOT. How to Conserve Your Energy 2021. Available online: https://www.rcot.co.uk/conserving-energy (accessed on 28 July 2021).
- NHS. Your COVID Recovery 2021. Available online: http://yourcovidrecovery.nhs.uk (accessed on 2 August 2021).
- Moy, K.L.; Scragg, R.K.; McLean, G.; Carr, H. The New Zealand Physical Activity Questionnaires: Validation by heart-rate moni-toring in a multiethnic population. J. Phys. Act. Health 2008, 5 (Suppl. S1), S45–S61. [Google Scholar] [CrossRef]
- Maddison, R.; Ni Mhurchu, C.; Jiang, Y.; Vander Hoorn, S.; Rodgers, A.; Lawes, C.M.; Rush, E. International Physical Activity Ques-tionnaire (IPAQ) and New Zealand Physical Activity Questionnaire (NZPAQ): A doubly labelled water validation. Int. J. Behav. Nutr. Phys. Act. 2007, 4, 62. [Google Scholar] [CrossRef] [Green Version]
- Department of Health and Social Care. Physical Activity Guidelines: UK Chief Medical Officers’ Report 2019. Available online: https://www.gov.uk/government/publications/physical-activity-guidelines-uk-chief-medical-officers-report (accessed on 15 September 2021).
- Holtzman, C.S.; Bhatia, S.; Cotler, J.; Jason, L.A. Assessment of Post-Exertional Malaise (PEM) in Patients with Myalgic Encepha-lomyelitis (ME) and Chronic Fatigue Syndrome (CFS): A Patient-Driven Survey. Diagnostics 2019, 9, 26. [Google Scholar] [CrossRef] [Green Version]
- Tremblay, M.S.; Aubert, S.; Barnes, J.D.; Saunders, T.J.; Carson, V.; Latimer-Cheung, A.E.; Chastin, S.F.; Altenburg, T.M.; Chinapaw, M.J. Sedentary Behavior Research Network (SBRN)—Terminology Consensus Project process and outcome. Int. J. Behav. Nutr. Phys. Act. 2017, 14, 75. [Google Scholar] [CrossRef] [Green Version]
- Booth, F.W.; Roberts, C.K.; Laye, M.J. Lack of exercise is a major cause of chronic diseases. Compr. Physiol. 2012, 2, 1143–1211. [Google Scholar]
- Cunningham, C.; O’Sullivan, R.; Caserotti, P.; Tully, M.A. Consequences of physical inactivity in older adults: A systematic review of reviews and meta-analyses. Scand. J. Med. Sci. Sports 2020, 30, 816–827. [Google Scholar] [CrossRef]
- de Rezende, L.F.; Rodrigues Lopes, M.; Rey-López, J.P.; Matsudo, V.K.; Luiz Odo, C. Sedentary behavior and health outcomes: An overview of systematic reviews. PLoS ONE 2014, 9, e105620. [Google Scholar] [CrossRef] [PubMed]
- John Hopkins University. COVID-19 Map: JohnsHopkins. 2021. Available online: https://coronavirus.jhu.edu/map.html (accessed on 12 August 2021).
- Rajan, S.; Khunti, K.; Alwan, N.; Steves, C.; MacDermott, N.; Morsella, A.; Angulo, E.; Winkelmann, J.; Bryndová, L.; Fronteira, I.; et al. In the Wake of the Pandemic: Preparing for Long COVID; European Observatory Policy Briefs; European Observatory on Health Systems and Policies: Copenhagen, Denmark, 2021. [Google Scholar]
- WHO. Support for Rehabilitation: Self-Management after COVID-19-Related Illness: World Health Organization. 2021. Available online: https://www.euro.who.int/en/health-topics/health-emergencies/coronavirus-covid-19/publications-and-technical-guidance/2020/support-for-rehabilitation-self-management-after-covid-19-related-illness-2020-produced-by-whoeurope (accessed on 27 September 2021).
- Blomberg, B.; Mohn, K.G.-I.; Brokstad, K.A.; Zhou, F.; Linchausen, D.W.; Hansen, B.-A.; Lartey, S.; Onyango, T.B.; Kuwelker, K.; Sævik, M.; et al. Long COVID in a prospective cohort of home-isolated patients. Nat. Med. 2021, 27, 1607–1613. [Google Scholar] [CrossRef] [PubMed]




| Characteristic | Total Sample (n = 477) |
|---|---|
| Age (years), mean (SD) | 45.69 (10.02) |
| Gender (female), n (%) | 425 (89.10) |
| BMI, median (IQR) | 25.71 (22.51, 30.47) |
| Ethnicity, n (%) | |
| White (British, Irish, Irish Traveller or other White backgrounds) | 442 (92.70) |
| Black (African, Caribbean or other Black backgrounds) | 3 (0.60) |
| Asian (Indian, Pakistani, Bangladeshi, Chinese or other Asian backgrounds) | 18 (3.80) |
| Mixed (White and Asian, White and Black African, White and Black Caribbean, Other) | 7 (1.50) |
| Other | 4 (0.80) |
| Country, n (%) | |
| England | 358 (78.34) |
| Scotland | 43 (9.41) |
| Wales | 26 (5.69) |
| USA | 11 (2.41) |
| Canada | 5 (1.09) |
| Northern Ireland | 5 (1.09) |
| Ireland | 3 (0.66) |
| Finland | 2 (0.44) |
| France | 1 (0.22) |
| India | 1 (0.22) |
| Netherlands | 1 (0.22) |
| Sweden | 1 (0.22) |
| Number of LC symptoms, median (IQR) | 11 (8,14) |
| Time since COVID-19 symptom onset (months), n (%) | |
| 0–6 | 132 (27.67) |
| 6–12 | 91 (19.08) |
| 12–18 | 247 (51.78) |
| Method of COVID-19 diagnosis, n (%) | |
| PCR test | 226 (47.4) |
| Antibody test | 50 (10.5) |
| Based on symptoms alone (including retrospectively) | 177 (37.1) |
| No testing available at the time | 12 (2.5) |
| Other | 7 (1.5) |
| Co-morbidities prior to LC, n (%) | |
| Allergies * | 12 (2.5) |
| Autoimmune diseases | 42 (8.8) |
| Cardiovascular disease | 20 (4.2) |
| Chronic neurological conditions | 10 (2.1) |
| Chronic pain | 13 (2.7) |
| Chronic respiratory conditions | 94 (19.7) |
| Diabetes (type 1 or 2) | 17 (3.6) |
| Mental health ** | 12 (2.5) |
| Migraines | 10 (2.1) |
| No diagnosed co-morbidities | 230 (48.2) |
| Osteoarthritis | 11 (2.3) |
| Other (any co-morbidity with a frequency of <2%) | 64 (13.4) |
| Unspecified hypo or hyperthyroidism | 12 (2.5) |
| F | df | p | η2 | |
|---|---|---|---|---|
| ADLs | ||||
| Pre-post | 225.97 | 1467 | <0.001 | 0.32 |
| Pre-post × LC duration | 0.16 | 2467 | 0.86 | 0.01 |
| LC duration Ϯ | 0.20 | 1467 | 0.82 | 0.00 |
| PA | ||||
| Pre-post | 286.31 | 1467 | <0.001 | 0.38 |
| Pre-post × LC duration | 0.11 | 2467 | 0.89 | 0.00 |
| Intensity | 51.67 | 1.61, 751.29 | <0.001 | 0.10 |
| Intensity × LC duration | 1.83 | 3.22, 751.29 | 0.14 | 0.01 |
| Pre-post × intensity | 36.85 | 1.72, 802.36 | <0.001 | 0.07 |
| Pre-post × intensity × LC duration | 0.61 | 3.44, 802.36 | 0.63 | 0.00 |
| LC duration Ϯ | 0.13 | 2467 | 0.88 | 0.00 |
| Intensity | Minutes per Week, Mean (SD) | Mean Difference (95% CI) | Paired t-Test, p Value | |
|---|---|---|---|---|
| Pre-COVID-19 Baseline | In the Last 7 Days | |||
| Brisk walking | 418.56 (507.18) | 24.33 (62.76) | 394.23 (348.67–439.8) | <0.001 |
| MPA | 480.11 (715.06) | 52.19 (195.17) | 427.92 (366.11–489.74) | <0.001 |
| VPA | 186.36 (316.36) | 4.11 (22.95) | 182.25 (153.84–210.65) | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wright, J.; Astill, S.L.; Sivan, M. The Relationship between Physical Activity and Long COVID: A Cross-Sectional Study. Int. J. Environ. Res. Public Health 2022, 19, 5093. https://doi.org/10.3390/ijerph19095093
Wright J, Astill SL, Sivan M. The Relationship between Physical Activity and Long COVID: A Cross-Sectional Study. International Journal of Environmental Research and Public Health. 2022; 19(9):5093. https://doi.org/10.3390/ijerph19095093
Chicago/Turabian StyleWright, Jack, Sarah L. Astill, and Manoj Sivan. 2022. "The Relationship between Physical Activity and Long COVID: A Cross-Sectional Study" International Journal of Environmental Research and Public Health 19, no. 9: 5093. https://doi.org/10.3390/ijerph19095093
APA StyleWright, J., Astill, S. L., & Sivan, M. (2022). The Relationship between Physical Activity and Long COVID: A Cross-Sectional Study. International Journal of Environmental Research and Public Health, 19(9), 5093. https://doi.org/10.3390/ijerph19095093








