First Known Report of mcr-Harboring Enterobacteriaceae in the Dominican Republic
Abstract
1. Introduction
2. Materials and Methods
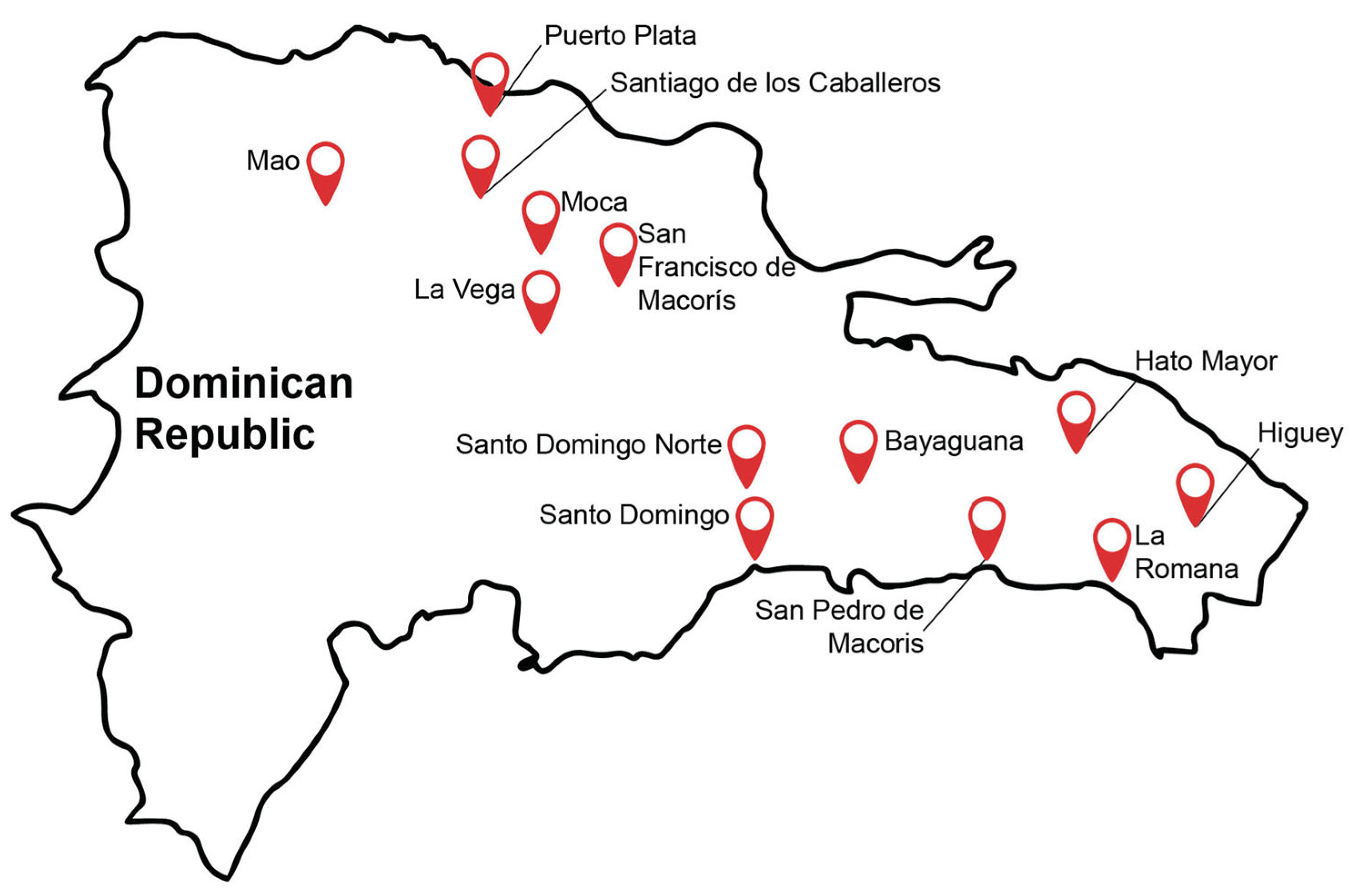
2.1. Sample Collection
2.2. Sample Preparation and Enrichment
2.3. Enterobacteriaceae Detection and Isolation
2.4. Salmonella Detection and Isolation
2.5. DNA Extraction from Samples
2.6. DNA Extraction from the Isolates
2.7. Multiplex RT-PCR for mcr Gene Detection
2.8. Whole Genome Sequencing
3. Results
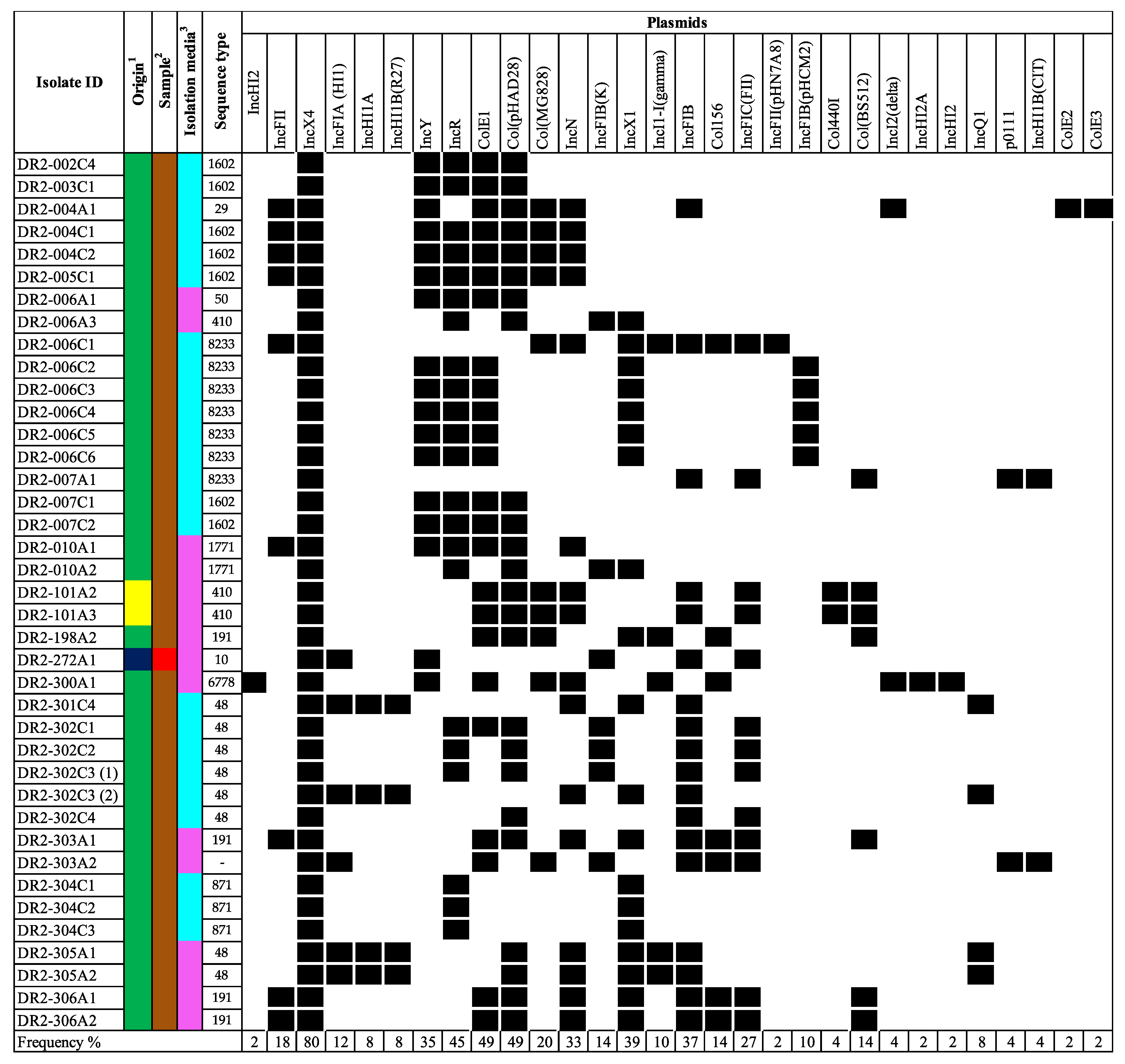
3.1. mcr Prevalence at the Isolate Level
3.2. Whole Genome Sequencing Analysis for mcr Confirmation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aslam Khurshid, M.; Arshad, M.I.; Muzammil, S.; Rasool, M.; Yasmeen, N.; Shah, T.; Chaudhry, T.H.; Rasool, M.H.; Shahid, A.; Xueshan, X.; et al. Ntibiotic Resistance: One Health One World Outlook. Front. Cell. Infect. Microbiol. 2021, 11, 771510. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.J.; Thottathil, S.E.; Newman, T.B. Antibiotics Overuse in Animal Agriculture: A Call to Action for Health Care Providers. Am. J. Public Health 1971, 105, 2409–2410. [Google Scholar] [CrossRef] [PubMed]
- Thumbi Njenga, M.K.; Marsh, T.L.; Noh, S.; Otiang, E.; Munyua, P.; Ochieng, L.; Ogola, E.; Yoder, J.; Audi, A.; Montgomery, J.M.; et al. Linking Human Health and Livestock Health: A “One-Health” Platform for Integrated Analysis of Human Health, Livestock Health, and Economic Welfare in Livestock Dependent Communities. PLoS ONE 2015, 10, e0120761. [Google Scholar] [CrossRef]
- Scott Acuff, G.; Bergeron, G.; Bourassa, M.W.; Gill, J.; Graham, D.W.; Kahn, L.H.; Morley, P.S.; Salois, M.J.; Simjee, S.; Singer, R.S.; et al. Critically Important Antibiotics: Criteria and Approaches for Measuring and Reducing Their Use in Food Animal Agriculture. Ann. N. Y. Acad. Sci. 2019, 1441, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.Y.; Wang, Y.; Walsh, T.R.; Yi, L.X.; Zhang, R.; Spencer, J.; Doi, Y.; Tian, G.; Dong, B.; Huang, X.; et al. Emergence of Plasmid-Mediated Colistin Resistance Mechanism mcr-1 in Animals and Human Beings in China: A Microbiological and Molecular Biological Study. Lancet Infect. Dis. 2016, 16, 161–168. [Google Scholar] [CrossRef]
- Gelbíčová Baráková, A.; Florianová, M.; Jamborová, I.; Zelendová, M.; Pospíšilová, L.; Koláčková, I.; Karpíšková, R. Dissemination and Comparison of Genetic Determinants of mcr-Mediated Colistin Resistance in Enterobacteriaceae via Retailed Raw Meat Products. Front. Microbiol. 2019, 10, 2824. [Google Scholar] [CrossRef]
- Alba Leekitcharoenphon, P.; Franco, A.; Feltrin, F.; Ianzano, A.; Caprioli, A.; Stravino, F.; Hendriksen, R.S.; Bortolaia, V.; Battisti, A. Molecular Epidemiology of mcr-Encoded Colistin Resistance in Enterobacteriaceae from Food-Producing Animals in Italy Revealed through the EU Harmonized Antimicrobial Resistance Monitoring. Front. Microbiol. 2018, 9, 1217. [Google Scholar] [CrossRef]
- Carroll Gaballa, A.; Guldimann, C.; Sullivan, G.; Henderson, L.O.; Wiedmann, M. Identification of Novel Mobilized Colistin Resistance Gene mcr-9 in a Multidrug-Resistant, Colistin-Susceptible Salmonella enterica Serotype Typhimurium Isolate. mBio 2019, 10, e00853-19. [Google Scholar] [CrossRef]
- Wang, C.; Feng, Y.; Liu, L.; Wei, L.; Kang, M.; Zong, Z. Identification of Novel Mobile Colistin Resistance Gene mcr-10. Emerg. Microbes Infect. 2020, 9, 508–516. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Discovery of First mcr-1 Gene in E. coli Bacteria Found in a Human in United States; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2016. [Google Scholar]
- Centers for Disease Control and Prevention. Newly Reported Gene, mcr-1, Threatens Last-Resort Antibiotics; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2016. [Google Scholar]
- Webb Kim, J.Y.; Tagg, K.A.; Kapsak, C.J.; Tobolowsky, F.; Birhane, M.G.; Francois Watkins, L.; Folster, J.P. Fourteen mcr-1-Positive Salmonella enterica Isolates Recovered from Travelers Returning to the United States from the Dominican Republic. Microbiol. Resour. Announc. 2022, 11, e0011822. [Google Scholar] [CrossRef]
- Tobolowsky Friedman, C.R.; Ryan, M.; Birhane, M.; Chen, J.; Webb, H.E.; Beukelman, R.A.; Bokanyi, R.; Byrd, D.J.; Connor, D.; Hanna, S.; et al. The Emergence of Mobile Colistin Resistance (mcr) Genes among Enteric Pathogens in the United States—2008–2019. Open Forum Infect. Dis. 2020, 7, S416. [Google Scholar] [CrossRef]
- Sharafi, T.; Ardebili, A. Plastic Binding Feature of Polymyxins: The Effect on MIC Susceptibility Measurements. Infect. Drug Resist. 2019, 12, 2649–2653. [Google Scholar] [CrossRef]
- Webb, H.E.; Granier, S.A.; Marault, M.; Millemann, Y.; den Bakker, H.C.; Nightingale, K.K.; Bugarel, M.; Ison, S.A.; Scott, H.M.; Loneragan, G.H. Dissemination of the mcr-1 Colistin Resistance Gene. Lancet Infect. Dis. 2016, 16, 144–145. [Google Scholar] [CrossRef]
- Perrin-Guyomard Granier, S.A.; Slettemeås, J.S.; Anjum, M.; Randall, L.; AbuOun, M.; Pauly, N.; Irrgang, A.; Hammerl, J.A.; Kjeldgaard, J.S.; Hammerum, A.; et al. Multicentre Evaluation of a Selective Isolation Protocol for Detection of mcr-Positive E. coli and Salmonella spp. in Food-Producing Animals and Meat. Lett. Appl. Microbiol. 2022, 75, 224–233. [Google Scholar] [CrossRef]
- Jolley, K.A.; Maiden, M.C. BIGSdb: Scalable analysis of bacterial genome variation at the population level. BMC Bioinform. 2010, 11, 595. [Google Scholar] [CrossRef]
- Migura-Garcia González-López, J.J.; Martinez-Urtaza, J.; Aguirre Sánchez, J.R.; Moreno-Mingorance, A.; Perez de Rozas, A.; Höfle, U.; Ramiro, Y.; Gonzalez-Escalona, N. mcr-Colistin Resistance Genes Mobilized by IncX4, IncHI2, and IncI2 Plasmids in Escherichia coli of Pigs and White Stork in Spain. Front. Microbiol. 2019, 10, 3072. [Google Scholar] [CrossRef]
- Zhang Chen, L.; Wang, J.; Yassin, A.K.; Butaye, P.; Kelly, P.; Gong, J.; Guo, W.; Li, J.; Li, M.; Yang, F.; et al. Molecular Detection of Colistin Resistance Genes (mcr-1, mcr-2 and mcr-3) in Nasal/oropharyngeal and Anal/cloacal Swabs from Pigs and Poultry. Sci. Rep. 2018, 8, 3705–3709. [Google Scholar] [CrossRef]
- Schoenmakers, K. How China Is Getting Its Farmers to Kick Their Antibiotics Habit. Nature 2020, 586, S60–S62. [Google Scholar] [CrossRef]
- Kieffer, N.; Aires-De-Sousa, M.; Nordmann, P.; Poirel, L. High Rate of mcr-1-Producing Escherichia coli and Klebsiella pneumoniae among Pigs, Portugal. Emerg. Infect. Dis. 2017, 23, 2023–2029. [Google Scholar] [CrossRef]
- Rhouma, M.; Beaudry, F.; Thériault, W.; Letellier, A. Colistin in Pig Production: Chemistry, Mechanism of Antibacterial Action, Microbial Resistance Emergence, and One Health Perspectives. Front. Microbiol. 2016, 7, 1789. [Google Scholar] [CrossRef]
- Rhouma, M.; Fairbrother, J.M.; Beaudry, F.; Letellier, A. Post Weaning Diarrhea in Pigs: Risk Factors and Non-Colistin-Based Control Strategies. Acta Vet. Scand. 2017, 59, 31. [Google Scholar] [CrossRef] [PubMed]
- Perez, F.; (Food Safety Department, Ministry of Agriculture, Santo Domingo, Dominican Republic). Personal communication, 2017.
- Sun Fang, L.-X.; Wu, Z.; Deng, H.; Yang, R.-S.; Li, X.-P.; Li, S.-M.; Liao, X.-P.; Feng, Y.; Liu, Y.-H. Genetic Analysis of the IncX4 Plasmids: Implications for a Unique Pattern in the mcr-1 Acquisition. Sci. Rep. 2017, 7, 424–429. [Google Scholar] [CrossRef]
- Maciuca Cummins, M.L.; Cozma, A.P.; Rimbu, C.M.; Guguianu, E.; Panzaru, C.; Licker, M. Genetic Features of mcr-1 Mediated Colistin Resistance in CMY-2-Producing Escherichia coli from Romanian Poultry. Front. Microbiol. 2019, 10, 2267. [Google Scholar] [CrossRef] [PubMed]
- Shafiq Huang, J.; Sadeeq Ur Rahman Jan Mohammad Shah Chen, L.; Gao, Y.; Wang, M.; Wang, L. High Incidence of Multidrug-Resistant Escherichia coli Coharboring mcr-1 and blaCTX-M-15 Recovered from Pigs. Infect. Drug Resist. 2019, 12, 2135–2149. [Google Scholar] [CrossRef] [PubMed]
- Matamoros van Hattem, J.; Arcilla, M.; Willemse, N.; Melles, D.; Penders, J.; Vinh, T.; Hoa, N.; Jong, M.; Schultsz, C. Global Phylogenetic Analysis of Escherichia coli and Plasmids Carrying the mcr-1 Gene Indicates Bacterial Diversity but Plasmid Restriction. Sci. Rep. 2017, 7, 15364. [Google Scholar] [CrossRef]
- García-Meniño García, V.; Mora, A.; Díaz-Jiménez, D.; Flament-Simon, S.C.; Alonso, M.P.; Blanco, J.E.; Blanco, M.; Blanco, J. Swine Enteric Colibacillosis in Spain: Pathogenic Potential of mcr-1 ST10 and ST131 E. coli Isolates. Front. Microbiol. 2018, 9, 2659. [Google Scholar] [CrossRef]
- Day, M.J.; Hopkins, K.L.; Wareham, D.W.; Toleman, M.A.; Elviss, N.; Randall, L.; Teale, C.; Cleary, P.; Wiuff, C.; Doumith, M.; et al. Extended-Spectrum β-lactamase-Producing Escherichia coli in Human-Derived and Foodchain-Derived Samples from England, Wales, and Scotland: An Epidemiological Surveillance and Typing Study. Lancet Infect. Dis. 2019, 19, 1325–1335. [Google Scholar] [CrossRef]
- Collignon, P.; McEwen, S. One Health-Its Importance in Helping to Better Control Antimicrobial Resistance. Trop. Med. Infect. Dis. 2019, 4, 22. [Google Scholar] [CrossRef]


| Primer/Probe | Sequence |
|---|---|
| * mcr126-F1 | 5′-GTAYTCTGTGCCGTGTATGTT-3′ |
| * mcr126-R2 | 5′-TCCATCACGCCTTTTGAGTC-3′ |
| mcr3-F1 | 5′-AAAACTGCACRGATGAAGAGC-3′ |
| mcr3-R2 | 5′-TCATCTCTSCAATCACKAAATC-3′ |
| mcr4-F1 | 5′-TTCAAAATTGCAGTCAAGAAGAAC-3′ |
| mcr4-R2 | 5′-ACCACTTCACTGAGAATAAAATC-3′ |
| mcr5-F1 | 5′-ACACGGCGCTGCTGTAC-3′ |
| mcr5-R2 | 5′-TATGCCATGGAGATACAGGC-3′ |
| mcr7-F1 | 5′-AACTGCAGCGATGAAGAGC-3′ |
| mcr7-R2 | 5′-ATCATCTCTGCTATGACAAAATC-3′ |
| mcr8-F1 | 5′-GATGCGTGACGTTGCTATG-3′ |
| mcr8-R2 | 5′-TGTGCCATGAAGATATATTCCG-3′ |
| ** mcr126-P3 | 5′/56ROXN/TATGATGTCGATACCGCCAAATACCAAG/3IAbRQSp-3′ |
| *** mcr347-P3 | 5′-/5HEX/AACACCTAYGACAACACYAT/3IABkFQ/-3′ |
| **** mcr58-P3 | 5′-/56-FAM/TTTCYGATCATGGRGAATCGCT/3IABkFQ/-3′ |
| Animal Source 1 | Feces % (Positive/Total) | Feed % (Positive/Total) | Water % (Positive/Total) | Meat % (Positive/Total) | Total % (Positive/Total) |
|---|---|---|---|---|---|
| Beef cattle | 52.5 (21/40) | 0.0 (0/2) | 60.0 (3/5) | 65.2 (30/46) | 58.1 (54/93) |
| Swine | 97.4 (38/39) | 46.2 (6/13) | 50.0 (3/6) | 66.7 (34/51) | 74.3 (81/109) |
| Poultry | 88.9 (32/36) | 81.8 (9/11) | 75.0 (3/4) | 70.7 (41/58) | 78.0 (85/109) |
| Total per sample type | 79.1 (91/115) | 57.7 (15/26) | 60.0 (9/15) | 67.7 (105/155) | 70.7 (220/311) |
| Animal Source | Feces % (Positive/Total) | Feed % (Positive/Total) | Water % (Positive/Total) | Meat % (Positive/Total) | Total per Animal Source % (Positive/Total) |
|---|---|---|---|---|---|
| Beef | 4.2 (3/72) | 0.0 (0/0) | 0.0 (0/13) | 0.9 (2/219) | 1.6 (5/304) |
| Swine | 14.7 (33/225) | 18.8 (3/16) | 0.0 (0/18) | 0.0 (0/244) | 7.2 (36/503) |
| Poultry | 1.1 (2/187) | 0.0 (0/47) | 0.0 (0/7) | 0.3 (1/306) | 0.5 (3/547) |
| Total per Sample type | 7.9 (38/484) | 4.8 (3/63) | 0.0 (0/38) | 0.4 (3/769) | 3.2 (44/1354) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perdomo, A.; Webb, H.E.; Bugarel, M.; Friedman, C.R.; Francois Watkins, L.K.; Loneragan, G.H.; Calle, A. First Known Report of mcr-Harboring Enterobacteriaceae in the Dominican Republic. Int. J. Environ. Res. Public Health 2023, 20, 5123. https://doi.org/10.3390/ijerph20065123
Perdomo A, Webb HE, Bugarel M, Friedman CR, Francois Watkins LK, Loneragan GH, Calle A. First Known Report of mcr-Harboring Enterobacteriaceae in the Dominican Republic. International Journal of Environmental Research and Public Health. 2023; 20(6):5123. https://doi.org/10.3390/ijerph20065123
Chicago/Turabian StylePerdomo, Angela, Hattie E. Webb, Marie Bugarel, Cindy R. Friedman, Louise K. Francois Watkins, Guy H. Loneragan, and Alexandra Calle. 2023. "First Known Report of mcr-Harboring Enterobacteriaceae in the Dominican Republic" International Journal of Environmental Research and Public Health 20, no. 6: 5123. https://doi.org/10.3390/ijerph20065123
APA StylePerdomo, A., Webb, H. E., Bugarel, M., Friedman, C. R., Francois Watkins, L. K., Loneragan, G. H., & Calle, A. (2023). First Known Report of mcr-Harboring Enterobacteriaceae in the Dominican Republic. International Journal of Environmental Research and Public Health, 20(6), 5123. https://doi.org/10.3390/ijerph20065123







