Panic in the Pandemic: Determinants of Vaccine Hesitancy and the Dilemma of Public Health Information Sharing during the COVID-19 Pandemic in Sri Lanka
Abstract
:1. Introduction
1.1. Overview of Vaccine Hesitancy
- Confidence—Lack of confidence in the vaccine’s effectiveness, efficacy, and safety, as well as the competency of health care workers (HCWs) who deliver the vaccines. The motivation and objectives behind the vaccination campaigns are often questioned.
- Convenience—Questions on being physically able to visit the site of vaccine delivery, affordability of the vaccine, understanding the instructions given (Health Literacy), and acceptance of vaccination within the cultural norms.
- Complacency—Ability to perceive the risk of getting infected with the disease vs. being protected by the vaccine.
- Collective Responsibility—Willingness to protect others.
- Calculations—Engagement in gathering extensive information.
- Contextual—Communication and media environment, influential leaders/gatekeepers, religion/culture/gender/economy/social status, politics, and policies.
- Individual/Group—Beliefs and attitudes on prevention, personal or family experiences in previous vaccinations, perceived risk and benefit, trust in the health care system and HCWs, etc.
- Vaccine/Vaccine Specific Influencers—Vaccine schedule, cost, design, and mode of delivery in the vaccination programme, reliability of vaccination equipment and the strength of recommendation by a health care professional.
1.2. COVID-19 Pandemic and Vaccination Status in Sri Lanka
2. Materials and Methods
2.1. Household Survey
2.2. Statistical Analysis
2.3. Semi-Structured Interviews
2.4. Ethical Considerations
3. Results
3.1. Factors Affecting Delayed Vaccination and Rejection of Vaccination by the Respondents
“I am newly married, and my husband and I are expecting a baby. We have been planning a baby for the past three years. We met several doctors and did some religious and ritualistic performances to have a child without further delay. Several of our colleagues and relatives advised us to consider this situation before getting a vaccine because many people suspect that vaccine impacts people’s fertility. This may be false or a rumour, but we have no option. No one knows what is happening…”(Female, age 27, urban, higher education, a public servant)
“I am suffering from high blood pressure, cholesterol and blood sugar. Also, I have gone through a stem treatment for blocking a valve in the heart. Several educated people, including some professionals, said that some vaccines make blood clots, which will harm patients with cardiovascular treatments. So, I went completely insane because of this information and even had a long time to get the first vaccine. After seeing people getting vaccines fearlessly, I decided to take the first two doses and did not get the third vaccine due to various concerns over different channels…”(Male, age 66, urban, higher education, a retired executive officer of public service)
“I was waiting to accept the second dose because many information sources informed that there were discrepancies of vaccine’s effectiveness as the vaccine has produced without enough trials…”(Male, age 25, higher education, urban)
“In the first stage, our community members hated to accept the vaccine because they thought it was against some religious principles. However, once cremation was started by the authorities, no one wanted to die as cremation was an extreme barrier to seeing the god. Thus, vaccine acceptance increased significantly…”(Religious leader, age 64, rural)
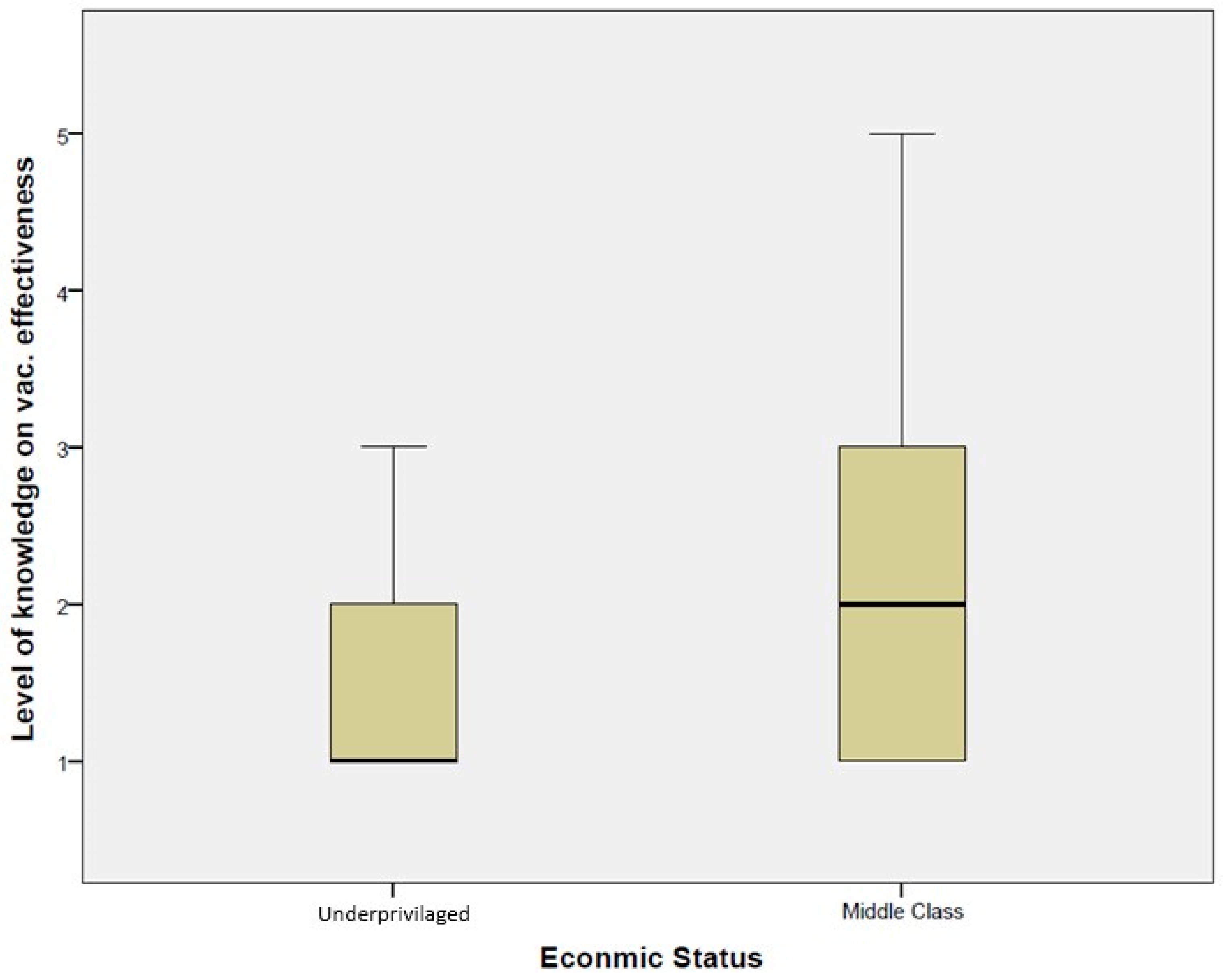
3.2. Knowledge of Vaccines and the Vaccination Process
3.2.1. Knowledge of Vaccine Development, Country of Origin, and Manufacturer Process
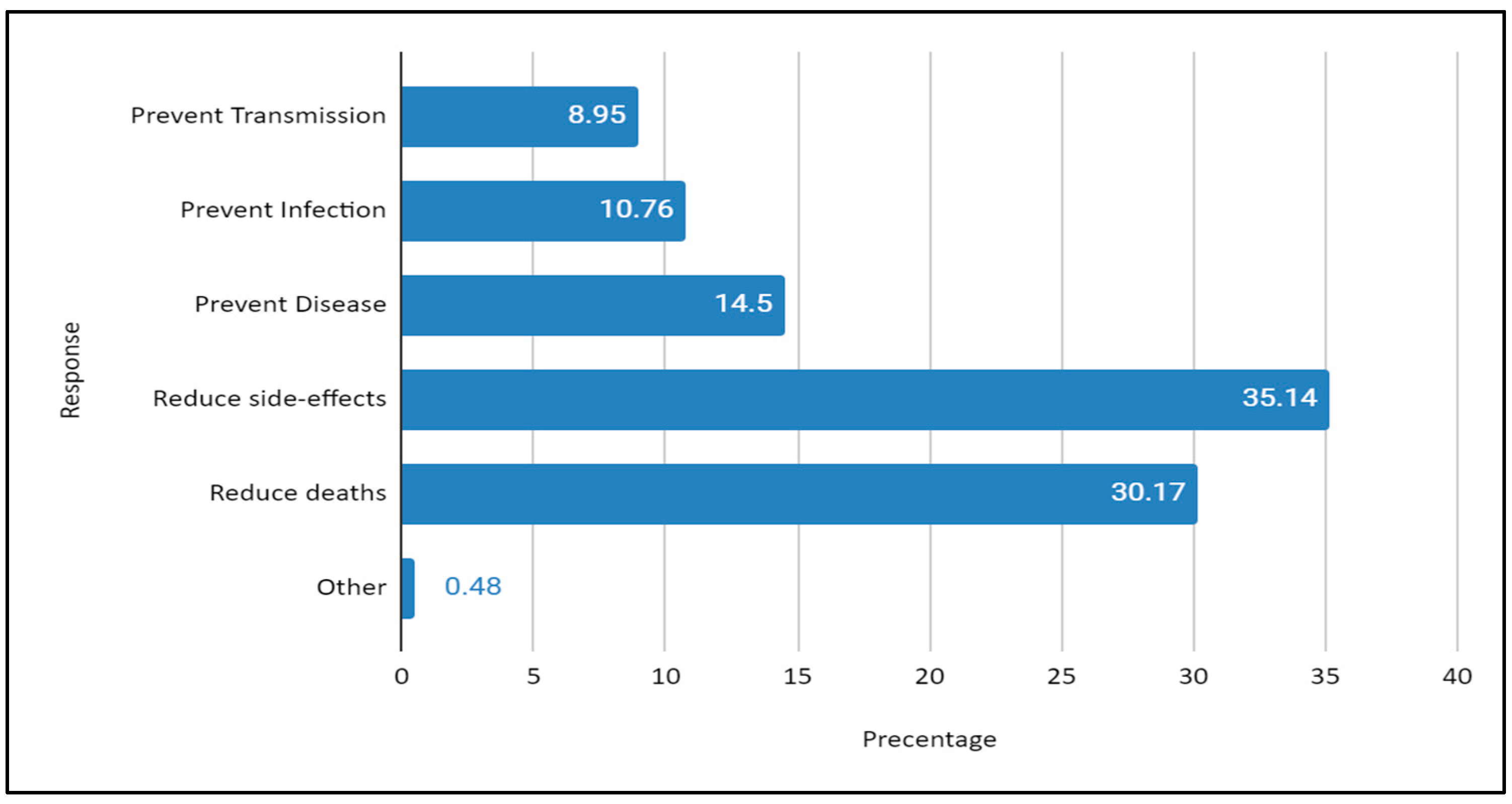
3.2.2. Knowledge of the Vaccine Outcomes
3.2.3. Knowledge of Potential Side Effects of the Vaccine
“I am 6 months pregnant. This is my first baby. I am scared because of side effects of vaccines that people talk about. I have a fear that if I take the vaccine, that will impact my baby…”(Female, age 25, rural, secondary education, no occupation)
“How can we believe the vaccine? There may be unseen side effects. Even paracetamol has side effects. Our peers were discussing the risk of malfunction or dysfunction of organs, especially genital organs (smiling). Many of my friends are afraid to take the vaccine due to this fact. We need to get married and have kids in the future. Who can guarantee that there are no such effects of the vaccines…(smiling)”(female, age 22, urban, tertiary education, IT officer in profession)
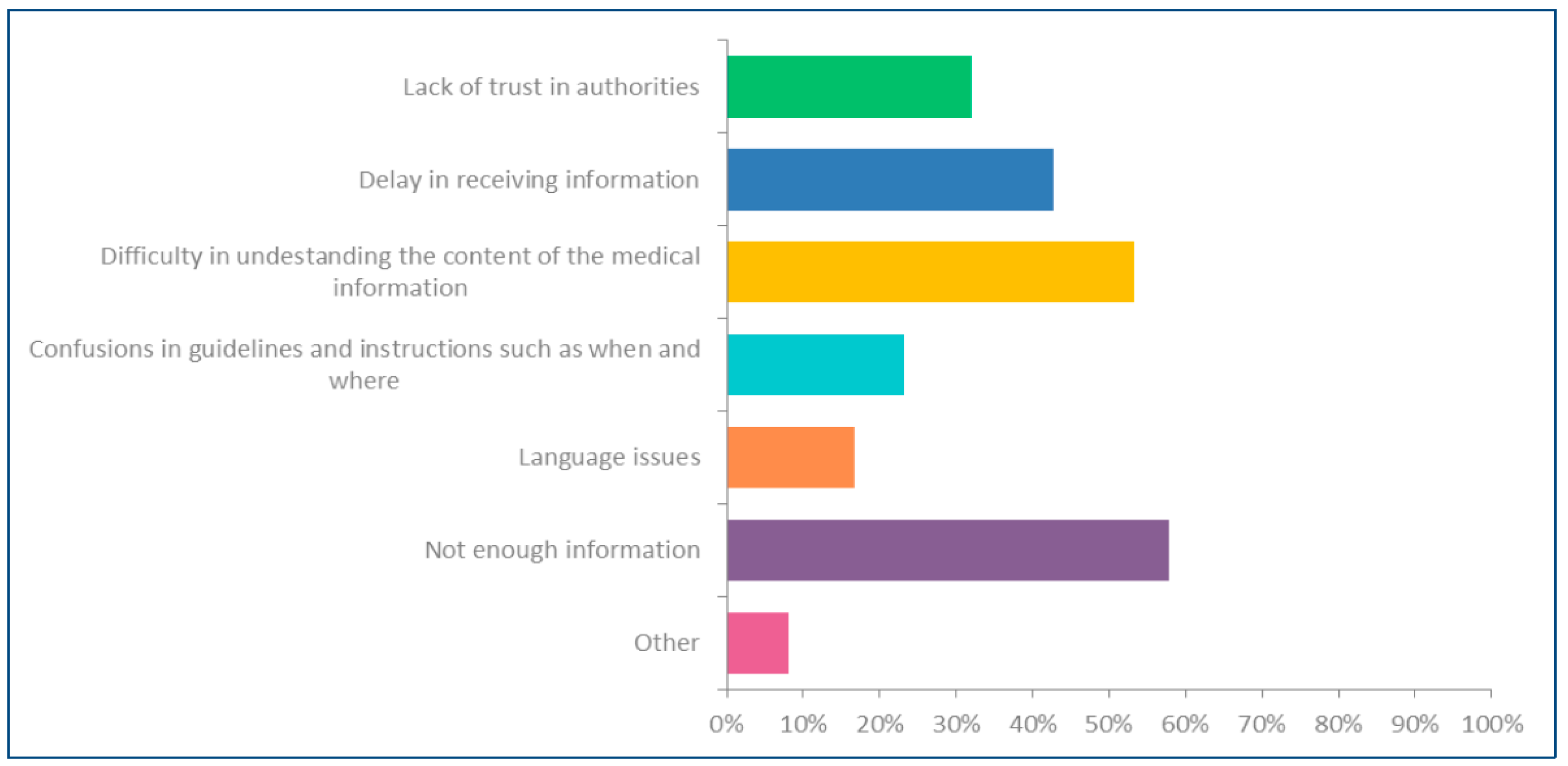
3.3. Public Health Information and Related Factors Affecting Vaccine Intake by the Respondents
“We have no idea about vaccines, the impact of vaccines, and presumed side effects, which people discuss in some informal forums. When we had something to confirm, we used to ask our younger son. He is searching Facebook and telling us what is right and wrong…”(Female, age 54, rural, secondary education, housemate).
Factors Affecting Satisfaction with Public Health Information Sharing among Respondents Regarding Vaccines
“I am involved in public health-related activities at the local level. We are a group that works closely with the community. People ask all doubtful matters from us as they trust us. However, we also faced a challenging situation due to the inconsistency of messages delivered by authorities and officers. We know that COVID-19 is a new challenge, and many of us are learning things by doing. However, we also experienced pathetic contradictions since several officials publicly shared different views on some significant issues associated with COVID-19 in the first half of the pandemic…”(Male, age 52, urban, higher education, public health inspector)
4. Discussion
- Low density of information: estate communities, Indigenous communities, and some communities in remote rural areas and low-income settings have comparatively limited access to information sources due to a lack of information infrastructures, such as internet facilities, devices, and accessibility to reliable data, and lack of public health professionals to engage in such information sharing and discussions. Further, people with insufficient education levels to understand health information shared by different sources also suffer from a scarcity of information on the cause. Hence, rumours are highly prevalent in such settings due to inadequate information, which causes a dilemma of “what to believe and what not”.
- High density of information: Youth, educated people, and some communities, such as urban, semi-urban, and rural areas near urban centres, have too much information coming from various sources, such as internet-based channels, electronic media, social media, professional networks, and primary community networks. In this milieu, people have too much information, complicating their decision-making.
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Piret, J.; Boivin, G. Pandemics Throughout History. Front. Microbiol. 2021, 11, 631736. [Google Scholar] [CrossRef] [PubMed]
- WHO. SARS-CoV-2 Variants-Disease Outbreak News: 31 December 2020; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- El-Shabasy, R.M.; Nayel, M.A.; Taher, M.M.; Abdelmonem, R.; Shoueir, K.R.; Kenawy, E.R. Three waves changes, new variant strains, and vaccination effect against COVID-19 pandemic. Int. J. Biol. Macromol. 2022, 204, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Dubé, E.; Laberge, C.; Guay, M.; Bramadat, P.; Roy, R.; Bettinger, J.A. Vaccine hesitancy. Hum. Vaccines Immunother. 2013, 9, 1763–1773. [Google Scholar] [CrossRef] [PubMed]
- Kamalrathne, T.; Amaratunga, D.; Haigh, R.; Kodituwakku, L. Need for effective detection and early warnings for epidemic and pandemic preparedness planning in the context of multi-hazards: Lessons from the COVID-19 pandemic. Int. J. Disaster Risk Reduct. 2023, 92, 103724. [Google Scholar] [CrossRef] [PubMed]
- Watson, O.J.; Barnsley, G.; Toor, J.; Hogan, A.B.; Winskill, P.; Ghani, A.C. Global impact of the first year of COVID-19 vaccination: A mathematical modelling study. Lancet Infect. Dis. 2022, 22, 1293–1302. [Google Scholar] [CrossRef]
- WHO. WHO Coronavirus (COVID-19) Dashboard-Vaccination Status; [Online]; World Health Organization, 2023; Available online: https://covid19.who.int/?mapFilter=vaccinations (accessed on 4 May 2023).
- Edouard, M.; Ritchie, H.; Rodés-Guirao, L.; Appel, C.; Giattino, C.; Hasell, J.; Macdonald, B.; Dattani, S.; Beltekian, D.; Ortiz-Ospina, E.; et al. Coronavirus Pandemic (COVID-19). Our World in Data. 2020. Available online: https://ourworldindata.org/coronavirus (accessed on 12 July 2021).
- WHO. Infodemic Management: An Overview of Infodemic Management during COVID-19; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- Broniatowski, D.A.; Dredze, M.; Ayers, J.W. “First Do No Harm”: Effective Communication About COVID-19 Vaccines. Am. J. Public Health 2021, 111, 1055–1057. [Google Scholar] [CrossRef]
- Borges Do Nascimento, I.J.; Pizarro, A.B.; Almeida, J.M.; Azzopardi-Muscat, N.; Gonçalves, M.A.; Björklund, M.; Novillo-Ortiz, D. Infodemics and health misinformation: A systematic review of reviews. Bull. World Health Organ. 2022, 100, 544–561. [Google Scholar] [CrossRef]
- Cornwall, W. Officials gird for a war on vaccine misinformation. Science 2020, 369, 14–15. [Google Scholar] [CrossRef]
- Lee, S.K.; Sun, J.; Jang, S.; Connelly, S. Misinformation of COVID-19 vaccines and vaccine hesitancy. Sci. Rep. 2022, 12, 13681. [Google Scholar] [CrossRef]
- Carpiano, R.M.; Callaghan, T.; Diresta, R.; Brewer, N.T.; Clinton, C.; Galvani, A.P.; Lakshmanan, R.; Parmet, W.E.; Omer, S.B.; Buttenheim, A.M.; et al. Confronting the evolution and expansion of anti-vaccine activism in the USA in the COVID-19 era. Lancet 2023, 401, 967–970. [Google Scholar] [CrossRef]
- To, Q.G.; To, K.G.; Huynh, V.N.; Nguyen, N.T.; Ngo, D.T.; Alley, S.; Tran, A.N.; Tran, A.N.; Pham, N.T.; Bui, T.X.; et al. Anti-vaccination attitude trends during the COVID-19 pandemic: A machine learning-based analysis of tweets. Digit. Health 2023, 9, 20552076231158033. [Google Scholar] [CrossRef] [PubMed]
- Bechini, A.; Bonanni, P.; Zanella, B.; Di Pisa, G.; Moscadelli, A.; Paoli, S.; Ancillotti, L.; Bonito, B.; Boccalini, S. Vaccine Production Process: How Much Does the General Population Know about This Topic? A Web-Based Survey. Vaccines 2021, 9, 564. [Google Scholar] [CrossRef] [PubMed]
- Calnan, M. Health policy and controlling COVID-19 in England: Sociological insights. Emerald Open Res. 2020, 1. [Google Scholar] [CrossRef]
- Raballo, A.; Poletti, M.; Preti, A. Vaccine Hesitancy, Anti-Vax, COVID-Conspirationism: From Subcultural Convergence to Public Health and Bioethical Problems. Front. Public Health 2022, 10, 877490. [Google Scholar] [CrossRef]
- Burki, T. The online anti-vaccine movement in the age of COVID-19. Lancet Digit. Health 2020, 2, e504–e505. [Google Scholar] [CrossRef]
- Cinelli, M.; Quattrociocchi, W.; Galeazzi, A.; Valensise, C.M.; Brugnoli, E.; Schmidt, A.L.; Zola, P.; Zollo, F.; Scala, A. The COVID-19 social media infodemic. Sci. Rep. 2020, 10, 16598. [Google Scholar] [CrossRef]
- Tasnim, S.; Hossain, M.M.; Mazumder, H. Impact of Rumors and Misinformation on COVID-19 in Social Media. J. Prev. Med. Public Health 2020, 53, 171–174. [Google Scholar] [CrossRef]
- Wilson, S.L.; Wiysonge, C. Social media and vaccine hesitancy. BMJ Glob. Health 2020, 5, e004206. [Google Scholar] [CrossRef]
- Maldonado, A.Y. Lessons From a House on Fire—From Smallpox to Polio. J. Infect. Dis. 2023, 227, 1025–1027. [Google Scholar] [CrossRef]
- Nuwarda, R.F.; Ramzan, I.; Weekes, L.; Kayser, V. Vaccine Hesitancy: Contemporary Issues and Historical Background. Vaccines 2022, 10, 1595. [Google Scholar] [CrossRef]
- Razai, M.S.; Chaudhry, U.A.R.; Doerholt, K.; Bauld, L.; Majeed, A. COVID-19 vaccination hesitancy. BMJ 2021, 373, n1138. [Google Scholar] [CrossRef] [PubMed]
- WHO. Vaccine Hesitancy: A Growing Challenge for Immunization Programmes; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Hickler, B.; Guirguis, S.; Obregon, R. Vaccine Special Issue on Vaccine Hesitancy. Vaccine 2015, 33, 4155–4156. [Google Scholar] [CrossRef] [PubMed]
- Macdonald, N.E. Vaccine hesitancy: Definition, scope and determinants. Vaccine 2015, 33, 4161–4164. [Google Scholar] [CrossRef] [PubMed]
- SAGE. Report of the Sage Working Group on Vaccine Hesitancy. 2014. Available online: https://www.asset-scienceinsociety.eu/sites/default/files/sage_working_group_revised_report_vaccine_hesitancy.pdf (accessed on 12 July 2021).
- Butler, R.; Macdonald, N.E. Diagnosing the determinants of vaccine hesitancy in specific subgroups: The Guide to Tailoring Immunization Programmes (TIP). Vaccine 2015, 33, 4176–4179. [Google Scholar] [CrossRef] [PubMed]
- Schmid, P.; Rauber, D.; Betsch, C.; Lidolt, G.; Denker, M.L. Barriers of Influenza Vaccination Intention and Behavior—A Systematic Review of Influenza Vaccine Hesitancy, 2005–2016. PLoS ONE 2017, 12, e0170550. [Google Scholar] [CrossRef]
- Lazarus, J.V.; Wyka, K.; White, T.M.; Picchio, C.A.; Gostin, L.O.; Larson, H.J.; Rabin, K.; Ratzan, S.C.; Kamarulzaman, A.; EL-Mohandes, A. A survey of COVID-19 vaccine acceptance across 23 countries in 2022. Nat. Med. 2023, 29, 366–375. [Google Scholar] [CrossRef]
- Sallam, M. COVID-19 Vaccine Hesitancy Worldwide: A Concise Systematic Review of Vaccine Acceptance Rates. Vaccines 2021, 9, 160. [Google Scholar] [CrossRef]
- Shakeel, C.S.; Mujeeb, A.A.; Mirza, M.S.; Chaudhry, B.; Khan, S.J. Global COVID-19 Vaccine Acceptance: A Systematic Review of Associated Social and Behavioral Factors. Vaccines 2022, 10, 110. [Google Scholar] [CrossRef]
- MOH. Coronavirus Disease 2019 (COVID-19)-Situation Report-7.5.2023; Epidemiology Unit, Ministry of Health: Colombo, Sri Lanka, 2023. [Google Scholar]
- Amaratunga, D.; Fernando, N.; Haigh, R.; Jayasinghe, N. The COVID-19 outbreak in Sri Lanka: A synoptic analysis focusing on trends, impacts, risks and science-policy interaction processes. Prog. Disaster Sci. 2020, 8, 100133. [Google Scholar] [CrossRef]
- Galappaththi, E.K.; Perera, C.D.; Dharmasiri, I.P.; Ford, J.D.; Kodithuwakku, S.S.; Chicmana-Zapata, V.; Zavaleta-Cortijo, C.; Pickering, K.; Van Bavel, B.; Hyams, K.; et al. Policy responses to COVID-19 in Sri Lanka and the consideration of Indigenous Peoples. Environ. Sci. Policy 2023, 144, 110–123. [Google Scholar] [CrossRef]
- Kamalrathne, T.; Amaratunga, D.; Haigh, R.; Amaratunga, D.; Haigh, R. A more decentralized governance framework for pandemic response: A multi-stakeholder approach for COVID-19 preparedness and planning in Sri Lanka. In COVID 19: Impact, Mitigation, Opportunities and Building Resilience: From Adversity to Serendipity-Perspectives of Global Relevance Based on Research, Experience and Successes in Combating COVID-19 in Sri Lanka; Ranjith Senaratne, D.A., Shanthi, M., Premachandra, A., Eds.; National Science Foundation: Colombo, Sri Lanka, 2021. [Google Scholar]
- Rannan-Eliya, R.P.; Ghaffoor, A.; Amarasinghe, S.; Nirmani, M.D.; Wijemunige, N.; Perera, S.; Samarage, S.; Dalpatadu, K.; Wisidagama, N.; Fonseka, S.; et al. Sri Lanka’s COVID-19 response and maintaining health services: Implications for future pandemics. BMJ Glob. Health 2024, 8 (Suppl. S6), e013286. [Google Scholar] [CrossRef] [PubMed]
- MOH. High Endemic Diseases/Outbreaks; [Online]; Epidemiology Unit, Ministry of Health: Colombo, Sri Lanka, 2023. Available online: https://www.epid.gov.lk/high-endemic-diseases-outbreaks (accessed on 8 May 2023).
- Swarnamali, H.; Francis, T.V.; Sooriyaarachchi, P.; Jayawardena, R. COVID-19 vaccine hesitancy in Sri Lanka: A national level survey. Int. J. Health Sci. 2023, 17, 3–11. [Google Scholar]
- Wijesinghe, M.S.D.; Weerasinghe, W.M.P.C.; Gunawardana, I.; Perera, S.N.S.; Karunapema, R.P.P. Acceptance of COVID-19 Vaccine in Sri Lanka: Applying the Health Belief Model to an Online Survey. Asia Pac. J. Public Health 2021, 33, 598–602. [Google Scholar] [CrossRef] [PubMed]
- DCS. Estimated Mid Year Population by District; [Online]. Department of Census and Statistics in Sri Lanka; 2023. Available online: https://www.rgd.gov.lk/web/images/2023/vitalStat/MidYearPopulation/Mid-year-population-by-district--Sex.pdf (accessed on 6 April 2024).
- Barlett, J.E.; Kotrlik, J.W.; Higgins, C.C. Organizational research: Determining appropriate sample size in survey research. Inf. Technol. Learn. Perform. J. 2001, 19, 43–50. [Google Scholar]
- MCFADDEN, D. Quantitative methods for Analysing travel behaviour of individuals: Some recent developments. In Behavioural Travel Modelling; Hensher, D., Stopher, P., Eds.; Routledge: London, UK, 1979; pp. 279–318. [Google Scholar]
- Marsoof, A. The Disposal of COVID-19 Dead Bodies: Impact of Sri Lanka’s Response on Fundamental Rights. J. Hum. Rights Pract. 2022, 13, 669–689. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, C.M.C.; Plotkin, S.A. Impact of Vaccines; Health, Economic and Social Perspectives. Front. Microbiol. 2020, 11, 1526. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tan, S.T.; Kwan, A.T.; Rodríguez-Barraquer, I.; Singer, B.J.; Park, H.J.; Lewnard, J.A.; Sears, D.; Lo, N.C. Infectiousness of SARS-CoV-2 breakthrough infections and reinfections during the Omicron wave. Nat. Med. 2023, 29, 358–365. [Google Scholar] [CrossRef]
- Heath, P.T.; Galiza, E.P.; Baxter, D.N.; Boffito, M.; Browne, D.; Burns, F.; Chadwick, D.R.; Clark, R.; Cosgrove, C.A.; Galloway, J.; et al. Safety and Efficacy of the NVX-CoV2373 Coronavirus Disease 2019 Vaccine at Completion of the Placebo-Controlled Phase of a Randomized Controlled Trial. Clin. Infect. Dis. 2023, 76, 398–407. [Google Scholar] [CrossRef]
- Andrews, N.; Stowe, J.; Kirsebom, F.; Toffa, S.; Rickeard, T.; Gallagher, E.; Gower, C.; Kall, M.; Groves, N.; O’Connell, A.M.; et al. covid-19 Vaccine Effectiveness against the Omicron (B.1.1.529) Variant. N. Engl. J. Med. 2022, 386, 1532–1546. [Google Scholar] [CrossRef]
- Wong, L.P.; Alias, H.; Danaee, M.; Ahmed, J.; Lachyan, A.; Cai, C.Z.; Lin, Y.; Hu, Z.; Tan, S.Y.; Lu, Y.; et al. COVID-19 vaccination intention and vaccine characteristics influencing vaccination acceptance: A global survey of 17 countries. Infect. Dis. Poverty 2021, 10, 122. [Google Scholar] [CrossRef]
- Azarpanah, H.; Farhadloo, M.; Vahidov, R.; Pilote, L. Vaccine hesitancy: Evidence from an adverse events following immunization database, and the role of cognitive biases. BMC Public Health 2021, 21, 1686. [Google Scholar] [CrossRef] [PubMed]
- Manilgama, S.R.; Hettiarachchi, N.M.; Jayasinghe, K.I.; De Silva, S.; Wanigaratne, T.; Jayalath, T.; Bandusiri, R.P.; Suganthan, N.; Sudarshan, P.; Pathirage, M.; et al. Prevalence of Covishield COVID-19 Vaccine (ChAdOx1 nCoV-19) Adverse Effects among Health Care Workers in Sri Lanka. Int. J. Infect. Dis. 2022, 116, S61. [Google Scholar] [CrossRef]
- Palmer, R.D. COVID 19 vaccines and the misinterpretation of perceived side effects clarity on the safety of vaccines. BioMedicine 2022, 12, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Wong, M.C.S.; Wong, E.L.Y.; Huang, J.; Cheung, A.W.L.; Law, K.; Chong, M.K.C.; Ng, R.W.Y.; Lai, C.K.C.; Boon, S.S.; Lau, J.T.F.; et al. Acceptance of the COVID-19 vaccine based on the health belief model: A population-based survey in Hong Kong. Vaccine 2021, 39, 1148–1156. [Google Scholar] [CrossRef]
- Seara-Morais, G.J.; Avelino-Silva, T.J.; Couto, M.; Avelino-Silva, V.I. The pervasive association between political ideology and COVID-19 vaccine uptake in Brazil: An ecologic study. BMC Public Health 2023, 23, 1606. [Google Scholar] [CrossRef]
- Hâncean, M.-G.; Lerner, J.; Perc, M.; Molina, J.L.; Geantă, M. Assortative mixing of opinions about COVID-19 vaccination in personal networks. Sci. Rep. 2024, 14, 3385. [Google Scholar] [CrossRef]
- Alsuwaidi, A.R.; Hammad, H.A.A.-K.; Elbarazi, I.; Sheek-Hussein, M. Vaccine hesitancy within the Muslim community: Islamic faith and public health perspectives. Hum. Vaccines Immunother. 2023, 19, 2190716. [Google Scholar] [CrossRef]
- Benoit, S.L.; Mauldin, R.F. The “anti-vax” movement: A quantitative report on vaccine beliefs and knowledge across social media. BMC Public Health 2021, 21, 2106. [Google Scholar] [CrossRef]
- Lin, F.; Chen, X.; Cheng, E.W. Contextualized impacts of an infodemic on vaccine hesitancy: The moderating role of socioeconomic and cultural factors. Inf. Process. Manag. 2022, 59, 103013. [Google Scholar] [CrossRef]
- Rocha, Y.M.; de Moura, G.A.; Desidério, G.A.; de Oliveira, C.H.; Lourenço, F.D.; de Figueiredo Nicolete, L.D. The impact of fake news on social media and its influence on health during the COVID-19 pandemic: A systematic review. J. Public Health 2021, 31, 1007–1016. [Google Scholar] [CrossRef]
- Cervi, L.; Tejedor Calvo, S.; Robledo-Dioses, K. Digital communication and the city. Analysis of the websites of the most visited cities in the world in the COVID-19 era. Rev. Lat. Comun. Soc. 2023, 81, 81–108. [Google Scholar] [CrossRef]
- Nayakarathna, S. Freedom Fights Fake: Sri Lanka’s Fight against the ‘Infodemic’ during the Pandemic. 2021. Available online: https://www.freiheit.org/sri-lanka/sri-lankas-fight-against-infodemic-during-pandemic (accessed on 26 March 2023).
- Balgiu, B.A.; Sfeatcu, R.T.; Âncu, A.M.C.; Imre, M.; Petre, A.; Tribus, L. The Multidimensional Vaccine Hesitancy Scale: A Validation Study. Vaccines 2022, 10, 1755. [Google Scholar] [CrossRef] [PubMed]
- Howard, M.C. A more comprehensive measure of vaccine hesitancy: Creation of the Multidimensional Vaccine Hesitancy Scale (MVHS). J. Health Psychol. 2022, 27, 2402–2419. [Google Scholar] [CrossRef] [PubMed]
- Kotta, I.; Kalcza-Janosi, K.; Szabo, K.; Marschalko, E.E. Development and Validation of the Multidimensional COVID-19 Vaccine Hesitancy Scale. Hum. Vaccines Immunother. 2022, 18, 1–10. [Google Scholar] [CrossRef] [PubMed]

| Vaccine | Dose | Number |
|---|---|---|
| AstraZeneca | 1st dose | 1,479,631 |
| 2nd dose | 1,418,593 | |
| Sinopharm | 1st dose | 12,054,824 |
| 2nd dose | 11,267,138 | |
| Sputnik V | 1st dose | 159,110 |
| 2nd dose | 155,812 | |
| Pfizer | 1st dose | 2,645,395 |
| 2nd dose | 1,123,923 | |
| 1st booster | 8,220,002 | |
| 2nd booster | 202,571 | |
| Moderna | 1st dose | 804,801 |
| 2nd dose | 787,361 |
| District | DS Divisions |
|---|---|
| Kandy | Delthota, Pathadumbara, Yatinuwara, Nawalapitiya |
| Galle | Baddegama, Karandeniya, Kosgoda, Ahungalle |
| Matara | Mulatiyana |
| Anuradhapura | Mihinthale, Anuradhapura |
| Puttalam | Dankotuwa, Wennappuwa, Naththandiya |
| Kurunegala | Ibbagamuwa, Mallwapitiya, Kurunegala |
| Colombo | Padukka, Seethawaka, Thibirigasyaya, Athulkotte, Mahawatta |
| Kalutara | Dodangoda, Walaliawita |
| Badulla | Dambana, Rideemaliyadda |
| Vaccine Dose | Responses |
|---|---|
| Only first dose | 7.53% |
| Only up to the second dose | 55.25% |
| Only up to the first booster | 32.47% |
| None | 4.75% |
| Reasons Admitted by Respondents | n | |
|---|---|---|
| 1 | Lack of knowledge of vaccination | 4.27% |
| 2 | Fear of presumed side effects | 46.29% |
| 3 | Believing in traditional medicine or religious faith | 3.95% |
| 4 | Waiting to see the results of vaccines (wait and see) | 17.40% |
| 5 | Advised by someone not to take the vaccine | 4.44% |
| 6 | Receiving negative information about the vaccine | 12.46% |
| 7 | No vaccine centres nearby | 3.69% |
| 8 | No vaccines available | 3.06% |
| 9 | Lack of encouragement from authorities | 2.37% |
| 10 | Other | 1.67% |
| Variables | Male (Reference) | Female | COR (95% CI) | p | AOR (95% CI) | p | |||
|---|---|---|---|---|---|---|---|---|---|
| Vaccine effectiveness | No knowledge (Ref) | 725 (53.19) | 927 (52.97) | ||||||
| Very low | 308 (22.60) | 391 (22.34) | 0.99 | (0.81–1.17) | 0.94 | 1.05 | (0.87–1.24) | 0.58 | |
| Low | 253 (18.56) | 294 (16.80) | 0.91 | (0.71–1.10) | 0.33 | 0.92 | (0.71–1.13) | 0.43 | |
| Moderate | 69 (5.06) | 126 (7.20) | 1.43 | (1.12–1.74) | 0.02 | 1.31 | (0.96–1.65) | 0.13 | |
| High | 8 (0.59) | 12 (0.69) | 1.17 | (0.27–2.07) | 0.73 | 1.29 | (0.38–2.20) | 0.59 | |
| Side effects of the vaccine | No knowledge (Ref) | 527 (38.66) | 756 (43.20) | ||||||
| Very low | 404 (29.64) | 457 (26.11) | 0.79 | (0.61–0.96) | 0.01 | 0.84 | (0.66–1.02) | 0.06 | |
| Low | 269 (19.74) | 265 (15.14) | 0.69 | (0.48–0.89) | <0.01 | 0.74 | (0.53–0.95) | 0.01 | |
| Moderate | 127 (9.32) | 194 (11.09) | 1.06 | (0.81–1.31) | 0.62 | 1.00 | (0.71–1.28) | 0.98 | |
| High | 36 (2.64) | 78 (4.46) | 1.51 | (1.10–1.92) | 0.05 | 1.23 | (0.78–1.68) | 0.37 | |
| Vaccine suitability against COVID-19 | No knowledge (Ref) | 895 (65.66) | 1101 (62.91) | ||||||
| Very low | 254 (18.64) | 311 (17.77) | 1.00 | (0.81–1.18) | 0.96 | 1.02 | (0.83–1.22) | 0.83 | |
| Low | 154 (11.30) | 205 (11.71) | 1.08 | (0.86–1.31) | 0.50 | 1.04 | (0.79–1.28) | 0.77 | |
| Moderate | 50 (3.67) | 115 (6.57) | 1.87 | (1.53–2.21) | <0.01 | 1.81 | (1.43–2.18) | <0.01 | |
| High | 10 (0.73) | 18 (1.03) | 1.46 | (0.69–2.24) | 0.34 | 1.50 | (0.71–2.29) | 0.32 | |
| Decisions based on scientific information | Yes | 353 (25.90) | 561 (32.06) | ||||||
| No | 553 (40.57) | 561 (32.06) | 0.64 | (0.46–0.82) | <0.01 | 0.654 | (0.47–0.84) | <0.01 | |
| No idea | 457 (33.53) | 628 (35.89) | 0.87 | (0.69–1.04) | 0.11 | 0.829 | (0.63–1.03) | 0.065 | |
| Variables | Underprivileged (Reference) | Middle Income | COR (95% CI) | p | AOR (95% CI) | p | |||
|---|---|---|---|---|---|---|---|---|---|
| Vaccine effectiveness | No knowledge (Ref) | 1004 (64.73) | 648 (41.49) | ||||||
| Very low | 308 (19.86) | 391 (25.03) | 1.97 | (1.79–2.15) | <0.01 | 1.58 | (1.39–1.78) | <0.01 | |
| Low | 206 (13.28) | 341 (21.83) | 2.56 | (2.37–2.76) | <0.01 | 2.12 | (1.90–2.34) | <0.01 | |
| Moderate | 31 (2.00) | 164 (10.50) | 8.20 | (7.80–8.59) | <0.01 | 8.16 | (7.72–8.59) | <0.01 | |
| High | 2 (0.13) | 18 (1.15) | 13.94 | (12.48–15.41) | <0.01 | 9.85 | (8.37–11.33) | <0.01 | |
| Side effects of the vaccine | No knowledge (Ref) | 795 (51.26) | 488 (31.24) | ||||||
| Very low | 400 (25.79) | 461 (29.51) | 1.88 | (1.70–2.05) | <0.01 | 1.33 | (1.14–1.52) | <0.01 | |
| Low | 225 (14.51) | 309 (19.78) | 2.24 | (2.03–2.44) | <0.01 | 1.49 | (1.27–1.71) | <0.01 | |
| Moderate | 77 (4.96) | 244 (15.62) | 5.16 | (4.88–5.44) | <0.01 | 4.12 | (3.80–4.44) | <0.01 | |
| High | 54 (3.48) | 60 (3.84) | 1.81 | (1.43–2.19) | <0.01 | 1.33 | (0.91–1.76) | 0.19 | |
| Vaccine suitability against COVID-19 | No knowledge (Ref) | 1154 (74.40) | 842 (53.91) | ||||||
| Very low | 234 (15.09) | 331 (21.19) | 1.94 | (1.75–2.13) | <0.01 | 1.85 | (1.64–2.06) | <0.01 | |
| Low | 128 (8.25) | 231 (14.79) | 2.47 | (2.24–2.71) | <0.01 | 2.33 | (2.07–2.59) | <0.01 | |
| Moderate | 32 (2.06) | 133 (8.51) | 5.70 | (5.30–6.09) | <0.01 | 6.06 | (5.63–6.50) | <0.01 | |
| High | 3 (0.19) | 25 (1.60) | 11.42 | (10.22–12.62) | <0.01 | 16.37 | (15.00–17.73) | <0.01 | |
| Decisions based on scientific information | Yes | 313 (20.18) | 601 (38.48) | ||||||
| No | 625 (40.30) | 489 (31.31) | 0.41 | (0.23–0.59) | <0.01 | 0.42 | (0.23–0.62) | <0.01 | |
| No idea | 613 (39.52) | 472 (30.22) | 0.40 | (0.22–0.58) | <0.01 | 0.64 | (0.43–0.85) | <0.01 | |
| Variables | Rural (Ref) | Urban | Estate | Urban | Estate | Urban | Estate | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| COR (95% CI) | p | COR (95% CI) | p | AOR (95% COI) | p | AOR (95% COI) | p. | |||||||||
| Vaccine effectiveness | No knowledge (Ref) | 1043 (50.46) | 233 (40.66) | 376 (79.49) | ||||||||||||
| Very low | 489 (23.66) | 143 (24.96) | 67 (14.16) | 1.31 | (1.07–1.54) | 0.02 | 0.38 | (0.10–0.66) | <0.01 | 1.29 | (1.04–1.54) | 0.04 | 1.01 | (0.66–1.37) | 0.94 | |
| Low | 379 (18.34) | 147 (25.65) | 21 (4.44) | 1.74 | (1.50–1.97) | <0.01 | 0.15 | (−0.30–0.61) | <0.01 | 1.54 | (1.27–1.80) | <0.01 | 0.48 | (−0.04–1.01) | 0.01 | |
| Moderate | 140 (6.77) | 46 (8.03) | 9 (1.90) | 1.47 | (1.11–1.83) | 0.04 | 0.18 | (−0.51–0.86) | <0.01 | 1.45 | (1.03–1.86) | 0.08 | 1.69 | (0.90–2.49) | 0.19 | |
| High | 16 (0.77) | 4 (0.70) | 0 (0.0) | 1.12 | (0.01–2.22) | 0.84 | 0.00 | (0.00–0.00) | <0.01 | 1.41 | (0.28–2.54) | 0.55 | 0.00 | (0.00–0.00) | <0.05 | |
| Side effects of the vaccine | No knowledge (Ref) | 769 (37.20) | 155 (27.05) | 359 (75.90) | ||||||||||||
| Very low | 599 (28.98) | 192 (33.51) | 70 (14.80) | 1.59 | (1.35–1.83) | <0.01 | 0.25 | (−0.03–0.53) | <0.01 | 1.53 | (1.28–1.78) | <0.01 | 0.53 | (0.18–0.89) | <0.01 | |
| Low | 394 (19.06) | 111 (19.37) | 29 (6.13) | 1.40 | (1.13–1.67) | 0.02 | 0.16 | (−0.24–0.56) | <0.01 | 1.38 | (1.09–1.67) | 0.03 | 0.38 | (−0.09–0.85) | <0.01 | |
| Moderate | 219 (10.60) | 89 (15.53) | 13 (2.75) | 2.02 | (1.72–2.32) | <0.01 | 0.13 | (−0.45–0.70) | <0.01 | 1.92 | (1.57–2.26) | <0.01 | 0.87 | (0.21–1.53) | 0.68 | |
| High | 86 (4.16) | 26 (4.54) | 2 (0.42) | 1.50 | (1.03–1.97) | 0.09 | 0.05 | (−1.36–1.46) | <0.01 | 1.21 | (0.68–1.74) | 0.48 | 0.24 | (−1.34–1.81) | 0.07 | |
| Vaccine suitability against COVID-19 | No knowledge (Ref) | 1300 (62.89) | 334 (58.29) | 362 (76.53) | ||||||||||||
| Very low | 383 (18.53) | 101 (17.63) | 81 (17.12) | 1.03 | (0.78–1.28) | 0.84 | 0.76 | (0.49–1.03) | 0.04 | 1.10 | (0.83–1.36) | 0.49 | 2.68 | (2.32–3.03) | <0.01 | |
| Low | 248 (12.00) | 91 (15.88) | 20 (4.23) | 1.43 | (1.16–1.70) | 0.01 | 0.29 | (−0.18–0.76) | <0.01 | 1.46 | (1.16–1.76) | 0.01 | 1.61 | (1.04–2.18) | 0.10 | |
| Moderate | 117 (5.66) | 41 (7.16) | 7 (1.48) | 1.36 | (0.99–1.74) | 0.11 | 0.21 | (−0.56–0.99) | <0.01 | 1.48 | (1.05–1.91) | 0.07 | 3.09 | (2.24–3.94) | 0.01 | |
| High | 19 (0.92) | 6 (1.05) | 3 (0.63) | 1.23 | (0.30–2.15) | 0.66 | 0.57 | (−0.66–1.79) | 0.36 | 1.49 | (0.54–2.45) | 0.41 | 3.97 | (2.42–5.53) | 0.08 | |
| Decisions based on scientific information | Yes | 663 (32.08) | 203 (35.43) | 48 (10.15) | ||||||||||||
| No | 767 (37.11) | 232 (40.49) | 115 (24.31) | 0.99 | (0.77–1.20) | 0.91 | 2.07 | (1.72–2.42) | <0.01 | 1.06 | (0.82–1.30) | 0.63 | 0.79 | (0.33–1.25) | 0.31 | |
| No idea | 637 (30.82) | 138 (24.08) | 310 (65.54) | 0.71 | (0.47–0.95) | 0.01 | 6.72 | (6.40–7.05) | <0.01 | 0.79 | (0.52–1.06) | 0.09 | 2.97 | (2.55–3.39) | <0.01 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamalrathne, T.; Jayasekara, J.R.; Amaratunga, D.; Haigh, R.; Kodituwakku, L.; Rupasinghe, C. Panic in the Pandemic: Determinants of Vaccine Hesitancy and the Dilemma of Public Health Information Sharing during the COVID-19 Pandemic in Sri Lanka. Int. J. Environ. Res. Public Health 2024, 21, 1268. https://doi.org/10.3390/ijerph21101268
Kamalrathne T, Jayasekara JR, Amaratunga D, Haigh R, Kodituwakku L, Rupasinghe C. Panic in the Pandemic: Determinants of Vaccine Hesitancy and the Dilemma of Public Health Information Sharing during the COVID-19 Pandemic in Sri Lanka. International Journal of Environmental Research and Public Health. 2024; 21(10):1268. https://doi.org/10.3390/ijerph21101268
Chicago/Turabian StyleKamalrathne, Thushara, Jayasekara R. Jayasekara, Dilanthi Amaratunga, Richard Haigh, Lahiru Kodituwakku, and Chintha Rupasinghe. 2024. "Panic in the Pandemic: Determinants of Vaccine Hesitancy and the Dilemma of Public Health Information Sharing during the COVID-19 Pandemic in Sri Lanka" International Journal of Environmental Research and Public Health 21, no. 10: 1268. https://doi.org/10.3390/ijerph21101268









