Hypoxia Pathway in Osteoporosis: Laboratory Data for Clinical Prospects
Abstract
:1. Introduction
2. Osteoporosis
3. Bone Remodeling
4. Hypoxia
4.1. Discovery and Introduction of Hypoxia-Related Molecules
4.2. The Role of Hypoxia Environment in the Body
4.3. Regulatory Mechanism of Hypoxia
5. Hypoxia Pathway in OP, Osteoblast, Osteoclast and Osteocytes
5.1. Research Progress and the Regulation between the Hypoxia Pathway and OP
5.2. Research Progress and the Regulation between Hypoxia Pathway and OB
5.3. Research Progress and the Regulation between the Hypoxia Pathway and OC
5.4. Research Progress and the Regulation between the Hypoxia Pathway and Osteocytes
6. Application of O2, Hypoxia Pathway in OP/Iron Overload Diseases
6.1. Hyperbaric Oxygen Chamber Therapy and OP
6.2. Mechanical Stress, Hypoxia and Bone Remodeling
6.3. HIF-Related Drugs and Iron Metabolism Diseases
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Thompson, C.B. Into Thin Air: How We Sense and Respond to Hypoxia. Cell 2016, 167, 9–11. [Google Scholar] [CrossRef]
- Whitham, S.; Dinesh-Kumar, S.; Choi, D.; Hehl, R.; Corr, C.; Baker, B. Transcriptional regulation of genes encoding glycolytic enzymes by hypoxia-inducible factor 1. J. Biol. Chem. 1994, 269, 23757–23763. [Google Scholar] [CrossRef]
- Moore, L.G. Measuring High-attitude Adaptation. J. Appl. Physiol. 2017, 123, 1371–1385. [Google Scholar] [CrossRef] [PubMed]
- Fandrey, J.; Schödel, J.; Eckardt, K.-U.; Katschinski, D.M.; Wenger, R.H. Now a Nobel gas: Oxygen. Pflugers. Arch. 2019, 471, 1343–1358. [Google Scholar] [CrossRef]
- Locatelli, F.; Del Vecchio, L.; Luise, M.C. Current and future chemical therapies for treating anaemia in chronic kidney disease. Expert. Opin. Pharmacother. 2017, 18, 781–788. [Google Scholar] [CrossRef]
- Kabei, K.; Tateishi, Y.; Shiota, M.; Osada-Oka, M.; Nishide, S.; Uchida, J.; Nakatani, T.; Matsunaga, S.; Yamaguchi, T.; Tomita, S.; et al. Effects of orally active hypoxia inducible factor alpha prolyl hydroxylase inhibitor, FG4592 on renal fibrogenic potential in mouse unilateral ureteral obstruction model. J. Pharmacol. Sci. 2020, 142, 93–100. [Google Scholar] [CrossRef]
- Wu, J.; Contratto, M.; Shanbhogue, K.P.; Manji, G.A.; O’Neil, B.H.; Noonan, A.; Tudor, R.; Lee, R. Evaluation of a locked nucleic acid form of antisense oligo targeting HIF-1α in advanced hepatocellular carcinoma. World J. Clin. Oncol. 2019, 10, 149–160. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, Y.; Jia, Y.; Xu, J.; Chai, Y. Roxadustat promotes angiogenesis through HIF-1α/VEGF/VEGFR2 signaling and accelerates cutaneous wound healing in diabetic rats. Wound Repair Regen. 2019, 27, 324–334. [Google Scholar] [CrossRef]
- Carreau, A.; Hafnyrahbi, B.E.; Matejuk, A.; Grillon, C.; Kieda, C. Why is the partial oxygen pressure of human tissues a crucial parameter? Small molecules and hypoxia. J. Cell. Mol. Med. 2011, 15, 1239–1253. [Google Scholar] [CrossRef]
- Poussa, M.; Rubak, J.; Ritsil, V. Differentiation of the Osteochondrogenic Cells of the Periosteum in Chondrotrophic Environment. Acta. Orthop. Scand. 1981, 52, 235–239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kusumbe, A.P.; Ramasamy, S.K.; Adams, R.H. Coupling of angiogenesis and osteogenesis by a specific vessel subtype in bone. Nature 2014, 507, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.W.; Ueharu, H.; Mishina, Y.J. Energy metabolism: A newly emerging target of BMP signaling in bone homeostasis. Bone 2020, 138, 115467. [Google Scholar] [CrossRef] [PubMed]
- Long, H.; Sun, B.; Cheng, L.; Zhao, S.; Zhu, Y.; Zhao, R.; Zhu, J. miR-139-5p Represses BMSC Osteogenesis via Targeting Wnt/β-Catenin Signaling Pathway. DNA Cell Biol. 2017, 36, 715–724. [Google Scholar] [CrossRef] [PubMed]
- Serocki, M.; Bartoszewska, S.; Janaszak-Jasiecka, A.; Ochocka, R.J.; Collawn, J.F.; Bartoszewski, R. miRNAs regulate the HIF switch during hypoxia: A novel therapeutic target. Angiogenesis 2018, 21, 183–202. [Google Scholar] [CrossRef]
- Yang, T.L.; Shen, H.; Liu, A.; Dong, S.S.; Zhang, L.; Deng, F.Y.; Zhao, Q.; Deng, H.W. A road map for understanding molecular and genetic determinants of osteoporosis. Nat. Rev. Endocrinol. 2020, 16, 91–103. [Google Scholar] [CrossRef]
- Compston, J.E.; McClung, M.R.; Leslie, W.D. Osteoporosis. Lancet 2019, 393, 364–376. [Google Scholar] [CrossRef]
- Vico, L.; Hargens, A. Skeletal changes during and after spaceflight. Nat. Rev. Rheumatol. 2018, 14, 229–245. [Google Scholar] [CrossRef]
- Yang, J.C.; Yang, Z.Q.; Li, W.B.; Xue, Y.R.; Xu, H.Y.; Li, J.B.; Shang, P. Glucocorticoid: A potential role in microgravity-induced bone loss. Acta Astronaut. 2017, 140, 206–212. [Google Scholar] [CrossRef]
- Sugimoto, T.; Inoue, D.; Maehara, M.; Oikawa, I.; Shigematsu, T.; Nishizawa, Y. Efficacy and safety of once-monthly risedronate in osteoporosis subjects with mild-to-moderate chronic kidney disease: A post hoc subgroup analysis of a phase III trial in Japan. J. Bone Miner. Metab. 2019, 37, 730–740. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Hui, K.; Hao, C.; Peng, Z.; Gao, Q.X.; Jin, Q.; Lei, G.; Min, J.; Qi, Z.; Bo, C. Low bone turnover and reduced angiogenesis in streptozotocin-induced osteoporotic mice. Connect. Tissue Res. 2016, 57, 277–289. [Google Scholar] [CrossRef]
- Hamed, S.A. Markers of bone turnover in patients with epilepsy and their relationship to management of bone diseases induced by antiepileptic drugs. Expert. Rev. Clin. Pharmacol. 2016, 9, 267–286. [Google Scholar] [CrossRef] [PubMed]
- Ireland, A.; JRittweger, J. Exercise for osteoporosis: How to navigate between overeagerness and defeatism. J. Musculoskelet. Neuronal Interact. 2017, 17, 155–161. [Google Scholar]
- Oganov, V.S.; Cann, C.; Rakhmanov, A.S.; Ternovoĭ, S.K. Study of the musculoskeletal system of the spine in humans after long-term space flights by the method of computerized tomography. Kosm Biol. Aviakosm. Med. 1990, 24, 20–21. [Google Scholar]
- Sarko, J. Bone and mineral metabolism. Emerg. Med. Clin. N. Am. 2005, 23, 703–721. [Google Scholar] [CrossRef]
- McKee, H.; Ioannidis, G.; Lau, A.; Treleaven, D.; Gangji, A.; Ribic, C.; Wong-Pack, M.; Papaioannou, A.; Adachi, J.D. Comparison of the clinical effectiveness and safety between the use of denosumab vs bisphosphonates in renal transplant patients. Osteoporos. Int. 2020, 31, 973–980. [Google Scholar] [CrossRef]
- Stone, J.A.; McCrea, J.B.; Witter, R.; Zajic, S.; Stoch, S.A. Clinical and translational pharmacology of the cathepsin K inhibitor odanacatib studied for osteoporosis. Br. J. Clin. Pharmacol. 2019, 85, 1072–1083. [Google Scholar] [CrossRef] [PubMed]
- Yamada, H.; Ochi, Y.; Mori, H.; Nishikawa, S.; Hashimoto, Y.; Tanaka, M.; Deacon, S.; Kawabata, K. Cortical bone mineral density is increased by the cathepsin K inhibitor ONO-5334, which leads to a robust increase in bone strength: Results from a 16-month study in ovariectomised cynomolgus monkeys. J. Bone Miner. Metab. 2019, 37, 636–647. [Google Scholar] [CrossRef]
- Markham, A. Romosozumab: First Global Approval. Drugs. 2019, 79, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Stolina, M.; Dwyer, D.; Niu, Q.T.; Villasenor, K.S.; Kurimoto, P.; Grisanti, M.; Han, C.Y.; Liu, M.; Li, X.; Ominsky, M.S.; et al. Temporal changes in systemic and local expression of bone turnover markers during six months of sclerostin antibody administration to ovariectomized rats. Bone 2014, 67, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Samadfam, R.; Jacobs, S.; Van Roy, M.; McGuire, A.; Schoen, P.; Baumeister, J.; Smith, S.Y.; Holz, J.B. ALX-0141, an anti-RANK-L targeting Nanobody®, increases bone mass in cynomolgus monkeys. Bone 2012, 50, 46. [Google Scholar] [CrossRef]
- Dam, C.; Jung, U.W.; Park, K.M.; Huh, J.; Park, W. Effect of teriparatide on early sinus graft healing in the ovariectomized rabbit. Clin. Oral. Implant. Res. 2020, 31, 264–273. [Google Scholar] [CrossRef]
- Stunes, A.K.; Erben, R.G.; Schüler, C.; Eriksen, E.F.; Tice, M.; Vashishth, D.; Syversen, U.; Mosti, M.P. Skeletal effects of plyometric exercise and metformin in ovariectomized rats. Bone 2020, 132, 115193. [Google Scholar] [CrossRef]
- Palermo, A.; Tuccinardi, D.; D’Onofrio, L.; Watanabe, M.; Maggi, D.; Maurizi, A.R.; Greto, V.; Buzzetti, R.; Napoli, N.; Pozzilli, P.; et al. Vitamin K and osteoporosis: Myth or reality? Metabolism 2017, 70, 57–71. [Google Scholar] [CrossRef]
- Gatti, D.; Fassio, A. Pharmacological management of osteoporosis in postmenopausal women: The current state of the art. J. Popul. Ther. Clin. Pharmacol. 2019, 26, e1–e17. [Google Scholar] [CrossRef]
- Ringe, J.D. Plain vitamin D or active vitamin D in the treatment of osteoporosis: Where do we stand today? Arch. Osteoporos. 2020, 15, 182. [Google Scholar] [CrossRef]
- Wang, H.; Wan, Y.; Tam, K.F.; Ling, S.; Bai, Y.; Deng, Y.; Liu, Y.; Zhang, H.; Cheung, W.H.; Qin, L.; et al. Resistive vibration exercise retards bone loss in weight-bearing skeletons during 60 days bed rest. Osteoporos. Int. 2012, 23, 2169–2178. [Google Scholar] [CrossRef]
- Bassett, C.A.; Pilla, A.A.; Pawluk, R.J. A non-operative salvage of surgically-resistant pseudarthroses and non-unions by pulsing electromagnetic fields. A preliminary report. Clin. Orthop. Relat. Res. 1977, 128–143, 598067. [Google Scholar]
- Gujjalapudi, M.; Anam, C.; Mamidi, P.; Chiluka, R.; Kumar, A.G.; Bibinagar, R. Effect of Magnetic Field on Bone Healing around Endosseous Implants-An In-vivo Study. J. Clin. Diagn. Res. 2016, 10, ZF01–ZF04. [Google Scholar] [CrossRef] [PubMed]
- Lewjecki, E.M. Pulse-echo Ultrasound Identifies Caucasian and Hispanic Women at Risk for Osteoporosis. J. Clin. Densitom. 2021, 24, 175–182. [Google Scholar] [CrossRef]
- Khosla, S.; Hofbauer, L.C. Osteoporosis treatment: Recent developments and ongoing challenges. Lancet Diabetes Endocrinol. 2017, 5, 898–907. [Google Scholar] [CrossRef]
- Rozenberg, S.; Bruyère, O.; Bergmann, P.; Cavalier, E.; Gielen, E.; Goemaere, S.; Kaufman, J.M.; Lapauw, B.; Laurent, M.R.; De Schepper, J.; et al. How to manage osteoporosis before the age of 50. Maturitas 2020, 138, 14–25. [Google Scholar] [CrossRef]
- Dirckx, N.; Moorer, M.C.; Clemens, T.L.; Riddle, R.C. The role of osteoblasts in energy homeostasis. Nat. Rev. Endocrinol. 2019, 15, 651–665. [Google Scholar] [CrossRef]
- Eriksen, E.F. Cellular mechanisms of bone remodeling. Rev. Endocr. Metab. Disord. 2010, 11, 219–227. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Li, J.Y.; Zhang, X.Z.; Liu, L.; Wan, Z.M.; Li, R.X.; Guo, Y. Involvement of p38MAPK/NF-κB signaling pathways in osteoblasts differentiation in response to mechanical stretch. Ann. Biomed. Eng. 2012, 40, 1884–1894. [Google Scholar] [CrossRef]
- Zhang, J.; Ding, C.; Ren, L.; Zhou, Y.; Shang, P. The effects of static magnetic fields on bone. Prog. Biophys. Mol. Biol. 2014, 114, 146–152. [Google Scholar] [CrossRef]
- Bassett, C.A.L.; Pawluk, R.J.; Pilla, A.A. Augmentation of bone repair by inductively coupled electromagnetic fields. Science 1974, 184, 575–577. [Google Scholar] [CrossRef]
- Zhou, J.; Ma, X.N.; Gao, Y.H.; Yan, J.L.; Shi, W.G.; Xian, C.J.; Chen, K.M. Sinusoidal electromagnetic fields promote bone formation and inhibit bone resorption in rat femoral tissues in vitro. Electromagn. Biol. Med. 2016, 35, 75–83. [Google Scholar] [CrossRef]
- Yang, J.; Zhou, S.; Lv, H.; Wei, M.; Fang, Y.; Shang, P. Static magnetic field of 0.2-0.4 T promotes the recovery of hindlimb unloading-induced bone loss in mice. Int. J. Radiat. Biol. 2021, 97, 746–754. [Google Scholar] [CrossRef]
- Guan, S.; Zhang, Z.; Wu, J. Non-coding RNA delivery for bone tissue engineering: Progress, challenges, and potential solutions. iScience 2022, 25, 104807. [Google Scholar] [CrossRef]
- Li, Z.; Xue, H.; Tan, G.; Xu, Z. Effects of miRNAs, lncRNAs and circRNAs on osteoporosis as regulatory factors of bone homeostasis (Review). Mol. Med. Rep. 2021, 24, 788. [Google Scholar] [CrossRef]
- Bellavia, D.; De Luca, A.; Carina, V.; Costa, V.; Raimondi, L.; Salamanna, F.; Alessandro, R.; Fini, M.; Giavaresi, G. Deregulated miRNAs in bone health: Epigenetic roles in osteoporosis. Bone 2019, 122, 52–75. [Google Scholar] [CrossRef]
- Bellavia, D.; Salamanna, F.; Raimondi, L.; De Luca, A.; Carina, V.; Costa, V.; Alessandro, R.; Fini, M.; Giavaresi, G. Deregulated miRNAs in osteoporosis: Effects in bone metastasis. Cell Mol. Life Sci. 2019, 76, 3723–3744. [Google Scholar] [CrossRef]
- Zhao, W.; Shen, G.; Ren, H.; Liang, D.; Yu, X.; Zhang, Z.; Huang, J.; Qiu, T.; Tang, J.; Shang, Q.; et al. Therapeutic potential of microRNAs in osteoporosis function by regulating the biology of cells related to bone homeostasis. J. Cell Physiol. 2018, 233, 9191–9208. [Google Scholar] [CrossRef]
- Suttamanatwong, S. MicroRNAs in bone development and their diagnostic and therapeutic potentials in osteoporosis. Connect Tissue Res. 2017, 58, 90–102. [Google Scholar] [CrossRef]
- Ji, X.; Chen, X.; Yu, X. MicroRNAs in Osteoclastogenesis and Function: Potential Therapeutic Targets for Osteoporosis. Int. J. Mol. Sci. 2016, 17, 349. [Google Scholar] [CrossRef]
- Ma, Y.; Shan, Z.; Ma, J.; Wang, Q.; Chu, J.; Xu, P.; Qin, A.; Fan, S. Validation of downregulated microRNAs during osteoclast formation and osteoporosis progression. Mol. Med. Rep. 2016, 13, 2273–2280. [Google Scholar] [CrossRef]
- Yu, F.Y.; Xie, C.Q.; Sun, J.T.; Peng, W.; Huang, X.W. Overexpressed miR-145 inhibits osteoclastogenesis in RANKL-induced bone marrow-derived macrophages and ovariectomized mice by regulation of Smad3. Life Sci. 2018, 202, 11–20. [Google Scholar] [CrossRef]
- Yan, J.; Guo, D.; Yang, S.; Sun, H.; Wu, B.; Zhou, D. Inhibition of miR-222-3p activity promoted osteogenic differentiation of hBMSCs by regulating Smad5-RUNX2 signal axis. Biochem. Biophys. Res. Commun. 2016, 470, 498–503. [Google Scholar] [CrossRef]
- Tian, L.; Zheng, F.; Li, Z.; Wang, H.; Yuan, H.; Zhang, X.; Ma, Z.; Li, X.; Gao, X.; Wang, B. miR-148a-3p regulates adipocyte and osteoblast differentiation by targeting lysine-specific demethylase 6b. Gene 2017, 627, 32–39. [Google Scholar] [CrossRef]
- Kenkre, J.S.; Bassett, J. The bone remodelling cycle. Ann. Clin. Biochem. 2018, 55, 308–327. [Google Scholar] [CrossRef]
- Grewe, J.M.; Knapstein, P.R.; Donat, A.; Jiang, S.; Smit, D.J.; Xie, W.; Keller, J. The role of sphingosine-1-phosphate in bone remodeling and osteoporosis. Bone Res. 2022, 10, 34. [Google Scholar] [CrossRef]
- Wang, L.; You, X.; Zhang, L.; Zhang, C.; Zou, W. Mechanical regulation of bone remodeling. Bone Res. 2022, 10, 16. [Google Scholar] [CrossRef]
- Siddiqui, J.A.; Partridge, N.C. Physiological Bone Remodeling: Systemic Regulation and Growth Factor Involvement. Physiology 2016, 31, 233–245. [Google Scholar] [CrossRef]
- Kim, J.M.; Lin, C.; Stavre, Z.; Greenblatt, M.B.; Shim, J.H. Osteoblast-Osteoclast Communication and Bone Homeostasis. Cells 2022, 9, 2073. [Google Scholar] [CrossRef]
- Yellowley, C.E.; Genetos, D.C. Hypoxia Signaling in the Skeleton: Implications for Bone Health. Curr. Osteoporos. Rep. 2019, 17, 26–35. [Google Scholar] [CrossRef]
- Semenza, G.L.; Wang, G.L. A nuclear factor induced by hypoxia via de novo protein synthesis binds to the human erythropoietin gene enhancer at a site required for transcriptional activation. Mol. Cell. Biol. 1992, 12, 5447–5454. [Google Scholar] [CrossRef]
- Wang, G.L.; Semenza, G.L. Purification and Characterization of Hypoxia-inducible Factor 1. J. Biol. Chem. 1995, 270, 1230–1237. [Google Scholar] [CrossRef]
- Iliopoulos, O.; Levy, A.P.; Jiang, C.; KaelinJr, W.G.; Goldberg, M.A. Negative regulation of hypoxia-inducible genes by the von Hippel-Lindau protein. Proc. Natl. Acad. Sci. USA 1996, 93, 10595–10599. [Google Scholar] [CrossRef]
- Maxwell, P.H.; Wiesener, M.S.; Chang, G.W.; Clifford, S.C.; Vauxx, E.C.; Cockman, M.E.; Wykoff, C.C.; Pugh, C.W.; Maher, E.R.; Ratcliffe, P.J.; et al. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature 1999, 399, 271–275. [Google Scholar] [CrossRef]
- Jaakkola, P.; Mole, D.R.; Tian, Y.M.; Wilson, M.I.; Gielbert, J.; Gaskell, S.J.; von Kriegsheim, A.; Hebestreit, H.F.; Mukherji, M.; Schofield, C.J.; et al. Targeting of HIF-α to the von Hippel-Lindau Ubiquitylation Complex by O2-Regulated Prolyl Hydroxylation. Science 2001, 292, 468–472. [Google Scholar] [CrossRef]
- Epstein, A.C.; Gleadle, J.M.; McNeill, L.A.; Hewitson, K.S.; O’Rourke, J.; Mole, D.R.; Mukherji, M.; Metzen, E.; Wilson, M.I.; Dhanda, A.; et al. elegans EGL-9 and mammalian homologs define a family of dioxygenases that regulate HIF by prolyl hydroxylation. Cell 2001, 107, 43–54. [Google Scholar] [CrossRef]
- Strowitzki, M.J.; Cummins, E.P.; Taylor, C.T. Protein Hydroxylation by Hypoxia-Inducible Factor (HIF) Hydroxylases: Unique or Ubiquitous? Cells 2019, 8, 384. [Google Scholar] [CrossRef] [PubMed]
- Bruick, R.K.; McKnight, S.L. A Conserved Family of Prolyl-4-Hydroxylases That Modify HIF. Science 2001, 294, 1337–1340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hurst, J.H. William Kaelin, Peter Ratcliffe, and Gregg Semenza receive the 2016 Albert Lasker Basic Medical Research Award. J. Clin. Investig. 2016, 126, 3628–3638. [Google Scholar] [CrossRef]
- Appelhoff, R.J.; Tian, Y.M.; Raval, R.R.; Turley, H.; Harris, A.L.; Pugh, C.W.; Ratcliffe, P.J.; Gleadle, J.M. Differential Function of the Prolyl Hydroxylases PHD1, PHD2, and PHD3 in the Regulation of Hypoxia-inducible Factor. J. Biol. Chem. 2004, 279, 38458–38465. [Google Scholar] [CrossRef] [PubMed]
- Semenza, G.L. Regulation of Mammalian O2 Homeostasis by Hypoxia-Inducible Factor 1. Annu. Rev. Cell. Dev. Biol. 1999, 15, 551–578. [Google Scholar] [CrossRef]
- Wang, G.L.; Jiang, B.H.; Rue, E.A.; Semenza, G.L. Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. Proc. Natl. Acad. Sci. USA 1995, 92, 5510–5514. [Google Scholar] [CrossRef]
- Hirose, K.; Morita, M.; Ema, M.; Mimura, J.; Hamada, H.; Fujii, H.; Saijo, Y.; Gotoh, O.; Sogawa, K.; Fujii-Kuriyama, Y.; et al. cDNA cloning and tissue-specific expression of a novel basic helix-loop-helix/PAS factor (Arnt2) with close sequence similarity to the aryl hydrocarbon receptor nuclear translocator (Arnt). Mol. Cell. Biol. 1996, 16, 1706–1713. [Google Scholar] [CrossRef]
- Takahata, S.; Sogawa, K.; Kobayashi, A.; Ema, M.; Mimura, J.; Ozaki, N.; Fujii-Kuriyama, Y. Transcriptionally active heterodimer formation of an Arnt-like PAS protein, Arnt3, with HIF-1a, HLF, and clock. Biochem. Biophys. Res. Commun. 1998, 248, 789–794. [Google Scholar] [CrossRef]
- Hu, C.J.; Wang, L.Y.; Chodosh, L.A.; Keith, B.; Simon, M.C. Differential roles of hypoxia-inducible factor 1alpha (HIF-1alpha) and HIF-2alpha in hypoxic gene regulation. Mol. Cell. Biol. 2003, 23, 9361–9374. [Google Scholar] [CrossRef]
- Yang, S.L.; Wu, C.; Xiong, Z.F.; Fang, X.F. Progress on hypoxia-inducible factor-3: Its structure, gene regulation and biological function. Mol. Med. Rep. 2015, 122, 411–2416. [Google Scholar] [CrossRef]
- Loboda, A.; Jozkowicz, A.; Dulak, J. HIF-1 and HIF-2 transcription factors-Similar but not identical. Mol. Cells 2010, 29, 435–442. [Google Scholar] [CrossRef]
- Keith, B.; Johnson, R.S.; Simon, M.C. HIF1α and HIF2α: Sibling rivalry in hypoxic tumour growth and progression. Nat. Rev. Cancer 2011, 12, 9–22. [Google Scholar] [CrossRef] [Green Version]
- Mastrogiannaki, M.; Matak, P.; Keith, B.; Simon, M.C.; Vaulont, S.; Peyssonnaux, C. HIF-2α, but not HIF-1α, promotes iron absorption in mice. J. Clin. Investig. 2009, 119, 1159–1166. [Google Scholar] [CrossRef]
- Hung, S.P.; Ho, J.H.; Shih, Y.R.; Lo, T.; Lee, O.K. Hypoxia Promotes Proliferation and Osteogenic Differentiation Potentials of Human Mesenchymal Stem Cells. J. Orthop. Res. 2011, 30, 260–266. [Google Scholar] [CrossRef]
- Spencer, J.A.; Ferraro, F.; Roussakis, E.; Spencer, J.A.; Ferraro, F.; Roussakis, E.; Klein, A.; Wu, J.; Runnels, J.M.; Zaher, W.; et al. Direct measurement of local oxygen concentration in the bone marrow of live animals. Nature 2014, 508, 269–273. [Google Scholar] [CrossRef]
- Stegen, S.; van Gastel, N.V.; Eelen, G.; Ghesquière, B.; D’Anna, F.; Thienpont, B.; Goveia, J.; Torrekens, S.; Van Looveren, R.; Luyten, F.P.; et al. HIF-1α Promotes Glutamine-Mediated Redox Homeostasis and Glycogen-Dependent Bioenergetics to Support Postimplantation Bone Cell Survival. Cell. Metab. 2016, 23, 265–279. [Google Scholar] [CrossRef]
- Chan, M.C.; Ilott, N.E.; Schödel, J.; Sims, D.; Tumber, A.; Lippl, K.; Mole, D.R.; Pugh, C.W.; Ratcliffe, P.J.; Ponting, C.P.; et al. Tuning the Transcriptional Response to Hypoxia by Inhibiting Hypoxia-inducible Factor (HIF) Prolyl and Asparaginyl Hydroxylases. J. Biol. Chem. 2016, 291, 20661–20673. [Google Scholar] [CrossRef]
- Oki, M.; Ando, K. Hematopoietic growth factors, cytokines, and bone-marrow microenvironment. Nihon. Rinsho. 2008, 66, 444–452. [Google Scholar] [PubMed]
- Weng, T.J.; Xie, Y.L.; Huang, J.L.; Luo, F.; Yi, L.; He, Q.; Chen, D.; Chen, L. Inactivation of Vhl in osteochondral progenitor cells causes high bone mass phenotype and protects against age-related bone loss in adult mice. J. Bone Miner. Res. 2014, 29, 820–829. [Google Scholar] [CrossRef]
- Wan, C.; Shao, J.; Gilbert, S.R.; Riddle, R.C.; Long, F.; Johnson, R.S.; Schipani, E.; Clemens, T.L. Role of HIF-1alpha in skeletal development. Ann. N. Y. Acad. Sci. 2010, 1192, 322–326. [Google Scholar] [CrossRef]
- Stegen, S.; Carmeliet, G. Hypoxia, hypoxia-inducible transcription factors and oxygen-sensing prolyl hydroxylases in bone development and homeostasis. Curr. Opin. Nephrol. Hypertens. 2019, 28, 328–335. [Google Scholar] [CrossRef]
- Fujimoto, H.; Fujimoto, K.; Ueda, A.; Ohata, M. Hypoxemia is a risk factor for bone mass loss. J. Bone Miner. Metab. 1999, 17, 211–216. [Google Scholar] [CrossRef]
- Yang, Y.X.; Fan, X.R.; Tao, J.Y.; Xu, T.; Zhang, Y.; Zhang, W.; Li, L.; Li, X.; Ding, H.; Sun, M.; et al. Impact of prenatal hypoxia on fetal bone growth and osteoporosis in ovariectomized offspring rats. Reprod. Toxicol. 2018, 78, 1–8. [Google Scholar] [CrossRef]
- Kaluz, S.; Tan, C.; Van Meir, E.G. Taking a HIF pill for old age diseases? Aging 2018, 10, 290–292. [Google Scholar] [CrossRef]
- Ade, C.J.; Broxterman, R.M.; Moore, A.D.; Barstow, T.J. Decreases in maximal oxygen uptake following long-duration spaceflight: Role of convective and diffusive O2 transport mechanisms. J. Appl. Physiol. 2017, 122, 968–975. [Google Scholar] [CrossRef]
- Stevens, H.Y.; Meays, D.R.; Frangos, J.A. Pressure gradients and transport in the murine femur upon hindlimb suspension. Bone 2006, 39, 565–572. [Google Scholar] [CrossRef]
- Zeng, G.Z.; Pan, X.L.; Tan, N.H.; Xiong, J.; Zhang, Y.M. Natural biflavones as novel inhibitors of cathepsin B and K. Eur. J. Med. Chem. 2006, 41, 1247–1252. [Google Scholar] [CrossRef]
- Liu, X.D.; Tu, Y.H.; Zhang, L.F.; Qi, J.; Ma, T.; Deng, L. Prolyl hydroxylase inhibitors protect from the bone loss in ovariectomy rats by increasing bone vascularity. Cell Biochem. Biophys. 2014, 69, 141–149. [Google Scholar] [CrossRef]
- Xue, Y.R.; Yang, J.C.; Luo, J.; Ren, L.; Shen, Y.; Dong, D.; Fang, Y.; Hu, L.; Liu, M.; Liao, Z.; et al. Disorder of Iron Metabolism Inhibits the Recovery of Unloading-Induced Bone Loss in Hypomagnetic Field. J. Bone Miner. Res. 2020, 35, 1163–1173. [Google Scholar] [CrossRef]
- Zhao, Q.; Shen, X.; Zhang, W.; Zhu, G.; Qi, J.; Deng, L. Mice with increased angiogenesis and osteogenesis due to conditional activation of HIF pathway in osteoblasts are protected from ovariectomy induced bone loss. Bone 2012, 50, 763–770. [Google Scholar] [CrossRef]
- Zhang, P.; Ha, N.Y.; Dai, Q.G.; Zhou, S.; Yu, C.; Jiang, L. Hypoxia suppresses osteogenesis of bone mesenchymal stem cells via the extracellular signal-regulated 1/2 and p38-mitogen activated protein kinase signaling pathways. Mol. Med. Rep. 2017, 16, 5515–5522. [Google Scholar] [CrossRef] [PubMed]
- Miyauchi, Y.; Sato, Y.; Kobayashi, T.; Yoshida, S.; Mori, T.; Kanagawa, H.; Katsuyama, E.; Fujie, A.; Hao, W.; Miyamoto, K.; et al. HIF1α is required for osteoclast activation by estrogen deficiency in postmenopausal osteoporosis. Proc. Natl. Acad. Sci. USA 2013, 110, 16568–16573. [Google Scholar] [CrossRef] [Green Version]
- Stegen, S.; Stockmans, I.; Moermans, K.; Thienpont, B.; Maxwell, P.H.; Carmeliet, P.; Carmeliet, G. Osteocytic oxygen sensing controls bone mass through epigenetic regulation of sclerostin. Nat. Commun. 2018, 9, 2557. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Xiao, L.Y.; Min, W.; Yuan, W.; Lu, S.; Huang, G. Bu-Shen-Tong-Luo decoction prevents bone loss via inhibition of bone resorption and enhancement of angiogenesis in ovariectomy-induced osteoporosis of rats. J. Ethnopharmacol. 2018, 220, 228–238. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Qu, Y.; Jin, X.; Guo, X.Q.; Wang, Y.; Qi, L.; Yang, J.; Zhang, P.; Li, L.Z. Protective effect of salidroside against bone loss via hypoxia-inducible factor-1α pathway-induced angiogenesis. Sci. Rep. 2016, 6, 32131. [Google Scholar] [CrossRef]
- Fayed, H.A.; Barakat, B.M.; Elshaer, S.S.; Abdel-Naim, A.B.; Menze, E.T. Antiosteoporotic activities of isoquercitrin in ovariectomized rats: Role of inhibiting hypoxia inducible factor-1 alpha. Eur. J. Pharmacol. 2019, 865, 172785. [Google Scholar] [CrossRef]
- Zhu, N.Q.; Hou, J.Y. Exploring the mechanism of action Xianlingubao Prescription in the treatment of osteoporosis by network pharmacology. Comput. Biol. Chem. 2020, 85, 107240. [Google Scholar] [CrossRef] [PubMed]
- Shao, J.; Zhang, Y.; Yang, T.Y.; Qi, J.; Zhang, L.; Deng, L. HIF-1α disturbs osteoblasts and osteoclasts coupling in bone remodeling by up-regulating OPG expression. Vitr. Cell. Dev. Biol. Anim. 2015, 51, 808–814. [Google Scholar] [CrossRef]
- Tomlinson, R.E.; Silva, M.J. HIF-1α regulates bone formation after osteogenic mechanical loading. Bone 2015, 73, 98–104. [Google Scholar] [CrossRef]
- Riddle, R.C.; Leslie, J.M.; Gross, T.S.; Clemens, T.L. Hypoxia-inducible factor-1α protein negatively regulates load-induced bone formation. J. Biol. Chem. 2011, 286, 44449–44456. [Google Scholar] [CrossRef]
- Lee, S.Y.; Park, K.H.; Lee, G.; Kim, S.J.; Song, W.H.; Kwon, S.H.; Koh, J.T.; Huh, Y.H.; Ryu, J.H. Hypoxia-inducible factor-2α mediates senescence-associated intrinsic mechanisms of age-related bone loss. Exp. Mol. Med. 2021, 53, 591–604. [Google Scholar] [CrossRef]
- Hannah, S.S.; McFadden, S.; McNeilly, A.; McClean, C. "Take My Bone Away?" Hypoxia and bone: A narrative review. J. Cell. Physiol. 2021, 236, 721–740. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Wang, M.M.; Feng, H.H.; Peng, Y.; Sun, J.; Qu, X.; Li, C. Lactate induces osteoblast differentiation by stabilization of HIF1α. Mol. Cell. Endocrinol. 2017, 452, 84–92. [Google Scholar] [CrossRef]
- Liu, T.L.; Zou, W.W.; Shi, G.D.; Xu, J.; Zhang, F.; Xiao, J.; Wang, Y. Hypoxia-induced MTA1 promotes MC3T3 osteoblast growth but suppresses MC3T3 osteoblast differentiation. Eur. J. Med. Res. 2015, 20, 10. [Google Scholar] [CrossRef]
- Kubota, M. Study on proliferation and function of periodontal ligament fibroblasts and osteoblastic cells under hypoxia. Kokubyo. Gakkai. Zasshi 1989, 56, 473–484. [Google Scholar] [CrossRef]
- Genetos, D.C.; Toupadakis, C.A.; Raheja, L.F.; Wong, A.; Papanicolaou, S.E.; Fyhrie, D.P.; Loots, G.G.; Yellowley, C.E. Hypoxia decreases sclerostin expression and increases Wnt signaling in osteoblasts. J. Cell. Biochem. 2010, 110, 457–467. [Google Scholar] [CrossRef]
- Tseng, W.P.; Yang, S.N.; Lai, C.H.; Tang, C.H. Hypoxia induces BMP-2 expression via ILK, Akt, mTOR, and HIF-1 pathways in osteoblasts. J. Cell. Physiol. 2010, 223, 810–818. [Google Scholar] [CrossRef]
- Rankin, E.B.; Wu, C.; Khatri, R.; Wilson, T.L.; Andersen, R.; Araldi, E.; Rankin, A.L.; Yuan, J.; Kuo, C.J.; Schipani, E.; et al. The HIF signaling pathway in osteoblasts directly modulates erythropoiesis through the production of EPO. Cell 2012, 149, 63–74. [Google Scholar] [CrossRef]
- Wang, Y.; Wan, C.; Deng, L.F.; Liu, X.; Cao, X.; Gilbert, S.R.; Bouxsein, M.L.; Faugere, M.C.; Guldberg, R.E.; Gerstenfeld, L.C.; et al. The hypoxia-inducible factor α pathway couples angiogenesis to osteogenesis during skeletal development. J. Clin. Investig. 2007, 117, 1616–1626. [Google Scholar] [CrossRef]
- Zhu, K.; Jiao, H.L.; Li, S.; Cao, H.; Galson, D.L.; Zhao, Z.; Zhao, X.; Lai, Y.; Fan, J.; Im, H.J.; et al. ATF4 promotes bone angiogenesis by increasing VEGF expression and release in the bone environment. J. Bone Miner. Res. 2013, 28, 1870–1884. [Google Scholar] [CrossRef]
- Costa, V.; Raimondi, L.; Conigliaro, A.; Salamanna, F.; Carina, V.; De Luca, A.; Bellavia, D.; Alessandro, R.; Fini, M.; Giavaresi, G. Hypoxia-inducible factor 1A may regulate the commitment of mesenchymal stromal cells toward angio-osteogenesis by mirna-675-5P. Cytotherapy 2017, 19, 1412–1425. [Google Scholar] [CrossRef] [PubMed]
- Bellavia, D.; Conigliaro, A.; Salamanna, F.; Alessandro, R.; Fini, M.; Giavaresi, G. MiR-33a Controls hMSCS Osteoblast Commitment Modulating the Yap/Taz Expression Through EGFR Signaling Regulation. Cells 2019, 8, 1495. [Google Scholar] [CrossRef] [Green Version]
- Costa, V.; Carina, V.; Conigliaro, A.; Raimondi, L.; De Luca, A.; Bellavia, D.; Salamanna, F.; Setti, S.; Alessandro, R.; Fini, M.; et al. miR-31-5p Is a LIPUS-Mechanosensitive MicroRNA that Targets HIF-1α Signaling and Cytoskeletal Proteins. Int. J. Mol. Sci. 2019, 20, 1569. [Google Scholar] [CrossRef]
- Liu, X.D.; Deng, L.F.; Wang, J.; Qi, J.; Zhou, Q.; Wang, J.S.; Wei, L.; Zhu, Y.P.; Clemens, T. The regulation of hypoxia inducible factor-1alpha on osteoblast function in postmenopausal osteoporosis. Zhonghua Wai Ke Za Zhi 2007, 45, 1274–1278. [Google Scholar] [PubMed]
- Wang, Y.; Wan, C.; Gilbert, S.R.; Clemens, T.L. Oxygen sensing and osteogenesis. Ann. N. Y. Acad. Sci. 2007, 1117, 1–11. [Google Scholar] [CrossRef]
- Kim, H.H.; Lee, S.E.; Chung, W.J.; Choi, Y.; Kwack, K.; Kim, S.W.; Kim, M.S.; Park, H.; Lee, Z.H. Stabilization of hypoxia-inducible factor-1alpha is involved in the hypoxic stimuli-induced expression of vascular endothelial growth factor in osteoblastic cells. Cytokine 2002, 17, 14–27. [Google Scholar] [CrossRef]
- Wei, S.; Zhang, R.G.; Wang, Z.Y. Deferoxamine/magnesium modified β-tricalcium phosphate promotes the bone regeneration in osteoporotic rats. J. Biomater. Appl. 2022, 37, 838–849. [Google Scholar] [CrossRef]
- Peng, J.; Lai, Z.G.; Fang, Z.L.; Xing, S.; Hui, K.; Hao, C.; Jin, Q.; Qi, Z.; Shen, W.J.; Dong, Q.N.; et al. Dimethyloxalylglycine Prevents Bone Loss in Ovariectomized C57BL/6J Mice through Enhanced Angiogenesis and Osteogenesis. PLoS ONE 2014, 9, e112744. [Google Scholar] [CrossRef]
- Chen, D.F.; Li, Y.; Zhou, Z.Y.; Xing, Y.; Zhong, Y.G.; Zou, X.N.; Tian, W.; Zhang, C. Synergistic Inhibition of Wnt Pathway by HIF-1α and Osteoblast-Specific Transcription Factor Osterix (Osx) in Osteoblasts. PLoS ONE 2012, 7, e52948. [Google Scholar] [CrossRef]
- Yin, N.; Zhu, L.Z.; Ding, L.; Yuan, J.; Du, L.; Pan, M.; Xue, F.; Xiao, H. MiR-135-5p promotes osteoblast differentiation by targeting HIF1AN in MC3T3-E1 cells. Cell. Mol. Biol. Lett. 2019, 24, 51. [Google Scholar] [CrossRef] [PubMed]
- Cui, M.; Kanemoto, S.; Cui, X.; Kaneko, M.; Asada, R.; Matsuhisa, K.; Tanimoto, K.; Yoshimoto, Y.; Shukunami, C.; Imaizumi, K. OASIS modulates hypoxia pathway activity to regulate bone angiogenesis. Sci. Rep. 2015, 5, 16455. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, D.E.; Bosch-Marce, M.; Semenza, G.L.; Hadjiargyrou, M. Enhanced bone regeneration associated with decreased apoptosis in mice with partial HIF-1 alpha deficiency. J. Bone Miner. Res. 2006, 22, 366–374. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.D.; Cai, F.; Liu, L.; Zhang, Y.; Yang, A.L. microRNA-210 is involved in the regulation of postmenopausal osteoporosis through promotion of VEGF expression and osteoblast differentiation. Biol. Chem. 2015, 396, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Qiu, M.; Yang, L.; Yang, L.; Zhang, Y.; Mu, S.; Song, H. MicroRNA-1-3p enhances osteoblast differentiation of MC3T3-E1 cells by interacting with hypoxia-inducible factor 1α inhibitor (HIF1AN). Mech. Dev. 2020, 162, 103613. [Google Scholar] [CrossRef] [PubMed]
- Merceron, C.; Ranganathan, K.; Wang, E.; Tata, Z.; Makkapati, S.; Khan, M.P.; Mangiavini, L.; Yao, A.Q.; Castellini, L.; Levi, B.; et al. Hypoxia-inducible factor 2α is a negative regulator of osteoblastogenesis and bone mass accrual. Bone Res. 2019, 7, 91–104. [Google Scholar] [CrossRef]
- Arnett, T.R.; Gibbons, D.C.; Utting, J.C.; Orriss, I.R.; Hoebertz, A.; Rosendaal, M.; Meghji, S. Hypoxia is a major stimulator of osteoclast formation and bone resorption. J. Cell. Physiol. 2003, 196, 2–8. [Google Scholar] [CrossRef]
- Hsieh, T.P.; Sheu, S.Y.; Sun, J.S.; Chen, M.H. Icariin inhibits osteoclast differentiation and bone resorption by suppression of MAPKs/NF-κB regulated HIF-1α and PGE(2) synthesis. Phytomedicine 2011, 18, 176–185. [Google Scholar] [CrossRef]
- Knowles, H.J. Hypoxia-Induced Fibroblast Growth Factor 11 Stimulates Osteoclast-Mediated Resorption of Bone. Calcif. Tissue Int. 2017, 100, 382–391. [Google Scholar] [CrossRef]
- Knowles, H.J. The Adenosine A Receptor Drives Osteoclast-Mediated Bone Resorption in Hypoxic Microenvironments. Cells 2019, 8, 624. [Google Scholar] [CrossRef]
- Hulley, P.A.; Bishop, T.; Vernet, A.; Schneider, J.E.; Edwards, J.R.; Athanasou, N.A.; Knowles, H.J. Hypoxia-inducible factor 1-alpha does not regulate osteoclastogenesis but enhances bone resorption activity via prolyl-4-hydroxylase 2. J. Pathol. 2017, 242, 322–333. [Google Scholar] [CrossRef]
- Knowles, H.J. Hypoxic regulation of osteoclast differentiation and bone resorption activity. Hypoxia 2015, 373–382. [Google Scholar] [CrossRef]
- Sun, K.T.; Chen, M.Y.; Tu, M.G.; Wang, I.K.; Chang, S.S.; Li, C.Y. MicroRNA-20a regulates autophagy related protein-ATG16L1 in hypoxia-induced osteoclast differentiation. Bone 2015, 73, 145–153. [Google Scholar] [CrossRef]
- Morita, M.; Sato, Y.; Iwasaki, R.; Kobayashi, T.; Watanabe, R.; Oike, T.; Miyamoto, K.; Toyama, Y.; Matsumoto, M.; Nakamura, M.; et al. Selective Estrogen Receptor Modulators Suppress Hif1α Protein Accumulation in Mouse Osteoclasts. PLoS ONE 2016, 11, e0165922. [Google Scholar] [CrossRef]
- Tando, T.; Sato, Y.; Miyamoto, K.; Morita, M.; Kobayashi, T.; Funayama, A.; Kanaji, A.; Hao, W.; Watanabe, R.; Oike, T.; et al. Hif1α is required for osteoclast activation and bone loss in male osteoporosis. Biochem. Biophys. Res. Commun. 2016, 470, 391–396. [Google Scholar] [CrossRef]
- Hulley, P.A.; Papadimitriou-Olivgeri, I.; Knowles, H.J. Osteoblast-Osteoclast Coculture Amplifies Inhibitory Effects of FG-4592 on Human Osteoclastogenesis and Reduces Bone Resorption. JBMR. Plus 2020, 4, e10370. [Google Scholar] [CrossRef] [PubMed]
- Divieti Pajevic, P.; Krause, D.S. Osteocyte regulation of bone and blood. Bone 2019, 119, 13–18. [Google Scholar] [CrossRef]
- Gross, T.S.; Akeno, N.; Clemens, T.L.; Komarova, S.; Srinivasan, S.; Weimer, D.A.; Mayorov, S. Selected Contribution: Osteocytes upregulate HIF-1alpha in response to acute disuse and oxygen deprivation. J. Appl. Physiol. 2001, 90, 2514–2519. [Google Scholar] [CrossRef]
- Sherk, V.D.; Rosen, C.J. Senescent and apoptotic osteocytes and aging: Exercise to the rescue? Bone 2019, 121, 255–258. [Google Scholar] [CrossRef]
- Weinstein, R.S.; Wan, C.; Liu, Q.L.; Wang, Y.; Almeida, M.; O’Brien, C.A.; Thostenson, J.; Roberson, P.K.; Boskey, A.L.; Clemens, T.L.; et al. Endogenous glucocorticoids decrease skeletal angiogenesis, vascularity, hydration, and strength in aged mice. Aging Cell 2010, 9, 147–161. [Google Scholar] [CrossRef]
- Zhu, J.; Tang, Y.; Wu, Q.; Ji, Y.C.; Feng, Z.F.; Kang, F.W. HIF-1α facilitates osteocyte-mediated osteoclastogenesis by activating JAK2/STAT3 pathway in vitro. J. Cell. Physiol. 2019, 234, 21182–21192. [Google Scholar] [CrossRef]
- Song, X.; Tang, Y.; Zhu, J.; Tian, Y.; Song, Z.; Hu, X.; Hong, C.; Cai, Y.; Kang, F. HIF-1α induces hypoxic apoptosis of MLO-Y4 osteocytes via JNK/caspase-3 pathway and the apoptotic-osteocyte-mediated osteoclastogenesis in vitro. Tissue. Cell 2020, 67, 101402. [Google Scholar] [CrossRef]
- Zuo, G.L.; Zhang, L.F.; Jin, Q.; Kang, H.; Jia, P.; Chen, H.; Shen, X.; Guo, L.; Zhou, H.B.; Wang, J.S.; et al. Activation of HIFα Pathway in Mature Osteoblasts Disrupts the Integrity of the Osteocyte/Canalicular Network. PLoS ONE 2015, 10, e0121266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Loots, G.G.; Robling, A.G.; Chang, J.C.; Murugesh, D.K.; Bajwa, J.; Carlisle, C.; Manilay, J.O.; Wong, A.; Yellowley, C.E.; Genetos, D.C. Vhl deficiency in osteocytes produces high bone mass and hematopoietic defects. Bone 2018, 116, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Seeley, T.W.; Sternlicht, M.D.; Klaus, S.J.; Neff, T.B.; Liu, D.Y. Induction of erythropoiesis by hypoxia-inducible factor prolyl hydroxylase inhibitors without promotion of tumor initiation, progression, or metastasis in a VEGF-sensitive model of spontaneous breast cancer. Hypoxia 2017, 5, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Salmón-González, Z.; Anchuelo, J.; Borregán, J.C.; Del Real, A.; Sañudo, C.; García-Unzueta, M.T.; Riancho, J.A.; Valero, C. Hyperbaric Oxygen Therapy Does Not Have a Negative Impact on Bone Signaling Pathways in Humans. Healthcare 2021, 9, 1714. [Google Scholar] [CrossRef] [PubMed]
- Hassan, K.H.; Oppo, G.M.; Roselli, G.; Parra, E.; Mahmoud, P.; Homsi, M.; Nori, J. Applications of low pressure hyperbaric chamber oxygen therapy in osteoporosis. In Proceedings of the 7th International Scientific Forum, ISF 2017, Oxford, UK, 7–9 February 2017; University of Oxford: Oxford, UK, 2009. [Google Scholar]
- Ververidis, A.N.; Paraskevopoulos, K.; Keskinis, A.; Ververidis, N.A.; Molla Moustafa, R.; Tilkeridis, K. Bone marrow edema syndrome/transient osteoporosis of the hip joint and management with the utilization of hyperbaric oxygen therapy. J. Orthop. 2021, 22, 29–32. [Google Scholar] [CrossRef] [PubMed]
- Takemura, A.; Pajevic, P.D.; Egawa, T.; Teshigawara, R.; Hayashi, T.; Ishihara, A. Effects of mild hyperbaric oxygen on osteoporosis induced by hindlimb unloading in rats. J. Bone Miner. Metab. 2020, 38, 631–638. [Google Scholar] [CrossRef]
- Mutluoglu, M.; Sonmez, G.; Sivrioglu, A.K.; Ay, H. There may be a role for hyperbaric oxygen therapy in transient osteoporosis of the hip. Acta Orthop. Belg. 2012, 78, 685–687. [Google Scholar] [PubMed]
- Yagishita, K.; Jinno, T.; Koga, D.; Kato, T.; Enomoto, M.; Kato, T.; Muneta, T.; Okawa, A. Transient osteoporosis of the hip treated with hyperbaric oxygen therapy: A case series. Undersea Hyperb. Med. 2016, 43, 847–854. [Google Scholar]
- Imerb, N.; Thonusin, C.; Pratchayasakul, W.; Arunsak, B.; Nawara, W.; Aeimlapa, R.; Charoenphandhu, N.; Chattipakorn, N.; Chattipakorn, S.C. Hyperbaric oxygen therapy improves age induced bone dyshomeostasis in non-obese and obese conditions. Life Sci. 2022, 295, 120406. [Google Scholar] [CrossRef]
- Wang, L.; Shi, Q.; Cai, Y.; Chen, Q.; Guo, X.; Li, Z. Mechanical-chemical coupled modeling of bone regeneration within a biodegradable polymer scaffold loaded with VEGF. Biomech. Model Mechanobiol. 2020, 19, 2285–2306. [Google Scholar] [CrossRef]
- Liu, Y.; Huang, X.; Yu, H.; Yang, J.; Li, Y.; Yuan, X.; Guo, Q. HIF-1α-TWIST pathway restrains cyclic mechanical stretch-induced osteogenic differentiation of bone marrow mesenchymal stem cells. Connect Tissue Res 2019, 60, 544–554. [Google Scholar] [CrossRef]
- Sun, R.; Zhang, C.; Liu, Y.; Chen, Z.; Liu, W.; Yang, F.; Zeng, F.; Guo, Q. Demethylase FTO promotes mechanical stress induced osteogenic differentiation of BMSCs with up-regulation of HIF-1α. Mol. Biol. Rep. 2022, 49, 2777–2784. [Google Scholar] [CrossRef]
- Shimomura, S.; Inoue, H.; Arai, Y.; Nakagawa, S.; Fujii, Y.; Kishida, T.; Shin-Ya, M.; Ichimaru, S.; Tsuchida, S.; Mazda, O.; et al. Mechanical stimulation of chondrocytes regulates HIF-1α under hypoxic conditions. Tissue Cell 2021, 71, 101574. [Google Scholar] [CrossRef]
- Jing, X.; Yang, X.; Zhang, W.; Wang, S.; Cui, X.; Du, T.; Li, T. Mechanical loading induces HIF-1α expression in chondrocytes via YAP. Biotechnol. Lett. 2020, 42, 1645–1654. [Google Scholar] [CrossRef]
- Milkiewicz, M.; Doyle, J.L.; Fudalewski, T.; Ispanovic, E.; Aghasi, M.; Haas, T.L. HIF-1alpha and HIF-2alpha play a central role in stretch-induced but not shear-stress-induced angiogenesis in rat skeletal muscle. J. Physiol. 2007, 583, 753–766. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, S.; Wang, T. How the mechanical microenvironment of stem cell growth affects their differentiation: A review. Stem. Cell Res. Ther. 2022, 13, 415. [Google Scholar] [CrossRef]
- Evstatiev, R.; Gasche, C. Iron sensing and signalling. Gut 2012, 61, 933–952. [Google Scholar] [CrossRef]
- Zhou, L.; Zhao, B.; Zhang, L.; Wang, S.; Dong, D.; Lv, H.; Shang, P. Alterations in Cellular Iron Metabolism Provide More Therapeutic Opportunities for Cancer. Int. J. Mol. Sci. 2018, 19, 1545. [Google Scholar] [CrossRef]
- Taylor, M.; Qu, A.J.; Anderson, E.R.; Matsubara, T.; Martin, A.; Gonzalez, F.J.; Shah, Y.M. Hypoxia-Inducible Factor-2α Mediates the Adaptive Increase of Intestinal Ferroportin During Iron Deficiency in Mice. Gastroenterology 2011, 140, 2044–2055. [Google Scholar] [CrossRef]
- Chavez, J.C.; Baranova, O.; Lin, J.; Pichiule, P. The transcriptional activator hypoxia inducible factor 2 (HIF-2/EPAS-1) regulates the oxygen-dependent expression of erythropoietin in cortical astrocytes. J. Neurosci. 2006, 26, 9471–9481. [Google Scholar] [CrossRef]
- Renassia, C.; Peyssonnaux, C. New insights into the links between hypoxia and iron homeostasis. Curr. Opin. Hematol. 2019, 26, 125–130. [Google Scholar] [CrossRef]
- Peyssonnaux, C.; Nizet, V.; Johnson, R.S. Role of the hypoxia inducible factors HIF in iron metabolism. Cell Cycle 2008, 7, 28–32. [Google Scholar] [CrossRef] [Green Version]
- Mastrogiannaki, M.; Matak, P.; Peyssonnaux, C. The gut in iron homeostasis: Role of HIF-2 under normal and pathological conditions. Blood 2013, 122, 885–892. [Google Scholar] [CrossRef]
- Duarte, T.L.; Talbot, N.P.; Drakesmith, H. NRF2 and Hypoxia-Inducible Factors: Key Players in the Redox Control of Systemic Iron Homeostasis. Antioxid. Redox. Signal. 2021, 35, 433–452. [Google Scholar] [CrossRef]
- Schwartz, A.J.; Das, N.K.; Ramakrishnan, S.K.; Jain, C.; Jurkovic, M.T.; Wu, J.; Nemeth, E.; Lakhal-Littleton, S.; Colacino, J.A.; Shah, Y.M. Hepatic hepcidin/intestinal HIF-2α axis maintains iron absorption during iron deficiency and overload. J. Clin. Investig. 2019, 129, 336–348. [Google Scholar] [CrossRef]
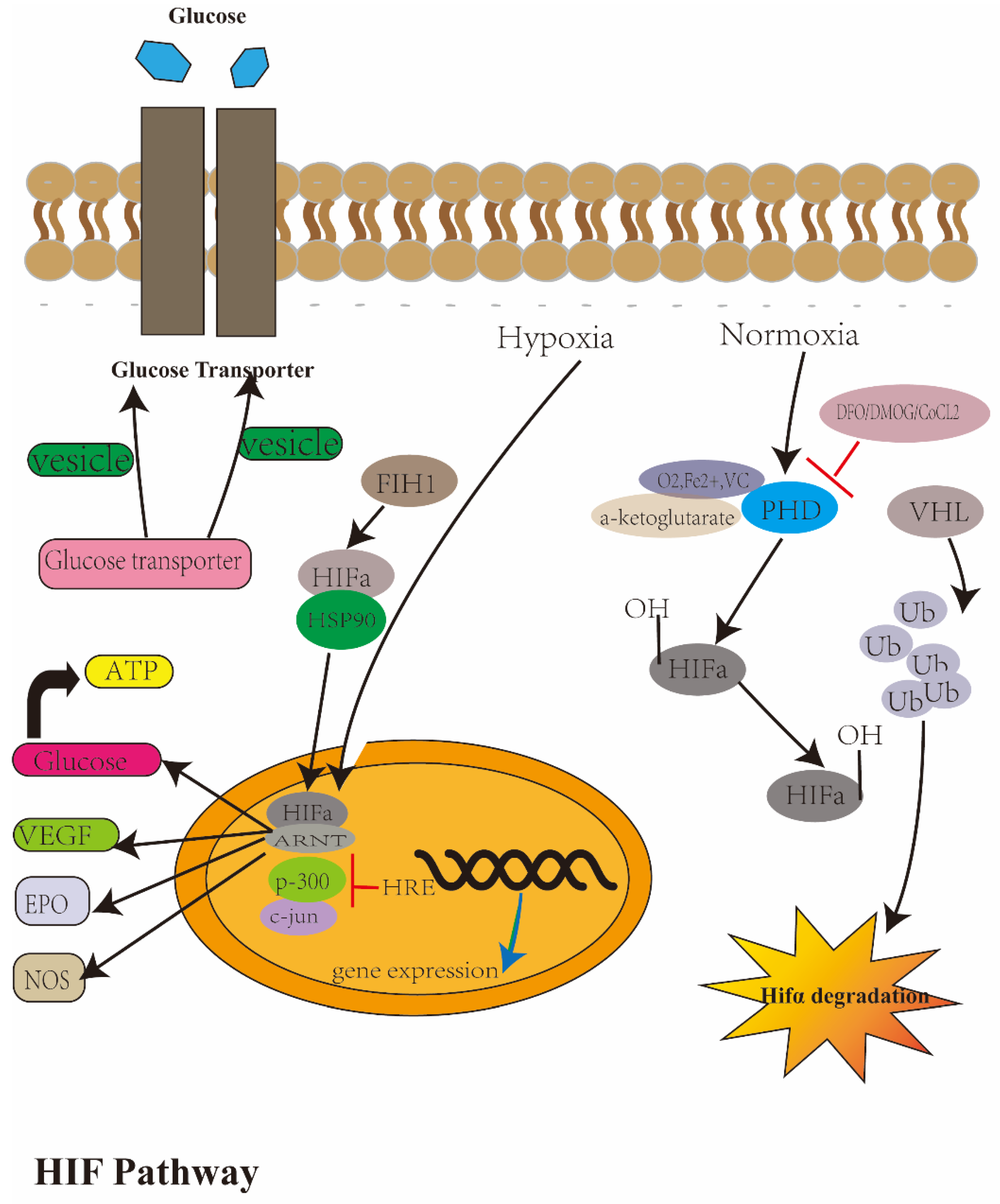

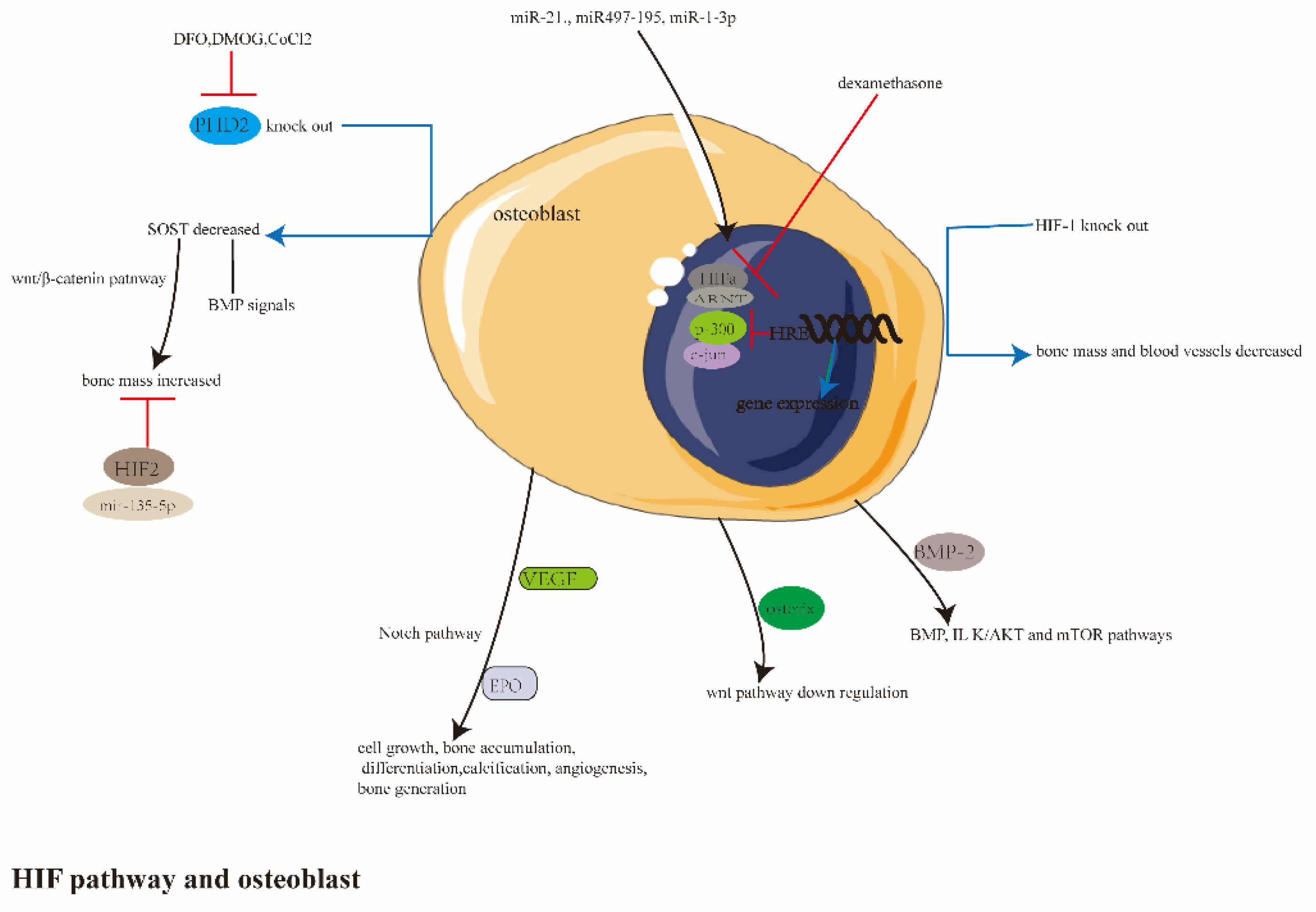


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Zhao, B.; Che, J.; Shang, P. Hypoxia Pathway in Osteoporosis: Laboratory Data for Clinical Prospects. Int. J. Environ. Res. Public Health 2023, 20, 3129. https://doi.org/10.3390/ijerph20043129
Wang J, Zhao B, Che J, Shang P. Hypoxia Pathway in Osteoporosis: Laboratory Data for Clinical Prospects. International Journal of Environmental Research and Public Health. 2023; 20(4):3129. https://doi.org/10.3390/ijerph20043129
Chicago/Turabian StyleWang, Jianping, Bin Zhao, Jingmin Che, and Peng Shang. 2023. "Hypoxia Pathway in Osteoporosis: Laboratory Data for Clinical Prospects" International Journal of Environmental Research and Public Health 20, no. 4: 3129. https://doi.org/10.3390/ijerph20043129




