Status of Oxidative Stress during Low-Risk Labour: Preliminary Data
Abstract
1. Introduction
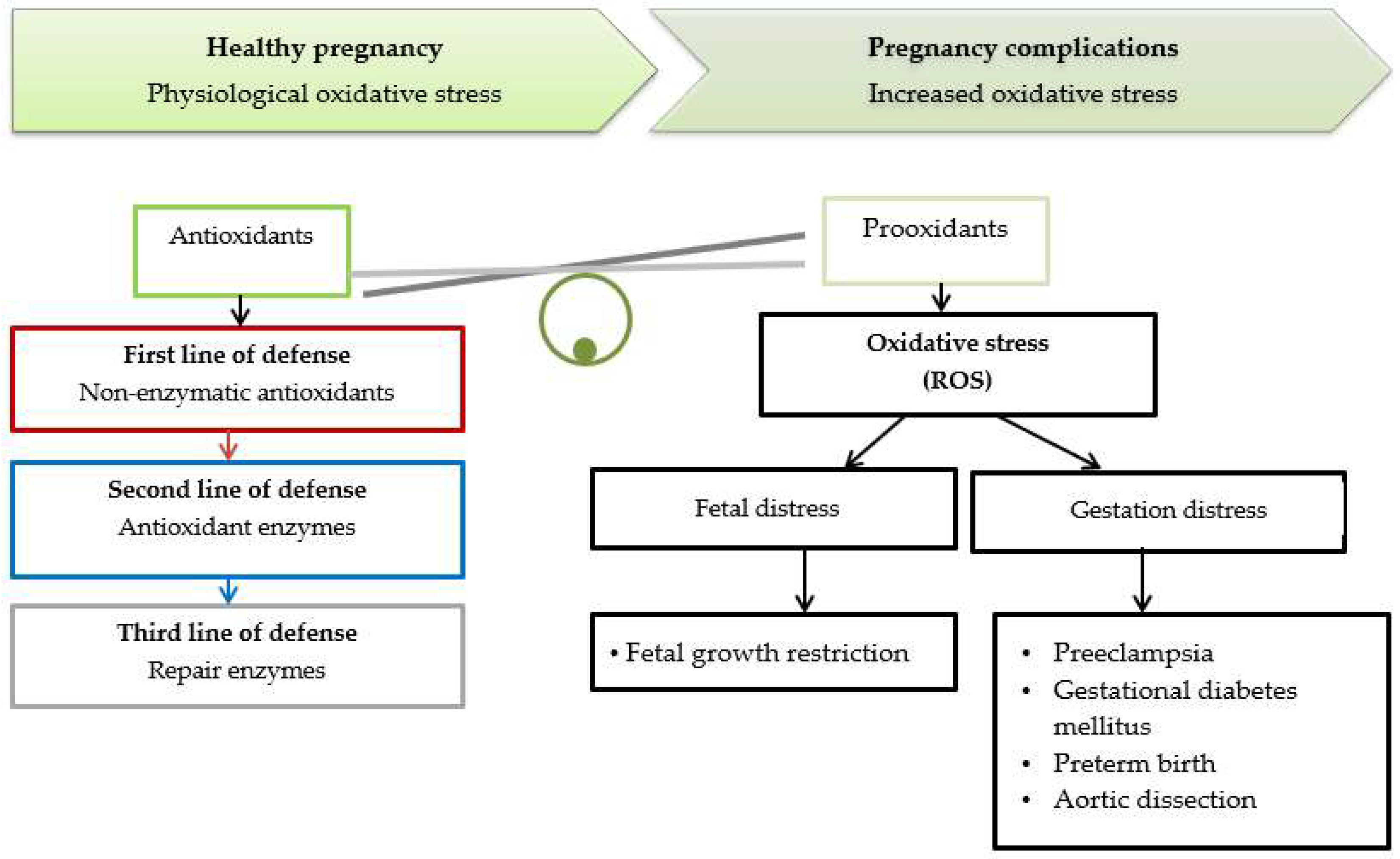
1.1. Oxidative Stress during Pregnancy and Childbirth
1.2. Selected Antioxidative Mechanisms
1.3. Aim
2. Materials and Methods
2.1. Design
2.2. Data Collection
2.3. Biological Material and Its Preparation for Biochemical Analyses
2.4. Oxidative Stress Analysis
2.4.1. Total Antioxidant Status (TAS)
2.4.2. Glutathione Peroxidase (GPx)
2.4.3. Superoxide Dismutase (SOD)
2.5. Analysis of Selected Cofactors of the Oxidation-Reduction Reactions
2.6. Data Analysis
- -
- dependent: mother’s venous blood, umbilical cord blood,
- -
- independent: total antioxidant potential (TAS), the activity of superoxide dismutase (SOD), the concentration of SOD cofactors (Mn, Cu and Zn), the activity of glutathione peroxidase (GPx).
2.7. Ethical Statement
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jiang, X.; Bar, H.Y.; Yan, J.; West, A.A.; Perry, C.A.; Malysheva, O.V.; Devapatla, S.; Pressman, E.; Vermeylen, F.M.; Wells, M.T.; et al. Pregnancy induces transcriptional activation of the peripheral innate immune system and increases oxidative DNA damage among healthy third trimester pregnant women. PLoS ONE 2012, 7, e46736. [Google Scholar] [CrossRef]
- Mistry, H.D.; Williams, P.J. The importance of antioxidant micronutrients in pregnancy. Oxid. Med. Cell. Longev. 2011, 2011, 841749. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Aranguren, L.; Nadeem, S. Bioenergetics adaptations and redox homeostasis in pregnancy and related disorders. Mol. Cell. Biochem. 2021, 476, 4003–4018. [Google Scholar] [CrossRef] [PubMed]
- Furness, D.L.; Dekker, G.A.; Roberts, C.T. DNA damage and health in pregnancy. J. Reprod. Immunol. 2011, 89, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Aponte-Mellado, A.; Premkumar, B.J.; Shaman, A.; Gupta, S. The effects of oxidative stress on female reproduction: A review. Reprod. Biol. Endocrinol. 2012, 29, 10–49. [Google Scholar] [CrossRef]
- Moore, T.A.; Ahmad, I.M.; Zimmerman, M.C. Oxidative Stress and Preterm Birth: An Integrative Review. Biol. Res. Nurs. 2018, 20, 497–512. [Google Scholar] [CrossRef]
- de Lucca, L.; Jantsch, L.B.; Vendrame, S.A.; Stein, C.D.S.; Klein, V.C.G.; Soares, K.B.; Gallarreta, F.M.P.; Moresco, R.N.; Gonçalves, T.L.G. Longitudinal Study of Delta-Aminolevulinate Dehydratase Activity and Oxidative Profile in Healthy Pregnant Women. Biomolecules 2019, 9, 18. [Google Scholar] [CrossRef]
- Leslie, K.; Whitley, G.S.; Herse, F.; Dechend, R.; Ashton, S.V.; Laing, K.; Thilaganathan, B.; Cartwright, J.E. Increased apoptosis, altered oxygen signaling, and antioxidant defenses in first-trimester pregnancies with high-resistance uterine artery blood flow. Am. J. Pathol. 2015, 185, 2731–2741. [Google Scholar] [CrossRef]
- Holland, O.J.; Hickey, A.J.R.; Alvsaker, A.; Moran, S.; Hedges, C.; Chamley, L.W.; Perkins, A.V. Changes in mitochondrial respiration in the human placenta over gestation. Placenta 2017, 57, 102–112. [Google Scholar] [CrossRef]
- Hussain, T.; Murtaza, G.; Metwally, E.; Kalhoro, D.H.; Kalhoro, M.S.; Rahu, B.A.; Sahito, R.G.A.; Yin, Y.; Yang, H.; Chughtai, M.I.; et al. The Role of Oxidative Stress and Antioxidant Balance in Pregnancy. Mediat. Inflamm. 2021, 2021, 9962860. [Google Scholar] [CrossRef]
- Chiarello, D.I.; Abad, C.; Rojas, D.; Toledo, F.; Vázquez, C.M.; Mate, A.; Sobrevia, L.; Marín, R. Oxidative stress: Normal pregnancy versus preeclampsia. Biochim. Biophys. Acta. Mol. Basis Dis. 2020, 1866, 165354. [Google Scholar] [CrossRef] [PubMed]
- Kota, S.K.; Gayatri, K.; Jammula, S.; Kota, S.K.; Krishna, S.V.; Meher, L.K.; Modi, K.D. Endocrinology of parturition. Indian. J. Endocrinol. Metab. 2013, 17, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, D.; Honnens, A.; Piechotta, M.; Lüttgenau, J.; Niemann, H.; Rath, D.; Bollwein, H. Effects of a protracted induction of parturition on the incidence of retained placenta and assessment of uterine artery blood flow as a measure of placental maturation in cattle. Theriogenology 2013, 80, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Alanazi, R.; Alotaibi, M.; Djouhri, L. In vitro effects of hydrogen peroxide on rat uterine contraction before and during pregnancy. Croat. Med. J. 2018, 59, 327–334. [Google Scholar] [CrossRef]
- Fragoso, M.B.T.; Ferreira, R.C.; Tenório, M.C.D.S.; Moura, F.A.; de Araújo, O.R.P.; Bueno, N.B.; Goulart, M.O.F.; de Oliveira, A.C.M. Biomarkers of Inflammation and Redox Imbalance in Umbilical Cord in Pregnancies with and without Preeclampsia and Consequent Perinatal Outcomes. Oxid. Med. Cell. Longev. 2021, 2021, 9970627. [Google Scholar] [CrossRef]
- Jîtcă, G.; Ősz, B.E.; Tero-Vescan, A.; Miklos, A.P.; Rusz, C.M.; Bătrînu, M.G.; Vari, C.E. Positive Aspects of Oxidative Stress at Different Levels of the Human Body: A Review. Antioxidants 2022, 11, 572. [Google Scholar] [CrossRef]
- Lu, J.; Wang, Z.; Cao, J.; Chen, Y.; Dong, Y. A novel and compact review on the role of oxidative stress in female reproduction. Reprod. Biol. Endocrinol. 2018, 16, 80. [Google Scholar] [CrossRef]
- Joo, E.H.; Kim, Y.R.; Kim, N.; Jung, J.E.; Han, S.H.; Cho, H.Y. Effect of Endogenic and Exogenic Oxidative Stress Triggers on Adverse Pregnancy Outcomes: Preeclampsia, Fetal Growth Restriction, Gestational Diabetes Mellitus and Preterm Birth. Int. J. Mol. Sci. 2021, 22, 10122. [Google Scholar] [CrossRef]
- Aquila, I.; Sacco, M.A.; Cordasco, F.; Ricci, P. Forensic case of a pregnant woman with Marfan syndrome. BMJ Case Rep. 2020, 13, e229959. [Google Scholar] [CrossRef]
- Cheng, X.; Chapple, S.J.; Patel, B.; Puszyk, W.; Sugden, D.; Yin, X.; Mayr, M.; Siow, R.C.; Mann, G.E. Gestational diabetes mellitus impairs Nrf2-mediated adaptive antioxidant defenses and redox signaling in fetal endothelial cells in utero. Diabetes 2013, 62, 4088–4097. [Google Scholar] [CrossRef]
- Zuo, J.; Zhang, Z.; Luo, M.; Zhou, L.; Nice, E.C.; Zhang, W.; Wang, C.; Huang, C. Redox signaling at the crossroads of human health and disease. MedComm 2022, 3, e127. [Google Scholar] [CrossRef] [PubMed]
- Oyewole, A.O.; Birch-Machin, M.A. Mitochondria-targeted antioxidants. FASEB J. 2015, 29, 4766–4771. [Google Scholar] [CrossRef] [PubMed]
- Spanidis, Y.; Veskoukis, A.S.; Papanikolaou, C.; Stagos, D.; Priftis, A.; Deli, C.K.; Jamurtas, A.Z.; Kouretas, D. Exercise-Induced Reductive Stress Is a Protective Mechanism against Oxidative Stress in Peripheral Blood Mononuclear Cells. Oxid. Med. Cell. Longev. 2018, 2018, 3053704. [Google Scholar] [CrossRef] [PubMed]
- Poggi, C.; Dani, C. Antioxidant strategies and respiratory disease of the preterm newborn: An update. Oxid. Med. Cell. Longev. 2014, 2014, 721043. [Google Scholar] [CrossRef] [PubMed]
- Aschner, J.L.; Aschner, M. Nutritional aspects of manganese homeostasis. Mol. Asp. Med. 2005, 26, 353–362. [Google Scholar] [CrossRef]
- Michaelis, V.; Aengenheister, L.; Tuchtenhagen, M.; Rinklebe, J.; Ebert, F.; Schwerdtle, T.; Buerki-Thurnherr, T.; Bornhorst, J. Differences and Interactions in Placental Manganese and Iron Transfer across an In Vitro Model of Human Villous Trophoblasts. Int. J. Mol. Sci. 2022, 23, 3296. [Google Scholar] [CrossRef]
- Bocca, B.; Ciccarelli, S.; Agostino, R.; Alimonti, A. Trace elements, oxidative status and antioxidant capacity as biomarkers in very low birth weight infants. Environ. Res. 2017, 156, 705–713. [Google Scholar] [CrossRef]
- Franco, M.C.; Kawamoto, E.M.; Gorjão, R.; Rastelli, V.M.; Curi, R.; Scavone, C.; Sawaya, A.L.; Fortes, Z.B.; Sesso, R. Biomarkers of oxidative stress and antioxidant status in children born small for gestational age: Evidence of lipid peroxidation. Pediatr. Res. 2007, 62, 204–208. [Google Scholar] [CrossRef]
- Willekens, J.; Runnels, L.W. Impact of Zinc Transport Mechanisms on Embryonic and Brain Development. Nutrients 2022, 14, 2526. [Google Scholar] [CrossRef]
- Linder, M.C. Ceruloplasmin and other copper binding components of blood plasma and their functions: An update. Metallomics 2016, 8, 887–905. [Google Scholar] [CrossRef]
- Nishi, H.; Inagi, R.; Kato, H.; Tanemoto, M.; Kojima, I.; Son, D.; Fujita, T.; Nangaku, M. Hemoglobin is expressed by mesangial cells and reduces oxidant stress. J. Am. Soc. Nephrol. 2008, 19, 1500–1508. [Google Scholar] [CrossRef] [PubMed]
- Kapralov, A.; Vlasova, I.I.; Feng, W.; Maeda, A.; Walson, K.; Tyurin, V.A.; Huang, Z.; Aneja, R.K.; Carcillo, J.; Bayir, H.; et al. Peroxidase activity of hemoglobin-haptoglobin complexes: Covalent aggregation and oxidative stress in plasma and macrophages. J. Biol. Chem. 2009, 284, 30395–30407. [Google Scholar] [CrossRef] [PubMed]
- Simon-Szabo, Z.; Fogarasi, E.; Nemes-Nagy, E.; Denes, L.; Croitoru, M.; Szabo, B. Oxidative stress and peripartum outcomes (Review). Exp. Ther. Med. 2021, 22, 771. [Google Scholar] [CrossRef] [PubMed]
- Farhat, Z.; Browne, R.W.; Bonner, M.R.; Tian, L.; Deng, F.; Swanson, M.; Mu, L. How do glutathione antioxidant enzymes and total antioxidant status respond to air pollution exposure? Environ. Int. 2018, 112, 287–293. [Google Scholar] [CrossRef]
- Paglia, D.E.; Valentine, W.N. Studies on the quantitative and qualitative characterisation of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 1967, 70, 158–169. [Google Scholar]
- Lotfollahi, H.; Mohammadi, M.; Ghaffari, S.; Badalzadeh, R.; Sohrabi, B.; Aslanabadi, N.; Separham, A.; Golmohammadi, A.; Abbasnejad, A.; Roshani, M. Effect of remote ischemic post-conditioning on oxidative stress in blood of STEMI patients treated with primary angioplasty. J. Cardiovasc. Thorac. Res. 2016, 8, 113–118. [Google Scholar] [CrossRef]
- Alirezaei, M.; Rezaei, M.; Hajighahramani, S.; Sookhtehzari, A.; Kiani, K. Oleuropein attenuates cognitive dysfunction and oxidative stress induced by some anesthetic drugs in the hippocampal area of rats. J. Physiol. Sci. 2017, 67, 131–139. [Google Scholar] [CrossRef]
- García-Rodríguez, C.E.; Mesa, M.D.; Olza, J.; Vlachava, M.; Kremmyda, L.S.; Diaper, N.D.; Noakes, P.S.; Miles, E.A.; Ramírez-Tortosa, M.C.; Liaset, B.; et al. Does consumption of two portions of salmon per week enhance the antioxidant defense system in pregnant women? Antioxid. Redox Signal. 2012, 16, 1401–1406. [Google Scholar] [CrossRef]
- Schlesier, K.; Harwat, M.; Böhm, V.; Bitsch, R. Assessment of antioxidant activity by using different in vitro methods. Free Radic. Res. 2002, 36, 177–187. [Google Scholar] [CrossRef]
- Toboła-Wróbel, K.; Pietryga, M.; Dydowicz, P.; Napierała, M.; Brązert, J.; Florek, E. Association of Oxidative Stress on Pregnancy. Oxid. Med. Cell. Longev. 2020, 2020, 6398520. [Google Scholar] [CrossRef]
- Buhimschi, I.A.; Buhimschi, C.S.; Pupkin, M.; Weiner, C.P. Beneficial impact of term labor: Nonenzymatic antioxidant reserve in the human fetus. Am. J. Obstet. Gynecol. 2003, 189, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Chełchowska, M.; Gajewska, J.; Ambroszkiewicz, J.; Mazur, J.; Ołtarzewski, M.; Maciejewski, T.M. Influence of Oxidative Stress Generated by Smoking during Pregnancy on Glutathione Status in Mother-Newborn Pairs. Antioxidants 2021, 10, 1866. [Google Scholar] [CrossRef] [PubMed]
- Sobrevia, L. Membrane transporters and receptors in pregnancy metabolic complications. Biochim. Biophys. Acta. Mol. Basis Dis. 2020, 1866, 165617. [Google Scholar] [CrossRef] [PubMed]
- Suhail, M.; Suhail, S.; Gupta, B.K.; Bharat, V. Malondialdehyde and Antioxidant Enzymes in Maternal and Cord Blood, and their Correlation in Normotensive and Preeclamptic Women. J. Clin. Med. Res. 2009, 1, 150–157. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Aydogan, U.; Durmaz, E.; Ercan, C.M.; Eken, A.; Ulutas, O.K.; Kavuk, S.; Gursel, O.; Alanbay, I.; Akay, C.; Kurekci, A.E.; et al. Effects of smoking during pregnancy on DNA damage and ROS level consequences in maternal and newborns’ blood. Arh. Hig. Rada Toksikol. 2013, 64, 35–46. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fall, C.H.; Yajnik, C.S.; Rao, S.; Davies, A.A.; Brown, N.; Farrant, H.J. Micronutrients and fetal growth. J. Nutr. 2003, 133 (5 Suppl. 2), 1747S–1756S. [Google Scholar] [CrossRef]
- Chiudzu, G.; Choko, A.T.; Maluwa, A. Pregnancy Are Associated with Risk of Spontaneous Preterm Birth: A Case-Control Study from Malawi. J. Pregnancy 2020, 2020, 9435972. [Google Scholar] [CrossRef]
- Srivastava, S.; Mehrotra, P.K.; Srivastava, S.P.; Siddiqui, M.K. Some essential elements in maternal and cord blood in relation to birth weight and gestational age of the baby. Biol. Trace Elem. Res. 2002, 86, 97–105. [Google Scholar]
- Takser, L.; Lafond, J.; Bouchard, M.; St-Amour, G.; Mergler, D. Manganese levels during pregnancy and at birh: Relation to environmental factors and smoking in a Southwest Quebec population. Environ. Res. 2004, 95, 119–125. [Google Scholar] [CrossRef]
- Yazbeck, C.; Moreau, T.; Sahuquillo, J.; Takser, L.; Huel, G. Effect of maternal manganese blood levels on erythrocyte calcium-pump activity in newborns. Sci. Total Environ. 2006, 354, 28–34. [Google Scholar] [CrossRef]
- Krachler, M.; Rossipal, E.; Micetic-Turk, D. Trace element transfer from the mother to the newborn--investigations on triplets of colostrum, maternal and umbilical cord sera. Eur. J. Clin. Nutr. 1999, 53, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Nandakumaran, M.; Al-Sannan, B.; Al-Sarraf, H.; Al-Shammari, M. Maternal-fetal transport kinetics of manganese in perfused human placental lobule in vitro. J. Matern. Fetal Neonatal Med. 2016, 29, 274–278. [Google Scholar] [CrossRef] [PubMed]
- Terrin, G.; Berni Canani, R.; Di Chiara, M.; Pietravalle, A.; Aleandri, V.; Conte, F.; De Curtis, M. Zinc in Early Life: A Key Element in the Fetus and Preterm Neonate. Nutrients 2015, 7, 10427–10446. [Google Scholar] [CrossRef] [PubMed]
- Irwinda, R.; Wibowo, N.; Putri, A.S. The Concentration of Micronutrients and Heavy Metals in Maternal Serum, Placenta, and Cord Blood: A Cross-Sectional Study in Preterm Birth. J. Pregnancy 2019, 2019, 5062365. [Google Scholar] [CrossRef]
- Bocca, B.; Madeddu, R.; Asara, Y.; Tolu, P.; Marchal, J.A.; Forte, G. Assessment of reference ranges for blood Cu, Mn, Se and Zn in a selected Italian population. J. Trace Elem. Med. Biol. 2011, 25, 19–26. [Google Scholar] [CrossRef]
- Komarova, T.; McKeating, D.; Perkins, A.V.; Tinggi, U. Trace Element Analysis in Whole Blood and Plasma for Reference Levels in a Selected Queensland Population, Australia. Int. J. Environ. Res. Public Health. 2021, 18, 2652. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, Y.; Piao, J.; Mao, D.; Li, Y.; Li, W.; Yang, L.; Yang, X. Reference Values of 14 Serum Trace Elements for Pregnant Chinese Women: A Cross-Sectional Study in the China Nutrition and Health Survey 2010–2012. Nutrients 2017, 9, 309. [Google Scholar] [CrossRef]
- Kot, K.; Łanocha-Arendarczyk, N.; Kupnicka, P.; Szymański, S.; Malinowski, W.; Kalisińska, E.; Chlubek, D.; Kosik-Bogacka, D. Selected Metal Concentration in Maternal and Cord Blood. Int. J. Environ. Res. Public Health 2021, 18, 12407. [Google Scholar] [CrossRef]
- Huang, S.H.; Weng, K.P.; Lin, C.C.; Wang, C.C.; Lee, C.T.; Ger, L.P.; Wu, M.T. Maternal and umbilical cord blood levels of mercury, manganese, iron, and copper in southern Taiwan: A cross-sectional study. J. Chin. Med. Assoc. 2017, 80, 442–451. [Google Scholar] [CrossRef]
- Pastore, A.; Federici, G.; Bertini, E.; Piemonte, F. Analysis of glutathione: Implication in redox and detoxifi cation. Clin. Chim. Acta 2003, 333, 19–39. [Google Scholar] [CrossRef]
- Wu, G.; Fang, Y.Z.; Yang, S.; Lupton, J.R.; Turner, N.D. Glutathione metabolism and its implications for health. J. Nutr. 2004, 134, 489–492. [Google Scholar] [CrossRef] [PubMed]
- Nagababu, E.; Chrest, F.J.; Rifkind, J.M. Hydrogen-peroxide-inducedheme degradation in red blood cells: The protective roles of catalase and glutathione peroxidase. Biochim. Biophys. Acta 2003, 16201, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.N.; Matharoo-Ball, B.; Shaw, R.W. Antioxidant enzyme expression, lipid peroxidation, and protein oxidation in human myometrium with parturition. Reprod. Sci. 2010, 17, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Abiaka, C.; Machado, L. Nitric oxide and antioxidant enzymes in venous and cord blood of late preterm and term omani mothers. Sultan Qaboos Univ. Med. J. 2012, 12, 300–305. [Google Scholar] [CrossRef]
- Wilking, M.; Ndiaye, M.; Mukhtar, H.; Ahmad, N. Circadian rhythm connections to oxidative stress: Implications for human health. Antioxid. Redox Signal. 2013, 19, 192–208. [Google Scholar] [CrossRef]
- Stritesky Larssen, K.; Lyberg, T. Oxidative status--age- and circadian variations?--a study in leukocytes/plasma. Neuro Endocrinol. Lett. 2006, 27, 445–452. [Google Scholar]
- Eick, S.M.; Geiger, S.D.; Alshawabkeh, A.; Aung, M.; Barrett, E.S.; Bush, N.; Carroll, K.N.; Cordero, J.F.; Goin, D.E.; Ferguson, K.K.; et al. Urinary oxidative stress biomarkers are associated with preterm birth: An ECHO program study. Am. J. Obstet. Gynecol. 2022, S0002-9378(22), 02170–021706. [Google Scholar] [CrossRef]
- Yang, J.; Chu, M.; Gong, C.; Gong, X.; Han, B.; Chen, L.; Wang, J.; Bai, Z.; Zhang, Y. Ambient fine particulate matter exposures and oxidative protein damage in early pregnant women. Environ. Pollut. 2022, 316 Pt 2, 120604. [Google Scholar] [CrossRef]
| Parameters | Mother’s Venous Blood (n = 168) | Umbilical Cord Blood (n = 168) | p Value | ||||
|---|---|---|---|---|---|---|---|
| ± SD | Median | Min-Max | ± SD | Median | Min-Max | ||
| TAS [mmol/L] | 1.65 ± 0.54 | 1.66 | 0.53–3.30 | 1.78 ± 0.37 | 1.76 | 0.65–4.00 | 0.034 |
| SOD [U/mL] | 205.02 ± 38.46 | 210.18 | 144.37–266.77 | 192.40 ± 29.28 | 194.07 | 132.24–281.41 | 0.013 |
| Mn [µmol/L] | 0.11 ± 0.24 | 0.06 | 0.01–2.87 | 0.11 ± 0.13 | 0.08 | 0.00–1.11 | 0.079 |
| Cu [µmol/L] | 32.86 ± 7.85 | 32.30 | 5.00–77.40 | 7.17 ± 4.35 | 6.60 | 0.50–33.40 | 0.000 |
| Zn [µmol/L] | 12.13 ± 8.50 | 10.75 | 1.00–58.50 | 15.17 ± 13.60 | 12.40 | 2.00–143.9 | 0.007 |
| GPx [U/g Hg] | 95.14 ± 42.97 | 88.49 | 2.81–211.40 | 151.13 ± 96.77 | 141.87 | 93.41–1057.14 | 0.000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zych, B.; Górka, A.; Myszka, A.; Błoniarz, D.; Siekierzyńska, A.; Błaż, W. Status of Oxidative Stress during Low-Risk Labour: Preliminary Data. Int. J. Environ. Res. Public Health 2023, 20, 157. https://doi.org/10.3390/ijerph20010157
Zych B, Górka A, Myszka A, Błoniarz D, Siekierzyńska A, Błaż W. Status of Oxidative Stress during Low-Risk Labour: Preliminary Data. International Journal of Environmental Research and Public Health. 2023; 20(1):157. https://doi.org/10.3390/ijerph20010157
Chicago/Turabian StyleZych, Barbara, Anna Górka, Aleksander Myszka, Dominika Błoniarz, Aleksandra Siekierzyńska, and Witold Błaż. 2023. "Status of Oxidative Stress during Low-Risk Labour: Preliminary Data" International Journal of Environmental Research and Public Health 20, no. 1: 157. https://doi.org/10.3390/ijerph20010157
APA StyleZych, B., Górka, A., Myszka, A., Błoniarz, D., Siekierzyńska, A., & Błaż, W. (2023). Status of Oxidative Stress during Low-Risk Labour: Preliminary Data. International Journal of Environmental Research and Public Health, 20(1), 157. https://doi.org/10.3390/ijerph20010157







