The Role of Plasminogen Activator Inhibitor 1 in Predicting Sepsis-Associated Liver Dysfunction: An Observational Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Design and Settings
2.2. Ethics
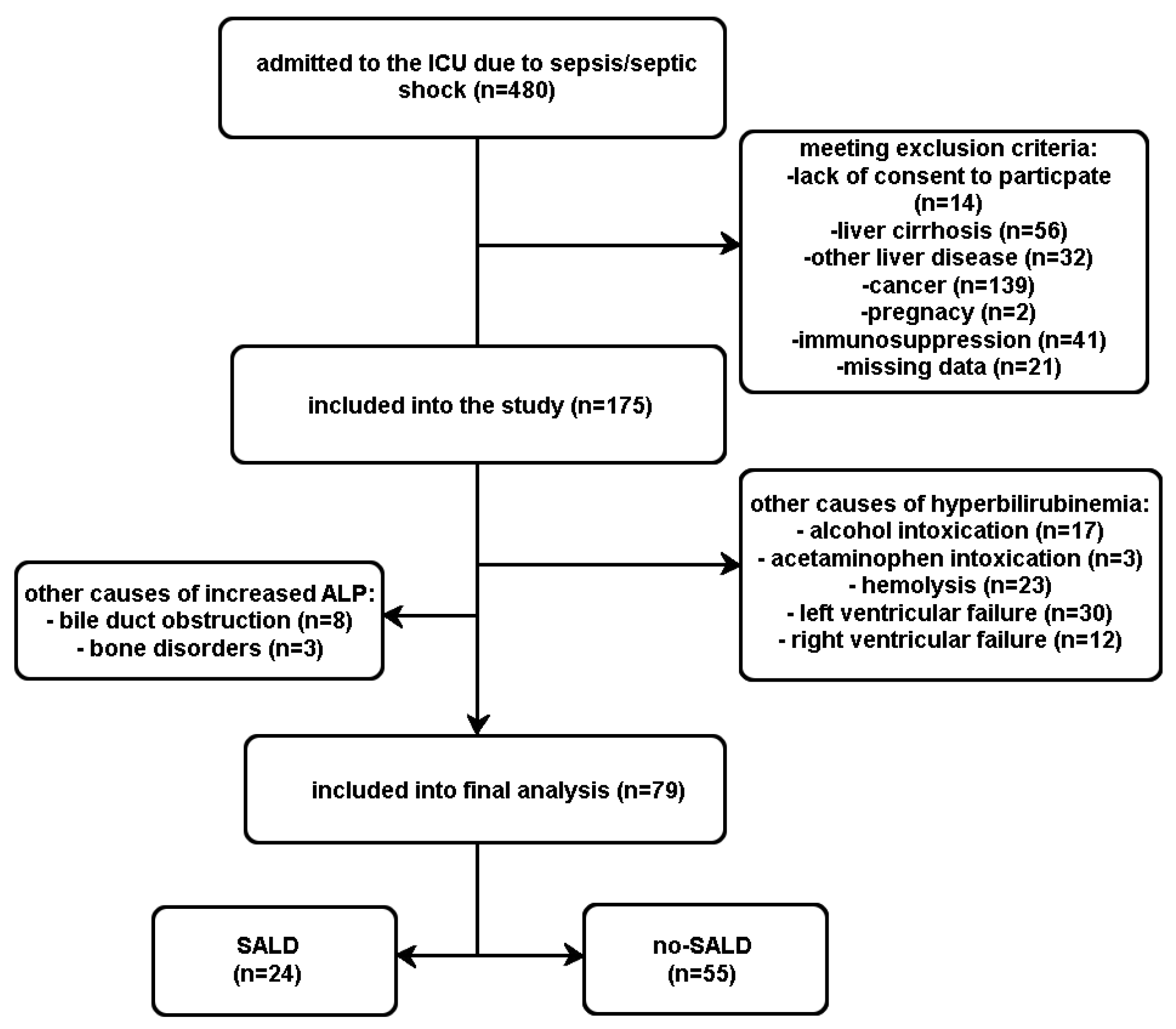
2.3. Participants
2.4. Outcomes
2.5. Analysed Biomarkers
2.5.1. IP-10
2.5.2. ET-1
2.5.3. Hepcidin
2.5.4. TAT
2.5.5. PAI-1
2.6. Statistical Analysis
3. Results
3.1. Comorbidities and Source of Sepsis
3.1.1. Comorbidities
3.1.2. Source of Sepsis
3.2. Routinely Measured Biomarkers
3.3. Analysed Biomarkers
3.4. PAI-1
3.4.1. PAI-1 Levels
3.4.2. PAI-1 as a Predictor of SALD
3.4.3. PAI-1 as a Predictor of Mortality
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kobashi, H.; Toshimori, J.; Yamamoto, K. Sepsis-associated liver injury: Incidence, classification and the clinical significance. Hepatol. Res. 2013, 43, 255–266. [Google Scholar] [CrossRef]
- Woznica, E.; Inglot, M.; Woznica, R.; Łysenko, L. Liver dysfunction in sepsis. Adv. Clin. Exp. Med. 2018, 27, 547–551. [Google Scholar] [CrossRef]
- Wang, D.; Yin, Y.; Yao, Y. Advances in sepsis-associated liver dysfunction. Burn. Trauma 2014, 2, 97–105. [Google Scholar] [CrossRef] [Green Version]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.D.; Coopersmith, C.M.; et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef]
- Lee, W.M.; Stravitz, R.T.; Larson, A.M. Introduction to the revised American Association for the Study of Liver Diseases Position Paper on acute liver failure 2011. Hepatology 2013, 55, 965–967. [Google Scholar] [CrossRef] [Green Version]
- Tapper, E.B.; Sengupta, N.; Bonder, A. The incidence and outcomes of ischemic hepatitis: A systematic review and metaanalisis. Am. J. Med. 2015, 128, 1314–1321. [Google Scholar] [CrossRef]
- Dellinger, R.P.; Levy, M.M.; Rhodes, A.; Annane, D.; Gerlach, H.; Opal, S.M.; Sevransky, J.E.; Sprung, C.L.; Douglas, I.S.; Jaeschke, R.; et al. Surviving sepsis campaign: Internationa guidelines for management of severe sepsis and septic shock: 2012. Crit. Care Med. 2013, 41, 580–637. [Google Scholar] [CrossRef]
- Taylor, F.B., Jr.; Toh, C.H.; Hoots, W.K.; Wada, H.; Levi, M. Scientific Subcommittee on Disseminated Intravascular Coagulation (DIC) of the International Society on Thrombosis and Haemostasis (ISTH): Towards definition, clinical and laboratory criteria, and a scoring system for disseminated intravascular coagulation. Thromb. Haemost. 2001, 86, 1327–1330. [Google Scholar] [CrossRef] [Green Version]
- Liu, M.; Guo, S.; Hibbert, J.M.; Jain, V.; Singh, N.; Wilson, N.O.; Stiles, J.K. CXCL10/IP-10 in Infectious Diseases Pathogenesis and Potential Therapeutic Implications. Cytokine Growth Factor Rev. 2011, 22, 121–130. [Google Scholar] [CrossRef]
- Herzig, S.D.; Luan, L.; Bohannon, J.K.; Toliver-Kinsky, T.E.; Guo, Y.; Sherwood, E.R. The role of CXCL10 in the pathogenesis of experimental septic shock. Crit. Care 2014, 18, R113. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Shen, J.; Man, K.; Chu, E.S.; Yau, T.O.; Sung, J.C.; Go, M.Y.; Deng, J.; Lu, L.; Wong, V.W.; et al. CXCL10 plays a key role as an inflammatory mediator and anon-invasive biomarker of non-alcoholic steatohepatitis. J. Hepatol. 2014, 61, 1365–1375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abe, T.; Fukuhara, T.; Wen, X.; Ninomiya, A.; Moriishi, K.; Maehara, Y.; Takeuchi, O.; Kawai, T.; Akira, S.; Matsuura, Y. CD44 participates in IP-10 induction in cells in which hepatitis C virus RNA is replicating, through an interaction with Toll-like receptor 2 and hyaluronan. J. Virol. 2012, 86, 6159–6170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, Y.; Wang, S.; Ma, J.W.; Lei, Z.; Zhu, H.F.; Lei, P.; Yang, Z.S.; Zhang, B.; Yao, X.X.; Shi, C.; et al. Hepatitis B virus protein X induced expression of the CXC chemokine IP-10 is mediated through activation of NF-kappaB and increases migration of leukocytes. J. Biol. Chem. 2010, 285, 12159–12168. [Google Scholar] [CrossRef] [Green Version]
- Piechota, M.; Banach, M.; Irzmanski, R.; Barylski, M.; Piechota-Urbanska, M.; Kowalski, J.; Pawlicki, L. Plasma Endothelin-1 Levels in Septic Patients. J. Intensive Care Med. 2007, 22, 232. [Google Scholar] [CrossRef] [PubMed]
- Kleniewska, P.; Piechota-Polanczyk, A.; Michalski, L.; Michalska, M.; Balcerczak, E.; Zebrowska, M.; Goraca, A. Influence of Block of NF-Kappa B Signaling Pathway on Oxidative Stress in the Liver Homogenates. Oxid. Med. Cell. Longev. 2013, 2013, 308358. [Google Scholar] [CrossRef] [Green Version]
- McKenna, S.; Gossling, M.; Bugarini, A.; Hill, E.; Anderson, A.L.; Rancourt, R.C.; Balasubramaniyan, N.; El Kasmi, K.C.; Wright, C.J. Endotoxemia induces IκBβ/NFκB dependent ET-1 expression in hepatic macrophages. J. Immunol. 2015, 195, 3866–3879. [Google Scholar] [CrossRef] [Green Version]
- Reynaert, H.; Thompson, M.G.; Thomas, T.; Geerts, A. Hepatic stellate cells: Role in microcirculation and pathophysiology of portal hypertension. Gut 2002, 50, 571–581. [Google Scholar] [CrossRef]
- Baveja, R.; Yokoyama, Y.; Korneszczuk, K.; Zhang, J.X.; Clemens, M.G. Endothelin-1 impairs oxygen delivery in livers from LPS-primed animals. Shock 2002, 17, 383–388. [Google Scholar] [CrossRef] [Green Version]
- Kali, A.; Charles, M.V.; Seetharam, R.S. Hepcidin—A novel biomarker with changing trends. Pharmacogn. Rev. 2015, 9, 35–40. [Google Scholar] [CrossRef] [Green Version]
- Sangkhae, V.; Nemeth, E. Regulation of the iron homeostatic hormone hepcidin. Adv. Nutr. 2017, 8, 126–136. [Google Scholar] [CrossRef] [Green Version]
- Michels, K.; Nemeth, E.; Ganz, T.; Mehrad, B. Hepcidin and Host Defense against Infectious Diseases. PLoS Pathog. 2015, 11, e1004998. [Google Scholar] [CrossRef] [Green Version]
- Spivak, I.; Arora, J.; Meinzer, C.; Durkalski-Mauldin, V.; Lee, W.M.; Trautwein, C.; Fontana, R.J.; Strnad, P.; Acute Liver Failure Study Group (ALFSG). Low serum hepcidin is associated with reduced short-term survival in adults with acute liver failure. Hepatology 2019, 69, 2136–2149. [Google Scholar] [CrossRef]
- Hoppensteadt, D.; Tsuruta, K.; Cunanan, J.; Hirman, J.; Kaul, I.; Osawa, Y.; Fareed, J. Thrombin generation mediators and markers in sepsis-associated coagulopathy and their modulation by recombinant thrombomodulin. Clin. Appl. Thromb. Hemost. 2014, 20, 129–135. [Google Scholar] [CrossRef]
- Iba, T.; Saitoh, D. Efficacy of antithrombin in preclinical and clinical applications for sepsis-associated disseminated intravascular coagulation. J. Intensive Care 2014, 2, 66. [Google Scholar] [CrossRef] [Green Version]
- Pelzer, H.; Schwarz, A.; Heimburger, N. Determination of human thrombin-antithrombin III complex in plasma with an enzyme-linked immunosorbent assay. Thromb. Haemost. 1988, 59, 101–106. [Google Scholar] [CrossRef]
- Kalambokis, G.N.; Oikonomou, A.; Baltayiannis, G.; Christou, L.; Kolaitis, N.I.; Tsianos, E.V. Thrombin generation measured as thrombin–antithrombin complexes predicts clinical outcomes in patients with cirrhosis. Hepatol. Res. 2016, 46, E36–E44. [Google Scholar] [CrossRef] [Green Version]
- Hack, C.E. Fibrinolysis in disseminated intravascular coagulation. Semin. Thromb. Hemost. 2001, 27, 633–638. [Google Scholar] [CrossRef]
- Dimova, E.Y.; Kietzmann, T. Metabolic, hormonal and environmental regulation of plasminogen activator inhibitor-1 (PAI-1) expression: Lessons from the liver. Thromb. Haemost. 2008, 100, 992–1006. [Google Scholar] [CrossRef]
- Ohkura, N.; Oishi, K.; Nakakuki, Y. Lipopolysaccharide-induced plasma PAI-1 increase does not correlate with PAI-1 synthesised de novo in the liver. Thromb. Res. 2013, 132, 398–399. [Google Scholar] [CrossRef]
- Imagawa, S.; Fujii, S.; Dong, J.; Furumoto, T.; Kaneko, T.; Zaman, T.; Satoh, Y.; Tsutsui, H.; Sobel, B.E. Hepatocyte Growth Factor Regulates E Box–Dependent Plasminogen Activator Inhibitor Type 1 Gene Expression in HepG2 Liver Cells. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 2407–2413. [Google Scholar] [CrossRef] [Green Version]
- Canbay, A.; Tacke, F.; Hadem, J.; Trautwein, C.; Gerken, G.; Manns, M.P. Acute Liver Failure. Dtsch. Arztebl. Int. 2011, 108, 714–720. [Google Scholar] [CrossRef] [PubMed]
- Koyama, K.; Madoiwa, S.; Nunomiya, S.; Koinuma, T.; Wada, M.; Sakata, A.; Ohmori, T.; Mimuro, J.; Sakata, Y. Combination of thrombin-antithrombin complex, plasminogen activator inhibitor-1, and protein C activity for early identification of severe coagulopathy in initial phase of sepsis: A prospective observational study. Crit. Care 2014, 18, R13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Katayama, S.; Koyama, K.; Shima, J.; Tonai, K.; Goto, Y.; Koinuma, T.; Nunomiya, S. Thrombomodulin, Plasminogen Activator Inhibitor-1 and Protein C Levels, and Organ Dysfunction in Sepsis. Crit. Care Explor. 2019, 1, e0013. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, K.; Kitamura, T.; Nakamura, Y.; Matsumoto, J.; Kawano, Y.; Ishikura, H. Usefulness of plasminogen activator inhibitor-1 as a predictive marker of mortality in sepsis. J. Int. Care 2017, 4, 42. [Google Scholar] [CrossRef]
- Hoshino, K.; Nakashio, M.; Maruyama, J.; Irie, Y.; Kawano, Y.; Ishikura, H. Validating plasminogen activator inhibitor-1 as poor prognostic factor in sepsis. Acute Med. Surg. 2020, 7, e581. [Google Scholar] [CrossRef]
- Madoiwa, S.; Nunomiya, S.; Ono, T.; Shintani, Y.; Ohmori, T.; Mimuro, J.; Sakata, Y. Plasminogen activator inhibitor 1 promotes a poor prognosis in sepsis-induced disseminated intravascular coagulation. Int. J. Hematol. 2006, 84, 398–405. [Google Scholar] [CrossRef]
- Saito, S.; Uchino, S.; Hayakawa, M.; Yamakawa, K.; Kudo, D.; Iizuka, Y.; Sanui, M.; Takimoto, K.; Mayumi, T.; Sasabuchi, Y. Epidemiology of disseminated intravascular coagulation in sepsis and validation of scoring systems. J. Crit. Care 2019, 50, 23–30. [Google Scholar] [CrossRef]



| All Patients (n = 79) | no-SALD (n = 55) | SALD (n = 24) | p | |
|---|---|---|---|---|
| Age (years) | 0.63 * | |||
| Me (Q1–Q3) | 66 (57–76) | 67 (60–74) | 61 (46–78) | |
| Min–Max | 19–88 | 23–88 | 19–88 | |
| Sex | 0.83 ** | |||
| (male/female) | 31/48 | 22(40%)/33(60%) | 9(38%)/15(62%) | |
| SOFA (pts.) | 0.09 * | |||
| Me (Q1–Q3) | 10 (7–13) | 9 (7–13) | 12(9–14) | |
| Min–Max | 3–17 | 3–17 | 3–15 | |
| APACHE II (pts.) | 0.98 | |||
| Me (Q1–Q3) | 24 (15–30) | 24 (15–30) | 24 (15–28) | |
| Min–Max | 15–30 | 8–44 | 7–37 | |
| Sepsis/Septic shock | 32/47 | 25 (78%)/30 (64%) | 7 (22%)/17 (36%) | 0.18 ** |
| 28 day survival | 49 (62 ± 5.5%) | 37 (67.3 ± 6.3%) | 12 (50.0 ± 10.2%) | 0.12 *** |
| Comorbidities | ||||
|---|---|---|---|---|
| All Patients (n = 79) | no-SALD (n = 55) | SALD (n = 24) | p | |
| HTA | 37 (46.84%) | 26 (47.27%) | 11 (45.83%) | 0.89 |
| DM | 23 (29.11%) | 19 (34.55%) | 4 (16.67%) | 0.18 |
| CCD | 3 (3.8%) | 1 (1.82%) | 2 (8.33%) | 0.22 |
| CA | 8 (10.13%) | 6 (10.91%) | 2 (8.33%) | 0.99 |
| OBS | 8 (10.13%) | 5 (9.09%) | 3 (12.5%) | 0.69 |
| CKD | 18 (23.68%) | 15 (27.27%) | 3 (12.5%) | 0.15 |
| CPD | 10 (12.66%) | 8 (14.55%) | 2 (8.33%) | 0.72 |
| ARY | 16 (20.25%) | 11 (22%) | 5 (20.83%) | 0.99 |
| ALC | 10 (12.66%) | 7 (12.73%) | 3 (12.5%) | 0.99 |
| NEUR | 4 (5.06%) | 4 (7.27%) | 0 | 0.31 |
| OTH | 8 (10.13%) | 5 (9.09%) | 3 (12.5%) | 0.69 |
| Source of Sepsis | ||||
|---|---|---|---|---|
| All Patients (n = 79) | No-SALD (n = 55) | SALD (n = 24) | p | |
| Abdominal infection (ABD) | 32 (40.51%) | 23 (41.82%) | 9 (37.5%) | 0.91 |
| Pulmonary infection (PNEU) | 27 (34.18%) | 17 (30.91%) | 10 (41.67%) | 0.50 |
| Soft tissue infection (TISS) | 3 (3.8%) | 2 (3.64%) | 1 (4.17%) | 0.99 |
| Neuroinfection (NEUR) | 4 (5.06%) | 3 (5.45%) | 1 (4.17%) | 0.99 |
| Bloodstream infection (BSI) | 3 (3.8%) | 0 | 3 (12.5%) | 0.026 |
| Urinary tract infection (UTI) | 6 (7.59%) | 6 (10.91%) | 0 | 0.17 |
| UTI. PNEU | 1 (1.27%) | 1 (1.82%) | 0 | 0.99 |
| NEUR. BSI | 1 (1.27%) | 1 (1.82%) | 0 | 0.99 |
| PNEU. BSI | 2 (2.53%) | 2 (3.64%) | 0 | 0.99 |
| Normal Plasma Values | no-SALD (n = 55) | SALD (n = 24) | p | |
|---|---|---|---|---|
| Me (Q1–Q3) | Me (Q1–Q3) | |||
| AST (U/L) | 5–34 | 51 (29–155) | 124 (38–325) | 0.020 |
| ALT (U/L) | 0–55 | 46 (20–81) | 61.5 (27–102) | 0.16 |
| Albumin (g/dL) | 3.2–4.6 | 2.35 (2.0–2.65) | 2.3 (1.8–2.7) | 0.85 |
| AT III (%) | 80–120 | 58.5 (45–81.1) | 53.3 (41.3–61.3) | 0.08 |
| INR | 0.9–1.3 | 1.26 (1.12–1.45) | 1.35 (1.16–1.83) | 0.13 |
| Prothrombin ratio (%) | 80–114 | 79.7 (66.6–87.8) | 73.7 (55.8–86.3) | 0.27 |
| APTT (s) | 21–30.1 | 36.9 (30.9–46.7) | 44.3 (33.9–53.6) | 0.09 |
| ALP (U/L) | <270 | 75 (54–148) | 147.5 (94–206) | 0.003 |
| Lactate (mmol/L) | <2 | 2 (1.3–3.6) | 4.75 (1.6–8.95) | 0.041 |
| PCT (ng/mL) | <0.05 | 4 (1.7–17.8) | 28 (3.1–71.3) | 0.0144 |
| BIL (mg/dL) | 0.3–1.2 | 0.70 (0.50–1.30) | 2.25 (1.10–4.16) | <0.001 |
| CRP (mg/L) | <5 | 239.8 (141.8–322.9) | 247.2 (155–318) | 0.44 |
| Normal Plasma Values | All Patients (n = 79) | no-SALD (n = 55) | SALD (n = 24) | p | |
|---|---|---|---|---|---|
| IP-10 (pg/mL) | 47–382 | 932.7 ± 1643.5 | 321.9 (198.7–796.9) | 315.4 (140.7–961.1) | 0.99 |
| ET-1 (pg/mL) | 0.58–1.96 | 2.06 ± 1.60 | 1.72 (1.19–2.45) | 1.54 (1.06–2.39) | 0.79 |
| PAI-1 (ng/mL) | 0.99–16.9 | 38.3 ± 53.3 | 13.1 (6.5–29.9) | 51.6 (9.9–74.9) | 0.020 |
| TAT (ng/mL) | 0.5–10 | 21.3 ± 116.6 | 4.3 (2.6–6.5) | 3.75 (2.83–5.75) | 0.81 |
| Hepcidin (ng/mL) | 82.4–56700 | 221.8 ± 167.1 | 195 (101.1–321.9) | 158.2 (93.5–217.9) | 0.35 |
| Variables | Univariate | |||
|---|---|---|---|---|
| HR | 95% CI | p | ||
| Age | 1.04 | 1.01 | 1.07 | 0.011 |
| SOFA | 1.14 | 1.03 | 1.27 | 0.009 |
| APACHE II | 1.06 | 1.02 | 1.10 | 0.003 |
| Lactate (mmol/L) | 1.14 | 1.06 | 1.22 | <0.001 |
| CRP (mg/L) | 1.00 | 1.00 | 1.00 | 0.53 |
| PCT (ng/mL) | 1.00 | 1.00 | 1.00 | 0.98 |
| BIL (mg/dL) | 1.12 | 0.94 | 1.35 | 0.21 |
| AST (U/L) | 1.00 | 1.00 | 1.00 | 0.82 |
| ALP (U/L) | 1.00 | 1.00 | 1.00 | 0.96 |
| PAI-1 (ng/mL) | 1.01 | 1.00 | 1.01 | 0.007 |
| Sex (ref. M) | 1.78 | 0.87 | 3.65 | 0.11 |
| SALD (ref. no) | 1.80 | 0.87 | 3.75 | 0.12 |
| PAI-1 (ref. ≤ 48.73) | 2.97 | 1.42 | 6.19 | 0.004 |
| Multivariate | ||||
| Age | 1.04 | 1.02 | 1.07 | 1.002 |
| Lactate (mmol/L) | 1.18 | 1.09 | 1.29 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Woźnica-Niesobska, E.; Leśnik, P.; Janc, J.; Zalewska, M.; Łysenko, L. The Role of Plasminogen Activator Inhibitor 1 in Predicting Sepsis-Associated Liver Dysfunction: An Observational Study. Int. J. Environ. Res. Public Health 2023, 20, 4846. https://doi.org/10.3390/ijerph20064846
Woźnica-Niesobska E, Leśnik P, Janc J, Zalewska M, Łysenko L. The Role of Plasminogen Activator Inhibitor 1 in Predicting Sepsis-Associated Liver Dysfunction: An Observational Study. International Journal of Environmental Research and Public Health. 2023; 20(6):4846. https://doi.org/10.3390/ijerph20064846
Chicago/Turabian StyleWoźnica-Niesobska, Ewa, Patrycja Leśnik, Jarosław Janc, Małgorzata Zalewska, and Lidia Łysenko. 2023. "The Role of Plasminogen Activator Inhibitor 1 in Predicting Sepsis-Associated Liver Dysfunction: An Observational Study" International Journal of Environmental Research and Public Health 20, no. 6: 4846. https://doi.org/10.3390/ijerph20064846






