The Search for Reliable Biomarkers of Disease in Multiple Chemical Sensitivity and Other Environmental Intolerances
Abstract
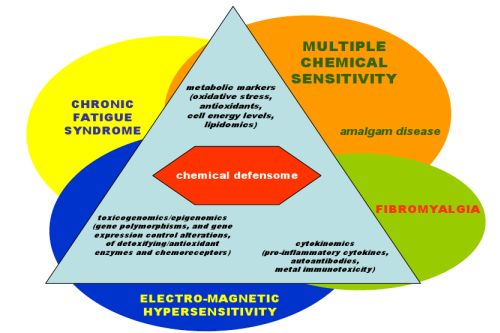
:1. Introduction
2. Controversial Case Definitions
2.1. Multiple Chemical Sensitivity
2.2. Other SRI/IEI
3. State-of-the-Art of Diagnosis for Environmental Intolerances
3.1. Clinical Criteria
3.2. Diagnostic Testing-Molecular Biomarkers of Disease
Genetic markers
Immunologic markers
Metabolic markers
3.3. Diagnostic Testing-Functional Brain Imaging
4. State-of-the-Art of Treatments
5. Conclusions: Sanitary System and Public Social Awareness
References and Notes
- Genuis, SJ. Sensitivity-related illness: the escalating pandemic of allergy, food intolerance and chemical sensitivity. Sci. Total Environ 2010, 408, 6047–6061. [Google Scholar]
- De Luca, C; Scordo, G; Cesareo, E; Raskovic, D; Genovesi, G; Korkina, L. Idiopathic environment-associated diseases: from molecular epidemiology to molecular medicine. Indian J. Exp. Biol 2010, 48, 625–635. [Google Scholar]
- Bornschein, S; Förstl, H; Zilker, T. Idiopathic environmental intolerances (formerly multiple chemical sensitivity) psychiatric perspectives. J. Intern. Med 2001, 250, 309–321. [Google Scholar]
- Staudenmayer, H. Idiopathic environmental intolerances (IEI): myth and reality. Toxicol. Lett 2001, 120, 333–342. [Google Scholar]
- International Programme on Chemical Safety/World Health Organization (IPCS/WHO). Conclusion and recommendations of a wokshop on multiple chemical sensitivities (MCS). Regul. Toxicol. Pharmacol 1996, 24, S79–S86.
- Mayou, R. Somatization. Psycother. Psychom 1993, 59, 69–83. [Google Scholar]
- Rademaker, M. Do Women Have More Adverse Drug Reactions? Am. J. Clin. Dermatol 2001, 2, 349–351. [Google Scholar]
- Cullen, MR. The worker with multiple chemical sensitivities: an overview. Occup. Med 1987, 2, 655–661. [Google Scholar]
- Miller, CS; Prihoda, TJ. A controlled comparison of symptoms and chemical intolerances reported by Gulf War veterans, implant recipients and persons with multiple chemical sensitivity. Toxicol. Ind. Health 1999, 15, 386–397. [Google Scholar]
- Gralewicz, S. Multiple chemical sensitivity: A new type of toxicity? Med. Pr 1998, 49, 473–481. [Google Scholar]
- Nogué Xarau, S; Alarcón Romay, M; Martínez Martínez, JM; Delclós Clanchet, J; Rovira Prat, E; Fernández Solà, J. Multiple chemical sensitivity: Epidemiological, clinical and prognostic differences between occupational and non-occupational cases. Med. Clin. (Barc) 2010, 135, 52–58. [Google Scholar]
- Park, J; Knudson, S. Medically unexplained physical symptoms. Health Rep 2007, 18, 43–47. [Google Scholar]
- Fukuyama, T; Ueda, H; Hayashi, K; Tajima, Y; Shuto, Y; Saito, TR; Harada, T; Kosaka, T. Detection of low-level environmental chemical allergy by a long-term sensitization method. Toxicol. Lett 2008, 180, 1–8. [Google Scholar]
- Shannon, M; Woolf, A; Goldman, R. Children’s environmental health: one year in a pediatric environmental health specialty unit. Ambul. Pediatr 2003, 3, 53–56. [Google Scholar]
- Jedrychowski, W; Galas, A; Whyatt, R; Perera, F. The prenatal use of antibiotics and the development of allergic disease in one year old infants. A preliminary study. Int. J. Occup. Med. Environ. Health 2006, 19, 70–76. [Google Scholar]
- Spencer, TR; Schur, PM. The challenge of multiple chemical sensitivity. J. Environ. Health 2008, 70, 24–27. [Google Scholar]
- De Luca, C; Scordo, MG; Cesareo, E; Pastore, S; Mariani, S; Maiani, G; Stancato, A; Loreti, B; Valacchi, G; Lubrano, C; Raskovic, D; De Padova, L; Genovesi, G; Korkina, L. Biological Definition of Multiple Chemical Sensitivity from Redox State and Cytokine Profiling and not from Polymorphisms of Xenobiotic-Metabolizing Enzymes. Toxicol. Appl. Pharmacol 2010, 248, 285–292. [Google Scholar]
- Korkina, L; Scordo, G; Deeva, I; Cesareo, E; De Luca, C. The chemical defensive system in the pathobiology of idiopathic environment-associated diseases. Curr. Drug Metab 2009, 10, 914–931. [Google Scholar]
- Mattson, MP; Calabrese, EJ. Hormesis: A Revolution in Biology, Toxicology and Medicine; Springer: New York, NY, USA, 2009; p. 213. [Google Scholar]
- Australian Government, Department of Health and Ageing, Office of Chemical Safety and Environmental Health, Scientific Review Report: Multiple Chemical Sensitivity: Identifying Key Research Needs; OCSEH: Canberra, Australia, 2010; p. 101.
- Lipson, JG; Doiron, N. Environmental issues and work: women with multiple chemical sensitivities. Health Care Women Int 2006, 27, 571–584. [Google Scholar]
- Wiesmüller, GA; Ebel, H; Hornberg, C; Kwan, O; Friel, J. Are syndromes in environmental medicine variants of somatoform disorders? Med. Hypotheses 2003, 61, 419–430. [Google Scholar]
- Kipen, HM; Fiedler, N. Environmental Factors in Medically Unexplained Symptoms and Related Syndromes: The Evidence and the Challenge. Environ. Health Perspect 2002, 110, 597–599. [Google Scholar]
- Aaron, LA; Buchwald, D. A review of the evidence for overlap among unexplained clinical conditions. Ann. Intern. Med 2001, 1, 868–881. [Google Scholar]
- Kanaan, RA; Lepine, JP; Wessely, SC. The association or otherwise of the functional somatic syndromes. Psychosom. Med 2007, 69, 855–859. [Google Scholar]
- Karlson, EW; Watts, J; Singorovitch, J; Bonetti, M; Wright, E; Cooper, GS; McAlindon, TE; Costenbader, KH; Massarotti, EM; Fitzgerald, LM; Jajoo, R; Husni, ME; Fossel, AH; Pankey, H; Ding, WZ; Knorr, R; Condon, S; Fraser, PA. Effect of glutathione S-transferase polymorphisms and proximity to hazardous waste sites on time to systemic lupus erythematosus diagnosis: results from the Roxbury lupus project. Arthritis Rheum 2007, 56, 244–254. [Google Scholar]
- Yun, BR; El-Sohemy, A; Cornelis, MC; Bae, SC. Glutathione S-transferase M1, T1, and P1 genotypes and rheumatoid arthritis. J. Rheumatol 2005, 32, 992–997. [Google Scholar]
- Bolt, HM; Kiesswetter, E. Is multiple chemical sensitivity a clinically defined entity? Toxicol. Lett 2002, 128, 99–106. [Google Scholar]
- Moen, BE. Chemical sensitivity and the work place environment: research needs. Psychoneuroendocrinology 2005, 30, 1039–1042. [Google Scholar]
- Sears, ME. The Medical Perspective on Environmentasl Sensitivities; Canadian Human Rights Commission: Ottawa, Canada, 2007. [Google Scholar]
- Waickman, FJ; Vojdani, A. Putting chemical and environmental sensitivities in perspective. Otolaryngol. Clin. North Am 1998, 31, 55–67. [Google Scholar]
- American Lung Association, Environmental Protection Agency, Consumer Product Safety Commission and Americal Medical Association, Indoor Air Pollution: an Introductioni for Health Professionals; EPA: Washington, DC, USA, 1997.
- Lacour, M; Zunder, T; Schmidtke, K; Vaith, P; Scheidt, C. Multiple Chemical Sensitivity Syndrome (MCS)—suggestions for an extension of the US MCS-case definition. Int. J. Hyg. Environ. Health 2005, 208, 141–151. [Google Scholar]
- Kreutzer, R; Neutra, RR; Lashuay, N. Prevalence of people reporting sensitivities to chemicals in a population-based survey. Am. J. Epidemiol 1999, 150, 1–12. [Google Scholar]
- Ashford, NA; Miller, CS. Chemical Exposures: Low Levels and High Stakes, 2nd ed; Van Nostrand Reinhold: New York, NY, USA, 1998. [Google Scholar]
- Hodgson, M. Sick building syndrome. Occup. Med 2000, 15, 571–585. [Google Scholar]
- Andersson, MJ; Andersson, L; Bende, M; Millqvist, E; Nordin, S. The idiopathic environmental intolerance symptom inventory: development, evaluation, and application. J. Occup. Environ. Med 2009, 51, 838–847. [Google Scholar]
- Das-Munshi, J; Rubin, GJ; Wessely, S. Multiple chemical sensitivities: A systematic review of provocation studies. J. Allergy Clin. Immunol 2006, 118, 1257–1264. [Google Scholar]
- Ariga, K; Ji, Q; Hill, JP; Kawazoe, N; Chen, G. Supramolecular approaches to biological therapy. Expert Opin. Biol. Ther 2009, 9, 307–320. [Google Scholar]
- Goldstone, JV; Hamdoun, A; Cole, BJ; Howard-Ashby, M; Nebert, DW; Scally, M; Dean, M; Epel, D; Hahn, ME; Stegeman, JJ. The chemical defensome: environmental sensing and response genes in the Strongylocentrotus purpuratus genome. Dev. Biol 2006, 300, 366–384. [Google Scholar]
- Meyer, UA. Drugs in special patient groups: clinical importance of genomics in drug effects. In Melmon and Morrelli’s Clinical Pharmacology, 4th ed; Carruthers, GS, Hoffmann, BB, Melmon, KL, Nieremberg, DW, Eds.; McGraw Hill: New York NY, USA, 2000; pp. 1179–1205. [Google Scholar]
- Black, DW; Doebbeling, BN; Voelker, MD; Clarke, WR; Woolson, RF; Barrett, DH; Schwartz, DA. Multiple Chemical Sensitivity Syndrome Symptom Prevalence and Risk Factors in a Military Population. Arch. Intern. Med 2000, 160, 1169–1176. [Google Scholar]
- Thomas, HV; Stimpson, NJ; Weightman, AL; Dunstan, F; Lewis, G. Systematic review of multi-symptom conditions in Gulf War veterans. Psychol. Med 2006, 36, 735–747. [Google Scholar]
- Eis, D; Helm, D; Mühlinghaus, T; Birkner, N; Dietel, A; Eikmann, T; Gieler, U; Herr, C; Lacour, M; Nowak, D; Pedrosa Gil, F; Podoll, K; Renner, B; Andreas Wiesmüller, G; Worm, M. The German Multicentre Study on Multiple Chemical Sensitivity (MCS). Int. J. Hyg. Environ. Health 2008, 211, 658–681. [Google Scholar]
- Wolfe, F; Smythe, HA; Yunus, MB; Bennett, RM; Bombardier, C; Goldenberg, DL; Tugwell, P; Campbell, SM; Abeles, M; Clark, P; et al. The American College of Rheumatology 1990 Criteria for the Classification of Fibromyalgia: Report of the Multicenter Criteria Committee. Arthritis Rheum 1990, 33, 160–172. [Google Scholar]
- Bradley, LA. Pathophysiologic mechanisms of fibromyalgia and its related disorders. J. Clin. Psychiatr 2008, 69, 6–13. [Google Scholar]
- George, SZ; Dover, GC; Wallace, MR; Sack, BK; Herbstman, DM; Aydog, E; Fillingim, RB. Biopsychosocial influence on exercise-induced delayed onset muscle soreness at the shoulder: pain catastrophizing and catechol-o-methyltransferase (COMT) diplotype predict pain ratings. Clin. J. Pain 2008, 24, 793–801. [Google Scholar]
- Centers for Disease Control and Prevention (CDC). Chronic Fatigue Syndrome. Available online: http://www.cdc.gov/cfs/ accessed on 18 November 2008.
- Lorusso, L; Mikhaylova, SV; Capelli, E; Ferrari, D; Ngonga, GK; Ricevuti, G. Immunological aspects of chronic fatigue syndrome. Autoimmun. Rev 2009, 8, 287–291. [Google Scholar]
- Maes, M. Inflammatory and oxidative and nitrosative stress pathways underpinning chronic fatigue, somatization and psychosomatic symptoms. Curr. Opin. Psychiatr 2009, 22, 75–83. [Google Scholar]
- Stejskal, VD; Danersund, A; Lindvall, A; Hudecek, R; Nordman, V; Yaqob, A; Mayer, W; Bieger, W; Lindh, U. Metal-specific lymphocytes: biomarkers of sensitivity in man. Neuroendocrinol. Lett 1999, 20, 289–298. [Google Scholar]
- Sterzl, I; Procházková, J; Hrdá, P; Bártová, J; Matucha, P; Stejskal, VD. Mercury and nickel allergy: risk factors in fatigue and autoimmunity. Neuroendocrinol. Lett 1999, 20, 221–228. [Google Scholar]
- Mitchell, RJ; Osborne, PB; Haubenreich, JE. Dental amalgam restorations: daily mercury dose and biocompatibility. J. Long Term Eff. Med. Implants 2005, 15, 709–721. [Google Scholar]
- Hahn, LJ; Kloiber, R; Leininger, RW; Vimy, M; Lorscheider, FL. Whole-body imaging of the distribution of mercury released from dental fillings into monkey tissues. FASEB J 1990, 4, 3256–3260. [Google Scholar]
- Harris, HH; Vogt, S; Eastgate, H; Legnini, DG; Hornberger, B; Cai, Z; Lai, B; Lay, PA. Migration of mercury from dental amalgam through human teeth. J. Synchrotron Radiat 2008, 15, 123–128. [Google Scholar]
- Heintze, U; Edwardsson, S; Derand, T; Birkhed, D. Methylation of mercury from dental amalgam and mercuric chloride by oral streptococci in vitro. Scand. J. Dent. Res 1983, 91, 150–152. [Google Scholar]
- Leistevuo, J; Leistevuo, T; Helenius, H; Pyy, L; Osterblad, M; Huovinen, P; Tenovuo, J. Dental amalgam fillings and the amount of organic mercury in human saliva. Caries Res 2001, 35, 163–166. [Google Scholar]
- Pigatto, PD; Brambilla, L; Guzzi, G. Mercury pink exanthem after dental amalgam placement. J. Eur. Acad. Dermatol. Venereol 2008, 22, 377–378. [Google Scholar]
- Moen, B; Hollund, B; Riise, T. Neurological symptoms among dental assistants: a crosssectional study. J. Occup. Med. Toxicol 2008, 3, 10. [Google Scholar]
- Martin, M; Broughton, S; Drangsholt, M. Oral lichen planus and dental materials: a case-control study. Contact Dermatitis 2003, 48, 331–336. [Google Scholar]
- Bartova, J; Prochazkova, J; Kratka, Z; Benetkova, K; Venclikova, Z; Sterzl, I. Dental amalgam as one of the risk factors in autoimmune diseases. Neuroendocrinol. Lett 2003, 24, 65–67. [Google Scholar]
- Hultman, P; Johansson, U; Turley, S; Lindh, U; Enestrom, S; Pollard, K. Adverse immunological effects and autoimmunity induced by dental amalgam and alloy in mice. FASEB J 1994, 8, 1183–1190. [Google Scholar]
- Bates, MN. Mercury amalgam dental fillings: an epidemiologic assessment. Int. J. Hyg. Environ. Health 2006, 209, 309–316. [Google Scholar]
- Mutter, J; Curth, A; Naumann, J; Deth, R; Walach, H. Does Inorganic Mercury Play a Role in Alzheimer’s Disease? A Systematic Review and an Integrated Molecular Mechanism. J. Alzheimers Dis 2010, 22, 357–374. [Google Scholar]
- Reinhardt, JW. Side effects: mercury contribution to body burden from dental amalgam. Adv. Dent. Res 1992, 6, 110–113. [Google Scholar]
- Zimmer, H; Ludwig, H; Bader, M. Determination of mercury in blood, urine and saliva for the biological monitoring of an exposure from amalgam fillings in a group with self-reported adverse health effects. Int. J. Hyg. Environ. Health 2002, 205, 205–211. [Google Scholar]
- Wiesmüller, GA; Niggemann, H; Weissbach, W; Riley, F; Maarouf, Z; Dott, W; Kunert, HJ; Zerres, K; Eggermann, T; Blömeke, B. Sequence variations in subjects with self-reported multiple chemical sensitivity (sMCS): a case-control study. J. Toxicol. Environ. Health A 2008, 71, 786–794. [Google Scholar]
- Lorscheider, FL; Vimy, MJ; Summers, AO. Mercury exposure from “silver” tooth fillings: emerging evidence questions a traditional dental paradigm. FASEB J 1995, 9, 504–508. [Google Scholar]
- Drasch, G; Böse-O’Reilly, S; Beinhoff, C; Roider, G; Maydl, S. The Mt. Diwata study on the Philippines 1999—assessing mercury intoxication of the population by small scale gold mining. Sci. Total Environ 2001, 267, 151–168. [Google Scholar]
- Jones, L; Bunnell, J; Stillman, J. A 30-year follow-up of residual effects on New Zealand School Dental Nurses, from occupational mercury exposure. Hum. Exp. Toxicol 2007, 26, 367–374. [Google Scholar]
- Mutter, J. Is dental amalgam safe for humans? The opinion of the scientific committee of the European Commission. J. Occup. Med. Toxicol 2011, 6, 2. [Google Scholar]
- Levallois, P. Hypersensitivity of human subjects to environmental electric and magnetic field exposure: a review of the literature. Environ Health Perspect 2002, 110(Suppl 4), 613–618. [Google Scholar]
- Portier, CJ; Wolfe, MS. Assessment of Health Effects from Exposure to Power-Line Frequency Electric and Magnetic Fields; National Institute of Environmental Health Sciences of the National Institutes of Health: Research Triangle Park, NC, USA, 1998. [Google Scholar]
- Eriksson, NM; Stenberg, BG. Baseline prevalence of symptoms related to indoor environment. Scand. J. Public Health 2006, 34, 387–396. [Google Scholar]
- Gangi, S; Johansson, O. A theoretical model based upon mast cells and histamine to explain the recently proclaimed sensitivity to electric and/or magnetic fields in humans. Med. Hypotheses 2000, 54, 663–671. [Google Scholar]
- Belyaev, I. Non-thermal Biological Effects of Microwaves. Microwave Rev 2005, 11, 13–29. [Google Scholar]
- Ziskin, MC. Electromagnetic hypersensitivity: a COMAR technical information statement. IEEE Eng. Med. Biol 2002, 21, 173–175. [Google Scholar]
- Havas, M. Electromagnetic hypersensitivity: biological effects of dirty electricity with emphasis on diabetes and multiple sclerosis. Electromagn. Biol. Med 2006, 25, 259–268. [Google Scholar]
- Ghezel-Ahmadi, D; Engel, A; Weidemann, J; Budnik, LT; Baur, X; Frick, U; Hauser, S; Dahmen, N. Heavy metal exposure in patients suffering from electromagnetic hypersensitivity. Sci. Total Environ 2010, 408, 774–778. [Google Scholar]
- Costa, A; Branca, V; Minoia, C; Pigatto, PD; Guzzi, G. Heavy metals exposure and electromagnetic hypersensitivity. Sci. Total Environ 2010, 408, 4919–4920. [Google Scholar]
- Barbara, G. Editorial: toll-like receptor expression in irritable bowel syndrome: on the alert for a microbial threat? Am. J. Gastroenterol 2011, 106, 337–339. [Google Scholar]
- Yale, SH; Musana, AK; Kieke, A; Hayes, J; Glurich, I; Chyou, PH. Applying case definition criteria to irritable bowel syndrome. Clin. Med. Res 2008, 6, 9–16. [Google Scholar]
- Cash, BD; Chey, WD. Irritable bowel syndrome—an evidence-based approach to diagnosis. Aliment. Pharmacol. Ther 2004, 19, 1235–1245. [Google Scholar]
- Saito, YA; Mitra, N; Mayer, EA. Genetic approaches to functional gastrointestinal disorders. Gastroenterology 2010, 138, 1276–1285. [Google Scholar]
- Domschke, K; Freitag, CM; Kuhlenbäumer, G; Schirmacher, A; Sand, P; Nyhuis, P; Jacob, C; Fritze, J; Franke, P; Rietschel, M; Garritsen, HS; Fimmers, R; Nöthen, MM; Lesch, KP; Stögbauer, F; Deckert, J. Association of the functional V158M catechol-O-methyltransferase polymorphism with panic disorder in women. Int. J. Neuropsychopharmacol 2004, 7, 183–138. [Google Scholar]
- Katz, RS; Wolfe, F; Michaud, K. Fibromyalgia diagnosis: a comparison of clinical, survey, and American College of Rheumatology criteria. Arthritis Rheum 2006, 54, 169–176. [Google Scholar]
- Miller, CS; Prihoda, TJ. The Environmental Exposure and Sensitivity Inventory (EESI): a standardized approach for measuring chemical intolerances for research and clinical applications. Toxicol. Ind. Health 1999, 15, 370–385. [Google Scholar]
- Andersson, MJ; Andersson, L; Bende, M; Millqvist, E; Nordin, S. The idiopathic environmental intolerance symptom inventory: development, evaluation, and application. Occup. Environ. Med 2009, 51, 838–847. [Google Scholar]
- Hojo, S; Kumano, H; Yoshino, H; Kakuta, K; Ishikawa, S. Application of Quick Environment Exposure Sensitivity Inventory (QEESI) for Japanese population: study of reliability and validity of the questionnaire. Toxicol. Ind. Health 2003, 19, 41–49. [Google Scholar]
- Their, R; Brüning, T; Roos, PH; Rihs, HP; Golka, K; Ko, Y; Bolt, HM. Markers of genetic susceptibility in human environmental hygiene and toxicology: the role of selected CYP, NAT and GST genes. Int. J. Hyg. Environ. Health 2003, 206, 149–171. [Google Scholar]
- Wourmhoudt, LW; Commandeur, JN; Vermeulen, NP. Genetic polymorphism of human NAcetyltrasferase, cytochrom P450, glutathione-S-transferase, and epoxide hydrolases emzymes: relevance to xenobiotic metabolism and toxicity. Crit. Review Toxicol 1999, 29, 59–127. [Google Scholar]
- Pavek, P; Dvorak, Z. Xenobiotic-induced transcriptional regulation of xenobiotic metabolizing enzymes of the cytochrome P450 superfamily in human extrahepatic tissues. Curr. Drug Metab 2008, 9, 129–143. [Google Scholar]
- Kerkvliet, NI. AHR-mediated immunomodulation: the role of altered gene transcription. Biochem. Pharmacol 2009, 77, 746–760. [Google Scholar]
- Puga, A; Ma, C; Marlowe, JL. The aryl hydrocarbon receptor cross-talks with multiple signal transduction pathways. Biochem. Pharmacol 2009, 77, 713–722. [Google Scholar]
- Korkina, L; Pastore, S. The role of redox regulation in the normal physiology and inflammatory diseases of skin. Front. Biosci. (Elite Ed) 2009, 1, 123–141. [Google Scholar]
- Jaiswal, AK. Nrf2 signaling in coordinated activation of antioxidant gene expression. Free Radic. Biol. Med 2004, 36, 1199–1207. [Google Scholar]
- Scandalios, JG. Oxidative stress: molecular perception and transduction of signals triggering antioxidant gene defenses. Braz. J. Med. Biol. Res 2005, 38, 995–1014. [Google Scholar]
- McKeown-Eyssen, G; Baines, C; Cole, DE; Riley, N; Tyndale, RF; Marshall, L; Jazmaji, V. Case-control study of genotypes in multiple chemical sensitivity: CYP2D6, NAT1, NAT2, PON1, PON2 and MTHFR. Int. J. Epidemiol 2004, 33, 971–978. [Google Scholar]
- Schnakenberg, E; Fabig, KR; Stanulla, M; Strobl, N; Lustig, M; Fabig, N; Schloot, W. A cross-sectional study of self-reported chemical-related sensitivity is associated with gene variants of drug-metabolizing enzymes. Environ. Health 2007, 6, 6. [Google Scholar]
- Skeith, KJ; Hussain, MS; Coutts, RT; Ramos-Remus, C; Avina-Zubieta, JA; Russell, AS. Adverse drug reactions and debrisoquine/sparteine (P450IID6) polymorphism in patients with fibromyalgia. Clin. Rheumatol 1997, 16, 291–295. [Google Scholar]
- Bolt, HM; Their, R. Relevance of the deletion polymorphisms of the glutathione S-transferases GSTT1 and GSTM1 in pharmacology and toxicology. Curr. Drug Metab 2006, 7, 613–628. [Google Scholar]
- Buskila, D. Genetics of chronic pain states. Best Pract. Res. Clin. Rheumatol 2007, 21, 535–547. [Google Scholar]
- Mannisto, PT; Kaakkola, S. Catechol-O-methyltransferase (COMT): biochemistry, molecular biology, pharmacology, and clinical efficacy of the new selective COMT inhibitors. Pharmacol. Rev 1999, 51, 593–628. [Google Scholar]
- Berg, M; Arnetz, BB; Lidén, S; Eneroth, P; Kallner, A. Techno-stress. A psychophysiological study of employees with VDU-associated skin complaints. J. Occup. Med 1992, 34, 698–701. [Google Scholar]
- Baines, CJ; McKeown-Eyssen, GE; Riley, N; Cole, DEC; Marshall, L; Loescher, B; Jazmaji, V. Case-control study of multiple chemical sensitivity, comparing haematology, biochemistry, vitamins and serum volatile organic compound measures. Occup. Med 2004, 54, 408–418. [Google Scholar]
- Maes, M; Twisk, FN. Why myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) may kill you: disorders in the inflammatory and oxidative and nitrosative stress (IO&NS) pathways may explain cardiovascular disorders in ME/CFS. Neuroendocrinol. Lett 2009, 30, 677–693. [Google Scholar]
- Maes, M; Kubera, M; Uytterhoeven, M; Vrydags, N; Bosmans, E. Increased plasma peroxides as a marker of oxidative stress in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). Med Sci Monit 2011, 17, SC11–15. [Google Scholar]
- Terlecky, SR; Koepke, JI; Walton, PA. Peroxisomes and aging. Biochim. Biophys. Acta 2006, 1763, 1749–1754. [Google Scholar]
- Haley, RW; Billecke, S; La Du, BN. Association of low PON1 type Q (type A) arylesterase activity with neurologic symptom complexes in Gulf War veterans. Toxicol. Appl. Pharmacol 1999, 157, 227–233. [Google Scholar]
- Hüppe, M; Ohnesorge, P; Krauss, B; Schmucker, P. Der MCS Fragebogen: Erste Befunde eines neuen Verfahrens zur Beschreibung MCS-ausloesender Stoffe und Symptome. Umweltmed. Forsch. Prax 2000, 5, 143–153. [Google Scholar]
- Müller, KE; Schnakenberg, E. Die Bedeutung der Glukuronidierung bei umweltmedizinischen Erkrankungen am Beispiel der UDP-Glukuronosyltransferase 1A1. Umwelt Med. Ges 2008, 21, 295–300. [Google Scholar]
- Berg, ND; Rasmussen, HB; Linneberg, A; Brasch-Andersen, C; Fenger, M; Dirksen, A; Vesterhauge, S; Werge, T; Elberling, J. Genetic susceptibility factors for multiple chemical sensitivity revisited. Int. J. Hyg. Environ. Health 2010, 213, 131–139. [Google Scholar]
- Genuis, SJ. The chemical erosion of human health: adverse environmental exposure and in-utero pollution-determinants of congenital disorders and chronic disease. J. Perinat. Med 2006, 34, 185–195. [Google Scholar]
- Rea, WJ. Chemical Sensitivity (Volume 4): Tools of Diagnosis and Methods of Treatment; Lewis Publishers: Boca Raton, FL, USA, 1997. [Google Scholar]
- Buffington, T. Developmental influences on medically unexplained symptoms. Psychother. Psychosom 2009, 78, 139–144. [Google Scholar]
- Genuis, SJ. Our genes are not our destiny: incorporating molecular medicine into clinical practice. J. Eval. Clin. Pract 2008, 14, 94–102. [Google Scholar]
- Patarca, R. Cytokines and chronic fatigue syndrome. Ann. NY Acad. Sci 2001, 933, 185–200. [Google Scholar]
- Dearman, RJ; Kimber, I. Cytokine profiling and chemical allergy. Toxicol. Appl. Pharmacol 2002, 185, 228–229. [Google Scholar]
- Jammes, Y; Steinberg, JG; Delliaux, S; Brégeon, F. Chronic fatigue syndrome combines increased exercise inducedoxidative stress and reduced cytokine and Hsp responses. J. Intern. Med 2009, 266, 196–206. [Google Scholar]
- Levin, AS; Byers, VS. Environmental illness: a disorder of immune regulation. Occup. Med 1987, 2, 669–681. [Google Scholar]
- Valentine-Thon, E; Müller, K; Guzzi, G; Kreisel, S; Ohnsorge, P; Sandkamp, M. LTT-MELISA is clinically relevant for detecting and monitoring metal sensitivity. Neuroendocrinol. Lett 2006, 27, 17–24. [Google Scholar]
- Klein, R; Schwenk, M; Heinrich-Ramm, R; Templeton, DM. The lymphocyte transformation test for sensitation to berillium and other metals. Pure Appl. Chem 2004, 76, 1269–1281. [Google Scholar]
- Hybenova, M; Hrda, P; Procházková, J; Stejskal, V; Sterzl, I. The role of environmental factors in autoimmune thyroiditis. Neuroendocrinol. Lett 2010, 31, 283–289. [Google Scholar]
- Pall, ML. Elevated nitric oxide/peroxynitrite theory of multiple chemical sensitivity: central role of N-methyl-D-aspartate receptors in the sensitivity mechanism. Environ. Health Perspect 2003, 111, 1461–1464. [Google Scholar]
- Jammes, Y; Steinberg, JG; Mambrini, O; Brégeon, F; Delliaux, S. Chronic fatigue syndrome: assessment of increased oxidative stress and altered muscle excitability in response to incremental exercise. J. Intern. Med 2005, 257, 299–310. [Google Scholar]
- Kennedy, G; Spence, VA; McLaren, M; Hill, A; Underwood, C; Belch, JJ. Oxidative stress levels are raised in chronic fatigue syndrome and are associated with clinical symptoms. Free Radic. Biol. Med 2005, 39, 584–589. [Google Scholar]
- Pizzichini, M; Fonzi, M; Sugherini, L; Fonzi, L; Gasparoni, A; Comporti, M; Pompella, A. Release of mercury from dental amalgam and its influence on salivary antioxidant activity. Sci. Total Environ 2002, 284, 19–25. [Google Scholar]
- Di Pietro, A; Visalli, G; La Maestra, S. Biomonitoring of DNA damage in peripheral blood lymphocytes of subjects with dental restorative fillings. Mutat. Res 2008, 650, 115–122. [Google Scholar]
- Logan, AC; Wong, C. Chronic fatigue syndrome: oxidative stress and dietary modifications. Altern. Med. Rev 2001, 6, 450–459. [Google Scholar]
- Cordero, MD; Moreno-Fernandez, AM; deMiguel, M; Bonal, P; Campa, F; Jimenez-Jimenez, LM; Ruiz-Losada, A; Sánchez-Domínguez, B; Sánchez Alcázar, JA; Salviati, L; Navas, P. Coenzyme Q10 distribution in blood is altered in patients with fibromyalgia. Clin. Biochem 2009, 42, 732–735. [Google Scholar]
- Bagis, S; Tamer, L; Sahin, G; Bilgin, R; Guler, H; Ercan, B; Erdogan, C. Free radicals and antioxidants in primary fibromyalgia: an oxidative stress disorder? Rheumatol. Int 2005, 25, 188–190. [Google Scholar]
- Pall, ML. Common etiology of posttraumatic stress disorder, fibromyalgia, chronic fatigue syndrome and multiple chemical sensitivity via elevated nitric oxide/peroxynitrite. Med. Hypotheses 2001, 57, 139–145. [Google Scholar]
- Chung, CP; Titova, D; Oeser, A; Randels, M; Avalos, I; Milne, GL; Morrow, JD; Stein, CM. Oxidative stress in fibromyalgia and its relationship to symptoms. Clin. Rheumatol 2009, 28, 435–438. [Google Scholar]
- Fulle, S; Mecocci, P; Fano, G; Vecchiet, I; Vecchini, A; Ricciotti, D; Cherubini, A; Pizzigallo, E; Vecchiet, L; Senin, U; Beal, MF. Specific oxidative alterations in vastus lateralis muscle of patients with the diagnosis of chronic fatigue syndrome. Free Rad. Biol. Med 2000, 29, 1252–1259. [Google Scholar]
- Bossuyt, PM; Reitsma, JB; Bruns, DE; Gatsonis, CA; Glasziou, PP; Irwig, LM; Lijmer, JG; Moher, D; Rennie, D; de Vet, HC. Towards complete and accurate reporting of studies of diagnostic accuracy: the STARD initiative. Clin. Chem 2003, 49, 1–6. [Google Scholar]
- Kumar, D. From evidence-based medicine to genomic medicine. Genomic Med 2007, 1, 95–104. [Google Scholar]
- Heuser, G; Wu, JC. Deep subcortical (including limbic) hypermetabolism in patients with chemical intolerance: human PET studies. Ann. NY Acad. Sci 2001, 933, 319–322. [Google Scholar]
- Mayberg, H. Critique: SPECT studies of multiple chemical sensitivity. Toxicol. Ind. Health 1994, 10, 661–665. [Google Scholar]
- Bornschein, S; Hausteiner, C; Drzezga, A; Bartenstein, P; Schwaiger, M; Förstl, H; Zilker, T. PET in patients with clear-cut multiple chemical sensitivity (MCS). Nuklearmedizin 2002, 41, 233–239. [Google Scholar]
- Bartenstein, P; Grünwald, F; Herholz, K; Kuwert, T; Tatsch, K; Sabri, O; Weiller, C. Role of positron emission tomography (PET) and single photon emission tomography (SPECT) in so-called “multiple chemical sensitivity”. Nuklearmedizin 1999, 38, 297–301. [Google Scholar]
- Gibson, PR; Elms, AN; Ruding, LA. Perceived treatment efficacy for conventional and alternative therapies reported by persons with multiple chemical sensitivity. Environ. Health Perspect 2003, 111, 1498–1504. [Google Scholar]
- Johansson, O. Electrohypersensitivity: state-of-the-art of a functional impairment. Electromagn. Biol. Med 2006, 25, 245–258. [Google Scholar]
- Scordo, MG; Caputi, AP; D’Arrigo, C; Fava, G; Spina, E. Allele and genotype frequencies of CYP2C9, CYP2C19 and CYP2D6 in an Italian population. Pharmacol. Res 2004, 50, 195–200. [Google Scholar]
- Riedl, MA; Casillas, AM. Adverse drug reactions: types and treatment. Am. Fam. Physician 2003, 68, 1781–1790. [Google Scholar]
- Guttman-Yassky, E; Weltfriend, S; Bergman, R. Resolution of orofacial granulomatosis with amalgam removal. J. Eur. Acad. Dermatol. Venerol 2003, 17, 344–347. [Google Scholar]
- Dunsche, A; Kastel, I; Terheyden, H; Springer, I; Christophers, E; Brasch, J. Oral lichenoid reactions associated with amalgam: improvement after amalgam removal. Br. J. Dermatol 2003, 148, 70–76. [Google Scholar]
- Hargreaves, RJ; Evans, JG; Janota, I; Magos, L; Cavanagh, JB. Persistant mercury in nerve cells 16 years after metallic mercury poisoning. Neuropath. Appl. Neurobiol 1988, 14, 443–452. [Google Scholar]
- Opitz, H; Schweinsberg, F; Grossmann, T; Wendt-Gallitelli, MF; Meyermann, R. Demonstration of mercury in the human brain and other organs 17 years after metallic mercury exposure. Clin. Neuropath 1996, 15, 139–144. [Google Scholar]
- Rooney, JP. The role of thiols, dithiols, nutritional factors and interacting ligands in the toxicology of mercury. Toxicology 2007, 234, 145–156. [Google Scholar]
- Aposhian, HV; Morgan, DL; Queen, HL; Maiorino, RM; Aposhian, MM. Vitamin C, glutathione, or lipoic acid did not decrease brain or kidney mercury in rats exposed to mercury vapor. J. Toxicol. Clin. Toxicol 2003, 41, 339–347. [Google Scholar]
- Goyer, RA. Nutrition and metal toxicity. Am. J. Clin. Nutr 1995, 61, 646S–650S. [Google Scholar]
- Ishibashi, M; Tonori, H; Miki, T; Miyajima, E; Kudo, Y; Tsunoda, M; Sakabe, K; Aizawa, Y. Classification of patients complaining of sick house syndrome and/or multiple chemical sensitivity. Tohoku J. Exp. Med 2007, 211, 223–233. [Google Scholar]
- Canadian Centre for Occupational Health and Safety, Indoor Air Quality: A Legitimate OSH Concern; Canadian Centre for Occupational Health and Safety: Hamilton, Canada, 2008.
- US Department of Education, Information Memorandum: Multiple Chemical Sensitivity; RSA-IM-02-04; US Department of Education: Washington, DC, USA, 2001.
- Rappaport, SM; Kim, S; Lan, Q; Vermeulen, R; Waidyanatha, S; Zhang, L; Li, G; Yin, S; Hayes, RB; Rothman, N; Smith, MT. Evidence hat humans metabolize benzene via two pathways. Environ. Health Perspect 2009, 117, 946–952. [Google Scholar]
- Cooper, C. Multiple chemical sensitivity in the clinical setting. Am. J. Nurs 2007, 107, 40–47. [Google Scholar]
- Randolph, TG. The ecologic unit—part I. Hosp. Manag 1964, 97, 45–47. [Google Scholar]
- Anema, S. Hospitalization for the Chemically Sensitive Patient Hospital Protocol Guidelines Dallas 1999—Multiple Chemical Sensitivity Guidelines for South Australian Hospitals; SA Health: Adelaide, Australia, 2010. [Google Scholar]
- Fisher, MM; Rose, M. Anaesthesia for patients with idiopathic environmental intolerance and chronic fatigue syndrome. Br. J. Anaesth 2008, 101, 486–491. [Google Scholar]
- Ishikawa, S; Miyata, M. Chemical sensitivity and its clinical characteristics in Japan. Asian Med. J 2010, 43, 7–15. [Google Scholar]
- Joffres, MR; Sampalli, T; Fox, RA. Physiologic and symptomatic responses to low-level substances in individuals with and without chemical sensitivities: a randomized controlled blinded pilot booth study. Environ. Health Perspect 2005, 113, 1178–1183. [Google Scholar]
- Fox, RA; Joffres, MR; Sampalli, T; Casey, J. The impact of a multidisciplinary, holistic approach to management of patients diagnosed with multiple chemical sensitivity on health care utilization costs: an observational study. J. Altern. Complement. Med 2007, 13, 223–229. [Google Scholar]
- Sampalli, T; Shepherd, M; Fox, R. Boundary objects in the multidisciplinary care management of chronic conditions: multiple chemical sensitivity. Stud. Health Technol. Inform 2009, 143, 534–539. [Google Scholar]
- Eltiti, S; Wallace, D; Zougkou, K; Russo, R; Joseph, S; Rasor, P; Fox, E. Development and evaluation of the electromagnetic hypersensitivity questionnaire. Bioelectromagnetics 2007, 28, 137–151. [Google Scholar]
- Skovbjerg, S; Johansen, JD; Rasmussen, A; Thorsen, H; Elberling, J. General practitioners’ experiences with provision of healthcare to patients with self-reported multiple chemical sensitivity. Scand. J. Prim. Health Care 2009, 27, 148–152. [Google Scholar]
| gene variant | population | diagnostic criteria | patients | healthy controls | Ref. |
|---|---|---|---|---|---|
| PON1 Q/R | GWV | - | 25 | 20 | Haley et al. [109] |
| PON1-55, PON1-192, PON2-148, CYP2D6, NAT1,NAT2, MHTFR C677T | MCS women | University of Toronto Health Survey (UTHS) reproducible, self-administered questionnaire | 203 | 162 | McKeown-Eyssen et al. [98] |
| NAT2, GSTM1, GSTT1 | Chemically sensitive | Modified EESI questionnaire | 273 | 248 | Schnackenberg et al. [99] |
| 5HTT, NAT1, NAT2, PON1, PON2, SOD2 | Self-reported MCS | Self-administered Environmental Medicine Questionnaire questionnaire (Hüppe et al. [110]) | 59 | 40 | Wiesműller et al. [67] |
| UGTA1 | MCS and/or FCS and/or FM | 42 | - | Műller et al. [111] | |
| CYP2C9, CYP2C19, CYP2D6, CYP3A5, UGTA1, UGTA, GSTT1, GSTM1, GSTP1 | MCS | Cullen’s criteria, QEESI | 144 | 218 | De Luca et al. [17] |
| CYP2D6, NAT2, PON1, MHTFR, CCK2R | MCS | Cullen’s criteria | 96 | 1,027 | Berg et al. [112] |
| Treatment | Condition |
|---|---|
| Trigger removal (trigger-free living space, chemical avoidance, antifungal sanitization, working place change or abandon, selected food removal, dental amalgam removal | MCS, CFS, IBS, EHS, SBS, SHS |
| Nutritional supplements | MCS, FM, FCS |
| Prescription therapies (psychotropic drugs, antimycotics, antiobiotics, hormone replacement) | MCS, FM, FCS, IBS, GWV |
| Psychotherapy | MCS, FM, CFS, GWV, dental amalgam disease, EHS |
| Detoxification treatments | MCS, FM, CFS, GWV, dental amalgam disease |
| Holistic treatments | MCS, FM, CFS, GWV, dental amalgam disease |
| Body therapies | MCS, FM, FCS |
© 2011 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
De Luca, C.; Raskovic, D.; Pacifico, V.; Thai, J.C.S.; Korkina, L. The Search for Reliable Biomarkers of Disease in Multiple Chemical Sensitivity and Other Environmental Intolerances. Int. J. Environ. Res. Public Health 2011, 8, 2770-2797. https://doi.org/10.3390/ijerph8072770
De Luca C, Raskovic D, Pacifico V, Thai JCS, Korkina L. The Search for Reliable Biomarkers of Disease in Multiple Chemical Sensitivity and Other Environmental Intolerances. International Journal of Environmental Research and Public Health. 2011; 8(7):2770-2797. https://doi.org/10.3390/ijerph8072770
Chicago/Turabian StyleDe Luca, Chiara, Desanka Raskovic, Valeria Pacifico, Jeffrey Chung Sheun Thai, and Liudmila Korkina. 2011. "The Search for Reliable Biomarkers of Disease in Multiple Chemical Sensitivity and Other Environmental Intolerances" International Journal of Environmental Research and Public Health 8, no. 7: 2770-2797. https://doi.org/10.3390/ijerph8072770